Abstract
With the increased importance of CT radiation dose to health care providers, patients and the general public, there is an increased responsibility to minimize patient dose effectively. Bismuth shields offer a simple strategy to reduce dose to certain anterior radiosensitive organs such as breasts and eyes. However, in order to reduce organ dose they must be used properly; improper use can lead to an actual increase in the patient dose. They also create a proportional increase in image noise in the section of the body adjacent to the shield and further reduce the quantitative precision of CT numbers. In addition, shielding can degrade the overall efficiency (by an order of approximately 10%) of the imaging process, reducing the theoretical image quality that can be expected from a certain level of patient dose. However, in spite of their significant disadvantages, there are certain clinical situations and practice considerations that provide qualified justification for their continued use.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In the last decade radiation dose associated with CT has become the topic of major public interest and concern. At a population level, there is a growing mandate to minimize radiation dose to as low as reasonably achievable [1]. Although this mandate addresses the radiation burden to the patient as a whole, there is also an established understanding that not all organs have the same level of radiosensitivity [2]. For example, breasts are more radiosensitive than the liver. The lens of the eye is more prone to radiation effects (in the form of cataract formation) than the brain. Further, imaging is often done in the context of a particular body region that exposes certain organs and not others. As such, patient dosimetry can be grossly oversimplified if it is based on radiation burden associated with individual organs, a topic addressed elsewhere in this issue [3]. Furthermore, there is a justifiable need to modulate the radiation field to minimize the exposure to certain radiosensitive organs.
Organ dose minimization can be accomplished via alteration of the radiation field from the scanner in a process known as organ-based tube current modulation, or virtual shielding [4]. In this process, the X-ray beam can be turned off when the beam is impinging on a superficial radiosensitive organ but alternatively increased when the beam is on the other side of the patient (Fig. 1). Combined with appropriate reconstruction, without a major impact on overall dose or image quality, this technology can reduce the dose to the targeted organ. But the major drawback of virtual shielding is the fact that it is offered by only one manufacturer and only in certain models. It is also less effective for breast dose reduction in adults who have large breasts [5].
In a conventional diagnostic thoracic CT scan, the breast is always included (a), but it is rarely an organ of interest. In the virtual shielding scheme (b), the mA is severely reduced when the beam is over the breasts and increased elsewhere, reducing the breast dose by 30–40% without an increase in image noise. (Dose maps courtesy of Juan Carlos Ramirez, Siemens Healthcare, Erlangen, Germany)
Alternatively, select organ shielding can be done with the application of an actual shield [6]. The shields are usually made of bismuth-impregnated latex with typical 0.85–3.4 g/cm of bismuth in the shield. They are designed to be placed over an organ of interest during the CT examination, typically targeting the eyes, breasts, thyroid or gonads (Fig. 2). By attenuating the X-ray beam entering the patient, the shield can reduce the dose to the targeted organ. This application of X-ray shielding should not be confused with the use of protective layers to block the X-ray from the patient or the operator through more absorptive lead shields when radiation is not needed. The bismuth shields are meant to be applied within the imaging field of view, and while offering a modest level of protection, they still allow enough X-ray penetration to enable image formation. The goal is to reduce the X-ray flux to provide beam modulation for the purpose of dose reduction to the organ.
Demonstration of the use of eye (a) and thyroid (b) shields for a pediatric examination (from J.D. Honigberg International, www.jdhmedical.com)
Bismuth organ shields can provide a meaningful reduction of dose to anterior organs as indicated in a number of studies; combined with their ease of use, they may be considered a method to mitigate the radiation risk to certain radiosensitive organs [6–10]. However, there are a number of significant drawbacks in the use of these shields that should be carefully considered. In 2012, the American Association of Physicists in Medicine (AAPM) released a position statement about the use of bismuth shields [11]. The statement noted “several disadvantages associated with the use of bismuth shields” and recommended that “alternatives to bismuth shielding be carefully considered, and implemented when possible” [11]. Using the AAPM justification for this statement, this article aims to summarize the disadvantages associated with the use of bismuth shields. However, there are certain considerations governing when the use of these shields is justified. This article further outlines these considerations.
The cons: potential problems associated with bismuth shields
There are three major disadvantages associated with the use of bismuth shields. First, in their exact deployment they can be applied improperly, leading to an actual over-exposure of the patient. Second, the shield can impact the quantitative accuracy of the CT numbers in the regions most proximate to the shield. Finally, there is in a sense wasted dose when the shield is applied. We detail each of these below.
One of the major concerns with the use of bismuth shields is that they may be deployed inappropriately when automatic exposure control (AEC) in the form of tube current modulation is used. The purpose of the AEC is to adaptively adjust the flux of X-rays based on the patient attenuation as the tube travels around the patient. A higher-attenuation section of the body results in higher X-ray flux. The scheme of the AEC for a given scan is most frequently based on the localizer image that is acquired in advance of the actual CT acquisition. Here lies the improper use of the bismuth shield. As illustrated in Fig. 3, to be effective the shield should be placed over the organ of interest after the localizer image is acquired. In doing so, the system scans the patient with no regard to the presence of the shield; the tube current is applied as if the shield were not present, leading to the desired dose reduction. If, however, the shield is placed before the localizer acquisition, the system adaptively increases the tube current in response to the attenuation of the shield, nullifying the very dose reduction purpose of the shield and possibly even increasing both the organ dose and the total patient dose [8, 12].
The top row demonstrates how the axial automatic exposure control (AEC) mA profile (yellow line) is determined from the localizer and how the addition of the shield (red line) after the localizer image acquisition does not impact the AEC profile. In the bottom row, the AEC is incorrectly applied by being placed before the localizer acquisition, leading to a prospective change in the AEC profile to a higher mA and increased patient dose
The above illustration is applicable to CT systems in which the AEC tube current modulation profile is fully determined from the localizer prior to the actual CT acquisition. In contrast, in certain CT systems the AEC profile is dynamically changed based on the attenuation level that the X-ray beam encounters as it rotates around the patient. For such systems, the organ shield should never be used because it would inevitably lead to an increase in patient dose.
A second concern about the use of the bismuth shield is the associated change in quantitative CT numbers [13]. Beam hardening is one of the major artifacts in CT imaging, affecting the accuracy and quantifiability of CT numbers. It manifests most prominently when highly attenuating objects are in the imaging field of view. Metal objects (e.g., metal implants), bones and higher concentrations of iodine create the most notable artifacts, affecting the CT numbers. The presence of a bismuth attenuating layer in the field-of-view similarly impacts the CT numbers, particularly in the proximity of the shield. The effect can be minimized by placing additional spacers between the shield and the body, but it cannot be fully eliminated.
Figure 4 illustrates this artifact, where the addition of the shield alters the CT numbers and appearance, most prominently in the proximity of the shield but even as far off as the lesion in mid-lung. Also illustrated in Fig. 4 is the impact of the shield on image noise. The shielded image is noisier. The increased noise can potentially impact diagnostic quality. However this enhancement of noise with the addition of a breast shield cannot necessarily be considered a disadvantage of the shield because dose reduction via the use of a shield or a corresponding change in the CT technique (e.g., mAs) can lead to a comparable impact on image noise. As such, the change in noise is not a major advantage or disadvantage of the use of the bismuth shield. Figure 5 illustrates the quantification of the change in noise and the CT numbers in a pediatric CT phantom with the addition of a shield compared to an alternative of reducing the overall mAs.
Axial CT of a lung nodule in a 17-year-old girl with history of ovarian dysgerminoma. CT scan obtained with breast shield in place (a) and without (b) shows lung nodule (arrow) in right lower lobe. The image acquired with the shield demonstrates a greater noise magnitude. (Used with permission from Fricke et al. [9])
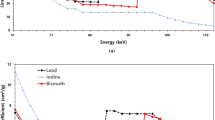
Assessment of noise and CT number in four regions of a pediatric phantom (a) without shield (Reference), with shield (Bi shielding) and without shield but with the mA reduced to achieve the same dose reduction as that of the shielded condition (Low-mAs). ROI-1 and ROI-2 are in the lungs and ROI-3 and ROI-4 in the mediastinum. The noise results (b) are largely equivalent with shielding and mA reduction, yielding similar results. The CT numbers in the lung (c) and mediastinum (d) can be affected by the addition of the shield but not by similar dose reduction achieved with reduced mAs. (Courtesy of Jia Wang, Mayo Clinic, Rochester, MN). ROI region of interest
The third concern associated with the use of bismuth shields is the ineffective use of radiation [14, 15]. Fig. 6 demonstrates this concern in a simplified illustration. For the anteriorly oriented X-ray beam, the figure shows how the presence of the shield reduces the radiation impinging onto the body, most effectively for the anterior organs (e.g., the breasts). The detector proportionally receives less flux as well and thus a corresponding increase in image noise. As such, from the perspective of the anterior beam, the shield gives rise to a largely proportionate effect on dose and image noise.
The situation is somewhat different for the posteriorly oriented beam. Here the X-ray beam that has gone through the patient and already deposited its dose in the patient is now blocked by the shield from reaching the detector. The beam has “dosed” the patient but is not being effectively captured by the detector to contribute to image formation with lowest possible noise. Therefore, the dose is partly wasted. This effect is more quantitatively illustrated in Fig. 7 through a simulation that shows how the addition of a shield (without changing the CT technique) can reduce the overall patient dose in the thorax by 4% and the breast by 17% (with presumed increase in noise). However, if one were to adjust the CT technique (e.g., increase the mA) such that the same noise level is maintained as that of the unshielded condition, but with the shield present, the breast dose remains unchanged and the overall dose to the thorax is increased by 11%. This example illustrates how the shield essentially reduces the ratio of image quality per dose: if by technique adjustments the dose is kept unchanged, the shield can lead to nosier images; conversely if the noise is held constant, the shield can increase dose to the patient.
Graph demonstrates the location-dependent depth dose from a posterior to an anterior surface of a cylindrical phantom emulating the thorax. Without shield, the dose is largely symmetrical. A bismuth shield when anteriorly located reduces the hypothetical breast dose by 17% and the thorax dose by 4%. With shield in place, if the mA is adjusted to achieve the same noise as that of the unshielded exam, the breast dose remains unchanged, but the thorax dose increases by 11%. The modeling was performed using an X-ray simulation platform (Xspect, Henry Ford Health System, Detroit, MI)
The pros: potential advantages of bismuth shields
With the notable disadvantages associated with the bismuth shields outlined above, the use of these shields seems hard to justify. However, there are a number of conditions and considerations when the use of such shields is warranted. These pertain to the ability to control radiation dose and image noise over a small section of the imaging field-of-view, the simplicity of the application, and the patient’s perception of safety.
As noted above, the application of the shield reduces the radiation dose but also involves a corresponding increase in image noise. Because radiation dose and image noise are generally inversely related to each other, it follows that if one is willing to tolerate a higher level of image noise, one can simply reduce the radiation dose instead of using the shield [16]. In doing so, one would be able to reduce the dose to all the organs during the examination, and not just a select organ. However, the shield enables the dose/noise manipulation to be carried out over a very limited section of the patient’s body. If a large anatomical region is being imaged, a clinician might not be willing to reduce the image quality across the entire region by setting a low global dose level. An example of this might be in whole-body PET/CT exams. Generally a sufficiently high level of dose is necessary to capture diagnostic-quality CT data from the patient’s body. However, that might unnecessarily expose the lens of the eye to excessive radiation dose. A simple application of an eye shield enables an effective reduction of dose to the lens of the eye (with a tolerable enhancement of noise only in that region) without setting the entire exam to a lower dose level and degrading the image quality across the entire field-of-view.
The second advantage of bismuth shields is in the simplicity of the application. In the very heterogeneous and complicated clinical environments of today, it is always preferable to use optimization strategies that are simple and easy to implement. In the example above, if a clinician decides to apply a global reduction of dose instead of using a shield, the exact level of adjustment to the CT technique factors that can achieve that dose reduction is not easy to determine. It requires detailed knowledge of technique factors associated with the CT system that are highly manufacturer-dependent and a high level of imaging physics expertise that is not readily accessible at a large number of non-academic imaging centers. Under these conditions the use of a shield can offer a simple and effective solution.
Finally, regardless of how practitioners might view the disadvantages of the shield, a patient or caretaker might have a significantly different perception of the shield’s role in mitigating radiation risk. One may argue that a health care provider should be able to provide a convincing explanation of the role and limitations of the shield. However, our collective failure as a community to convey even the most basic concepts of radiation risk to our patients and to the general public is evidence that the ability of the provider should not be overestimated in this regard. Furthermore, the actual risk associated with a single (reasonably optimized) CT exam is so small and so difficult to quantify that it is arguably more of a perceived risk than an actual risk. As such, an effort to mitigate this perceived risk may be more relevant than mitigating any theoretical “actual” risk. Efforts to impact or reduce the perception of risk on the part of the patient or caregiver could be as or more significant in the context of clinical practice than any small disadvantage to a portion of the image that results from the use of a shield. A parent might simply feel more comfortable about subjecting a child to CT radiation with the use of a shield, and the ~10% disadvantage that such use entails might be an acceptable price to pay for patient comfort and confidence.
Conclusion
If properly used, bismuth shields offer a methodology to reduce dose to targeted superficial organs. The dose reduction is associated with a corresponding increase in image noise in the section covered by the shield. It also involves a degradation of the quantitative accuracy of the CT numbers and a lower level of image quality per overall patient dose compared to when the shield is not used. Therefore, the use of such shields in clinical practice should be carefully examined. Factors that can impact the shield use consideration include the importance of quantitative precision, the specific anatomical application (e.g., whole-body PET/CT), the ability of the scanner to apply different-quality indexing protocols for different regions of the body, and the availability of virtual shielding. Ease of use and patient perception or preference are significant factors in the clinical practice that may favor the use of shields.
References
McCollough CH, Chen GH, Kalender W et al (2012) Achieving routine submillisievert CT scanning: report from the summit on management of radiation dose in CT. Radiology 264:567–580
International Commission on Radiological Protection (2007) The 2007 recommendations of the international commission on radiological protection, ICRP publication 103. Ann ICRP 37:2–4
Samei E, Tian X (2014) Determining organ dose: the holy grail. Pediatr Radiol [In press]
Duan X, Wang J, Christner JA et al (2011) Dose reduction to anterior surfaces with organ-based tube-current modulation: evaluation of performance in a phantom study. AJR Am J Roentgenol 197:689–695
Lungren MP, Yoshizumi TT, Brady SM et al (2012) Radiation dose estimations to the thorax using organ-based dose modulation. AJR Am J Roentgenol 199:W65–W73
Hopper KD, King SH, Lobell ME et al (1997) The breast: inplane x-ray protection during diagnostic thoracic CT — shielding with bismuth radioprotective garments. Radiology 205:853–858
Mukundan S, Wang PI, Frush DP et al (2007) MOSFET dosimetry for radiation dose assessment of bismuth shielding of the eye in children. AJR Am J Roentgenol 188:1648–1650
Coursey C, Frush DP, Yoshizumi T et al (2008) Pediatric chest MDCT using tube current modulation: effect on radiation dose with breast shielding. AJR Am J Roentgenol 190:W54–61
Fricke BL, Donnelly LF, Frush DP et al (2003) In-plane bismuth breast shields for pediatric CT: effects on radiation dose and image quality using experimental and clinical data. AJR Am J Roentgenol 180:407–411
Hohl C, Wildberger JE, Sü C et al. (2006) Radiation dose reduction to breast and thyroid during MDCT: effectiveness of an in-plane bismuth shield. Acta Radiologica 47:562-567
American Association of Physicists in Medicine (2012) Bismuth Shielding — AAPM position statement. http://www.aapm.org/publicgeneral/BismuthShielding.pdf. Accessed 27 May 2014
Leswick DA, Hunt MM, Webster ST et al (2008) Thyroid shields versus z-axis automatic tube current modulation for dose reduction at neck CT. Radiology 249:572–580
Kalra MK, Dang P, Singh S et al (2009) In-plane shielding for CT: effect of off-centering, automatic exposure control and shield-to-surface distance. Korean J Radiol 10:156–163
Geleijns J, Salvado Artells M et al (2006) Quantitative assessment of selective in-plane shielding of tissues in computed tomography through evaluation of absorbed dose and image quality. Eur Radiol 16:2334–2340
Vollmar SV, Kalender WA (2008) Reduction of dose to the female breast in thoracic CT: a comparison of standard-protocol, bismuth-shielded, partial and tube-current-modulated CT examinations. Eur Radiol 18:1674–1682
Wang J, Duan X, Christner JA et al (2011) Radiation dose reduction to the breast in thoracic CT: comparison of bismuth shielding, organ-based tube current modulation and use of a globally decreased tube current. Med Phys 38:6084–6092
Conflicts of interest
Dr. Samei receives grant support for research from GE Healthcare, Siemens Healthcare and Carestream Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Samei, E. Pros and cons of organ shielding for CT imaging. Pediatr Radiol 44 (Suppl 3), 495–500 (2014). https://doi.org/10.1007/s00247-014-3084-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-014-3084-z