Abstract
Permanent reclamation of tailings generated by surface mining in the Canadian oil sands may be achieved through the creation of end pit lakes (EPLs) in which tailings are stored in mined-out pits and capped with water. However, these tailings contain high concentrations of dissolved organics, metals, and salts, and thus surface water quality of EPLs is a significant concern. This is the first study to investigate the chronic toxicity of surface water from Base Mine Lake (BML), the Canadian oil sands first large-scale EPL, to aquatic invertebrates that play a vital role in the early development of aquatic ecosystems (Chironomus dilutus and Ceriodaphnia dubia). After exposure of C. dilutus larvae for 23 days and C. dubia neonates for 8 days, no mortality was observed in any treatment with whole BML surface water. However, the emergence of C. dilutus adults was delayed by nearly 1 week, and their survival was significantly reduced (36%) compared with the controls. Reproduction (fecundity) of C. dubia was reduced by 20% after exposure to 2014 BML surface water; however, the effect was not observed after exposure to BML surface water collected a year later in 2015. Despite some adverse effects, the results of this study indicate that BML surface water quality is improving over time and is able to support certain salt-tolerant aquatic organisms. Because salinity within BML will persist for decades without manual intervention, the ecological development of the lake will likely resemble that of a brackish or estuarine ecosystem with reduced diversity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
The extraction and separation of bitumen from surface-mined ore in the Athabasca region of Canada’s Alberta oil sands results in the generation of vast quantities of oil sands process-affected water (OSPW) and fluid fine tailings (FFT). These waste products contain high concentrations of dissolved organic acids, metals, and salts that adversely affect a range of physiological endpoints in aquatic organisms, such as fish and invertebrates (Alharbi et al. 2016a; Anderson et al. 2012a; Bartlett et al. 2017; Bauer et al. 2017, 2019; Kavanagh et al. 2011; Lari et al. 2017, 2018, 2019; Leclair et al. 2013; MacDonald et al. 2013; McNeill et al. 2012; van den Heuvel et al. 2012; Wiseman et al. 2013a). In many cases, the aquatic toxicity of whole OSPW has been mitigated or eliminated following the reduction or removal of the dissolved organic fraction via physical and chemical treatments, such as ozonation and activated charcoal (Anderson et al. 2012b; He et al. 2010, 2011, 2012a, b; Lyons et al. 2018; McQueen et al. 2017; Wiseman et al. 2013b). Thus, the dissolved organic fraction of OSPW is now generally accepted as being the primary driver of aquatic toxicity. Historically, this has been attributed to the high concentrations of naphthenic acids (NAs) found in OSPW. However, more recent studies have shown that NAs (O2− compounds) comprise only a portion of the total organic fraction and that other compounds, such as the O2+, NO+, and SO+ classes, also contribute to the toxicity of OSPW (Alharbi et al. 2016b; Morandi et al. 2015, 2016; Sun et al. 2017).
Due to a lack of effective and economical large-scale treatment methods, surface mine operators have accumulated nearly 1 trillion L of fluid tailings (AEP 2015a) stored in ponds covering more than 88 km2 of land (AEP 2015b). One of the proposed methods for permanent reclamation is the establishment of end pit lakes (EPLs) in which FFT is stored in mined-out ore pits and capped with a mixture of OSPW and fresh water. In doing so, oil sands mine operators can use these readily available waste products as backfill materials to solve the issues of accumulation and storage, while relying on natural aging processes as well as manual intervention to improve surface water quality over time. In order to test the large-scale effectiveness of EPLs within the Canadian oil sands, Syncrude established Base Mine Lake (BML) in December 2012—an 8 km2 EPL containing 186 billion L of FFT capped with 58 billion L of fresh water and OSPW (Dompierre et al. 2016, 2017). Detailed geochemical and hydrological characterization of BML is ongoing and described elsewhere (Arriaga et al. 2019; Dompierre et al. 2016, 2017; Risacher et al. 2018; Tedford et al. 2019; White and Liber 2018). Once acceptable surface water quality has been achieved within BML, the lake may be incorporated into the surrounding watershed for permanent reclamation or discharged into another regional water body, such as the Athabasca River. However, while the toxicity of OSPW may be reduced from aging due to natural degradation and transformation of organic acids, some toxicity is still observed in OSPW samples taken from smaller-scale tailings ponds that have been aging for over a decade (Anderson et al. 2012b; Wiseman et al. 2013a). Thus, the time required for BML to reach acceptable surface water quality remains a significant concern.
In a year-long wetland microcosm study, Toor et al. (2013a) showed that although NA concentrations naturally decreased over time leading to reduced acute toxicity, some residual fraction of NAs were recalcitrant and associated with persistent chronic toxicity. It was discovered that natural in situ degradation was dependent upon structure, with small and slightly unsaturated NAs degrading significantly faster than large and highly unsaturated NAs (Toor et al. 2013b). Thus, the use of natural biodegradation processes within small-scale tailings ponds or large-scale EPLs will likely be insufficient to completely remove dissolved organic-associated toxicity from OSPW in a timely manner (Anderson et al. 2012a; Toor et al. 2013b). Nonetheless, in extremely long reclamation strategies in which OSPW will age for decades (such as EPLs), the contribution of the organic fraction to toxicity is expected to slowly diminish over time. However, most inorganic constituents, such as major ions, will likely persist in systems such as BML indefinitely (Dompierre et al. 2016, 2017; White and Liber, 2018). Many OSPW toxicity studies acknowledge elevated concentrations of major ions, such as sodium (Na+), chloride (Cl−), sulfate (SO42−), and bicarbonate (HCO3−), but rarely discuss their influence on the observed toxicity. Results for the specific toxicity of OSPW-associated metals are even more limited. However, recent studies have begun to highlight the toxicity of inorganic constituents in aged OSPW, especially to sensitive aquatic invertebrates, and the potential significance this may have for long-term oil sands reclamation (Bauer et al. 2019; McQueen et al. 2017; White and Liber 2018).
Although the aquatic toxicity of whole OSPW has already been well characterized, historically most of these studies have examined the effects of raw untreated water on fish and focused on short exposures for release scenarios. In the case of BML, it is more appropriate to examine the potential long-term effects of the more dilute surface water on lower trophic level organisms that will play a role in the colonization and ecological development of aquatic EPL ecosystems. In this study, we investigated the chronic toxicity of whole BML surface water as well as reconstituted BML saltwater to Chironomus dilutus and Ceriodaphnia dubia, two species of freshwater invertebrates ubiquitous throughout Canadian aquatic ecosystems. Along with other common aquatic invertebrates, such as Daphnia and Hyalella, these organisms fulfill a vital role in aquatic ecosystems by converting primary producers (phytoplankton and bacteria) into animal protein. Thus, they form an important link in aquatic food chains and comprise a significant portion of the diet of higher trophic level organisms, such as juvenile fish and waterfowl (Environment Canada 1997, 2007). We examined apical endpoints, such as survival, growth, and reproduction, to assess the early surface water quality of BML and to predict potential population-level effects as the lake develops over time.
Materials and Methods
Test Organisms
All tests were conducted using organisms from in-house cultures at the University of Saskatchewan Toxicology Centre (Saskatoon, SK, Canada) maintained according to Environment Canada Biological Test Method guidelines (1997, 2007). Chironomus dilutus mass cultures were raised in 15-L aquaria containing aerated, carbon- and bio-filtered municipal water from the City of Saskatoon, and a thin layer of silica sand (particle size 200–400 μm). Cultures were fed 15 mL of Nutrafin fish flake slurry (Rolf C. Hagen Inc., Montreal, QC) three times per week, and water was changed at least once per week. Ceriodaphnia dubia mass cultures were raised in 2-L glass jars containing moderately hard reconstituted water (MHRW) and fed 15 mL of algae (Raphidocelis subcapitata, 3.0-3.5x107 cells/mL) and 4 mL of YCT (yeast, cerophyll, and trout chow) daily; water was changed at least three times per week. Both C. dilutus and C. dubia cultures were kept in an environmental chamber maintained at 23 ± 1 °C with a 16:8 h light:dark photoperiod (< 600 lx for C. dubia).
Exposure Waters
Surface water samples were collected from BML in October 2014 and used as exposure water in the C. dilutus toxicity test. Additional samples were obtained in August 2015, and as a result, both 2014 and 2015 BML surface waters were used in the subsequent C. dubia toxicity test. Detailed information on the sampling and chemical composition of these BML surface water samples is reported by White and Liber (2018). Based on the lack of observed effects on survival and growth in diluted 25% and 50% 2014 BML surface water treatments in a C. dilutus pilot study (not shown), only the fully concentrated (100%) 2014 BML surface water was used in the partial lifecycle test. Conversely, the C. dubia toxicity tests were performed with additional 25% and 50% BML dilution treatments due to the higher sensitivity expected of C. dubia from the results of a preliminary NaCl salinity toxicity test (refer to Supplementary Material). Culture water for each test organism was used as the freshwater control and dilution water in each of the respective toxicity tests to remove the need for acclimation. As a result, the water chemistry of the freshwater control in the C. dilutus test varied slightly from that of the C. dubia test. To account for the potential effects of the elevated salinity found within BML surface water, reconstituted saltwater treatments were also prepared and tested to mimic the major ion composition of each BML surface water treatment (100% saltwater for the 2014 BML surface water in the C. dilutus test, and 25%, 50%, and 100% saltwaters in the C. dubia test for both 2014 and 2015 BML surface waters). Recipes, water quality parameters, and major ion concentrations for culture freshwaters and reconstituted saltwaters are listed in Tables S1 and S2. All chemical salts were obtained from Fisher Scientific (Ottawa, ON, Canada). Conventional water quality parameters (temperature, conductivity, pH, hardness, alkalinity, ammonia, and dissolved oxygen) were measured in the Water Quality Laboratory at the University of Saskatchewan Toxicology Centre. Major ion concentrations were analyzed by Maxxam Analytics (Calgary, AB, Canada). More detailed information on chemical analyses is described by White and Liber (2018).
Chironomus dilutus Partial Lifecycle Assessment
Although C. dilutus egg masses have been found to be very resistant to contaminants, first-instar larvae are the most sensitive life stage, with tolerance progressively increasing with age and instar (Gauss et al. 1985). To be as environmentally relevant as possible, the partial lifecycle test was initiated (Day 0) with an egg mass placed directly into each of three 15-L aquaria containing silica sand and one of the exposure waters (culture freshwater, 100% reconstituted 2014 BML saltwater and 100% 2014 BML surface water). The three egg masses were all < 24 h old, approximately the same size, and isolated from adults from a single in-house culture and breeding jar. The aquaria were fed 15 mL of Nutrafin slurry approximately every other day, following normal culturing procedures, and daily observations were made on the appearance of each egg mass. After 11 days of exposure, the second phase of the exposure was initiated with 100 larvae randomly isolated from each aquarium. An additional five groups of ten larvae were randomly selected and isolated from each aquarium and then oven dried at 60 °C for 72 h to determine average 11-day larval dry weights for each treatment (Supplementary Material).
The second phase of the exposure period was conducted in accordance with standard Environment Canada Biological Test guidelines (1997) with 100 second-instar larvae randomly isolated from each exposure aquarium and transferred in groups of 10 into 300-mL lidded tall form glass beakers containing 50 mL of silica sand and 200 mL of exposure water. Since the organisms had already been exposed for 11 days at this point in the test, they were transferred from the exposure aquaria into the glass test vessels with their larval cases intact. To investigate both larval and adult endpoints, ten replicates of ten organisms (100 organisms per treatment) were used so that half of the test vessels could be terminated before pupation (Day 23), whereas the remaining five test vessels in each treatment continued the exposure through to adult emergence (until Day 39). Each beaker of ten organisms was continuously aerated and fed 60 µL of Nutrafin slurry daily; water changes (80–90% of volume, approximately 175 mL) and water chemistry (temperature, dissolved oxygen, pH, conductivity, total hardness, alkalinity, and total ammonia) were performed every other day. All test vessels were maintained under the same environmental conditions as previously described for the culture aquaria. Observations on survival as well as larval activity and behavior were recorded daily. Once the first pupation was observed (Day 23), half of the test vessels in each treatment were terminated and final observations of larval survival were recorded. These larvae were then removed from their sediment cases and oven dried. The remaining five test vessels in each treatment continued with the exposure unchanged, and daily observations were expanded to take note of the timing of larval pupation and adult emergence. Subsets of individual organisms from each treatment, as well as larval cases, were collected from test vessels at test termination and stored in 70% ethanol for morphological and structural characterization.
Ceriodaphnia dubia Survival and Reproduction Assessment
The C. dubia toxicity test was conducted in accordance with standard Environment Canada Biological Test guidelines (2007) using 25-mL borosilicate glass test tubes containing 15 mL of exposure water, 250 µL of algae (R. subcapitata), 25 µL of YCT, 1.2 µg of sodium selenate (Na2SeO4), and 0.5 µg of vitamin B12. Each test tube contained a single < 24-h-old C. dubia neonate obtained from in-house cultures that had produced a minimum of three healthy broods prior to test initiation. All exposure waters were aerated for at least 48 h before setup and were continuously aerated throughout the entire test before each daily renewal. Each treatment contained ten replicates (10 organisms total) held in test tube racks maintained under the same environmental conditions as the mass cultures (experimental design is depicted in Fig. S1). Test organisms were transferred to clean test tubes containing fresh exposure water, food, and nutrient solutions daily using transfer pipettes. During this process, the orientation and order of treatment groups were randomized to minimize the potential effects of small variations in chamber light intensity. Observations on mortality, brood counts, and general appearance were recorded daily before water changes. Water-quality parameters (temperature, dissolved oxygen, pH, conductivity, total hardness, and alkalinity) were measured daily during water changes: new test solutions on Days 0, 2, 4, and 6; old test solutions on Days 1, 3, 5, and 7. Due to the small volume of water contained within each test tube, water quality for “old” water was measured as a composite of all ten replicates per treatment. Test endpoints were survival (8 days) and the total number of neonates produced in the first three broods (≤ 8 days).
Data and Statistical Analysis
Due to the percentage-based concentrations of the treatments in both tests (0–100%) no dose–response curves were calculated. Instead, to compare the effects among the freshwater control, saltwater, and BML surface water exposures (as well as differences between 2014 and 2015), endpoints from each treatment of the C. dilutus (survival, larval, and adult dry weights) and C. dubia (survival, brood counts) toxicity tests were analyzed using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc tests. If results did not pass the Shapiro–Wilk normality test, significant differences among treatments were instead determined using Kruskal–Wallis one-way ANOVA on Ranks followed by Dunn’s post hoc tests. All data and statistical analyses were performed and plotted with Sigmaplot version 12.0 software (Systat Software Inc., San Jose, CA) and conducted with α = 0.05.
Results and Discussion
Chironomus dilutus Partial Lifecycle Assessment
Chironomids were exposed to the treatment waters directly as egg masses to mimic potential introduction to BML via surface water oviposition and to capture effects on the earliest life stage and initial case building activity. Even with the longer and more environmentally relevant exposure scenario, no significant effects on larval survival were observed in any treatment. Furthermore, exposure to the reconstituted saltwater control had no statistically significant effect on any endpoint, which was consistent with the lack of effects observed in the saltwater controls of Anderson et al. (2012a, b). This apparent lack of effect of salinity was expected, as chironomids are generally much more tolerant of elevated major ion concentrations than other organisms (C. dilutus 20-day IC10 of 2316 mg Cl−/L compared to C. dubia 7-day IC25 of 454 mg Cl−/L; Elphick et al. 2011). Complete survival in the BML treatment was also consistent with exposures of chironomids to aged OSPW from similar reclamation ponds (Anderson et al. 2012b) but in contrast to the significant 45% larval mortality observed in exposure to fresh OSPW (Anderson et al. 2012a, b).
Although there were no effects on survival, hatching of the egg masses in the BML surface water treatment appeared delayed and corresponded with a small but significant 27% reduction in larval biomass after 23 days of exposure compared with the freshwater and saltwater controls (Fig. S2). This was also similar to the reductions observed in other oil sands reclamation ponds (approximately 20%; Anderson et al. 2012b), but significantly less than the 64% reduction in larval biomass observed after 10 days of exposure to fresh OSPW (Anderson et al. 2012a, b). Slight decreases in larval activity, biomass, and structural integrity of sediment cases in the BML surface water treatment were also consistent with observations by Anderson et al. (2012b) for other reclamation ponds. Despite these reductions, no statistically significant differences were observed in the final biomass of successfully emerged adults after accounting for sex (females are approximately 2.5 × larger than males due to morphological differences; Fig. S3). However, it is important to note that the time required to reach final biomass varied depending on when adult emergence occurred. It is also worth noting that exposure to environmental stressors often leads to greater proportions of males in chironomid populations (Liber et al. 1996); however, no effects on sex ratio were observed in either this study (Fig. S4) or that of Anderson et al. (2012b).
Adult Emergence
Adult emergence in the saltwater control was delayed by 2 days compared with the freshwater control, but still followed the same pattern and rate, and resulted in a similar number of successfully emerged adults (Fig. 1). Conversely, adult emergence in the BML surface water treatment was reduced by 40% and delayed by up to 5 days compared with the controls. Thus, even though the adult chironomids in the BML surface water treatment were the same size as those in the freshwater and saltwater controls, they required nearly an additional week of growth to achieve it. A similar effect was observed in organisms exposed to fresh OSPW, although the delay in emergence was significantly longer (approximately 11 days), while no significant delay was observed in organisms exposed to other oil sands reclamation pond waters (Anderson et al. 2012a, b).
Cumulative successful emergence of Chironomus dilutus adults after exposure to a freshwater control, saltwater control (100% reconstituted 2014 BML saltwater), and 100% 2014 BML surface water. Emergence counts were conducted daily. Exposure duration ranged from 23 to 39 days depending on the day of emergence
To more accurately identify effects on the survival and emergence of C. dilutus adults, organisms were grouped categorically at test termination based on those that: (A) emerged completely from their exuvia and were able to leave the water’s surface (survival, successful emergence); (B) emerged completely from their exuvia but were unable to leave the water’s surface and drowned (mortality, successful emergence); (C) were unable to completely emerge from their exuvia (mortality, failed emergence); (D) died as pupae (mortality, failed emergence); and (E) died as larvae (mortality, failed pupation). Average percentages of organisms observed within each category of each treatment are shown in Fig. 2. Average successful emergence (survival A) in the freshwater control, saltwater control, and BML surface water were 60%, 64%, and 36%, respectively. This successful emergence rate in BML surface water was almost identical to that of chironomids exposed to the other reclamation pond waters (approximately 35%; Anderson et al. 2012b), and significantly higher than those exposed to fresh OSPW (13%; Anderson et al. 2012a). It should be noted that the successful emergence rate of the freshwater control in this study was lower than desired at only 60%, which may have been due to overcrowding of test vessels during later stages of the exposure. The studies by Anderson et al. (2012a, b) purposely deviated from standard testing procedure by using larger 1-L beakers containing 500 mL of test solution and observed control emergence rates of approximately 80%. This suggests that improvements to test design may lead to an increase in the emergence success of organisms exposed to BML surface water. This is further supported by the fact that the successful pupation rate in BML surface water (90%; A + B + C + D) was approximately 15–20% higher than the rates observed in the other reclamation pond waters (approximately 70–75%; Anderson et al. 2012b).
Average percentage of Chironomus dilutus organisms observed within five defined categories of survival (A) and mortality (B, C, D, E) after exposure to a freshwater control, saltwater control (100% reconstituted 2014 BML saltwater), and 100% 2014 BML surface water. Exposure duration ranged from 23 to 39 days depending on the day of emergence (A, B, C). Error bars represent one standard deviation of the mean. Significant differences among treatments within each category were determined using one-way ANOVA followed by Tukey’s post hoc test and are denoted by lowercase letters
Organic Acids
Based on the results of a previous study by Liber et al. (1996), Anderson et al. (2012b) suggested that reductions in pupation and adult emergence were a result of reduced larval biomass caused by NA-induced stress or alterations in bioenergetics. Reductions in larval activity and integrity of cases were also attributed to stress and decreased energy stores (Anderson et al. 2012a). A later study by Wiseman et al. (2013a) found increased abundances of the oxidative stress-related transcripts gst, cat, gpx, increased abundances of the endocrine disruption-related transcripts usp, esr, err, and increased lipid peroxidation in C. dilutus exposed to fresh OSPW. As a result, oxidative stress-induced apoptosis was proposed as the cause for decreased survival and growth via metabolism of organic compounds (NAs) in larvae resulting in the production of reactive oxygen species (Wiseman et al. 2013a). Additionally, effects on development (reduced pupation and emergence) were attributed to disruption of endocrine function, likely due to smaller organic acids with estrogen-like properties (Wiseman et al. 2013a). These effects were not observed in C. dilutus larvae exposed to reclamation pond water, which supports the conclusion that aging reduces concentrations of NAs and their associated toxicity (Wiseman et al. 2013a). The results of this study support the organic acid fraction as being the primary driver of OSPW toxicity to C. dilutus as similar effects between surface water from BML and the other water-capped oil sands reclamation ponds corresponded with similar concentrations of NAs (13–25 mg/L; Anderson et al. 2012b; White and Liber 2018). It is important to note that even though the toxicity of aged reclamation pond water was significantly reduced compared with fresh OSPW, it still contained an appreciable concentration of NAs and caused a significant reduction in the success of C. dilutus adult emergence (Wiseman et al. 2013a). Anderson et al. (2012a) also found that ozonation of fresh OSPW significantly reduced the concentration of NAs and their associated toxicity, but a small concentration and some effects remained. This suggests that some recalcitrant fraction of NAs will likely persist within BML surface water and continue to affect the growth and development of sensitive invertebrate species for some time.
Surface Tension
The most interesting result from this study was that the reduction in emergence success of organisms exposed to BML surface water appeared to be due to a significant increase in the proportion of fully emerged adults that were unable to leave the surface of the water and drowned (mortality B). The average mortality values for these organisms within the freshwater control, saltwater control, and BML surface water treatments were 6%, 10%, and 32%, respectively. Unfortunately, Anderson et al. (2012a, b) did not qualitatively assess adult emergence and therefore it is unknown whether a similar phenomenon occurred in their studies. Based on qualitative observations of water samples throughout the duration of this test, the surface tension of BML surface water was noticeably lower than that of the freshwater and saltwater controls, which may have been at least partly responsible for the observed effects on emergence. Studies characterizing OSPW and its dissolved organic fraction have frequently made note of the surfactant-like properties and structure of NAs (Alharbi et al. 2016a; Clemente and Fedorak 2005; Frank et al. 2008; Pourrezai et al. 2011; Schramm and Smith 1985). However, this surfactant effect in reducing surface water tension of OSPW and its impact on aquatic organisms has not been previously demonstrated. This is unsurprising, as this effect would have the most impact on insects, especially those that require surface tension to transition from an aquatic pupal stage to a terrestrial adult stage (such as C. dilutus), which have only been briefly examined in the context of OSPW toxicity. This apparent inability to escape the surface of BML water may also be a result of decreased energy, as previously discussed, or a combination of this effect with reduced surface tension.
Ceriodaphnia dubia Survival and Reproduction
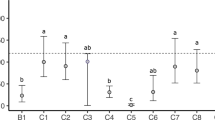
Aside from 90% survival in the freshwater control, 100% survival was observed in all C. dubia test treatments after 8 days of exposure (Fig. S5). Furthermore, no statistically significant differences were observed in reproduction among any of the treatments, or between the two years, at the 25% and 50% dilution levels, except for 2015 BML surface water which caused a small but statistically significant increase in reproduction. Conversely, statistically significant rates of reproduction inhibition were observed in both the 2014 and 2015 100% BML saltwater controls (53% and 52% inhibition, respectively). Some inhibition of reproduction (19%) was observed in the 100% 2014 BML surface water treatment; however, the 100% 2015 BML surface water did not cause any statistically significant inhibition of reproduction (Fig. 3).
Average number of neonates produced per Ceriodaphnia dubia adult in the first three broods (A), and calculated reproduction inhibition (B), after exposure to a freshwater control (moderately hard reconstituted water, MHRW), saltwater control (reconstituted 2014 and 2015 BML saltwater), and 2014 and 2015 BML surface water. The BML surface water treatments were tested at 25%, 50%, and 100% strength (dilution in MHRW) with paired saltwater controls at the same concentrations. Error bars represent 1 standard deviation of the mean. Significant differences among treatments were determined using one-way ANOVA followed by Tukey’s post hoc test (n = 6–10, α = 0.05) and are denoted by letters
Salinity
We have previously identified that Na+, Cl−, and HCO3− are the major ions that comprise the vast majority of the salinity within BML surface water (92% of total osmolarity) and also pose the highest potential toxicological risk to sensitive freshwater aquatic organisms such as C. dubia (White and Liber 2018). Studies of chronic toxicity of major ions and salinity on the growth and reproduction of cladocerans, such as C. dubia, generally attribute effects to altered energy allocation due to impairment of osmoregulation (Erickson et al. 2017; Freitas and Rocha 2011; Lasier and Hardin 2010; Mount et al. 2016; Simmons 2012; Soucek and Dickinson 2016). The hemolymph of freshwater cladocerans, such as Ceriodaphnia, is hyperosmotic relative to the external environment; thus, osmoregulation is largely dependent on the active uptake and concentration of major ions against a concentration gradient, such as Cl− exchange for HCO3− via active Na+/K+ ATP transport pumps (Aladin 1991; Aladin and Potts 1995; Hoke et al. 1992). Because the body wall of cladocerans has very low permeability to water and ions, these transport processes occur via specialized, highly permeable membranes containing mitochondria-rich ion-transporting cells (also known as chloride cells or ionocytes) localized at the epipodites—gill-like structures attached to the base of the legs (Aladin 1991). In environments with increased salinity, such as BML, concentration gradients across these membranes are altered, resulting in greater energy required to maintain homeostasis and leading to stress. Several other related mechanisms of effect also have been identified, including direct damage to embryos (Freitas and Rocha 2011), altered membrane permeability (Baker et al. 2017), competition at uptake sites (Simmons 2012; Soucek and Dickinson 2016), and decreased filter-feeding rates (Soucek 2007).
The effects of salinity within BML surface water on C. dubia can be predicted as a function of total osmolarity and Ca2+ concentration using the acute ‘general ion toxicity submodel’ developed by Erickson et al. (2018; Eq. 16), as previously demonstrated (White and Liber 2018). Based on the water chemistry reported in Table S2, the model provided C. dubia 48-h osmolarity LC50 values of 74 mM for the 100% BML surface water and 78 mM for the 100% saltwater controls. These values were approximately 30% higher than the measured osmolarity in the 2014 and 2015 100% BML surface water and saltwater treatments (51–55 mM), which seemed to support the lack of mortality observed in the saltwater treatments. A subsequent model for the chronic toxicity of major ions to C. dubia was recently published by the same group (Mount et al. 2019; Eq. 6). The chronic toxicity model is based on the same “general ion toxicity submodel” parameters (total osmolarity and Ca2+ concentration) described by Erickson et al. (2018), but instead estimates a 7-day IC50 for C. dubia reproduction. Based on the water chemistry reported in Table S2, the model provided C. dubia 7-day osmolarity IC50 values of 36–37 mM for the 100% BML surface waters and 100% saltwater controls. These values were consistent with the results of Mount et al. (2019), which indicate that the acute-to-chronic ratio for the toxicity of Na salts is approximately 2 (1.9, 1.8, and 2.3 for NaCl, Na2SO4, and NaHCO3, respectively). Compared with the acute toxicity model, the predicted chronic toxicity IC50 values were approximately 30% lower than the measured osmolarity in the 100% BML surface water and saltwater controls, indicating > 50% inhibition of reproduction. However, the results of the preliminary NaCl toxicity test yielded a somewhat higher 7-day C. dubia osmolarity IC50 of 52 mM, which indicated approximately 50% inhibition of reproduction in the 100% BML surface waters and 100% saltwater controls (refer to Supplementary Material for the full discussion of the NaCl toxicity test results; Figs. S6 and S7). Overall, these predictions were relatively consistent with the 52–53% inhibition observed in the 100% saltwater controls but significantly higher than the 0–19% inhibition observed in the 100% BML surface water treatments.
Water Hardness and Trace Elements
These discrepancies may be at least partially explained by differences in water hardness among the different treatments and tests. Water hardness has been shown to have a significant effect on mitigating the toxicity of salinity to sensitive freshwater organisms, such as C. dubia (Elphick et al. 2011; Erickson et al. 2017; Lasier and Hardin 2010; Mount et al. 2019, 2016). The NaCl toxicity test was performed using a stock solution spiked directly into the 60 mg CaCO3/L MHRW C. dubia culture and dilution water, whereas the reconstituted BML saltwater was prepared to have a water hardness as close to natural BML surface water as possible (100 mg CaCO3/L). However, due to the high pH and HCO3−/CO32− concentration within the reconstituted saltwaters, it was difficult to maintain the desired water hardness of 100 mg CaCO3/L, because the Ca and Mg visibly precipitated out of solution over time as carbonates. The same phenomenon was observed in the reconstituted saltwater prepared in the C. dilutus test, although it had no effect on the results due to the higher tolerance of C. dilutus to salinity. In an attempt to correct for this decrease, the 100% saltwater solutions used in the C. dubia toxicity test were prepared at a higher initial water hardness (120 mg CaCO3/L), but still dropped to 70–80 mg CaCO3/L after a few days of aeration. Substantial precipitation and settling of CaCO3 also occurred in C. dubia tests at high pH by Mount et al. (2016), with a 92% reduction observed in the concentration of dissolved CaCO3 after only 48 h (1500 mg/L to 120 mg/L).
It also is worth noting that Mount et al. (2016) used amended saltwaters instead of reconstituted saltwaters in their toxicity tests (chemical salts were added to natural field-collected water instead of laboratory water). Natural field-collected water contains background alkalinity without the need for the excessive addition of chemical NaHCO3 salt used in this study, while also containing trace nutrients which provide beneficial effects and a more realistic exposure scenario (Mount et al. 2016). This is supported by the hormesis-like effects observed in the 25% and 50% dilution 2015 BML surface water treatments, which were likely due to the very low concentrations of trace elements measured within BML surface water (White and Liber 2018), many of which are essential micronutrients for C. dubia (Naddy et al. 1995). It is therefore reasonable to assume that improving the saltwater formulation to more adequately mimic the chemical composition of BML surface water would further reduce the observed inhibition of C. dubia reproduction. However, only increasing the water hardness by 20 mg CaCO3/L and adding some trace nutrients to the 100% saltwater controls seems insufficient to achieve the substantial amelioration of reproduction success observed in the 100% BML surface water treatments.
Dissolved Organic Carbon
The only other difference in the chemical composition between the reconstituted saltwaters and BML surface waters was the absence of dissolved organic carbon (DOC). It was therefore hypothesized that the presence of DOC within BML surface water (23–24 mg/L) may have had an attenuating effect on the toxicity of the major ions. This apparent antagonism between major ion toxicity and dissolved organic acids (NAs) has previously been observed in pilot studies of OSPW exposure to both C. dubia and chironomids (Kennedy 2012; Turcotte et al. 2009), as well as in a recent study by Bauer et al. (2019). Attenuating salt-organic acid interactions also have been observed in toxicity tests of major ions to C. dubia (Lasier and Hardin 2010) and NAs to fathead minnows (Kavanagh et al. 2012). In both cases, the effect was only observed in tests with HCO3 salts, but not Cl or SO4 salts. The conservation of this bicarbonate-organic acid interaction between different species and research groups (and the apparent lack of similar interactions with Cl and SO4) suggests a specific mechanism of action and supports the conclusions of both groups that the effect may be due to altered uptake (Kavanagh et al. 2012; Lasier and Hardin 2010). More recent studies of HCO3− toxicity (Harper et al. 2014; Vera et al. 2014) attribute effects to the disruption of active Cl−/HCO3− exchange and regulation resulting in the inhibition of Cl− uptake by cladocerans based on the mechanism proposed by Hoke et al. (1992). High concentrations of HCO3− also have been observed to cause significant reductions in Na+/K+ ATP pump activity in fathead minnows (Farag and Harper 2014). However, the influence of dissolved organic compounds on this mechanism and its potential effect within BML surface water remain unknown.
Ecological Development of Base Mine Lake
Although salinity within BML surface water likely poses the highest concern for freshwater organisms with rapid development and reproduction, our results suggest that BML surface water quality has reached an acceptable level to support populations of certain salt-tolerant invertebrates. Furthermore, the reduction in reproduction inhibition observed in the C. dubia test (19% in 2014, 0% in 2015) suggests that BML surface water quality continues to improve over time. This is supported by the discovery of wild Daphnia pulex in a sample of BML surface water collected in August 2016 (White and Liber 2018). Recent studies also have discovered a relatively dynamic and diverse microbial community within the surface water of BML (Mori et al. 2019; Richardson et al. 2020), particularly in the upper oxygenated zone where they may serve as a potential food source for higher trophic level organisms such as daphnids. However, Richardson et al. (2020) also detected a low relative abundance of photosynthesizers, attributed to the high turbidity measured within BML surface water (Tedford et al. 2019), which may ultimately limit the potential for robust ecological development within BML.
For salt-tolerant organisms with longer and more complex lifecycles, such as C. dilutus, long-term exposure to BML surface water may adversely affect development due to the presence of persistent dissolved organic acids. Unfortunately, neither the exposure duration used in this study, nor those of Anderson et al. (2012a, b), extended for the full lifecycle of C. dilutus. Without knowledge of the effects of BML surface water exposure on adult reproduction and the development of subsequent progeny, it is difficult to predict population-level effects in the field. It has previously been shown that a reduction in larval growth can be associated with a decline in the reproductive output of female chironomids (Sibley et al. 1997), although the ovipositing success of females and their second-generation offspring may be unaffected (Liber et al. 1996). Despite observed delays and reductions in larval growth, the similar biomass of all successfully emerged adults in this study suggests that exposure to BML surface water may not significantly affect reproductive success. It has been proposed that delays in the time to emergence of C. dilutus adults, as well as altered behavior and case structure, may increase the susceptibility of larvae and pupae to predators (Anderson et al. 2012b; Sibley et al. 1997). However, this will only be of concern once BML surface water is able to support higher trophic level organisms, such as fish, at which point the toxicity of BML surface water and prevalence of these effects will likely be even further reduced. Delays in adult emergence may also affect mating success if they are sex-specific, such as those observed by Anderson et al. (2012a), although the lack of effect on sex ratio in successfully emerged adults in BML surface water suggests otherwise. Of all test endpoints, the reduced emergence success (36%) of adults exposed to BML surface water is therefore the most concerning with respect to long-term ecosystem health. Despite this reduction, adults who did successfully emerge appeared normal and healthy, which may indicate that reproduction and population growth will still occur in BML, albeit at lower rates compared with natural aquatic ecosystems. This prediction is supported by the results of previous surveys of aquatic insect populations and colonization in OSPW-affected constructed wetlands of the Canadian oil sands region. Whelly (2000) observed similar reductions in the growth and survival of OSPW-exposed Chironomus larvae in these systems but no significant effects on the rates of oviposition or total adults. However, both Whelly (2000) and Leonhardt (2003) observed a general trend of decreased species abundance and richness that indicated functional, but low-diversity ecosystems.
Conclusions
This is the first study to examine the toxicity of BML surface water to freshwater invertebrates involved in the colonization and early development of aquatic ecosystems. Due to the more dilute nature of BML surface water compared with raw OSPW, no statistically significant mortality was observed after chronic exposure of C. dilutus larvae and C. dubia neonates to either whole BML surface water or reconstituted BML saltwater. However, a significant delay in the growth and time to emergence, as well as a reduction in emergence success, was observed in adult C. dilutus over the course of the partial lifecycle test (approximately 25–40 days). These effects were attributed to the dissolved organic compounds within BML surface water, because no similar effects were observed in the saltwater controls. As a result, these effects are expected to diminish over time as BML continues to age and will likely only influence the total number of organisms within the ecosystem, because adults who did emerge successfully appeared to be healthy and were of the same size, shape, and sex ratio as those in the controls. In the C. dubia tests, a 20% reduction in reproductive output (fecundity) was observed in organisms exposed for 8 days to whole BML surface water collected in 2014 but was not observed in organisms exposed to BML surface water collected the following year (2015). These results, in addition to the discovery of wild Daphnia pulex adults and neonates in a BML surface water sample collected in 2016 (White and Liber 2018), indicate that the surface water quality of BML continues to improve over time and has reached an acceptable level to support populations of certain salt-tolerant aquatic organisms. However, salinity within BML will continue to be a persistent concern for decades, and thus the ecological development of the lake will likely resemble that of a brackish or estuarine ecosystem with reduced diversity and possibly reduced abundance.
References
AEP (2015a) Mineable oil sands fluid tailings volume by facility as of the end of 2013. Oil Sands Information Portal, Alberta Environment and Parks, Edmonton, AB. Dataset no. 540. http://osip.alberta.ca/library/Dataset/Details/540. Accessed 27 Feb 2015
AEP (2015b) Total area of the oil sands tailings ponds over time. Oil Sands Information Portal, Alberta Environment and Parks, Edmonton, AB. Dataset no. 542. http://osip.alberta.ca/library/Dataset/Details/542. Accessed 4 Mar 2015
Aladin NV (1991) Salinity tolerance and morphology of the osmoregulation organs in Cladocera with special reference to Cladocera from the Aral Sea. Hydrobiologia 225:291–299
Aladin NV, Potts WTW (1995) Osmoregulatory capacity of the Cladocera. J Comp Physiol B 164:671–683
Alharbi HA, Morandi G, Giesy JP, Wiseman SB (2016a) Effect of oil sands process-affected water on toxicity of retene to early life-stages of Japanese medaka (Oryzias latipes). Aquat Toxicol 176:1–9
Alharbi HA, Saunders DVM, Al-Mousa A, Alcorn J, Pereira AS, Martin JW, Giesy JP, Wiseman SB (2016b) Inhibition of ABC transport proteins by oil sands process affected water. Aquat Toxicol 170:81–88
Anderson JC, Wiseman SB, Wang N, Moustafa A, Perez-Estrada L, Gamal El-Din M, Martin JW, Liber K, Giesy JP (2012a) Effectiveness of ozonation treatment in eliminating toxicity of oil sands process-affected water to Chironomus dilutus. Environ Sci Technol 46:486–493
Anderson JC, Wiseman SB, Moustafa A, Gamal El-Din M, Liber K, Giesy JP (2012b) Effects of exposure to oil sands process-affected water from experimental reclamation ponds on Chironomus dilutus. Water Res 46:1662–1672
Arriaga D, Nelson TC, Risacher FF, Morris PK, Goad C, Slater GF, Warren LA (2019) The co-importance of physical mixing and biogeochemical consumption in controlling water cap oxygen levels in Base Mine Lake. Appl Geochem 111:104442
Baker JA, Gilron G, Chalmers BA, Elphick JR (2017) Evaluation of the effect of water type on the toxicity of nitrate to aquatic organisms. Chemosphere 168:435–440
Bartlett AJ, Frank RA, Gillis PL, Parrott JL, Marentette JR, Brown LR, Hooey T, Vanderveen R, McInnis R, Brunswick P, Shang D, Headley JV, Peru KM, Hewitt LM (2017) Toxicity of naphthenic acids to invertebrates: extracts from oil sands process-affected water versus commercial mixtures. Environ Pollut 227:271–279
Bauer AE, Frank RA, Headley JV, Peru KM, Farwell AJ, Dixon DG (2017) Toxicity of oil sands acid-extractable organic fractions to freshwater fish: Pimephales promelas (fathead minnow) and Oryzias latipes (Japanese medaka). Chemosphere 171:168–176
Bauer AE, Hewitt LM, Parrott JL, Bartlett AJ, Gillis PL, Deeth LE, Rudy MD, Vanderveen R, Brown L, Campbell SD, Rodrigues MR, Farwell AJ, Dixon DG, Frank RA (2019) The toxicity of organic fractions from aged oil sands process-affected water to aquatic species. Sci Total Environ 669:702–710
Environment Canada (1997) Biological test method: test for survival and growth in sediment using the larvae of freshwater midges (Chironomus tentans or Chironomus riparius). Method Development and Applications Section, Environment Canada, Ottawa, ON
Environment Canada (2007) Biological test method: test of reproduction and survival using the cladoceran Ceriodaphnia dubia. Method Development and Applications Section, Environment Canada, Ottawa, ON
Clemente JS, Fedorak PM (2005) A review of the occurrence, analyses, toxicity, and biodegradation of naphthenic acids. Chemosphere 60:585–600
Dompierre KA, Lindsay MBJ, Cruz-Hernandez P, Halferdahl GM (2016) Initial geochemical characteristics of fluid fine tailings in an oil sands end pit lake. Sci Total Environ 556:196–206
Dompierre KA, Barbour SL, North RL, Carey SK, Lindsay MBJ (2017) Chemical mass transport between fluid fine tailings and the overlying water cover of an oil sands end pit lake. Water Resour Res 53:4725–4740
Elphick JRF, Bergh KD, Bailey HC (2011) Chronic toxicity of chloride to freshwater species: effects of hardness and implications of water quality guidelines. Environ Toxicol Chem 30:239–246
Erickson RJ, Mount DR, Highland TL, Hockett JR, Hoff DJ, Jenson CT, Norberg-King TJ, Peterson KN (2017) The acute toxicity of major ion salts to Ceriodaphnia dubia. II. Empirical relationships in binary salt mixtures. Environ Toxicol Chem 36:1525–1537
Erickson RJ, Mount DR, Highland TL, Hockett JR, Hoff DJ, Jenson CT, Norberg-King TJ, Peterson KN (2018) The acute toxicity of major ion salts to Ceriodaphnia dubia. III. Mathematical models for mixture toxicity. Environ Toxicol Chem 37:247–259
Farag AM, Harper DD (2014) The chronic toxicity of sodium bicarbonate, a major component of coal bed natural gas produced waters. Environ Toxicol Chem 33:532–540
Frank RA, Kavanagh R, Burnison BK, Arsenault G, Headley JV, Peru KM, Solomon KR (2008) Toxicity assessment of collected fractions from an extracted naphthenic acid mixture. Chemosphere 72:1309–1314
Freitas EC, Rocha O (2011) Acute and chronic effects of sodium and potassium on the tropical freshwater cladoceran Pseudosida ramosa. Ecotox 20:88–96
Gauss JD, Woods PE, Winner RW, Skillings JH (1985) Acute toxicity of copper to three life stages of Chironomus tentans as affected by water hardness/alkalinity. Environ Pollut 37:149–157
Harper DD, Farag AM, Skaar D (2014) Acute toxicity of sodium bicarbonate, a major component of coal bed natural gas produced waters, to 13 aquatic species as defined in the laboratory. Environ Toxicol Chem 33:525–531
He Y, Wiseman SB, Zhang X, Hecker M, Jones PD, Gamal El-Din M, Martin JW, Giesy JP (2010) Ozonation attenuates the steroidogenic disruptive effects of sediment free oil sands process water in the H295R cell line. Chemosphere 80:578–584
He Y, Wiseman SB, Hecker M, Zhang X, Wang N, Perez LA, Jones PD, Gamal El-Din M, Martin JW, Giesy JP (2011) Effect of ozonation on the estrogenicity and androgenicity of oil sands process-affected water. Environ Sci Technol 45:6268–6274
He Y, Patterson S, Wang N, Hecker M, Martin JW, Gamal El-Din M, Giesy JP, Wiseman SB (2012a) Toxicity of untreated and ozone-treated oil sands process-affected water (OSPW) to early life stages of the fathead minnow (Pimephales promelas). Water Res 46:6359–6368
He Y, Wiseman SB, Wang N, Perez-Estrada LA, Gamal El-Din M, Martin JW, Giesy JP (2012b) Transcriptional responses of the brain-gonad-liver axis of fathead minnows exposed to untreated and ozone-treated oil sands process-affected water. Environ Sci Technol 46:9701–9708
Hoke RA, Gala WR, Drake JB, Giesy JP (1992) Bicarbonate as a potential confounding factor in Cladoceran toxicity assessments of pore water from contaminated sediments. Can J Fish Aquat Sci 49:1633–1640
Kavanagh RJ, Frank RA, Oakes KD, Servos MR, Young RF, Fedorak PM, MacKinnon MD, Solomon KR, Dixon DG, van der Kraak G (2011) Fathead minnow (Pimephales promelas) reproduction is impaired in aged oil sands process-affected waters. Aquat Toxicol 101:214–220
Kavanagh RJ, Frank RA, Burnison BK, Young RF, Fedorak PM, Solomon KR, Van Der Kraak G (2012) Fathead minnow (Pimephales promelas) reproduction is impaired when exposed to a naphthenic acid extract. Aquat Toxicol 116–117:34–42
Kennedy KD (2012) Growth, survival, and community composition of Chironomidae (Diptera) larvae in selected Athabasca oil sands process-affected wetlands waters of north-eastern Alberta. M.Sc. Dissertation, Department of Biological Sciences, University of Windsor, Windsor, ON, Canada. https://scholar.uwindsor.ca/etd/4820/. Accessed 21 Apr 2017
Lari E, Steinkey D, Mohaddes E, Pyle GG (2017) Investigating the chronic effects of oil sands process-affected water on growth and fitness of Daphnia magna Straus 1820. Sci Total Environ 595:594–600
Lari E, Steinkey D, Pyle GG (2018) Effects of seasonal changes on the toxic impacts of oil sands process-affected water on Daphnia magna. Arch Environ Contam Toxicol 74:408–413
Lari E, Steinkey D, Razmara P, Mohaddes E, Pyle GG (2019) Oil sands process-affected water impairs the olfactory system of rainbow trout (Oncorhynchus mykiss). Ecotox Environ Saf 170:62–67
Lasier PJ, Hardin IR (2010) Observed and predicted reproduction of Ceriodaphnia dubia exposed to chloride, sulfate, and bicarbonate. Environ Toxicol Chem 29:347–358
Leclair LA, MacDonald GZ, Phalen LJ, Kollner B, Hogan NS, van den Heuvel MR (2013) The immunological effects of oil sands surface waters and naphthenic acids on rainbow trout (Oncorhynchus mykiss). Aquat Toxicol 142–143:185–194
Leonhardt CL (2003) Zoobenthic succession in constructed wetlands of the Fort McMurray oil sands region: developing a measure of zoobenthic recovery. M.Sc. dissertation. University of Windsor, Windsor, ON, Canada. https://scholar.uwindsor.ca/etd/4612. Accessed 21 Apr 2017
Liber K, Call DJ, Dawson TD, Whiteman FW, Dillon TM (1996) Effects of Chironomus tentans larval growth retardation on adult emergence and ovipositing success: implications for interpreting freshwater sediment bioassays. Hydrobiologia 323:155–167
Lyons DD, Philibert DA, Zablocki T, Qin R, Huang R, Gamal El-Din M, Tierney KB (2018) Assessment of raw and ozonated oil sands process-affected water exposure in developing zebrafish: associating morphological changes with gene expression. Environ Pollut 241:959–968
MacDonald GZ, Hogan NS, Kollner B, Thorpe KL, Phalen LJ, Wagner BD, van den Heuvel MR (2013) Immunotoxic effects of oil sands-derived naphthenic acids to rainbow trout. Aquat Toxicol 126:95–103
McNeill SA, Arens CJ, Hogan NS, Kollner B, van den Heuvel MR (2012) Immunological impacts of oil sands-affected waters on rainbow trout evaluated using an in situ exposure. Ecotoxicol Environ Saf 84:254–261
McQueen AD, Kinley CM, Hendrikse M, Gaspari DP, Calomeni AJ, Iwinski KJ, Castle JW, Haakensen MC, Peru KM, Headley JV, Rodgers JH (2017) A risk-based approach for identifying constituents of concern in oil sands process-affected water from the Athabasca Oil Sands region. Chemosphere 173:340–350
Morandi GD, Wiseman SB, Pereira A, Mankidy R, Gault IGM, Martin JW, Giesy JP (2015) Effects-directed analysis of dissolved organic compounds in oil sands process-affected water. Envrion Sci Technol 49:12395–12404
Morandi GD, Zhang K, Wiseman SB, Pereira A, Martin JW, Giesy JP (2016) Effect of lipid partitioning on predictions of acute toxicity of oil sands process affected water to embryos of fathead minnow (Pimephales promelas). Environ Sci Technol 50:8858–8866
Mori JF, Chen LX, Jessen GL, Rudderham SB, McBeth JM, Lindsay MBJ, Slater GF, Banfield JF, Warren LA (2019) Putative mixotrophic nitrifying-denitrifying gammaproteobacteria implicated in nitrogen cycling within the ammonia/oxygen transition zone of an oil sands pit lake. Front Microbiol 10:1–15
Mount DR, Erickson RJ, Highland TL, Hockett JR, Hoff DJ, Jenson CT, Norberg-King TJ, Peterson KN, Polaske ZM (2016) The acute toxicity of major ion salts to Ceriodaphnia dubia: I. influence of background water chemistry. Environ Toxicol Chem 35:3039–3057
Mount DR, Erickson RJ, Forsman BB, Highland TL, Hockett JR, Hoff DL, Jenson CT, Norberg-King TJ (2019) Chronic toxicity of major ion salts and their mixtures to Ceriodaphnia dubia. Environ Toxicol Chem 38:769–783
Naddy RB, La Point TW, Klaine SJ (1995) Toxicity of arsenic, molybdenum and selenium combinations to Ceriodaphnia dubia. Environ Toxicol Chem 14:329–336
Pourrezai P, Drzewicz P, Wang Y, Gamal El-Din M, Perez-Estrada LA, Martin JW, Anderson J, Wiseman S, Liber K, Giesy JP (2011) The impact of metallic coagulants on the removal of organic compounds from oil sands process-affect water. Environ Sci Technol 45:8452–8459
Richardson E, Bass D, Smirnova A, Paoli L, Dunfield P, Dacks JB (2020) Phylogenetic estimation of community composition and novel eukaryotic lineages in base mine lake: an oil sands tailings reclamation site in Northern Alberta. J Eukaryot Microbiol 67:86–99. https://doi.org/10.1111/jeu.12757
Risacher FF, Morris PK, Arriaga D, Goad C, Nelson TC, Slater GF, Warren LA (2018) The interplay of methane and ammonia as key oxygen consuming constituents in early stage development of Base Mine Lake, the first demonstration oil sands pit lake. Appl Geochem 93:49–59
Schramm LL, Smith RG (1985) The influence of natural surfactants on interfacial charges in the hot-water process for recovering bitumen from the Athabasca oil sands. Colloid Surf 14:61–85
Sibley PK, Benoit DA, Ankley GT (1997) The significance of growth in Chironomus tentans sediment toxicity tests: relationship to reproduction and demographic endpoints. Environ Toxicol Chem 16:336–345
Simmons JA (2012) Toxicity of major cations and anions (Na+, K+, Ca2+, Mg2+, Cl− and SO42−) to a macrophyte and an alga. Environ Toxicol Chem 31:1370–1374
Soucek DJ (2007) Bioenergetic effects of sodium sulfate on the freshwater crustacean, Ceriodaphnia dubia. Ecotox 16:317–325
Soucek DJ, Dickinson A (2016) Influence of chloride on the chronic toxicity of sodium nitrate to Ceriodaphnia dubia and Hyalella azteca. Ecotox 25:1406–1416
Sun J, Peng H, Alharbi HA, Jones PD, Giesy JP (2017) Identification of chemicals that cause oxidative stress in oil sands process-affected water. Environ Sci Technol 51:8773–8781
Tedford E, Halferdahl G, Pieters R, Lawrence GA (2019) Temporal variations in turbidity in an oil sands pit lake. Environ Fluid Mech 19:457–473
Toor NS, Franz ED, Fedorak PM, MacKinnon MD, Liber K (2013a) Degradation and aquatic toxicity of naphthenic acids in oil sands process-affected waters using simulated wetlands. Chemosphere 90:449–458
Toor NS, Han X, Franz E, MacKinnon MD, Martin JW, Liber K (2013b) Selective biodegradation of naphthenic acids and a probable link between mixture profiles and aquatic toxicity. Environ Toxicol Chem 32:2207–2216
Turcotte D, Kautzman M, Peru K, Headley J, Liber K (2009) Characterizing salt and naphthenic acids interaction in the toxicity of oil sands process water to freshwater invertebrates. Conference presentation, University of Saskatchewan, unpublished pilot study
van den Heuvel MR, Hogan NS, Roloson SD, van der Kraak GJ (2012) Reproductive development of yellow perch (Perca flavescens) exposed to oil sands-affected waters. Environ Toxicol Chem 31:654–662
Vera CL, Hyne RV, Patra R, Ramasamy S, Pablo F, Julli M, Kefford BJ (2014) Bicarbonate toxicity to Ceriodaphnia dubia and the freshwater shrimp Paratya australiensis and its influence on zinc toxicity. Environ Toxicol Chem 33:1179–1186
Whelly MP (2000) Aquatic invertebrates in wetlands of the oil sands region of northeast Alberta, Canada, with emphasis on Chironomidae (Diptera). M.Sc. Dissertation. University of Windsor, Windsor, ON, Canada. https://scholar.uwindsor.ca/etd/1207. Accessed 21 Apr 2017
White KB, Liber K (2018) Early chemical and toxicological risk characterization of inorganic constituents in surface water from the Canadian oil sands first large-scale end pit lake. Chemosphere 211:745–757
Wiseman SB, Anderson JC, Liber K, Giesy JP (2013a) Endocrine disruption and oxidative stress in larvae of Chironomus dilutus following short-term exposure to fresh or aged oil sands process-affected water. Aquat Toxicol 142–143:414–421
Wiseman SB, He Y, Gamal-El Din M, Martin JW, Jones PD, Hecker M, Giesy JP (2013b) Transcriptional responses of male fathead minnows exposed to oil sands process-affected water. Comp Biochem Physiol C 157:227–235
Acknowledgements
This research was funded by the University of Saskatchewan Toxicology Centre and the Toxicology Graduate Program. The authors thank Stephanie Schiffer, Katherine Raes, and Erin Maloney for their assistance in the culturing and care of test organisms used in this study.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
White, K.B., Liber, K. Chronic Toxicity of Surface Water from a Canadian Oil Sands End Pit Lake to the Freshwater Invertebrates Chironomus dilutus and Ceriodaphnia dubia. Arch Environ Contam Toxicol 78, 439–450 (2020). https://doi.org/10.1007/s00244-020-00720-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-020-00720-3