Abstract
Mercury (Hg) is a ubiquitous and highly toxic contaminant that can have negative effects on wildlife. Only a few studies have measured Hg concentrations in birds from the south central United States, and the potential threat of Hg contamination to birds in this region is largely unknown. In the present study, we assess Hg concentrations in blood and feathers from five bird species [eastern bluebird (Sialis sialis), Carolina wren (Thryothorus ludovicianus), wood duck (Aix sponsa), great egret (Ardea alba), and great blue heron (Ardea herodias)] that occupy different trophic levels at Caddo Lake and Lewisville Lake, located in northeast and north central Texas, respectively. Both sites are contaminated with Hg from the atmosphere. Adult passerines had higher Hg concentrations in their blood than conspecific nestlings. Mercury concentrations in feathers differed between species by more than an order of magnitude with large piscivorous species having higher concentrations than smaller insectivorous species. Mercury concentrations in eastern bluebirds were higher at Caddo Lake than Lewisville Lake. The present study represents one of the first studies of Hg concentrations in multiple bird species in north Texas and suggests that Hg concentrations in birds from atmospherically polluted sites in this region may be high enough to compromise fitness in those species.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Methyl mercury (MeHg) is a ubiquitous and highly toxic contaminant that negatively effects the nervous, reproductive, and immune systems of vertebrates, including birds (Scheuhammer et al. 2007; Franceschini et al. 2009). Although mercury (Hg) can be discharged into aquatic systems directly from point sources resulting in local contamination issues, the source of Hg in most ecosystems is atmospheric deposition (Selin 2009). Atmospherically deposited Hg originates from natural and anthropogenic emission sources, the largest of which include fossil fuel combustion and artisanal or small-scale gold mining (Krabbenhoft and Sunderland 2013). Once emitted, mercury can remain in the atmosphere for long periods of time (months to more than a year) and be deposited far from the original source, which has resulted in most areas of the planet becoming contaminated with levels of Hg that exceed pre-industrial baselines (Selin 2009). Mercury is deposited in relatively non-toxic inorganic forms but it can be converted to the toxic and accumulative methyl form by microbes in aquatic systems (Selin 2009). Primary producers concentrate MeHg directly from the water (Miles et al. 2001; Pickhardt and Fisher 2007), but consumers are exposed to MeHg through their diet (Hall et al. 1997; Tsui and Wang 2004; Pickhardt et al. 2006). MeHg biomagnifies in food webs reaching concentrations in higher order organisms, like birds, that are millions of times greater than that found near the base of the food web (Lavoie et al. 2013).
Concern exists that Hg contamination has contributed to population declines in birds (Braune et al. 2006; Edmonds et al. 2010; Seewagen 2010), but relatively few studies have examined Hg concentrations in birds compared to well-studied taxa like fish. Previous surveys of Hg concentrations in North American birds at sites contaminated by atmospheric deposition have focused on the northeastern U.S. (Evers et al. 1998, 2007), the Great Lakes (Evers et al. 1998), the Everglades (Frederick et al. 2004), and sites in western Canada (Hipfner et al. 2011; Guigueno et al. 2012). Little information is available regarding Hg concentrations in birds from other areas in North America not directly linked to a point source. In particular, relatively few studies have quantified concentrations of Hg in bird species from the south central U.S. (but see King et al. 1985; King and Cromartie 1986; Mora et al. 2008), though previous work has shown that the region experiences high levels of Hg deposition and that fish are highly contaminated with Hg, with concentrations similar to those observed in the northeastern U.S. (Drenner et al. 2011, 2013).
In the present study, we assessed Hg concentrations in birds sampled from two sites in north central and northeast Texas, Lewisville Lake and Caddo Lake, respectively and determined if differences in Hg concentrations exist based on age, trophic level, and location. The present study is the first to document Hg concentrations in multiple bird species in north Texas at sites polluted with atmospheric Hg.
Our study suggests that Hg concentrations in birds from atmospherically polluted sites in this region have concentrations of Hg as high as in other studies where negative impacts on behavior, reproductive success, or other aspects of life history have been observed (Heinz 1979; Hallinger et al. 2010; Jackson et al. 2011; Frederick and Jayasena 2011; Jayasena et al. 2011).
Methods
Study Areas
Lewisville Lake
Lewisville Lake, located in north central Texas within the northern portion of the Dallas-Fort Worth metro area, is an open water reservoir with an adjacent protected wetland habitat managed by the U.S. Army Corps of Engineers (Fig. 1). Mercury deposition is relatively low and the surrounding ecoregion does not have landscape characteristics associated with elevated deposition or methylation of Hg (Drenner et al. 2011, 2013). A previous study at Lewisville Lake found relatively low Hg concentrations in largemouth bass (Micropterus salmoides; Drenner et al. 2011).
Caddo Lake
Caddo Lake is a forested wetland ecosystem on the border between Texas and Louisiana (Fig. 1). The region is known to have high levels of Hg wet deposition (>11 μg/m2/year, Drenner et al. 2013), landscape characteristics associated with increased Hg methylation (Drenner et al. 2011, 2013), and Hg levels in largemouth bass high enough to pose a significant hazard to human health (Drenner et al. 2013). Consequently, high Hg concentrations have been documented in multiple trophic levels of non-avian taxa, including invertebrates, fish, amphibians, reptiles, and mammals from Caddo Lake (Giggleman et al. 1998; Chumchal and Hambright 2009; Chumchal et al. 2011; Drenner et al. 2011). Mercury contamination has been found to negatively effect piscivorous spotted gar (Lepisosteus occulatus) from Caddo Lake (Barst et al. 2011). Fish and invertebrates from Caddo Lake have Hg concentrations (Chumchal et al. 2011; Gann et al. 2015) that are similar to concentrations shown to adversely affect birds when consumed (Heinz 1979; Frederick and Jayasena 2011; Jayasena et al. 2011; Varian-Ramos et al. 2014) and a risk analysis found that Hg concentrations in shoreline spiders were high enough to pose a potential risk to arachnivorous nestling songbirds (Gann et al. 2015). The only previous study to document Hg concentrations in birds at Caddo Lake examined great blue heron nestlings (n = 7) and found that individuals possessed liver Hg concentrations as high as 19,900 ng/g ww (Giggleman et al. 1998).
Nest Monitoring, Capture, and Sampling
All sampling at Lewisville Lake was conducted at the Lewisville Lake Environmental Learning Area (LLELA) in Denton County in north central Texas (Fig. 1). At Caddo Lake samples were collected from the Caddo Lake National Wildlife Refuge, Caddo Lake Wildlife Management Area, and a lakefront residential area in north Harrison and south Marion counties (Fig. 1).
We used bluebird and wood duck nest boxes at both sites to attract nesting birds during the 2010 breeding season. From March to July, we collected breast feathers (adults) and sampled blood (adults and nestlings) directly from Carolina wrens and eastern bluebirds that utilized the nest boxes (Table 1). Female wood ducks line their nests with feathers plucked from their breasts. We also collected wood duck nest-lining feathers from nest boxes (Table 1). Blood was collected in capillary tubes after puncturing the brachial vein on adults or the tibial vein on nestlings using a sterile 26-guage needle. We also opportunistically collected adult great egret feathers (i.e., flight feathers, coverts, and breeding plumes) and great blue heron feathers (i.e., flight and body feathers; Table 1). These feathers were assumed to be from different individuals but this was not verified. Sampled species represented different trophic guilds (Table 1; Hepp and Belrose 2013, Gowaty and Plissner 2015; Haggerty and Morton 2014, McCrimmon et al. 2011; Vennesland and Butler 2011) and occupy each study site year round, thus minimizing potential issues related to differences in Hg concentrations due to the timing of molt and Hg deposition at unknown locations. Wood ducks, great egrets, and great blue herons primarily feed directly from the aquatic environment (McCrimmon et al. 2011; Vennesland and Butler 2011; Hepp and Belrose 2013). Eastern bluebirds and Carolina wrens feed directly in terrestrial habitats, but may consume emergent aquatic prey items (e.g. Odonata, Ephemeroptera, some Diptera), especially for those birds nesting closer to the waterfront (Gowaty and Plissner 2015; Haggerty and Morton 2014). Our sampling methodology was approved by the Institutional Animal Care and Use Committee (IACUC) at the University of North Texas.
Mercury Analysis
All samples were transported from the field and stored at −20 °C until analysis. All feather samples were rinsed 3× with ddH2O to remove dirt and debris, blotted dry with a Kimwipe tissue and allowed to dry at room temperature prior to analysis. Blood samples were dried at 55 °C and removed from capillary tubes with a stainless steel plunger for dry weight (dw) analysis.
Samples were analyzed for total Hg concentration with a direct Hg analyzer (DMA-80; Milestone) that used thermal decomposition, gold amalgamation, and atomic absorption spectrometry (U.S. EPA 1998). Total Hg was used as a proxy for MeHg as previous studies have found that >95 % of total Hg in feathers and avian blood is in the form of MeHg (Thompson and Furness 1989; Spalding et al. 2000; Rimmer et al. 2005). Quality assurance included reference and duplicate samples. Samples of National Research Council Canada reference materials (MESS-3, marine sediment, certified value = 91 ± 9 ng Hg/g dw, or DOLT-3, dogfish liver, certified value = 3370 ± 140 ng Hg/g dw) were analyzed approximately every 10 samples. The mean percentage recovery of reference materials was 102.1 % (n = 23). Duplicate samples were analyzed approximately every 25 samples, and the mean relative percent difference was 12.8 % (n = 6). Concentrations are reported as ng of total Hg per g dw for both feathers and blood. The limit of detection (LOD) for the Hg analyzer was 0.2174 ng of Hg (i.e., ~40 and ~8 ng/g dw based on mean sample weight for blood and feather samples, respectively). Mercury concentrations in blood were below the LOD for 100 and 26 % of eastern bluebird nestlings at Lewisville Lake and Caddo Lake, respectively. Mercury concentrations were above the LOD for all other blood samples and all feather samples examined in the study. For samples with Hg concentrations below the LOD proxy Hg concentrations were calculated using one-half the LOD (i.e., 0.1087 ng) divided by the sample weight (g).
Statistical Analysis
At each site, we determined if Hg concentrations were significantly different between (1) age classes (adult vs. nestling) of a given species and (2) species of a given age-class. We also determined whether Hg concentrations in a given species and age-class differed between sites. The two tissue types (i.e., blood and feather) were analyzed separately in all comparisons.
Nonparametric statistics were used to compare Hg concentrations, because the datasets were not normally distributed or possessed small sample sizes. Mann–Whitney U two-sample test with Z approximation and continuity correction and Kruskal–Wallis tests were used to test for differences in Hg concentrations between two and more than two samples, respectively. Significant results for Kruskal–Wallis tests were followed with Tukey multiple comparison tests on ranked data. Because blood Hg concentrations from nestmates may be considered pseudoreplicates (Hurlbert 1984), comparisons that included nestlings were conducted with the median value from each nest. All statistical analyses were conducted using SAS® 9.3 (SAS Institute, Cary, NC). Findings were considered to be statistically significant when p < 0.05.
In the present study, total Hg data is presented on a dw and estimated wet weight (ww) basis to facilitate comparisons with other studies. Water accounts for approximately 75–80 % of the weight of blood in birds (Scanlon 1982; Rimmer et al. 2010). We estimated ww total Hg concentrations by multiplying dw total Hg concentrations by a conversion factor of 0.2 to account for weight change due to water loss after drying.
Results
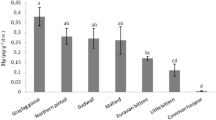
Median Hg concentrations in blood ranged from 21.7 ng/g dw in nestling eastern bluebirds from Lewisville Lake to 3360 ng/g dw in adult Carolina Wrens from Caddo Lake (Fig. 2). Median Hg concentration in feathers ranged from 279 ng/g dw in adult eastern bluebirds from Lewisville Lake to 13,923 ng/g dw in adult great blue herons at Caddo Lake (Fig. 3).
Mercury concentrations in blood for eastern bluebird and Carolina wren. Actual measurements presented in dry weight (ng/g). Estimated wet weight (ng/g) presented on right axis; conversion based on Scanlon (1982). Sample size indicated in parentheses. Boxes represent first quartile, median, and third quartile and whiskers represent the range. The letter “L” on the figure indicates that nesting eastern bluebirds were collected from Lewisville Lake. Note break in y-axis
Mercury concentrations in feathers from adult birds from Lewisville Lake and Caddo Lake. Sample sizes are given in parentheses. Boxes represent first quartile, median, and third quartile and whiskers represent the range. Within each site, species with Hg concentrations that were significantly different are indicated with different lowercase (Lewisville Lake) or uppercase (Caddo Lake) letters
Age-Class Comparisons
At each site, adult passerines had higher concentrations of Hg in their blood than nestlings but the difference was not always significant (Fig. 2; Table S1). Mercury in blood from adult eastern bluebirds were ~1–3 orders of magnitude higher than in nestlings (df = 9, Z = −2.78, p = 0.006 and df = 16, Z = −3.32, p = 0.0009 at Lewisville Lake and Caddo Lake, respectively). Although the ranges of Hg concentration in blood for nestlings (240–983 ng/g dw) and adult (2100–8805 ng/g dw) Carolina wrens did not overlap (Fig. 2; Table S1), we did not detect a significant difference in Hg concentrations between age classes (df = 4, Z = 1.75, p = 0.08). The lack of significance is likely due to lack of power from low sample sizes (n = 3 from both age classes).
Within Site Comparisons
Lewisville Lake
Differences in the concentrations of Hg in feathers were detected between species from Lewisville Lake (H = 6.37, df = 2, p = 0.04; Fig. 3; Table S2). Great egrets had higher concentrations of Hg in feathers than eastern bluebirds (post hoc analysis with Tukey grouping on ranked data, p < 0.05), whereas wood duck Hg concentrations were intermediate and not statistically different from either of the other two species (p > 0.05).
Caddo Lake
At Caddo Lake, we observed differences in the concentrations of Hg in blood between species. Adult and nestling Carolina wrens had significantly higher concentrations of Hg in blood than adult and nesting eastern bluebirds, respectively (df = 13, Z = 2.19, p = 0.03 and df = 7, Z = 2.09, p = 0.04, respectively; Fig. 2; Table S1). Differences in concentrations of Hg in feathers were also detected between species (Kruskal–Wallis test, H = 38.7, df = 4, p < 0.0001). Herons and egrets had higher concentrations of Hg in feathers than passerines and wood ducks (post hoc analysis with Tukey grouping on ranked data, p > 0.05; Fig. 3; Table S2).
Among Site Comparison
For eastern bluebirds, Hg concentrations in blood were significantly higher at Caddo Lake than Lewisville Lake for both nestlings (df = 9, Z = 2.78, p = 0.006) and adults (df = 16, Z = −3.32, p = 0.0009; Fig. 2; Table S1). The same pattern was observed for eastern bluebird feather samples (df = 18, Z = −2.89, p = 0.004; Fig. 3; Table S2). Average Hg concentrations of feathers for adult wood ducks and great egrets were higher at Caddo Lake than Lewisville Lake but the difference was not significant (df = 32, Z = −0.97, p = 0.33 and df = 16, Z = −1.75, p = 0.08 for wood ducks and great egrets respectively; Fig. 3; Table S2). The power to test for statistical differences between the two locations was low, however, due to the small sample sizes from Lewisville Lake.
Discussion
Age-Class Comparisons
Blood Hg concentrations were consistently lower in eastern bluebird and Carolina wren nestlings compared with conspecific adults by nearly an order of magnitude, a finding that is consistent with previous studies that have compared Hg concentrations in nestling and adult birds (Scheuhammer et al. 1998; Evers et al. 2005; Brasso and Cristol 2008; Condon and Cristol 2009). This pattern is likely the result of rapid feather growth during the nestling period when continuous Hg sequestration from the blood occurs until feather growth has ceased (Fournier et al. 2002; Condon and Cristol 2009). In addition, differences in total Hg-exposure time between adults and nestlings also may contribute to the observed differences. Blood Hg concentrations in several bird species have been shown to gradually increase over time during the intermolt period when feather growth is not occurring (Bearhop et al. 2000b; Ackerman et al. 2008; Varian-Ramos et al. 2014). As both eastern bluebird and Carolina wren undergo molt from July through October, we collected samples fairly late in adults’ intermolt period when elevated concentrations of Hg in the blood are expected (Haggerty and Morton 2014; Gowaty and Plissner 2015).
Within Site Comparisons
Mercury concentrations differed among species within sites and mostly corresponded with trophic guild. Piscivores had the highest concentrations of Hg, followed by insectivores and omnivores. It is well established that MeHg concentrations increase with trophic level and is highest in fish-eating birds (Campbell et al. 2005; Evers et al. 2005; Rimmer et al. 2010; Lavoie et al. 2010; Keller et al. 2014).
Within trophic guilds, more fine-scale feeding specialization may have influenced Hg concentrations (Bearhop et al. 2000a; Keller et al. 2014). For example, Carolina wrens consume a greater percentage of animal-derived tissues as opposed to plant material in their diet compared to eastern bluebirds (94 vs. 59 % on annual basis, respectively, although insects may consist of up to ~85 % of the bluebird diet during spring and summer months; Beal et al. 1941). Carolina wrens are also known to consume more spiders than eastern bluebirds (Beal et al. 1941). Because animal-derived tissue would be expected to have a higher concentration of Hg than plant-derived tissue (Chumchal et al. 2011), and because spiders may contain higher Hg concentrations due to increased trophic position (Cristol et al. 2008; Rimmer et al. 2010), feeding specialization may have contributed to the differences observed between the two passerine species in the present study even though both species are commonly considered to be primarily insectivorous (Haggerty and Morton 2014; Gowaty and Plissner 2015).
Additionally, the differences in Hg concentrations between eastern bluebirds and Carolina wrens may have been impacted by foraging micro-habitat (Evers et al. 2005; Chumchal et al. 2008; Chumchal and Hambright 2009; Walters et al. 2009). Terrestrial organisms that forage closer to contaminated sites may be exposed to greater contaminant loads (Evers et al. 2005; Cristol et al. 2008; Walters et al. 2009; Barnes and Gerstenberger 2015). The three Carolina wren nests sampled in the present study were located immediately adjacent (e.g. 1–15 m) to aquatic habitat at Caddo Lake, whereas the eastern bluebird nests were located between 0 and 5 km from Caddo Lake. Because songbirds are known to forage in the vicinity of their nest, Carolina wrens may have fed on insects and spiders connected to the aquatic food web with high concentrations of Hg (Cristol et al. 2008; Walters et al. 2009; Keller et al. 2014; Gann et al. 2015).
Among Site Comparison
The majority of species sampled tended to have higher Hg concentrations at Caddo Lake than at Lewisville Lake. This pattern is in agreement with previous studies that identified a west to east gradient in Hg concentrations of largemouth bass in north Texas (Drenner et al. 2011, 2013). Largemouth bass sampled at Caddo Lake had 3–4× higher Hg concentrations as compared to those sampled at Lewisville Lake (Drenner et al. 2011). This gradient in Hg contamination of fish was correlated with Hg in wet deposition and conifer coverage, both of which increase from west to east across this region (Drenner et al. 2013). Additional studies with sampling sites in and between both regions should be conducted to assess definitively whether the west to east gradient in Hg contamination observed with fish also exists for birds.
Implications of Observed Hg Concentrations
Our results suggest that some birds sampled at both Caddo Lake and Lewisville Lake may be negatively impacted by high Hg exposure. Piscivorous wading birds at both sampling locations had feather Hg concentrations similar to those associated with endocrine disruption in white ibis (Frederick and Jayasena 2011; Jayasena et al. 2011). Likewise, Hg concentrations in wood duck feather samples from both Caddo Lake and Lewisville Lake were high, >5000 ng/g dw, with one sample >11,000 ng/g dw, similar to concentrations associated with negative effects in mallards (Heinz 1979). It should be noted that the adult wood duck feathers with the highest Hg concentration in the present study (11,426 ng/g dw) were collected from a nest box at Caddo Lake. The nest box contained three chick carcasses with feather Hg concentrations that ranged from 8605 to 18,310 ng/g dw (data on Hg concentration in nestlings was not included in Fig. 3 or Table S2).
Mercury concentrations may be high enough to pose risk and compromise fitness in populations of some songbird species at Caddo Lake. Two adult Carolina wrens had a blood Hg concentration that approached or exceeded 700 ng/g ww (i.e., 670 and 1760 ng/g ww) and a Carolina wren and an eastern bluebird had feather Hg concentrations that approached 2400 ng/g dw (i.e., 2117 and 2383 ng/g dw, respectively), which are all within the range at which adverse effects have been documented in previous studies focused on birds. For example, in free-living Carolina wrens in Virginia, Jackson et al. (2011) modeled the response of nest survival to female Hg concentration among other competing risks and showed that a 10 % reduction in nest success was associated with 700 ng/g ww in blood and 2400 ng/g dw Hg in body feathers, whereas a 30 % reduction in nest success was associated with 1700 ng/g ww in blood. Additionally, tonal frequency, note diversity, and length were reduced in the songs of both Carolina wrens and house wrens (Troglodytes aedon) at the same Hg contaminated study site in Virginia (Hallinger et al. 2010).
Our results indicate that high Hg concentrations may be a cause for concern in some bird species at both Lewisville Lake and Caddo Lake. This finding is notable because both sites are polluted with Hg from the atmosphere, rather than a direct point source. Given that all aquatic ecosystems in the region are contaminated with atmospheric Hg (http://nadp.sws.uiuc.edu/mdn/), the present study suggests that Hg may pose a risk to birds residing in ecosystems throughout the South Central United States.
References
Ackerman JT, Eagles-Smith CA, Takekawa JY, Bluso JD, Adelsbach TL (2008) Mercury concentrations in blood and feathers of prebreeding Forster’s terns in relation to space use of San Francisco Bay, California, USA, habitats. Environ Toxicol Chem 27:897–908
Barnes J, Gerstenberger S (2015) Using feathers to determine mercury contamination in peregrine falcons and their prey. J Raptor Res 49:43–58
Barst BD, Gevertz AK, Chumchal MM, Smith JD, Rainwater TR, Drevnick PE, Hudelson K, Hart A, Verbeck GF, Roberts AP (2011) Laser ablation ICP-MS co-localization of mercury and immune response in fish. Environ Sci Technol 45:8982–8988
Beal FEL, McAtee WL, Kalmback ER (1941) Common birds of Southeastern United States in relation to agriculture. US Fish Wild Pub paper 246
Bearhop S, Phillips RA, Thompson DR, Waldron S, Furness RW (2000a) Variability in mercury concentrations of Great Skuas Catharacta skua: the influence of colony, diet and trophic status inferred from stable isotope signatures. Mar Ecol Prog Ser 195:261–268
Bearhop S, Ruxton GD, Furness RW (2000b) Dynamics of mercury in blood and feathers of great skuas. Environ Toxicol Chem 19:1638–1643
Brasso RL, Cristol DA (2008) Effects of mercury exposure on the reproductive success of tree swallows (Tachycineta bicolor). Ecotoxicology 17:133–141
Braune BM, Mallory ML, Gilchrist HG (2006) Elevated mercury levels in a declining population of ivory gulls in the Canadian Arctic. Mar Pollut Bull 52:978–982
Campbell LM, Norstrom RJ, Hobson K, Muir DCG, Backus S, Fisk AT (2005) Mercury and other trace elements in a pelagic Arctic marine food web (Northwater Polynya, Baffin Bay). Sci Total Environ 351–352:247–263
Chumchal MM, Hambright KD (2009) Ecological factors regulating mercury contamination of fish from Caddo Lake, Texas, U.S.A. Environ Toxicol Chem 28:962–972
Chumchal MM, Drenner RW, Fry B, Hambright KD, Newland LW (2008) Habitat-specific differences in mercury concentration in a top predator from a shallow lake. Trans Am Fish Soc 137:195–208
Chumchal MM, Rainwater TR, Osborn SC, Roberts AP, Abel MT, Cobb GP, Smith PN, Bailey FC (2011) Mercury speciation and biomagnification in the food web of Caddo Lake, Texas and Louisiana, USA, a subtropical freshwater ecosystem. Environ Toxicol Chem 30:1153–1162
Condon AM, Cristol DA (2009) Feather growth influences blood mercury level of young songbirds. Environ Toxicol Chem 28:395–401
Cristol D, Brasso RL, Condon AM, Fovargue RE, Friedman SL, Hallinger KK, Monroe AP, White AE (2008) The movement of aquatic mercury through terrestrial food webs. Science 320:335
Drenner RW, Chumchal MM, Wente SP, McGuire M, Drenner SM (2011) Landscape-level patterns of mercury contamination of fish in North Texas, USA. Environ Toxicol Chem 30:2041–2045
Drenner RW, Chumchal MM, Jones CM, Lehmann CM, Gay DA, Donato DI (2013) Effects of mercury deposition and coniferous forests on the mercury contamination of fish in the South Central United States. Environ Sci Technol 47:1274–1279
Edmonds ST, Evers DC, Cristol D, Mettke-Hofmann C, Powell LL, McGann AJ, Armiger JW, Lane OP, Tessler DF, Newell P, Heyden K, O’Driscoll NJ (2010) Geographic and seasonal variation in mercury exposure of the declining rusty blackbird. Condor 112:789–799
Evers DC, Kaplan JD, Meyer MW, Reaman PS, Braselton WE, Major A, Burgess N, Scheuhammer AM (1998) Geographic trend in mercury measured in common loon feathers and blood. Environ Toxicol Chem 17:173–183
Evers DC, Burgess NM, Champoux L, Hoskins B, Major A, Goodale WM, Taylor RJ, Poppenga R, Daigle T (2005) Patterns and interpretation of mercury exposure in freshwater avian communities in northeastern North America. Ecotoxicology 14:193–221
Evers DC, Han YJ, Driscoll CT, Kamman NC, Goodale MW, Lambert KF, Holsen TM, Chen CY, Clair TA, Butler T (2007) Biological mercury hotspots in the northeastern United States and southeastern Canada. Bioscience 57:1–15
Fournier F, Karasov WH, Kenow KP, Meyer MW, Hines RK (2002) The oral bioavailability and toxicokinetics of methylmercury in common loon (Gaviaimmer) chicks. Comp Biochem Phys A 133:703–714
Franceschini MD, Lane OP, Evers DC, Reed JM, Hoskins B, Romero LM (2009) The corticosterone stress response and mercury contamination in free-living tree swallows, Tachycineta bicolor. Ecotoxicology 18:514–521
Frederick P, Jayasena N (2011) Altered pairing behaviour and reproductive success in white ibises exposed to environmentally relevant concentrations of methylmercury. Proc R Soc B 278:1851–1857
Frederick PC, Hylton B, Heath JA, Spalding MG (2004) A historical record of mercury contamination in southern Florida (USA) as inferred from avian feather tissue. Environ Toxicol Chem 23:1474–1478
Gann G, Powell C, Chumchal M, Drenner R (2015) Hg-contaminated terrestrial spiders pose a potential risk to songbirds at Caddo Lake (Texas/Louisiana, USA). Environ Toxicol Chem 34:303–306
Giggleman CM, Baker DM, Lusk JD (1998) A contaminants survey of three lentic systems within the Cypress Creek Watershed, Texas, 1993–1995. 9320005/2F27. U.S. Fish and Wildlife Service, Region 2, Arlington Ecological Services Field Office, Arlington
Gowaty PA, Plissner JH (2015) Eastern bluebird (Sialia sialis), In: Poole A (eds) The birds of North America online. Cornell Lab of Ornithology, Ithaca. Retrieved from the birds of North America online: http://bna.birds.cornell.edu.bnaproxy.birds.cornell.edu/bna/species/381. doi:10.2173/bna.381
Guigueno MF, Elliott KH, Levac J, Wayland M, Elliott JE (2012) Differential exposure of alpine ospreys to mercury: melting glaciers, hydrology or deposition patterns? Environ Int 40:24–32
Haggerty TM, Morton ES (2014) Carolina Wren (Thryothorus ludovicianus). In: Poole A (Ed) The birds of North America online. Cornell Lab of Ornithology, Ithaca. Retrieved from the birds of North America online: http://bna.birds.cornell.edu.bnaproxy.birds.cornell.edu/bna/species/188. doi:10.2173/bna.188
Hall BD, Bodaly RA, Fudge RP, Rudd JM, Rosenberg DM (1997) Food as the dominant pathway of methylmercury uptake by fish. Water Air Soil Pollut 100:13–24
Hallinger KK, Zabransky DJ, Kazmer KA, Cristol DA (2010) Birdsong differs between mercury-polluted and reference sites. Auk 127:156–161
Heinz GH (1979) Methylmercury: reproductive and behavioral effects on three generations of mallard ducks. J Wildl Manag 43:394–401
Hepp GR, Bellrose FC (2013) Wood duck (Aix sponsa). In: Poole A (Ed) The birds of North America online. Cornell Lab of Ornithology, Ithaca. Retrieved from the birds of North America online: http://bna.birds.cornell.edu.bnaproxy.birds.cornell.edu/bna/species/169. doi:10.2173/bna.169
Hipfner JM, Hobson KA, Elliott JE (2011) Ecological factors differentially affect mercury levels in two species of sympatric marine birds of the North Pacific. Sci Total Environ 409:1328–1335
Hurlbert SH (1984) Pseudoreplication and the design of ecological field experiments. Ecol Monogr 54:187–211
Jackson AK, Evers DC, Etterson MA, Condon AM, Folson SB, Detweiler J, Schmerfeld J, Cristol DA (2011) Mercury exposure affects the reproductive success of a free-living terrestrial songbird, the Carolina wren (Thryothorus ludovicianus). Auk 128:759–769
Jayasena N, Frederick PC, Larkin ILV (2011) Endocrine disruption in white ibises (Eudocimus albus) caused by exposure to environmentally relevant levels of methylmercury. Aquat Toxicol 105:321–327
Keller RH, Xie L, Buchwalter DB, Franzreb KE, Simons TR (2014) Mercury bioaccumulation in Southern Appalachian birds, assessed through feather concentrations. Ecotoxicology 23:304–316
King KA, Cromartie E (1986) Mercury, cadmium, lead, and selenium in three waterbird species nesting in Galveston Bay, Texas, USA. Colon Waterbird 9:90–94
King KA, Blankinship DR, Payne E, Krynitsky AJ, Hensler GL (1985) Brown pelican populations and pollutants in Texas 1975–1981. Wilson Bull 97:201–214
Krabbenhoft DP, Sunderland EM (2013) Global change and mercury. Science 341:1457–1458
Lavoie RA, Hebert CE, Rail JF, Braune BM, Yumvihoze E, Hill LG, Lean DR (2010) Trophic structure and mercury distribution in a Gulf of St. Lawrence (Canada) food web using stable isotope analysis. Sci Total Environ 408:5529–5539
Lavoie RA, Jardine TD, Chumchal MM, Kidd KA, Campbell LM (2013) Biomagnification of mercury in aquatic food webs: a worldwide meta-analysis. Environ Sci Technol 47:13385–13394
McCrimmon DA Jr, Ogden JC, Bancroft GT (2011) Great egret (Ardea alba). In: Poole A (Ed) The birds of North America online. Cornell Lab of Ornithology, Ithaca. Retrieved from the birds of North America online: http://bna.birds.cornell.edu.bnaproxy.birds.cornell.edu/bna/species/570. doi:10.2173/bna.570
Miles CJ, Moye HA, Phlips EJ, Sargent B (2001) Partitioning of monomethylmercury between freshwater algae and water. Environ Sci Technol 35:4277–4282
Mora M, Montoya AB, Lee MC, Macias-Duarte A, Rodriguez-Salazar R, Juergens PW, Lafón-Terrazas A (2008) Persistent environmental pollutants in eggs of aplomado falcons from Northern Chihuahua, Mexico, and South Texas, USA. Environ Int 34:44–50
Pickhardt PC, Fisher NS (2007) Accumulation of inorganic and methylmercury by freshwater phytoplankton in two contrasting water bodies. Environ Sci Technol 41:125–131
Pickhardt PC, Stepanova M, Fisher NS (2006) Contrasting uptake routes and tissue distributions of inorganic and methylmercury in mosquitofish, Gambusia affinis, and redear sunfish, Lepomis microlophus. Environ Toxicol Chem 25:2132–2142
Rimmer CC, McFarland KP, Evers DC, Miller EK, Aubry Y, Busby D, Taylor RJ (2005) Mercury concentrations in Bicknell’s thrush and other insectivorous passerines in montane forests of northeastern North America. Ecotoxicology 14:223–240
Rimmer CC, Miller EK, McFarland KP, Taylor RJ, Faccio SD (2010) Mercury bioaccumulation and trophic transfer in the terrestrial food web of a montane forest. Ecotoxicology 19:697–709
Scanlon PF (1982) Wet and dry weight relationships of mallard (Anas platyrhynchos) tissues. Bull Environ Contam Toxicol 29:615–617
Scheuhammer AM, Atchison CM, Wong AH, Evers DC (1998) Mercury exposure in breeding common loons (Gavia immer) in central Ontario, Canada. Environ Toxicol Chem 17:191–196
Scheuhammer AM, Meyer MW, Sandheinrich MB, Murray MW (2007) Effects of environmental methylmercury on the health of wild birds, mammals, and fish. Ambio 36:12–18
Seewagen CL (2010) Threats of environmental mercury to birds: knowledge gaps and priorities for future research. Bird Conserv Int 20:112–123
Selin NE (2009) Global biogeochemical cycling of mercury: a review. Annu Rev Environ Resour 34:43–63
Spalding MG, Frederick PC, McGill HC, Bouton SN, McDowell LR (2000) Methylmercury accumulation in tissues and its effects on growth and appetite in captive great egrets. J Wildl Dis 36:411–422
Thompson DR, Furness RW (1989) Comparison of the levels of total and organic mercury in seabird feathers. Mar Pollut Bull 20:577–579
Tsui MT, Wang WX (2004) Uptake and elimination routes of inorganic mercury and methylmercury in Daphnia magna. Environ Sci Technol 38:808–816
U.S. Environmental Protection Agency (1998) Method 7473: mercury in solids and solutions by thermal decomposition, amalgamation, and atomic absorption spectrophotometry. Washington, D.C
Varian-Ramos CW, Swaddle JP, Cristol DA (2014) Mercury reduces avian reproductive success and imposes selection: an experimental study with adult-or lifetime-exposure in zebra finch. PLoS One 9:e95674
Vennesland R, Butler R (2011) Great blue heron (Ardea herodias). In: Poole A (Ed) The birds of North America online. Cornell Lab of Ornithology, Ithaca. Retrieved from the birds of North America online: http://bna.birds.cornell.edu.bnaproxy.birds.cornell.edu/bna/species/025. doi:10.2173/bna.25
Walters DM, Mills MA, Fritz KM, Raikow DF (2009) Spider-mediated flux of PCBs from contaminated sediments to terrestrial ecosystems and potential risks to Arachnivorous birds. Environ Sci Technol 44:2849–2856
Acknowledgments
This project was funded with a University of North Texas Research Initiative Grant. Ryan Williams provided invaluable assistance in sample collection. The authors thank Vanessa Adams and Jason Roesner (Caddo Lake Wildlife Management Area, Caddo Lake National Wildlife Refuge) and Dr. Kenneth Steigman (Lewisville Lake Environmental Learning Area) for sampling permits, assisting with sample collection, and generously providing field housing. A special thank you to many private property owners of Karnack and Uncertain, TX, for permission to sample birds and/or providing information central to the project, namely Tom Walker, John and Diane Winn, Jeff Thompson, and Sam and Randy Canup.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Schulwitz, S.E., Chumchal, M.M. & Johnson, J.A. Mercury Concentrations in Birds from Two Atmospherically Contaminated Sites in North Texas, USA. Arch Environ Contam Toxicol 69, 390–398 (2015). https://doi.org/10.1007/s00244-015-0189-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-015-0189-9