Abstract
The aim of this study was to determine concentrations of heavy metals (cadmium [Cd] and mercury [Hg]) and metallothioneins (MTs) in blood plasma and caudal scutes of Morelet’s crocodile (Crocodylus moreletii) from Rio Hondo, a river and natural border between Mexico and Belize. Three transects of the river (approximately 20 km each) were surveyed in September 2012 and April 2013, and samples were collected from 24 crocodiles from these areas. In blood plasma, Cd (7.6 ± 9.6 ng/ml) was detected in 69 % of samples (n = 9); Hg (12.2 ± 9.2 ng/ml) was detected in 46 % of samples (n = 6); and MTs (10,900 ± 9,400 ng/ml) were detected in 92 % of samples (n = 12). In caudal scutes samples, Cd (31.7 ± 39.4 ng/g) was detected in 84 % of samples (n = 12) and Hg (374.1 ± 429.4 ng/g) in 83 % of samples (n = 20). No MTs were detected in caudal scutes. Hg concentrations in scutes from the Rio Hondo were 2- to 5-fold greater than those previously reported in scutes from other localities in northern Belize. In blood plasma, a significant positive relationship between Hg and body size was observed. Mean concentrations of Cd and MTs in size classes suggest that MTs may be related to Cd exposure. This is the first report of MT presence in crocodile blood.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Heavy-metal pollution in terrestrial and aquatic ecosystems has increased in the last 50 years (Schneider et al. 2013). Because of the persistence of metals in the environment, there is a concern about the impact of agricultural activities and untreated sewage on ecosystems (Defew et al. 2005; Schneider et al. 2013). Heavy metals are nonbiodegradable and can be toxic to both vertebrate and invertebrate fauna. After absorption by an organism, metals can biomagnify through the food chain (Heaton-Jones et al. 1997) and affect regulatory and metabolic processes, such as cellular respiration, metabolism of lipids and carbohydrates, hormonal secretion and synthesis, as well as reproduction and growth, in exposed organisms (Fingerman et al. 1996). Depending on the exposure concentration, metal toxicity can also result in mortality (Wolfe et al. 1998; Vázquez 2005).
Although environmental pollution has been recognized as one of the major threats to reptiles, research regarding the impacts of heavy metals on reptile populations is limited (Smith et al. 2007). The primary routes of heavy-metal exposure in crocodilians are ingestion, dermal contact, maternal transfer, and potentially egg contact with metals in nesting media (Smith et al. 2007). Several studies have reported heavy metals in various crocodilian tissues (Delany et al. 1988; Brisbin et al. 1998; Burger et al. 2000; Almli et al. 2005; Xu et al. 2006; Campbell et al. 2010). In Morelet´s crocodile (Crocodylus moreletii), Rainwater et al. (2002, 2007) measured mercury (Hg), cadmium (Cd), copper (Cu), lead (Pb), and zinc (Zn) concentrations from animals sampled in northern Belize.
Metallothioneins (MTs) are heavy metal-binding proteins that play an important role in the transport of metals in internal tissues and have been related to heavy-metal detoxification in mammals (Margoshes and Vallee 1957; Nordberg and Nordberg 2009). Studies with laboratory mice suggest that even after a single exposure to Cd, significant proportions of the Cd-MT complex may be found in plasma (Nordberg and Nordberg 2009). Some studies have noted that Cd and Hg induce expression of MTs in reptiles just as in mammals and birds (Hidalgo et al. 2009). However, there is a significant lack of information regarding MTs in reptiles, although they have been identified in Asian tortoises (Clemmys mutica) (Yamamura and Suzuki 1984) and American alligators (Alligator mississippiensis) (Bell and Lopez 1985).
In humans, the MT isoform MT-IV is expressed in the stratified squamous epithelium of the skin, tongue, and upper alimentary tract (Santucci et al. 2000). In addition, expression of the MT-I and MT-II isoforms have been observed in basal keratinocytes of the epidermis (Van-deer-Oord and DeLey 1994). Reptiles are characterized by a hard, dry skin that consists of a thin epidermis and a thicker dermis (Ortiz-Cervantes and Rubio-Lozano 2012); their scales are mainly epidermal and formed by keratin. In crocodilians, scutes can remain throughout life and grow gradually (Ortiz-Cervantes and Rubio-Lozano 2012). Therefore, although not previously reported, it is possible that MTs may also be found in caudal scutes of crocodiles.
Studies in the Rio Hondo, a transboundary river between southern Mexico and northern Belize, and Chetumal Bay, in which the Rio Hondo drains, have reported heavy metals in water and sediments and have suggested that pollution sources are human settlements and the use of agrochemicals in the Rio Hondo basin (Euan-Avila et al. 2002; Garcia-Rios and Gold-Bouchot 2003; Buenfil-Rojas and Flores-Cuevas 2007). Morelet’s crocodile inhabits the Rio Hondo and associated wetlands (Cedeño-Vázquez et al. 2006), but the accumulation and impact of heavy metals in this population remains uncertain. The aim of the present study was to determine the presence of Cd and Hg in blood plasma and caudal scutes of Morelet’s crocodiles from Rio Hondo and that of MTs as a heavy-metal detoxification mechanism.
Materials and Methods
Study Area
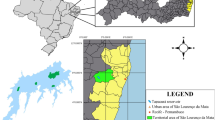
The Rio Hondo is located in the southeast of the Yucatan Peninsula and is considered to be the natural border between Mexico and Belize (Fig. 1) (Magnon Basnier 2002). It extends approximately 115 km from La Union village to Chetumal Bay (Comisión Nacional del Agua 2012). Morelet’s crocodiles were sampled along three transects of the Rio Hondo (approximately 20 km each),with each named according to a representative human settlement: as follows P = Pucte (18°11′N, 88°41′W–18°18′N, 88°35′W), C = Cocoyol (18°02′N, 88°45′W–18°10′N, 88°40′W), and L = La Union (17°54′N, 88°53′W–18°01′N, 88°46′W) (Fig. 1). These transects were selected considering potential sources of heavy-metal pollution from agricultural areas and discharges of wastewater (Euan-Avila et al. 2002).
Sample Collection
Caudal scutes and blood plasma samples were collected from crocodiles in September 2012 and April 2013. Crocodiles were captured by traditional active methods using snares (Messel et al. 1981; Woodward and Marion 1978). Total length (TL), body mass, and sex were determined for each animal. Size classes were based on TL as yearlings (≤50 cm), juveniles (51–100 cm), subadults (101–150 cm), and adults (>150 cm) (Sigler et al. 2011).
Blood was obtained from the dorsal postcranial sinus, stored in tubes containing lithium heparin, mixed by inversion (Sánchez-Herrera et al. 2011), and stored on ice in the field. Plasma was obtained by centrifugation at 3,157 RPM for 15 min in a portable centrifuge (Mobilespin; Vulcon Technologies, Inc., Grandview, Missouri).
Caudal scutes were removed with a scalpel and placed in labeled plastic containers (previously treated with 5 % HNO3). Iodine solution was applied to scute removal sites to prevent potential infection. Plasma and scutes samples were stored on ice until the arrival to the laboratory where they were stored frozen until analysis (Rainwater et al. 2007).
Metals Analysis
Before use, all glassware was washed with phosphate-free detergent (1:1), rinsed with tap water, and then kept in acid solution for 24 h (5 % HNO3 for Cd and HCl 1 N for Hg and MTs). Glassware was subsequently rinsed with distilled water (Andreani et al. 2008; Perry et al. 1975). Analysis of Cd and Hg was adapted from several methods (Andreani et al. 2008; Delany et al. 1988; Perry et al. 1975; Rainwater et al. 2007). Samples were thawed at room temperature, and then 0.5–1 ml of plasma and 0.2–0.5 g of scutes were transferred to 100-ml Teflon beakers. Before analysis, scutes were rinsed with deionized water to remove any possible external contamination.
Acid digestion was performed by adding 2 ml of concentrated HNO3 to each beaker and allowing samples to predigest for 2 days. Samples were then heated for 4 h at 90 °C and cooled for 30 min. They were then treated with 30 % H2O2 and reheated for 2 h. Samples were cooled and filtered with no. 2 Whatman filter paper in 10-ml volumetric flasks and brought to volume with 1 % HNO3 for Cd analysis. Filtered samples for Hg were placed in 25-ml volumetric flasks and treated with 1.25 ml of KMnO4 until there was persistence of a purple color (Sanchez-Rodriguez 2009; Triunfante et al. 2009). Solutions were clarified with 12 % NH2OH·HCl and brought to volume with deionized water.
Samples were analyzed by graphite furnace–atomic absorption spectrophotometry (Avanta PM–GF3000, GBC) for Cd and hydride generation–atomic absorption spectrophotometry (Avanta PM–HG 3000, GBC) for Hg. Accuracy of the analysis was determined using certified reference material TORT-2 (lobster hepatopancreas) from the National Research Council of Canada and sample blanks. The detection limits were 3.9 ng Cd/g and 2.3 ng Hg/g, and recovery percentages were 92.1 % ± 3 % for Cd and 94.3 % ± 9.5 % for Hg.
MT Analysis
To obtain the cytosol from scutes, 0.2–0.5 g of frozen tissue were placed into a 5-ml tube with 4 volumes of sucrose solution (0.25 M). Samples were homogenized at high-speed with a Tissue Tearor (Model 985370-395; Biospec Products Inc.) for 5 min (due to the hardness of the tissue). Resulting homogenate was centrifuged at 18,000×g for 20 min at 4 °C (Scheuhammer and Cherian 1991). The supernatant fluid was transferred to an Eppendorf tube and frozen until analysis. Plasma aliquots were analyzed directly.
Scute cytosol and 0.1–0.5 ml of plasma were placed in Eppendorf tubes. The MT assay was performed according to the silver (Ag)-saturation method (Scheuhammer and Cherian 1991; Bienengräber et al. 1995). The final supernatants were aspirated, transferred to 5-ml volumetric flasks, and brought to volume with 1 % HNO3. Ag was measured by flame atomic absorption spectrophotometry (Avanta PM, GBC); for the stoichiometry, it was assumed that 1 μg of Ag+ represented 3.55 μg of MTs. Accuracy of the analysis was determined using the reference standard MT COP03 95 % (metallothionein from rabbit liver) from Creative Biomart (New York, USA) and sample blanks. The detection limits were 0.7 µg Ag/ml, and the recovery percentage for MTs was 89.2 % ± 1.9 %.
Statistical Analyses
Statistical analyses were performed using the statistical software Statistica (Version 7; Statsoft). Due to a small sample size (n < 30) and failure to achieve normality and homogeneity of variance, nonparametric procedures were used. Kruskal–Wallis test was used to identify differences in metals and MT concentrations between size classes. Spearman correlations were performed to detect relationships between concentrations of metals and MTs. Finally, linear regression analyses were performed to detect relationships between metals and MT concentrations and TL, snout-to-vent length (SVL), and body mass. For statistical purposes, concentrations decreasing lower than detection limits were assigned values of one-half the detection limit for that particular analyte (Rainwater et al. 2007). All tests were considered significant when p ≤ 0.05.
Results
Twenty-four Morelet´s crocodiles, ranging from 32 to 190.5 cm TL, were captured along the surveyed transects, (Table 1). Caudal scutes were collected from all crocodiles, whereas blood plasma samples were obtained only from 13 individuals (64–190 cm TL).
Heavy Metals
In blood plasma, Cd was detected in 100 % of samples from Pucte (n = 5) and Cocoyol (n = 2) and in 33 % of samples from La Union (n = 2); Hg was only detected in 40 % of samples from Pucte (n = 2) and in 67 % in samples from La Union (n = 4). Significant differences were observed in Hg concentrations between adults and the other size classes (p = 0.04). A significant positive relationship (p < 0.05) was observed between concentrations of Hg and TL, SVL, and body mass.
In caudal scutes, Hg was detected in 100 % (n = 8) of samples from La Union, in 71 % (n = 5) from Cocoyol, and in 78 % (n = 7) from Pucte. Due to the small amount of tissue obtained from caudal scutes of small crocodiles, it was possible to analyze Cd from only 13 individuals (Pucte [n = 4], Cocoyol [n = 4], and La Union [n = 5]). This metal was detected in 100 % of crocodiles from Pucte and La Union and in 50 % from Cocoyol. No significant relations were detected between Hg or Cd concentrations and TL or body mass of crocodiles. Mean concentrations of Cd in adults (16.04 ± 1.45 ng/g wet weight [ww]) were considerably lower compared with the other size classes, whereas concentrations of Hg in juveniles (630.4 ± 806.8 ng/g ww) were the highest (Table 2). Scute samples from yearlings contained detectable amounts of Hg (271.3 ± 162.1 ng/g ww) with a maximum concentration of 435 ng/g.
Metallothioneins
In blood plasma, MTs were detected in 100 % of samples from Pucté (n = 5) and La Union (n = 6) and one sample (50 %) from Cocoyol. No MTs were detected in caudal scutes. No significant relationships between MT concentrations and TL, SVL, and body mass were observed. The disparities in sample size of juveniles, subadults, and adults (4:7:2) precluded some statistical analyses (Table 2). Nevertheless, mean concentrations of Cd and MTs in the three size classes were closely related but not significant (Fig. 2). Finally, Spearman correlation test showed no relationships between metals (Cd and Hg) and MT concentrations.
Discussion
Heavy Metals in Blood Plasma
Because there have been no studies of heavy-metal concentrations in blood plasma of crocodiles, the results for Morelet’s crocodile in this study can only be compared with similar studies of other reptile groups. The presence of Cd and Hg in blood plasma may be due to recent exposure (Shaikh and Smith 1984). However, studies in water snakes (Nerodia sipedon) suggest that Hg in blood of these organisms is directly proportional to the accumulation in internal tissues (liver, kidney, and muscle) (Burger et al. 2005). Therefore, Hg present in plasma could be an indicator of chronic exposure.
Some studies have noted Hg in blood is directly related to the body burden of Hg in organisms (i.e., the total amount before excretion). When exposure is recurrent, this becomes a persistent body burden (Burger et al. 2005). Hg detected in plasma of Morelet’s crocodiles in this study is directly proportional to the TL, SVL and body mass of individuals. This suggests that body burdens of Hg increase with increasing body size (Andreani et al. 2008), a pattern also observed in wild cottonmouths (Agkistrodon piscivorus) using liver, kidney, and tail tips (Rainwater et al. 2005). It is noteworthy that the concentrations of Hg obtained in this study (4–29 ppb) are within the values reported for other wildlife species (<50 ppb) including birds, mammals, and reptiles (Lehner et al. 2013). If Hg concentrations in plasma and internal tissues are directly proportional (Burger et al. 2005), it is likely that Hg in plasma represents a persistent body burden and not a recent exposure. To corroborate this theory, it would be pertinent to analyze relationships between plasma and internal tissues.
The highest values of Hg in blood plasma of adults (according to significant differences between size classes) confirm that there is a correlation between this metal and crocodiles size. However, because Hg was only analyzed in two adults, we recommend a larger sample size and a similar number of individuals by size classes to increase the statistical power for these correlations. Although values are within those reported in reptiles (Lehner et al. 2013), biological and genetic effects of chronic exposure to Hg in crocodiles remain unknown (Schneider et al. 2012). Even if Hg in plasma represented the total body burden, it must be noted that crocodilians appear to possess a remarkable capacity to accumulate Hg without presenting toxic effects, e.g., emaciation, malformations, or abnormal motor coordination (Peters 1983; Rainwater et al. 2002).
Heavy Metals in Caudal Scutes
The results obtained for caudal scutes indicate that Morelet’s crocodiles in the Rio Hondo have been exposed to Cd and Hg and that these metals are accumulated in their tissues. Some studies in Alligator mississippiensis (Heaton-Jones et al. 1997; Jagoe et al. 1998; Burger et al. 2000) and water snakes (Nerodia sipedon) (Burger et al. 2005) also reported accumulation of heavy metals in internal and external tissues (scutes). Burger et al. (2000) mentioned the strong correlation between scutes and internal tissues in A. mississippiensis and suggested that high concentrations of Cd and Hg may represent internal accumulation.
Overall, Cd and Hg concentrations detected in scutes obtained in this study were greater than those reported for Morelet’s crocodiles in northern Belize (Table 3), suggesting that exposure to these metals is greater in the Rio Hondo basin. It was noted that Hg concentrations from La Union (561.63 ± 224.73 ng/g) were greater than those at the other transects as well as at two sites previously sampled in northern Belize (98.7 ± 21.6 ng/g) (Rainwater et al. 2007). A possible explanation could be presence of Booth’s River in Belize, which drains into the Rio Hondo (very close from La Union). This river may receive Hg coming from an area with intense use of agrochemicals by Mennonites from Blue Creek village (Magnon Basnier 2002).
Some studies suggest that Cd concentrations must exceed 200 ppm in the kidneys to produce nephrotoxic effects (for example, Shaikh and Hirayama 1979). Cd concentrations obtained from caudal scutes of C. moreletii from Rio Hondo do not exceed 145 ppb. Assuming that Cd in scutes is directly proportional to Cd in liver and kidneys (Burger et al. 2000), crocodiles sampled in the present study may not be at risk of hepatic or renal toxicity.
Metal accumulation varies among aquatic and terrestrial species depending on a variety of factors (Smith et al. 2007); thus, the finding that the highest Cd and Hg concentrations observed in this study were in caudal scutes of juveniles may be in part due to the diet, which is based mostly on aquatic gastropods, crustaceans, fish, and nonfish vertebrates (Platt et al. 2006). In fact, gastropods usually inhabit substrates rich in organic matter where contaminants are accumulated in greater concentrations (Bryan and Langston 1992). Another possible explanations could be due to transfer from mother to offspring (Smith et al. 2007; Schneider et al. 2013) or due to chronic pollution such as constant agrochemical inputs to the river (Magnon Basnier 2002). In the last scenario, juveniles with rapid metabolism accumulate greater concentration of metals than adults; however, in the case of long-term constant exposure, the relationship between metals concentration and size of the crocodiles could change (Trillanes et al. 2014).
Metallothioneins
Our results indicate that MTs are present in blood plasma but not in the caudal scutes of Morelet’s crocodiles and that MT concentrations are greater than those of metals. MTs in blood plasma may appear long before the effects of nephrotoxicity are manifested (Shaikh and Hirayama 1979). Once toxic metals are found in the tissues of the organisms, MTs bind these metals to prevent any toxic effect (Amiard et al. 2006). High concentrations of metal–MT complexes in internal organs tend to retard the transfer of metals into the blood; therefore, MT concentrations in plasma is greater (Deb and Fukushima 1999). It should also be considered MTs bind multiple metal ions; thus, they could not only regulate Cd and Hg toxicity but also that of essential metals to perform metabolic processes in the organism (Amiard et al. 2006). Similarly, metals in blood plasma not only bind to MTs but also to other proteins such as albumin and globulins (Shaikh and Hirayama 1979).
MTs in our study were not correlated with the concentrations of heavy metals, TL, SVL, or weight. In addition, no significant differences in MT concentrations between size classes were observed. Nordberg and Nordberg (2009) described that the main function of MTs in blood plasma consists of transporting metal–MT complexes to other organs. Therefore, MT synthesis does not occur in blood plasma but rather n response to the presence of Cd in the liver. Depending on the type of exposure, the release of MTs in the bloodstream may be fast (recent exposure) or slow (chronic exposure). If a chronic exposure exists, MT synthesis becomes continuous. Our findings suggest that concentrations of MTs in crocodiles may not depend on their age or size but the type of exposure. The greatest concentrations of MTs were observed in males. Previously, it was noted male organisms tend to hold greater amounts of MTs than females, though this trend has not been clearly described (Nordberg and Nordberg 2009).
The trend observed in Cd and MTs may be due to this protein induction and the formation of Cd–MT complexes, whereas in Hg no trend related to MTs was noted. Chan et al. (1992) performed a controlled assay (7 days) in which they injected Cd, Hg, Cd–MT and Hg–MT in rats and observed that both Hg as well as Hg–MT induce synthesis of MTs only in kidneys, unlike Cd and Cd–MT, which induce synthesis in both liver (Cd) and kidneys (Cd–MT). In addition, MT levels in blood plasma increased after 3 days of injecting Cd, which did not occur with injections of Hg. These findings suggest that hepatic MT significantly influences MTs in blood plasma and that Cd is the metal inducing the synthesis of MTs in the liver. Because Hg does not induce MTs in the liver, the presence of Hg or Hg–MT in plasma is not significant (Chan et al. 1992).
Although some studies have demonstrated the presence of MTs in skin (Santucci et al. 2000), in this study no MTs in scutes of Morelet’s crocodile were detected. This may be an artifact of improper extraction of scute cytosol due to their hardness. Nevertheless, MTs may not express in crocodile’s skin. Dobrovoljc et al. (2012) examined MT in different organs of salamanders (Necturus maculosus) and toads (Bufo bufo). Interestingly, both species presented concentrations of MTs and Cd-MT in liver and kidneys; however, although MTs were not expressed in the skin, Cd concentrations were detected. This proves that heavy metals are stored in caudal scutes, but MTs are not synthesized; thus, metal–MT complexes are not formed.
Conclusion
The results of the present study showed that Morelet’s crocodile is exposed to heavy-metal pollution in the study area as well as its capacity, like other crocodilians, to accumulate toxic metals (Delany et al. 1988; Odierna 1995; Yanochko et al. 1997; Jagoe et al. 1998; Jeffree et al. 2001, 2005; Campbell 2003, Rainwater et al. 2002, 2007; Trillanes et al. 2014). Although crocodiles sampled in the study area did not show apparent signs of toxicity or disease, and despite that a recent study determined that most of the crocodiles from Rio Hondo exhibit a good body condition (Cedeño-Vázquez et al. 2011), contamination by metals such as Cd and Hg may represent a threat to the C. moreletii population in this area in the future. Crocodilians are susceptible to chronic effects of contaminants on reproduction and long-term health (Brisbin et al. 1998; Guillette et al. 2000; Lance et al. 2006) that are not visually or externally measurable (i.e., decreases in fertility, egg quality, hatchling health and survival). For example, in reptiles Hg may cause adverse effects such as behavioral and endocrine disruptions (Schneider et al. 2013).
In this study, heavy metals and MTs in blood plasma suggest recent exposure (Shaikh and Hirayama 1979; Shaikh and Smith 1984). However, Hg in plasma suggests an accumulation in inner tissues in addition to chronic exposure to this metal (Burger et al. 2005). Therefore, to determine the type of exposure (recent or chronic), further studies of heavy metals and MTs in blood fractions, urine, feces, caudal scutes, and internal tissues are required.
Interestingly, we observed a similar pattern in the concentrations of Cd and MTs (Fig. 2). The presence of MTs in blood plasma may fulfill the role of detoxification by binding Cd to form the complex Cd–MT. However to support this finding, Cd–MT analysis is needed for a better understanding of detoxification strategies.
Finally, because this is the first report of heavy metals and MTs in plasma of Morelet’s crocodile in its natural environment, we recommend conducting studies with a larger and similar sample size by size classes including egg and consideration of other metals (Pb, Cu, Zn, selenium, and iron) in the analyses. In addition, according to the results of our study, the first for this species in Rio Hondo, long-term monitoring is essential to assess health and quality of habitat in the basin.
References
Almli B, Mwase M, Sivertsen T, Musonda MM, Flåøyen A (2005) Hepatic and renal concentrations of 10 trace elements in crocodiles (Crocodylus niloticus) in the Kafue and Luangwa rivers in Zambia. Sci Total Environ 337:75–82
Amiard JC, Amiard-Triquet C, Barka S, Pellerin J, Rainbow PS (2006) Metallothioneins in aquatic invertebrates: their role in metal detoxification and their use as biomarkers. Aquat Toxicol 76(2):160–202
Andreani G, Santoro M, Cottignoli S, Fabbri M, Carpenè E, Isani G (2008) Metal distribution and metallothionein in loggerhead (Caretta caretta) and green (Chelonia mydas) sea turtles. Sci Total Environ 390:287–294
Bell JU, Lopez JM (1985) Isolation and partial characterization of a cadmium binding protein from the liver of alligators exposed to cadmium. Comp Biochem Physiol C 82(1):123–128
Bienengräber M, Forderkunz S, Klein D, Summer KH (1995) Determination of Cu-containing metallothionein: comparison of Ag saturation assay, thiomolybdate assay, and enzyme-linked immunosorbent assay. Anal Biochem 228(1):69–73
Brisbin Jr IL, Jagoe CH, Gaines KF, Gariboldi JC (1998) Environmental contaminants as concerns for the conservation biology of crocodilians. In: Proceedings of the 14th Working meeting of the Crocodile Specialist Group of the SSC of the IUCN—The World Conservation Union, Gland, Switzerland, pp 155–173
Bryan GW, Langston WJ (1992) Bioavailability, accumulation and effects of heavy metals in sediments with special reference to United Kingdom estuaries: a review. Environ Pollut 76(2):89–131
Buenfil-Rojas M, Flores-Cuevas N (2007) Determinación de metales pesados (As, Cd, Hg y Pb) presentes en el Río Hondo, Quintana Roo. In: VI Congreso Internacional y XII Nacional de Ciencias Ambientales Chihuahua, Mexico, pp 435–439
Burger J, Gochfeld M, Rooney AA, Orlando EF, Woodward AR, Guillette LJ (2000) Metals and metalloids in tissues of American alligators in three Florida lakes. Arch Environ Contam Toxicol 38:501–508
Burger J, Campbell KR, Campbell TS, Shukla T, Jeitner C, Gochfeld M (2005) Use of skin and blood as nonlethal indicators of heavy metal contamination in northern water snakes (Nerodia sipedon). Arch Environ Contam Toxicol 49(2):232–238
Campbell KR (2003) Ecotoxicology of crocodilians. Appl Herpetol 1:45–163
Campbell JW, Waters MN, Tarter A, Jackson J (2010) Heavy metal and selenium concentrations in liver tissue from wild American alligator (Alligator mississippiensis) livers near Charleston, South Carolina. J Wildl Dis 46(4):1234–1241
Cedeño-Vázquez JR, Ross JP, Calmé S (2006) Population status and distribution of Crocodylus acutus and C. moreletii in southeastern Quintana Roo, Mexico. Herpetol. Nat Hist 10(1):17–30
Cedeño-Vázquez JR, González-Ávila F, Castro-Pérez J (2011) Condición corporal del cocodrilo de pantano (Crocodylus moreletii) en el Rio Hondo, Quintana Roo, Mexico. Quehacer Científico en Chiapas 1(11):19–26
Chan HM, Satoh M, Zalups RK, Cherian MG (1992) Exogenous metallothionein and renal toxicity of cadmium and mercury in rats. Toxicology 76(1):15–26
Comisión Nacional del Agua (2012) Atlas digital de agua México. Sistema Nacional de Información del Agua. Subdirección Técnica, CONAGUA. http://www.conagua.gob.mx/atlas/ciclo20.html. Accessed 08 April 2014
Deb SC, Fukushima T (1999) Metals in aquatic ecosystems: Mechanisms of uptake, accumulation and release-ecotoxicological. Int J Environ Stud 56:385–417
Defew LH, Mair JM, Guzman HM (2005) An assessment of metal contamination in mangrove sediments and leaves from Punta Mala Bay, Pacific Panama. Mar Pollut Bull 50(5):547–552
Delany MF, Bell JU, Sundlof SF (1988) Concentrations of contaminants in muscle of the American alligator in Florida. J Wildl Dis 24(1):62–66
Dobrovoljc K, Falnoga I, Žnidarič MT, Mazej D, Ščančar J, Bulog B (2012) Cd, Cu, Zn, Se, and metallothioneins in two amphibians, Necturus maculosus (Amphibia, Caudata) and Bufo bufo (Amphibia, Anura). Biol Trace Elem Res 150(1–3):178–194
Euan-Avila JI, Liceaga-Correa MA, Rodriguez-Sanchez H (2002) Caracterización de fuentes no puntuales de contaminación agrícola en el municipio de Othón P. Blanco en Quintana Roo y su potencial influencia en la Bahía de Chetumal. In: Rosado-May FJ, Romero-Mayo R, De-Jesús-Navarrete A (eds) Contribuciones de la ciencia al manejo costero integrado de la Bahía de Chetumal y su área de influencia. UQROO, Chetumal, Quintana Roo, pp 197–204
Fingerman M, Devi M, Reddy PS, Katyayani R (1996) Impact of heavy metal exposure on the nervous system and endocrine-mediated processes in crustaceans. Zool Stud 35(1):1–8
Garcia-Rios V, Gold-Bouchot G (2003) Trace metals in sediments from Bahia de Chetumal, Mexico. Bull Environ Contam Toxicol 70(6):1228–1234
Guillette LJ Jr, Crain DA, Gunderson MP, Kools SAE, Milnes MR, Orlando EF et al (2000) Alligators and endocrine disrupting contaminants: a current perspective. Am Zool 40:438–452
Heaton-Jones TG, Homer BL, Heaton-Jones DL, Sundlof SF (1997) Mercury distribution in American alligators (Alligator mississippiensis) in Florida. J Zoo Wildl Med 28(1):62–70
Hidalgo J, Chung R, Penkowa M, Vašák M (2009) Structure and function of vertebrate metallothioneins. In: Sigel A, Sigel H, Sigel RKO (eds) Metallothioneins and related chelators. Metal ions in life science, vol 5. Royal Society of Chemistry, Cambridge, pp 279–317
Jagoe CH, Arnold-Hill B, Yanochko GM, Winger PV, Brisbin IL Jr (1998) Mercury in alligators (Alligator mississippiensis) in the southeastern United States. Sci Total Environ 213(1):255–262
Jeffree RA, Markich SJ, Twining JR (2001) Element concentrations in the flesh and osteoderms of estuarine crocodiles (Crocodylus porosus) from the Alligator Rivers region, northern Australia: Biotic and geographic effects. Arch Environ Contam Toxicol 40:236–245
Jeffree RA, Markich SJ, Tucker AD (2005) Patterns of metal accumulation in osteoderms of the Australian freshwater crocodile, Crocodylus johnstoni. Sci Total Environ 336:71–80
Lance VA, Horn TR, Elsey RM, de Peyster A (2006) Chronic incidental lead ingestion in a group of captive reared alligators (Alligator mississippiensis): possible contribution to reproductive failure. Comp Biochem Physiol C 142:30–35
Lehner AF, Rumbeiha W, Shlosberg A, Stuart K, Johnson M et al (2013) Diagnostic analysis of veterinary dried blood spots for toxic heavy metals exposure. J Anal Toxicol 37(7):406–422
Magnon Basnier C (2002) El río Hondo como componente hidrológico de la Bahía de Chetumal y como corredor biológico compartido amenazado. In: Rosado-May FJ, Romero-Mayo R, De Jesús-Navarrete A (eds) Contribuciones de la ciencia al manejo costero integrado de la Bahía de Chetumal y su área de influencia. UQROO, Chetumal, Quintana Roo, pp 23–32
Margoshes M, Vallee BL (1957) A cadmium protein from equine kidney cortex. J Am Chem Soc 79:4813–4814
Messel H, Vorlicek GC, Wells AC, Green WJ (1981) Surveys of tidal rivers systems in the Northern Territory of Australia and their crocodile populations. Monograph No 1. Pergamon, Sidney, Australia
Nordberg M, Nordberg G (2009) Metallothioneins: Historical development and overview. Metal ions in life sciences, vol 5. Royal Society of Chemistry, Cambridge, pp 1–29
Odierna E (1995) The occurrence of lead in Brazilian caiman as determined by atomic absorption spectrometry: a potential ecological disaster. Dissertation, Manhattan College, USA
Ortiz-Cervantes A, Rubio-Lozano R (2012) Reptiles. Características generales, biología y diversidad de especies. In: Curso de cría, mantenimiento y patología de animales exóticos. University of Murcia, Spain, pp 27–30
Perry EF, Koirtyohann SR, Perry HM Jr (1975) Determination of cadmium in blood and urine by graphite furnace atomic absorption spectrophotometry. Clin Chem 21(4):626–629
Peters LJ (1983) Mercury accumulation in the American alligator. Dissertation, University of Florida
Platt S, Rainwater T, Finger A, Thorbjarnarson JB, Anderson T, McMurry S (2006) Food habits, ontogenetic dietary partitioning and observations of foraging behaviour of Morelet’s crocodile (Crocodylus moreletii) in Northern Belize. Herpetol J 16:281–290
Rainwater TR, Adair BM, Platt SG, Anderson TA, Cobb GP, McMurry ST (2002) Mercury in Morelet’s crocodile eggs from Northern Belize. Arch Environ Contam Toxicol 324(1):319–324
Rainwater TR, Reynolds KD, Cañas JE, Cobb GP, Anderson TA, McMurry ST et al (2005) Organochlorine pesticides and mercury in cottonmouths (Agkistrodon piscivorus) from northeastern Texas, USA. Environ Toxicol Chem 24(3):665–673
Rainwater TR, Wu TH, Finger AG, Cañas JE, Yu L, Reynolds KD et al (2007) Metals and organochlorine pesticides in caudal scutes of crocodiles from Belize and Costa Rica. Sci Total Environ 373(1):146–156
Sánchez-Herrera O, Lopez-Segurajauregui G, Garcia-Naranjo-Ortiz de la Huerta A, Benitez-Diaz H (2011) Programa de Monitoreo del Cocodrilo de Pantano (Crocodylus moreletii) México Belice-Guatemala, CONABIO
Sanchez-Rodriguez LH (2009) Comparación de dos métodos de determinación de mercurio total en cabello por espectroscopías de absorción atómica con generador de hidruros y diferencial de efecto zeeman con pirolizador. Dissertation, National University of Colombia
Santucci B, Amantea A, Giuliano MC, Valenzano C, Cristaudo A (2000) Expression of metallothioneins-I and -II isoforms at positive patch-test sites. Contact Dermatitis 43:103–106
Scheuhammer AM, Cherian MG (1991) Quantification of metallothionein by silver saturation. Methods Enzymol 205:78–83
Schneider L, Pacheco-Peleja R, Kluczkovski A Jr, Martinez-Freire G, Marioni B, Vogt RC et al (2012) Mercury concentration in the spectacled caiman and black caiman (Alligatoridae) of the Amazon: implications for human health. Arch Environ Contam Toxicol 63:270–279
Schneider L, Maher W, Green A, Vogt RC (2013) Mercury contamination in reptiles: an emerging problem with consequences for wild life and human health. In: Kim KH, Brown RJC (eds) Mercury: sources, applications and health impacts. Nova Science Publishers, Hauppauge, NY, pp 173–232
Shaikh ZA, Hirayama K (1979) Metallothionein in the extracellular fluids as an index of cadmium toxicity. Environ Health Perspect 28:267–271
Shaikh ZA, Smith LM (1984) Biological indicators of cadmium exposure and toxicity. Experientia 40(1):36–43
Sigler L, Cedeño-Vázquez JR, Cupul-Magaña FG (2011) Método de detección visual nocturna (DVN). In: Sánchez-Herrera O, López-Segurajáuregui G, García-Naranjo-Ortiz-de la-Huerta A, Benítez-Díaz H (comps) Programa de Monitoreo del Cocodrilo de Pantano (Crocodylus moreletii). Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, México Belice-Guatemala, México
Smith PN, Cobb GP, Godard-Codding C, Hoff D, McMurry ST, Rainwater TR et al (2007) Contaminant exposure in terrestrial vertebrates. Environ Pollut 150:41–64
Trillanes CE, Pérez-Jiménez JC, Rosíles-Martínez R, González-Jáuregui M (2014) Metals in the caudal scutes of Morelet’s crocodile (Crocodylus moreletii) from the Southern Gulf of Mexico. Bull Environ Contam Toxicol 93:423–428
Triunfante P, Soares ME, Santos A, Tavares S, Carmo H, Bastos ML (2009) Mercury fatal intoxication: two case reports. Forensic Sci Int 184:e1–e6
Van-deer-Oord JJ, DeLey M (1994) Distribution of metallothionein in normal and pathological human skin. Arch Dermatol Res 286:62–68
Vázquez FJ (2005) Toxicidad comparada de zinc, plomo y mercurio para Zoea I de Chasmagnathus granulatus (Brachyura). Tesina de Ciencias Exactas y Naturales. Universidad de Belgrano. Argentina
Wolfe MF, Schwarzbach S, Sulaiman RA (1998) Effects of mercury on wildlife: a comprehensive review. Environ Toxicol Chem 17(2):146–160
Woodward AR, Marion WR (1978) An evaluation of factors affecting night-light counts of alligators. Proc Annu Conf Southeast Assoc Fish Wildl Agencies 32:291–302
Xu Q, Fang S, Wang Z, Wang Z (2006) Heavy metal distribution in tissues and eggs of Chinese alligator (Alligator sinensis). Arch Environ Contam Toxicol 50:580–586
Yamamura M, Suzuki KT (1984) Isolation and characterization of metallothionein from the tortoise Clemmys mutica. Comp Biochem Physiol C 79:63–69
Yanochko GM, Jagoe CH, Brisbin IL Jr (1997) Tissue mercury concentrations in alligators (Alligator mississippiensis) from the Florida Everglades and the Savannah River Site, South Carolina. Arch Environ Contam Toxicol 32:323–328
Acknowledgments
This research was supported by the Consejo Nacional de Ciencia y Tecnologia, El Colegio de la Frontera Sur, and Comisión Nacional para el Conocimiento y Uso de la Biodiversidad (Project “Monitoreo del cocodrilo de pantano (Crocodylus moreletii) MéxicoBelice-Guatemala”). We thank Fernando Gonzalez Avila, Raymundo Mineros Ramírez, Roberto Herrera Pavón, and Irving Rosas López for field assistance and the family Rosas Carmona for providing support and accommodation during the surveys. We thank Adriana Zavala for assistance in laboratory analyses. We thank Thomas R. Rainwater and two anonymous reviewers for valuable comments on a previous version of the manuscript. Research permits for this study were issued by the Dirección General de Vida Silvestre of the Secretaría del Medio Ambiente y Recursos Naturales (OFICIO NÚM/SGPA/DGVS/04288/12 and OFICIO NÚM/SGPA/DGVS/00911/13).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Buenfil-Rojas, A.M., Álvarez-Legorreta, T. & Cedeño-Vázquez, J.R. Metals and Metallothioneins in Morelet’s Crocodile (Crocodylus moreletii) from a Transboundary River Between Mexico and Belize. Arch Environ Contam Toxicol 68, 265–273 (2015). https://doi.org/10.1007/s00244-014-0088-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-014-0088-5