Abstract
Prevalence of kidney stones is increasing worldwide, flexible ureterorenoscopy (f-URS) is the most common surgical treatment. Postoperative urinary tract infection (PUTI) is the primary complication. Some risk factors are classically associated with PUTI, especially preoperative positive urinalysis (POPU). We aimed to identify risk factors for PUTI after f-URS for urolithiasis in patients with treated POPU, and to identify the different pre and postoperative pathogens. Retrospective, single-center study of all f-URS for urolithiasis between January 2004 and December 2020. Procedures with treated POPU were categorized as PUTI or no PUTI (NPUTI). We examined demographics, preoperative, perioperative and postoperative characteristics in each group. Among 1934 procedures analyzed, 401 (20.7%) had POPU; these were categorized into NPUTI (n = 352, 87.8%) and PUTI (n = 49, 12.2%). By univariate analysis, only preoperative stenting duration (76.3 in NPUTI group vs 107.7 days in PUTI group, p = 0.001) was significantly associated with a higher risk of PUTI in univariate analysis. Germ distribution was similar in both groups. We compared pre- and postoperative microbiological data for interventions with PUTI, and found that only 8.7% of pathogens were identical between pre and postoperative urinalysis. Our study shows that the rate of PUTI is higher for patients with a POPU and that preoperative stent duration is the sole risk factor in patients with POPU. The low concordance rate (8.7%) between POPU and post-operative pathogens highlights the need for further research on obtaining sterile preoperative urinalysis, or performing intraoperative culture (urines, stent or stone), to treat PUTI early with an adapted antibiotic therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The prevalence and incidence of kidney stone disease have increased over the past few decades, affecting up to 20% of the population in some countries [1, 2]. One of the main associated risks is recurrence. For urological stones requiring intervention, flexible ureterorenoscopy (f-URS) has been the most commonly used surgical treatment for over 30 years [3]. Because it is effective, minimally invasive and associated with few adverse events, [4] many scientific communities worldwide recommend it [5,6,7]. Urinary tract infection (UTI) is one of the main complications of f-URS, [4, 8,9,10,11] occurring in up to 8.5% of patients [12]. Several risk factors are classically associated with UTI, such as comorbidities, presence of material in the urinary tract, and preoperative presence of bacterial or fungal urinary colonization [12,13,14]. To screen for such preoperative urinary tract colonizations, preoperative urinalysis is recommended and commonly performed, along with antibiotic prophylaxis administered perioperatively [7, 15]. Despite these precautions, post f-URS UTI remains a cause of severe complications. It is well established that positive preoperative urinary tract cultures (POPU), despite being treated, is a risk factor for postoperative urinary tract infection (PUTI) as shown by numerous studies [12,13,14, 16, 17]. In this respect, surgical practices vary, with some surgeons only performing f-URS in case of a sterile or sterilized urinalysis, whereas others do not routinely perform a urinalysis.
In this study, we aimed to evaluate the associated factors for UTI in patients with treated POPU who underwent f-URS. The secondary objective was to determine the infectious microorganisms responsible for PUTI.
Methods
Study design and patient
We retrospectively analyzed all f-URS procedures performed for urolithiasis between January 2004 and December 2020 in our tertiary care university department. Only adults over 18 years were included. Procedures performed for diagnostic purposes, or for upper urinary tract tumors were excluded. Then, among procedures performed in patients with treated, positive preoperative urinalysis, we separated patients into a group who experienced PUTI (PUTI group) and those who did not (no PUTI (NPUTI) group) (Fig. 1).
The primary outcome was PUTI within 30 days after surgery. PUTI was defined as fever (temperature above 38.5 °C) and need for antibiotic treatment, with bacteriological documentation on either urine or blood cultures.
Pre-, per and postoperative evaluation
The following data were recorded: demographics (age, gender, body mass index (BMI), Charlson comorbidity index (CCI), history of diabetes, anticoagulant or antiplatelet agent), stone characteristics (localization, cumulative stone diameter, stone density), surgical history (presence of a preoperative stent, preoperative urine culture, preoperative antibiotic therapy, operative time, ureteral access sheath use, ureteral dilatation, laser use, complete fragmentation, stone localization, postoperative stent), surgical and anesthetic complications classed according to the Clavien–Dindo classification [18] and the additional antibiotic prophylaxis during surgery.
We identified the spectrum of antibiotics used preoperatively, and compared it to the spectrum of antibiotics used for prophylaxis during surgery. If the preoperative antibiotics had a narrower spectrum, the antibiotic prophylaxis during surgery was considered complementary.
The complete fragmentation and stone free (SF) rate was defined as the total absence of residual stone after rigorous examination at the end of the surgery and imaging evaluation.
Surgical and perioperative procedures
According to the department protocol, all patients with POPU were treated with an antibiotic appropriate for the identified pathogen 48 h before surgery and continued for 3 days thereafter. Procedures were performed by several surgeons from our urological department following a standardized protocol [4]. Patients were placed in the dorsal lithotomy position, and all surgeries were performed under general anesthesia [19]. During surgery, for additional antibiotic prophylaxis, with cefazoline, it was carried out at the discretion of the surgeon and anesthesiologist.
All procedures were preceded by careful skin preparation, comprising cleaning and disinfection according to local protocols.
Statistical analysis
Qualitative variables were analyzed by Chi-square or Fisher’s test and quantitative variables by the Mann–Whitney U test. A p-value < 0.05 was considered statistically significant. All analysis were performed using XLSTAT® 2021.3.1 (Addinsoft Inc., Paris, France).
Results
Between January 2004 and December 2020, 1934 procedures were performed: 781 in women (40.4%) and 1153 in men (59.6%). Overall, PUTI occurred in 118 interventions (6.1%). Eight patients (0.41%) had postoperative septic shock.
Among the 1934 procedures performed, 401 (20.7%) had positive preoperative urinalysis. These 401 patients were categorized according to PUTI occurrence: 352 (87.8%) interventions had no PUTI (NPUTI group) and 49 (12.2%) had PUTI. The rate of PUTI among patients with POPU was significantly higher than that observed in the overall population (6.1 vs 12.2%, p < 0.001). Patient demographics and peroperative data were comparable in both groups; these data are summarized, as well as stone characteristics and peroperative data in Tables 1 and 2. The hospitalization duration was longer in the PUTI group (4.98 vs 2.96 days, p < 0.001). In the NPUTI group, 324 (92.0%) interventions were complication free. Nineteen (5.4%) Clavien–Dindo stage I complications, 9 (2.6%) stage II, and 0 stage III, IV or V complications were observed. In the PUTI group, all procedures had PUTI and therefore were classified at least as Clavien–Dindo stage II complications (94%), additionally, one stage III (2.0%), one stage IV (2.0%) and one stage V corresponding to a death (2.0%) were observed.
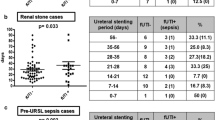
By univariate analysis, only the duration of preoperative stent differed significantly between groups (107.7 vs 76.3 days, p = 0.001).
We analyzed microbiological data from treated preoperative urinalysis in both groups. The germs identified were distributed in similar proportions in both groups, with Gram-Negative Bacilli (GNB) being the predominant family (accounting for 59.1% in the NPUTI group and 57.1% in the PUTI group), followed by Gram-Positive Cocci (GPC) (21.0% and 16.3%, respectively), fungus (2.6% and 4.1%, respectively) and Gram-Positive Bacilli (GPB) (1.1% in the NPUTI group). Two germs were identified in 7.7% in the PUTI group, and 16.3% in the NPUTI group (Table 1). There was no difference between the preoperative antibiotic therapy used (Table 1).
We compared the microorganisms from preoperative urinalysis with those found during postoperative infectious episodes for all procedures with PUTI. The microorganisms responsible for PUTI were as follows: 55.1% GNB, 20.4% GPC, 14.3% a combination of two germs and 2.0% fungus (Table 3).
In 65.2% (n = 30) of cases, the germs identified in the pre- and postoperative samples were different. In the remaining 34.8% (n = 16) the pre- and postoperative germs were identical. Of these, 26.1% (n = 12) had the same germ with a different antibiogram between pre- and postoperative urinalysis, while only 8.7% (n = 4) had PUTI with complete agreement between the pre- and postoperative urinalysis microorganism (Fig. 2).
Discussion
In this cohort of procedures with POPU, even properly treated, the only persistent associated factor was the duration of the preoperative ureteric stenting. These predisposing factors have been highlighted in different systematic reviews, and while age, BMI, sex gender, diabetes mellitus have a controversial role according to different studies, POPU is uniformly found [17, 20,21,22]. The microorganism identified in the POPU versus those responsible for PUTI were only minimally concordant: the same germ was found in 26.1%, but the antibiogram was identical in only 8.7% of cases.
We observed a PUTI rate of 6.1% in overall cohort of 1934 procedures, which is in the lower range of average, compared to existing studies, where infectious complications are reported to occur in 6 to 8.5% [17, 21]. A recent large study including 71,305 procedures in England showed a 6.8% rate of postoperative urinary tract infection [11]. A study examining patients who had f-URS after obstructive acute pyelonephritis found a higher rate of PUTI, at 16%. In our study, this criterion did not emerge as significant but for procedures with POPU, the PUTI rate was 12.2% [23].
To the best of our knowledge, no study has specifically assessed PUTI rates in patients with preoperative positive urinalysis. The absence of associated factors in this population, except for the duration of the preoperative ureteric stent, may suggest that preoperative positive urinalysis, although correctly treated, could be an important risk factor. It may be worth considering, as previously recommended by the Committee of Infectiology of the French Urologist Association in 2015, performing f-URS only after obtaining a sterile urine culture [24].
Both the European Association of Urology and the French Society of Anesthesia and Intensive Care (SFAR) recommend performing urological endoscopic surgeries with a sterile urinalysis or one that has been treated curatively. Perioperative antibiotic prophylaxis is also recommended; however, no specific guidelines address the necessity for antibiotic prophylaxis in cases where a positive urinalysis has already been treated. The impact of perioperative antibiotic prophylaxis when the preoperative urinalysis is positive remains to be investigated. Indeed, our study does not reveal any statistical association between the group with or without additional prophylaxis. This could potentially be explained by reduced number of participants; however, as far as we know, this is the only study to compares additional antibiotic prophylaxis in patients with a positive preoperative urinalysis [19, 25].
We can underline that the distribution of preoperative pathogens was similar in both groups (PUTI and NPUTI). GNB were predominant, followed by GPC, fungus, and GPB. Cole et al. recently studied unplanned hospitalizations following URS. They found that 2.4% of patients were hospitalized with a postoperative infectious complication. The main risk factors were a higher CCI, history of recurrent urinary tract infection, stone size, intraoperative complication and non-SF status. What is interesting in this study is the distribution of postoperative infection germs, which was very similar to ours, namely 61.5% Gram-negative (55.1% in our study), 19.2% Gram-positive (20.4% of GPC and 2% of GPB), with the exception of 15.4% fungus in Cole’s study (vs 2.0% in our work), and 3.8% of patients with two germs (14.3%) [26].
Finally, we examined the agreement between preoperative germs and those found during postoperative infections. We found that 65.2% of germs differed between preoperative urine culture and postoperative infection identification. Nevo et al. studied the concordance of germs between the preoperative urinalysis, ureteric stent culture and postoperative infection in patients who all had previous ureteric stenting. They found no agreement between preoperative urine culture and stent culture, possibly due to the biofilm that can form on the preoperative ureteric stent. Urine cultures do not seem to be a reliable predictor of the complete absence of bacteria in the urinary tract. They also showed that a positive stent culture was a risk factor for PUTI and could be used to choose the antibiotic therapy for septic patients [27].
In our work, when pre and postoperative germ identification was concordant, 26.1% had a different antibiogram. Finally, only 8.7% of the interventions with PUTI had complete agreement between pre and postoperative urinalysis. A recent study focused on the discordance between urine culture, stent culture, stone culture and postoperative blood culture. They showed a statistical association between Foreign Body Colonization (FBC) and the risk for PUTI complication. FBC seems to be a better indicator of the germ responsible for PUTI [16]. These data suggest that preoperative urinalysis is not a reliable indicator of possible germs during PUTI. It may be necessary to discuss the utility of systematic intraoperative pyelic urinalysis, stent culture or analysis of a stone fragment to better anticipate PUTI.
Our work has several limitations. First, this is a retrospective, single-center study with potential selection bias due to missing data. Moreover, if a patient had mild infection, they may have consulted a general practitioner, and not informed us of the complication during follow-up consultation. However, as the designated emergency center in our region, and with some patients undergoing outpatient surgery receiving a systematic follow-up call on postoperative day one, the large majority of PUTI case were referenced. Despite these limitations, our study has several strengths. We have a large cohort of f-URS interventions, and even in the subgroup of interventions with a positive preoperative treated urinalysis, we totaled 401 procedures. Our general results are in agreement with the existing literature, indicating that our population is representative of general population. Our work is one of the few to study the pre and postoperative microbiological identification.
Conclusion
Our study shows a PUTI rate similar to that reported in the literature, at 6.1% overall. However, this rate was significantly higher in the specific population of patients with POPU, at 12.2%. Among patients with POPU, preoperative duration of the ureteric stent differed significantly between those with, and those without PUTI. These results suggest that POPU and a prolonged duration of ureteral stenting preoperatively may be important risk factors for PUTI.
Our findings reveal a low rate of agreement between peroperative urinalysis and the pathogens responsible for postoperative infections, with only 26.1% having similar bacteria and 8.7% having similar antibiogram.
Data availability
The datasets generated and analyzed durent the current study are available from the corresponding author on reasonable request.
References
Ziemba JB, Matlaga BR (2017) Epidemiology and economics of nephrolithiasis. Investig Clin Urol 58(5):299. https://doi.org/10.4111/icu.2017.58.5.299
Romero V, Akpinar H, Assimos DG (2010) Kidney stones: a global picture of prevalence, incidence, and associated risk factors. Rev Urol 12(2–3):e86-96
Reis Santos JM (2018) Ureteroscopy from the recent past to the near future. Urolithiasis 46(1):31–37. https://doi.org/10.1007/s00240-017-1016-8
Delorme G, Huu YN, Lillaz J, Bernardini S, Chabannes E, Guichard G, Bittard H, Kleinclauss F (2012) Ureterorenoscopy with holmium-yttrium-aluminum-garnet fragmentation is a safe and efficient technique for stone treatment in patients with a body mass index superior to 30 kg/m 2. J Endourol 26(3):239–243. https://doi.org/10.1089/end.2011.0391
Chabannes É, Bensalah K, Carpentier X, Bringer JP, Conort P, Denis É, Dore B, Estrade V, Gautier JR, Hadjadj H, Hubert J, Hoznek A, Lechevallier E, Meria P, Mozer P, Saussine C, Yonneau L, Traxer O (2013) Prise en charge urologique des calculs rénaux et urétéraux de l’adult mise au point du comité lithiase de l’association française d’urologie (CLAFU). Asp généraux. Prog En Urol 23(16):1389–1399. https://doi.org/10.1016/j.purol.2013.08.315
Estrade V, Bensalah K, Bringer JP, Chabannes E, Carpentier X, Conort P, Denis E, Doré B, Gautier JR, Hadjadj H, Hubet J, Hoznek A, Lechevallier E, Meria P, Mozer P, Saussine C, Yonneau L, Traxer O (2013) Place de l’urétérorénoscopie souple en première intention pour le traitement des calculs du rein résultats de l’enquête de pratique du comité lithiase de l’AFU réalisée en 2011. Prog En Urol 23(1):22–28. https://doi.org/10.1016/j.fpurol.2017.03.002
Türk C, Petřík A, Sarica K, Seitz C, Skolarikos A, Straub M, Knoll T (2016) EAU guidelines on diagnosis and conservative management of urolithiasis. Eur Urol 69(3):468–474. https://doi.org/10.1016/j.eururo.2015.07.040
Du K, Wang RS, Vetter J, Paradis AG, Figenshau RS, Venkatesh R, Desai AC (2018) Unplanned 30-Day encounters after ureterorenoscopy for urolithiasis. J Endourol 32(12):1100–1107. https://doi.org/10.1089/end.2018.0177
Moses RA, Ghali FM, Pais VM, Hyams ES (2016) Unplanned hospital return for infection following ureteroscopy-can we identify modifiable risk factors? J Urol 195(4 Pt 1):931–936. https://doi.org/10.1016/j.juro.2015.09.074
Jacquemet B, Martin L, Pastori J, Bailly V, Guichard G, Bernardini S, Chabannes E, Bittard H, Kleinclauss F (2014) Comparison of the efficacy and morbidity of flexible ureterorenoscopy for lower pole stones compared with other renal locations. J Endourol 28(10):1183–1187. https://doi.org/10.1089/end.2014.0286
Veeratterapillay R, Gravestock P, Harding C, Shaw M, Fitzpatrick J, Keltie K, Cognigni P, Sims A, Rogers A (2023) Infection after ureteroscopy for ureteric stones: analysis of 71 305 cases in the hospital episode statistics database. BJU Int 131(1):109–115. https://doi.org/10.1111/bju.15850
Senocak C, Ozcan C, Sahin T, Yilmaz G, Ozyuvali E, Sarikaya S, Resorlu B, Oguz U, Bozkurt OF, Unsal A, Adsan O (2018) Risk factors of infectious complications after flexible uretero-renoscopy with laser lithotripsy. Urol J 15(4):158–163
Ma YC, Jian ZY, Yuan C, Li H, Wang KJ (2020) Risk factors of infectious complications after ureteroscopy: a systematic review and meta-analysis based on adjusted effect estimate. Surg Infect 21(10):811–822. https://doi.org/10.1089/sur.2020.013
Blackmur JP, Maitra NU, Marri RR, Housami F, Malki M, McIlhenny C (2016) Analysis of factors’ association with risk of postoperative urosepsis in patients undergoing ureteroscopy for treatment of stone disease. J Endourol 30(9):963–969. https://doi.org/10.1089/end.2016.0300
Cuvelier G, Legrand G, Vignes B, Lechevallier É, Rébillard X, Coloby P (2013) Recommandations AFU/SFAR/AFCA/ANAP Chirurgie ambulatoire en urologie.;23:69. http://sfar.org/wp-content/uploads/2015/10/2_PUROL_Recommandations-chirurgie-ambulatoire-en-urologie.pdf
Nevo A, Golomb D, Lifshitz D, Yahav D (2019) Predicting the risk of sepsis and causative organisms following urinary stones removal using urinary versus stone and stent cultures. Eur J Clin Microbiol Infect Dis 38(7):1313–1318
Sun J, Xu J, OuYang J (2020) Risk factors of infectious complications following ureteroscopy: a systematic review and meta-analysis. Urol Int 104(1–2):113–124
Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, Schulick RD, de Santibanes E, Pekolj J, Slankamenac K, Bassi C, Vonlanthen R, Padbury R, Cameron J, Makuuchi M (2009) The clavien-dindo classification of surgical complications: five-year experience. Ann Surg 250(2):187–196
Martin C, Auboyer C, Dupont H, Gauzit R, Kitzis M, Lepape A, Mimoz O, Montravers P, Pourriat JL (2011) Société française d’anesthésie et de réanimation antibioprophylaxie en chirurgie et médecine interventionnelle (patients adultes) actualisation 2010. Ann Fr Anesth Réanimation 30(2):168–190
Chugh S, Pietropaolo A, Montanari E, Sarica K, Somani BK (2020) Predictors of urinary infections and urosepsis after ureteroscopy for stone disease: a systematic review from EAU section of urolithiasis (EULIS). Curr Urol Rep 21(4):16. https://doi.org/10.1007/s11934-020-0969-2
Baboudjian M, Gondran-Tellier B, Abdallah R, Sichez PC, Akiki A, Gaillet S, Delaporte V, Karsenty G, Lechevallier E, Boissier R (2020) Predictive risk factors of urinary tract infection following flexible ureteroscopy despite preoperative precautions to avoid infectious complications. World J Urol 38(5):1253–1259. https://doi.org/10.1007/s00345-019-02891-8
Laclergerie F, Jacquemet B, Guichard G, Bernardini S, Chabannes E, Martin L, Pastori J, Bailly V, Bittard H, Kleinclauss F (2014) Urétérorénoscopie souple avec laser holmium-YAG dans la prise en charge des lithiases urinaires chez le patient obèse : résultats d’une cohorte monocentrique. Prog En Urol 24(10):634–639. https://doi.org/10.1016/j.purol.2014.03.005
Ito K, Takahashi T, Somiya S, Kanno T, Higashi Y, Yamada H (2022) Risk factors for infectious complications of ureteroscopy after obstructive acute pyelonephritis. Urol J. https://doi.org/10.22037/uj.v19i.7241
Bruyère F, Sotto A, Escaravage L, Cariou G, Mignard JP, Hoznek CP, A, Bernard L, Boiteux JP, Thibault M, Soussy CJ, Bugel H, (2010) Recommandations de bonnes pratiques cliniques : l’antibioprophylaxie en chirurgie urologique, par le comité d’infectiologie de l’association française d’urologie (CIAFU). Prog En Urol 20(2):101–108. https://doi.org/10.1016/j.purol.2009.11.003
Bonkat G, Bartoletti R, Bruyère F, Cai T (2017) EAU Guidelines on Urological Infections. Uro - Eur Association of Urol. https://doi.org/10.1016/j.euf.2018.03.004
Cole A, Telang J, Kim TK, Swarna K, Qi J, Dauw C, Seifman B, Abdelhady M, Roberts W, Hollingsworth J, Ghani KR (2020) Infection-related hospitalization following ureteroscopic stone treatment: results from a surgical collaborative. BMC Urol 20(1):176. https://doi.org/10.1186/s12894-020-00720-4
Nevo A, Mano R, Schreter E, Lifshitz DA (2017) Clinical implications of stent culture in patients with indwelling ureteral stents prior to ureteroscopy. J Urol 198(1):116–121. https://doi.org/10.1016/j.juro.2017.01.064
Funding
No funding was received for this work.
Author information
Authors and Affiliations
Contributions
J SIMON : Data collection, data analysis, manuscrit writing and editing F KLEINCLAUSS : Manuscript editing E CHABANNES : Manuscript editing K BOUILLER : Manuscript editing A FRONTCZAK : Data analysis, manuscript writing and editing.
Corresponding author
Ethics declarations
Conflict of interest
No competing financial interests exist.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Simon, J., Kleinclauss, F., Chabannes, É. et al. Urinary tract infection after flexible ureterorenoscopy for urolithiasis in patients with positive treated preoperative urinalysis. Urolithiasis 52, 45 (2024). https://doi.org/10.1007/s00240-024-01546-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00240-024-01546-7