Abstract
Purpose
Beta-lactams (BL), the most commonly prescribed class of antibiotics, are recommended as the first-line therapy for multiple indications in infectious disease guidelines. Meropenem (MERO) is frequently used in intensive care units (ICU) to treat bacterial infections with or without sepsis. The pharmacokinetics of MERO display a large variability in patients admitted to ICUs due to altered pathophysiology. The aim of this study was to perform an external evaluation of published population pharmacokinetic models of MERO in order to test their predictive performance in a cohort of ICU adult patients.
Methods
A literature search in PubMed/Medline database was made following the PRISMA statement. External evaluation was performed using NONMEM software, and the bias and inaccuracy values were calculated.
Results
An external validation dataset from the Timone Hospital in Marseille, France, included 84 concentration samples from 27 patients. Four models of MERO were identified according to the inclusion criteria of the study. None of the models presented acceptable values of bias and inaccuracy.
Conclusion
While performing external evaluations on some populations may confirm a model’s suitability to diverse groups of patients, there is still some variability that cannot be explained nor solved by the procedure. This brings to light the difficulty to develop only one model for ICU patients and the need to develop one specific model to each population of critically ill patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Meropenem (MERO) is broad-spectrum antibiotics of the carbapenem class, with activity against both Gram-positive and Gram-negative bacteria that are widely used in critically ill patients [1]. The antimicrobial activity of MERO is time-dependent, meaning that its efficacy is related to the fraction of the dosing interval that the free plasma concentration is above the minimum inhibitory concentration (fT>MIC) [2]. New recommendations have been made on the optimization of beta-lactam treatment in ICU patients, such as the use of continuous or prolonged infusion of beta-lactam antibiotics in order to maximize its clinical efficacy for a target attainment of 100% fT>4xMIC [3]. Indeed, suboptimal drug exposure can lead to mortality and, possibly, the emergence of antibiotic resistance, while overexposure may be neurotoxic [4].
Population pharmacokinetic (PK) analyses develop mathematical models using specific patient group that then describe drug’s concentration in various populations and allow to optimize drug-dosing regimens [5]. Multiple population PK models have been developed during the last few years. However, these models often do not involve an external evaluation procedure, although it is recommended in order to apply the model to a clinical setting [6]. An external evaluation involves using an independent dataset to assess the accuracy of an existing model and to predict its outcome using patients with similar characteristics to those the model was developed with [7]. Knowing if the model will fit a given population of ICU patients proves to be a challenge. Besides, studies have shown that patients admitted to intensive care units (ICUs) display large variability in PK primarily due to altered pathophysiology, as opposed to healthy individuals or non-critically ill patients [8]. Due to this, optimal dosage is particularly difficult in these patients. Population pharmacokinetic models can be used in dosing simulations to predict the probability of target attainment of different dosing regimens. This proves to be useful for decision-making in a clinical setting.
The aim of this study was to perform an external evaluation of published population PK models of MERO in order to test their predictive performance in a cohort of ICU patients.
Material and methods
Patients
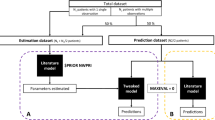
This study was a prospective, multicenter observational study performed between October 2015 and May 2017, in 2 ICUs (multidisciplinary critical care (20 beds), cardiovascular critical care (20 beds)), Timone University Hospital, Marseille, France. Ethical approval was obtained from the Institutional Review Board (Comité de Protection des Personnes Sud-Est I, Saint-Etienne, France, protocol n° 2017-A01446-47) which waived the need for written consent. Patients or next of kin was orally informed of the goal and design of the study. The antimicrobial agent assays were performed by the university-affiliated pharmacological laboratory (Department of Clinical Pharmacology and Pharmacovigilance of Timone University Hospital, Marseille, France). Blood samples were collected from patients who received a standard loading dose of 2 g of MERO and subsequently a maintenance dose of 4 g as a continuous infusion. Antibiotic maintenance dose was adjusted according to impaired renal function. Therapeutic drug monitoring was done on day 1, day 4, day 7, and at the end of treatment, and dosage was modified according to the clinician to optimize treatment. Samples were transported in 5 h to the pharmacological laboratory and were centrifuged for 10 min at 3000g and 4 °C. If necessary, plasma samples were stored at − 20 °C until analysis and MES buffer (4-Morpholineethanesulfonic acid, 1 M) was added to plasma (1/10). The assay protocol was adapted according to the method of Verdier et al. [9] and validated according to the EMA guidelines [10]. Briefly, before high-performance liquid chromatography (HPLC) analysis (Dionex Ultimate 3000 HPLC, Ultraviolet detection), sample preparation consisted of protein precipitation using acetonitrile and then liquid-liquid extraction by dichloromethane. The MIAA (5-Methoxyindole-3-acetic acid) has been chosen as internal standard. The limit of quantification was 0.5 μg/ml and the upper limit of linearity has been validated at 50 μg/ml. In case of higher concentrations observed, the sample was diluted before reanalysis.
Published models
The PRISMA statement for reporting systematic reviews and meta-analyses was used to conduct the literature review included in the study [11] (Fig. 1). A literature search in PubMed/Medline database was made using the following search terms: (meropenem AND pharmacokinetics/ OR renal elimination/ OR (pharmacokinetic* OR ((pharmaco OR drug) ADJ kinetic*) OR area under curve? OR AUC OR (renal ADJ (elimination? OR excretion? OR clearance?))) OR (((nonlinear OR non-linear) ADJ mixed effect model*) OR NONMEM OR WinNonMix OR P-PHARM OR NLMIXED OR ADAPT) AND EXP population/ OR population groups/ OR (population? or ethnic group?)). Additional relevant studies were manually screened from the identified publications.
The inclusion criteria for the published models were as follows: (1) the study was a population pharmacokinetic analysis of MERO in ICU adult patients; and (2) the publication was written in English. The articles were excluded if (1) the model was not developed with software using parametric approach or (2) the non-availability of sufficient data to rewrite the models.
Model evaluation
The external evaluation was conducted using the nonlinear mixed effect modeling (NONMEM version 7.4; ICON Development Solutions, Ellicott City, MD, USA) software. The plots were made using the R version 3.5.1. The final population PK models were re-established based on the formulas and parameters reported for each of the included publications. The external evaluation was performed without any additional fitting of the model to the data (MAXEVAL = 0 option in NONMEM). To assess the predictive performance of this model, we first compared goodness-of-fit plots of the predicted concentration and the observed concentration. The global fit of the population pharmacokinetic models was also evaluated by visual predictive checks (VPC). Prediction error (PE) was used to assess the predictive performance of the models. PE was determined by the following equation:
where Cpred and Cobs are the predicted and observed concentration, respectively [12]. The median prediction error (MDPE) and the median absolute prediction error were used as a measure of bias and inaccuracy, respectively [13].
-
1.
Inaccuracy (i.e., the size of the typical miss) of model predictions is given for the ith individual, by the median absolute performance errors (MADPEi).
-
2.
Bias (i.e., the direction and the size of the deviation from the predicted concentration) of model predictions is given for the ith individual by the median prediction error (MDPEi):
A MDPE between – 20 and 20% and a MDAPE ≤ 30% are considered as acceptable criteria for bias and inaccuracy [14]. A Bland-Altman diagram was also produced for comparison purposes and to show the trend in prediction error. If a model presented acceptable bias and accuracy, its predictive performance based on simulation would be evaluated and the statistics of the observed and simulated time-concentration profiles would be compared using VPC and NPDE tests [15].
Evaluation of predicted concentration 24 h after the first dose
As the meropenem clinical efficacy is related to concentration greater to the minimal inhibitory concentration during 100% of time interval, the predicted C24h using each of the 4 population pharmacokinetic models was examined. The C24h (after the first dose) was calculated for all patients using covariates only (a priori prediction) or by using observed meropenem concentration(s) (Bayesian estimation) [16]. The predicted C24h estimated using the a priori and Bayesian approach were compared.
Results
Patients
Thirty ICU patients on MERO infusion were enrolled in this study. Three patients were excluded because of incomplete information. The final database consisted of 27 patients (15 men and 12 women) (Table 1). In total, 84 concentrations were used for external evaluation. Creatinine clearance was estimated using the Cockcroft-Gault formula. The median Simplified Acute Physiology Score (SAPS) II was 38 (range 17–87) and the median Sepsis-related Organ Failure Assessment (SOFA) score was 5 (range 2–15). Seventeen patients had documented infections (Enterobacter aerogenes (n = 1), Enterobacter cloacae (n = 3), Enterococcus faecium (n = 2), Escherichia coli (n = 7), Klebsiella pneumoniae (n = 2), Pseudomonas aeruginosa (n = 2), Morganella morganii (n = 1), Raoultella ornithicolithica (n = 1), Serratia marcescens (n = 1), Staphylococcus aureus (n = 1)). Comedication included amikacin (n = 8), gentamicin (n = 1), and vancomycin (n = 2).
Published models
Seven models were screened [17,18,19,20, 22,23,24], but three were excluded due to insufficient information. We tried to reach the author of one article in order to obtain the values of the residual variability without any success. The predictive performance of four models was evaluated using the external evaluation dataset [17,18,19,20]. The characteristics of the patients in the included studies are summarized in Table 1. The details of the evaluated models are listed in Table 2. Three studies described MERO pharmacokinetics with two-compartment models. Several covariates were used: creatinine clearance (n = 3), bodyweight (n = 2), age (n = 2), and albumin (n = 1). Estimated values of clearance (CL) and central volume of distribution (V or V1) were between 9.38 and 13.6 L/h and 7.9 and 26.2 L, respectively.
Model evaluation
Figure 2 shows plots of predicted concentrations versus observed concentrations for each evaluated model. All evaluated models presented an under-prediction of concentrations with large inaccuracy. The values of bias and inaccuracy of the evaluated models are described in Table 3. The values ranged between − 14.4 and − 33.6 for the bias and 32.4 and 52.8 for the inaccuracy. The Bland-Altman diagram confirmed the trend in prediction error (Supplementary material, Figure S1). The predictive performance of the predicted meropenem concentration time profiles as revealed from the VPC of the population pharmacokinetic models was highly variable (Supplementary material, Figure S2).
Evaluation of predicted concentration 24 h after the first dose
The C24h of a course of therapy was predicted a priori (covariates only) and estimated from the observed meropenem concentrations (Bayesian) with each model. The distribution of the predicted meropenem C24h was highly heterogeneous (Fig. 3). Models such as that of Roberts et al. [20] demonstrated similar ranges for the a priori and Bayesian estimated C24h. This indicated that an a priori prediction of this model is promising, but can be further improved with the inclusion of observed meropenem concentrations and Bayesian.
Discussion
Several population PK models of MERO [17,18,19,20, 22,23,24,25,26,27,28,29,30,31] in critically ill patients have been published over the past decades, and predictability was often not fully evaluated [32]. After evaluation, none of the selected models in this study was found to be suitable for clinical application as they did not meet the defined criteria. Four population PK models of MERO [17,18,19,20] corresponded to the inclusion criteria and were evaluated, but none turned out to be valid for the external validation dataset as bias and inaccuracy were outside of the determined values. Amongst the four models, two were outright eliminated, while the model of Roberts et al. [20] had a MDPE and MDAPE outside of the acceptable values, but near the limits. However, this model showed a low difference between the a priori and Bayesian estimated C24h. This indicated that an a priori prediction of this model could be promising. The best predictive model of MERO was developed by Delattre et al. [17], as its bias was within the criteria, and the inaccuracy was close to the determined value, albeit outside of the range. However, bias was close to the limits of admissibility and showed the risk of under-prediction of this model. Moreover, the difference between the a priori and Bayesian estimated C24h was as high. This indicates that an a priori prediction of this model will not be promising. Only an integrated assessment of all predictive performance markers (bias, inaccuracy, goodness-of-fit plots, and forecasting performance) can provide evidence of a model’s relevance. Although we initially had four possibly models for MERO, none ended up being appropriate for clinical use under the herein defined criteria.
Our population consisted of patients with severe infections, which corresponded to the patients diagnosed with severe sepsis or septic shock that all studies recruited. However, as bias and inaccuracy were either near or out the threshold, there may be other factors that vary between both populations that have to be considered, such as the severity of the illness, comedications, and the underlying pathologies common in each group. Moreover, the high values of the between-subject variability and residual variability in all the included publications also indicate how different each population of patients is from the other. This could be due to several factors, including the patient population that the PK models were derived from and the methods used in the PK modeling. Even though commonly used covariates such as creatinine clearance and body weight were incorporated in most of the models, a significant variability still remains, which would explain the difficulty to validate its accuracy using a given population. The different methods used to estimate covariates such as the serum creatinine can affect the transferability of the model to a clinical setting [6]. Amongst the four evaluated models, certain covariates are more commonly used than others. Renal clearance plays a significant role in the predicted levels of MERO, as the estimated creatinine clearance was incorporated as a covariate in the PK equation for CL in all evaluated models except for Mattioli et al. [19]. As MERO is mainly eliminated by renal route, changes in renal functions would affect the concentrations of the beta-lactam antibiotics [33]. Therefore, evaluating a patient’s kidney function is primordial to the validity of the model. Estimation of renal function in critically ill patients remains an issue; it is clear that creatinine level is an imperfect tool in assessing kidney function. All studies evaluated creatinine clearance using Cockcroft-Gault formula. But, as previously described, the Cockcroft-Gault formula is highly inadequate in intensive care unit [34] but remains largely used in clinical practice and could be explained the large observed variability. Future investigations should evaluated the Cystatin C as marker of renal function to reduce this variability [34]. Body weight was also used as a covariate by Delattre et al. [17] and Li et al. [18]. As described by Marsot et al., the body weight has an effect on dose optimization, which could be explained by its relationship with the volume of distribution [35]. The value of the latter can lower or increase the concentrations of the antibiotics. Mattioli et al. [19] included albumin level in their PK equation for the volume of distribution. Indeed, hypoalbuminaemia is a common but frequently neglected condition in intensive care unit [35]. The incidence is reportedly as high as 40–50% [36]. With decreasing albumin concentrations, an increase in the unbound fraction drugs can occur [37]. However, the inclusion of that covariate is not common in other studied models. Additionally, the patients from the external validation dataset were administered meropenem by continuous infusion (CI), which differed from the mode of administration of some of the selected studies (Table 1). However, Roberts et al. [20] compared the mode of administration between patients that received meropenem either intermittent bolus administration or an equal dose administered by CI. They concluded that while the administration by CI maintained higher concentrations in subcutaneous tissue and plasma, it did not have a major impact on the final data. The discrepancy between mode of administration of the population groups could still be a potential weakness of the study.
The design of the study also can explain a part of the observed variability [38]. Indeed, the number of patients from whom the model is derived also plays a significant role in assessing the model’s robustness [38]. In this study, the number of patients and the number of samples differed greatly between each model and the external validation dataset. Roberts et al. [20] only used ten patients to develop their population PK model, which could suggest a decreased reliability due to the small numbers of patients. Compared with Roberts et al. [20], Delattre et al. [17] collected data from 88 ICU patients treated with amikacin but only 19 of these were coadministered with MERO. Only internal evaluations such as bootstraps and/or visual predictive checks (VPC) were done to assess the chosen models. Moreover, half of the selected models [17, 18] were developed based on routine clinical data, possibly leading to incorrect documentation or a lack of information. A study by Alihodzic et al. [39] demonstrated that erroneous records could have a considerable impact on the precision and accuracy of the estimated PK parameters in population PK modeling.
Recently, Dhaese et al. [31] also realized an external evaluation in which eight population PK models of MERO were compared. We found similar results, concluding that there is a large variability in the predictive performance of the models when performing an external validation using an independent dataset of ICU patients. Moreover, Dhaese et al. [31] suggested that an external evaluation should also be based on its tendency to under- or over-predict concentrations and that the consequences of under-dosing of MERO are probably more important than the consequences of overdosing. However, several recent studies showed that neurotoxicity exists with beta-lactams [40, 41]. High such as low MERO concentrations must be avoided. There is a need to develop a population PK model for each cohort of patients, as every intensive care unit has a different population, which can affect the pharmacokinetics.
Some remarks need to be made regarding the design of this study. First, this study included therapeutic drug monitoring data, resulting in the limited number of blood samples per patient collected, and the data were collected from a clinical setting. Another limitation for this study was the low number of patients (n = 27) used for the external evaluation. This element is also considered to be a limitation in assessing the forecasting performance. Indeed, forecasting performance can also be a criterion for evaluation [16]. Furthermore, the fact that the exclusion criteria restricted the models chosen to those developed by a parametric approach might be considered a limitation.
Conclusion
In conclusion, the predictive performance of evaluated PK models in ICU patients showed a wide variability. External evaluation in the population of interest is a necessary step before defining new dose adaptation. This brings to light the difficulty to generalize model in ICU population and the need to develop one specific model to each population of critically ill patients.
References
Wiseman LR, Wagstaff AJ, Brogden RN, Bryson HM (1995) Meropenem. A review of its antibacterial activity, pharmacokinetic properties and clinical efficacy. Drugs. 50(1):73–101
Mouton JW, Dudley MN, Cars O, Derendorf H, Drusano GL (2005) Standardization of pharmacokinetic/pharmacodynamic (PK/PD) terminology for anti-infective drugs: an update. J Antimicrob Chemother 55(5):601–607
Guilhaumou R, Benaboud S, Bennis Y, Dahyot-Fizelier C, Dailly E, Gandia P et al (2019) Optimization of the treatment with beta-lactam antibiotics in critically ill patients-guidelines from the French Society of Pharmacology and Therapeutics (Société Française de Pharmacologie et Thérapeutique-SFPT) and the French Society of Anaesthesia and Intensive Care Medicine (Société Française d’Anesthésie et Réanimation-SFAR). Crit Care (London, England) 23(1):104
Chow KM, Hui AC, Szeto CC (2005) Neurotoxicity induced by beta-lactam antibiotics: from bench to bedside. Eur J Clin Microbiol Infect Dis 24(10):649–653
Mould DR, Upton RN (2013) Basic concepts in population modeling, simulation, and model-based drug development-part 2: introduction to pharmacokinetic modeling methods. CPT Pharmacometrics Syst Pharmacol 2(4):e38-e
Zhao W, Kaguelidou F, Biran V, Zhang D, Allegaert K, Capparelli EV, Holford N, Kimura T, Lo YL, Peris JE, Thomson A, van den Anker JN, Fakhoury M, Jacqz-Aigrain E (2013) External evaluation of population pharmacokinetic models of vancomycin in neonates: the transferability of published models to different clinical settings. Br J Clin Pharmacol 75(4):1068–1080
Lee JY, Garnett CE, Gobburu JV, Bhattaram VA, Brar S, Earp JC et al (2011) Impact of pharmacometric analyses on new drug approval and labelling decisions: a review of 198 submissions between 2000 and 2008. Clin Pharmacokinet 50(10):627–635
Varghese JM, Roberts JA, Lipman J (2010) Pharmacokinetics and pharmacodynamics in critically ill patients. Curr Opin Anaesthesiol 23(4):472–478
Verdier MC, Tribut O, Tattevin P, Le Tulzo Y, Michelet C, Bentue-Ferrer D (2011) Simultaneous determination of 12 beta-lactam antibiotics in human plasma by high-performance liquid chromatography with UV detection: application to therapeutic drug monitoring. Antimicrob Agents Chemother 55(10):4873–4879
EMA. guideline-bioanalytical-method-validation_en.pdf [Internet]. Available from: https://wwwemaeuropaeu/en/documents/scientific-guideline/guideline-bioanalytical-method-validation_enpdf. [cited 2019 Apr 24].
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6(7):e1000097
Guang W, Baraldo M, Furlanut M (1995) Calculating percentage prediction error: a user’s note. Pharmacol Res 32(4):241–248
Varvel JR, Donoho DL, Shafer SL (1992) Measuring the predictive performance of computer-controlled infusion pumps. J Pharmacokinet Biopharm 20(1):63–94
Hara M, Masui K, Eleveld DJ, Struys M, Uchida O (2017) Predictive performance of eleven pharmacokinetic models for propofol infusion in children for long-duration anaesthesia. Br J Anaesth 118(3):415–423
Brendel K, Comets E, Laffont C, Laveille C, Mentre F (2006) Metrics for external model evaluation with an application to the population pharmacokinetics of gliclazide. Pharm Res 23(9):2036–2049
Broeker A, Nardecchia M, Klinker KP, Derendorf H, Day RO, Marriott DJ et al (2019) Towards precision dosing of vancomycin: a systematic evaluation of pharmacometsric models for Bayesian forecasting. Clin Microbiol Infect 25(10):1286.e1–1286.e7
Delattre IK, Musuamba FT, Jacqmin P, Taccone FS, Laterre P-F, Verbeeck RK, Jacobs F, Wallemacq P (2012) Population pharmacokinetics of four β-lactams in critically ill septic patients comedicated with amikacin. Clin Biochem 45(10):780–786
Li C, Kuti JL, Nightingale CH, Nicolau DP (2006) Population pharmacokinetic analysis and dosing regimen optimization of meropenem in adult patients. J Clin Pharmacol 46(10):1171–1178
Mattioli F, Fucile C, Del Bono V, Marini V, Parisini A, Molin A et al (2016) Population pharmacokinetics and probability of target attainment of meropenem in critically ill patients. Eur J Clin Pharmacol 72(7):839–848
Roberts JA, Kirkpatrick CM, Roberts MS, Robertson TA, Dalley AJ, Lipman J (2009) Meropenem dosing in critically ill patients with sepsis and without renal dysfunction: intermittent bolus versus continuous administration? Monte Carlo dosing simulations and subcutaneous tissue distribution. J Antimicrob Chemother 64(1):142–150
Delattre IK, Musuamba FT, Nyberg J, Taccone FS, Laterre P-F, Verbeeck RK, Jacobs F, Wallemacq PE (2010) Population pharmacokinetic modeling and optimal sampling strategy for Bayesian estimation of amikacin exposure in critically ill septic patients. Ther Drug Monit 32(6):749–756
Jaruratanasirikul S, Thengyai S, Wongpoowarak W, Wattanavijitkul T, Tangkitwanitjaroen K, Sukarnjanaset W, Jullangkoon M, Samaeng M (2015) Population pharmacokinetics and Monte Carlo dosing simulations of meropenem during the early phase of severe sepsis and septic shock in critically ill patients in intensive care units. Antimicrob Agents Chemother 59(6):2995–3001
Kees MG, Minichmayr IK, Moritz S, Beck S, Wicha SG, Kees F, Kloft C, Steinke T (2016) Population pharmacokinetics of meropenem during continuous infusion in surgical ICU patients. J Clin Pharmacol 56(3):307–315
Minichmayr IK, Roberts JA, Frey OR, Roehr AC, Kloft C, Brinkmann A (2018) Development of a dosing nomogram for continuous-infusion meropenem in critically ill patients based on a validated population pharmacokinetic model. J Antimicrob Chemother 73(5):1330–1339
Lodise TP, Nau R, Kinzig M, Drusano GL, Jones RN, Sorgel F (2007) Pharmacodynamics of ceftazidime and meropenem in cerebrospinal fluid: results of population pharmacokinetic modelling and Monte Carlo simulation. J Antimicrob Chemother 60(5):1038–1044
Crandon JL, Ariano RE, Zelenitsky SA, Nicasio AM, Kuti JL, Nicolau DP (2010) Optimization of meropenem dosage in the critically ill population based on renal function. Intensive Care Med 37(4):632–638
Lodise TP, Sorgel F, Melnick D, Mason B, Kinzig M, Drusano GL (2011) Penetration of meropenem into epithelial lining fluid of patients with ventilator-associated pneumonia. Antimicrob Agents Chemother 55(4):1606–1610
Mathew SK, Mathew BS, Neely MN, Naik GS, Prabha R, Jacob GG, K S, Fleming DH (2016) A nonparametric pharmacokinetic approach to determine the optimal dosing regimen for 30-minute and 3-hour meropenem infusions in critically ill patients. Ther Drug Monit 38(5):593–599
Cojutti P, Sartor A, Righi E, Scarparo C, Bassetti M, Pea F (2017) Population pharmacokinetics of high-dose continuous-infusion meropenem and considerations for use in the treatment of infections due to KPC-producing <span class=“named-content genus-species” id=“named-content-1”>Klebsiella pneumoniae</span>. Antimicrob Agents Chemother 61(10):e00794–e00717
Sjovall F, Alobaid AS, Wallis SC, Perner A, Lipman J, Roberts JA (2018) Maximally effective dosing regimens of meropenem in patients with septic shock. J Antimicrob Chemother 73(1):191–198
Dhaese SAM, Farkas A, Colin P, Lipman J, Stove V, Verstraete AG, Roberts JA, de Waele JJ (2019) Population pharmacokinetics and evaluation of the predictive performance of pharmacokinetic models in critically ill patients receiving continuous infusion meropenem: a comparison of eight pharmacokinetic models. J Antimicrob Chemother 74(2):432–441
Brendel K, Dartois C, Comets E, Lemenuel-Diot A, Laveille C, Tranchand B et al (2007) Are population pharmacokinetic and/or pharmacodynamic models adequately evaluated? A survey of the literature from 2002 to 2004. Clin Pharmacokinet 46(3):221–234
Roberts JA, Abdul-Aziz MH, Lipman J, Mouton JW, Vinks AA, Felton TW, Hope WW, Farkas A, Neely MN, Schentag JJ, Drusano G, Frey OR, Theuretzbacher U, Kuti JL, International Society of Anti-Infective Pharmacology and the Pharmacokinetics and Pharmacodynamics Study Group of the European Society of Clinical Microbiology and Infectious Diseases (2014) Individualised antibiotic dosing for patients who are critically ill: challenges and potential solutions. Lancet Infect Dis 14(6):498–509
Sunder S, Jayaraman R, Mahapatra HS, Sathi S, Ramanan V, Kanchi P, Gupta A, Daksh S, Ram P (2014) Estimation of renal function in the intensive care unit: the covert concepts brought to light. J Intensive Care 2(1):31
Marsot A, Guilhaumou R, Riff C, Blin O (2017) Amikacin in critically ill patients: a review of population pharmacokinetic studies. Clin Pharmacokinet 56(2):127–138
Finfer S, Bellomo R, Boyce N, French J, Myburgh J, Norton R (2004) A comparison of albumin and saline for fluid resuscitation in the intensive care unit. N Engl J Med 350(22):2247–2256
Roberts JA, Pea F, Lipman J (2013) The clinical relevance of plasma protein binding changes. Clin Pharmacokinet 52(1):1–8
Duffull S, Waterhouse T, Eccleston J (2005) Some considerations on the design of population pharmacokinetic studies. J Pharmacokinet Pharmacodyn 32(3-4):441–457
Alihodzic D, Broeker A, Baehr M, Kluge S, Langebrake C, Wicha SG (2020) Impact of inaccurate documentation of sampling and infusion time in model-informed precision dosing. Front Pharmacol 11:172. https://doi.org/10.3389/fphar.2020.00172
Grahl JJ, Stollings JL, Rakhit S, Person AK, Wang L, Thompson JL, Pandharipande PP, Ely EW, Patel MB (2018) Antimicrobial exposure and the risk of delirium in critically ill patients. Crit Care 22(1):337
Bhattacharyya S, Darby RR, Raibagkar P, Gonzalez Castro LN, Berkowitz AL (2016) Antibiotic-associated encephalopathy. Neurology. 86(10):963–971
Funding
Internal funding supported this study.
Author information
Authors and Affiliations
Contributions
YW and AM analyzed the data. AM performed population pharmacokinetic analysis. LV included patient. YW and AM wrote the manuscript. OB validated the manuscript. AM and RG conceived and designed the study. AM supervised the work. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 67.1 kb)
Rights and permissions
About this article
Cite this article
Wang, Y., Guilhaumou, R., Blin, O. et al. External evaluation of population pharmacokinetic models for continuous administration of meropenem in critically ill adult patients. Eur J Clin Pharmacol 76, 1281–1289 (2020). https://doi.org/10.1007/s00228-020-02922-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-020-02922-z