Abstract
Purpose
The study aimed to measure the percentage of preventable adverse drug reactions that lead to the hospitalization (PADRAd) and to explore the heterogeneity in its estimation through subgroup analysis of study characteristics.
Methods
Two investigators independently searched in electronic databases and related bibliography for prospective studies involving PADRAd. We excluded studies investigating medication errors and spontaneous and retrospective reporting. The primary outcome was PADRAd percentage. To explore the heterogeneity, we performed subgroup analysis based on study region, wards, age groups, adverse drug reaction (ADR) definitions, preventability assessment, ADR identification methods, study duration and sample size. We explored fatal PADRAd and causative drugs as a secondary outcome. We used the generic inverse variance method with random effect model to compute meta-analytic summary.
Results
Of the 68 full-text articles assessed, we included 22 studies. The mean PADRAd percentage was 45.11 % (95 % CI = 33.06–57.15; I 2 = 99 %). Studies including elderly (63.31 %) and all age groups (49.03 %) showed higher percentages than paediatric population (16.40 %). Studies examining all hospital populations showed higher percentages than specific wards. We observed high percentages in studies using Edwards and Aronson as an ADR definition and Hallas et al. as a preventability assessment tool. After age group adjustment, ADR detection methods did not show significant difference. The fatal PADRAd percentage was 1.58 % (95 % CI = −0.60 to 3.76; I 2 = 47 %). Paediatric and elderly studies showed a different causative drug pattern.
Conclusion
Variation in PADRAd across the studies can be explained by difference in study populations and data collection methods. Extrapolation of preventable reactions should be carried out considering all these factors with caution.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Drug-related problems are important public health problems. They include adverse drug reactions (ADRs), inappropriate drug selection, drug use without indications, non-compliance, drug interactions and use of subtherapeutic and supra-therapeutic dosage [1]. ADRs are an important cause of hospital admissions [2, 3]. They account for almost two thirds of all drug-related hospital admissions and emergency department visits [4, 5]. Beijer et al. estimated 4.9 % (range = 0.2–41.3 %) of hospital admissions were due to ADRs. The authors observed about 30 % ADRs as preventable [6]. Leendertse et al. observed low weighted mean prevalence (0.46 %) for drug-related hospitalization. The authors observed variations in prevalence based on study setting, population and data collection methods. So, many characteristics of the study can influence the ADR prevalence [7].
In an earlier systematic review, Hakkarainen et al. observed 52 and 45 % of ADRs were preventable among outpatients and inpatients, respectively [8]. ADRs resulting in hospital admission (ADRAd) generally represent more severe reactions than ADRs occurring during hospitalization (ADRIn) and are one of the leading causes of death in the population [4, 9, 10]. Preventable ADRs cause unnecessary loss of health, quality of life and money [6]. Earlier systematic reviews of preventable ADRs considered both prospective and retrospective studies and did not investigate the heterogeneity for the various study characteristics like population, data collection methods, etc. [6, 8]. So, we conducted this updated systematic review to measure the percentage of preventable ADRAd (PADRAd) among the prospective studies and to explore the heterogeneity in its estimation based on study characteristics.
Materials and methods
Search strategy
Two investigators independently searched in electronic databases (PubMed, Google Scholar and Cochrane Database of Systematic Reviews). PubMed searches only journals, while Google Scholar searches all internet resources [11]. PubMed database searches abstract, while Google Scholar searches the full text to retrieve the articles. So, we choose different search terms for PubMed and Google Scholar. We used synonyms and truncation in PubMed search only. The key terms of PubMed search were ‘adverse drug reaction’ OR ‘adverse drug event’ OR ‘adverse reaction’ OR ‘adverse event’ OR ‘drug related problem’ OR ‘drug induced problem’ AND (‘avoid*’ OR ‘prevent*’) AND hospital*. The key terms of Google Scholar search were ‘Adverse drug reaction’ AND ‘Preventable’ AND ‘Hospitalization’. We also searched the bibliographies of relevant articles. We only searched English language articles. We searched the studies published between January 2000 and June 2015 for review (last search on 18 June 2015).
We assessed title, abstract and if necessary full-text articles from retrieving references according to selection criteria. Any disagreement between the two investigators was discussed and resolved by consensus or by a third investigator.
Selection criteria
We included all prospective studies related to ADRs that provide sufficient data to calculate PADRAd. All studies should have identified ADRAd through WHO or “Edwards and Aronson” [12] or other similar definitions and specified preventability assessment tools. We excluded studies of retrospective or spontaneous reporting design; those did not differentiate ADRs from adverse drug events or medication errors; considered all augmented reactions as preventable without applying specified measurement tools; and focused on particular reactions or specific drug exposure only. Detailed selection criteria are in Supplementary file 1.
Data extraction
We extracted the following information from each study in Excel sheet: study year, its duration, number of the study centre, level of care provided by a hospital, study specific departments, region, country, demographic data, ADR definition used, total number of patients studied, number of patients with ADRAd, number of preventable ADRAd, fatal reactions, causative drugs, causality, preventability and severity. Two investigators cross checked the accuracy of data entry. We assessed the methodological quality of the included studies as per Smyth et al. considering study design, methods for identifying ADRs, causality, preventability and severity assessment [13].
Primary outcome assessment
The primary outcome was to assess the percentage of PADRAd. We used an Excel sheet to estimate PADRAd percentage and its standard error (SE). Based on an expectation of high heterogeneity from earlier ADR-related studies, we used generic inverse variance method with random effect model to calculate mean PADRAd percentage and its 95 % confidence interval [14]. There is no ideal approach in the presence of high heterogeneity. We selected random effect model which incorporates heterogeneity in overall analysis of effects and gives a more conservative estimate with wider confidence intervals. We assessed the heterogeneity for the percentage of PADRAd using I 2 test—25 % as low, 50 % moderate and 75 % as high [15]. We observed high heterogeneity for the percentage of PADRAd (I 2 = 99 %).
Subgroup analysis of primary outcome assessment
To explore the heterogeneity, we did the subgroup analysis of primary outcome based on the study characteristics like study population (region, wards, age groups), data collection methods (ADR definition, preventability assessment tools, ADR identification method), study duration (≤3, 4–11 and ≥12 months) and sample size (<1000 and ≥1000 patients). Study region was based on continents. Study ward includes admission wards. Age group includes paediatric, elderly and all age groups. We considered Karch and Lasagna definition similar to WHO definition [16]. The ADR identification method includes a medical record review or combined approach (interview plus medical record review). Detailed study characteristics for subgroup analysis are in Supplementary file 2.
Based on the results of subgroup analysis, we again explored the percentage of PADRAd in ‘all age group studies’ for the different study characteristics.
Secondary outcome assessments
Prevalence of PADRAd and fatal ADRAd
We used number of patients with PADRAd as numerator and total number of hospitalized patients as denominator to calculate prevalence from each study. We used an Excel sheet to estimate the PADRAd prevalence, percentage of fatal ADRAd, preventable fatal ADRAd and their SE through Excel sheet. We used generic inverse variance method with random effect model to compute meta-analytic summary and its 95 % confidence interval. We assessed the heterogeneity using I 2 test.
Suspected drugs
We extracted the causative drugs of PADRAd and summarized using absolute numbers of cases and percentage. We labelled the causative drugs as per anatomical therapeutic chemical classification system [17]. We also presented the data of causative drugs from paediatric and elderly studies separately.
The meta-analysis was performed through ‘Review manager software version 5.0’.
Results
Literature search
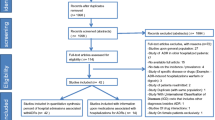
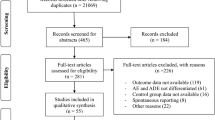
As shown in Fig. 1, we evaluated a total of 68 full-text articles as per selection criteria and included 22 studies.
Characteristics and methodological quality of the included studies
Table 1 shows the characteristics of all included studies [18–39]. Two studies included both ADRAd and ADRIn data [24, 25]. Chan et al. studied adverse drug event. Easton et al. and Al-Arifi et al. analysed all drug-related problems. We only included ADRAd data of these studies. Two studies included ADRAd and ADRs concurrently present during admission [23, 24]. We only extracted ADRAd data. Fourteen studies identified ADRs using WHO definition. Doshi et al. communicated use of WHO definition on contact of our previous systematic review [25]. One study used Karch and Lasagna ADR definition [20]. Six studies used Edwards and Aronson ADR definition. The studies detected ADRs using interview (one study), medical record review (nine studies) and combined approach (ten studies). Two studies did not state the methods used to detect ADRs [25, 31]. Total 21 studies used the causality assessment. The preventability assessment tools used were Schumock and Thornton (eight studies), Hallas et al. (nine studies), French ADR preventability scale (two studies), Nelson and Talbert (one study), Nebeker et al. (one study) and Livio et al. (one study). Detailed study characteristics of included studies according to age are presented in Supplementary file 3.
Prevalence of PADRAd
Among the 15 studies [18–20, 24, 26–28, 31, 32, 34–39], 3948 out of 95,278 patients were admitted due to PADRAd. The number of patients hospitalized due to PADR Ad varied from 2 to 1852 in the included studies. The PADRAd prevalence varied from 0.10 to 6.41 %. The mean PADRAd prevalence was 2.45 % (95 % CI = 1.77–3.12 %; I 2 = 99 %) (Supplementary Fig. 1).
Percentage of PADRAd
In 22 included studies, a total 4797 ADRAd observed in 4462 patients and 1886 were reported as preventable. The PADRAd percentage varied from 4.26 to 83.33 %. The mean percentage of PADRAd was 45.11 % (95 % CI = 33.06–57.15; I 2 = 99 %) (Fig. 2).
Subgroup analysis of the percentage of PADRAd based on the study characteristics
Based on the findings of subgroup analysis of study characteristics, we identified age group as a most important heterogeneity modifier. As shown in Fig. 3, age group showed important influence on PADRAd percentage ranging from 16.40 (paediatric population) to 63.31 (elderly population). So, we further compared the study characteristics by adjusting the age groups.
Age groups
As shown in Table 2, paediatric age group studies showed a low PADRAd percentage (16.40 %) and high heterogeneity (I 2 = 87 %). Two paediatric studies used Hallas et al. preventability tool and Edwards and Aronson definition. By combining these studies, we observed relatively high percentage and reduction in the heterogeneity [24.67 % (95 % CI = 15.38–34.14), I 2 = 37 %]. Other two paediatric studies used Schumock and Thornton algorithm and WHO definition. By combining these studies, we observed relatively low percentage and reduction in the heterogeneity [5.76 % (95 % CI = 0.10–11.43), I 2 = 0 %] (Supplementary Fig. 2).
Elderly studies showed a high PADRAd percentage (63.31 %) and heterogeneity (I 2 = 91 %). Both elderly studies used Hallas et al. preventability tool. We could not explore it further due to the presence of only two studies in elderly group.
All age group studies showed high PADRAd percentage (49.03 %) and heterogeneity (I 2 = 96 %). As shown in Table 2, we explored all age group studies for each study characteristics and described its finding along with each characteristic description.
Study wards
Whole hospital studies (61.65 %) showed a high PADRAd percentage followed by emergency department studies (42.45 %) and internal medicine (40.24 %). Like age group, paediatric ward studies (12.46 %) showed a low PADRAd percentage.
Adjustment for age groups
All age group studies showed similar patterns in percentage of PADRAd. Internal medicine studies showed low heterogeneity (I 2 = 0 %). Their other similar characteristics were WHO definition, all age groups and no study of less than 3 months duration. We also observed a reduction in heterogeneity in emergency studies (I 2 = 42 %). Their other similar characteristic was WHO definition (Fig. 4).
Study regions
European (44.37 %) and Asian studies (45.38 %) showed comparable percentages of PADRAd. Asian studies showed low heterogeneity (I 2 = 0 %). Their other common characteristic was WHO definition. However, Australian studies showed lower prevalence (29.34 %) and high heterogeneity (I 2 = 94 %). Both Australian studies used the WHO definition.
Adjustment for age groups
There is no effect of adjustment of age group in European studies on PADRAd (Supplementary Fig. 3).
ADR definition
We observed a slightly high PADRAd percentage in studies using Edwards and Aronson (51.66 %) than WHO (42.32 %) definition. This trend continues in all age group studies (60.13 vs. 46.13 %; I 2 ≥ 86 %) (Supplementary Fig. 4).
Preventability assessment tools
Hallas et al. (54.65 %) showed a high PADRAd percentage followed by French preventability scale (42.17 %) and Schumock and Thornton algorithm (35.25 %).
Adjustment for age groups
We observed a high percentage in Hallas et al. (65.21 %) group. All three studies of Hallas et al. group were of the European continent. Two of them belonged to whole hospital setup and two used WHO definition. Both Schumock and Thornton algorithm (42.28 %) and French preventability scale (42.17 %) showed comparable percentage. We also observed moderate heterogeneity in Schumock and Thornton algorithm group. Their other similar characteristics were European continent, WHO definition and study duration >12 months. Similar characteristics of French preventability scale studies were European continent, WHO definition, medical record review and study duration 4–11 months (Fig. 5).
ADR identification method
We observed comparable PADRAd percentage between studies using medical record review and combined approach (interview plus medical record review). We observed similar trend in percentage in all age group studies.
However, we observed significant reduction in heterogeneity in studies using medical record review (I 2 = 18 %). Their similar characteristics were European continent and WHO definition (Supplementary Fig. 5).
Findings of study duration and sample size along with their inference are presented in a Supplementary file 4.
Fatal reactions
Ten out of 21 included studies mentioned the number of fatal reactions [21–23, 30–32, 34, 36, 37, 39], 4 out of 21 studies mentioned zero fatal reactions [25, 28, 29, 33] and 4 out of 8 studies mentioned data about preventable fatal reactions [21, 22, 31, 37]. The meta-analytic summary percentage of fatal reactions was 2.69 % [(95 % CI = 1.32–4.06 %), I 2 = 82 %]. The percentage of preventable fatal reactions was 1.58 % (95 % CI = –0.60 to 3.76) with moderate heterogeneity (I 2 = 47 %) (Supplementary Fig. 6).
Suspected drugs
Nine studies mentioned the drugs causing PADRAd [27, 29–31, 33, 35, 37–39]. We observed cardiovascular drugs in one fourth of cases. Other major incriminated groups were musculoskeletal (16.13 %) and nervous system (16.89 %). Commonly implicated drugs were non-steroidal anti-inflammatory drugs (NSAIDs) (16.13 %), antithrombotics (12.14 %), cardiac glycosides (9.87 %) and diuretics (7.97 %). Paediatric and elderly studies showed a different causative drug pattern. Nervous system (25 %) and alimentary tract and metabolism (25 %) were major causative class in paediatric studies, while musculoskeletal (44.87 %) and cardiovascular systems (28.21 %) were major class in elderly [Table 3].
Discussion
In the present study, we analysed the PADRAd percentage based on prospective ADR-related studies of the last one and half decade. We observed PADRAd as an important cause of morbidity and mortality. We observed a wide range of preventability (4.26–83.33 %) among the included studies. This variation can be explained by study population and data collection methods.
We observed higher (45.11 %) PADRAd percentage than Beijer et al. (28.1 %) and Goettler et al. (31 %) [6, 40]. Both systematic reviews included retrospective studies, did not specify the preventability assessment tools in included studies and were based on studies mainly conducted before 2000. Their primary objective was not the preventable ADRAd. We observed low PADRAd percentage than Hakkarainen et al. (52 %) [8]. One possible reason could be exclusion of paediatric population in earlier systematic review. As compared to Hakkarainen et al., we restricted our search to last one and half decade and did not include retrospective studies [8]. We also observed low PADRAd percentage than Winterstein et al. (59 %) [41]. The earlier systematic review included all undesirable events related to drug therapy to assess preventability and was based on studies conducted before 2000. We observed high PADRAd prevalence than Hakkarainen et al. (2.0 %) [8] and lower than Howard et al. (3.7 %) and Winterstein et al. (4.3 %) [41, 42]. This difference in prevalence can be explained by the difference in study methodology. None of these studies explored the heterogeneity in PADRAd percentage estimation through subgroup analysis of study characteristics.
We observed lower percentage of PADRAd in paediatric age groups. Even after adjusting for ADR definition and preventability assessment methods, this percentage remains low as compared to elderly and all age group studies. The earlier systematic review suggested a wide range of preventable ADRs (7–98 %) in paediatric population [13]. This could be due to the inclusion of all setting studies (community as well as inpatient) and all drug-related problems in assessing preventability by Smyth et al. [13]. Earlier systematic reviews on ADRAd observed low prevalence of ADRs in paediatric age group as compared to elderly and all age group studies [6, 7, 43]. Leendertse et al. explained low prevalence with the use of the few drugs in paediatric admission studies [7]. This could also be the possible reason for the low percentage of PADRAd. Another reason could be a different drug utilization pattern that leads to different classes of drugs causing PADRAd in paediatrics. In our study, we observed less or no PADRAd due to cardiovascular, antithrombotic and NSAIDs in paediatric than other age groups. Finding of elderly subgroup is in line with earlier systematic reviews [6, 41]. Beijer et al. observed high rate of PADRAd in elderly than non-elderly population (87.9 vs. 24.0 %) [6]. Winterstein et al. observed high PADRAd prevalence in age group >70 than ≤70 years (7.6 vs. 3.9 %) [41]. The elderly are more prone to develop ADRs related to use of inappropriate drugs and polypharmacy [44, 45], which could result into a high percentage of PADRAd.
With respect to study wards, we observed a high percentage of PADRAd in studies including entire hospital populations instead of subpopulations. This suggests PADRAd as an important cause of morbidity in the hospitalized patients. We also observed relatively low heterogeneity in all age group studies adjusting for specific subpopulations (internal medicine and emergency ward). High heterogeneity in whole hospital setting could be due to mixture of study populations.
Leendertse et al. observed smaller ADRAd prevalence in European studies than North American and Australian studies [7]. Miguel et al. observed lower ADRIn prevalence in Asian studies than European and American studies [46]. We also observed low rate of PADRAd in Australian studies without age group adjustment. This reinforces the importance of other study characteristics in the ADR assessment. We did not observe any effect of age group adjustment alone on European studies in terms of percentage and heterogeneity. We could not further evaluate the impact of different geographical locations on PADRAd percentage due to availability of only one Asian study in all age group setting.
We observed slightly high PADRAd percentage with studies using ‘Edwards and Aronson’ than ‘WHO definition’ for the ADR detection. The ‘Edwards and Aaronson’ uses ‘medicinal product’ as a causative agent and considers ADRs only to those events which predict hazard from future administration and warrants prevention or specific treatment or alteration of the dosage regimen or product withdrawal. The WHO definition considers ADR also to the minor reactions which do not have any consequences [12]. However, impact of ADR definition needs to be interpreted cautiously due to the presence of high heterogeneity.
PADRAd percentages and heterogeneity could also vary due to its assessment tool. We observed moderate to very high heterogeneity for the different methods. This was expected based on an earlier systematic review suggesting lack of valid and reliable method to assess preventability [47]. We observed higher PADRAd percentage as well as heterogeneity with ‘Hallas et al.’ than other methods. Unlike other methods, ‘Hallas et al.’ is not based on a fixed set of questionnaires and allows the investigator to label all events as preventable which are inconsistent with present-day knowledge of good medical practice, was clearly unrealistic, taking the known circumstances into account or could have been avoided by an effort exceeding the obligatory demands [48]. The reason for high heterogeneity of ‘Hallas et al.’ tool could be due to a mixture of different ADR definition studies. French preventability assessment method is an algorithm and assigns scores based on knowledge about the drug and its possible role, patient-related risk factors and conditions of prescription. Olivier et al. observed poor agreement among experts due to difficulty in terms of a clear understanding of the items [49]. This could be the reason for the moderate to high heterogeneity despite of having many similar characteristics in this group. We observed moderate heterogeneity among studies using Schumock and Thornton algorithm. Unlike the other two methods, this algorithm is more explicit and defines the preventability based on the history of previous allergy reactions, inappropriate drug selection according to diagnosis and patient characteristics, toxic serum drug concentration, lack of the required therapeutic drug monitoring or other necessary laboratory tests, known ADR treatment, drug interactions, poor compliance and lack of preventive measures [50].
In a paediatric population, we observed the impact of data collection methods on percentage and heterogeneity. Studies using Hallas et al. preventability tool and Edwards and Aronson definition yield high PADRAd percentage than those using Schumock and Thornton algorithm and WHO definition. Both paediatric subpopulations also showed reduction in heterogeneity. However, we should interpret cautiously due to the presence of a small number of studies for this comparison and should be considered as an exploratory finding for the future studies.
We observed no difference of ADR detection methods (combined approach or medical record review alone) on PADRAd percentage after the age group adjustment. There is little possibility of over- or underestimation due to ADR detection methods. This suggests the medical record review is an important tool to detect ADRs [7, 51]. Finding of low heterogeneity among medical record review group could be due to the similar ADR definition and same geographical location giving relatively homogenous setting.
The literature suggests ADR as an important cause of death in hospitalized patients and in general populations [52, 53]. We observed 2.69 % of reactions as fatal. Our analysis does not include ADRs leading to death outside the hospital. There is a possible window of ADR severity. So, ADRAd in this study represents ADRs are those severe enough to cause admission but not severe enough to cause death outside the hospital. There is a possibility of underestimation in the measuring of fatal reaction prevalence and its percentage. One population-based study of deceased subjects reported that one fourth of fatal ADRs are preventable [54]. One fatal ADRAd-based study in internal medicine reported one fifth of reactions as preventable [55]. We observed more than half of fatal ADRAd as preventable which suggests ADR as an important preventable cause of mortality in the hospitalized patients.
NSAIDs, antithrombotic drugs, cardiac glycosides and diuretics accounted for almost 50 % incriminated drugs in PADRAd cases. Earlier systematic review of preventable drug-related hospital admissions observed antithrombotic, diuretics and NSAIDs accounting for 50 % of incriminated drugs [42]. This could be due to difference in drug utilization pattern over a time. All except one study providing data about suspected drugs in our study are published after 2005. The earlier systematic review was mainly based on studies published before 2005. Drug class that requires attention for PADRAd in paediatrics are analgesics, psycholeptics, antidiabetic drugs, antinauseants, NSAIDs and corticosteroids. Interventions on NSAIDs, cardiac glycosides, antithrombotic and antihypertensive agents could reduce PADRAd in elderly patients. However, findings on drugs classes have to be weighed against their potential benefits.
The strengths of this study are its selection criteria and thorough literature search for more recent studies. We only focused on ADRs that lead to the hospitalization and did not include the ADRs that concurrently present during the admission. We have analysed only prospective studies and excluded the spontaneous reporting and retrospective studies. We have included only ADR data excluding adverse drug events and/or drug-related problems. We did a subgroup analysis to identify the factors associated with the heterogeneity and the impact of various study characteristics like populations and data collection methods on proportion of PADRAd. We could find causative drugs for the preventable reactions and suggest their pattern as per the age groups.
This systematic review has several limitations also. We acknowledge that some may consider exclusion of literature published before the year 2000 as arbitrary. Our decision was based on most of the earlier systematic reviews on this subject which were before this period. We searched only freely accessed databases PubMed and Google Scholar and could not search EMBASE. This systematic review is based on observational studies which usually had inherent biases and differences in study design. So, we explored our primary outcome to understand and quantify heterogeneity sources across studies. We could only compare study characteristics by adjusting for the age groups only. We could not adjust the multiple characteristic of study population and data collection methods due to mixture of study characteristics in the included studies. So, we could not do an analysis of risk factors for the PADRAd and answer whether it is old age or the type of department or certain drug classes which result in the highest risk for a preventable ADR. This could have led to over- or underestimation in percentage of PADRAd. Moreover, subgroup analysis requires cautious interpretation in terms of small sample for each study characteristics. All included studies had preventability as a secondary objective. Future studies need to focus on these areas to suggest remedial measures for reducing the burden of PADRAd.
Conclusion
PADRAd is an important cause of morbidity and mortality in the hospitalized patients and burdens to the health care system. We identified age groups, study wards, ADR definition and preventability assessment tools as important heterogeneity modifiers. PADRAd percentages are higher in all age groups and elderly than paediatric studies. PADRAd percentages are higher in the whole hospital than specific ward setting. With respect to data collection methods, studies using ‘Hallas et al.’ preventability assessment tool and ‘Edwards and Aronson’ ADR definition are likely to give high PADRAd percentages. Paediatric and elderly studies showed a different causative drug pattern of PADRAd.
References
Nivya K, Sri S, Ragoo N, Ayaprakash B, Sonal SM (2015) Systemic review on drug related hospital admissions—a PubMed based search. Saudi Pharm J 23(1):1–8
Brvar M, Fokter N, Bunc M, Mozina M (2009) The frequency of adverse drug reaction related admissions according to method of detection, admission urgency and medical department specialty. BMC Clin Pharmacol 9:8
Davies EC, Green CF, Taylor S, Williamson PR, Mottram DR, Pirmohamed M (2009) Adverse drug reactions in hospital in-patients: a prospective analysis of 3695 patient-episodes. PLoS One 4(2):e4439
Lazarou J, Pomeranz BH, Corey PN (1998) Prevalence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. JAMA 279(15):1200–1205
Patel H, Bell D, Molokhia M, Srishanmuganathan J, Patel M, Car J et al (2007) Trends in hospital admissions for adverse drug reactions in England: analysis of national hospital episode statistics 1998–2005. BMC Clin Pharmacol 7:9
Beijer HJM, De Blaey CJ (2002) Hospitalisations caused by adverse drug reactions (ADR): a meta-analysis of observational studies. Pharm World Sci 24(2):46–54
Leendertse AJ, Visser D, Egberts AC, van den Bemt PM (2010) The relationship between study characteristics and the prevalence of medication-related hospitalizations: a literature review and novel analysis. Drug Saf 33(3):233–244
Hakkarainen KM, Hedna K, Petzold M, Hägg S (2012) Percentage of patients with preventable adverse drug reactions and preventability of adverse drug reactions—a meta-analysis. PLoS One 7(3):e33236
Fattinger K, Roos M, Vergères P, Holenstein C, Kind B, Masche U et al (2000) Epidemiology of drug exposure and adverse drug reactions in two Swiss departments of internal medicine. Br J Clin Pharmacol 49(2):158–167
Moore N, Lecointre D, Noblet C, Mabille M (1998) Frequency and cost of serious adverse drug reactions in a department of general medicine. Br J Clin Pharmacol 45(3):301–308
Shultz M (2007) Comparing test searches in PubMed and Google Scholar. J Med Libr Assoc 95(4):442–445
Edwards IR, Aronson JK (2000) Adverse drug reactions: definitions, diagnosis and management. Lancet 356(9237):1255–1259
Smyth RMD, Gargon E, Kirkham J, Cresswell L, Golder S, Smyth R et al (2012) Adverse drug reactions in children—a systematic review. PLoS One 7(3):e24061
Higgins JPT, Green S (2011). Cochrane handbook for systematic reviews of interventions version 5.1.0 [updated March 2011]. The Cochrane Collaboration.
Rücker G, Schwarzer G, Carpenter JR, Schumacher M (2008) Undue reliance on I(2) in assessing heterogeneity may mislead. BMC Med Res Methodol 8(1):79
Karch FE, Lasagna L (1975) Adverse drug reactions—a critical review. JAMA 234(12):1236–1241
ATC/DDD index 2016. WHO Collaborating Centre for Drug Statistics Methodology. WHO collaborating centre, Oslo, Norway. Available from: http://www.whocc.no/atc_ddd_index/ [Last accessed on: 20th November, 2016].
Ahern F, Sahm LJ, Lynch D, McCarthy S (2014) Determining the frequency and preventability of adverse drug reaction-related admissions to an Irish university hospital: a cross-sectional study. Emerg Med J 31(1):24–29
Alexopoulou A, Dourakis SP, Mantzoukis D, Pitsariotis T, Kandyli A, Deutsch M et al (2008) Adverse drug reactions as a cause of hospital admissions: a 6-month experience in a single centre in Greece. Eur J Intern Med 19(7):505–510
Al-Arifi M, Abu-Hashem H, Al-Meziny M, Said R, Aljadhey H (2014) Emergency department visits and admissions due to drug related problems at Riyadh military hospital (RMH), Saudi Arabia. Saudi Pharm J 22(1):17–25
Chan M, Nicklason F, Vial JH (2001) Adverse drug events as a cause of hospital admission in the elderly. Intern Med J 31(4):199–205
Davies EC, Green CF, Mottram DR, Rowe PH, Pirmohamed M (2010) Emergency re-admissions to hospital due to adverse drug reactions within 1 year of the index admission. Br J Clin Pharmacol 70(5):749–755
Dormann H, Criegee-Rieck M, Neubert A, Egger T, Geise A, Krebs S et al (2003) Lack of awareness of community-acquired adverse drug reactions upon hospital admission dimensions and consequences of a dilemma. Drug Saf 26(5):353–362
Dormann H, Neubert A, Criegee-Rieck M, Egger T, Radespiel-Troger M, Azaz-Livshits T et al (2004) Readmissions and adverse drug reactions in internal medicine: the economic impact. J Intern Med 255(6):653–663
Doshi MS, Patel PP, Shah SP, Dikshit RK (2012) Intensive monitoring of adverse drug reactions in hospitalized patients of two medical units at a tertiary care teaching hospital. J Pharmacol Pharmacother 3(4):308–313
Easton KL, Chapman CB, Brien JA (2004) Frequency and characteristics of hospital admissions associated with drug-related problems in paediatrics. Br J Clin Pharmacol 57(5):611–615
Franceschi M, Scarcelli C, Niro V, Seripa D, Pazienza AM, Pepe G et al (2008) Prevalence, clinical features and avoidability of adverse drug reactions as cause of admission to a geriatric unit a prospective study of 1756 patients. Drug Saf 31(6):545–556
Gallagher RM, Bird KA, Mason JR, Peak M, Williamson PR, Nunn AJ et al (2011) Adverse drug reactions causing admission to a paediatric hospital: a pilot study. J Clin Pharm Ther 36(2):194–199
Gallagher RM, Mason JR, Bird KA, Kirkham JJ, Peak M, Williamson PR et al (2012) Adverse drug reactions causing admission to a paediatric hospital. PLoS One 7(12):e50127
Grenouillet-Delacre M, Verdoux H, Moore N, Haramburu F, Miremont-Salamé G, Etienne G et al (2007) Life-threatening adverse drug reactions at admission to medical intensive care: a prospective study in a teaching hospital. Intensive Care Med 33(12):2150–2157
Hopf Y, Watson M, Williams D (2008) Adverse-drug-reaction related admissions to a hospital in Scotland. Pharm World Sci 30(6):854–862
Benard-Laribierea A, Miremont-Salamea G, Perault-Pochatc MC, Noizea P, Haramburua F (2015) EMIR Study Group on behalf of the French network of pharmacovigilance centres. Incidence of hospital admissions due to adverse drug reactions in France: the EMIR study. Fundam Clin Pharmacol 29(1):106–111
Olivier P, Boulbés O, Tubery M, Lauque D, Montastruc JL, Lapeyre-Mestre M (2002) Assessing the feasibility of using an adverse drug reaction preventability scale in clinical practice a study in a French emergency department. Drug Saf 25(14):1035–1044
Pirmohamed M, James S, Meakin S, Green C, Scott AK, Walley TJ et al (2004) Adverse drug reactions as cause of admission to hospital: prospective analysis of 18820 patients. BMJ 329(7456):15–19
Posthumus AAG, Alingh CCW, Zwaan CCM, van Grootheest KK, Hanff LL, Witjes BB et al (2012) Adverse drug reaction related admissions in paediatrics, a prospective single-centre study. BMJ Open 2(4):e000934
Rottenkolber D, Schmiedl S, Rottenkolber M, Farker K, Saljé K, Mueller S et al (2011) Adverse drug reactions in Germany: direct costs of internal medicine hospitalizations. Pharmacoepidemiol Drug Saf 20(6):626–634
Rivkin A (2007) Admissions to a medical intensive care unit related to adverse drug reactions. Am J Health-Syst Pharm 64(17):1840–1843
Ruiz B, García M, Aguirre U, Aguirre C (2008) Factors predicting hospital readmissions related to adverse drug reactions. Eur J Clin Pharmacol 64(7):715–722
Wasserfallen J, Livio F, Buclin T, Tillet L, Yersin B, Biollaz J (2001) Rate, type, and cost of adverse drug reactions in emergency department admissions. Eur J Intern Med 12(5):442–447
Goettler M, Schneeweiss S, Hasford J (1997) Adverse drug reaction monitoring—cost and benefit considerations. Part II: cost and preventability of adverse drug reactions leading to hospital admission. Pharmacoepidemiol Drug Saf Suppl 3:S79–S90
Winterstein AG, Sauer BC, Hepler CD, Poole C (2002) Preventable drug-related hospital admissions. Ann Pharmacother 36(7–8):1238–1248
Howard RL, Avery AJ, Slavenburg S, Royal S, Pipe G, Lucassen P et al (2007) Which drugs cause preventable admissions to hospital? A systematic review. Br J Clin Pharmacol 63(2):136–147
Kongkaew C, Noyce PR, Ashcroft DM (2008) Hospital admissions associated with adverse drug reactions: a systematic review of prospective observational studies. Ann Pharmacother 42(7):1017–1025
Taché SV, Sönnichsen A, Ashcroft DM (2011) Prevalence of adverse drug events in ambulatory care: a systematic review. Ann Pharmacother 45(7–8):977–989
Passarelli MC, Jacob-Filho W, Figueras A (2005) Adverse drug reactions in an elderly hospitalised population: inappropriate prescription is a leading cause. Drugs Aging 22(9):767–777
Miguel A, Azevedo LF, Araújo M, Pereira AC (2012) Frequency of adverse drug reactions in hospitalized patients: a systematic review and meta-analysis. Pharmacoepidemiol Drug Saf 21(11):1139–1154
Hakkarainen KM, Andersson Sundell K, Petzold M, Hägg S (2012) Methods for assessing the preventability of adverse drug events: a systematic review. Drug Saf 35(2):105–126
Hallas J, Harvald B, Gram LF, Grodum E, Brosen K, Haghfelt T et al (1990) Drug related hospital admissions: the role of definitions and intensity of data collection, and the possibility of prevention. J Intern Med 228(2):83–90
Olivier P, Caron J, Haramburu F, Imbs JL, Jonville-Béra AP, Lagier G et al (2005) Validation of a measurement scale: example of a French Adverse Drug Reactions Preventability Scale. Therapie 60(1):39–45
Schumock GT, Thornton JP (1992) Focusing on the preventability of adverse drug reactions. Hosp Pharm 27(6):538
Field TS, Gurwitz JH, Harrold LR, Rothschild JM, Debellis K, Seger AC et al (2004) Strategies for detecting adverse drug events among older persons in the ambulatory setting. J Am Med Inform Assoc 11(6):429–498
Pardo Cabello AJ, González Contreras LG, ManzanoGamero MV, Gómez Jiménez FJ, Puche Cañas E (2009) Prevalence of fatal adverse drug reactions in hospitalized patients. Int J Clin Pharmacol Ther 47(10):596–602
Wester K, Jönsson AK, Spigset O, Druid H, HäggS (2008) Incidence of fatal adverse drug reactions: a population based study. Br J Clin Pharmacol 65(4):573–579.
Jönsson AK, Hakkarainen KM, Spigset O, Druid H, Hiselius A, HäggS (2010) Preventable drug related mortality in a Swedish population. Pharmacoepidemiol Drug Saf 9(2):211–215.
Zoppi M, Braunschweig S, Kuenzi UP, Maibach R, Hoigné R (2000) Incidence of lethal adverse drug reactions in the comprehensive hospital drug monitoring, a 20-year survey, 1974-1993, based on the data of Berne/St. Gallen Eur J Clin Pharmacol 56(5):427–430
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Electronic supplementary material
Supplementary file 1
(DOCX 13 kb)
Supplementary file 2
(DOCX 13 kb)
Supplementary file 3
(DOCX 41 kb)
Supplementary file 4
(DOCX 11 kb)
Supplementary Fig. 1
Meta-analytic summary of prevalence of PADRAd. Abbreviation is as follows: PADR Ad preventable ADRs resulting in hospital admission. (JPEG 685 kb)
Supplementary Fig. 2
Meta-analytic summary of subgroup analysis of percentage of PADRAd in paediatric patients for ADR definition and preventability assessment tool. Abbreviation is as follows: PADR Ad preventable ADRs resulting in hospital admission. (JPEG 592 kb)
Supplementary Fig. 3
Meta-analytic summary of subgroup analysis of percentage of PADRAd according to study region. Abbreviation is as follows: PADR Ad preventable ADRs resulting in hospital admission. (JPEG 3014 kb)
Supplementary Fig. 4
Meta-analytic summary of subgroup analysis of percentage of PADRAd according to ADR definition. Abbreviation is as follows: PADR Ad preventable ADRs resulting in hospital admission. (JPEG 2978 kb)
Supplementary Fig. 5
Meta-analytic summary of subgroup analysis of percentage of PADRAd according to ADR identification method. Abbreviation is as follows: PADR Ad preventable ADRs resulting in hospital admission. (JPEG 2797 kb)
Supplementary Fig. 6
Meta-analytic summary of fatal ADRAd and preventable fatal ADRAd. (JPEG 1169 kb)
Rights and permissions
About this article
Cite this article
Patel, N.S., Patel, T.K., Patel, P.B. et al. Hospitalizations due to preventable adverse reactions—a systematic review. Eur J Clin Pharmacol 73, 385–398 (2017). https://doi.org/10.1007/s00228-016-2170-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-016-2170-6