Abstract
Purpose
To estimate the risk of mortality and length of stay in hospitalised patients who have experienced suspected adverse drug reactions (ADRs) as compared to patients who did not experience suspected ADRs.
Methods
A systematic literature search was conducted on databases for observational and randomised controlled studies conducted in any inpatient setting that reported deaths and/or length of hospital stay in patients who had suspected ADRs and did not have suspected ADRs during hospitalisation. PRISMA guidelines were strictly followed during the review. The methodological quality of included studies was assessed using a tool designed by Smyth et al. for the studies of adverse drug reactions. The meta-analytic summary of all-cause mortality was estimated using odds ratio—OR (95% CI) and length of stay using mean difference—MD (95% CI). Both outcomes were pooled using a random effect model (DerSimonian and Laird method). Subgroup and meta-regression were performed based on study variables: study design, age group, study ward, study region, types of suspected ADRs (ADRAd—suspected ADRs that lead to hospitalisation and ADRIn—suspected ADRs that occur following hospitalisation), study duration, sample size and study period. The statistical analysis was conducted through the ‘Review manager software version 5.4.1 and JASP (Version 0.14.1)’.
Results
After screening 475 relevant articles, 55 studies were included in this meta-analysis. Patients having suspected ADRs had reported significantly higher odds of all-cause mortality [OR: 1.50 (95% CI: 1.21–1.86; I2 = 100%) than those patients who did not have suspected ADRs during hospitalisation. Study wards, types of suspected ADRs and sample size were observed as significant predictors of all-cause mortality (p < 0.05). Patients having suspected ADRs had reported significantly higher mean difference in hospital stay [MD: 3.98 (95% CI: 2.91, 5.05; I2 = 99%) than those patients who did not have suspected ADRs during hospitalisation. Types of suspected ADRs and study periods were observed as significant predictors of length of stay (p < 0.05).
Conclusion
Suspected ADRs significantly increase the risk of mortality and length of stay in hospitalised patients.
Systematic review registration.
CRD42020176320.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
An adverse event is any untoward medical occurrence in a patient or clinical trial subject administered a medicinal product which does not necessarily have a causal relationship with this treatment. It can be any symptom, abnormal laboratory finding or disease temporally associated with the use of a medicinal product, whether or not considered related to the medicinal product [1]. A suspected adverse drug reaction (ADR) is defined as a noxious and unintended response to a medicine [1]. In contrast to an adverse event, a causal relationship between a medicinal product and an occurrence is suspected in the case of ADR. This definition includes overdose, off-label use, abuse, misuse, occupational exposure to a medicinal product and medication error [1, 2]. ADRs are an important cause of hospital admission as well as could prolong hospitalisation. The prevalence of suspected ADRs in hospitalised patients could vary from 0.2 to 58% [3,4,5,6,7]. An earlier meta-analysis has estimated that suspected ADRs are the six leading causes of death in the USA in hospitalised patients [8]. The prevalence of mortality among patients due to suspected ADRs in hospitalised patients could vary from 0.0 to 5.2% [9, 10]. The drugs were suspected in 0.0 to 18.2% of inpatient deaths [11].
All-cause mortality refers to the number of people who died from any cause in a given period. It is used to compare additional deaths compared with the baseline. It could provide a measure of the excess mortality, directly and indirectly, attributable to drug-related harm to patients who had suspected ADRs during their hospital stay compared with those who did not have suspected ADRs. Length of stay is an indicator of the use of medical services. Increased hospital stay has been associated with economic burden, risks of complications and mortality [12, 13]. The ADRs may be life threatening and often lead to emergency department visits, hospital admission and prolongation of hospital stay. All earlier meta-analyses on suspected ADRs had focused on the prevalence estimation due to ADR-related mortality in hospitalised patients [3, 7,8,9,10,11] or ADR leading to hospital admission [6, 7, 14] to highlight associated mortality and hospital admission burden. However, no data are available on all-cause mortality due to suspected ADRs and length of stay among the inpatients. In this meta-analysis, we want to estimate the risk of mortality and length of hospital stay in hospitalised patients who have experienced suspected ADRs as compared to patients who do not experience suspected ADRs.
Methods
PRISMA guidelines were strictly adhered to during the systematic review and the study protocol was registered on PROSPERO (CRD42020176320).
Information sources and search strategy
Two investigators (TKP and PBP) independently searched PubMed, Google Scholar, LILACS, SCOPUS, Cochrane Database of Systematic Reviews and a bibliography of relevant articles, systematic reviews and meta-analyses. The keywords used for PubMed and Google Scholar search were (adverse drug event OR adverse drug effect OR adverse drug reaction OR drug related problem OR medication error) AND (inpatient OR hospital*) AND (fatal* OR death OR lethal OR mortality). There were no time and language restrictions. The last search was carried out on 6 May 2021.
Working definition
Suspected ADR: A noxious and unintended response to a medicine. This definition extends beyond suspected reactions at appropriate use of medicine and includes harm from an overdose, off-label use, abuse, misuse, occupational exposure to a medicinal product and medication error [1].
ADRAd: Patient should be admitted to the hospital because of suspected ADR.
ADRIn: Patient develops suspected ADRs following hospitalisation.
Population, intervention, comparator, outcome and study design (PICOS) criteria for the systematic review are presented in Table 1.
Study participants
The study population comprised patients of any age group in the inpatient setting. The study population did not comprise patients who received treatment in ambulatory care or emergency care setting without requiring hospitalisation. The emergency care studies which provided data of patients on subsequent hospitalisation were included.
Study arm (ADR arm)
-
Patients who had at least one suspected ADR in the inpatient setting
Control arm (non-ADR arm)
-
Patients who did not have suspected ADR in the inpatient setting
Exposure
The exposure included administration of drugs to the patients regardless of dose, setting (inpatient or outpatient), administrator (health care professional, caregiver or patient) or use (appropriate or inappropriate) and subsequent occurrence of adverse drug events.
Study selection criteria
We included all observational (cross-sectional, case–control and cohort designs) and randomised controlled studies conducted in the inpatient setting on any study wards. Studies should have reported all-cause deaths and/or length of hospital stay in study arms (suspected ADR and non-suspected ADR arms) or should have provided sufficient data to compute them. We excluded studies not differentiating suspected ADRs from adverse events due to non-drug-related interventions or complications of surgical or medical procedures (e.g. low oxygen saturation after tracheostomy, infection in the surgical wound). Studies also excluded if solely depended on voluntary or spontaneous reporting methods to detect suspected ADRs, outpatient or ambulatory care settings, focused on specific drugs (e.g. antiepileptic drugs) or clinical conditions (e.g. renal failure) or events (e.g. anaphylactic reaction) and duplicate studies.
Primary outcome
-
All-cause mortality: meta-analytic summary of all-cause mortality between suspected ADR and the non-suspected ADR groups was the primary outcome
Secondary outcome
-
Length of stay: meta-analytic summary of the mean length of hospital stay between suspected ADR and the non-suspected ADR group was the secondary outcome.
Study screening
Initially, two investigators independently screened the title and abstract as per a predefined questionnaire. In the next stage, retrieving full texts were assessed as per the selection criteria and availability of outcome data. Any disagreements in study selection were resolved through discussion and consensus.
Data extraction
The following data were extracted in the predefined Excel sheet: first author, publication year, geographical location, study design, study period, study duration, study ward, age group, data collection methods, suspected ADR definition used, types of suspected ADR studied, personnel who identified suspected ADR, causality, severity and preventability assessment, baseline data of population in study arms (age, gender, number of drugs received), total number of inpatients in a study ward, number of patients in suspected ADR and non-suspected ADR arm, total number of deaths in suspected ADR and non-suspected ADR arm, length of hospital stay in suspected ADR and non-suspected ADR arm patients. All extracted variables were cross-checked to ensure the quality of data extraction.
Risk of bias assessment of included studies
The risk of bias was assessed using the tool designed by Smyth et al. for the studies of adverse drug reactions [15]. The assessment was based on a description of the study design, methods for identifying suspected ADRs and methods for determining causality, preventability and severity [15]. The publication bias was assessed through visual inspection of the funnel plot and Egger’s regression test.
Data synthesis
The meta-analytic summary of all-cause mortality was estimated using odds ratio—OR (95% CI) and length of stay using mean difference—MD (95% CI). High heterogeneity was anticipated, and a random effect model (DerSimonian and Laird method) was preferred over a fixed effect for the meta-analysis.
A forest plot was generated to display OR (95% CIs) and MD (95% CI) for each study. An I2 test was used to evaluate heterogeneity. An I2 value of 25%, 50% and 75% was considered low, medium and high heterogeneity, respectively. The sensitivity analysis of the primary and secondary outcomes was performed using a low risk of bias studies.
Subgroup analysis
The subgroup analysis of both outcome parameters (mortality and length of stay) was performed to explore the possible sources of heterogeneity. The following study variables were used: study design, age group, study ward, study region, types of suspected ADRs based on their setting of occurrence (ADRAd—suspected ADRs that lead to hospitalisation and ADRIn—suspected ADRs that occur following hospitalisation), study duration, sample size and study period.
Meta-regression
Initially, the influence of all study variables on mortality and length of stay was assessed through univariable meta-regression. The subgroup with a minimum of 4 studies was selected as a moderator [16]. Subsequently, the study variables showing a significance level of p < 0.10 were further explored through multivariable regression using the random effect model [17].
Statistical package
Review Manager software (RevMan version 5.4.1) was used for meta-analysis and subgroup analysis. JASP (Version 0.14.1) was used for univariable and multivariable meta-regression analysis.
Result
Literature search
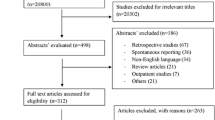
A total of 55 full-text articles were included from 21,919 retrieved references from the literature of databases and other sources [18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72]. A PRISMA flow chart of included studies is presented in Fig. 1. The study designs of excluded full-text studies were cross-sectional (215), case–control (2), cohort (3) and randomised controlled trial (1).
Study characteristics
The general characteristics of included studies are presented in Table 2. The included studies had used cross-sectional (prospective—31, retrospective—17, prospective and retrospective—1), case–control (5) and cohort (1) study designs. Studies were conducted in the internal medicine ward (13), whole hospital (13), emergency department (8), intensive care unit—ICU (6), multispecialty wards (6), geriatric ward (2), paediatric ward and ICU (2), paediatric ward (2), internal medicine and ICU (1), medical and surgical wards and ICU (1) and surgical ward (1). The emergency department studies admitted patients through emergency units [25, 54, 57,58,59] or had admission in emergency wards [62, 68, 72]. A total of 21 studies focused on ADRAd and 19 studies on ADRIn. Ten studies included both ADRAd and ADRIn, while 5 studies did not specify the type of suspected ADRs. Studies used different data collection methods to detect suspected ADRs: medical record review (28), interview and medical record review (19), ADE trigger tool–based medical record review (2), ADE trigger tool–based medical record review through computerised record system (1), medical record review with voluntary reporting by health care professionals (1) and medical record review through computerised record system (1). A total of 15 studies used WHO or a similar definition, 7 studies Edwards and Aronson definition, 3 studies ICD-9 E codes and 2 studies Aronson and Ferner definition to identify suspected ADRs. Claret et al. only focused on medication errors [25]. In the case of Darchy et al., data of iatrogenic disease due to drug exposure were considered a suspected ADR, while data of iatrogenic disease due to medical and surgical procedures were excluded [28]. In the case of ICD-9 and ICD-10 code–based studies, only data on suspected ADRs [19, 21, 48, 60, 71] were considered. The detailed general characteristics of included studies are presented in Supplementary Table 1.
Risk of bias assessment in individual studies
All studies clearly described the study design. One study did not specify the suspected ADR identification method [63], while three studies did not describe data collection methods [45, 50, 63]. Seven studies did not specify individuals who identified suspected ADRs [19, 21, 35, 45, 48, 60, 71]. A total of 39 studies specified the methods of causality, 24 preventability and 38 severity assessment of suspected ADRs [Supplementary Table 2]. The risk of bias summary is described in Supplementary Fig. 1.
All-cause mortality
A total of 31 studies (1,577,946 all-cause deaths; 38,377,918 patients) contributed to all-cause mortality outcome. The suspected ADR arm reported 35,644 all-cause deaths (5.50%) out of 648,289 inpatients, while the non-suspected ADR arm reported 1,542,302 all-cause deaths (4.09%) out of 37,729,629 inpatients. As shown in Fig. 2, patients having suspected ADR had reported significantly higher odds of mortality [OR: 1.50 (95% CI: 1.21–1.86; I2 = 100%) than those patients who did not have suspected ADR during hospitalisation. On sensitivity analysis, the OR of all-cause mortality varied from 1.25 to 1.61 with a low risk of bias studies [Supplementary table 3].
Length of hospital stay
A total of 40 studies (8,282,929 patients) contributed to the length of hospital stay outcome. As shown in Fig. 3, patients having suspected ADR had reported significantly higher mean difference in hospital stay [MD: 3.98 (95% CI: 2.91–5.05; I2 = 99%) than those patients who did not have suspected ADR during hospitalisation. On sensitivity analysis, MD in the length of hospital stay varied from 3.06 to 3.98 with a low risk of bias studies [Supplementary table 3].
Subgroup analysis of all-cause mortality and length of hospital stay based on the study characteristics
Study design: Retrospective studies showed a trend of higher odds of all-cause mortality and mean length of stay than prospective (Tables 3 and 4). Prospective studies showed low heterogeneity (7%).
Age groups: Age groups showed different trends for all-cause mortality and hospital stay. ‘Adults and elderly’ studies showed a trend of higher odds of all-cause mortality (Table 3), while ‘paediatric’ and ‘elderly’ age group studies showed a trend of higher mean length of stay (Table 4). ‘All age’ studies (n = 2) also showed low heterogeneity (I2 = 0%) for a hospital stay. Both ‘all age’ studies used a retrospective study design.
Study wards: Whole hospital and multispecialty studies showed a trend of higher odds of all-cause mortality and mean length of hospital stay than intensive care unit and emergency department studies. Emergency department studies showed low heterogeneity for all-cause mortality (I2 = 0%; n = 4) and length of stay (I2 = 44%; n = 5). Their common characteristics were prospective design, study region and shorter duration.
Study regions: South American and Asian studies showed a trend of higher odds of all-cause mortality and mean length of stay than other region studies.
Types of suspected ADRs: Studies focusing on ADRIn showed a trend of higher odds of all-cause mortality [OR: 2.01 (95% CI: 1.68–2.42); I2 = 58%, n = 9 vs. OR: 0.96 (95% CI: 0.72–1.27); I2 = 95%, n = 13] and mean length of stay [MD: 7.13 (95% CI: 3.90–10.37); I2 = 99%, n = 16 vs. OR: 0.58 (95% CI: − 0.21 to 1.38); I2 = 91%, n = 14] than studies focusing on ADRAd (Tables 3 and 4). Both groups showed high heterogeneity and differed in study characteristics.
Study duration: Longer duration (≥ 12 months) studies showed a trend of higher odds of all-cause mortality and mean length of stay than shorter duration (< 12 months) studies.
Sample size: Large sample size (≥ 1000) studies showed a trend of higher odds of all-cause mortality and mean length of stay than small sample size (< 1000) studies.
Study period: The study period showed different trends for all-cause mortality and hospital stay. Studies conducted ‘before 2000’ showed a trend of higher odds of all-cause mortality (Table 3), while studies conducted ‘after 2010’ showed a trend of longer length of stay (Table 4).
Meta-regression
The univariable analysis showed that all-cause mortality was higher in ‘Medical and surgical wards, ICU’ studies (regression coefficient: 1.37), ADRIn (regression coefficient: 0.89), ‘ADRAd and ADRIn’ studies (regression coefficient: 0.81) and large sample size studies (regression coefficient: 0.63). Study wards, types of suspected ADRs and sample size were further explored through a multivariable regression model. All three study characteristics were observed as significant predictors of all-cause mortality (p < 0.05) (Table 3).
The univariable analysis showed that length of stay was higher in ‘multispecialty wards’ (regression coefficient: 6.17), whole Hospital studies (regression coefficient: 4.39), ADRIn (regression coefficient: 6.28) and ‘ADRAd and ADRIn’ studies (regression coefficient: 4.29) and studies conducted between 2000 and 2010 (regression coefficient: 2.85). Study wards, types of suspected ADRs and study periods were further explored through a multivariable regression model. As shown in Table 4, types of suspected ADRs and study periods were observed as significant predictors of length of stay (p < 0.05).
Discussion
The findings of our meta-analysis suggest that suspected ADRs are significantly associated with the risk of mortality and length of stay in hospitalised patients. This is the first meta-analysis that reported odds of all-cause mortality and extra days of the length of hospital stay associated with suspected ADRs. All earlier meta-analyses had estimated the prevalence of suspected ADR-related mortality among inpatients [2, 6,7,8,9,10,11, 14]. None of them compared the odds of mortality and length of hospital stay between patients with suspected ADRs with those who did not develop suspected ADRs.
This meta-analysis presents two main findings. First, the odds of mortality in patients who had suspected ADRs is one and half times higher than those patients who did not have suspected ADRs during their hospitalisation. Second, patients who had suspected ADRs are 4 days likely to stay more in the hospital than those patients who did not have suspected ADRs during their hospital stay.. This suggests prevention of ADRs will significantly reduce the burden on the patient and hospital. A sizable proportion of inpatients develops suspected ADRs during their hospitalisation. Earlier meta-analyses suggest one out of five to seven patients had ADRs during their hospitalisation [4, 5]. Almost half of suspected ADRs among inpatients are preventable [73, 74].
Study wards, types of suspected ADRs and sample size could accurately predict higher odds of all-cause mortality in patients having suspected ADRs. An earlier meta-analysis observed study wards were a significant predictor of the percentage of drug-related deaths out of the total inpatient mortality [11]. Panagioti et al. observed medical care settings (general hospitals, primary and advanced hospital specialities) as a significant predictor of the prevalence of preventable patient harm in medical care [17]. However, Martins et al. in an earlier meta-analysis did not find any study characteristics as a significant predictor of the percentage of suspected ADRs among adults [75]. The predictor of heterogeneity among meta-analysis of suspected ADR studies could be varied depending upon the variability in the study population (all age vs adults), the denominator (all inpatients vs inpatient deaths) and types of suspected ADRs (all suspected ADRs vs preventable adverse events). The preventive strategies should focus on all these factors to identify the priority areas.
One of the important findings of this meta-analysis is the difference in the impact of types of suspected ADRs based on their origin in outpatient (ADRAd) or inpatient settings (ADRIn) on all-cause mortality and length of hospital stay. The patients who experienced ADRIn had two times higher odds of all-cause mortality and 7 days higher length of hospital stay than those patients who did not experience ADRIn. All-cause of mortality and length of stay do not differ between patients who were admitted due to ADRs and other disease conditions. This could be because of circumstances involved in the occurrence of ADRs based on the setting and the need for different preventive strategies for ADRAd and ADRIn.
Sample size rather than study design (prospective or retrospective) is an important factor in assessing suspected ADR-related all-cause mortality. Large (≥ 1000) sample size studies had shown significantly higher odds of mortality than the small sample size (< 1000) studies. Small sample studies do not yield precise or reliable effect size estimates [76, 77]. Our findings suggest suspected ADR-related all-cause mortality data should be cautiously interpreted from the small sample size studies. We did not observe the impact of study design (prospective or retrospective) on all all-cause mortality and length of hospital stay. Seven out of fifteen retrospective and one out of fifteen prospective studies had large sample sizes.
Though the trend of all-cause mortality has reduced in studies conducted in the twenty-first century over the twentieth century, the pattern of a rising trend in hospital stay in the last two decades should be of concern. Patients experiencing a suspected ADR during hospitalisation stay 4 days longer in the hospital. This is important because the extra length of stay contributes to the economic burden on the patients [78]. Higher hospital stay enhances the risk of opportunistic infections, worse treatment outcomes and the economic burden on the patients. Higher stay decreases the bed turnover rate, which can reduce the profit margin and enhance the social costs [79, 80]. Suspected ADRs are a significant burden to patients and healthcare systems.
The study admission wards are the important predictors of suspected ADR-related mortality. Patients admitted to the whole hospital and ‘medical and surgical wards and ICU’ experienced higher odds of mortality in the suspected ADR arm. Patients experiencing suspected ADRs had more than 5 days of additional hospital stay in studies conducted in the whole hospital, multispeciality wards, paediatric wards and PICU. There is a critical need to understand the nature of suspected ADRs and their unique complexities in different admission wards to provide safe patient care. An earlier meta-analysis has shown that pharmacist-led interventions, a brief educational session and a technology intervention could significantly reduce the risk of acquiring serious ADRs [81,82,83]. Such interventions should be validated in different admission wards and settings to devise the best ADR preventive strategies to reduce the mortality and length of hospital stay. Patients admitted to ICUs are considered most at risk of errors and adverse outcomes [84]. ICUs and emergency department studies did not show high mortality and longer lengths of stay due to suspected ADRs. This could be because four out of six ICU and seven out of eight emergency department studies focused on ADRAd.
This meta-analysis has several limitations. The definition of suspected ADRs varied among the included studies. Studies that followed the WHO definition could have excluded medication errors and unintentional drug overdoses related to ADRs. Edwards and Aaronson Edwards and Aaronson’s ADR definition includes ADRs at therapeutic doses and medication errors. This definition also covers reactions related to excipients. ICD-9 E code–based studies could have included ADRs at therapeutic doses, accidental overdoses and poisoning. Few studies also included non-compliance as a suspected ADR. We could not assess the impact of a computerised medical record system as a data collection tool due to the few numbers of included studies. Twelve out of thirty-one included studies in all-cause mortality outcome assessment did not specify the causality assessment methods between suspected ADRs and suspected drugs. However, we did not observe its impact on sensitivity analysis after excluding these studies on sensitivity analysis. All-cause mortality and length of hospital stay varied among included studies. Meta-regression and subgroup analyses could only partially explain the variability in the estimation of the length of stay. There is a possibility that other relevant factors could also account for the unexplained heterogeneity. Both outcomes could have been affected by the underlying condition for admission, its severity and co-morbidity. The findings of subgroup analysis and meta-regression require cautious interpretation due to the small number of studies on each characteristic.
Conclusion
Patients having suspected ADRs in the hospital setting are at increased risk of all-cause mortality and longer hospital stay than those who did not experience suspected ADRs. Study wards, types of suspected ADRs and sample size are heterogeneity modifiers in the case of all-cause mortality, while types of suspected ADRs and admission wards are heterogeneity modifiers in the case of length of hospital stay.
Data availability
The study data are included in the main text/supplementary data file; further inquiries can be directed to the corresponding author.
References
European Medicines Agency (2017) Guideline on good pharmacovigilance practices (GVP). Available from: https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-good-pharmacovigilance-practices-annex-i-definitions-rev-4_en.pdf. Last assessed on 20 Nov 2022
Baldo P, Francescon S, Fornasier G (2018) Pharmacovigilance workflow in Europe and Italy and pharmacovigilance terminology. Int J Clin Pharm 40(4):748–753
Angamo MT, Chalmers L, Curtain CM, Bereznicki LR (2016) Adverse-drug-reaction-related hospitalisations in developed and developing countries: a review of prevalence and contributing factors. Drug Saf 39(9):847–857
Laatikainen O, Miettunen J, Sneck S, Lehtiniemi H, Tenhunen O, Turpeinen M (2017) The prevalence of medication-related adverse events in inpatients-a systematic review and meta-analysis. Eur J Clin Pharmacol 73(12):1539–1549
Miguel A, Azevedo LF, Araújo M, Pereira AC (2012) Frequency of adverse drug reactions in hospitalized patients: a systematic review and meta-analysis. Pharmacoepidemiol Drug Saf 21(11):1139–1354
Oscanoa TJ, Lizaraso F, Carvajal A (2017) Hospital admissions due to adverse drug reactions in the elderly. A meta-analysis Eur J Clin Pharmacol 73(6):759–770
Patel TK, Patel PB (2016) Incidence of adverse drug reactions in Indian hospitals: a systematic review of prospectives. Curr Drug Saf 11(2):128–136
Lazarou J, Pomeranz BH, Corey PN (1998) Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. JAMA 279(15):1200–1205
Patel TK, Patel PB (2018) Mortality among patients due to adverse drug reactions that lead to hospitalization: a meta-analysis. Eur J Clin Pharmacol 74(6):819–832
Patel PB, Patel TK (2019) Mortality among patients due to adverse drug reactions that occur following hospitalisation: a meta-analysis. Eur J Clin Pharmacol 75(9):1293–1307
Patel TK, Patel PB, Bhalla HL, Kishore S (2022) Drug-related deaths among inpatients: a meta-analysis. Eur J Clin Pharmacol 78(2):267–278
Baek H, Cho M, Kim S, Hwang H, Song M, Yoo S (2018) Analysis of length of hospital stay using electronic health records: a statistical and data mining approach. PLoS ONE 13(4):e0195901
Lingsma HF, Bottle A, Middleton S, Kievit J, Steyerberg EW, Marang-van de Mheen PJ (2018) Evaluation of hospital outcomes: the relation between length-of-stay, readmission, and mortality in a large international administrative database. BMC Health Serv Res 18(1):116
Beijer HJ, de Blaey CJ (2002) Hospitalisations caused by adverse drug reactions (ADR): a meta-analysis of observational studies. Pharm World Sci 24(2):46–54
Smyth RM, Gargon E, Kirkham J, Cresswell L, Golder S, Smyth R et al (2012) Adverse drug reactions in children–a systematic review. PLoS ONE 7(3):e24061
Fu R, Gartlehner G, Grant M, Shamliyan T, Sedrakyan A, Wilt TJ et al. (2010) Conducting quantitative synthesis when comparing medical interventions: AHRQ and the effective health care program. 2010 Oct 25. In: Methods guide for effectiveness and comparative effectiveness reviews [Internet]. Rockville (MD): Agency for Healthcare Research and Quality (US); 2008-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK49407/
Panagioti M, Khan K, Keers RN, Abuzour A, Phipps D, Kontopantelis E et al (2019) Prevalence, severity, and nature of preventable patient harm across medical care settings: systematic review and meta-analysis. BMJ 366:l4185
Alexopoulou A, Dourakis SP, Mantzoukis D, Pitsariotis T, Kandyli A, Deutsch M et al (2008) Adverse drug reactions as a cause of hospital admissions: a 6-month experience in a single center in Greece. Eur J Intern Med 19(7):505–510
Amann C, Hasford J, Stausberg J (2012) Stationäre Aufnahmen wegen unerwünschter Arzneimittelereignisse (UAE): Analyse der DRG-Statistik 2006 [Hospital admission due to adverse drug events (ADE): an analysis of German routine hospital data of 2006]. Gesundheitswesen 74(10):639–644
Angamo MT, Chalmers L, Curtain CM, Yilma D, Bereznicki L (2018) Mortality from adverse drug reaction-related hospitalizations in south-west Ethiopia: a cross-sectional study. J Clin Pharm Ther 43(6):790–798
Bond CA, Raehl CL (2006) Adverse drug reactions in United States hospitals. Pharmacotherapy 26(5):601–608
Brvar M, Fokter N, Bunc M, Mozina M (2009) The frequency of adverse drug reaction related admissions according to method of detection, admission urgency and medical department specialty. BMC Clin Pharmacol 9:8
Camargo AL, Cardoso Ferreira MB, Heineck I (2006) Adverse drug reactions: a cohort study in internal medicine units at a university hospital. Eur J Clin Pharmacol 62(2):143–149
Chan M, Nicklason F, Vial JH (2001) Adverse drug events as a cause of hospital admission in the elderly. Intern Med J 31(4):199–205
Claret PG, Bobbia X, Renia R, Stowell A, Crampagne J, Flechet J et al (2016) Prescription errors by emergency physicians for inpatients are associated with emergency department length of stay. Therapie S0040–5957(16):30053–30061
Classen DC, Pestotnik SL, Evans RS, Lloyd JF, Burke JP (1997) Adverse drug events in hospitalized patients. Excess length of stay, extra costs, and attributable mortality. JAMA 277(4):301–306
Damen NL, Baines R, Wagner C, Langelaan M (2017) Medication-related adverse events during hospitalization: a retrospective patient record review study in The Netherlands. Pharmacoepidemiol Drug Saf 26(1):32–39
Darchy B, Le Mière E, Figuérédo B, Bavoux E, Domart Y (1999) Iatrogenic diseases as a reason for admission to the intensive care unit: incidence, causes, and consequences. Arch Intern Med 159(1):71–78
Davies EC, Green CF, Mottram DR, Pirmohamed M (2006) Adverse drug reactions in hospital in-patients: a pilot study. J Clin Pharm Ther 31(4):335–341
Davies EC, Green CF, Taylor S, Williamson PR, Mottram DR, Pirmohamed M (2009) Adverse drug reactions in hospital in-patients: a prospective analysis of 3695 patient-episodes. PLoS ONE 4(2):e4439
de Boer M, Boeker EB, Ramrattan MA, Kiewiet JJ, Dijkgraaf MG, Boermeester MA et al (2013) Adverse drug events in surgical patients: an observational multicentre study. Int J Clin Pharm 35(5):744–752
de Las SR, Díaz-Agudelo D, Burgos-Flórez FJ, Vaca C, Serrano-Meriño DV (2016) Adverse drug reactions in hospitalized Colombian children. Colomb Med (Cali) 47(3):142–147
Dequito AB, Mol PG, van Doormaal JE, Zaal RJ, van den Bemt PM, Haaijer-Ruskamp FM et al (2011) Preventable and non-preventable adverse drug events in hospitalized patients: a prospective chart review in the Netherlands. Drug Saf 34(11):1089–1100
Dormann H, Neubert A, Criegee-Rieck M, Egger T, Radespiel-Tröger M, Azaz-Livshits T et al (2004) Readmissions and adverse drug reactions in internal medicine: the economic impact. J Intern Med 255(6):653–663
Esteban Jiménez Ó, Navarro Pemán C, González Rubio F, Lanuza Giménez FJ, Montesa Lou C (2017) Análisis de la incidencia y de las características clínicas de las reacciones adversas a medicamentos de uso humano en el medio hospitalario [A study of incidence and clinical characteristics of adverse drug reactions in hospitalized patients.]. Rev Esp Salud Publica 91:e201712050
Fattinger K, Roos M, Vergères P, Holenstein C, Kind B, Masche U et al (2000) Epidemiology of drug exposure and adverse drug reactions in two Swiss departments of internal medicine. Br J Clin Pharmacol 49(2):158–167
Giordani F, Rozenfeld S, Martins M (2014) Adverse drug events identified by triggers at a teaching hospital in Brazil. BMC Pharmacol Toxicol 15:71
Grenouillet-Delacre M, Verdoux H, Moore N, Haramburu F, Miremont-Salamé G, Etienne G et al (2007) Life-threatening adverse drug reactions at admission to medical intensive care: a prospective study in a teaching hospital. Intensive Care Med 33(12):2150–2157
Haffner S, von Laue N, Wirth S, Thürmann PA (2005) Detecting adverse drug reactions on paediatric wards: intensified surveillance versus computerised screening of laboratory values. Drug Saf 28(5):453–464
Haukland EC, Mevik K, von Plessen C, Nieder C, Vonen B (2019) Contribution of adverse events to death of hospitalised patients. BMJ Open Qual 8(1):e000377
Hofer-Dueckelmann C, Prinz E, Beindl W, Szymanski J, Fellhofer G, Pichler M, Schuler J (2011) Adverse drug reactions (ADRs) associated with hospital admissions - elderly female patients are at highest risk. Int J Clin Pharmacol Ther 49(10):577–586
Hu Q, Qin Z, Zhan M, Chen Z, Wu B, Xu T (2020) Validating the Chinese geriatric trigger tool and analyzing adverse drug event associated risk factors in elderly Chinese patients: a retrospective review. PLoS ONE 15(4):e0232095
Ji HH, Song L, Xiao JW, Guo YX, Wei P, Tang TT et al (2018) Adverse drug events in Chinese pediatric inpatients and associated risk factors: a retrospective review using the Global Trigger Tool. Sci Rep 8(1):2573
Jolivot PA, Pichereau C, Hindlet P, Hejblum G, Bigé N, Maury E et al (2016) An observational study of adult admissions to a medical ICU due to adverse drug events. Ann Intensive Care 6(1):9
Kojima T, Matsui T, Suzuki Y, Takeya Y, Tomita N, Kozaki K et al (2020) Risk factors for adverse drug reactions in older inpatients of geriatric wards at admission: multicenter study. Geriatr Gerontol Int 20(2):144–149
Liao PJ, Mao CT, Chen TL, Deng ST, Hsu KH (2019) Factors associated with adverse drug reaction occurrence and prognosis, and their economic impacts in older inpatients in Taiwan: a nested case-control study. BMJ Open 9(5):e026771
Mehta U, Durrheim DN, Blockman M, Kredo T, Gounden R, Barnes KI (2008) Adverse drug reactions in adult medical inpatients in a South African hospital serving a community with a high HIV/AIDS prevalence: prospective observational study. Br J Clin Pharmacol 65(3):396–406
Miguel A, Marques B, Freitas A, Lopes F, Azevedo L, Pereira AC (2013) Detection of adverse drug reactions using hospital databases-a nationwide study in Portugal. Pharmacoepidemiol Drug Saf 22(8):907–913
Mjörndal T, Boman MD, Hägg S, Bäckström M, Wiholm BE, Wahlin A et al (2002) Adverse drug reactions as a cause for admissions to a department of internal medicine. Pharmacoepidemiol Drug Saf 11(1):65–72
Moore N, Lecointre D, Noblet C, Mabille M (1998) Frequency and cost of serious adverse drug reactions in a department of general medicine. Br J Clin Pharmacol 45(3):301–308
Mouton JP, Njuguna C, Kramer N, Stewart A, Mehta U, Blockman M et al (2016) Adverse drug reactions causing admission to medical wards: a cross-sectional survey at 4 hospitals in South Africa. Medicine (Baltimore) 95(19):e3437
Mouton JP, Fortuin-de Smidt MC, Jobanputra N, Mehta U, Stewart A, de Waal R et al (2020) Serious adverse drug reactions at two children’s hospitals in South Africa. BMC Pediatr 20(1):3
Nazer LH, Eljaber R, Rimawi D, Hawari FI (2013) Adverse drug events resulting in admission to the intensive care unit in oncology patients: incidence, characteristics and associated cost. J Oncol Pharm Pract 19(4):298–304
Olivier P, Bertrand L, Tubery M, Lauque D, Montastruc JL, Lapeyre-Mestre M (2009) Hospitalizations because of adverse drug reactions in elderly patients admitted through the emergency department: a prospective survey. Drugs Aging 26(6):475–482
Park S, In Y, Suh GY, Sohn K, Kim E (2013) Evaluation of adverse drug reactions in medical intensive care units. Eur J Clin Pharmacol 69(1):119–131
Passarelli MC, Jacob-Filho W, Figueras A (2005) Adverse drug reactions in an elderly hospitalised population: inappropriate prescription is a leading cause. Drugs Aging 22(9):767–777
Pedrós C, Quintana B, Rebolledo M, Porta N, Vallano A, Arnau JM (2014) Prevalence, risk factors and main features of adverse drug reactions leading to hospital admission. Eur J Clin Pharmacol 70(3):361–367
Pedrós C, Formiga F, Corbella X, Arnau JM (2016) Adverse drug reactions leading to urgent hospital admission in an elderly population: prevalence and main features. Eur J Clin Pharmacol 72(2):219–226
Phillips AL, Nigro O, Macolino KA, Scarborough KC, Doecke CJ, Angley MT et al (2014) Hospital admissions caused by adverse drug events: an Australian prospective study. Aust Health Rev 38(1):51–57
Riaz M, Brown JD (2019) Association of adverse drug events with hospitalization outcomes and costs in older adults in the USA using the nationwide readmissions database. Pharmaceut Med 33(4):321–329
Rozenfeld S, Giordani F, Coelho S (2013) Eventos adversos a medicamentos em hospital terciário: estudo piloto com rastreadores [Adverse drug events in hospital: pilot study with trigger tool]. Rev Saude Publica 47(6):1102–1111
Rydberg DM, Holm L, Engqvist I, Fryckstedt J, Lindh JD, Stiller CO et al (2016) Adverse drug reactions in a tertiary care emergency medicine ward - prevalence, preventability and reporting. PLoS ONE 11(9):e0162948
Sánchez Muñoz-Torrero JF, Barquilla P, Velasco R, Fernández Capitan Mdel C, Pacheco N, Vicente L et al (2010) Adverse drug reactions in internal medicine units and associated risk factors. Eur J Clin Pharmacol 66(12):1257–1264
Suh DC, Woodall BS, Shin SK, Hermes-De Santis ER (2000) Clinical and economic impact of adverse drug reactions in hospitalized patients. Ann Pharmacother 34(12):1373–1379
Takahashi Y, Sakuma M, Murayama H, Morimoto T (2018) Effect of baseline renal and hepatic function on the incidence of adverse drug events: the Japan Adverse Drug Events study. Drug Metab Pers Ther 33(4):165–173
Tangiisuran B, Davies JG, Wright JE, Rajkumar C (2012) Adverse drug reactions in a population of hospitalized very elderly patients. Drugs Aging 29(8):669–679
Toscano Guzmán MD, Banqueri MG, Otero MJ, Fidalgo SS, Noguera IF, Guerrero MCP (2021) Validating a trigger tool for detecting adverse drug events in elderly patients with multimorbidity (TRIGGER-CHRON). J Patient Saf 17(8):e976–e982
Trifirò G, Calogero G, Ippolito FM, Cosentino M, Giuliani R, Conforti A et al (2005) Adverse drug events in emergency department population: a prospective Italian study. Pharmacoepidemiol Drug Saf 14(5):333–340
van der Hooft CS, Sturkenboom MC, van Grootheest K, Kingma HJ, Stricker BH (2006) Adverse drug reaction-related hospitalisations: a nationwide study in The Netherlands. Drug Saf 29(2):161–168
Vargas E, Simón J, Martin JC, Puerro M, Gonzalez-Callejo MA, Jaime M et al (1998) Effect of adverse drug reactions on length of stay in intensive care units. Clin Drug Investig 15(4):353–360
Vitorino M, Aguiar P, Sousa P (2020) In-hospital adverse drug events: analysis of trend in Portuguese public hospitals. Cad Saude Publica 36(3):e00056519
Zed PJ, Abu-Laban RB, Balen RM, Loewen PS, Hohl CM, Brubacher JR et al (2008) Incidence, severity and preventability of medication-related visits to the emergency department: a prospective study. CMAJ 178(12):1563–1569
Hakkarainen KM, Hedna K, Petzold M, Hägg S (2012) Percentage of patients with preventable adverse drug reactions and preventability of adverse drug reactions–a meta-analysis. PLoS ONE 7(3):e33236
Patel NS, Patel TK, Patel PB, Naik VN, Tripathi CB (2017) Hospitalizations due to preventable adverse reactions-a systematic review. Eur J Clin Pharmacol 73(4):385–398
Martins AC, Giordani F, Rozenfeld S (2014) Adverse drug events among adult inpatients: a meta-analysis of observational studies. J Clin Pharm Ther 39(6):609–620
Hackshaw A (2008) Small studies: strengths and limitations. Eur Respir J 32(5):1141–1143
Case LD, Ambrosius WT (2007) Power and sample size. Methods Mol Biol 404:377–408
Barnett AG, Page K, Campbell M, Martin E, Rashleigh-Rolls R, Halton K et al (2013) The increased risks of death and extra lengths of hospital and ICU stay from hospital-acquired bloodstream infections: a case-control study. BMJ Open 3(10):e003587
Bueno H, Ross JS, Wang Y, Chen J, Vidán MT, Normand SL et al (2010) Trends in length of stay and short-term outcomes among Medicare patients hospitalized for heart failure, 1993–2006. JAMA 303(21):2141–2147
Rotter T, Kinsman L, James E, Machotta A, Gothe H, Willis J, et al. (2010) Clinical pathways: effects on professional practice, patient outcomes, length of stay and hospital costs. Cochrane Database Syst Rev (3):CD006632
Gray SL, Hart LA, Perera S, Semla TP, Schmader KE, Hanlon JT (2018) Meta-analysis of interventions to reduce adverse drug reactions in older adults. J Am Geriatr Soc 66(2):282–288
O’Mahony D (2016) Pharmacists and prevention of inappropriate prescribing in hospital. Age Ageing 45(2):181–183
Rieckert A, Teichmann AL, Drewelow E, Kriechmayr C, Piccoliori G, Woodham A et al (2019) Reduction of inappropriate medication in older populations by electronic decision support (the PRIMA-eDS project): a survey of general practitioners’ experiences. J Am Med Inform Assoc 26(11):1323–1332
Prgomet M, Li L, Niazkhani Z, Georgiou A, Westbrook JI (2017) Impact of commercial computerized provider order entry (CPOE) and clinical decision support systems (CDSSs) on medication errors, length of stay, and mortality in intensive care units: a systematic review and meta-analysis. J Am Med Inform Assoc 24(2):413–422
Author information
Authors and Affiliations
Contributions
TKP contributed to all aspects of this manuscript, including conception and design; acquisition, analysis, interpretation of data and drafting of the manuscript. PBP contributed to the design, acquisition, analysis and interpretation of data. HLB contributed to the analysis, interpretation of data and drafting of the manuscript. PD, VB and SK contributed to the interpretation of data and drafting of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Patel, T.K., Patel, P.B., Bhalla, H.L. et al. Impact of suspected adverse drug reactions on mortality and length of hospital stay in the hospitalised patients: a meta-analysis. Eur J Clin Pharmacol 79, 99–116 (2023). https://doi.org/10.1007/s00228-022-03419-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-022-03419-7