Abstract
Understanding natural movement patterns and ecological roles of marine megafauna is a research priority best studied in areas with minimal human impact. The spatial distribution patterns specifically for immature turtles at foraging grounds have been highlighted as a research gap for effective management and conservation strategies for sea turtle populations. Capture–mark–recapture (CMR) records (n = 2287) of 1672 immature green (Chelonia mydas) (n = 1158) and hawksbill turtles (Eretmochelys imbricata) (n = 514) from a long-term (1981–2021) in-water CMR program at Aldabra Atoll, Seychelles, were analyzed for 10 sites (0.35–25 km apart). Site fidelity was not correlated with either season or turtle size. Green turtles had lower site fidelity than hawksbill turtles. Green turtles showed avoidance (i.e., opposite of fidelity) of three sites, while hawksbill turtles displayed high fidelity to two sites. Sites displaying non-random behavior (avoidance and/or fidelity) did not share the same benthic habitat types. Results indicate that fidelity can be detected at a fine scale with CMR, but that further exploration into the habitat characteristics of the sites and the ecological roles of both species at the atoll is needed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Marine megafauna substantially influence coastal ecosystems, as contributors to both top-down (e.g., as predation and herbivory; Burkholder et al. 2013; Atwood et al. 2015) and bottom-up (e.g., nutrient cycling; Bouchard and Bjorndal 2000; Roman et al. 2014) processes. Understanding the movement patterns and habitat use of these frequently migratory and wide-ranging species is crucial both for identifying their ecological roles (Hays et al. 2016) and for improving their conservation management (Hooker et al. 2011; Hays et al. 2019), since to effectively protect a species, we need to know where it occurs and what it needs. Marine ecosystems are threatened globally (Halpern et al. 2015), and marine protected areas (MPAs) have been an instrumental tool for conserving biodiversity on a large scale (Grorud-Colvert et al. 2021). While MPAs usually do not cover the spatial extent of a migratory species, they can provide important protection at aggregation sites (such as for foraging or breeding; Hooker et al. 1999; Hays et al. 2021; Conners et al. 2022). The movement patterns of megafauna within protected areas offer valuable insight into their natural movements, habitat requirements and ecological roles, which is particularly important in the context of climate change (Gilmour et al. 2022). Most atolls are predicted to be uninhabitable by the mid-twenty-first century from the combination of sea level rise and wave-driven impacts from increased storm intensity (Storlazzi et al. 2018). Also, extreme temperatures have led to changes in coral reefs, across ocean basins (Tebbett et al. 2023), and have led to large seagrass bed collapses (Shark Bay, Australia; Thomson et al. 2015). Climate change is predicted to cause marine megafauna range shifts by affecting habitat and places of refuge, impacting reproduction and disease, and changing how food sources are distributed (Grose et al. 2020).
Sea turtles are long-lived, migratory megafauna with complex life cycles, having ontogenetic shifts throughout their lifespan and using both pelagic and coastal habitats (Musick and Limpus 1997; Godley et al. 2008). Post-hatchlings spend several years in pelagic waters (Bolten 2003) before recruiting to neritic foraging habitats (Musick and Limpus 1997). Immature turtles can stay in the same foraging grounds for decades (Chaloupka and Limpus 2005) or move between developmental habitats (Meylan et al. 2011). Once sexually mature, they migrate between foraging and breeding grounds, to which they can show high fidelity (Bowen and Karl 2007; Shimada et al. 2020). Site fidelity is the tendency to return to/remain in the same area repeatedly over time. At foraging grounds, site fidelity within their home range could enhance survival, since they are returning to a predictable, familiar environment (Hawkes et al. 2011; Shimada et al. 2020). Fidelity is one way to describe movements and behavior at foraging grounds. Immature turtles are known to display strong site fidelity, which has been described at different spatial scales (Bresette et al. 1998; González Carman et al. 2016) but they also show nomadic (e.g., Luschi and Casale 2014; Baldi et al. 2023) and avoidance behaviors (Cardona et al. 2005; Senko et al. 2010). Understanding spatial distribution patterns at foraging grounds has been identified as a research priority for developing effective management and conservation strategies for immature turtles (Wildermann et al. 2018).
Green turtles (Chelonia mydas) and hawksbill turtles (Eretmochelys imbricata) share the same immature foraging grounds in various parts of the world (e.g., Bjorndal and Bolten 2010; Martins et al. 2020; Stokes et al. 2023; Sanchez et al. 2023). Both species have experienced major population declines and are on the IUCN Red List as Endangered for green turtles and Critically Endangered for hawksbill turtles (Seminoff 2004; Mortimer and Donnelly 2008). While populations are now rebounding in many areas (Allen et al. 2010; Mazaris et al. 2017), the habitats they rely on (such as seagrass beds) are declining globally (Waycott et al. 2009), leading to demographic changes in juvenile aggregations at some foraging grounds (Long et al. 2021; Meylan et al. 2022). Within these foraging aggregations, various behaviors are influenced by factors such as competition (Schofield et al. 2022), prey resources and foraging preferences (Gillis et al. 2018), size (Pillans et al. 2021), predators (Heithaus et al. 2007), human disturbance, and individual differences (Chambault et al. 2020). These factors contribute to smaller-scale movements and migrations, highlighting the intricate dynamics of turtle behavior and their potential response to environmental changes.
Aldabra Atoll, a United Nations Educational, Scientific and Cultural Organization (UNESCO) site in the Western Indian Ocean, hosts substantial foraging aggregations of immature green and hawksbill turtles (Sanchez et al. 2023). The atoll has been protected for over 50 years and is remote, with the closest landmasses being Madagascar, ca. 400 km to the south, and East Africa, 700 km to the west. The atoll is considered an important benchmark site for natural processes because of minimal human impact. A 40-year capture–mark–recapture (CMR) monitoring program has been collecting information on immature turtles at several sites within Aldabra’s lagoon accumulating a long-term and large dataset in the context of turtle CMR studies. We use this dataset to investigate small-scale spatial movement patterns of immature green and hawksbill turtles in Aldabra’s lagoon. Our objectives were to determine: (i) whether turtles show foraging site fidelity at such a small scale; (ii) whether site fidelity is related to turtle size or season; and (iii) movement connectivity among sites within the lagoon.
Materials and methods
Study area and field work
Aldabra Atoll comprises four main islands that surround a protected shallow lagoon (Fig. 1). The lagoon covers 203 km2 and experiences a tidal change up to 3 m (Hamylton et al. 2018). The outside reef has steep seaward slopes (Stoddart et al. 1971). Due to Aldabra’s remote and inaccessible location, it has experienced minimal human impact, and has served as a refuge for species and subspecies with high endemism; it has therefore been recognized as an important site to study ecological and evolutionary processes (UNESCO 2022). Two trade-wind seasons occur at the atoll, with a wetter and warmer northwest monsoon season, and a drier southeast trade-wind season (Stoddart and Mole 1977). The atoll is uninhabited except by personnel of the research station run by a local public trust, the Seychelles Islands Foundation (SIF). A maximum of 20 staff resides at the station, conducting monitoring and research activities and providing research support.
The ten turtle capture sites (A–J) at Aldabra Atoll, Seychelles, for the in-water turtle capture program during 1981–2021. The Seychelles are in the Western Indian Ocean, north of Madagascar (see inset map) and Aldabra is marked by the star, in the southern part of the Seychelles Economic Exclusive Zone (dotted lines; Flanders Marine Institute 2022). Map source: QGIS Development Team (2022)
An in-water sea turtle CMR monitoring program was conducted from July 1981 to March 2021, concentrating on capturing immature turtles within the lagoon, and focusing in the west lagoon, primarily for logistical reasons. Sampling sessions were not standardized spatially or temporally, and turtles were hand-captured either by walking at low tide through partially exposed tidal pools of seagrass/macroalgal mats or at high tide by jumping from a boat onto or next to turtles (Ehrhart and Ogren 1999). High tide captures mostly occurred in sandy sites. A turtle was pursued until caught or another was pursued if the first was not captured. The location was recorded each time a turtle was captured, and capture locations were grouped into ten capture sites, A–J (Fig. 1). The minimum straight-line distance (between polygon edges) between the ten capture sites at Aldabra, calculated in QGIS, was ca. 0.35–25 km. Most sites, however, were 0.4–6 km apart (sites A–I), with only one site in the eastern part of the lagoon (J), 22–25 km from all other sites (Table S1). The approximate capture areas also varied in size (when looking at QGIS polygons) from 0.14 to 0.95 km2: A = 0.14 km2; B = 0.95 km2; C = 0.23 km2; D = 0.68 km2; E = 0.32 km2; F = 0.17 km2; G = 0.73 km2; H = 0.58 km2; I = 0.23 km2; J = 0.22 km2 (Table S1). Turtles were released immediately after tagging for the most part in the same area they were captured. If a more sheltered area was needed to tag and measure the turtles, turtles were then released there. These are reported in the results.
Curved carapace length notch to tip (CCLn-t) and minimum curved carapace length (CCLmin; Bolten 1999) were measured, and the weight of animals were measured with a spring balance (Bolten 1999). The first time a turtle was captured, two flipper tags were applied, one in each front flipper (Balazs 1999). Each time a turtle was recaptured, its tag numbers were recorded and missing tags replaced with new ones. From the 1980s to 1995, Monel (National Band & Tag Co. styles 681, 49) and plastic yellow roto-tags (Dalton Supplies Ltd.) were used. From mid-1995 forward, Inconel tags (National Band & Tag Co. style 681) were used on smaller turtles while larger turtles (> 15 kg before 2020 and > 10 kg after 2020) received titanium tags (Aust. ‘turtle’ titanium tag/Stockbrands Co.). In cases where CCLn-t was not recorded and CCLmin was, CCLmin was converted using Aldabra specific equations (see Sanchez et al. 2023).
Capture rate at a site is here defined as the number of turtles caught per site divided by the total number of capture sessions at that site. A ‘capture session’ is defined as a visit to a site that resulted in at least one turtle capture. If more than one site was visited in the same day, each visit was counted as a separate session. The monitoring program did not record if a capture session occurred and no turtles were captured, and it did not record the start or end time when searching for turtles at a site. Although a session with no captures did not happen often, it is known to have occurred at least a handful of times in the history of the program. Therefore, capture rate will be an underestimation of effort. To take into account this limitation, the following analyses are designed not to be affected by different site features.
Data analysis
Site fidelity
All analyses were performed separately for each turtle species in R (v4.2.1; R Core Team 2021) using RStudio (v1.2.5; Posit team 2022). Generalized linear models (GLMs) and generalized linear mixed models (GLMMs) were fit using R package “glmmTMB” (Brooks et al. 2017). The family of response residuals was assessed with the “check_distribution” and “check_overdispersion” functions from the “performance” package (Lüdecke et al. 2021). Goodness-of-fit tests on scaled residuals were evaluated through the “DHARMa” package (Hartig 2022) by plotting function “simulateResiduals” and with functions “testUniformity” and “testOutliers”. Model assumptions were evaluated through QQ plot residual tests, the within-group deviation from uniformity, the Levene test for homogeneity of variance and dispersion tests from those functions. Each of these evaluations have built-in significance testing. Models were evaluated as a good fit if all tests were not significant.
Site fidelity was investigated using three approaches. Since effort could not be incorporated into models, the three approaches were designed to not be affected by this limitation. First, we checked if turtle size affects the probability of being captured multiple times, with the following GLMM: glmmTMB(cbind(main, others) ~ sizegroup + (1 | site), family = binomial(), where: main is the number of times captured in the site of most captures within each individuals’ recapture history, others is the number of times captured at all other sites, sizegroup is larger or smaller size category, and site is the site of most captures of the turtle, set as a random factor to account for heterogeneity in the sites. The variable sizegroup was a factor with two levels (small and large) with a cutoff of 50 cm CCLn-t. The cutoff was based on the mean size of all captured turtles (48.0 and 47.1 cm CCLn-t for green turtles and hawksbill turtles, respectively). In support of the 50-cm CCLn-t threshold, growth rates calculated from the same long-term dataset in a different study found that the 10-cm size classes of 50–60 and 60–70 cm CCLn-t size classes were the highest (Sanchez et al. 2023); thereby, the cutoff at 50-cm kept those similar growth rates grouped together. As no significant relationships were found in this study between size and site fidelity (see Results), size was not further considered.
In the second approach, site fidelity was defined in relation to a site, assessing whether turtles frequent a certain site more than would be expected by perfect nomadism among sites. In the latter case, the following relation is expected:
where Nx is the number of individual turtles originally tagged at each site (e.g., Na, Nb, Nc, etc.), T is their sum (total number of turtles tagged at all sites), and nxX is the number of turtles originally tagged at that site that have been recaptured at the same site (e.g., naA, nbB, etc.). By comparing Nx/T to nxX/Nx, we removed the effect of site-specific capture effort. In other words, for Ho (random mix), we expected the proportion of turtles recaptured at the same site of tagging to be equal to the overall proportion of individual turtles tagged at that site. On the contrary, for H1 (non-random mix), we expected this proportion to be greater than the overall proportion if turtles are showing site fidelity (e.g., naA/Na > Na/T), or less in the case of avoidance (i.e., the opposite of fidelity). These terms are here used just to describe these two opposite cases and do not necessarily imply a specific behavior of the turtle. A Fisher’s exact test was used to test these hypotheses.
In case of site fidelity, the turtle may settle at a certain site by chance or because of specific site features. In the first case, fidelity (F) to a certain site x, defined as \(Fx=\frac{nxX}{Nx}\), is expected to be similar in all sites (i.e., Fa = Fb = Fc…Fj), while in the second case F is expected to differ among sites. To assess which is the most likely case, a Fisher’s pairwise test (with Bonferroni correction) from R package “rstatix” (Kassambara 2023) was used.
In the third approach, site fidelity was investigated in relation to seasonality. We assessed the number of times each individual was caught and the number of different capture sites within its capture history (e.g., DD = 1 site, FDF = 2 sites). For each capture history, individuals were categorized as captured only during the northwest monsoon season (NW), only the southeast trade-wind season (SE) or a mixture of the two. For green turtles, if a turtle was recaptured multiple times, each subsequent recapture pair was considered a separate event (i.e., for capture history FDF, recapture pairs are FD and DF). Any event without a known capture location was excluded from this analysis. For each capture–recapture event, we determined if both the capture and recapture were from the same season or from different seasons. The relationship between site fidelity events—whether a turtle is captured and then recaptured at the same site or not—and season was explored with GLMMs. For green turtles, the model glmmTMB(Capture_event_fidelity ~ season + (1|TurtleID), family = binomial) was used, with site fidelity (1 = capture–recapture in the same location; 0 = capture–recapture in different locations) as the response and season (1 = capture–recapture in the same season; 2 = in different seasons) as the predictor variable. Using proportions for site and season removed the effect of site-specific capture effort. Turtle ID was used as a random effect to account for multiple capture–recapture events from the same individual. For hawksbill turtles, there were fewer individuals and more observations than for green turtles, and the model failed to converge. Therefore, the model was further simplified by only including the first capture event of each individual, instead of the whole capture history, with the following model: glmmTMB(Capture_event_fidelity ~ season, family = binomial).
Connectivity
Since a connectivity analysis is affected by capture effort but it cannot be accounted for in this study, the following analysis aimed to provide qualitative information on the connectivity among sites rather than quantitative estimates. To look at patterns of connectivity between sites, a network analysis was performed using R packages “igraph” (for network analysis; Csardi and Nepusz 2006) and “ggraph” (for network representation; Pedersen 2022). Due to the structure of the capture program (effort unevenly spread across sites) and being unable to account for imperfect or heterogeneous detection (Gimenez et al. 2019), only very simple analyses were applied. Capture events were analyzed as non-directional, and QGIS (QGIS Development Team 2022) was used for mapping. Centroids were created for each site polygon in QGIS. The centroids/nodes represented spatial regions (the capture sites), the edges/links represented the movement between sites, and loops represented a capture and recapture at the same site. Edges were not representative of actual paths taken by turtles but instead represent the connection between two nodes with a straight line (distances among sites are provided in Table S1).
Node size was proportional to the number of times a capture event occurred at that site, including capture events where the capture–recapture occurred at the same site (loops). ‘Edge width’ represented the magnitude of capture events between nodes. Centrality measures of degree, eigenvector, and betweenness, were calculated for each node to identify more common sites or relative importance of sites from the movement network, using the “igraph” package (Csardi and Nepusz 2006) through functions “centr_degree”, “centr_eigen”, and “centr_betw”. They were set as non-directional and normalized. ‘Degree’ represents the number of nodes a node is connected to; ‘eigenvector’ is similar to degree but takes into account the centrality of the nodes it is connected to, thereby giving a further indication of which nodes have influence over the whole network. ‘Betweenness’ shows the importance of a node based on how important the other nodes are that it is in direct contact with, thereby showing which nodes influence the network flow. Node and edge network values were visualized spatially through QGIS. R package “ggraph” (Pedersen 2022) was used to create arc diagrams with function “geom_edge_arc”. Self-loops were included through function “geom_edge_loop.” It automatically multiplies them by two (for the capture and recapture; Pedersen 2022).
Benthic composition of sites
The lagoon habitat map (Hamylton et al. 2018) indicated eight categories of habitat for the sites: bare carbonate sand, fine silts with organic content, dense macroalgal mat on sand, sparse macroalgae and seagrass assemblage on sand, seagrass beds, coral patches, bedrock, and coral rubble. Benthic composition was explored by spatially overlaying the polygon representing each site onto the benthic habitat map (Hamylton et al. 2018) to assess whether sites with fidelity had consistent benthic habitat compositions. The proportion area of each habitat category was calculated per site using QGIS.
Results
A total of 1158 green turtles and 514 hawksbill turtles (immature individuals) were tagged on Aldabra as part of the in-water CMR program from 1981 to 2021, with 236 (20.3%) green turtles and 276 (53.7%) hawksbill turtles recaptured at least once (Table 1). Recaptured individuals were re-caught from one to nine times, and the number of captures per site was 8–565 for green turtles and 4–273 for hawksbill turtles.
The mean carapace length (CCLn-t) of captured green turtles was 48.6 ± 8.1 SD cm (range 35.4‒74.5, n = 163), and hawksbill turtles 46.1 ± 11.0 SD cm (range 30.1‒80.5, n = 146). Since 96% (n = 1342) of the green turtle captures and 97% (n = 773) of the hawksbill turtle captures were released in the same location as capture, these were not analyzed in a separate analysis.
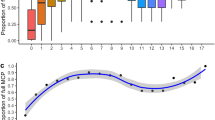
Overall, the number of capture sessions per site ranged from two (site I) to 154 (site G) for green turtles and two (site E) to 87 (site G) for hawksbill turtles. Capture sessions varied per season, with 2–39 and 1–115 sessions for green turtles in the NW and SE seasons, respectively, and 2–29 and 2–63 for hawksbills in the NW and SE seasons, respectively (Table S2). Our site-specific capture rate (number of turtles caught per site divided by the total number of sessions at that same site) was highest at site E (4.5 ± 2.1 SD captures/session) followed by site F (4.26 ± 2.6 SD captures/session) for green turtles, and highest for site B (3.81 ± 3.2 SD captures/session) followed by site G (3.14 ± 2.6 SD captures/session) for hawksbill turtles (Table S3, Figs. S1, S2).
Site fidelity
Assessing site fidelity with the first approach using GLMs, all model evaluations listed in the methods were insignificant (p > 0.05) and therefore were considered to meet all underlying assumptions. Size did not affect the probability of recapture for green turtles (estimate = − 0.00, std. error = 0.40, z = − 0.001, p = 0.99; n = 168) or for hawksbill turtles (estimate = − 0.59, std error = 0.78, z = − 0.76, p = 0.45; n = 156). The random effect of site indicated some variability for both species (variance = 1.92, 4.85; std. dev = 1.39, 2.20, respectively, for green and hawksbill turtles). Since size group was not significant for both species, all sizes were pooled together in the other analyses.
The second approach, of assessing whether turtles frequent a certain site more than would be expected by random selection, suggested that for green turtles, three (C, G, H) of the ten capture sites had a lower ratio of recaptures than the overall ratio (Table 2), indicating avoidance. When comparing these three sites with each other, they had different proportions of recaptures (p < 0.001) suggesting that settling probability differs by site, with significant differences between C–G (n = 649, p < 0.001) and G–H (n = 737, p < 0.01). For hawksbills, two (G, J) of the ten capture sites had higher proportions than the overall proportion (Table 2), indicating site fidelity. However, their proportions of recaptures did not differ from each other, suggesting that turtles settle there by chance.
The third approach, looking at the effect of season on site fidelity, indicated that individuals tended to be recaptured at only one site for both green turtles and hawksbill turtles. No individual capture history included more than three sites. For green turtles, 45% of the individuals had capture histories within the SE season and 45% had capture histories within both seasons. Hawksbill turtles were similar for the SE and both seasons (40% and 56%, respectively). Both species had high proportions of recaptures at one site (65% for green turtles, 90% for hawksbill turtles; Fig. S3, Table S4). Additionally, 68% and 65% of the green turtle and hawksbill turtle capture sessions, respectively, were in the SE.
When determining whether capture–recapture events occurred in the same season or not, for green turtles, same/different season was significant (GLMM, n = 236; estimate = − 0.80, std. error = 0.35, z = − 2.31, p = 0.02). This suggests that the season in which the capture–recapture occurs has a significant effect on the likelihood of being recaptured in the same location. The negative coefficient for season (− 0.80) implies that turtles are less likely to be recaptured in the same location in different seasons (n = 87 events) compared to the same season (n = 149 events: southeast season = 121, northwest season = 28). The random effect of TurtleID accounted for some of the variability (variance = 0.84, std. dev = 0.91; n = 168). For hawksbill turtles, when using the original capture–recapture event of each individual, the same/different season of capture–recapture did not affect the capture event fidelity (GLM, n = 148; estimate = 0.01, std. error = 0.65, z = 0.01, p = 0.99). For both species, all model evaluations were insignificant (p > 0.05) and therefore were considered to meet the underlying assumptions.
Connectivity
Capture–recapture events at the same site (e.g., AA, BB, CC, etc.) were most common for site G for green turtles and sites G and J for hawksbill turtles (Fig. 2; Tables S5 and S6). Green turtles had the highest ‘degree’ of centrality at site G (score = 8), followed by sites D and F (score 6), which was reinforced by the highest ‘eigenvector’ centrality scores at sites G (1), D (0.87), and F (0.81). These results suggest sites G, D, and F had the most influence over the network in terms of events. For ‘betweenness’ centrality (the highest influence on the network for captures), site H had the highest score (11) followed by G (1.5; Table S7).
a, c Capture–recapture event movement network for spatial representation and b, d arc diagram to show capture–recapture in the same location (loops) of a, b green turtle and c, d hawksbill turtle events at the different capture sites (sites A–J) over the long-term dataset from Jul 1981 to Mar 2021. Node strength is represented by the node size, based on the number of times a turtle was captured in that site. Edges are non-directional and weighted based on capture event frequency. Capture effort and size of capture site were not included in the analysis
Hawksbill turtles had the highest ‘degree’ centrality at site B (score = 6) followed by G (score = 5), and ‘eigenvector’ scores were highest in B (1.0) followed by G (0.88) and H (0.72). In combination, these suggest that sites B and G had the most influence over the network for capture events. ‘Betweenness’ centrality was highest for site B (score 5.5) followed by J (1.0; Table S7).
Site benthic composition
The recapture sites varied in their habitat composition, including six to eight benthic categories of the lagoon habitat map (Fig. 3). Only sites B and J had the category ‘coral rubble’ (0.2% and 6.5%, respectively).
Percent of the habitat categories (see Hamylton et al. 2018) found at each site (B–J). Site A was not covered in the habitat map and is therefore not included here. Along the top, white ( +) for hawksbill turtles and black ( +) for green turtles indicate the sites with the highest degree centrality scores. However, capture effort and size of capture site were not factored into the centrality scores
Discussion
Within the context of a foraging area with minimal human impacts, we examined site fidelity of two sympatric sea turtle species at a fine scale (most sites < 6 km apart, and one site 22–26 km from the others). Low site fidelity might have indicated that the scale examined was too small relative to the mobility of these species; however, we found both significant site fidelity and avoidance at this scale.
Site fidelity and connectivity
In the Aldabra lagoon, most individuals of both species were recaptured at sites different from their original capture locations. Site fidelity was observed at two sites for hawksbill turtles and three sites for green turtles; however, the two species behaved differently. Green turtles showed an avoidance and hawksbills a significant site fidelity. Other studies assessing fine-scale movements using CMR mostly recapture turtles at the same sites (although they use different scales to define sites). In Bermuda, for example, nearly 90% of juvenile green turtle recaptures were at the same site as original capture, and turtles that switched sites traveled from nearly 0.2 km to over 2.0 km to their next site (Meylan et al. 2022). A 9-year study in Martinique, with ten capture sites 0.4–40 km apart, found a ca. 90% probability of recapturing a juvenile green turtle in original capture site (Siegwalt et al. 2020). The data from our study suggest there may be less fidelity at Aldabra or home ranges are larger, leading to lower rates of recapture.
Spatial movements of juvenile green and hawksbill turtles have been explored through active (Van Dam and Diez 1998) and passive acoustic telemetry (Selby et al. 2019; Pillans et al. 2021) and satellite tracking (Wood et al. 2017; Chambault et al. 2020). Comparing results from these studies is challenging because of differences between regions, sites, sample sizes, study design, and analyses, but they do provide insight to the species. Home ranges for immature green turtles and hawksbill turtles have been estimated to be on average < 3 km2 at sites in the Atlantic and Caribbean (Makowski et al. 2006; Chevis et al. 2017; Griffin et al. 2020; Matley et al. 2021). Within these home ranges, high-use areas can vary in size (Florida; Makowski et al. 2006) with larger space use (> 16 km2) presumably due to food resources being widely distributed (Gulf of California; Seminoff et al. 2002). For hawksbills, studies in the Caribbean have shown home ranges to range from 0.07–2 km for immature hawksbill turtles (Van Dam and Diez 1998; Blumenthal et al. 2009). Highly variable individual movements have been observed with some turtles using a single site and others using multiple sites, with no clear pattern (Chevis et al. 2017).
We predicted that green turtles’ avoidance behavior at three sites in the west lagoon was likely due to site features, but this does not seem to have been the case. Two sites (C and H) had similar proportions of sand, macroalgae, and seagrass, with differences in coral patch coverage (20% and < 1% for C and H, respectively), while site G was quite different, comprised predominantly of sand. This seems to suggest that green turtles were avoiding these sites for a reason other than the benthic habitat composition. Predation risk, availability of resting sites, turtle size, competition, and prey availability can influence movement (further discussed below). Additionally, C and H are areas where opportunistic dugong sightings have been recorded (Appoo et al. 2019). Green turtle avoidance at these sites may reduce competition with dugongs for similar foraging resources, since dugongs mostly rely on seagrasses or algae (when seagrass is scarce) (Marsh et al. 1982; Wirsing et al. 2022). Although these additional factors were not possible to analyze with the CMR data, they point the way for future investigations at the atoll. Conversely, hawksbill turtles had strong site fidelity to two sites (G, J), suggesting that settling (indicated by showing strong site fidelity/high site recaptures), at these sites which may happen by chance or by factors not accounted for here. Of all the sites, sites G (west lagoon) and J (east lagoon) differed the most in terms of their benthic habitat composition, with site G dominated by sand, while J was mainly seagrass and coral, suggesting that these sites are used for different purposes. At Lighthouse Reef Atoll in Belize, which also has a shallow, protected lagoon, habitat type and location (forereef vs. lagoon) correlated with juvenile hawksbill turtle abundance, with fewer hawksbills captured in the lagoon. The lagoon was probably a transit area between foraging sites (Scales et al. 2011). It is possible this is also the case for the Aldabra hawksbill turtles. Benthic complexity, such as coral and ledges, is known to be important for hawksbill habitat use (Witt et al. 2010; Selby et al. 2019), but hawksbills also occur in seagrass beds and sandy areas (Bjorndal and Bolten 2010; Selby et al. 2019), and utilize mangrove estuaries and shallow rocky reefs near mangroves as foraging grounds (Gaos et al. 2012; Martinez-Estevez et al. 2021). Habitat availability and use needs to be more closely assessed to understand how both species are using these sites.
Connectivity between sites was explored using capture–recapture events (not individuals) with a network analysis. We could not account for capture effort in the connectivity analysis; therefore, our results provide qualitative information on the connectivity among sites. Size of capture sites was also not included in the analysis, which may also affect recapture of individuals. The sites of highest importance for immature green turtles were G, D, and F, and for immature hawksbill turtles, sites B, G, and J (Fig. 2). The shared importance of site G was also seen in site fidelity among individuals. Additionally, all green turtle capture events occurred in the west of the lagoon, while hawksbill turtles showed connectivity between the west and east sides of the lagoon. High fidelity and wider connectivity within the lagoon was also found for hawksbill turtles at Lighthouse Reef Atoll in Belize (Chevis et al. 2017). The connectivity of both species between the sites also shows their overlap in habitat use. Niche partitioning has been seen where green and hawksbill turtles co-occur, via foraging in different microhabitats (Martins et al. 2020). A decrease in food quantity or quality can result in a lower site fidelity due to an increased foraging effort (Meylan et al. 2022), and turtles in urbanized coastal areas with pollution and habitat loss may consume their less preferred prey compared to when in protected environments with less human impact and a greater diversity of prey (Santos et al. 2011; Martins et al. 2020). As Aldabra has been protected for decades and experiences minimal human impact, direct anthropogenic disturbance is unlikely to have reduced availability of prey items, but increases in the size of the foraging aggregations in response to conservation efforts in the region might result in competition for prey items and/or changes in the turtle aggregation composition. Over-grazing by recovering foraging green turtle aggregations has been documented in places such as Lakshadweep, India, and Bermuda, USA (Gangal et al. 2021; Meylan et al. 2022). Similarly, models of recovering whale populations indicate that reduced krill biomass caused by climate change will lead to competition for food and result in population declines (Tulloch et al. 2019). Also, in Great Britain, gray seals are possibly contributing to harbor seal population declines in areas of overlap through prey competition (Wilson and Hammond 2019).
Movement patterns vary in other regions, similar to our findings at Aldabra. In Belize, juvenile hawksbill turtle movements followed three patterns: ‘residents’ spent 60–100% of their time at a single location; ‘sequential residents’ spent several weeks/months at a site (with small exploratory movements) before moving to a different one for weeks/months (i.e., showing strong site fidelity); and ‘transients’ did not stay at any site for an extended period (Chevis et al. 2017). Green turtles off the coast of Brazil (juveniles and adults) showed three similar patterns, with turtles foraging on macroalgae having larger ranges (Godley et al. 2003).
The fact that movements within the west lagoon were recorded for both species plus transits of hawksbill turtles across the lagoon may be explained either by high numbers of untagged turtles resulting in low recapture rates, or turtle movements that are influenced by environmental factors we have not studied. For example, diel movements have been found to influence juvenile turtle movements (Makowski et al. 2006; Hart et al. 2016; Selby et al. 2019; Matley et al. 2020). At St. Croix, in the United States Virgin Islands, green turtles were found using both lagoon and reef habitat at night (Griffin et al. 2020), while in the nearby Culebra and Culebrita islands, Puerto Rico, green turtles mostly remained within the lagoon at night (Griffin et al. 2019). Both locations are in MPAs. In the Cayman Islands, turtles used the lagoon during the day and moved to the outside reef at night (Blumenthal et al. 2010). Chambault et al. (2020) investigated diel patterns and daily movement drivers for juvenile green turtles in five sites in the southwest Indian Ocean and found strong inter-individual and inter-site differences. At Europa, which has a semi-enclosed lagoon, like Aldabra, most tracked turtles remained in the mangroves, and had the same high-use areas regardless of day or night (Chambault et al. 2020). Strong tidal areas may not have the same diurnal–nocturnal turtle movements as seen in areas without strong tides, with turtles using the tides to access habitats (Senko et al. 2010). This may be a likely scenario for Aldabra turtles.
Turtle size has been shown to affect turtle distribution at other locations. In the atoll-like system of the Dry Tortugas, FL, larger green turtles rested at night in deeper water while smaller turtles rested in shallow water < 1 m deep (Hart et al. 2016). Likewise, larger turtles used deeper, open waters, and smaller immature turtles used shallow protected bays in the Florida Keys and Guinea Bissau (Welsh and Mansfield 2022; Madeira et al. 2022). In north-western Australia, there was spatial segregation between foraging juvenile, sub-adult and adult green turtles’ life stages: larger turtles were found in areas of the lagoon that were more open and smaller turtles were closer to the shoreline. The shift from shoreline to further offshore within the lagoon increased the range of area used and capture habitats as turtles matured (Pillans et al. 2022). In our study, we found no relationship between turtle size and probability of being recaptured.
Season can also affect distribution. For green turtles at Aldabra, we recaptured a higher proportion of turtles in the same location when the initial capture event occurred during the same season. As Aldabra is tropical, seasonal migrations seen in temperate latitudes (e.g., Williard et al. 2017) are not expected. A possible explanation for our results for green turtles may be sampling or site bias. The northwest season is calmer with clearer water, making it easier to pursue turtles further from land. The southeast season is windier and the water more turbid; turtles are pursued closer to land and in more sheltered areas. Potential bias could be reduced by recording more information during capture sessions, including recording the number of turtles seen for each species and if no turtles were captured during a session at a site.
Dugongs and green turtles rely on similar prey; therefore, competition between them has been investigated (reviewed in Wirsing et al. 2022). In the Torres Strait and Shoalwater Bay (Australia), there was a wide spatial overlap between the two species with a difference between their core use areas (Gredzens et al. 2014). Studies have suggested that the two species partition resources by foraging at different water depths (e.g., Chilvers et al. 2004; Hazel et al. 2009). It has also been suggested that turtles minimize competition with dugongs by adjusting their diet (André et al. 2005), since in places where both turtles and dugongs occur, turtles have been found to include ctenophores, jellyfish, sponges and/or macroalgae (Heithaus et al. 2002; André et al. 2005). When both species are consuming seagrasses, their differences in foraging strategies may also reduce competition (Wirsing et al. 2022). To understand the dynamics between dugongs and green turtles in the Aldabra lagoon, further investigations into foraging ecology of both species would need to be conducted.
Finally, predation and social interactions influence animal movement. For example, green turtles show altered habitat choice with increased tiger shark predation risk (Heithaus et al. 2007). Tiger sharks (Galeocerdo curvier) are common around Aldabra (Stevens 1984, SIF unpubl.) and are known to prey on green turtles there (Frazier 1971). Although very little is known about tiger shark distribution and movement around the atoll, the spatio-temporal distribution of the two turtle species could be affected by such predators.
Limitations and recommendations
Our study uses a simple approach to investigate fine-scale site fidelity with CMR, by evaluating site-specific ratios compared to overall, expected ratios for each site. We combine those results with other methods (network analysis, GLM/GLMMs) to maximize the information content from this long-term dataset. CMR is a low-cost method which can provide long-term views on site fidelity and connectivity, which can otherwise be difficult to obtain (Godley et al. 2003) and has contributed valuable long-term information on turtle ecology (e.g., Meylan et al. 2022). The disadvantages of CMR include not knowing extent of occupancy and movement paths in between captures and extracting information from low recapture rates. Low recapture rates, common in other CMR studies (e.g., Long et al. 2021), could be due to turtles having large home ranges with multiple foraging areas, being transient to the area, or having tagged too few turtles to produce a meaningful recapture rate. Low recapture rates can also be species-specific: immature green turtles are more abundant at Aldabra than immature hawksbills, but they swim faster than hawksbills, making green turtles more difficult to capture.
Although we attempted to standardize the data, the number of captures will still be affected by capture effort, but unfortunately sessions with no captures were not recorded. Sampling sessions where no turtles were caught are known to have occurred in the history of the program, and although it is thought to be a small number, there is no way of calculating the number of sessions that ended in zero captures. In the future, and for other similar programs, GPS coordinates and searching effort should be recorded for every session (including sessions with no captures) to improve information on spatial use and density. Since GPS coordinates were not available, capture site areas were conservatively estimated, likely over-estimating the size of the capture areas. Sites ranged in size from 0.14 to 0.95 km2. When looking at capture site size and network importance for green turtles, sites G, D, and F had the highest ‘degree’ centrality scores. G (0.73 km2) and D (0.68 km2) had two of the largest estimated capture areas, while F (0.17 km2) had one of the smallest capture areas. For hawksbill turtles, sites B and H had the highest ‘degree’ centrality scores: B had the largest area of all the sites (0.95 km2), while H (0.58 km2) was near the middle. While results of analyses indicated that even the smaller sites were important, it is possible that sites covering larger areas could have a bias on a turtle being recaptured. Capture effort at the different sites will also have a bias on recaptures. The connectivity analysis would have been affected by capture effort and possibly by the size of the capture areas, therefore it aimed to provide qualitative information on the connectivity among sites.
Small-scale studies on the spatial ecology of turtles could be strengthened, in conjunction with CMR programs, with more precise but shorter-term methods such as acoustic (Chevis et al. 2017; Griffin et al. 2020) or satellite tracking (Shimada et al. 2020; Siegwalt et al. 2020) to define core foraging areas and spatial movement. These methods would work well in the enclosed lagoon of Aldabra and would substantially improve the quality of the information from such studies.
Conclusions
Our fine-scale analysis of immature turtle recaptures in a relatively undisturbed environment has provided insight into their distribution and site fidelity at Aldabra. Both green turtles and hawksbill turtles use the same sites; however, we show opposing site-fidelity behavior in the two species: avoidance for green turtles and high site fidelity for hawksbill turtles. Such findings have implications for the scale of conservation measures and can inform human use, e.g., areas where tourism could be increased or should be avoided.
With Aldabra’s turtle population increasing (Pritchard et al. 2022), resources may eventually become limited (e.g., through increased competition or habitat degradation; see Meylan et al. 2022). The current high site fidelity of hawksbill turtles at a few sites at Aldabra suggests that these turtles do not have to move far to find food. Increasing foraging efforts could, however, lead to reduced site fidelity. Aldabra is an exemplary location to study how turtles respond to increasing turtle populations and climate change. Follow-on research to improve our understanding of sea turtle ecology and habitat use at Aldabra could include fine-scale investigation of resource partitioning and the impact of environmental factors, such as tides, on their movement and behavior through methods such as acoustic or satellite telemetry (e.g., Selby et al. 2019; Chambault et al. 2020), and/or acceleration data loggers (Hart et al. 2016). Our findings provide a better understanding of the ecology and habitat use of immature sea turtles. This will help to guide conservation efforts at different geographic scales, and provide insight into how turtles might adapt to environmental change.
Data availability
The datasets analyzed from the current study are available from the corresponding author upon reasonable request.
References
Allen Z, Shah N, Grant A, Derand G, Bell D (2010) Hawksbill turtle monitoring in Cousin Island Special Reserve, Seychelles: an eight-fold increase in annual nesting numbers. Endanger Species Res 11:195–200. https://doi.org/10.3354/esr00281
André J, Gyuris E, Lawler IR (2005) Comparison of the diets of sympatric dugongs and green turtles on the Orman Reefs, Torres Strait, Australia. Wildl Res 32:53. https://doi.org/10.1071/WR04015
Appoo J, Sanchez C, Burt AJ, Richards H, van de Crommenacker J, Currie J, Fleischer-Dogley F (2019) The use of Aldabra and its protected waters by marine mammals. Seychelles Res J 1:166–179
Atwood TB, Connolly RM, Ritchie EG, Lovelock CE, Heithaus MR, Hays GC, Fourqurean JW, Macreadie PI (2015) Predators help protect carbon stocks in blue carbon ecosystems. Nat Clim Chang 5:1038–1045. https://doi.org/10.1038/nclimate2763
Balazs GH (1999) Factors to consider in the tagging of sea turtles. In: Eckert KL, Bjorndal KA, Abreu-Grobois A, Donnelly M (eds) Research and management techniques for the conservation of sea turtles. IUCN/SSC Marine Turtle Specialist Group, Washington, pp 101–109
Baldi G, Furii G, Del Vecchio M, Salvemini P, Vallini C, Angelini V, Pari S, Lombardi Moraes K, Profico C, Olivieri V, Margaritoulis D, Rees AF, Çurri A, Hochscheid S, Freggi D, Lazar B, Luschi P, Casale P (2023) Behavioural plasticity in the use of a neritic foraging area by loggerhead sea turtles: insights from 37 years of capture–mark–recapture in the Adriatic Sea (Mediterranean Sea). ICES J Mar Sci 80:210–217. https://doi.org/10.1093/icesjms/fsac227
Bjorndal KA, Bolten AB (2010) Hawksbill sea turtles in seagrass pastures: success in a peripheral habitat. Mar Biol 157:135–145. https://doi.org/10.1007/s00227-009-1304-0
Blumenthal JM, Austin TJ, Bell CDL, Bothwell JB, Broderick AC, Ebanks-Petrie G, Gibb JA, Luke KE, Olynik JR, Orr MF, Solomon JL, Godley BJ (2009) Ecology of hawksbill turtles, Eretmochelys imbricata, on a Western Caribbean foraging ground. Chelonian Conserv Biol 8:1–10. https://doi.org/10.2744/CCB-0758.1
Blumenthal JM, Austin TJ, Bothwell JB, Broderick AC, Ebanks-Petrie G, Olynik JR, Orr MF, Solomon JL, Witt MJ, Godley BJ (2010) Life in (and out of) the lagoon: fine-scale movements of green turtles tracked using time-depth recorders. Aquat Biol 9:113–121. https://doi.org/10.3354/ab00222
Bolten AB (1999) Techniques for measuring sea turtles. In: Eckert KL, Bjorndal KA, Abreu-Grobois FA, Donnelly M (eds) Res Manag Tech Conserv Sea Turtles IUCN/SSC Mar Turt Spec Gr Publ No 4 1–5
Bolten AB (2003) Life history patterns of sea turtles: consequences of an oceanic juvenile stage. In: Lutz PL, Musick J, Wyneken J (eds) The biology of sea turtles. CRC Press, Boca Raton, FL, pp 243–257
Bouchard SS, Bjorndal KA (2000) Sea turtles as biological transporters of nutrients and energy from marine to terrestrial ecosystems. Ecology 81:2305. https://doi.org/10.2307/177116
Bowen BW, Karl SA (2007) Population genetics and phylogeography of sea turtles. Mol Ecol 16:4886–4907. https://doi.org/10.1111/j.1365-294X.2007.03542.x
Bresette M, Gorham J, Peery B (1998) Site fidelity and size frequencies of juvenile green turtles (Chelonia mydas) utilizing near shore reefs in St. Lucie County, Florida. In: Mar. Turt. Newsl. http://www.seaturtle.org/mtn/archives/mtn82/mtn82p5.shtml?nocount. Accessed 12 May 2023
Brooks ME, Kristensen K, Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Mächler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400. https://doi.org/10.32614/RJ-2017-066
Burkholder DA, Heithaus MR, Fourqurean JW, Wirsing A, Dill LM (2013) Patterns of top-down control in a seagrass ecosystem: could a roving apex predator induce a behaviour-mediated trophic cascade? J Anim Ecol 82:1192–1202. https://doi.org/10.1111/1365-2656.12097
Cardona L, Revelles M, Carreras C, San Félix M, Gazo M, Aguilar A (2005) Western Mediterranean immature loggerhead turtles: habitat use in spring and summer assessed through satellite tracking and aerial surveys. Mar Biol 147:583–591. https://doi.org/10.1007/s00227-005-1578-9
Chaloupka M, Limpus C (2005) Estimates of sex- and age-class-specific survival probabilities for a southern Great Barrier Reef green sea turtle population. Mar Biol 146:1251–1261. https://doi.org/10.1007/s00227-004-1512-6
Chambault P, Dalleau M, Nicet JB, Mouquet P, Ballorain K, Jean C, Ciccione S, Bourjea J (2020) Contrasted habitats and individual plasticity drive the fine scale movements of juvenile green turtles in coastal ecosystems. Mov Ecol 8:1–15. https://doi.org/10.1186/s40462-019-0184-2
Chevis MG, Godley BJ, Lewis JP, Lewis JJ, Scales KL, Graham RT (2017) Movement patterns of juvenile hawksbill turtles Eretmochelys imbricata at a Caribbean coral atoll: long-term tracking using passive acoustic telemetry. Endanger Species Res 32:309–319. https://doi.org/10.3354/esr00812
Chilvers BL, Delean S, Gales NJ, Holley DK, Lawler IR, Marsh H, Preen AR (2004) Diving behaviour of dugongs, Dugong dugon. J Exp Mar Bio Ecol 304:203–224. https://doi.org/10.1016/j.jembe.2003.12.010
Conners MG, Sisson NB, Agamboue PD, Atkinson PW, Baylis AMM, Benson SR, Block BA, Bograd SJ, Bordino P, Bowen WD, Brickle P, Bruno IM, González Carman V, Champagne CD, Crocker DE, Costa DP, Dawson TM, Deguchi T, Dewar H, Doherty PD, Eguchi T, Formia A, Godley BJ, Graham RT, Gredzens C, Hart KM, Hawkes LA, Henderson S, Henry RW, Hückstädt LA, Irvine LM, Kienle SS, Kuhn CE, Lidgard D, Loredo SA, Mate BR, Metcalfe K, Nzegoue J, KouereyOliwina CK, Orben RA, Ozaki K, Parnell R, Pike EP, Robinson PW, Rosenbaum HC, Sato F, Shaffer SA, Shaver DJ, Simmons SE, Smith BJ, Sounguet GP, Suryan RM, Thompson DR, Tierney M, Tilley D, Young HS, Warwick-Evans V, Weise MJ, Wells RS, Wilkinson BP, Witt MJ, Maxwell SM (2022) Mismatches in scale between highly mobile marine megafauna and marine protected areas. Front Mar Sci 9:1236. https://doi.org/10.3389/FMARS.2022.897104/BIBTEX
Csardi G, Nepusz T (2006) The igraph software package for complex network research. InterJournal, Complex Syst 1695. https://igraph.org
Ehrhart LM, Ogren LH (1999) Studies in foraging habitats: capturing and handling turtles. In: Research and management techniques for the conservation of sea turtles, vol 4. IUCN/SSC Marine Turtle Specialist Group Publication. pp 61–65
Frazier J (1971) Observations on sea turtles at Aldabra Atoll. Philos Trans R Soc Lond B Biol Sci 260:373–410
Gangal M, Gafoor A-B, D’Souza E, Kelkar N, Karkarey R, Marbà N, Arthur R, Alcoverro T (2021) Sequential overgrazing by green turtles causes archipelago-wide functional extinctions of seagrass meadows. Biol Conserv 260:109195. https://doi.org/10.1016/j.biocon.2021.109195
Gaos A, Lewison R, Wallace B, Yañez I, Liles M, Nichols W, Baquero A, Hasbún C, Vasquez M, Urteaga J, Seminoff J (2012) Spatial ecology of critically endangered hawksbill turtles Eretmochelys imbricata: implications for management and conservation. Mar Ecol Prog Ser 450:181–194. https://doi.org/10.3354/meps09591
Gillis AJ, Ceriani SA, Seminoff JA, Fuentes MMPB (2018) Foraging ecology and diet selection of juvenile green turtles in the Bahamas: insights from stable isotope analysis and prey mapping. Mar Ecol Prog Ser 599:225–238. https://doi.org/10.3354/meps12635
Gilmour ME, Adams J, Block BA, Caselle JE, Friedlander AM, Game ET, Hazen EL, Holmes ND, Lafferty KD, Maxwell SM, McCauley DJ, Oleson EM, Pollock K, Shaffer SA, Wolff NH, Wegmann A (2022) Evaluation of MPA designs that protect highly mobile megafauna now and under climate change scenarios. Glob Ecol Conserv 35:e02070. https://doi.org/10.1016/j.gecco.2022.e02070
Gimenez O, Mansilla L, Klaich MJ, Coscarella MA, Pedraza SN, Crespo EA (2019) Inferring animal social networks with imperfect detection. Ecol Modell 401:69–74. https://doi.org/10.1016/j.ecolmodel.2019.04.001
Godley BJ, Luschi P, Lima EHSM, Akesson S, Broderick AC, Glen F, Godfrey MH, Hays GC (2003) Movement patterns of green turtles in Brazilian coastal waters described by satellite tracking and flipper tagging. Mar Ecol Prog Ser 253:279–288
Godley BJ, Blumenthal JM, Broderick AC, Coyne MS, Godfrey MH, Hawkes LA, Witt MJ (2008) Satellite tracking of sea turtles: where have we been and where do we go next? Endanger Species Res 4:3–22. https://doi.org/10.3354/esr00060
González Carman V, Bruno I, Maxwell S, Álvarez K, Albareda D, Acha EM, Campagna C (2016) Habitat use, site fidelity and conservation opportunities for juvenile loggerhead sea turtles in the Río de la Plata, Argentina. Mar Biol 163:1–13. https://doi.org/10.1007/s00227-015-2795-5
Gredzens C, Marsh H, Fuentes MMPB, Limpus CJ, Shimada T, Hamann M (2014) Satellite tracking of sympatric marine megafauna can inform the biological basis for species co-management. PLoS One 9:e98944. https://doi.org/10.1371/journal.pone.0098944
Griffin LP, Finn JT, Diez C, Danylchuk AJ (2019) Movements, connectivity, and space use of immature green turtles within coastal habitats of the Culebra Archipelago, Puerto Rico: implications for conservation. Endanger Species Res 40:75–90. https://doi.org/10.3354/ESR00976
Griffin LP, Smith BJ, Cherkiss MS, Crowder AG, Pollock CG, Hillis-Starr Z, Danylchuk AJ, Hart KM (2020) Space use and relative habitat selection for immature green turtles within a Caribbean marine protected area. Anim Biotelemetry 8:1–13. https://doi.org/10.1186/s40317-020-00209-9
Grorud-Colvert K, Sullivan-Stack J, Roberts C, Constant V, Costa BHE, Pike EP, Kingston N, Laffoley D, Sala E, Claudet J, Friedlander AM, Gill DA, Lester SE, Day JC, Gonçalves EJ, Ahmadia GN, Rand M, Villagomez A, Ban NC, Gurney GG, Spalding AK, Bennett NJ, Briggs J, Morgan LE, Moffitt R, Deguignet M, Pikitch EK, Darling ES, Jessen S, Hameed SO, Di Carlo G, Guidetti P, Harris JM, Torre J, Kizilkaya Z, Agardy T, Cury P, Shah NJ, Sack K, Cao L, Fernandez M, Lubchenco J (2021) The MPA guide: a framework to achieve global goals for the ocean. Science 373(6560):861. https://doi.org/10.1126/science.abf0861
Grose SO, Pendleton L, Leathers A, Cornish A, Waitai S (2020) Climate change will re-draw the map for marine megafauna and the people who depend on them. Front Mar Sci 7:1–13. https://doi.org/10.3389/fmars.2020.00547
Halpern BS, Frazier M, Potapenko J, Casey KS, Koenig K, Longo C, Lowndes JS, Rockwood RC, Selig ER, Selkoe KA, Walbridge S (2015) Spatial and temporal changes in cumulative human impacts on the world’s ocean. Nat Commun 6:7615. https://doi.org/10.1038/ncomms8615
Hamylton S, Hagan A, Bunbury N, Fleischer-Dogley F, Spencer T (2018) Mapping the lagoon at Aldabra Atoll, Western Indian Ocean. Atoll Res Bull 45–59. https://doi.org/10.17863/CAM.38477
Hart K, White C, Iverson A, Whitney N (2016) Trading shallow safety for deep sleep: juvenile green turtles select deeper resting sites as they grow. Endanger Species Res 31:61–73. https://doi.org/10.3354/esr00750
Hartig F (2022) _DHARMa: residual diagnostics for hierarchical (multi-level/ mixed) regression models_. R package version 0.4.6. https://CRAN.R-project.org/package=DHARMa
Hawkes LA, Witt MJ, Broderick AC, Coker JW, Coyne MS, Dodd M, Frick MG, Godfrey MH, Griffin DB, Murphy SR, Murphy TM, Williams KL, Godley BJ (2011) Home on the range: spatial ecology of loggerhead turtles in Atlantic waters of the USA. Divers Distrib 17:624–640. https://doi.org/10.1111/J.1472-4642.2011.00768.X
Hays GC, Ferreira LC, Sequeira AMM, Meekan MG, Duarte CM, Bailey H, Bailleul F, Bowen WD, Caley MJ, Costa DP, Eguíluz VM, Fossette S, Friedlaender AS, Gales N, Gleiss AC, Gunn J, Harcourt R, Hazen EL, Heithaus MR, Heupel M, Holland K, Horning M, Jonsen I, Kooyman GL, Lowe CG, Madsen PT, Marsh H, Phillips RA, Righton D, Ropert-Coudert Y, Sato K, Shaffer SA, Simpfendorfer CA, Sims DW, Skomal G, Takahashi A, Trathan PN, Wikelski M, Womble JN, Thums M (2016) Key questions in marine megafauna movement ecology. Trends Ecol Evol 31:463–475. https://doi.org/10.1016/j.tree.2016.02.015
Hays GC, Bailey H, Bograd SJ, Bowen WD, Campagna C, Carmichael RH, Casale P, Chiaradia A, Costa DP, Cuevas E, Nico de Bruyn PJ, Dias MP, Duarte CM, Dunn DC, Dutton PH, Esteban N, Friedlaender A, Goetz KT, Godley BJ, Halpin PN, Hamann M, Hammerschlag N, Harcourt R, Harrison AL, Hazen EL, Heupel MR, Hoyt E, Humphries NE, Kot CY, Lea JSE, Marsh H, Maxwell SM, McMahon CR, Notarbartolo di Sciara G, Palacios DM, Phillips RA, Righton D, Schofield G, Seminoff JA, Simpfendorfer CA, Sims DW, Takahashi A, Tetley MJ, Thums M, Trathan PN, Villegas-Amtmann S, Wells RS, Whiting SD, Wildermann NE, Sequeira AMM (2019) Translating marine animal tracking data into conservation policy and management. Trends Ecol Evol 34:459–473. https://doi.org/10.1016/j.tree.2019.01.009
Hays GC, Mortimer JA, Rattray A, Shimada T, Esteban N (2021) High accuracy tracking reveals how small conservation areas can protect marine megafauna. Ecol Appl 31(7):e02418. https://doi.org/10.1002/eap.2418
Hazel J, Lawler IR, Hamann M (2009) Diving at the shallow end: green turtle behaviour in near-shore foraging habitat. J Exp Mar Bio Ecol 371:84–92. https://doi.org/10.1016/j.jembe.2009.01.007
Heithaus MR, McLash JJ, Frid A, Dill LM, Marshall GJ (2002) Novel insights into green sea turtle behaviour using animal-borne video cameras. J Mar Biol Assoc UK 82:1049–1050. https://doi.org/10.1017/S0025315402006689
Heithaus MR, Frid A, Wirsing AJ, Dill LM, Fourqurean JW, Burkholder D, Thomson J, Bejder L (2007) State-dependent risk-taking by green sea turtles mediates top-down effects of tiger shark intimidation in a marine ecosystem. J Anim Ecol 76:837–844. https://doi.org/10.1111/j.1365-2656.2007.01260.x
Hooker SK, Whitehead H, Gowans S (1999) Marine protected area design and the spatial and temporal distribution of cetaceans in a submarine canyon. Conserv Biol 13:592–602. https://doi.org/10.1046/j.1523-1739.1999.98099.x
Hooker SK, Cañadas A, Hyrenbach KD, Corrigan C, Polovina JJ, Reeves RR (2011) Making protected area networks effective for marine top predators. Endanger Species Res 13:203–218. https://doi.org/10.3354/ESR00322
Kassambara A (2023) _rstatix: pipe-friendly framework for basic statistical tests_. R package version 0.7.2. https://CRAN.R-project.org/package=rstatix
Long CA, Chabot RM, El-Khazen MN, Kelley JR, Mollet-Saint Benoît C, Mansfield KL (2021) Incongruent long-term trends of a marine consumer and primary producers in a habitat affected by nutrient pollution. Ecosphere 12:e03553. https://doi.org/10.1002/ecs2.3553
Lüdecke D, Ben-Shachar M, Patil I, Waggoner P, Makowski D (2021) performance: an R package for assessment, comparison and testing of statistical models. J Open Source Softw 6(60):3139. https://doi.org/10.21105/joss.03139
Luschi P, Casale P (2014) Movement patterns of marine turtles in the Mediterranean Sea: a review. Ital J Zool 81:478–495. https://doi.org/10.1080/11250003.2014.963714
Madeira FM, Rebelo R, Catry P, Neiva J, Barbosa C, Regalla A, Patrício AR (2022) Fine-scale foraging segregation in a green turtle (Chelonia mydas) feeding ground in the Bijagós archipelago, Guinea Bissau. Front Mar Sci 9:1–13. https://doi.org/10.3389/fmars.2022.984219
Makowski C, Seminoff JA, Salmon M (2006) Home range and habitat use of juvenile Atlantic green turtles (Chelonia mydas L.) on shallow reef habitats in Palm Beach, Florida, USA. Mar Biol 148:1167–1179. https://doi.org/10.1007/s00227-005-0150-y
Marsh H, Channells P, Heinsohn G, Morrissey J (1982) Analysis of stomach contents of dugongs from Queensland. Wildl Res 9:55. https://doi.org/10.1071/WR9820055
Martinez-Estevez L, Amador JPC, Amador FC, Zilliacus KM, Pacheco AM, Seminoff JA, Lucero J, Oceguera K, Tershy BR, Croll DA (2021) Spatial ecology of hawksbill sea turtles (Eretmochelys imbricata) in foraging habitats of the Gulf of California, Mexico. Glob Ecol Conserv 27:e01540. https://doi.org/10.1016/j.gecco.2021.e01540
Martins RF, Andrades R, Nagaoka SM, Martins AS, Longo LL, Ferreira JS, Bastos KV, Joyeux JC, Santos RG (2020) Niche partitioning between sea turtles in waters of a protected tropical island. Reg Stud Mar Sci 39:101439. https://doi.org/10.1016/j.rsma.2020.101439
Matley J, Jossart J, Johansen L, Jobsis P (2020) Environmental drivers of diving behavior and space-use of juvenile endangered Caribbean hawksbill sea turtles identified using acoustic telemetry. Mar Ecol Prog Ser 652:157–171. https://doi.org/10.3354/meps13466
Matley J, Johansen L, Klinard N, Eanes S, Jobsis P (2021) Habitat selection and 3D space use partitioning of resident juvenile hawksbill sea turtles in a small Caribbean bay. Mar Biol 168:1–15. https://doi.org/10.1007/s00227-021-03912-0
Mazaris AD, Schofield G, Gkazinou C, Almpanidou V, Hays GC (2017) Global sea turtle conservation successes. Sci Adv 3:1–7. https://doi.org/10.1126/sciadv.1600730
Meylan PA, Meylan AB, Gray JA (2011) The ecology and migrations of sea turtles 8. Tests of the developmental habitat hypothesis. Bull Am Museum Nat Hist 2011:1–70. https://doi.org/10.1206/357.1
Meylan PA, Hardy RF, Gray JA, Meylan AB (2022) A half-century of demographic changes in a green turtle (Chelonia mydas) foraging aggregation during an era of seagrass decline. Mar Biol 169:1–20. https://doi.org/10.1007/s00227-022-04056-5
Mortimer JA, Donnelly M (2008) Eretmochelys imbricata. The IUCN red list of threatened species.https://doi.org/10.2305/IUCN.UK.2008.RLTS.T8005A12881238.en
Musick JA, Limpus CJ (1997) Habitat utilization and migration in juvenile sea turtles. In: Lutz PL, Musick JA (eds) The biology of sea turtles. CRC Press, Boca Raton, FL, pp 137–163
Pedersen T (2022) _ggraph: an implementation of grammar of graphics for graphs and networks_. R package version 2.1.0. https://CRAN.R-project.org/package=ggraph
Pillans RD, Fry GC, Haywood MDE, Rochester W, Limpus CJ, Patterson T, Babcock RC (2021) Residency, home range and tidal habitat use of green turtles (Chelonia mydas) in Port Curtis, Australia. Mar Biol 168:1–18. https://doi.org/10.1007/s00227-021-03898-9
Pillans RD, Whiting SD, Tucker AD, Vanderklift MA (2022) Fine-scale movement and habitat use of juvenile, subadult, and adult green turtles (Chelonia mydas) in a foraging ground at Ningaloo Reef, Australia. Aquat Conserv Mar Freshw Ecosyst 32:1323–1340. https://doi.org/10.1002/aqc.3832
Posit Team (2022) RStudio: integrated development environment for R. Posit Software, PBC, Boston, MA. http://www.posit.co/
Pritchard A, Sanchez C, Bunbury N, Burt A, Currie J, Doak N, Fleischer-Dogley F, Metcalfe K, Mortimer JA, Richards H, van de Crommenacker J, Godley B (2022) Green turtle population recovery at Aldabra Atoll continues after 50 yr of protection. Endanger Species Res 47:205–215. https://doi.org/10.3354/esr01174
QGIS Development Team (2022) QGIS geographic information system. Open Source Geospatial Foundation Project. Version 3.12.1
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Roman J, Estes JA, Morissette L, Smith C, Costa D, Mccarthy J, Nation JB, Nicol S, Pershing A, Smetacek V (2014) Whales as marine ecosystem engineers. Front Ecol Environ 12:377–385. https://doi.org/10.1890/130220
Sanchez CL, Bunbury N, Mortimer JA, A’Bear L, Betts M, von Brandis R, Burt AJ, Cooke L, van de Crommenacker J, Currie JC, Doak N, Fleischer-Dogley F, Mederic E, Mels B, Pistorius P, Richards H, Casale P (2023) Growth rate and projected age at sexual maturity for immature hawksbill turtles and green turtles foraging in the remote marine protected area of Aldabra Atoll, Seychelles. Mar Biol 170:49. https://doi.org/10.1007/s00227-023-04197-1
Santos RG, Martins AS, Farias JN, Horta PA, Pinheiro HT, Torezani E, Baptistotte C, Seminoff JA, Balazs GH, Work TM (2011) Coastal habitat degradation and green sea turtle diets in Southeastern Brazil. Mar Pollut Bull 62:1297–1302. https://doi.org/10.1016/j.marpolbul.2011.03.004
Scales KL, Lewis JA, Lewis JP, Castellanos D, Godley BJ, Graham RT (2011) Insights into habitat utilisation of the hawksbill turtle, Eretmochelys imbricata (Linnaeus, 1766), using acoustic telemetry. J Exp Mar Bio Ecol 407:122–129. https://doi.org/10.1016/j.jembe.2011.07.008
Schofield G, Papafitsoros K, Chapman C, Shah A, Westover L, Dickson LCD, Katselidis KA (2022) More aggressive sea turtles win fights over foraging resources independent of body size and years of presence. Anim Behav 190:209–219. https://doi.org/10.1016/J.ANBEHAV.2022.05.006
Selby TH, Hart KM, Smith BJ, Pollock CG, Hillis-Starr Z, Oli MK (2019) Juvenile hawksbill residency and habitat use within a Caribbean marine protected area. Endanger Species Res 40:53–64. https://doi.org/10.3354/esr00975
Seminoff JA (2004) Chelonia mydas. http://www.iucnredlist.org/details/4615/0. Accessed 17 Aug 2020
Seminoff J, Resendiz A, Nichols W (2002) Home range of green turtles Chelonia mydas at a coastal foraging area in the Gulf of California, Mexico. Mar Ecol Prog Ser 242:253–265. https://doi.org/10.3354/meps242253
Senko J, Koch V, Megill WM, Carthy RR, Templeton RP, Nichols WJ (2010) Fine scale daily movements and habitat use of East Pacific green turtles at a shallow coastal lagoon in Baja California Sur, Mexico. J Exp Mar Bio Ecol 391:92–100. https://doi.org/10.1016/j.jembe.2010.06.017
Shimada T, Limpus CJ, Hamann M, Bell I, Esteban N, Groom R, Hays GC (2020) Fidelity to foraging sites after long migrations. J Anim Ecol 89:1008–1016. https://doi.org/10.1111/1365-2656.13157
Siegwalt F, Benhamou S, Girondot M, Jeantet L, Martin J, Bonola M, Lelong P, Grand C, Chambault P, Benhalilou A, Murgale C, Maillet T, Andreani L, Campistron G, Jacaria F, Hielard G, Arqué A, Etienne D, Gresser J, Régis S, Lecerf N, Frouin C, Lefebvre F, Aubert N, Vedie F, Barnerias C, Thieulle L, Guimera C, Bouaziz M, Pinson A, Flora F, George F, Eggenspieler J, Woignier T, Allenou J-P, Louis-Jean L, Chanteur B, Béranger C, Crillon J, Brador A, Habold C, Le Maho Y, Robin J-P, Chevallier D (2020) High fidelity of sea turtles to their foraging grounds revealed by satellite tracking and capture-mark-recapture: new insights for the establishment of key marine conservation areas. Biol Conserv 250:108742. https://doi.org/10.1016/j.biocon.2020.108742
Stevens JD (1984) Life-history and ecology of sharks at Aldabra Atoll, Indian Ocean Author. Proc R Soc B Biol Sci 222:79–106
Stoddart DR, Mole L (1977) Climate of Aldabra Atoll. Atoll Res Bull 202:1–21
Stoddart DR, Taylor JD, Fosberg FR, Farrow GE (1971) Geomorphology of Aldabra Atoll. Philos Trans R Soc Lond B Biol Sci 260:31–66
Stokes HJ, Hays GC, Mortimer JA, Esteban N, Laloë J (2023) Synergistic use of UAV surveys, satellite tracking data, and mark-recapture to estimate abundance of elusive species. Ecosphere 14:e4444. https://doi.org/10.1002/ecs2.4444
Storlazzi CD, Gingerich SB, van Dongeren A, Cheriton OM, Swarzenski PW, Quataert E, Voss CI, Field DW, Annamalai H, Piniak GA, McCall R (2018) Most atolls will be uninhabitable by the mid-21st century because of sea-level rise exacerbating wave-driven flooding. Sci Adv 4:1–10. https://doi.org/10.1126/sciadv.aap9741
Tebbett SB, Connolly SR, Bellwood DR (2023) Benthic composition changes on coral reefs at global scales. Nat Ecol Evol 7:71–81. https://doi.org/10.1038/s41559-022-01937-2
Thomson JA, Burkholder DA, Heithaus MR, Fourqurean JW, Fraser MW, Statton J, Kendrick GA (2015) Extreme temperatures, foundation species, and abrupt ecosystem change: an example from an iconic seagrass ecosystem. Glob Chang Biol 21:1463–1474. https://doi.org/10.1111/gcb.12694
Tulloch VJD, Plagányi ÉE, Brown C, Richardson AJ, Matear R (2019) Future recovery of baleen whales is imperiled by climate change. Glob Chang Biol 25:1263–1281. https://doi.org/10.1111/gcb.14573
UNESCO (2022) Aldabra Atoll. https://whc.unesco.org/en/list/185. Accessed 12 Dec 2022
Van Dam RP, Diez CE (1998) Home range of immature hawksbill turtles (Eretmochelys imbricata (Linnaeus)) at two Caribbean islands. J Exp Mar Bio Ecol 220:15–24. https://doi.org/10.1016/S0022-0981(97)00080-4
Waycott M, Duarte CM, Carruthers TJB, Orth RJ, Dennison WC, Olyarnik S, Calladine A, Fourqurean JW, Heck KL, Hughes AR, Kendrick GA, Kenworthy WJ, Short FT, Williams SL (2009) Accelerating loss of seagrasses across the globe threatens coastal ecosystems. Proc Natl Acad Sci U S A 106:12377–12381. https://doi.org/10.1073/pnas.0905620106
Welsh RC, Mansfield KL (2022) Intraspecific spatial segregation on a green turtle foraging ground in the Florida Keys, USA. Mar Biol 169:22. https://doi.org/10.1007/s00227-021-04012-9
Wildermann NE, Gredzens C, Avens L, Barrios-Garrido HA, Bell I, Blumenthal J, Bolten AB, McNeill JB, Casale P, Di Domenico M, Domit C, Epperly SP, Godfrey MH, Godley BJ, González-Carman V, Hamann M, Hart KM, Ishihara T, Mansfield KL, Metz TL, Miller JD, Pilcher NJ, Read MA, Sasso C, Seminoff JA, Seney EE, Williard AS, Tomás J, Vélez-Rubio GM, Ware M, Williams JL, Wyneken J, Fuentes MMPB (2018) Informing research priorities for immature sea turtles through expert elicitation. Endanger Species Res 37:55–76. https://doi.org/10.3354/esr00916
Williard A, Hall A, Fujisaki I, McNeill J (2017) Oceanic overwintering in juvenile green turtles Chelonia mydas from a temperate latitude foraging ground. Mar Ecol Prog Ser 564:235–240. https://doi.org/10.3354/meps12013
Wilson LJ, Hammond PS (2019) The diet of harbour and grey seals around Britain: examining the role of prey as a potential cause of harbour seal declines. Aquat Conserv Mar Freshw Ecosyst 29:71–85. https://doi.org/10.1002/aqc.3131
Wirsing A, Kiszka J, Allen A, Heithaus M (2022) Ecological roles and importance of sea cows (Order: Sirenia): a review and prospectus. Mar Ecol Prog Ser 689:191–215. https://doi.org/10.3354/meps14031
Witt M, McGowan A, Blumenthal J, Broderick A, Gore S, Wheatley D, White J, Godley B (2010) Inferring vertical and horizontal movements of juvenile marine turtles from time-depth recorders. Aquat Biol 8:169–177. https://doi.org/10.3354/ab00221
Wood LD, Brunnick B, Milton SL (2017) Home range and movement patterns of subadult hawksbill sea turtles in southeast Florida. J Herpetol 51:58–67. https://doi.org/10.1670/15-133
Acknowledgements
We are very thankful to all the Aldabra staff of the Seychelles Islands Foundation for continuing this long-term monitoring program of over 40 years, in a very logistically difficult location. Thanks go particularly to the skippers who made it possible to catch many of these turtles. Thank you to the research officers, science coordinators, island managers and wardens, researchers, rangers, volunteers, and everyone else who assisted whether from Aldabra or from other locations who assisted in collecting the data. Thank you to the two anonymous reviewers who greatly improved this manuscript.
Funding
Open access funding provided by Università di Pisa within the CRUI-CARE Agreement. Seychelles Islands Foundation collected and provided all data for the study and provided logistical support for CLS.
Author information
Authors and Affiliations
Contributions
CLS: Conceptualization, Data Curation, Methodology, Investigation, Formal analysis, Writing—Original Draft. JAM: Design of field study and project supervision between 1981 and 2008. PC: Conceptualization, Methodology, Supervision, Writing—Review and Editing. NB: Administration, Conceptualization, Supervision, Writing—Review and Editing. FFD: Administration, Supervision, Writing—Review and Editing. JAM, LA, JA, MB, RvB, LC, JvdC, JCC, ND, TM, EM, BM, PP, HR, US: Investigation, Data Curation. JAM, JA, MB, LC, BM, PP, HR: Writing: Review and Editing.
Corresponding author
Ethics declarations
Conflict of interest
There are no known conflicts of interest.
Ethical standards
All data was collected by or through the Seychelles Islands Foundation, the managing body of Aldabra Atoll UNESCO World Heritage Site using standard protocols based on international best practice. No turtles were injured or killed during data collection for this study.
Additional information
Responsible Editor: L. Avens.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sanchez, C.L., Bunbury, N., Mortimer, J.A. et al. Small-scale movements and site fidelity of two sympatric sea turtle species at a remote atoll. Mar Biol 171, 91 (2024). https://doi.org/10.1007/s00227-024-04414-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-024-04414-5