Abstract
Understanding the processes that determine the occurrence of species, especially for those exposed to human activities, is key to appropriate management. The common bottlenose dolphins, Tursiops truncatus, is well studied worldwide, but very little attention has been given to transient groups of this species and how such groups are exposed to human activities. Here, we modeled and mapped how the environment and human activities drives bottlenose dolphin habitat suitability, and residence patterns in an anthropized area of the Southwestern Atlantic Ocean. To predict habitat suitability, we ran 300 distribution models, including six algorithms, and generated an ensemble model. In parallel, we used photo-identification techniques to investigate dolphins' residence patterns. Our results demonstrated that primary productivity, seabed slope and port activities explained dolphins’ habitat suitability. The most suitable areas included coastal waters, nearby port complexes and shipping routes. We also identified a low degree of residence in Cabo Frio and Rio de Janeiro city waters, but calves were constantly sighted there, indicating an important area for caring and nursing. The high overlap between the dolphins’ most suitable areas and human activities, such as ports, vessel traffic and fisheries spots, plus the presence of calves in these areas, highlights the need for safeguard measurements to protect these animals from anthropogenic threats. Our results can be used to support management decisions, such as fisheries regulations and the creation of new marine protected areas to conserve critical habitats for this species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Distribution studies are critical to assess the effects of the environment and human activities on several species (Morris and Doak 2002; Rodrigues et al. 2006). Species Distribution Models (SDM) have been increasingly used to determine the potential distribution and identify suitable habitats for conservation purposes (Guisan et al. 2017). However, the distribution of marine organisms is not well studied as it is of terrestrial organisms (Redfern et al. 2006; Robinson et al. 2011). In addition, the distribution of these organisms is mainly explained by environmental variables (e.g., di Tullio et al. 2016; McBride-Kebert et al. 2019; de Rock et al. 2019) neglecting the importance of human activities in affecting species distributions.
For years, studies of marine species with high movement ability have been challenging for researchers (Redfern et al. 2006). Marine mammals, like dolphins, living in open and fluid environments, have only a few or no physical barriers to limit their access to resources, such as highly mobile prey (Sims et al. 2008; Melo-Merino et al. 2020). Their distribution changes over time due to changes in their biological and ecological requirements (Forcada 2018). As a result, these animals used to have higher mobility and larger home ranges when compared to terrestrial mammals (Tucker et al. 2014). However, individuals or their entire populations can have a high degree of association with specific areas, which is in general related to resource availability that motivates the repeated visit or constant use of these areas (e.g., Nathan et al. 2008; Passadore et al. 2018; Akkaya Baş et al. 2019).
The common bottlenose dolphin (Tursiops truncatus), hereafter called bottlenose dolphin, is a cosmopolitan species found in coastal and oceanic environments (e.g., Milmann et al. 2016; Zanardo et al. 2017; Tardin et al. 2020). As top predators, this species is susceptible to several impacts on the environment, such as high concentration of contaminants that affect them and their ecosystems (Smith and Gangolli 2002). The species has ecological plasticity, i.e., worldwide populations vary considerably in terms of habitat use, residence pattern, behavior, and diet (Fruet et al. 2011; Lodi et al. 2014; Tardin et al. 2020; Carmen et al. 2021; Pace et al. 2021). However, information about some populations is still scarce. Although this species is found on almost the entire Brazilian coast (Lodi et al. 2017), most studies on this species investigated populations with temporary or permanent residence at a local scale (Simões-Lopes & Fabian 1999; Fruet et al. 2011). Recently, two distinct subspecies, a coastal and an offshore one, were recognized through genetics and morphological evidence in the Southwestern Atlantic (Costa et al. 2016; Simões-Lopes et al. 2019).
In the present study, we focus on the offshore ecotype, Tursiops truncatus truncatus, the unique subspecies in the study area, Rio de Janeiro State waters (Simões-Lopes et al. 2019). These waters are surrounded by the most populated area in Latin America, housing an important industrial pole, including oil and gas exploitation, and several port complexes (IBGE 2010; ANTAQ 2021). Due to the cumulative nature of different human activities combined with unique characteristics, such as habitat variability and the occurrence of threatened species, this area is considered a top conservation priority in Brazil (Magris et al. 2021). To support appropriate safeguard plans for bottlenose dolphin in such coastal area, we aimed to (i) model how the environment and human activities affect bottlenose dolphins habitat suitability, (ii) map the modeled suitable and unsuitable areas and (iii) analyze the individuals’ residence pattern. Considering that these animals have high energy requirements, we hypothesized that oceanographic characteristics related to prey distribution are the main drivers influencing the bottlenose dolphin habitat suitability.
Materials and methods
Study area
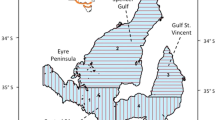
Our study area is defined by the geopolitical boundaries of the Rio de Janeiro State, from the coastline to the territorial sea of 200 NM (Fig. 1), in the Southeastern Brazilian coast located on the Southwestern Atlantic Ocean (SWAO). It comprises part of two sedimentary basins: Campos Basin and Santos Basin, both explored by the oil and gas industry, with vessel traffic and exploration, drilling, and extraction activities (ANP 2021). Industrial and artisanal fishing activities, including seine and gillnet fisheries in nearby coastal areas, are common in the area comprising approximately 50,000 t of fisheries resources captured per year (Fiperj 2020).
Study area encompassing the coastal limits of Rio de Janeiro State, Southeastern Brazil, subareas of Cabo Frio (CF) and Rio de Janeiro (RJ), and marine protected areas located in Rio de Janeiro, Brazil. APA Pau Brasil = Área de Proteção Ambiental do Pau Brasil (IUCN category VI); Resex Arraial do Cabo = Reserva Extrativista Marinha do Arraial do Cabo (IUCN category V); Resex Itaipu = Reserva Extrativista Marinha de Itaipu (IUCN category V); Mona Cagarras = Monumento Natural das Ilhas Cagarras (IUCN category III). For more details on categories, see Day et al. (2019, http://dx.doi.org/10.25607/OBP-694)
The Cabo Frio subarea (CF) has a narrow and irregular continental shelf showing depressions and steep slopes, with the 100 m isobath located at a maximum distance of 10 km from the coast (Duarte and Viana 2007; Reis et al. 2013). The warm and oligotrophic Tropical Water flows southward carried by the Brazil Current, but during spring and summer, the South Atlantic Central Water recurrently emerges due to a wind-driven upwelling, resulting in high primary and fish productivity (Carbonel 1998; Mazzoil et al. 2008).
The Rio de Janeiro subarea (RJ) has a more extensive continental shelf and consequently a less pronounced slope than CF. The 100 m isobath is located 80 km from the coast (Reis et al. 2013). Surrounded by the most populous cities in Latin America, Rio de Janeiro city (IBGE 2010), this subarea is continuously exposed to several human threats such as overfishing, submarine outfalls, and eutrophic waters from Guanabara Bay (Carreira and Wagener 1998; Rangel et al. 2007; Tubino et al. 2007; Amorim and Monteiro-Neto 2016).
Distribution
Data collection
We compiled occurrence records of common bottlenose dolphins from primary and secondary data, from 1983 to 2021 (Supplementary Table 1). All these data were obtained through visual detection from systematic sampling efforts during all seasons, covering coastal and offshore areas and following pre-established routes from vessels ranging from 6 to 36 m. Environmental layers (minimum, mean and maximum) for our study area were obtained from three different public databases: Bio-Oracle (Assis et al. 2018), Global Marine Datasets for Species Distribution Modeling and Environment Visualization (Basher et al. 2018) and Ocean Climate Layers for Marine Spatial Ecology (Sbrocco and Barber 2013) at 5 arcminutes resolution. Port activity layers were obtained from the ocean-based pollution layer (Halpern et al. 2015) at 30 arcseconds resolution (Table 1). This layer combines estimates of pollution coming from density of commercial shipping and from distance to ports (see Halpern et al. 2015 for more details). All layers were standardized for 5 arcminutes resolution and used as explanatory variables for the bottlenose dolphin habitat suitability. We have selected all layers that we had access to, and that may affect the suitability by changing prey distribution or, in some way, the dolphins’ behavior.
Data analyses
We carried out all the analyses in the R environment v4.1.1 (R Core Team 2021) and used the R package ‘biomod2’ v3.5.1 (Thuiller et al. 2020) to generate the Species Distribution Models. To avoid spatial autocorrelation, we randomly filtered occurrence records within a radius of 9.2 km (~ 5 arcminutes), the same resolution as the layers used as explanatory variables, using the ‘spThin’ v0.2.0 package (Aiello-Lammens et al. 2015). Thus, all occurrence records were at least 9.2 km from each other. We checked multicollinearity among the explanatory variables using the ‘usdm’ v1.1.18 package (Naimi et al. 2014) and those with Variance Inflation Factor (VIF) > 3 were excluded from the model (Zuur et al. 2010).
Since it was not possible to obtain true absence data, we generated five sets of 1000 pseudo-absences following the ‘random’ strategy, in which the choice is made randomly given the number of pseudo-absences. We used six algorithms for two types of data requirements: presence–absence models using regression (Generalized Linear Models—GLM and Generalized Additive Models—GAM), boosting (Random Forest—RF and Generalized Boosting Model—GBM), discrimination techniques (Flexible Discriminant Analysis—FDA), and presence-background models using Maximum Entropy models—MaxEnt (more details in Guisan et al. 2017). For model calibration, we used 70% of records for training and 30% for testing using cross-validation techniques at a constant prevalence at 0.5 (Guisan et al. 2017). Each algorithm was replicated ten times and the importance of each variable was retrieved running ten permutations using the ‘get_variables_importance’ function of the ‘biomod2’ package (Thuiller et al. 2020). This test shuffles a variable in the dataset and compares the predictions of the reference model and the shuffled model via Pearson’s correlation. Then, it is returned a score that, the higher it is, the more influence the variable has on the model (Thuiller et al. 2020).
We generated response curves for bottlenose dolphin habitat suitability as a function of each explanatory variable included in our final models (Supplementary Fig. S1). The metric used for the evaluation of each model was the Area Under the Curve (AUC) from the Receiver Operating Characteristic (ROC) (DeLeo 1993). All replicates with AUC > 0.7 were selected and aggregated for a final ensemble model using weighted-by-AUC mean (pAUC) (Araújo and New 2007). To include a measure of uncertainty, which is suggested for any SDMs (Zurell et al. 2020), we generated a committee averaging map that indicates the coefficient of variation of the algorithms (Supplementary Fig. S2).
Residence pattern
To investigate residence pattern in each subarea, we carried out systematic surveys and used photo-identification data collected between 2010 and 2018 from long-term cetacean monitoring projects conducted by the Laboratório de Bioacústica e Ecologia de Cetáceos, Projeto Baleias & Golfinhos do Rio and Projeto Ilhas do Rio, and by large scale scientific cruises, such as Projeto de Monitoramento de Cetáceos da Bacia de Santos (PMC-BS). The photo-identification technique consisted of taking dorsal fin images during surveys and comparing the natural marks (i.e., nicks, notches, scars) in each dorsal fin by photo-identification protocols that allowed a reliable individual identification (Hammond et al. 1990). Then, for identified individuals, we calculated the individual residence index as the number of days individual dolphins were sighted in the area divided by (i) the number of days of total effort and (ii) the number of days of effort from the first sighting to the last one. We also calculated residence as (iii) the number of seasons individual dolphins sighted divided by the number of seasons of effort.
These residence indices were standardized to have the same weight during the analysis using the “scale” function (“base” package v3.6.2), and individual dolphin identified was categorized into three residence degrees (low, medium or high, cut through the “rect.hclust” function from “base” package v3.6.2, and set out by us according to the average indices values of each degree) through the analysis of Agglomerative Hierarchical Clustering (“hclust” function from the “base” package v3.6.2) using the Ward distance method and the squared Euclidean distance measure. This analysis generates a matrix of dissimilarity through pre-established parameters. We calculated the cophenetic correlation coefficient through the “cor” function (“base” package v3.6.2) to evaluate whether the analysis distortion was significant, assuming a suitable clustering of the data when the value was above 0.7 (Rohlf 1970).
Results
The filtering technique used to avoid spatial autocorrelation retained 62 bottlenose dolphins occurrence records from a total of 445. After multicollinearity inspection, six of the 14 explanatory variables were selected: minimum and maximum current velocity (CURmin and CURmax, respectively), port activities (port), minimum primary productivity (PPmin), seabed slope (slope), and minimum sea surface temperature (SSTmin) (Supplementary Table 2).
For the final ensemble model (pAUC > 0.7), we considered 207 out of 300 models. Algorithms’ performances varied and, in general, FDA had the lowest pAUC values (mean = 0.76) and GBM the highest (0.81) (Table 2). The PPmin (0.65), slope (0.18), and port (0.10) were the most important variables (Table 2). Overall, bottlenose dolphin habitat suitability had a non-linear positive relationship with port, a negative relationship with slope, and was higher in moderate values of PPmin (Supplementary Fig. S1). The most suitable areas for the species distribution were in shallow waters within the continental shelf. The highest suitability values were in the coastal area displayed in the East–West direction, including the two subareas (CF and RJ) and three bays: Ilha Grande, Sepetiba, and Guanabara (Fig. 2).
Considering CF and RJ subareas, 614 individuals of bottlenose dolphins were cataloged between 2011 and 2018. Fifty-seven (9.3%) individuals were recaptured at least once and included in the residence analysis. Of these, 39 individuals had a low residence degree (68.4%), seven had a medium degree (12.3%) and 11 had a high degree (19.3%) (Supplementary Table 3 and Supplementary Fig. 3). The cophenetic correlation coefficient of 0.73 indicated that the dendrogram was well clustered.
Discussion
We showed that offshore bottlenose dolphins (Tursiops truncatus truncatus) are, in general, transient in Rio de Janeiro coastal waters (low degree of residence), but with a small subset of individuals with medium or high degree of residence in specific areas. These dolphins also occur in coastal areas and the continental shelf break, up to the slope, likely influenced by environmental conditions and human activities. We found that the most suitable areas for these dolphins occur in moderate primary productivity sites, along the continental shelf, and in more gentle slopes, from shallow water, less than 50 m deep. Shallow waters tend to be more productive, presenting a greater abundance of fishes (Fiperj 2020) that are typical prey for these dolphins. Indeed, the predicted suitable areas for bottlenose dolphins include CF and RJ subareas, which are surrounded by fishing landing ports that land more than 90% of the local fishing production (Fiperj 2020) reinforcing that these sites likely have high prey availability.
Primary productivity was an important predictor for explaining bottlenose dolphins’ suitable areas. However, bottlenose dolphins were not usually sighted in regions with the highest values for primary productivity, such as Ilha Grande, Sepetiba and Guanabara bays. At these bays, primary productivity is equivalent to those usually found in eutrophic waters (e.g., Marins et al. 2010; Aguiar et al. 2011; Castelo et al. 2021). The absence of bottlenose dolphins in those areas could be associated with the co-occurrence of other dolphins’ species. Indeed, these bays are also inhabited by resident populations of Guiana dolphins, Sotalia guianensis (e.g., Ribeiro-Campos et al. 2021). An aggressive interaction between bottlenose and Guiana dolphins was reported in Baía Norte, Southern Brazil (Wedekin et al. 2004), an area where both species overlap their niches by sharing consumptions of demersal mullet species (Teixeira et al. 2021). Such niche overlap suggests that potential interspecific competition between both dolphin populations may be a limiting factor for the occurrence of bottlenose dolphin (Teixeira et al. 2021), avoiding these areas even if the habitat is suitable. On the other hand, bottlenose dolphins are usually sighted within CF and RJ subareas, outside the three productive bays (e.g., Tardin et al. 2013, 2019; Laporta et al. 2017). The frequent occurrence of groups with calves (87.5%) at these two subareas suggests that both subareas may be important for feeding and breeding activities.
Considering CF subarea, bottlenose dolphins are already known to occur primarily in shallow and productive areas (Tardin et al. 2019). On a larger scale, on the shelf break of South and Southeast regions, the frequency of sightings is greater in areas close to the 500 m isobath than in deeper waters (di Tullio et al. 2016), but worldwide bottlenose dolphins show plasticity in habitats used. This species tends to use shallow water with higher primary productivity either in sheltered or open waters with gentle slopes occurring in Australia, Namibia, Spain, and the United States (Cañadas et al. 2002; Zanardo et al. 2017; McBride-Kebert et al. 2019; de Rock et al. 2019). However, this species is also found on steeper slopes with high primary productivity caused by upwelling in shelf break or as barriers during feeding tactics in shallow habitats (Cañadas et al. 2002; McBride-Kebert et al. 2019).
Residence patterns in a specific area also vary among populations worldwide, from high (e.g., Simões-Lopes and Fabian 1999; Laporta et al. 2017; Carmen et al. 2021; Bennington et al. 2021) to low (e.g., Zolman 2002; Balmer et al. 2008; Akkaya Baş et al. 2019; Pace et al. 2021). Low residence patterns may indicate that the species use larger habitats than the studied area (Zanardo et al. 2016; Cobarrubia-Russo et al. 2019), while a high residence pattern to specific locations may suggest critical habitats for vital activities (Simões-Lopes and Fabian 1999; Ingram and Rogan 2002).
Large-range movements (from 700 to ca. 1.700 km) were observed for bottlenose dolphins tagged with satellite tags or photo-identified in Brazil (Cremer et al. 2018). Thus, it is likely that the individuals analyzed in the present study belong to a large population and groups remain in a certain area for short-term periods or regularly visit it to feed or breed. An individual variance in terms of residence may suggest complex habitat, social or population structures (Zolman 2002; Blasi and Boitani 2014). Residence in a specific area may be linked to the high availability of food resources and low predation risks (Knip et al. 2012; Habel et al. 2016). On the other hand, resident individuals tend to be more exposed to local threats (Warkentin and Hernández 1996; Atkins et al. 2016). However, even those dolphins with low residence patterns might be exposed to local threats in our study area.
The most suitable areas for the species, for example, are areas close to port complexes and shipping routes. These areas are surrounded by four port complexes (located in Campos municipality, Guanabara, Sepetiba and Ilha Grande bays), and are affected by nearby ports along the coast (located on Macaé, Búzios, Cabo Frio municipalities and Guaíba Island) (ANTAQ 2021). There is also high vessel traffic associated with oil and gas exploration occurring in the Campos and Santos basins (ANP 2021). The effects of port complexes and related activities, such as vessel traffic, on dolphins’ populations are well reported worldwide (e.g., Halpern et al. 2015; Walker et al. 2019). Collisions of dolphins with vessels or their propellers, for example, may cause mutilation and even the death of individuals (van Waerebeek et al. 2007; Schoeman et al. 2020). Noisy areas, such as those near port complexes and shipping routes, may also change dolphin behavior and its acoustic repertoire, cause acoustic masking, and lead to temporary or permanent habitat abandonment (Guerra et al. 2014; Marley et al. 2017; Erbe et al. 2019).
As aforementioned, we also found that the highest suitable areas for bottlenose dolphins overlap with important gillnet fisheries (Fiperj 2020). Gillnets are the most dangerous fisheries for small cetaceans, increasing bycatch risks (e.g., Lewison et al. 2004; Read et al. 2006; Reeves et al. 2013). In Brazil, the bycatch risk of bottlenose dolphins is reported in several studies (e.g., Lodi et al. 2013; di Tullio et al. 2015; Zappes et al. 2016). Ways to mitigate bycatch are enforcing laws and creating no-fishing areas (Lodi et al. 2013; di Tullio et al. 2015; Zappes et al. 2016).
These suitable areas also have high levels of contaminants (Vidal et al. 2020), which may cause various adverse effects on cetaceans, such as toxins being passed to a calf through the placenta or via lactation, and immunosuppression of both calves and adults, which may result in skin diseases and even death (Moura et al. 2009; Bossart 2011; Vidal et al. 2020). A study on halogenated organic compounds shows that bottlenose dolphins are relatively more contaminated in Southeastern Brazil than in the Western United States, with most of the compounds of anthropogenic or unknown origin (Alonso et al. 2017). Therefore, individuals occurring in this area may be under all these risks.
Understanding residence patterns together with the predictions of suitable habitats may contribute to safeguarding critical areas for these dolphins. Our study identified the most suitable habitats for bottlenose dolphins in an area with multiple human activities that may expose them to several different impacts. We also found that these dolphins vary in how they use the area, suggesting complex social or populational age structures. By mapping these critical areas and characterizing how dolphins use them, our findings may support additional and more effective conservation actions. For instance, the creation of MPAs to better manage local human activities, and protect critical habitats for this important top predator since the MPAs network along Rio de Janeiro State does not encompass most of the highest suitable sites for bottlenose dolphins. Moreover, effective management of fisheries focusing on the protection of the ecosystem and reducing the bycatch of the bottlenose dolphin and any other marine species is urgently needed.
Data availability
Part of the data used in this study can be found at https://sispmcprd.petrobras.com.br/sispmc. Data not available on the previous website can be requested from the corresponding author.
References
Aiello-Lammens ME, Boria RA, Radosavljevic A, Vilela B, Anderson RP (2015) spThin: an R package for spatial thinning of species occurrence records for use in ecological niche models. Ecography 38:541–545. https://doi.org/10.1111/ecog.01132
Akkaya Baş A, Öztürk B, Amaha Öztürk A (2019) Encounter rate, residency pattern and site fidelity of bottlenose dolphins (Tursiops truncatus) within the Istanbul Strait, Turkey. J Mar Biol Assoc UK 99:1009–1016. https://doi.org/10.1017/S0025315418000577
Alonso MB, Maruya KA, Dodder NG, Lailson-Brito J, Azevedo A, Santos-Neto E, Torres JPM, Malm O, Hoh E (2017) Non-targeted screening of halogenated organic compounds in bottlenose dolphins (Tursiops truncatus) from Rio de Janeiro, Brazil. Environ Sci Technol 51:1176–1185. https://doi.org/10.1021/acs.est.6b04186
Amorim RB, Monteiro-Neto C (2016) Marine protected area and the spatial distribution of the gill net fishery in Copacabana, Rio de Janeiro, RJ, Brazil. Braz J Biol 76:1–9. https://doi.org/10.1590/1519-6984.06614
ANP (2021) Agência Nacional do Petróleo, Gás Natural e Biocombustíveis (ANP). https://www.gov.br/anp/pt-br. Accessed 26 Apr 2021
ANTAQ (2021) Agência Nacional de Transportes Aquaviários (ANTAQ). https://www.gov.br/antaq/pt-br. Accessed 26 Apr 2021
Araújo MB, New MG (2007) Ensemble forecasting of species distributions. Trends Ecol Evol 22:42–47. https://doi.org/10.1016/j.tree.2006.09.010
Assis J, Tyberghein L, Bosch S, Verbruggen H, Serrão EA, de Clerck O (2018) Bio-oracle v2.0: Extending marine data layers for bioclimatic modelling. Glob Ecol Biogeogr 27:277–284. https://doi.org/10.1111/geb.12693
Atkins S, Cantor M, Pillay N, Cliff G, Keith M, Parra G (2016) Net loss of endangered humpback dolphins: integrating residency, site fidelity, and bycatch in shark nets. Mar Ecol Prog Ser 555:249–260. https://doi.org/10.3354/meps11835
Balmer BC, Wells RS, Nowacek SM, Nowacek DP, Schwacke LH, Mclellan WA, Scharf SF, Rowles TK, Hansen LJ, Spradlin TR, Pabst DA (2008) Seasonal abundance and distribution patterns of common bottlenose dolphins (Tursiops truncatus) near St. Joseph Bay, Florida, USA. J Cetac Res Manage 10:157–167
Basher Z, Bowden D, Costello M (2018) Global Marine Environment Datasets (GMED). In: World Wide Web electronic. http://gmed.auckland.ac.nz.
Bennington S, Rayment W, Currey R, Oldridge L, Henderson S, Guerra M, Brough T, Johnston D, Corne C, Johnson D, Slooten L, Dawson S (2021) Long-term stability in core habitat of an endangered population of bottlenose dolphins (Tursiops truncatus): Implications for spatial management. Aquat Conserv Mar Freshwat Ecosyst 31:665–676. https://doi.org/10.1002/aqc.3460
Blasi MF, Boitani L (2014) Complex social structure of an endangered population of bottlenose dolphins (Tursiops truncatus) in the Aeolian Archipelago (Italy). PLoS ONE 9:e114849. https://doi.org/10.1371/journal.pone.0114849
Bossart GD (2011) Marine mammals as sentinel species for oceans and human health. Vet Pathol 48:676–690. https://doi.org/10.1177/0300985810388525
Cañadas A, Sagarminaga R, Garcı́a-Tiscar S, (2002) Cetacean distribution related with depth and slope in the Mediterranean waters off southern Spain. Deep Sea Res Part I 49:2053–2073. https://doi.org/10.1016/S0967-0637(02)00123-1
Carbonel C (1998) Modelling of upwelling in the coastal area of Cabo Frio (Rio de Janeiro - Brazil). Rev Bras Oceanogr 46:01–17. https://doi.org/10.1590/S1413-77391998000100001
Carmen M, Berrow SD, O’Brien JM (2021) Foraging behavior of bottlenose dolphins in the Shannon Estuary, Ireland as determined through static acoustic monitoring. J Mar Sci Eng 9:275. https://doi.org/10.3390/jmse9030275
Carreira RS, Wagener ADLR (1998) Speciation of sewage derived phosphorus in coastal sediments from Rio de Janeiro, Brazil. Mar Pollut Bull 36:818–827. https://doi.org/10.1016/S0025-326X(98)00062-9
Castelo WFL, Martins MVA, de Lima Ferreira PA, Figueira R, da Costa CF, da Fonseca LB, Bergamashi S, Pereira E, Terroso D, Pinto AFS, Simon MB, Socorro OAA, Frontalini F, da Silva LC, Rocha F, Geraldes M, Guerra JV (2021) Long-term eutrophication and contamination of the central area of Sepetiba Bay (SW Brazil). Environ Monit Assess 193:100. https://doi.org/10.1007/s10661-021-08861-1
Cobarrubia-Russo S, Barreto G, Quintero-Torres E, Molero-Lizarraga A, Wang X (2019) Occurrence, abundance, range, and residence patterns of Tursiops truncatus on the coast of Aragua, Venezuela. Mamm Res 64:289–297. https://doi.org/10.1007/s13364-018-0401-1
Costa APB, Rosel PE, Daura-Jorge FG, Simões-Lopes PC (2016) Offshore and coastal common bottlenose dolphins of the western South Atlantic face-to-face: What the skull and the spine can tell us. Mar Mamm Sci 32:1433–1457. https://doi.org/10.1111/mms.12342
Cremer MJ, Daura-Jorge FG, Cypriano-Souza AL, Wedekin LL (2018) Movements of offshore bottlenose dolphins in the South Brazil Bight, Southeastern Brazil. Abstr XII Congr De La Soci Latinoame De Espec En Mamíferos Acuáticos RT 18:103
de Aguiar VM, C, Neto JAB, Rangel CM, (2011) Eutrophication and hypoxia in four streams discharging in Guanabara Bay, RJ, Brazil, a case study. Mar Pollut Bull 62:1915–1919. https://doi.org/10.1016/j.marpolbul.2011.04.035
de Rock P, Elwen S, Roux J, Leeney R, James B, Visser V, Martin M, Gridley T (2019) Predicting large-scale habitat suitability for cetaceans off Namibia using MinxEnt. Mar Ecol Prog Ser 619:149–167. https://doi.org/10.3354/meps12934
de Tubino R, A, Monteiro-Neto C, Moraes LE de S, Paes ET (2007) Artisanal fisheries production in the coastal zone of Itaipu, Niterói, RJ, Brazil. Braz J Oceanogr 55:187–197. https://doi.org/10.1590/S1679-87592007000300003
DeLeo JM (1993) Receiver operating characteristic laboratory (ROCLAB): software for developing decision strategies that account for uncertainty. In: 2nd international symposium on uncertainty modeling and analysis. IEEE, pp 318–325
di Tullio J, Fruet P, Secchi E (2015) Identifying critical areas to reduce bycatch of coastal common bottlenose dolphins Tursiops truncatus in artisanal fisheries of the subtropical western South Atlantic. Endanger Species Res 29:35–50. https://doi.org/10.3354/esr00698
di Tullio JC, Gandra TBR, Zerbini AN, Secchi ER (2016) Diversity and distribution patterns of cetaceans in the Subtropical Southwestern Atlantic Outer continental shelf and slope. PLoS ONE 11:e0155841. https://doi.org/10.1371/journal.pone.0155841
Duarte CSL, Viana AR (2007) Santos drift system: stratigraphic organization and implications for late Cenozoic palaeocirculation in the Santos Basin, SW Atlantic Ocean. Geol Soc Lond Spec Publ 276:171–198. https://doi.org/10.1144/GSL.SP.2007.276.01.09
Erbe C, Marley SA, Schoeman RP, Smith JN, Trigg LE, Embling CB (2019) The effects of ship noise on marine mammals - a review. Front Mar Sci. https://doi.org/10.3389/fmars.2019.00606
Fiperj (2020) Projeto de Monitoramento da Atividade Pesqueira na Bacia de Santos (PMAP-BS). https://www.comunicabaciadesantos.com.br/programa-ambiental/projeto-de-monitoramento-da-atividade-pesqueira-pmap.html. Accessed 6 May 2021
Forcada J (2018) Distribution. In: Wursig B, Thewissen JGM, Kovacs K (eds) Encyclopedia of Marine Mammals, 3rd edn. Elsevier, London, pp 259–262
Fruet PF, Secchi ER, di Tullio JC, Kinas PG (2011) Abundance of bottlenose dolphins, Tursiops truncatus (Cetacea: Delphinidae), inhabiting the Patos Lagoon estuary, southern Brazil: implications for conservation. Zoologia (curitiba) 28:23–30. https://doi.org/10.1590/S1984-46702011000100004
Guerra M, Dawson S, Brough T, Rayment W (2014) Effects of boats on the surface and acoustic behaviour of an endangered population of bottlenose dolphins. Endanger Species Res 24:221–236. https://doi.org/10.3354/esr00598
Guisan A, Thuiller W, Zimmermann NE (2017) Habitat Suitability and Distribution Models. Cambridge University Press, Cambridge
Habel JC, Hillen J, Schmitt T, Fischer C (2016) Restricted movements and high site fidelity in three East African cloud-forest birds. J Trop Ecol 32:83–87. https://doi.org/10.1017/S0266467415000516
Halpern BS, Frazier M, Potapenko J, Casey KS, Koenig K, Longo C, Lowndes JS, Rockwood RC, Selig ER, Selkoe KA, Walbridge S (2015) Spatial and temporal changes in cumulative human impacts on the world’s ocean. Nat Commun 6:7615. https://doi.org/10.1038/ncomms8615
Hammond PS, Mizroch SA, Donovan GP (1990) Individual recognition of cetaceans: use of photo-identification and other techniques to estimate population parameters. Cambridge
IBGE (2010) Censo 2010. https://censo2010.ibge.gov.br/. Accessed 9 May 2021
Ingram S, Rogan E (2002) Identifying critical areas and habitat preferences of bottlenose dolphins Tursiops truncatus. Mar Ecol Prog Ser 244:247–255. https://doi.org/10.3354/meps244247
Knip DM, Heupel MR, Simpfendorfer CA (2012) Habitat use and spatial segregation of adult spottail sharks Carcharhinus sorrah in tropical nearshore waters. J Fish Biol 80:767–784. https://doi.org/10.1111/j.1095-8649.2012.03223.x
Laporta P, Martins CCA, Lodi L, Domit C, Vermeulen E, di Tullio JC (2017) Report of the working group on habitat use of Tursiops truncatus in the Southwest Atlantic Ocean. Latin Am J Aquat Mamm 11:47–61. https://doi.org/10.5597/00215
Lewison R, Crowder L, Read A, Freeman S (2004) Understanding impacts of fisheries bycatch on marine megafauna. Trends Ecol Evol 19:598–604. https://doi.org/10.1016/j.tree.2004.09.004
Lodi L, Zappes CA, dos Santos ÁSG (2013) Aspectos etnoecológicos e implicações para a conservação de Tursiops truncatus (Cetartiodactyla: Delphinidae) no Arquipélago das Cagarras, Rio de Janeiro, Brasil. Sitientibus Série Ciênc Biol. https://doi.org/10.13102/scb200
Lodi L, Cantor M, Daura-Jorge FG, Monteiro-Neto C (2014) A missing piece from a bigger puzzle: declining occurrence of a transient group of bottlenose dolphins off Southeastern Brazil. Mar Ecol 35:516–527. https://doi.org/10.1111/maec.12108
Lodi L, Domit C, Laporta P, di Tullio JC, Martins CCA, Vermeulen E (2017) Report of the working group on the distribution of Tursiops truncatus in the Southwest Atlantic Ocean. Latin Am J Aquat Mamm 11:29–46. https://doi.org/10.5597/lajam00214
Magris RA, Costa MDP, Ferreira CEL, Vilar CC, Joyeux J, Creed JC, Copertino MS, Horta PA, Sumida PYG, Francini-Filho RB, Floeter SR (2021) A blueprint for securing Brazil’s marine biodiversity and supporting the achievement of global conservation goals. Divers Distrib 27:198–215. https://doi.org/10.1111/ddi.13183
Marins FO, Novaes RLM, Rocha RM, Junqueira AOR (2010) Non indigenous ascidians in port and natural environments in a tropical Brazilian bay. Zoologia (curitiba) 27:213–221. https://doi.org/10.1590/S1984-46702010000200009
Marley SA, Salgado Kent CP, Erbe C, Parnum IM (2017) Effects of vessel traffic and underwater noise on the movement, behaviour and vocalisations of bottlenose dolphins in an urbanised estuary. Sci Rep 7:13437. https://doi.org/10.1038/s41598-017-13252-z
Mazzoil M, Reif JS, Youngbluth M, Murdoch ME, Bechdel SE, Howells E, McCulloch SD, Hansen LJ, Bossart GD (2008) Home ranges of bottlenose dolphins (Tursiops truncatus) in the Indian River Lagoon, Florida: environmental correlates and implications for management strategies. EcoHealth 5:278–288. https://doi.org/10.1007/s10393-008-0194-9
McBride-Kebert S, Taylor JS, Wilkinson KA, Lyn H, Moore FR, Sacco DF, Kar B, Kuczaj SA II (2019) Common bottlenose dolphin, Tursiops truncatus, seasonal habitat use and associations with habitat characteristics in Roanoke Sound, North Calo. Int J Comp Psychol. https://doi.org/10.46867/ijcp.2019.32.02.01
Melo-Merino SM, Reyes-Bonilla H, Lira-Noriega A (2020) Ecological niche models and species distribution models in marine environments: A literature review and spatial analysis of evidence. Ecol Model 415:108837. https://doi.org/10.1016/j.ecolmodel.2019.108837
Milmann L, Danilewicz D, Machado R, dos Santos RA, Ott PH (2016) Feeding ecology of the common bottlenose dolphin, Tursiops truncatus, in southern Brazil: analyzing its prey and the potential overlap with fisheries. Braz J Oceanogr 64:415–422. https://doi.org/10.1590/s1679-87592016116406404
Morris WF, Doak DF (2002) Quantitative conservation biology: theory and practice of population viability analysis. Sinauer Associates, Sunderland
Moura JF, Siciliano S, Sarcinelli PN, Hacon S (2009) Organochlorine pesticides in marine tucuxi dolphin milk incidentally captured with its calf in Barra de São João, east coast of Rio de Janeiro State. Braz Mar Biodivers Rec 2:e62. https://doi.org/10.1017/S1755267209000530
Naimi B, Hamm NAS, Groen TA, Skidmore AK, Toxopeus AG (2014) Where is positional uncertainty a problem for species distribution modelling? Ecography 37:191–203. https://doi.org/10.1111/j.1600-0587.2013.00205.x
Nathan R, Getz WM, Revilla E, Holyoak M, Kadmon R, Saltz D, Smouse PE (2008) A movement ecology paradigm for unifying organismal movement research. Proc Natl Acad Sci 105:19052–19059. https://doi.org/10.1073/pnas.0800375105
Pace DS, di Marco C, Giacomini G, Ferri S, Silvestri M, Papale E, Casoli E, Ventura D, Mingione M, Alaimo Di Loro P, Jona Lasinio G, Ardizzone G (2021) Capitoline dolphins: residency patterns and abundance estimate of Tursiops truncatus at the Tiber River Estuary (Mediterranean Sea). Biology (basel) 10:275. https://doi.org/10.3390/biology10040275
Passadore C, Möller L, Diaz-Aguirre F, Parra GJ (2018) High site fidelity and restricted ranging patterns in southern Australian bottlenose dolphins. Ecol Evol 8:242–256. https://doi.org/10.1002/ece3.3674
R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rangel CA, Chaves LCT, Monteiro-Neto C (2007) Baseline assessment of the reef fish assemblage from Cagarras Archipelago, Rio de Janeiro, southeastern Brazil. Braz J Oceanogr 55:7–17. https://doi.org/10.1590/S1679-87592007000100002
Read AJ, Drinker P, Northridge S (2006) Bycatch of marine mammals in U.S. and global fisheries. Conserv Biol 20:163–169. https://doi.org/10.1111/j.1523-1739.2006.00338.x
Redfern J, Ferguson M, Becker E, Hyrenbach K, Good C, Barlow J, Kaschner K, Baumgartner M, Forney K, Ballance L, Fauchald P, Halpin P, Hamazaki T, Pershing A, Qian S, Read A, Reilly S, Torres L, Werne F (2006) Techniques for cetacean-habitat modeling. Mar Ecol Prog Ser 310:271–295. https://doi.org/10.3354/meps310271
Reeves R, McClellan K, Werner T (2013) Marine mammal bycatch in gillnet and other entangling net fisheries, 1990 to 2011. Endanger Species Res 20:71–97. https://doi.org/10.3354/esr00481
Reis AT, Maia RMC, Silva CG, Rabineau M, Guerra JV, Gorini C, Ayres A, Arantes-Oliveira R, Benabdellouahed M, Simões I, Tardin R (2013) Origin of step-like and lobate seafloor features along the continental shelf off Rio de Janeiro State, Santos basin-Brazil. Geomorphology 203:25–45. https://doi.org/10.1016/j.geomorph.2013.04.037
Ribeiro-Campos A, Pereira KS, Quintana CEP, Barbosa M, Dias CP, Brião JA, dos Santos CVC, Castro C, Pedreira HS, Carvalho RR, Santos-Neto E, Bittencourt L, Bisi TL, Lailson-Brito J, Azevedo AF (2021) Habitat use by the Guiana dolphin (Sotalia guianensis) (Cetartiodactyla: Delphinidae) in southeastern Brazil. Regional Studies in Marine Science 44:101778. https://doi.org/10.1016/j.rsma.2021.101778
Robinson LM, Elith J, Hobday AJ, Pearson RG, Kendall BE, Possingham HP, Richardson AJ (2011) Pushing the limits in marine species distribution modelling: lessons from the land present challenges and opportunities. Glob Ecol Biogeogr 20:789–802. https://doi.org/10.1111/j.1466-8238.2010.00636.x
Rodrigues A, Pilgrim J, Lamoreux J, Hoffmann M, Brooks T (2006) The value of the IUCN Red List for conservation. Trends Ecol Evol 21:71–76. https://doi.org/10.1016/j.tree.2005.10.010
Rohlf FJ (1970) Adaptive Hierarchical Clustering Schemes. Syst Biol 19:58–82. https://doi.org/10.1093/sysbio/19.1.58
Sbrocco EJ, Barber PH (2013) MARSPEC: ocean climate layers for marine spatial ecology. Ecology 94:979–979. https://doi.org/10.1890/12-1358.1
Schoeman RP, Patterson-Abrolat C, Plön S (2020) A global review of vessel collisions with marine animals. Front Mar Sci. https://doi.org/10.3389/fmars.2020.00292
Simões-Lopes P, Daura-Jorge F, Lodi L, Bezamat C, Costa A, Wedekin L (2019) Bottlenose dolphin ecotypes of the western South Atlantic: the puzzle of habitats, coloration patterns and dorsal fin shapes. Aquat Biol 28:101–111. https://doi.org/10.3354/ab00712
Simões-Lopes PC, Fabian ME (1999) Residence patterns and site fidelity in bottlenose dolphins, Tursiops truncatus (Montagu) (Cetacea, Delphinidae) off Southern Brazil. Revista Brasileira De Zoologia 16:1017–1024. https://doi.org/10.1590/S0101-81751999000400012
Sims DW, Southall EJ, Humphries NE, Hays GC, Bradshaw CJA, Pitchford JW, James A, Ahmed MZ, Brierley AS, Hindell MA, Morritt D, Musyl MK, Righton D, Shepard ELC, Wearmouth VJ, Wilson RP, Witt MJ, Metcalfe JD (2008) Scaling laws of marine predator search behaviour. Nature 451:1098–1102. https://doi.org/10.1038/nature06518
Smith AG, Gangolli SD (2002) Organochlorine chemicals in seafood: occurrence and health concerns. Food Chem Toxicol 40:767–779. https://doi.org/10.1016/S0278-6915(02)00046-7
Tardin RH, Simão SM, Alves MAS (2013) Distribution of Tursiops truncatus in Southeastern Brazil: a modeling approach for summer sampling. Nat Conserv 11:65–74. https://doi.org/10.4322/natcon.2013.011
Tardin RH, Chun Y, Jenkins CN, Maciel IS, Simão SM, Alves MAS (2019) Environment and anthropogenic activities influence cetacean habitat use in southeastern Brazil. Mar Ecol Prog Ser 616:197–210. https://doi.org/10.3354/meps12937
Tardin RH, Maciel IS, Maricato G, Simão SM, Maria TF, Alves MAS (2020) Occurrence, residency patterns and habitat use of the bottlenose dolphin, Tursiops truncatus truncatus, on two Marine Protected Areas in Southeastern Brazil. An Acad Bras Ciênc 92:e20180843. https://doi.org/10.1590/0001-3765202020180843
Teixeira CR, Botta S, Daura-Jorge FG, Pereira LB, Newsome SD, Simões-Lopes PC (2021) Niche overlap and diet composition of three sympatric coastal dolphin species in the southwest Atlantic Ocean. Mar Mamm Sci 37:111–126. https://doi.org/10.1111/mms.12726
Thuiller W, Georges D, Engler R, Breiner F (2020) biomod2: Ensemble platform for Species Distribution Modeling.
Tucker MA, Ord TJ, Rogers TL (2014) Evolutionary predictors of mammalian home range size: body mass, diet and the environment. Glob Ecol Biogeogr 23:1105–1114. https://doi.org/10.1111/geb.12194
van Waerebeek K, Baker AN, Félix F, Gedamke J, Iñiguez M, Sanino GP, Secchi E, Sutaria D, van Helden A, Wang Y (2007) Vessel collisions with small cetaceans worldwide and with large whales in the Southern Hemisphere, an initial assessment. Latin Am J Aquat Mamm. https://doi.org/10.5597/lajam00109
Vidal LG, Vannuci-Silva M, Alonso MB, Feo ML, Corcellas C, Bisi TL, Flach L, Fragoso ABL, Lima Silva FJ, Carvalho VL, de Meirelles ACO, Domit C, Barbosa LA, Cremer MJ, Azevedo AF, Torres JPM, Malm O, Lailson-Brito J, Eljarrat E (2020) Pyrethroid insecticides along the Southwestern Atlantic coast: Guiana dolphin (Sotalia guianensis) as a bioindicator. Sci Total Environ 728:138749. https://doi.org/10.1016/j.scitotenv.2020.138749
Walker TR, Adebambo O, del Aguila Feijoo MC, Elhaimer E, Hossain T, Edwards SJ, Morrison CE, Romo J, Sharma N, Taylor S, Zomorodi S (2019) Environmental effects of marine transportation. In: World seas: an environmental evaluation. Volume III: ecological issues and environmental impacts, 2nd edn. Academic Press, pp 505–530
Warkentin IG, Hernández D (1996) The conservation implications of site fidelity: A case study involving nearctic-neotropical migrant songbirds wintering in a Costa Rican mangrove. Biol Cons 77:143–150. https://doi.org/10.1016/0006-3207(95)00146-8
Wedekin LL, Daura-Jorge FG, Simões-Lopes PC (2004) An aggressive interaction between bottlenose dolphins (Tursiops truncatus) and estuarine dolphins (Sotalia guianensis) in Southern Brazil. Aquat Mamm 30:391–397. https://doi.org/10.1578/AM.30.3.2004.391
Zanardo N, Parra GJ, Möller LM (2016) Site fidelity, residency, and abundance of bottlenose dolphins (Tursiops sp.) in Adelaide’s coastal waters. South Aust Mar Mamm Sci 32:1381–1401. https://doi.org/10.1111/mms.12335
Zanardo N, Parra GJ, Passadore C, Möller LM (2017) Ensemble modelling of southern Australian bottlenose dolphin Tursiops sp. distribution reveals important habitats and their potential ecological function. Mar Ecol Prog Ser 569:253–266. https://doi.org/10.3354/meps12091
Zappes CA, Simões-Lopes PC, Andriolo A, di Beneditto APM (2016) Traditional knowledge identifies causes of bycatch on bottlenose dolphins (Tursiops truncatus Montagu 1821): An ethnobiological approach. Ocean Coast Manag 120:160–169. https://doi.org/10.1016/j.ocecoaman.2015.12.006
Zolman ES (2002) Residence patterns of bottlenose dolphins (Tursiops truncatus) in the Stono River Estuary, Charleston County, South Carolina, U.S.A. Mar Mamm Sci 18:879–892. https://doi.org/10.1111/j.1748-7692.2002.tb01079.x
Zurell D, Franklin J, König C, Bouchet PJ, Dormann CF, Elith J, Fandos G, Feng X, Guillera-Arroita G, Guisan A, Lahoz-Monfort JJ, Leitão PJ, Park DS, Peterson AT, Rapacciuolo G, Schmatz DR, Schröder B, Serra-Diaz JM, Thuiller W, Yates KL, Zimmermann NE, Merow C (2020) A standard protocol for reporting species distribution models. Ecography 43:1261–1277. https://doi.org/10.1111/ecog.04960
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1:3–14. https://doi.org/10.1111/j.2041-210X.2009.00001.x
Acknowledgements
We would like to thank Laboratório de Bioacústica e Ecologia de Cetáceos, Projeto Baleias & Golfinhos do Rio de Janeiro, Projeto Ilhas do Rio, Projeto de Monitoramento de Cetáceos da Bacia de Santos (PMC-BS) and Projeto Talude for support in data collecting and analyses. We thank Petrobras for kindly authorizing the data use from PMC-BS. We also thank the reviewers who made valuable suggestions to improve the manuscript.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001. Laboratório de Bioacústica e Ecologia de Cetáceos was funded by Edital Universal / Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (number 479348/2010–3) and by Fundação Grupo Boticário de Proteção à Natureza (number 0997/2013–2). Projeto Baleias & Golfinhos do Rio de Janeiro is accomplished by Instituto Mar Adentro and funded by Programa Costa Atlântica / Fundação SOS Mata Atlântica (2013, 2016–2017 e 2019) and by Programa Marinho / WWF-Brasil (2016–2017, number CPT 00776–2016). Projeto Ilhas do Rio is accomplished by Instituto Mar Adentro, funded by Petrobras (concession number: phase II—6000.0086840.13.2 and phase III—5850.0106133.17.2), and with technical curation by WWF-Brasil and funded by Associação IEP e JGP (phase IV). Projeto de Monitoramento de Cetáceos is a monitoring program required by Brazil’s federal environmental agency, IBAMA, for the environmental licensing process of the oil production and transport by Petrobras at the Santos Basin pre-salt province (process number 02001.114279/2017–80, ACCTMB number 657/2015). Fábio G. Daura-Jorge received research grants from CNPq (308867/2019–0) and from CAPES (88887.374128/2019–00). Research grants to Maria Alice S. Alves were provided by CNPq research grant (PQ process #306.579/2018–9).
Author information
Authors and Affiliations
Contributions
Study conception and design were performed by GM, RT, TM and MASA. Data collection and analyses were performed by GM, RT, LL, LW, FD and IM. The first draft of the manuscript was written by GM and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
This is an observational study and no ethical approval is required.
Additional information
Responsible Editor: V. Paiva.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.





Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Maricato, G., Tardin, R., Lodi, L. et al. Identifying suitable areas for common bottlenose dolphin in anthropized waters. Mar Biol 169, 110 (2022). https://doi.org/10.1007/s00227-022-04095-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-022-04095-y