Abstract
The yellowmouth barracuda Sphyraena viridensis, a highly mobile predator, is considered a migratory seasonal visitor to the Azores Islands by local fishers. This perception is challenged here through a combined analysis of multi-annual commercial landings, underwater visual census and telemetry data from acoustically tagged fish to investigate the long-term movement patterns and residency (up to 4 years) in a seamount marine reserve. Although the landing patterns suggested a strong seasonal variation in abundance, the visual census and the telemetry experiment results were consistent with year-round residency in the Azores. Barracudas occurred consistently in underwater census throughout the year, and most tagged fish were almost continuously detected at the seamounts for up to four consecutive years. Detection probability did, however, decrease over time and showed seasonal variability as tagged fish were less mobile with longer absences during summer and fall. Given the intrinsic vulnerability of fish aggregations to exploitation, our results highlight the need to protect shallow seamount habitats and preserve sexually mature fish aggregations and their reproductive potential, contributing to the sustainable management and conservation of these vulnerable ecosystems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Barracudas (family Sphyraenidae) are iconic top pelagic predators of tropical, subtropical and some warm temperate reef habitats. Despite their ecological importance and economic value for commercial fisheries, sport fishing and leisure diving, our knowledge of their life history and behavior remains poor. The main exception is the great barracuda (Sphyraena barracuda), which has been the focus of various investigations relating to their behavior (Paterson 1998; Wilson et al. 2006), early life history (Auster et al. 2011; D’Alessandro et al. 2011; Habegger et al. 2011; D’Alessandro et al. 2013), biology (Kadison et al. 2010) and spatial ecology (O’Toole et al. 2010, 2011; Hansen and Kerstetter 2015). In contrast, the remaining 19 barracuda species were subject to only a handful of studies, mainly focusing on their distribution, ecology and biology derived either from fisheries data (e.g., Kara and Bourehail 2003; Kozul et al. 2005; Bearez 2008; Dulcic et al. 2009; Villegas-Hernandez et al. 2014) or from visual observations (e.g., Barreiros et al. 2002; Bertoncini et al. 2010; Doiuchi et al. 2011; Friedlander et al. 2014). Consequently, the spatial ecology for many of these species remains poorly documented.
The yellowmouth barracuda, Sphyraena viridensis, is only known to occur in the eastern Mediterranean and the Macaronesian archipelagos in the northeast Atlantic (Azores, Madeira and the Canary Islands) and is the only barracuda known to these islands (Afonso 2002; Wirtz et al. 2008; Brito et al. 2002). Yellowmouth barracuda is the most abundant pelagic schooling predator on local offshore reefs and shallow seamounts in the Azores (Barreiros et al. 2002; Bertoncini et al. 2010), where it is thought to be a migratory summer visitor (Barreiros et al. 2002). Until recently, the almaco jack (Seriola rivoliana) was also thought to be highly migratory, possibly emigrating from the Azores during the colder months. However, a recent investigation showed that this pelagic reef predator is resident year-round, at least on shallow seamounts (Fontes et al. 2014). This finding challenges the generalized perception that spatial management and the benefits of marine reserves are difficult to achieve for pelagic fishes, because the spatial protection offered by marine reserves seldom matches the scale of pelagic fish movements.
Many pelagic predators, including some reef fishes, can be found over remote seamounts (Abecasis et al. 2009), which are considered hot spots of biodiversity in the open ocean (Pitcher et al. 2007). Seamounts tend to accelerate water currents, enhancing the local flux of prey organisms, which in turn cause pelagic prey to aggregate and attract pelagic fish, seabirds and marine mammals (Morato et al. 2008). This particular dynamic enhances productivity and allows seamounts to sustain larger fish communities than surrounding areas (Koslow 1997; Genin and Dower 2007). Shallow seamounts may also harbor mixed communities, whereby deep-sea and open-ocean fishes coexist with coastal species which find there an opportunity to settle and survive far from coastal habitats. This is typical of many tropical atolls and shallow offshore reefs worldwide (Meyer et al. 2007; Monteiro et al. 2008), but is also true for some warm temperate shallow seamounts (Afonso 2002; Abecasis et al. 2009). Considering that pelagic predators may be attracted to and aggregate at seamounts to explore the increased availability of suitable prey, it is worth considering whether seasonal cycles in productivity, prey availability and environmental conditions can affect seamount attractiveness for pelagic predators and influence long-term residency.
In this paper, we combined information from commercial landings, underwater visual census and acoustic telemetry to investigate residency patterns of yellowmouth barracuda in the Azores, exploring the seasonal, inter-annual and fine-scale movements at the Formigas seamount reserve. Understanding the short- and long-term residency patterns in the region and within the reserve should help to assess the effectiveness of seamount reserves to manage and protect pelagic fishes like the yellowmouth barracuda.
Methods
Landings
Yellowmouth barracuda is a common catch in the Azorean artisanal mixed coastal fishery and is caught primarily using gill nets, trawling and pole-and-line with live bait. To search for possible seasonal trends in occurrence of barracuda in the fisheries catches, we analyzed the official landing statistics from Azorean regional fish auction authority database. All of the reported landings from 1978 to 2011 across the nine islands were aggregated to obtain the total landings by season (winter, January–March; spring, April to June; summer, July to September; and fall, October to December) of yellowmouth barracuda across the archipelago and years.
Underwater visual census
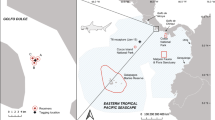
The seasonal pattern of occurrence of yellowmouth barracuda in the fishery-independent IMAR-UAz underwater visual census (UVC) program database was analyzed. The program uses standard belt transects conducted by SCUBA divers from the surface to 40 m depth at coastal and offshore reefs (for details, see Schmiing et al. 2013) across the archipelago. We selected sites for their representativeness of habitat, which included rocky reefs at the Formigas and D. João de Castro (DJC) shallow seamount reserve and at the islands of Corvo, Faial, Pico and Sta. Maria (Fig. 1). The period between July 1997 and November 2000 was used to produce a dataset with surveys across all months. A fish species list was extracted for each dive at rocky reefs deeper than 10 m, and the presence or absence of barracuda recorded. However, the number of monthly censuses varied, and some months could not be sampled in particular year, while surveys at seamounts were restricted to the summer months. To account for the unbalanced design, (1) we averaged the monthly frequency of occurrence of barracuda (from species lists per dive) from 1997 to 2000, and (2) we standardized these observations by dividing the number of fish lists containing yellowmouth barracuda by the total number of dives per month.
Acoustic telemetry
Study site
The seasonal and inter-annual residency of yellowmouth barracuda at the Formigas (FOR) shallow seamount was assessed using passive acoustic telemetry. The summit of the seamount is the sunken portion of a 5-km-wide old caldera, providing about 20 ha of shallow habitat (0–100 m) (Fig. 1). It has three major elevations: the Formigas islets on the west, the Dollabarat reef on the east and the “middle reef” (26 m) in the center of the caldera. The seamount was declared a marine protected area in 1988 with fishing prohibited in its shallow (0–200 m) reefs, except tuna fishing. In 2003, the whole seamount and surrounding area down to 1,800 m were declared a regional and international (OSPAR) no-take marine reserve (except for tuna fishing) covering a total area of 53,000 ha.
Capture and tagging
Fishing locations were selected based on the abundance of bait fish, through the interpretation of echo-sounder profiles. Jigged barracuda were hauled slowly (ca. 0.2 m/s) to promote the natural release of excess gas through the gut and reduce the probability of swim bladder rupture. Barracuda were captured by midwater (20–70 m) jigging or traditional surface pole-and-line using live bait [small (<20 cm) Trachurus picturatus].
In total, 17 yellowmouth barracuda were surgically implanted with ultrasonic transmitter tags (V16-4H Vemco Ltd., Halifax, Nova Scotia) over the three reefs of the Formigas seamount: 13 fish (65–95 cm FL) in September 2007 and four fish (68.5–104 cm FL) in August 2008 (Table 1). The tags were surgically implanted in the peritoneal cavity of selected individuals while keeping fish in inverted position and tonic immobility with continuous seawater flow through the mouth and gills for the entire procedure (for details on tagging procedure, see Afonso et al. 2009). Following measurement, all acoustically tagged fish were tagged externally with PDS small plastic tipped dart tags (Hallprint, Australia) for external recognition if recaptured. No fish presented evident signs of barotrauma (protruded eyes or gut, hemorrhagic eyes or gills) (Jarvis and Lowe 2008). Tagged fish were immediately released at the original site of capture (Table 1), the whole handling procedure lasting < 4 min.
Acoustic monitoring
In August 2007, we deployed three acoustic receivers (VR2 W, Vemco single frequency) at Formigas, one at each of the three shallow reefs, at about 25 m depth (Fig. 1). The receivers continuously monitored the presence of any coded transmitters in their vicinity, logging the exact time/date and code of a given tag when in range. Although listening range tests were not conducted at the study sites, similar listening ranges as observed on other Azorean reefs (Afonso et al. 2008a, b) and seamounts (for details see Afonso et al. 2012, 2014) of comparable characteristics were assumed. These tests showed maximum listening range of 800 m.
Acoustic transmitters randomly emitted a coded signal every 60–180 s for an estimated battery life of 1470 days, while the total monitoring period of the receivers was 1410 days, except for the middle reef station which stopped prematurely, due to failure, 1039 days after deployment. We moored the receivers approximately 2.5 m above the seafloor and retrieved them by SCUBA diving to download stored information. We retrieved receivers once a year to download stored information.
Data analysis
Seasonal landings and seasonal frequency of occurrence were analyzed using one-way ANOVA and post hoc Tukey–Kramer tests to compare all pairs. All statistical assumptions (normality, constant variances and presence of outliers) were checked prior to analysis. The long-term acoustic data were screened for spurious detections, which may occur whenever signals from different coded transmitter emissions collide and result in “false” detections (Heupel et al. 2006). To evaluate levels of residency of tagged fish, we calculated detection frequency indices after Fontes et al. (2014) where the total number of days a fish was detected by receiver x (I x ) or the whole array of receivers (I R) is divided by the maximum possible number of days that the fish could be detected. The denominator assumes the same value as the duration of the experiment since the transmitter’s predicted battery duration was longer than the duration of the experiment. We computed an additional residence index (I RD), based on the ratio between the total number of days a fish was detected and the number of days elapsed between first and last detection (Afonso et al. 2008a). We corrected Ix by using only effective duty periods, i.e., the sum of days that individual receivers/stations were active (Fontes et al. 2014).
We used linear distance between stations where a given fish was detected as a proxy for fish mobility within the receiver array, and minimum monthly travelled distance within the array was estimated as the sum of the minimum linear distances travelled between the stations sequentially visited over 1 month. Absence periods were defined as the number of days between two detections of a given transmitter/fish, at any station. For these analyses, we only used detections from September 2008 to September 2010, when all three shallow stations were active, in order to have the best available spatial cover of the seamount.
We modeled the probability of detection (i.e., residency) at the Formigas reserve as a function of explanatory covariates, fish length, number of days at liberty, water temperature, season, surface chlorophyll-a concentration and geostrophic current velocity, using a generalized linear mixed model, GLMM (package lme4, Bates et al. 2011) (see Fontes et al. 2014 for details).
Chlorophyll-a concentration and geostrophic current velocity were extracted from AVISO (http://www.aviso.oceanobs.com/en/home.html) using Arc-GIS, with the Marine Geospatial Ecology Tools (MGET), from a 20-by-20 km square centered at the Formigas reserve. Temperature was measured every 30 min with a temperature logger (StowAway® tidbit) moored at 25 m on the Formigas rocks. Individual detections were binned over 15-day periods, and a fish was considered as being present in a given bin if at least two valid detections were recorded within <24 h from each other across the Formigas receiver network.
In this analysis, the data set from the first batch of fish (2007) was truncated, taking the total period of monitoring of the second batch (1051 days) as the cutoff point. Initially, a thorough data exploration was conducted (i.e., visual inspection of data distribution and possible outliers, pairwise correlation of environmental covariates, variance inflation factor (VIF) analysis and interaction between variables) following Zuur et al. (2010). All covariates except season (categorical covariate) were averaged over each 15-day bin. Correlated covariates were excluded from the “full model” which included only time post-release, fish length, season and geostrophic current velocity as fixed effect parameters. Tag ID was used as random effect to account for repetitive measurements of each tag with time, and a stepwise backward model selection based on the minimization of the Akaike Information Criterion (Akaike 1973) used to identify the best model. Finally, we applied pairwise post hoc tests (repetitive GLMMs that used alternate seasons as the baseline for the estimation) to look for differences between seasons. Bonferroni corrections were used to correct the significance levels for repetitive testing. Statistical models and data exploration were executed using R software (R-Development-Core-Team 2012).
Results
Fisheries and Underwater Visual Census
Commercial catches of yellowmouth barracuda in the Azores were highly seasonal, with most landings occurring in spring and summer (Fig. 2). The average landed weight of yellowmouth barracuda was significantly different between seasons (ANOVA, F (3,4816) = 0.23; p < 0.0001) with average fall and winter landings significantly lower than spring and summer. Spring landings were lower than those in summer, while fall and winter landings were not significantly different (post hoc test, Tukey–Kramer HSD). In contrast, the frequency occurrence of barracuda in the UVC was not significantly different between seasons (ANOVA, p > 0.05) (Fig. 2).
Frequency of occurrence of yellowmouth barracuda (Shyraena viridensis) from underwater visual census (black columns) and cumulative landings in the Azores (gray columns). Numbers above black columns represent the number of transects performed in island locations (bold) and offshore seamounts (regular). Bars represent standard error
Seamount residency
We detected 13 of the 17 tagged barracuda in the Formigas acoustic receiver array (Table 1). None of the tagged barracuda were detected in any other receivers across the archipelago, including those at the neighboring island of Santa Maria (Fig. 1).
Most fish were detected over multiple years in Formigas. The residency index (I R ) ranged between 0.02 and 0.8 with an average residency of 0.33. The average individual residency period (I RD), i.e., considering only the period from release to the last detection was 0.5, ranging from 0.15 to 0.81 (Table 1). Three fish (6672, 6677 and 6678) were detected only during the first year (Fig. 3).
Detection plot of yellowmouth barracuda (Sphyraena viridensis) tagged at the Formigas seamount reserve, illustrating periods of presence and absence on any given day over the duration of the study. Black circles indicate daily detections in all three receivers combined, and the doted vertical line indicates the day when the middle reef receiver was deactivated
Some fish were detected regularly throughout the experiment or until they disappeared from the receiver array. Others were detected more intermittently, with variable absence duration, including one long absence of fish 6684 of over 2 years (Fig. 3). However, over 90% of the absent periods were shorter than a week (Fig. 4). The average absence period was longer in the warmer months (summer and fall) than in the colder months (winter and spring) (ANOVA with season nested within year, F 3,4 = 2.96; p < 0.005) (Fig. 5).
Box plots of average length of time intervals without detection by season for the yellowmouth barracuda (Sphyraena viridensis) at Formigas reserve. Boxes upper and lower limits represent the 25th and 75th percentiles, whiskers above the box indicate the 90th percentile, and dashed line represents the average. Dots represent the 5th and 95th percentiles
Most barracuda were detected at all the three receivers, while residency indexes were generally greater at the station closest to the site of tagging. Fish travelled frequently among the shallow receivers, and we observed an increase in mobility during colder months relative to the warmer months (ANOVA, with season nested within year, F 3,4 = 2.1; p < 0.05) (Fig. 6).
Box plots of monthly average distance travelled between stations, by season for yellowmouth barracuda (Sphyraena viridensis) from Formigas. Boxes upper and lower limits represent the 25th and 75th percentiles, whiskers above the box indicate the 90th percentile, and dashed line represents the average. The dots represent outliers
Detection probability
Individual size and environmental variables had no effect on detection probability and were not retained in the final model. Season and time post-release had a significant effect on the detection probability of yellowmouth barracuda (Table 2). Barracudas were more likely to be detected in winter and spring in comparison with summer and fall (Table 3), and detection probability was inversely related to time post-release.
Discussion
Here we present results of the first experimental investigation into the spatial ecology of yellowmouth barracuda. Our observations compliment two previous telemetric studies available for the Sphyraenidae (O’Toole et al. 2011) (Hansen and Kerstetter 2015), and our data provide the longest time series on the behavior of any barracuda species to date.
According to the perceived wisdom of Azorean fishers, the yellowmouth barracuda is a migratory fish, with peak abundances occurring in the summer. This notion is probably derived from the strong seasonal pattern observed in commercial landings, which supports the argument that local management/conservation strategies are unlikely to have a significant impact. However, the UVCs data analysis and the telemetry experiment presented here suggest that the yellowmouth barracuda may be resident in the Azores. The seasonal pattern observed in commercial landings probably does not reflect the abundance of yellowmouth barracuda throughout the year; rather, seasonal variation in landings is likely driven by changes in the artisanal fleet’s behavior, which is known to alternate from bottom fishing in the cold months to pelagic fishing in the warmer months (Carvalho et al. 2011).
The tagging experiment showed that about two-thirds of the tagged individuals were long-term residents (1–4 years) at the Formigas shallow seamount. Four barracudas (24%) were never detected, possibly due to either post-release mortality or emigration. Although emigration is a possibility, it seems unlikely when compared to other mobile reef fishes tagged in similar previous telemetry studies (e.g., Meyer et al. 2007; Afonso et al. 2009; Fontes et al. 2014). As a more probable explanation, immediate post-release mortality (within 24 h) of these four fish would yield a premature mortality rate (23.5%) comparable to that reported by O’Toole et al. (2011) for the great barracuda (21%). For one fish detected only during the first couple of months after release, and a second that was apparently absent during most of the study (almost 3 years), but which returned in the last year, it is possible that these two individuals emigrated from the seamount. Nevertheless, there is no definitive proof of long-distance emigration, as both of these fish were never detected elsewhere in our receiver array, including other seamounts and the neighboring island of Santa Maria (Fig. 1).
Although Formigas is a marine reserve, poaching is frequent (personal observation) and tuna fishing with live bait is permitted by request. As none of the six fish that ceased to be recorded before the end of the study were reported as recaptures, we suggest that they may have been fished illegally and would not have been reported.
In spite of the residency pattern, our tagged barracuda were frequently out of receiver detection range for short periods, 90% of which ranged between 1 day and 1 week. Although long-distance travels over short periods of time (82 km/day) have been reported for the great barracuda (Hansen and Kerstetter 2015), we suspect this is not the case for the yellowmouth barracuda, given the isolation of the Formigas reef and the lack of detections across the regional receiver array. During the summer months, yellowmouth barracuda are known to spend most of their time drifting passively, alternating with shorter active periods pursuing prey (Barreiros et al. 2002). Alternatively, we suspect that most absences may be the result of the limited receiver coverage at the seamount (<20% of available reef habitat) and, to some extent, the potential acoustic barrier effect caused by the islet ridge and shallow Dollabarat reef. If a tagged animal is on the east side of the islets, it is possible that it may not be detected by the receiver on the west side of the ridge. However, this effect was never tested or quantified.
Our findings are consistent with those found by previous studies on other pelagic reef predators in the Azores (Afonso et al. 2009; Fontes et al. 2014) and elsewhere (Meyer et al. 2007), suggesting that the residential nature of pelagic fishes may be more prevalent than previously perceived. Unsurprisingly, we noted that detection probability decreased over time, independent of individual size or environmental characteristics, possibly due to a combination of emigration, natural and fishing mortality. Although yellowmouth barracuda were detected year-round, residency patterns and mobility within the reef decreased during spring and summer. Barreiros et al. (2002) reported that summer schooling barracuda spend approximately 75% of their time drifting passively and the rest of the time either chasing prey or interacting socially. During summer, the higher abundance of the barracuda’s preferred prey (juvenile blue jack mackerel Trachurus picturatus, as per Barreiros et al. 2002) and the accessibility to spawning partners in schools could reduce their need for longer movements. Assuming equal probability of finding tagged fishes across the entire reef, lower mobility should translate to longer periods of individual disappearance simply as a result of fewer displacements across the reef reducing the likelihood of encountering a receiver. Unlike the yellowmouth barracuda, Fontes et al. (2014) using the same experimental regime found that almaco jacks were more mobile in the summer and suggested this could be related to an increase in mobility between core activity areas and spawning aggregations, as in other Carangidae (e.g., Meyer et al. 2007; Afonso et al. 2009).
Unlike the great barracuda, which migrates offshore to spawn during the warmer months (O’Toole et al. 2011), our data suggest that the spawning grounds of tagged yellowmouth barracuda are contained within the Formigas reserve, since they remained in the area throughout the spawning season (May through August, personal observation). This finding, although based on a relatively low number of individuals, was repeated over consecutive seasons. These results are consistent with the findings from other recent investigations (e.g., Afonso et al. 2009; Fontes et al. 2014) that suggest that pelagic predators such as barracudas and jacks are likely to benefit from long-term protection of these isolated seamount habitats by reducing the vulnerability of resident populations to overfishing and the protection of spawning biomass.
In conclusion, our results are consistent with the hypothesis that yellowmouth barracuda is a year-round resident in the Azores, while small-scale mobility and detection probability varied between seasons and over time. Novel insights into the spatial ecology of the yellowmouth barracuda in the Azores have strong management and conservation implications given the intrinsic higher vulnerability of seamount-aggregating fishes (Morato et al. 2006; Morato and Pitcher 2008; Hernandez-Salas 2015) and spawning aggregations in particular (Sadovy and Domeier 2005; Sadovy et al. 2008; Gruss et al. 2014). Our results suggest that seamount reserves can effectively contribute to the conservation of barracudas and the behavioral data presented may further be applied to better regulate the seasonal and spatial exploitation of seamount populations.
References
Abecasis D, Cardigos F, Alamada F, Gonçalves JMS (2009) New records on the ichthyofauna of the Gorringe seamount (Northeastern Atlantic). Mar Biol Res 5(605):611
Afonso P (2002) Spatial patterns in the littoral fish community of the Azores. MS thesis, Coimbra
Afonso P, Fontes J, Holland KN, Santos RS (2008a) Social status determines behaviour and habitat usage in a temperate parrotfish: implications for marine reserve design. Mar Ecol Prog Ser 359:215–227
Afonso P, Fontes J, Morato T, Holland KN, Santos RS (2008b) Reproduction and spawning habitat of white trevally, Pseudocaranx dentex, in the Azores, central north Atlantic. Sci Mar (Barc) 72:373–381
Afonso P, Fontes J, Holland KN, Santos RS (2009) Multi-scale patterns of habitat use in a highly mobile reef fish, the white trevally Pseudocaranx dentex, and their implications for marine reserve design. Mar Ecol Prog Ser 381:273–286. doi:10.3354/meps07946
Afonso P, Graca G, Berke G, Fontes J (2012) First observations on seamount habitat use of blackspot seabream (Pagellus bogaraveo) using acoustic telemetry. J Exp Mar Biol Ecol 436:1–10. doi:10.1016/j.jembe.2012.08.003
Afonso P, McGinty N, Graca G, Fontes J, Inacio M, Totland A, Menezes G (2014) Vertical migrations of a deep-sea fish and its prey. PLoS ONE. doi:10.1371/journal.pone.0097884
Akaike H (1973) Information theory and an extension of the maximum likelihood principle. In: Petrov BN, Csaki F (eds) 2nd international symposium information theory, Budapest, pp 267–281
Auster PJ, Grenda D, Godfrey J, Heupel E, Auscavitch S, Mangiafico J (2011) Behavioral observations of Lilliputian Piscivores: young-of-year Sphyraena barracuda at offshore sub-tropical reefs (NW Atlantic Ocean). Southeast Nat 10:563–569. doi:10.1656/058.010.0318
Barreiros JP, Santos RS, de Borba AE (2002) Food habits, schooling and predatory behaviour of the yellowmouth barracuda, Sphyraena viridensis (Perciformes: Sphyraenidae) in the Azores. Cybium 26:83–88
Bates D, Maechler M, Bolker B (2011) lme4: linear mixed-effects models using S4 classes. R package version 0999375-42. http://CRAN.R-project.org/package=lme4
Bearez P (2008) Occurrence of Sphyraena qenie (Sphyraenidae) in the tropical eastern Pacific, with a key to the species of barracudas occurring in the area. Cybium 32:95–96
Bertoncini AA, Machado LF, Barreiros JP, Hostim-Silva M, Verani JR (2010) Rocky reef fish community structure in two Azorean islands (Portugal) central North Atlantic. J Mar Biol Assoc UK 90:1353–1362. doi:10.1017/s0025315410000135
Brito A, Pascual PJ, Falcón JM, Sancho A, González G (2002) Peces de las Islas Canarias. Catálogo comentado e ilustrado, Tenerife
Carvalho N, Edwards-Jones G, Isidro E (2011) Defining scale in fisheries: small versus large-scale fishing operations in the Azores. Fish Res 109:360–369. doi:10.1016/j.fishres.2011.03.006
D’Alessandro EK, Sponaugle S, Llopiz JK, Cowen RK (2011) Larval ecology of the great barracuda, Sphyraena barracuda, and other sphyraenids in the Straits of Florida. Mar Biol 158:2625–2638. doi:10.1007/s00227-011-1771-y
D’Alessandro EK, Sponaugle S, Cowen RK (2013) Selective mortality during the larval and juvenile stages of snappers (Lutjanidae) and great barracuda Sphyraena barracuda. Mar Ecol Prog Ser 474:227–242. doi:10.3354/meps10114
Doiuchi R, Bogorodsky S, Nakabo T (2011) An underwater photograph of Sphyraena iburiensis (Perciformes: Sphyraenidae) from the Red Sea: first record outside Japanese waters. Ichthyol Res 58:99–100. doi:10.1007/s10228-010-0180-4
Dulcic J, Kovacic M, Dragicevic B (2009) Range extension and additional records of the yellowmouth barracuda, Sphyraena viridensis (Actinopterygii: Perciformes: Sphyraenidae) in the eastern Adriatic sea. Acta Ichthyol Pisc 39:59–61. doi:10.3750/aip2009.39.1.12
Fontes J, Schmiing M, Afonso P (2014) Permanent aggregations of a pelagic predator at shallow seamounts. Mar Biol 161:1349–1360. doi:10.1007/s00227-014-2423-9
Friedlander AM, Ballesteros E, Fay M, Sala E (2014) Marine communities on oil platforms in Gabon, West Africa: high biodiversity oases in a low biodiversity environment. PLoS ONE. doi:10.1371/journal.pone.0103709
Genin A, Dower JF (2007) Seamount plankton dynamics. In: Pitcher TJ, Morato T, Hart PJB, Clark MR, Haggan N, Santos RS (eds) Seamounts: ecology, fisheries & conservation. Blackwell, Oxford, p 527
Gruss A, Robinson J, Heppell SS, Heppell SA, Semmens BX (2014) Conservation and fisheries effects of spawning aggregation marine protected areas: what we know, where we should go, and what we need to get there. ICES J Mar Sci 71:1515–1534. doi:10.1093/icesjms/fsu038
Habegger ML, Motta PJ, Huber DR, Deban SM (2011) Feeding biomechanics in the Great Barracuda during ontogeny. J Zool 283:63–72. doi:10.1111/j.1469-7998.2010.00745.x
Hansen NR, Kerstetter D (2015) Habitat use and vertical distribution of the great barracuda Shpyraena barracuda (Edwards 1771) in the Western North Atlantic using electronic archival tags. Gulf Caribb Res 26:4–9. doi:10.18785/gcr.2601.06
Hernandez-Salas CR (2015) Seamounts protection in the Pacific Insular Region of Chile. Chin J Int Law 14:151–176. doi:10.1093/chinesejil/jmu046
Heupel MR, Simpfendorfer CA, Collins AB, Tyminski JP (2006) Residency and movement patterns of bonnethead sharks, Sphyrna tiburo, in a large Florida estuary. Environ Biol Fishes 76:47–67. doi:10.1007/s10641-006-9007-6
Jarvis ET, Lowe CG (2008) The effects of barotrauma on the catch-and release survival of southern California nearshore and shelf rockfish (Scorpaenidae, Sebastes spp.). Can J Fish Aquat Sci 65:1286–1296. doi:10.1139/F08-071
Kadison E, D’Alessandro EK, Davis GO, Hood PB (2010) Age, growth, and reproductive patterns of the Great Barracuda, Sphyraena barracuda, from the Florida keys. Bull Mar Sci 86:773–784. doi:10.5343/bms.2009.1070
Kara MH, Bourehail N (2003) Occurrence of the barracuda, Sphyraena viridensis (Sphyraenidae), off eastern coasts of Algeria. Cybium 27:59–60
Koslow JA (1997) Seamounts and the ecology of deep-sea fisheries. Am Sci 85:168–176
Kozul V, Tutman P, Glavic N, Skaramuca B, Bolotin J (2005) First record of the yellowmouth barracuda, Sphyraena viridensis (Sphyraenidae) from Adriatic Sea. Cybium 29:201–202
Meyer CG, Holland KN, Papastamatiou YP (2007) Seasonal and diel movements of giant trevally Caranx ignobilis at remote Hawaiian atolls: implications for the design of Marine Protected Areas. Mar Ecol Prog Ser 333:13–25. doi:10.3354/Meps333013
Monteiro P, Ribeiro D, Silva JA, Bispo J, Goncalves JMS (2008) Ichthyofauna assemblages from two unexplored Atlantic seamounts: Northwest Bank and Joao Valente Bank (Cape Verde archipelago). Sci Mar 72:133–143
Morato T, Pitcher TJ (2008) Reconciling fisheries with conservation on seamounts. In: Nielsen JL, Dodson JJ, Friedland K, Hamon TR, Musick J, Verspoor E (eds) Reconciling fisheries with conservation: proceedings of the Fourth World Fisheries Congress, Symposium 49. American Fisheries Society, Bethesda, Maryland
Morato T, Cheung WWL, Pitcher TJ (2006) Vulnerability of seamount fish to fishing: fuzzy analysis of life-history attributes. J Fish Biol 68:209–221. doi:10.1111/j.1095-8649.2005.00894.x
Morato T, Varkey DA, Dâmaso C, Machete M, Santos M, Prieto R, Santos RS, Pitcher TJ (2008) Evidence of a seamount effect on aggregating visitors. Mar Ecol Prog Ser. doi:10.3354/meps07269
O’Toole AC, Murchie KJ, Pullen C, Hanson KC, Suski CD, Danylchuk AJ, Cooke SJ (2010) Locomotory activity and depth distribution of adult great barracuda (Sphyraena barracuda) in Bahamian coastal habitats determined using acceleration and pressure biotelemetry transmitters. Mar Freshw Res 61:1446–1456. doi:10.1071/mf10046
O’Toole AC, Danylchuk AJ, Goldberg TL, Suski CD, Philipp DP, Brooks E, Cooke SJ (2011) Spatial ecology and residency patterns of adult great barracuda (Sphyraena barracuda) in coastal waters of The Bahamas. Mar Biol 158:2227–2237. doi:10.1007/s00227-011-1728-1
Paterson SE (1998) Group occurrence of great barracuda (Sphyraena barracuda) in the Turks and Caicos Islands. Bull Mar Sci 63:633–638
Pitcher TJ, Morato T, Hart PJB, Clark MR, Haggan N, Santos RS (2007) Seamounts: ecology, fisheries and conservation. Blackwell, Oxford
R-Development-Core-Team (2012) R: a language and environment for statistical computing. R-project.org, Vienna
Sadovy Y, Domeier M (2005) Are aggregation-fisheries sustainable? Reef fish fisheries as a case study. Coral Reefs 24:254–262. doi:10.1007/s00338-005-0474-6
Sadovy Y, De Mitcheson YS, Cornish A, Domeier M, Colin PL, Russell M, Lindeman KC (2008) A global baseline for spawning aggregations of reef fishes. Conserv Biol 22:1233–1244
Schmiing M, Afonso P, Tempera F, Santos RS (2013) Predictive habitat modelling of reef fishes with contrasting trophic ecologies. Mar Ecol Prog Ser 474:201–216. doi:10.3354/Meps10099
Villegas-Hernandez H, Munoz M, Lloret J (2014) Life-history traits of temperate and thermophilic barracudas (Teleostei: Sphyraenidae) in the context of sea warming in the Mediterranean Sea. J Fish Biol 84:1940–1957. doi:10.1111/jfb.12411
Wilson SK, Wilson DT, Lamont C, Evans M (2006) Identifying individual great barracuda Sphyraena barracuda using natural body marks. J Fish Biol 69:928–932. doi:10.1111/j.1095-8649.2006.01128.x
Wirtz P, Fricke R, Biscoito MJ (2008) The coastal fishes of Madeira Island - new records and an annotated check-list. Zootaxa 1715(1715):1–26
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1:3–14. doi:10.1111/j.2041-210X.2009.00001.x
Acknowledgements
We thank T. Morato, F. Cardigos, N. Serpa, M. Santos, J. Sousa, G. Graça, M. Machete, J. Botelho and the crews of R/V “Arquipélago” and R/V “Águas Vivas” for their help with field work; F. Tempera, R. Medeiros and G. Graça for providing data and support in the production of bathymetric maps; J. Santos for telemetry database management; and Mara Schmiing for helping with the statistical modeling. A special thank is due to R. S. Santos for stimulating discussions and for his continued support to this research. FCT/MCTES-MEC provided individual support to J.F., M.S. and P.A. (SFRH/BD/12788/2003, SFRH/BPD/66532/2009, SFRH/BD/66117/2009, SFRH/BPD/27057/2006 and Ciência 2008/POPH/QREN). IMAR-DOP/UAz is UI&D #531 and Associated Laboratory LARSyS funded by FCT/MEC and Azorean DRCTC (OE, COMPETE, Pro-Convergência). This study had the support of Fundação para a Ciência e Tecnologia (FCT), through the strategic project UID/MAR/04292/2013 granted to MARE. This paper is a contribution to the projects MARMACII (INTERREG/05/MAC/4.2/A4), SEAMOV (PTDC/MAR/108232/2008) and MoniZec (DRCT/0281M2.1.2/I/018/2011) and received additional funding through the FCT/MCTES and DRCT “Reequipamento” programs. Research in this study was conducted in agreement with Azorean and Portuguese laws and legal requirements for the handling of vertebrate animals.
Funding
This study was funded through the contributions of IMAR-DOP/UAz and LARSyS funded by FCT/MCTES and DRCTC (OE, COMPETE, Pro-Convergência). FCT/MCTES-MEC provided individual support to J.F., M.S. and P.A. (SFRH/BD/12788/2003, SFRH/BPD/66532/2009, SFRH/BD/66117/2009, SFRH/BPD/27057/2006 and Ciência 2008/POPH/QREN).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Responsible Editor: E. Hunter.
Reviewed by D. Kerstetter and an undisclosed expert.
Rights and permissions
About this article
Cite this article
Fontes, J., Afonso, P. Long-term residency and movements of yellowmouth barracuda (Sphyraena viridensis) at a shallow seamount. Mar Biol 164, 19 (2017). https://doi.org/10.1007/s00227-016-3045-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-016-3045-1