Abstract
We used a comprehensive database comprising a worldwide distribution of 3074 species of marine benthic harpacticoid copepods to explore Arctic (north to 60°N) and tropical (32°S–32°N) faunas. In addition to species and genus richness, we compared the number of observed genera with that expected from a neutral model (random samples from a corresponding species pool). This analysis was performed at three scales: global (Arctic vs. tropics), regional (three Arctic and four tropical regions) and local (78 assemblages). Overall, more than three times as many species and twice as many genera were found in the tropics compared with the Arctic. The number of taxa restricted to a given zone (“climatic endemics”) was much higher in the tropics. All zonal and regional faunas had significantly fewer genera than expected (phylogenetic clumping), but this depletion was more substantial in the Arctic. Phylogenetically, younger families included fewer widely distributed genera and were more species rich at low latitudes, in conformity with the “Out of tropics” hypothesis. At the local level, there was no latitudinal difference in the average species richness. However, the Arctic assemblages comprised considerably fewer genera than expected, whereas phylogenetic diversity in the tropics only slightly deviated from the expected level. Thus, compared with the tropics, the Arctic harpacticoid fauna shows lower global but similar local species diversity and a lower level of endemicity. The Arctic fauna also shows stronger phylogenetic clumping at all scales, indicating multiple underlying processes of species filtering (evolutionary and ecological).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Explaining the large-scale patterns in biodiversity, such as latitudinal diversity gradients, is among the most important problems in ecology. The causes and drivers of these patterns are still much debated (e.g., Gaston and Blackburn 2000, and references therein). Biodiversity, however, has until recently been measured solely in terms of species (or higher taxa) numbers, with little regard to the phylogenetic aspects, such as the distribution of species within higher taxa. At the same time, phylogeny-based biogeography can be crucial for understanding the processes that determine the contemporary patterns in diversity, including the latitudinal gradients (Wiens and Donoghue 2004).
Since many phylogenetic diversity measures, including the species-to-genera ratio (S/G) as the simplest one, are positively correlated with the number of species, one would expect that such measures should trace common patterns in diversity, e.g., latitudinal gradients. However, the actual values of phylogenetic diversity often deviate systematically from the expected level, demonstrating either joint occurrences of related species (high S/G, phylogenetic clumping) or their segregation (low S/G, phylogenetic over-dispersion). In particular, Roy et al. (1996) found a significant latitudinal increase in the S/G ratio for marine mollusks along the northeastern Pacific coast, even though the total number of species showed the opposite trend. Similar inverse relationships have also been found for angiosperms (Fenner et al. 1997) and vascular plants in Fennoscandia (Grytnes et al. 2010). Furthermore, Krug et al. (2008) and Harnik et al. (2010) found that the S/G ratios for tropical and polar, but not temperate, faunas of marine bivalves lay significantly above the expected level. The counterintuitive latitudinal increase in S/G ratios implies phylogenetic clumping at high latitudes, at least at the global and regional levels (local-scale assemblages were not considered). Several hypotheses have been proposed to explain this pattern, involving both large-scale (evolutionary) and local (ecological) forces, but empirical support remains limited, and the cross-scale comparisons are few (Azovsky 1996; Webb et al. 2002; Wiens and Donoghue 2004).
Furthermore, these findings, as many other macroecological studies, are based on macroorganisms, whereas data on global distribution of microscopic metazoans (meiofauna) are extremely scarce. This especially concerns harpacticoid copepods—one of the most diverse groups of marine meiofauna, inhabiting all types of sediments at all depths and latitudes. This group is of particular interest as a test of biogeographical principles, since their typical body size is close to the empirical 1-mm threshold value discriminating macroorganisms with biogeography and microorganisms “without biogeography” (Finlay 2002). Harpacticoids are just the “boundary group” between macro- and microfauna, but their biogeography is poorly known.
Here, we present the first study to compare the diversity of marine benthic harpacticoids from low-latitude (“tropical”) and high-latitude (“Arctic”) shallow waters. More particularly, our null hypothesis was that, at every scale and latitudinal zone considered, harpacticoid taxonomic diversity was statistically indistinguishable from the random subsamples from a larger species pool. Any deviation from the null hypothesis (either phylogenetic clumping or over-dispersion) at a certain scale would indicate some processes of allied species sorting. Furthermore, we hypothesize that difference in the diversity between the high- and low-latitude faunas would indicate the latitudinal variation in the sorting processes.
Materials and methods
Data
Global dataset
A worldwide, taxonomically standardized comprehensive database consisting of spatial occurrences of extant shallow-water marine benthic harpacticoids was used for the analysis (Chertoprud et al. 2010). The base currently includes the data on 3074 species from 533 genera and contains about 11,000 occurrence records grouped into 35 pre-defined geographical areas. The data were compiled primarily from an exhaustive literature search of over 1000 sources, though some original data were also used. Obligate deep-sea species (only occurring below 300 m) were not considered, since deep-sea harpacticoid fauna is highly specific and largely underexplored (Seifried 2003). While compiling the database, all data passed careful quality control for spelling errors, invalid combinations, synonymy and/or later ascertained misidentifications and were taxonomically standardized using recent revisions and redescriptions. The distribution records from summarizing publications or Web sites were cross-referenced as much as possible with the original papers. Regarding nomenclature, we primarily followed Wells’ (2007) system while accounting for later taxonomic changes and updates.
For the purpose of this study, all Arctic and sub-Arctic regions north of 60°N were considered as the “high-latitude zone.” The areas between 32°N and 32°S were treated as the “low-latitude” or tropical zone. Hereafter, these zones are referred to as “Arctic” and “tropics” for short, even though they also encompass sub-Arctic and subtropical regions, respectively. The fauna of the southern polar regions (Antarctic coasts) was included in the global database but was not analyzed separately because it is insufficiently studied and only a few appropriate local datasets are available (see below). Species and genera were classified according to their distribution across the climate zones. Following Krug et al. (2008), taxa occurred in only one zone (Arctic or tropics) are termed “climatic endemics”; taxa that range extends from tropical to Arctic zones are termed “climatic cosmopolitans.”
Regional datasets
The Arctic zone was provisionally subdivided into three regions: Central (White and Barents Seas including Spitsbergen), East Arctic (from Kara to Beaufort Sea) and West Arctic (Baffin Bay, Greenland, Iceland and Norway coasts). The tropical zone was further subdivided into four regions: central Atlantic (C_ATL), Eastern Pacific (E_PAC) (Americas’ coasts, Polynesia, Hawaii and Galapagos Islands), West Indian (W_IND) (East Africa coasts, Arabian Gulf and Red Sea) and East Indian–West Pacific (EIWP) (Bengal Gulf, Andaman Sea, South China Sea, Indonesia, Philippines, New Guinea and tropical Australia). At the regional level, the faunas of these regions, retrieved from the global database, were analyzed separately.
Local datasets
Seventy eight species lists for the local harpacticoid assemblages were compiled from the literature and harmonized according to the modern taxonomy. Faunistic studies based on more or less systematic sampling that explored a wide range of families were mainly chosen; taxonomical studies dealing with particular taxa were also accounted to amend the species lists. Among these local datasets, 29 sites were from the Arctic, and 49 were from the tropics (Fig. 1). The full list of the local datasets with source references is given in Online Resource 1 of the Supporting Information.
The data came from various surveys, which were very different in their scope, design, types of habitats and sampling effort. A more comprehensive survey of various habitats could potentially yield more species in the same genus. To take into account the possible effects of unequal sampling, we estimated the habitat diversity covered by each local dataset. The following habitats were distinguished: silt/mud, sand, gravel/cobbles, fouling of hard substrates (rocks, corals), near-bottom water/ice and macrophytes. Habitat diversity score was estimated as the number of habitats sampled. If the range of sampled depths exceeded ten meters, one additional point to the score was added to account for bathymetric zonality. Also, the sampling effort was rated as follows: (1) low (short-term local survey with limited number of samples); (2) medium (several sampling sessions in different sites or seasons); and (3) high (extensive long-term survey with large number of samples).
Phylogeny
The phylogenetic system of Harpacticoida is thus far questionable and is mainly based on morphology, and paleontologic and genetic data are scarce and fragmented and thus not helpful for evaluating the evolutionary age of a taxon. Indeed, the phylogenetic position of many taxa is in a state of flux (Seifried 2003; Boxshall and Halsey 2004). Nevertheless, we decided to use the morphology-based phylogenetic tree to estimate relative age of a taxon (Huys and Boxshall 1991). Based on the most comprehensive phylogenetic scheme proposed by Seifried (2003), we assessed three families of the most plesiomorphic clades: Polyarthra (comprising Longipediidae and Canuellidae) and Aegisthoidea (Aegisthidae)—as the “oldest” ones; the “relatively old” families included Neobradyidae, Ectinosomatidae and all representatives of the Podogennonta lineage, while later diverged Exanechentera members (Peltidiidae, Porcellidiidae, Tegastidae, Tisbidae, Novocriniidae, Paramesochridae, Superornatiremidae and Tachidiidae) were treated as “young” families.
Statistical analysis
The species-to-genera ratio (S/G) is often used as the simplest measure of phylogenetic diversity. Proposed as early as 1901 by the plant biogeographer Paul Jaccard, this ratio was used to identify emergent patterns in the structure of assemblages by, for example, directly relating its to the intensity of interspecific competition. However, this approach has been criticized repeatedly because the S/G ratio depends strongly on the species number and is thus not directly comparable between datasets acquired through variable and unknown sampling effort (see Azovsky 1996 for historical review). To overcome this pitfall, observed S/G values should be adjusted to the null expectation by applying the appropriate neutral model (Simberloff 1970; Azovsky 1996; Webb et al. 2002). In addition, such comparison does not imply any testable null hypothesis.
Therefore, we assessed taxonomic diversity against a null model derived from a randomization of the appropriate species pool. The implicit null hypothesis is that the taxonomic structure of a given species list is not distinguishable from a random sample from the species pool containing it. The model was obtained by randomly drawing species (without replacement) from a corresponding species pool (a “master list”), assuming that the species arrived randomly and independently into the sample from the pool. Given the number of species in a sample and the taxonomic composition of the master list, this null model represents the expected mean number of genera (Gexp) in that sample. The standard deviation (SD) for Gexp was also estimated by resampling. The Gexp and SD calculations were performed by applying the individual rarefaction procedure in the PAST software version 3.04 (Hammer et al. 2001).
We then compared the observed (Gobs) and expected genus numbers using the standardized effect size of genera (SESG), which was calculated as the difference between the observed and expected numbers of genera, divided by the SD of the expected value: SESG = (Gobs − Gexp)/SD. The SESG ranges from negative to positive infinity; positive values represent over-dispersion (when the species list includes more genera, i.e., fewer congeners, and has a lower S/G ratio than expected), whereas negative values represent under-dispersion (a list composed of species that are more related than those of a null model).
The use of SESG has many advantages. First, this estimator is robust to varying richness and uncontrolled sampling efforts and is thus comparable across various datasets (Fenner et al. 1997; Krug et al. 2008; Grytnes et al. 2010). Second, because its values are standardized relative to the expected distribution, SESG directly indicates the strength of deviation from neutrality. Values between 2 and −2 indicate that the taxonomic diversity deviates insignificantly from a random sample (i.e., the observed number of genera lies within a 95 % CI for the expected level), whereas values beyond this range indicate sufficient deviation.
This analysis was sequentially performed at three levels (Fig. 2):
-
1.
A global comparison was performed for the Arctic and tropical datasets using the entire fauna (3074 species) as the “master list”;
-
2.
A regional-to-global comparison was performed for each of seven regional datasets, with the Arctic or tropical fauna as the corresponding “master list”;
-
3.
A local-to-regional comparison was performed for 78 local datasets, with the corresponding regional fauna as the “master list.”
A nonparametric Mann–Whitney test and a paired Wilcoxon test for equal medians were used for the Arctic–tropics comparisons. To check the difference in the SESG values between regions, a Kruskal–Wallis multiple-group test was used with sequential Bonferroni correction of p values for multiple comparisons. We also performed nonparametric analysis (PERMANOVA) to test the effects of sampling effort, habitat diversity and latitude on local species richness and SESG values.
Additionally, the “average taxonomic distinctness” (AvTD, Δ+) was estimated for the regional datasets to assess whether the observed Δ+ values are consistent with a random distribution model (i.e., assuming that the species distribution is taxonomy independent). We performed the randomization test (N = 1000) available in PRIMER v. 6.0 (Clarke and Gorley 2006) to estimate the 95 % probability limits for the Δ+ values.
Results
Global diversity
Overall, 433 species (162 genera) have been reported from arctic shallow waters, and only a minor subset of these (96 species and 13 genera) are found exclusively in this zone and could thus be treated as potential “arctic endemics” (Fig. 3). More than three times as many species (1442) and twice as many genera (358) are found in the tropics. Among them, 91.6 % of “tropical” species and 68.0 % of genera are not present in the Arctic, and 60.1 % of these species (28.1 % of genera) are limited to the tropical zone in their distribution (“tropical climatic endemics”).
A total of 126 species and 115 genera have been recorded from the Arctic to the tropics (“climatic cosmopolitans”). In addition, 15 species and eight genera occur both in the Arctic and in anti-boreal zone and/or the Antarctic but not in the tropics. These taxa either have a bipolar distribution or may be potential cosmopolitans.
Thus, the tropical fauna of harpacticoids, considered as a whole, appears to be much richer and more specific than that of Arctic zone (Fig. 3). In addition, the tropical fauna has a higher S/G ratio (4.03 vs. 2.67 in the Arctic). When compared with the null model, however, we obtained an SESG value equal to −5.91 for the Arctic and −3.29 for the tropics. Both values (the large symbols in Fig. 4) are far beyond the ±2 band (95 % CI for the null model), indicating significantly fewer genera (higher S/G ratios) than expected. This depletion (“phylogenetic clumping”) is notably more substantial in the Arctic region.
Additionally, we separately compared the average species richness for the genera with a different type of distribution. The cosmopolitan genera comprised significantly more species in the tropics than in the Arctic, and the endemic genera had on average many fewer species than the widely distributed genera, both in the Arctic and in the tropics (Table 1).
Species richness and phylogenetic age of families
Phylogenetically, ancient clades were better represented in high latitudes than the recent ones. Percent of species presented in the Arctic fauna, decreased regularly from 21 % in the “oldest” families to 7 % in the “young” families (Fig. 5). In the tropics, all age groups were more or less equally represented, each having 50–60 % of its total richness. The “oldest” families comprised on average 10.7 species in the Arctic compared with 31.3 species in the tropics. Similar figures were observed for other “relatively old” families: 10.6 and 32.5 species, respectively. In contrast, the “young” families comprised only 4.6 species in the Arctic compared with 35.1 species in the tropics. Furthermore, an average 46.5 % of genera in the “oldest” families were climatic cosmopolitans, i.e., had representatives in all climatic zones, whereas only 20.4 % genera in the “young” families were cosmopolitans. In other words, phylogenetically older taxa were more successful in settling the Arctic zone, whereas younger taxa were more restricted to the tropics in their distribution.
Regional diversity
Some 55 % of the Arctic species were found in only one region; this value was much higher (76.5 %) in the tropics. With regard to genera, these values were almost identical (44.4 and 43.6 %). Thus, the Arctic fauna demonstrated lower regional endemicity at the species level but not at the genus level.
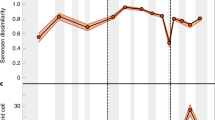
All three arctic regions have sufficiently lower negative SESG values than expected for random samples from Arctic fauna. This is also true for all tropical regions except the West Indian region (Fig. 4). Thus, almost all regional faunas consistently indicate “phylogenetic clumping” of species that constitute the zonal pools. The average taxonomic distinctness (Δ+) calculated up to the genus level (i.e., ignoring the grouping of genera by families, Fig. 6a) showed a pattern that was consistent with that of SESG. The Arctic fauna showed more pronounced declines in taxonomic diversity than the tropical fauna. The same procedure was applied for the full taxonomic range (accounting for the assignment of both species to genera and genera to families), and similar results were observed (Fig. 6b): all regions but one (tropical Atlantic) showed lower phylogenetic diversity than expected, though the difference between low and high latitudes became negligible.
Average taxonomic distinctness (Δ+) calculated up to genera level (a) and up to family level (b). A funnels represent the 95 % limits for a random draws from the World fauna master list. See “Materials and methods” for the regions’ abbreviations
Local diversity
Sampling effort turned to be the main factor influencing the local species richness (Fig. 7a). As expected, the more extensive surveys yielded more species (PERMANOVA test: p < 10−16). Local richness also slightly increased if several habitat types were investigated (p = 0.065), while the difference between low- and high-latitude localities was insignificant (Fig. 7a, b). On average, the Arctic localities included 48.2 species, and the tropical localities included almost the same number (46.4 species). The three outliers with unusually high species diversity were Kandalaksha Bay (the White Sea, 168 species), Inhaca Island near Mozambique (126 species) and the Andaman Islands in the Indian Ocean (119 species), the three sites of long-term systematic studies. Except for these three sites, the majority of localities exhibited from 30 to 50 species each, regardless of location.
Finally, we tested the genus diversity of local assemblages, comparing it with the expected values for random draws from the respective regional species pools. Neither sampling effort nor habitat diversity had any effect on SESG values (PERMANOVA tests: p = 0.33 and 0.14, correspondingly). It provides some assurance that further analysis was not affected by an inherent sampling bias (Fig. 7 c, d). The difference between high and low latitudes, however, was highly significant. Most of the tropical assemblages (41 out of 49) had SESG values within the 95 % scatter band of the null model, and only eight points showed significantly negative values (Fig. 8). In contrast, all Arctic communities strongly deviated from neutrality, again having fewer genera but more congeners than expected. The Mann–Whitney pairwise post hoc test confirmed a highly significant difference in SESG between the Arctic and tropical datasets, though differences between the tropical regions were insignificant (Table 2). Thus, there was a pronounced difference in phylogenetic diversity between high and low latitudes, even at the level of local assemblages.
Discussion
We have analyzed diversity of marine benthic harpacticoids at three levels: global (high and low-latitudinal bands entirely), regional (on spatial scale of 103–104 km) and local (assemblages from confined areas, on scale of 1–102 km). The analysis reveals noticeable latitudinal differences in all considered aspects of diversity, i.e., in species and genera richness, endemicity and species-to-genera ratio. Below we discuss these differences, with special attention to spatial scale of their manifestation.
Patterns in species and genus richness
There are three times as many harpacticoid species and twice as many genera in the tropics than in the Arctic. However, there is no difference in local richness between tropical and arctic localities. On the other hand, the local richness strongly correlates with diversity of habitats, indicating the prominent role of spatial heterogeneity. This is in good agreement with our previous findings that marine harpacticoids show a latitudinal trend in spatial turnover (beta diversity) but not in alpha diversity (Azovsky et al. 2012). Recently, Fonseca and Netto (2014) analyzed data for nematodes from 43 estuaries around the world and found that genus turnover decreased with increasing latitude, whereas local genus richness did not show a clear latitudinal trend. Similar latitudinal changes in species turnover but not in local diversity have also been reported for some other marine and terrestrial groups (see Azovsky et al. (2012) for additional references and Soininen et al. (2007) for an extensive meta-analysis). All these findings allowed us to consider spatial turnover (beta diversity) as the crucial component in forming a global latitudinal diversity gradients, while local richness seems to be driven by another set of factors.
Meanwhile, the above-mentioned difference in total harpacticoid richness may be partially caused by the insufficient exploration of polar fauna. The unequal number of the appropriate datasets (29 polar vs. 49 from tropics) indirectly confirms such insufficiency.
Patterns in climatic endemicity
Taxa with wide and restricted climatic ranges show sharply asymmetric distributions between high and low latitudes. Indeed, only 22 % of species and 8 % of genera recorded at high latitudes are potential endemic to this zone, whereas these figures are notably higher in the tropics: 60 % of endemic species and 28 % of genera. Eleven of 13 polar endemic genera are monotypic, though one-third of “tropical endemic” genera include several species. It was shown earlier that percent of endemic harpacticoids tended to be lower at high latitudes, even after correction for species richness (=sampling effort) (Chertoprud et al. 2010). This type of asymmetry in the distribution of climatic endemics is not a specific feature of harpacticoids. Similar pattern, known as Rapoport’s rule (Stevens 1989), has been demonstrated repeatedly for large metazoans (Gaston and Blackburn 2000; Ruggiero and Werenkraut 2007). Our findings allow the extension of this rule to small-sized meiofauna as well, whereas some protists, e.g., ciliates, show the reversed pattern (Azovsky and Mazei 2013).
Patterns in the S/G ratio
Perhaps the most intriguing and counterintuitive results were observed for the species-to-genera ratio. It would be natural to expect more species per genus in the tropics, area that is commonly assumed to have higher diversity and high speciation rates. However, we obtained quite the opposite: on the whole, the higher than expected S/G ratios (i.e., negative SESG values) were observed at high latitudes despite the low number of taxa at that zone. This difference is observed at both global and regional levels but is most clearly pronounced for the local-scale data (compare Figs. 4, 8). As mentioned in the Introduction section, similar results have also been reported for some other groups.
Several hypotheses could be proposed to explain these patterns.
-
1.
Null hypothesis: this pattern is an artifact of data selection, sampling or identification biases, or “distribution of taxonomists.” All the species lists, however, had been processed using the same checking and unification procedure to ensure their accordance with recent taxonomical revisions (see the “Materials and methods” section). Our analysis shows that neither sampling effort nor habitat diversity has significant effect on SESG values. Also, there are no reasons to suggest that more taxonomists “splitters” worked in the Arctic than in the tropics, and any “latitudinal” difference in their experience level is also doubtful. Nevertheless, for greater assurance, we performed an additional “pair-wise test” comparing Arctic versus tropical datasets reported by the same authors (or at least with the participation of the same authors). We found eight such pairs, and the SESG values were significantly lower for the Arctic datasets compared with their tropical “counterparts” (paired Wilcoxon test: p = 0.0078, n = 8). Thus, there is no evidence supporting the null hypothesis.
-
2.
The higher than expected S/G ratios near the pole might have resulted from relatively high net rates of speciation: the few lineages that manage to reach this zone are free to diversify there, producing more species per lineage than expected (the high-latitude diversification hypothesis, see Weir and Schluter 2007; Krug et al. 2008). However, this hypothesis is not supported by our data. There are precious few endemics at high latitudes (Fig. 1), and endemic genera in the Arctic have lower S/G ratios than those in the tropics (Table 1) (11 of 13 high-latitude endemic genera are monotypic, and all belong to the “old” families). Cosmopolitan genera have several more species, but they also tend to decline in species richness at high latitudes. These latitudinal differences in S/G ratios were most pronounced in the “young,” phylogenetically advanced families. Therefore, the higher than expected S/G ratios at polar zone are driven by genus depletion rather than by rapid diversification.
-
3.
“Out of tropics” hypothesis, or “equatorial pump” acting at the genus level. The low-latitude zone is considered the point of origin for many higher taxa that subsequently disperse polarward, though this dispersal is delayed and selective. This assumption, first made by Darlington (1957) and Meyen (1987) under the name of “equatorial pump,” was later developed and proved by Jablonski et al. (2006, 2013) as the mechanism of latitudinal diversity gradient formation (the “Out of tropics” hypothesis). Taking into consideration the high tropical origination rates and low polar extinction rates, this model treats polar diversity as a result of the accumulation of taxa that evolved elsewhere in the world (preferentially in the tropics) and then expanded their ranges poleward (Jablonski et al. 2006; Krug et al. 2008). The low total diversity of Arctic fauna indicates that not every taxon was able to pass through the climatic and geographical filters. If these successfully expanding taxa primarily belong to older and usually species-rich clades, this scenario should generally lead to higher S/G ratios at high latitudes than for rapidly diversifying, highly endemic tropical fauna.
Our results are consistent with this hypothesis. First, a lower percentage of endemics were found in the Arctic (both at the species and at genus levels, Fig. 3). Second, young families were the poorly represented in the Arctic and showed no endemic genera there, likely because they have had too little time for expansion and/or divergence. Third, it is the tendency for genera with large latitudinal ranges to be more species rich within each climatic zone (Table 1). Thus, those genera that reach the poles are not a random subset of the global biota.
Therefore, the global-level pattern agrees well with the “Out of tropics” model. However, this model appeals to evolutionary processes (i.e., speciation, extinction and range expansion) operating at large spatiotemporal scales, and thus, it is inapplicable to local-scale diversity patterns (Swenson 2011, Buschke et al. 2014). Our analysis reveals that Arctic and tropical assemblages do not differ in their average species richness (Fig. 7a, b); however, the Arctic assemblages are not simply an equivalent proportion of the regional diversity: they have significantly higher S/G ratios than expected (phylogenetic clumping, see Fig. 8; Table 2). This result was obtained by a comparison with random subsets from respective regional species pools and thus was exempted from global- and regional-level effects. Therefore, some additional, community-level mechanisms of ecological sorting should be suggested to explain this pattern.
Recently, Arita et al. (2014) applied a similar approach to analyze New World bats. They reported fewer genera than expected at high latitudes, whereas these figures were close to or slightly higher than expected in the tropics—the pattern similar to our “local-scale” one. Arita et al. grouped the data into one-degree latitudinal bins (approximately 111 km), a scale that is intermediate between our “local” and “regional” scales, but corresponding more closely to the local scale if accounting for the higher dispersal rate for bats than for benthic harpacticoids.
Ecological theory has yielded two main types of these sorting mechanisms:
-
1.
Competitive exclusion: closely related species, being also more similar ecologically, should compete most severely and thus should coexist more rarely than taxonomically distant ones, resulting in phylogenetic over-dispersion;
-
2.
Environmental filtering: closely related species with similar environmental preferences should co-occur more often in the same habitats, which should result in phylogenetic clustering.
Both mechanisms imply that more closely related species tend to show more similar ecological features than expected by chance (phylogenetic niche conservatism). In fact, both mechanisms can act simultaneously but are expected to have opposing effects; thus, the observed phylogenetic community pattern is a result of a balance between the two processes (Cavender-Bares et al. 2009; Mayfield and Levine 2010). A number of studies on various groups evidence that this balance depends on the spatial scale, taxonomic resolution and environmental heterogeneity (Azovsky 1992, 1996; Cavender-Bares et al. 2006, 2009; Stevens et al. 2011; Swenson 2011). Moreover, at certain scale the effects of these processes may compensate for each other, such that the resulting pattern becomes indistinguishable from the neutral model expectation (null balance, Azovsky 1996).
The results presented herein reveal the obvious latitudinal shift in this balance, with strong phylogenetic clumping at high latitudes. Within the aforementioned theoretical framework, two additional hypotheses can be proffered to explain this difference:
-
4.
Arctic communities face more severe environmental filtering than tropical ones;
and/or
-
5.
There is more severe competition in the tropics, which more frequently leads to local exclusion of relatives.
Our data do not allow us to prefer either (or both) of these hypotheses. More generally, both possibilities (i.e., latitudinal shifts in environmental filtering or in the strength of biotic interactions) have been discussed, but direct evidence is scarce and controversial. Tropical environments are widely assumed to be highly diversified, providing a greater variety of resources and thus more possibilities for fine niche specialization (Rohde 1978; Dyer et al. 2007; but see Beaver 1979; Novotny et al. 2006; Lewinsohn and Roslin 2008 for counter-evidence). Qiao et al. (2015) analyzed species abundance distribution data for 32 forest tree communities and found that the deviation from neutrality correlated with latitude; they interpreted this as an indication of an increase in the strength of environmental filtering in regions further from the tropics. Similarly, latitudinal differences in competition intensity are often assumed to be important (e.g., Luiselli 2006), but the evidence for this claim is again rare and ambiguous. Recent work synthesizing studies of marine sessile benthos (Barnes 2002; Barnes and Kukliński 2003) has suggested that interspecific competition at low latitudes is more frequent but less frequently leads to competitive exclusion. Notably, in their extensive review, Schemske et al. (2009) failed to offer any direct comparisons of the strength of interspecific competition across latitudes.
There are some evidences that local meiobenthic diversity is mainly driven by local environmental conditions, rather than by latitude (harpacticoids: Rybnikov et al. 2003; Azovsky et al. 2012, and the present study; nematodes: Mokievsky and Azovsky 2002; Fonseca and Netto 2014). So we prefer the environmental filtering as the most likely mechanism of local-scale phylogenetic clumping. Deeper studies are necessary, however, to make a reasonable judgment.
Conclusion
Our analysis reveals that the phylogenetic diversity of marine benthic harpacticoids is non-randomly filtered at all levels: zonal (i.e., high vs. low latitudes as a whole), regional and local. The signals of phylogenetic clumping are stronger at high (Arctic) latitudes. At zonal and regional levels, this difference could be explained within the framework of the “Out of tropics” model. The most obvious contrast between latitudes, however, appears at the local scale, indicating also the difference in some community-level mechanisms of species sorting. These results confirm that biodiversity is nested hierarchically across spatial scales (Buschke et al. 2014). The observed patterns in biodiversity, including latitudinal gradients, are therefore the result of multiple underlying evolutionary and ecological processes. Decomposing their effects and quantifying their relative roles would perhaps be more fruitful, rather than searching for a simple single-factor explanation unified across taxa, scales and regions. In particular, studies exploring biogeographical variations in local ecological processes are the next logical step toward this goal.
References
Arita HT, Vargas-Barón J, Villalobos F (2014) Latitudinal gradients of genus richness and endemism and the diversification of New World bats. Ecography 37:1024–1033. doi:10.1111/ecog.00720
Azovsky AI (1992) Co-occurrence of congeneric species of marine ciliates and the competitive exclusion principle: the effect of scale. Russ J Aquat Ecol 1:49–59
Azovsky AI (1996) The effect of scale on congeners coexistence: can mollusks and polychaetes reconcile beetles to ciliates? Oikos 77:117–126
Azovsky AI, Mazei YuA (2013) Do microbes have macroecology? Large-scale patterns in the diversity and distribution of marine benthic ciliates. Glob Ecol Biogeogr 22:163–172. doi:10.1111/j.1466-8238.2012.00776.x
Azovsky AI, Garlitska LA, Chertoprud ES (2012) Broad-scale patterns in local diversity of marine benthic harpacticoid copepods (Crustacea). Mar Ecol Prog Ser 460:63–77. doi:10.3354/meps09756
Barnes DKA (2002) Polarization of competition increases with latitude. Proc R Soc B 269:2061–2069. doi:10.1098/rspb.2002.2105
Barnes DKA, Kukliński P (2003) High polar spatial competition: extreme hierarchies at extreme latitude. Mar Ecol Prog Ser 259:17–28. doi:10.3354/meps259017
Beaver RA (1979) Non-equilibrium ‘island’ communities. A guild of tropical bark beetles. J Anim Ecol 48:987–1002
Boxshall GA, Halsey SH (2004) An introduction to copepod diversity. Ray Society, London
Buschke FT, Brendonck L, Vanschoenwinkel B (2014) Differences between regional and biogeographic species pools highlight the need for multi-scale theories in macroecology. Front Biogeogr 6:173–184
Cavender-Bares J, Keen A, Miles B (2006) Phylogenetic structure of Floridian plant communities depends on taxonomic and spatial scale. Ecology 87:S109–S122. doi:10.1890/0012-9658(2006)87[109:PSOFPC]2.0.CO;2
Cavender-Bares J, Kozak KH, Fine PVA, Kembel SW (2009) The merging of community ecology and phylogenetic biology. Ecol Lett 12:693–715. doi:10.1111/j.1461-0248.2009.01314.x
Chertoprud ES, Garlitska LA, Azovsky AI (2010) Large-scale patterns in marine harpacticoid (Crustacea, Copepoda) diversity and distribution. Mar Biodivers 40:301–315. doi:10.1007/s12526-010-0054-z
Clarke KR, Gorley RN (2006) PRIMER v6: user manual/tutorial. PRIMER-E, Plymouth
Darlington PJ (1957) Zoogeography: the geographical distribution of animals. Wiley, New York
Dyer LA, Singer MS, Lill JT et al (2007) Host specificity of Lepidoptera in tropical and temperate forests. Nature 448:696–699. doi:10.1038/nature05884
Fenner M, Lee WG, Bastow Wilson J (1997) A comparative study of the distribution of genus size in twenty angiosperm floras. Biol J Linn Soc 62:225–237. doi:10.1006/bijl.1997.0152
Finlay BJ (2002) Global dispersal of free-living microbial eukaryote species. Science 296:1061–1063. doi:10.1126/science.1070710
Fonseca G, Netto SA (2014) Macroecological patterns of estuarine nematodes. Estuaries Coasts 38:612–616. doi:10.1007/s12237-014-9844-z
Gaston KJ, Blackburn TM (2000) Pattern and process in macroecology. Blackwell, Oxford
Grytnes JA, Birks HJB, Heegaard E, Peglar SM (2010) Geographical trends in the species-to-family ratio of vascular plants in Fennoscandia. Nor Geogr Tidsskr 54:60–64. doi:10.1080/002919500423780
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontol Electron 4:1–9
Harnik PG, Jablonski D, Krug AZ, Valentine JW (2010) Genus age, provincial area and the taxonomic structure of marine faunas. Proc R Soc B 277:3427–3435. doi:10.1098/rspb.2010.0628
Huys R, Boxshall GA (1991) Copepod evolution. The Ray Society, London
Jablonski D, Roy K, Valentine JW (2006) Out of the tropics: evolutionary dynamics of the latitudinal diversity gradient. Science 314:102–106. doi:10.1126/science.1130880
Jablonski D, Belanger CL, Berke SK (2013) Out of the tropics, but how? Fossils, bridge species, and thermal ranges in the dynamics of the marine latitudinal diversity gradient. Proc Nat Acad Sci 110:10487–10494. doi:10.1073/pnas.1308997110
Krug AZ, Jablonski D, Valentine JW (2008) Species-genus ratios reflect a global history of diversification and range expansion in marine bivalves. Proc R Soc B 275:1117–1123. doi:10.1098/rspb.2007.1729
Lewinsohn TM, Roslin T (2008) Four ways towards tropical herbivore megadiversity. Ecol Lett 11:398–416. doi:10.1111/j.1461-0248.2008.01155.x
Luiselli L (2006) Resource partitioning and interspecific competition in snakes: the search for general geographical and guild patterns. Oikos 114:193–211. doi:10.1111/j.2006.0030-1299.14064.x
Mayfield MM, Levine JM (2010) Opposing effects of competitive exclusion on the phylogenetic structure of communities. Ecol Lett 13:1085–1093. doi:10.1111/j.1461-0248.2010.01509.x
Meyen SV (1987) Fundamentals of palaeobotany. Chapman and Hall, London
Mokievsky VO, Azovsky AI (2002) Re-evaluation of species diversity patterns of free-living marine nematodes. Mar Ecol Prog Ser 238:101–108. doi:10.3354/meps238101
Novotny V, Drozd P, Miller SE et al (2006) Why are there so many species of herbivorous insects in tropical rainforests? Science 313:1115–1118. doi:10.1126/science.1129237
Qiao X, Jabot F, Tang Z, Jiang M, Fang J (2015) A latitudinal gradient in tree community assembly processes evidenced in Chinese forests. Glob Ecol Biogeogr 24:314–323. doi:10.1111/geb.12278
Rohde K (1978) Latitudinal differences in host-specificity of marine Monogenea and Digenea. Mar Biol 47:125–134. doi:10.1007/BF00395633
Roy K, Jablonski D, Valentine JW (1996) Higher taxa in biodiversity studies: patterns from Eastern Pacific marine molluscs. Phil Trans R Soc B 351:1605–1613. doi:10.1098/rstb.1996.0144
Ruggiero A, Werenkraut V (2007) One-dimensional analyses of Rapoport’s rule reviewed through meta-analysis. Glob Ecol Biogeogr 16:401–414. doi:10.1111/j.1466-8238.2006.00303.x
Rybnikov PV, Kondar DV, Azovsky AI (2003) Properties of the White Sea littoral sediments and their influence on the fauna and distribution of Harpacticoida. Oceanology 43(1):91–102
Schemske DW, Mittelbach GG, Cornell HV, Sobel JM, Roy K (2009) Is there a latitudinal gradient in the importance of biotic interactions? Ann Rev Ecol Evol Syst 40:245–269. doi:10.1146/annurev.ecolsys.39.110707.173430
Seifried S (2003) Phylogeny of Harpacticoida (Copepoda): revision of “Maxillipedasphalea” and Exanechentera. Cuvillier Verlag, Göttingen
Simberloff DS (1970) Taxonomic diversity of island biotas. Evolution 24:23–47
Soininen J, Lennon JJ, Hillebrand H (2007) A multivariate analysis of beta diversity across organisms and environments. Ecology 88:2830–2838. doi:10.1890/06-1730.1
Stevens GC (1989) The latitudinal gradient in geographical range: how so many species coexist in the tropics. Am Nat 133:240–256
Stevens RD, Gavilanez MM, Tello JS, Ray DA (2011) Phylogenetic structure illuminates the mechanistic role of environmental heterogeneity in community organization. J Anim Ecol 81:455–462. doi:10.1111/j.1365-2656.2011.01900.x
Swenson NG (2011) The role of evolutionary processes in producing biodiversity patterns, and the interrelationships between taxonomic, functional and phylogenetic biodiversity. Am J Bot 98:472–480. doi:10.3732/ajb.1000289
Webb CO, Ackerly DD, McPeek MA, Donoghue MJ (2002) Phylogenies and community ecology. Ann Rev Ecol Syst 33:475–505. doi:10.1146/annurev.ecolsys.33.010802.150448
Weir JT, Schluter D (2007) The latitudinal gradient in recent speciation and extinction rates of birds and mammals. Science 315:1574–1576. doi:10.1126/science.1135590
Wells JBJ (2007) An annotated checklist and keys to the species of Copepoda Harpacticoida (Crustacea). Zootaxa 1568:1–872
Wiens JJ, Donoghue MJ (2004) Historical biogeography, ecology and species richness. Trends Ecol Evol 19:639–644. doi:10.1016/j.tree.2004.09.011
Acknowledgments
This study was supported by the Russian Foundation for Basic Research (Grant No. 15-04-02245) and the Russian Scientific Foundation (Grant No. 14-50-00029).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: S. Connell.
Reviewed by Undisclosed experts.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Azovsky, A., Garlitska, L. & Chertoprud, E. Multi-scale taxonomic diversity of marine harpacticoids: Does it differ at high and low latitudes?. Mar Biol 163, 94 (2016). https://doi.org/10.1007/s00227-016-2876-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-016-2876-0