Abstract
Motor unit recruitment was assessed in two muscles with similar muscle fiber-type compositions and that participate in skilled movements: the tongue muscle, genioglossus (GG), and the hand muscle, first dorsal interosseous (FDI). Our primary objectives were to determine in the framework of a voluntary movement whether muscle force is regulated in tongue as it is in limb, i.e., via processes of rate coding and recruitment. Recruitment in the two muscles was assessed within each subject in the context of ramp force (FDI) and in the tongue (GG) during vowel production and specifically, in the context of ramp increases in loudness, and subsequently expressed relative to the maximal. The principle findings of the study are that the general rules of recruitment and rate coding hold true for both GG and FDI, and second, that average firing rates, firing rates at recruitment and peak firing rates in GG are significantly higher than for FDI (P < 0.001) despite tasks performed across comparable force ranges (~2–40 % of max). The higher firing rates observed in the tongue within the context of phonation may be a function of that muscle’s dual role as (prime) mover and hydrostatic support element.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The human tongue and hand are capable of precisely controlled voluntary movement. For the tongue, these movements underpin critical functions including speech production and for the hand, grasp and manipulation of objects. The two structures have in common intrinsic and extrinsic musculatures that comprise roughly equivalent proportions of types I and II muscle fiber but differ in regard to their “skeletal” support. Thus, hand muscles attach to bone which supports posture and movement and serves to amplify force generated by muscle contraction. In contrast, tongue muscles lack bony support and thus serve both as prime movers and to generate internal pressure and stiffness to constitute a “hydrostatic skeleton” (Kier and Smith 1985; Smith and Kier 1989; Skierczynski et al. 1996).
Although there are numerous studies that document other (non-human) hydrostats performing skilled movements (Kier et al. 1989; Johnsen and Kier 1993; Gutfreund et al. 1996, 1998; Matzner et al. 2000), our understanding of the neuromuscular control of these structures in the context of voluntary movements is quite limited. Thus, it is unclear whether muscle force is regulated in the tongue as it is in limb, i.e., via processes of rate coding and recruitment. Here we perform a side-by-side comparison of muscle motor unit recruitment patterns in the tongue and in the hand in nine healthy young adults. Despite very different structures and functions, we find motor units in muscles of the hand and tongue are recruited in a similar manner over comparable effort ranges (%max). However, the absolute firing rate of motor units in the tongue muscle during articulation of a vowel is significantly higher than in a muscle of the hand during finger abduction. It is tempting to speculate that higher firing contributes to tongue muscle stiffness and in turn to the generation of the hydrostatic skeleton. This work was presented previously in abstract form (Shumway et al. 2014).
Methods
Thirty-two experiments were performed in nine healthy adult volunteers (six women and three men) between the ages of 20 and 34 years. Experimental procedures were approved by the University of Arizona, Human Subjects Committee. All subjects gave their written informed consent prior to participation in the study.
Procedures
We initially demonstrated the suitability of the protocol via intramuscular fine wire recordings of whole muscle GG and FDI EMG in performance of ramp force and loudness tasks (see Fig. 1). These recordings serve as verification that GG EMG is modulated as a function of increasing loudness and that ramp rise, envelope and duration for this task is comparable to the ramp rise, envelope and duration for force ramps performed by a hand muscle.
Representative whole muscle electromyographic recording obtained from the first dorsal interosseous (FDI) and genioglossus (GG) muscles in performance of force (%max) and loudness (%max) ramps, respectively. The whole muscle EMG output for FDI and GG is comparable in regard to trajectory, amplitude (mV) and time course
Each experimental session was preceded by a brief rehearsal period. Subjects subsequently performed 3–5 maximum voluntary maneuvers to establish the maximum force (FDI) and loudness (GG) EMG range generated in the tasks described below. The average of three maneuvers that were consistent to within ±10 % determined the subject’s maximum voluntary effort for each experiment.
Tongue tasks
Subjects sat in a dental chair with their head supported. Subjects were asked to produce the vowel /ee/ (as in the word /s ee d/) and to increase loudness or vocal effort over the course of the production. This vowel was selected because it is produced with slight mouth opening and with minimal jaw motion. Jaw opening is of significance for speech because aperture affects the radiation of sound and in turn perceived loudness. Moreover, jaw gape and movement reflexly modulate tongue muscle activity. Thus, jaw closure is a potent inhibitor of tongue protrusion (Ishiwata et al. 2000), presumably to prevent biting, whereas jaw depression or opening co-activates both tongue and jaw muscles (Ishiwata et al. 2000; Stanek et al. 2014). In selecting /ee/ as the target production, both sources of variability were effectively minimized.
Respiratory movements of the chest wall were monitored via two strain-gauge transducers (Pneumotrace, UFI, Morro Bay, CA) positioned around the thorax at the mid-sternal level and around the abdomen at the level of the umbilicus and which allowed us to distinguish inspiration from expiration-related segments of each breath cycle. Vowel productions commenced at end inspiration and were roughly 5.0 s in duration as determined by the individual’s available breath support. All utterances were recorded via a head-mounted omnidirectional microphone (Opus 55.18 MK II, BeyerDynamic, Long Branch, NJ) positioned ~2.0 mm from the left corner of the mouth and displayed in Spike2 (Cambridge Electronic Design, CED). Visual feedback of the time course and loudness of the utterance was displayed on a computer monitor positioned within the subject’s field of view.
Hand tasks
Subjects sat in a dental chair with their forearm supported on an adjustable platform. The upper arm was slightly abducted, and the elbow joint was flexed at 130°. The hand and forearm rested on a support with the palmar side of the hand turned downward and the thumb abducted and resting against a vertical aluminum rod. A leather cuff placed around the distal interphalangeal joint of the index finger was coupled to a force transducer. Each subject then was instructed to perform a slow ramp abduction of the index finger during which force increased over a ~5.0-s window. The force exerted by the subject was recorded by means of a force–displacement transducer (Grass FT10, Grass Instruments, Quincy MA), and the signal was amplified (×1000) (Transbridge 4M, WPI, Sarasota, FL) and displayed in Spike2 (Cambridge Electronic Design, CED). Subjects were provided visual feedback of the time course and amplitude of the force trace on the computer monitor.
Motor unit recording
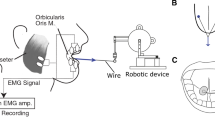
A total of 122 single motor unit recordings were obtained from the GG (N = 57 motor units) and FDI (N = 65 motor units) muscles. Each subject participated in both the GG and FDI experiments performed on separate occasions. Motor unit activities were recorded using tungsten microelectrodes (100–200 kΩ at 1 kHz, tip diameter, 250-μm shaft diameter; FHC, Bowdoinham, ME) inserted through the skin. For GG, depth to the muscle belly was determined via ultrasound and ranged between 1.5 and 2.0 cm, and for FDI, the electrode was inserted into the middle part of the muscle belly at depths ~1.0–1.5 cm. Surface electrodes (4 mm diameter Ag–AgCl) are attached to the skin overlying the mastoid process (GG), and the radial styloid (FDI) served as the reference electrode in each case. All intramuscular EMG signals were sampled at 20 kHz, amplified (2000) and band-pass filtered (0.1–3 kHz (Model P511, Grass Instruments, West Warwick, RI) and displayed on a digital oscilloscope.
In each experimental session, subjects were instructed either to produce the vowel /ee/ (GG) or to slowly abduct the index finger (FDI), while the microelectrode was manipulated until action potentials of motor units were identifiable on the electrode. Subsequently, single motor unit activities were recorded in each loudness or force ramp. Subjects completed 3–5 such ramps per trial and rested 1–2 min between trials.
Signal processing and statistical analysis
All data were acquired and analyzed using Spike2 and custom-designed software (CED). Average sound pressure level (dBSPL) and average force (N) attained in each ramp were expressed as a percentage of the maximum (%max). Quantitative comparisons of EMG activities were made as follows. Single motor unit action potentials were discriminated using a template-matching algorithm based on waveform shape and amplitude as discussed previously (Bailey et al. 2007b). For analysis of average discharge rate and variability, only those motor units whose activities could be followed throughout each series of loudness or force ramp tasks were included. The firing rate at recruitment was calculated from the first three interspike intervals (ISIs) in both muscles. In each muscle, the peak was identified as the highest firing rate recorded during each force or loudness ramp. Initial, average, peak and delta (peak firing rate–firing rate at recruitment) firing rates were calculated for each motor unit from three to five ramps recorded in each condition. Differences in single motor unit activities were assessed via a within-subject ANOVA testing main effects for muscle (%max GG vs. % max FDI) as a function of task (i.e., force or loudness). When a significant F value was obtained, differences were tested using post hoc contrasts with significance levels adjusted according to the Bonferroni procedure. A significance level of P < 0.05 was used for all comparisons.
Results
Figure 2 contains representative recordings obtained from one subject in two separate sessions and shows single motor unit activities recorded during force and loudness ramps, respectively. Despite the very different nature of the tasks performed by each muscle, the ramps share comparable trajectories and attain the same peak and motor units appear to be recruited into activity at roughly equivalent force/loudness levels (%max). The principle difference between the two recordings lies in peak motor unit firing rates which reach 20 Hz for GG and 14 Hz for FDI.
Representative recordings obtained from one subject performing ~5.0 s force and loudness ramps. a Representative recordings of two FDI muscle motor units (motor unit A and B) recruited into activity at ~2.0 and 8.0 % of max force, respectively. b Representative recordings of two GG muscle motor units also recruited into activity at ~2.0 and ~5.0 % of max loudness, respectively
Box plots and interquartile ranges displaying firing rate averages for initially recruited units are presented in each of the panels in Fig. 3 and confirm the results depicted in the individual recordings. As shown, firing rates at recruitment (GG: 18.6 ± 3.4 Hz and FDI: 10.9 ± 1.8 Hz) (Fig. 3a), average firing rates (GG: 12.9 ± 3.3 Hz and FDI:7.0 ± 2.3 Hz) (Fig. 3b), peak firing rates (GG: 29.0 ± 5.7 Hz and FDI:16.6 ± 3.96 Hz) (Fig. 3c) and the net change in firing rate from recruitment to peak firing rate (i.e., ∆ firing rate, Fig. 3d) all were higher in GG relative to FDI muscle motor units (P < 0.001). We also compared the firing rates of initially recruited motor units recorded (e.g., see MU A in Fig. 2) with the firing rates of subsequently recruited motor units in the same ramp (e.g., see MU B in Fig. 2). Peak firing rates for these later recruited motor units (see Fig. 3c) were characterized by slower average firing rates relative to initially recruited units (P < 0.001). Note that firing rates for both motor units A and B were consistently higher in GG than in FDI.
Panels a–d depict group means and interquartile ranges for motor unit firing rates at recruitment, average firing rate, peak firing rates and ∆ firing rate (Hz) for FDI and GG muscles. GG motor unit firing rates were consistently higher relative to FDI (indicated by asterisk, P < 0.001). Firing rates for initially recruited units (MU A) also were greater relative to subsequently recruited units (MU B) (indicated by asterisks P < 0.001) in both muscles. Note that firing rates of both FDI motor units (MU A and MU B) are lower than firing rates for both GG motor units (P < 0.001). The net change (∆) in discharge (i.e., peak firing rate–firing rate at recruitment) was greater for motor unit A in GG relative to motor unit A in FDI (indicated by asterisk, P < 0.001), but there was no difference in the ∆ firing rate between motor unit B in GG and motor unit B in FDI
Figure 4 shows the distribution of firing rates for all GG and FDI motor units as a function of force and loudness (%max). For the total number of units recorded in both experiments, recruitment occurred across a comparable force range encompassing ~2–40 (% of max). However, as shown in the frequency histograms, the majority of FDI motor units were recruited into activity relatively early in the force ramp and therefore at lower average forces, whereas GG motor units were recruited across a broader range of forces (P < 0.001).
GG and FDI muscle motor units (N = 122) displayed as a function of firing rate at recruitment (%max force/loudness). First recruited (MU A; N = 32) and subsequently recruited (MU B; N = 31) units in FDI muscle indicated by black squares and circles, respectively. First recruited (MU A; N = 32) and subsequently recruited (MU B; N = 27) units in GG muscle indicated by gray squares and circles, respectively. Histograms depict the distribution of firing rates for the two motor unit types. Although muscle motor units in both the hand and tongue were recruited across comparable force and loudness ranges (2–40 % of max), the majority of FDI units were recruited at a lower average force (4 % of max) relative to GG (14 % of max) motor units (P < 0.003)
Discussion
Summary
Muscular hydrostats including the elephant trunk, tentacles of cephalopods and the vertebrate tongue have been well studied from a kinematic perspective (Gilbert et al. 2007; Winkel and Schleichardt 2011; Zelman et al. 2013; Xing et al. 2014); however, much less is known about how they develop force and whether neuromuscular control in these structures is analogous to control in limb (Gutfreund et al. 1998). In this study, we characterize motor unit activity in a tongue muscle contrasting it with motor unit activity in a similarly specialized muscle of the hand. We show that muscle motor units of the GG and FDI are recruited over comparable loudness/force ranges encompassing ~2–40 % of maximum. A principle difference between the tongue and hand, however, lies in the absolute firing rates, which are significantly higher in GG than in FDI.
Critique of method
As muscle force increases, the activities of newly recruited motor units typically begin to overlap with and obscure the activities of initially recruited units. This poses a particular challenge to the experimentalist, and tracking single motor units beyond ~40 % of maximum seldom is exceedingly difficult to accomplish. Whether motor units recruited at forces in excess of 40 % behave similarly to motor units recruited at low and moderate force levels cannot be determined from the current data set. Second, although the electrodes used in both muscles were fabricated to moderate impedances, the pickup area for these electrodes is restricted to a region in the immediate vicinity of the electrode tip. Accordingly, we cannot exclude the possibility that motor units recruited into activity but which lie outside the pickup area may behave differently from those reported here.
Despite structural differences between the tongue and hand, the techniques used to assess muscle motor unit activities in each case are those devised for assessment of limb muscle motor unit activity. Accordingly, previous studies compared tongue and hand muscle function in regard to strength (Buehring et al. 2013), fatigue (Solomon et al. 2002; Adams et al. 2014a, b), movement precision (Sutton et al. 1977) and force regulation (Adams et al. 2014b). The results of this work indicate that the control is comparable in each structure although pinpoint accuracy of the hand appears somewhat greater—at least within the context of displacement (Sussman 1970).
Unlike previous studies in tongue in voluntary movements (Scardella et al. 1993; Mortimore et al. 1999; Solomon et al. 2000; Weijnen et al. 2000; Blumen et al. 2002; BuSha et al. 2002; Solomon and Munson 2004; Bailey et al. 2007a; Pittman and Bailey 2009), we asked subjects to produce the vowel /ee/ and to increase the loudness of the production over ~5.0 s. We show here that the magnitude of GG EMG in the loudness ramp approximated that for FDI during finger abduction (Fig. 1). Second, the magnitude of whole muscle EMG attained in the maximum loudness task is comparable to the magnitude of EMG attained in the maximum force task (range 0.6–1.0 mV) and falls within the range of forces reported for GG during tongue protrusion (range 0.5–1.5 mV) (Vranish and Bailey 2015). On this basis, the ramp loudness task appears to offer a suitable framework within which to assess tongue muscle activation and against which to compare muscle and motor unit activities in FDI.
Firing rate modulation
Importantly, the average and ∆ firing rates reported here in a loudness ramp are consistent with firing rates during protrusion and with firing rates reported in other cranial muscles including lateral pterygoid and temporalis MUs (12–24 Hz) (Phanachet et al. 2002) and laryngeal muscles during phonation (range, 5–30 Hz) (Luschei et al. 1999). One possible explanation for higher firing rates in GG relative to FDI may be in relation to the duration of the afterhyperpolarization (AHP). Because minimum firing rate is inversely related to the duration of the AHP after the action potential, GG motoneurons may possess briefer period AHPs than FDI motoneurons. Remarkably, although firing rate modulation is evident here in the context of phonation and in protrusion (Bailey et al. 2007b), there is little evidence of rate modulation in respiration-related contexts (Nicholas et al. 2010; Richardson and Bailey 2010; Saboisky et al. 2010). This disparity in GG motor unit firing rates for volitional versus respiratory conditions suggests that firing rates in the GG are more likely a function of the sources of drive impinging on the pool than whether the pool has a cranial/spinal location.
Regulating muscle force
Muscle force is generated by a combination of changes in the discharge rates of already active units and recruitment of previously silent motor units. In the GG, recruitment occurred across the breadth of the force range up to ~40 %, whereas in FDI, the majority of units were recruited early in the ramp at forces <20 % of maximum. The different recruitment distributions may reflect differences in how force is regulated in each case. Current estimates of motor unit number based on the compound muscle action potential indicate that there are 228 ± 45 motor units in FDI (Zhou et al. 2012) compared to ~1000–1500 motor units in GG (Atsumi and Miyatake 1987; O’Kusky and Norman 1995). The greater number of GG motor units may permit continued recruitment across the full range of forces, whereas in FDI, force increments may initially occur via recruitment and subsequently via mores subtle modulation of firing rate over the intermediate force range (Milner-Brown et al. 1973).
Overall, higher firing rates and an extended range of recruitment may also be a consequence of the GG’s role both as a prime mover and “skeletal” element. Muscular hydrostats are unique in regard to their soft-bodied composition because muscle activation moves but also confers stiffness required for structural stability against internal and external forces (Yekutieli and Flash 2009). Whereas speech sound articulation does not depend upon the generation of large forces against the palate or teeth, increases in GG firing rate and continuous recruitment observed in the context of a loudness task may underpin increases in lingual (internal) pressure and stiffness (Skierczynski et al. 1996). Internal pressure is critical in hydrostats for transference of force between muscles and underpins muscle co-activation (Yekutieli and Flash 2009). Thus, a pattern of motor unit activation that includes higher overall firing rates along with recruitment throughout the force range may contribute the “skeletal” stiffness against which other tongue muscles develop force.
Theoretical implications
Our understanding of the principles governing the nervous system control of skeletal motor behavior is derived from studies conducted in limb muscles that have clearly defined origins and insertions and that produce force across a joint. Cranial muscles differ from limb muscles in that they interdigitate extensively and do not span a joint. Although both GG and FDI are subject to voluntary control, control of tongue muscle motor units may differ from hand muscle motor units by virtue of the absence of a bony skeleton and the requirement that tongue muscles serve both as a prime mover and hydrostatic support element (Kier 2012). The present findings provide de novo observations regarding the voluntary modulation of muscle activities in a cranial motoneuron pool and demonstrate recruitment and firing rate modulation within the context of a behavior that begins to approximate the muscle’s function in life. In addition to providing insight into basic mechanisms of force regulation, the loudness task may be of use as an alternate framework within which to assess tongue muscle function.
References
Adams V, Mathisen B, Baines S, Lazarus C, Callister R (2014a) Reliability of measurements of tongue and hand strength and endurance using the Iowa Oral Performance Instrument with elderly adults. Disabil Rehabil. doi:10.3109/09638288.2014.921245
Adams V, Mathisen B, Baines S, Lazarus C, Callister R (2014b) Reliability of measurements of tongue and hand strength and endurance using the Iowa Oral Performance Instrument with healthy adults. Dysphagia 29:83–95. doi:10.1007/s00455-013-9486-5
Atsumi T, Miyatake T (1987) Morphometry of the degenerative process in the hypoglossal nerves in amyotrophic lateral sclerosis. Acta Neuropathol 73:25–31
Bailey EF, Rice AD, Fuglevand AJ (2007a) Firing patterns of human genioglossus motor units during voluntary tongue movement. J Neurophysiol 97:933–936. doi:10.1152/jn.00737.2006
Bailey EF, Rice AD, Fuglevand AJ (2007b) Firing patterns of human genioglossus motor units during voluntary tongue movement. J Neurophysiol 97:933–936. doi:10.1152/jn.00737.2006
Blumen MB, Perez de La Sota A, Quera-Salva MA, Frachet B, Chabolle F, Lofaso F (2002) Genioglossal electromyogram during maintained contraction in normal humans. Eur J Appl Physiol 88:170–177
Buehring B, Hind J, Fidler E, Krueger D, Binkley N, Robbins J (2013) Tongue strength is associated with jumping mechanography performance and handgrip strength but not with classic functional tests in older adults. J Am Geriatr Soc 61:418–422. doi:10.1111/jgs.12124
BuSha BF, Strobel RJ, England SJ (2002) The length-force relationship of the human genioglossus in patients with obstructive sleep apnea. Respir Physiol Neurobiol 130:161–168
Gilbert RJ, Napadow VJ, Gaige TA, Wedeen VJ (2007) Anatomical basis of lingual hydrostatic deformation. J Exp Biol 210:4069–4082
Gutfreund Y, Flash T, Yarom Y, Fiorito G, Segev I, Hochner B (1996) Organization of octopus arm movements: a model system for studying the control of flexible arms. J Neurosci 16:7297–7307
Gutfreund Y, Flash T, Fiorito G, Hochner B (1998) Patterns of arm muscle activation involved in octopus reaching movements. J Neurosci 18:5976–5987
Ishiwata Y, Ono T, Kuroda T, Nakamura Y (2000) Jaw-tongue reflex: afferents, central pathways, and synaptic potentials in hypoglossal motoneurons in the cat. J Dent Res 79:1626–1634
Johnsen S, Kier WM (1993) Intramuscular crossed connective-tissue fibers: skeletal support in the lateral fins of squid and cuttlefish (Mollusca, Cephalopoda). J Zool 231:311–338
Kier WM (2012) The diversity of hydrostatic skeletons. J Exp Biol 215:1247–1257. doi:10.1242/Jeb.056549
Kier WM, Smith KK (1985) Tongues, tentacles and trunks:the biomechanics and movement of muscular hydrostats. Zool J Linn Soc 83:207–324
Kier WM, Smith KK, Miyan JA (1989) Electromyography of the fin musculature of the cuttlefish Sepia officinalis. J Exp Biol 143:17–31
Luschei ES, Ramig LO, Baker KL, Smith ME (1999) Discharge characteristics of laryngeal single motor units during phonation in young and older adults and in persons with Parkinson disease. J Neurophysiol 81:2131–2139
Matzner H, Gutfreund Y, Hochner B (2000) Neuromuscular system of the flexible arm of the octopus: physiological characterization. J Neurophysiol 83:1315–1328
Milner-Brown HS, Stein RB, Yemm R (1973) Changes in firing rate of human motor units during linearly changing voluntary contractions. J Physiol 230:371–390
Mortimore IL, Fiddes P, Stephens S, Douglas NJ (1999) Tongue protrusion force and fatigability in male and female subjects. Eur Respir J 14(1):191–195
Nicholas CL, Bei B, Worsnop C et al (2010) Motor unit recruitment in human genioglossus muscle in response to hypercapnia. Sleep 33:1529–1538
O’Kusky JR, Norman MG (1995) Sudden infant death syndrome: increased number of synapses in the hypoglossal nucleus. J Neuropathol Exp Neurol 54:627–634
Phanachet I, Whittle T, Wanigaratne K, Murray GM (2002) Functional properties of single motor units in the inferior head of human lateral pterygoid muscle: task firing rates. J Neurophysiol 88:751–760
Pittman LJ, Bailey EF (2009) Genioglossus and intrinsic electromyographic activities in impeded and unimpeded protrusion tasks. J Neurophysiol 101:276–282
Richardson PA, Bailey EF (2010) Tonically discharging genioglossus motor units show no evidence of rate coding with hypercapnia. J Neurophysiol 103:1315–1321. doi:10.1152/jn.00686.2009
Saboisky JP, Jordan AS, Eckert DJ et al (2010) Recruitment and rate-coding strategies of the human genioglossus muscle. J Appl Physiol 109:1939–1949
Scardella AT, Krawciw N, Petrozzino JJ, Co MA, Santiago TV, Edelman NH (1993) Strength and endurance characteristics of the normal human genioglossus. Am Rev Respir Dis 148:179–184
Shumway K, Porfirio D, Bailey E (2014) Motor unit recruitment patterns in genioglossus and first dorsal interosseous. FASEB J 28(1102):1104
Skierczynski BA, Wilson RJ, Kristan WB Jr, Skalak R (1996) A model of the hydrostatic skeleton of the leech. J Theor Biol 181:329–342
Smith KK, Kier WM (1989) Trunks, tongues and tentacles: moving with skeletons of muscle. Am Sci 77:22–35
Solomon NP, Munson B (2004) The effect of jaw position on measures of tongue strength and endurance. J Speech Lang Hear Res 47:584–594
Solomon NP, Robin DA, Luschei ES (2000) Strength, endurance, and stability of the tongue and hand in Parkinson disease. J Speech Lang Hear Res 43:256–267
Solomon NP, Drager KD, Luschei ES (2002) Sustaining a constant effort by the tongue and hand: effects of acute fatigue. J Speech Lang Hear Res 45:613–624
Stanek E, Cheng S, Takatoh J, Han BX, Wang F (2014) Monosynaptic premotor circuit tracing reveals neural substrates for oro-motor coordination. Elife 3:e02511. doi:10.7554/eLife.02511
Sussman HM (1970) The role of sensory feedback in tongue movement control. J Aud Res 10:296–321
Sutton D, Larson CR, Taylor EM, Lindeman RC (1977) Isometric force control by human tongue and forefinger. Arch Oral Biol 22:33–36
Vranish JR, Bailey EF (2015) A comprehensive assessment of genioglossus electromyographic activity in healthy adults. J Neurophysiol:jn 00975:02014. doi:10.1152/jn.00975.2014
Weijnen FG, Kuks JB, van der Bilt A, van der Glas HW, Wassenberg MW, Bosman F (2000) Tongue force in patients with myasthenia gravis. Acta Neurol Scand 102:303–308
Winkel B, Schleichardt A (2011) On the derivation of passive 3D material parameters from 1D stress-strain data of hydrostats. J Biomech 44:2113–2117. doi:10.1016/j.jbiomech.2011.05.037
Xing F, Woo J, Ziemba JK, Stone M, Prince JL (2014) Tracking four dimensional muscle mechanics from high-resolution and tagged magnetic resonance imaging. J Acoust Soc Am 135:2196. doi:10.1121/1.4877164
Yekutieli Y, Flash T (2009) Biomechanics: hydroskeletal. In: Squire LR (ed) Encyclopedia of neuroscience, vol 2. Academic Press, Oxford, pp 189–200
Zelman I, Titon M, Yekutieli Y, Hanassy S, Hochner B, Flash T (2013) Kinematic decomposition and classification of octopus arm movements. Front Comput Neurosci 7:60. doi:10.3389/fncom.2013.00060
Zhou P, Li X, Rymer WZ (2012) Computing motor unit number index of the first dorsal interosseous muscle with two different contraction tasks. Med Eng Phys 34:1209–1212. doi:10.1016/j.medengphy.2012.06.011
Acknowledgements
We extend our thanks to Dr. Andrew Fuglevand for assistance in recording motor unit activities from the FDI and to Dr Gail Koshland for helpful comments on preliminary drafts of the manuscript. Special thanks to Jennifer Vranish M. S. and David Taska and to our subject participants. This work was supported by a Grant from the National Institute of Health (NIDCD 009587).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
All subjects gave their written informed consent prior to participation in the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shumway, K.R., Porfirio, D.J. & Bailey, E.F. Phonation-related rate coding and recruitment in the genioglossus muscle. Exp Brain Res 233, 2133–2140 (2015). https://doi.org/10.1007/s00221-015-4284-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-015-4284-y