Abstract
α-l-Arabinofuranosidases with an orchestral action of xylanolytic enzymes degrades the xylan in plant cell wall. In this study, heterologous expression, biochemical characterization, and synergistic action of α-l-Arabinofuranosidase from previously identified.Geobacillus vulcani GS90 (GvAbf) was investigated. The recombinant α-l-Arabinofuranosidase was overexpressed in Escherichia coli BL21 (λDE) and purified via His-tag Ni-affinity and size-exclusion chromatography. Optimum activity of the purified α-l-Arabinofuranosidase was obtained at pH 5 and at 70 °C. The GvAbf was active in a broad pH and temperature ranges; pH 4–9 and 30–90 °C, respectively. In addition, it retained most of its activity after an hour incubation at 70 °C and remained relatively stable at pH 3–6. GvAbf was quite stable against various metal ions. The kinetic parameters of GvAbf was obtained as Vmax and Km; 200 U/mg and 0.2 mM with p-nitrophenyl-α-l-arabinofuranoside and 526 U/mg and 0.1 mM with sugar beet arabinan, respectively. The synergistic action of GvAbf was studied with commercially available xylanase on juice enrichment of apples, grapes, oranges, and peaches. The best juice enrichment in terms of clarity, reducing sugar content, and yield, was achieved with GvAbf and xylanase together compared to treatment with xylanase and GvAbf alone in all fruits. The treatment with GvAbf and xylanase together lead to an increased juice yield by 26.56% (apple), 30.88% (grape), 40.00% (orange) and 32.20% (peach) as well as having a significant effect on juice clarity by an increase of % transmittance 47.26, 25.98, 41.77, and 44.97, respectively. The highest reducing sugar level of fruit juices also obtained with GvAbf and xylanase together compared to treatment with xylanase and GvAbf alone in all types of fruits. GvAbf and xylanase together as simultaneous synergistic manner may have an exciting potential for application in fruit juice processing.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Hemicellulose is one of the main components of plant cell wall and the second most abundant natural polysaccharide after cellulose [1]. To utilize hemicelluloses in a various biotechnological applications, the polymeric structure of hemicellulose has to be hydrolyzed into monosaccharides. Xylan, the main structure of hemicellulose, is degraded by an orchestral action of xylanolytic enzymes including α-l-Arabinofuranosidases (Abfs) (EC 3.2.1.55). The main role of α-l-Arabinofuranosidases is to break the terminal and non-reducing α-1,2-, α-1,3-, and/or α-1,5-l-arabinofuranoside substitutions into α-l-arabinosides such as arabinoxylan, l-arabinan and the other polysaccharides that include arabinose [2]. On the basis of amino acid sequence similarities and mode of action against different substrates, Abfs can be classified into five glycoside hydrolase (GH) families; GH3, 10, 43, 51, 54, and 62 [3].
Some reports have been documented about synergistic actions of Abf and other xylanolytic enzymes such as xylanase, β-xylosidase, acetyl esterase, and endo-(1,5)-α-l-arabinanase in a concerted manner against different type of heterogenous xylans. Accordingly, different combinations of enzyme coctail including Abf has a higher synergistic impact rather than individual Abf action [4,5,6,7,8]. Abf and xylanase together shows synergetically higher action than the combination of Abf and another hemicellulose against wheat arabinoxylan. This great synergy between α-l-Arabinofuranosidase and xylanase may be attributed that it can likely be one way for degradation of biomass using minimal enzyme loading [6].
Importantly, Abf and other hemicellulases like xylanase play synergistically a promising role in the agro-industrial process [5, 9, 10] containing beverage industry, wine industry, paper from pulp, increase of animal feedstuffs, improvement of bread and bioethanol production [11,12,13,14,15]. It has been shown that Abf and xylanase exhibit a synergistic manner in the pulp and paper industry using unbleached kraft pulp [16]. Another integration action among these enzymes has also shown in bioethanol production using wet arabinoxylan [6]. In this respect, although it is known that xylanase and Abf play a role individually on fruit juice clarification [17, 18] their synergistic action has not been reported for this process in beverage industry so far.
Abfs are widely distributed in bacteria, fungi and plants [4, 19, 20]. Recently, much effort has been devoted to identification of α-l-Arabinofuranosidases from extremophilic microorganisms, because the related industrial processes operate at high temperatures [21, 22]. For instance, in general, fermentation conditions are adjusted to minimum 60 °C, because high temperatures eliminate contamination problems and provide not only fluidic medium but also soluble substrates [23]. Up to date, many studies have been performed for isolation of Abfs from several sources [22, 24,25,26,27,28], but α-l-arabinofuranosidase activity has been shown in only three members of thermophilic Geobacillus genera; Geobacillus stearothermophilus, Geobacillus thermoleovorans and Geobacillus caldoxylolyticus [29,30,31]. So far, to our knowledge, Geobacillus vulcani (G. vulcani) has not been reported as a source for α-l-Arabinofuranosidase. Here, we reported the heterologous expression and biochemical characterization of α-l-Arabinofuranosidase from previously identified G. vulcani GS90 (GvAbf) by 16S rRNA [32] for the first time. In addition, the synergetical action of GvAbf with a commercially available xylanase was demonstrated as a novel application of apple, orange, grape and peach juice enrichment in the present study. Although GvAbf was effective on fruit juice enrichment process including clarification, reducing sugar content and yield, utilization of GvAbf with the xylanase enzyme as a synergistic manner displayed exciting potential for application in fruit juice processing.
Materials and methods
Materials
Sugar beet arabinan was purchased from Megazyme (Bray, Wirklow, Ireland). Genomic DNA Isolation Kit, PCR Amplification Kit, Gel Purification Kit, TA Cloning Kit and the restriction endonucleases were obtained from Fermentas (Fermentas-Life Science Technologies, Lithuania). All other chemicals including pNP-α-l-arabinofuranoside and endo β-1,4-xylanase were supplied by Sigma Chemical (St. Louis, MO, USA).
Bacterial strains and growth conditions
Previously identified G. vulcani GS90 (GvAbf) by 16S rRNA [32] was used as a source of α-l-Arabinofuranosidase and E. coli DH5α was used for molecular cloning purpose. This strain was grown in Luria–Bertani (LB) medium in the presence of ampicillin (100 µg ml−1). E. coli BL21 (λDE3) was used for expression of GvAbf in the presence of kanamycin (30 µg ml−1), chloramphenicol (20 µg ml−1) and isopropyl-β-d-thiogalactopyranoside (IPTG, 1 mM) in LB medium. The bacterial strain used in this study was from Balçova Geothermal region of İzmir and the xylanolytic activity of the strain was screened previously [33].
Cloning of GvAbf gene
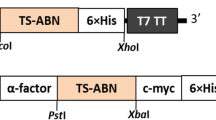
GvAbf gene was amplified from the genomic DNA of the bacterial strain using the following primers 5′-catatggctacaaaaaaagcaacc-3′ for forward and 5′-aagcttttatcgttttcctaaacg-3′ for reverse with the NdeI and HindIII sites (underlined), respectively. The amplified Abf gene was first cloned into pTZ57R/T vector (Fermentas-Life Science Technologies, Lithuania) and transformed to E. coli DH5α. After plasmid isolation, the DNA fragment was digested with NdeI and HindIII enzymes, gel-purified and cloned into NdeI/HindIII restricted pET-28a(+) expression vector (Novagen). The nucleotide sequence of the gene in the pET-28a(+) plasmid was determined and deposited in the EMBL Nucleotide Sequence Databases with the assigned accession number of HE653772.
Expression and purification of the GvAbf enzyme
Escherichia coli BL21 (DE3) harboring the IPTG inducible plasmid pET-28a(+) encoding for GvAbf enzyme was co-expressed with a chaperon protein; pG-Tf2 (Chaperone Plasmid, TAKARA Bio Inc., Japan), to increase recovery of the correctly folded target enzyme. For this purpose, this strain was grown at 200 rpm, 37 °C in LB medium (100 ml) with convenient antibiotics. When the OD600 reached to 0.5, co-expression is induced by addition of 1 mM IPTG and culture was more incubated at 225 rpm, 37 °C for 4 h. The cells were harvested by centrifugation at 10,000×g, 4 °C for 20 min. After resuspension of the cell pellet in 50 mM Na-P buffer pH 7.0, cell membranes were disrupted by sonication for 5 min on ice (Bandelin, Sonopuls Ultrasonic Homogenizers, HD 2070). Then, the cell debris was discarded by centrifugation at 16,000g, 4 °C for 15 min. The supernatant was subjected to nickel affinity chromatography for protein purification. The nickel column (HIS-Select® Nickel Affinity Gel, Sigma) was washed with 10 column volumes of ice cold 20 mM Tris–HCl buffer, pH 8.0 (300 mM NaCl, 10 mM of imidazole), and the bound proteins were eluted with elution buffer (250 mM imidazole, 100 mM NaCl in Tris–HCl buffer, pH 8.0). The proper fractions were pooled and concentrated using a cut-off filter (Amicon® Ultra-4 Centrifugal Filter Unit), and further purified by size-exclusion chromatography using Sepharose G-100 resin. The purified sample was checked for the purity of the target protein by SDS–PAGE analysis (10%). Quantitative protein determination was spectrophotometrically measured at 595 nm according to Bradford’s method [34]. Bovine serum albumin was used as a standard protein.
Biochemical characterization of GvAbf
Activity assay
Activity of GvAbf enzyme was determined via the previously described method by Canakci et al. with some modifications [31]. Briefly, the enzyme was assayed using the mixture contained 25 µl of 2 mM pNP-α-l-arabinofuranoside (pNP), 425 µl of McIlvaine buffer (pH 6.0) and 50 µl of enzyme solutions (0.1 µg). The hydrolysis reaction was carried out at 50 °C for 10 min and then terminated by the addition of 500 µl of 1.0 M Na2CO3 solution. The released pNP amount was determined at 420 nm with the extinction coefficient value of 1.78 × 104 M−1 cm−1. One unit of enzyme activity is defined as the amount of enzyme that releases 1 µmol of pNP per minute.
Sugar beet arabinan (2%, w/v) was also used as substrate to investigate the ability of GvAbf to release arabinose. For this purpose, the same assay procedure was used as described for pNP-α-l-arabinofuranoside, exceptionally at pH 5.5 and 65 °C. The dinitrosalicylic acid (DNS) method [35] was performed to investigate the reducing sugar content at the end of the reaction where l-arabinose was used as the standard. One unit of enzyme activity is defined as the amount of 1 µmol of arabinose that released by the enzyme per minute.
Optimum pH and temperature for enzyme activity
The effect of pH on enzyme activity was investigated using buffers at different pH, ranging from 3 to 12; Na–citrate buffer (pH 3–6), sodium phosphate buffer (pH 7–8), and glycine–NaOH buffer (pH 9–12). The optimum pH of the enzyme was determined under the standard assay conditions. The effect of temperature on enzyme activity was examined under standard assay conditions at different temperatures (30–90 °C) using the optimum buffer and the buffer was heated to the relevant temperature before the assay.
pH and temperature stability
GvAbf enzyme was also characterized by means of pH and temperature stability. To investigate the temperature stability, the enzyme was incubated in Na–citrate buffer (pH 5) at 50, 60, 70, 71, 72 and 75 °C for 1 h and the residual enzyme activity was measured after each incubation using standard activity assay. For the pH stability, GvAbf enzyme was incubated for 1 h at 70 °C in buffers; pH 3–10, and then the residual enzyme activity was determined by the standard activity assay.
Effect of metal ions and chemical reagents on the GvAbf activity
To assess the influence of metal ions (CuSO4, KCl, CaCl2, MgCl2, MnCl2, CdSO4, NaF, CoCl2, NaCl and NiCl2) and chemical reagents (DTT, EDTA and BME, ethanol, hexane, methanol, ethyl acetate, acetone, acetonitrile), GvAbf enzyme was incubated in the presence these chemicals at room temperature for 1 h. The reaction was initiated by addition of pNP-α-l-arabinofuranoside and then the remaining enzyme activity was determined by the standard activity assay.
Kinetic studies
Kinetic parameters of GvAbf enzyme were determined using Lineweaver–Burk plots and assuming that the reactions followed a simple Michaelis–Menten kinetics. Lineweaver–Burk plots were obtained both for pNP-α-L-arabinofuranoside and for sugar beet arabinan using standard activity assay.
Enzymatic hydrolysis in fruit juice enrichment
GvAbf enzyme was tested for its ability to enrich fruit juices in terms of clarity, yield end reducing sugar content. In addition, the synergetical action of GvAbf was investigated with a commercially available xylanase in fruit juice clarification, yield and reducing sugar content processes in a similar way. Fresh fruits (apple, orange, grape and peach) were used for preparing fruit juice. They were collected from local vendor, carefully washed, dried in air and homogenized. Homogenized fruit juices were treated with 1 mg/ml of pure GvAbf enzyme at 50 °C for 4 h. At the end of treatments, the juice samples were centrifuged at 5000g for 10 min. Supernatants were analyzed for clarity, reducing sugar and yield. The clarity of the fruit juices was determined by measuring the percent transmittance (%T) at a wavelength of 660 nm using UV–Vis spectrophotometer. The percent transmittance was considered a measure of juice clarity. The yield of juices was detected by measuring the volume of the fruit juice after centrifugation. Furthermore, release of reducing sugars from fruit juices was examined by DNS method [35, 36]. To compare the fruit juice enrichment effect of xylanase, the similar studies were performed with a commercially available endo β-1,4-xylanase (Sigma) under the same conditions. Each fruit juices after homogenization were treated with 20 IU/ml endo β-1,4-xylanase at 50 °C for 4 h. At the end of treatments, the juice samples were centrifuged and analysis of clarity, reducing sugar and yield were performed like GvAbf application. The synergistic effect of GvAbf enzyme with endo β-1,4-xylanase was also studied for the same fruit juice enrichment tests including clarity, yield and release of reducing sugar level. Both enzymes (1 mg/ml of pure GvAbf and 20 IU/ml endo β-1,4-xylanase) together were applied to the indicated fruit juices using the same procedures and analysis of clarity, reducing sugar and yield were determined.
Data presentation and statistical analysis
All experiments were conducted in triplicate. Statistical errors of the data were determined using Original Software, version 5.0 Original Lab and Excel Software, Microsoft.
Bioinformatic analysis
In this study, amino acid sequence alignment was used by BLAST to investigate the similarity of GvAbf with the other Abf enzymes from different sources. Phylogenetic and molecular evolutionary analyses were conducted using MEGA version 6 [37].
Results and discussion
Hemicelluloses are mainly composed of xylans, and degradation of this component is dependent on the orchestral action of xylanolytic enzymes such as Arabinofuranosidases and xylanases [2]. We reported an α-l-Arabinofuranosidase from previously identified thermophilic bacterium G. vulcani GS90 [32] which releases arabinose moiety from sugar beet arabinan. After the enzyme characterization, we investigated the synergetical action of GvAbf in fruit juice enrichment process.
Amino acid sequence of GvAbf
GvAbf gene was isolated from chromosomal DNA of G. vulcani GS90 and its length was determined as 1509 bp encoding 502 amino acids. Based on the sequence analysis, it was shown that GvAbf is an α-l-Arabinofuranosidase belonging to GH51 family. Significantly, GvAbf has a 98.6% amino acid sequence identity to the α-l-Arabinofuranosidase from G. stearothermophilus T-6. GvAbf also has Glu175 (acid–base catalytic residue) and Glu294 (catalytic nucleophile residue) which were reported to be responsible for the catalytic activity [38]. In addition, GvAbf showed 89.7% identity to α-l-Arabinofuranosidase from G. caldoxylosilyticus [31]. Yet, there was low identity between GvAbf and α-l-Arabinofuranosidase from T. xylanilyticus by 26.3% [21]. The phylogenetic tree was constructed considering the amino acid sequences of the α-l-Arabinofuranosidases in GH51 family by maximum likelihood (ML) method to determine the evolutionary connection of GvAbf. Apparently, GvAbf had the closest connection with α-l-Arabinofuranosidase of G. stearothermophilus T-6 (Fig. 1).
Expression and purification of GvAbf
GvAbf, consisting of 502 amino acids was co-expressed in the presence of a chaperon protein pG-Tf2 as described in the method section. The GvAbf was purified to homogeneity using conventional Ni–NTA affinity purification followed by size-exclusion chromatography. The recovery of the expressed GvAbf in the soluble fraction was increased significantly by the help of a chaperon protein. The sample purity was assessed by SDS–PAGE analysis confirming the molecular mass of the GvAbf around 60 kDa (Fig. 2).
Biochemical characterization of GvAbf
Purified recombinant GvAbf was characterized and the results were summarized in Fig. 3 and Tables 1 and 2. The GvAbf activity were evaluated in the temperature interval of 30–90 °C and pH interval of 3–12 (Fig. 3a, b). The enzyme showed hydrolytic activity in broad pH and temperature range, and the maximum activity was observed at 70 °C and pH 5. In addition, the effect of different pH and temperature values on the GvAbf stability were determined (Fig. 3c, d). GvAbf was quite stable at at high temperatures and the thermal stability tests showed that the enzyme remained relatively stable even after 1 h of incubation at 70 °C without a major activity loss (~ 90%). In terms of pH stability, GvAbf had an interesting profile. The enzyme was stable at pH 4, 5, 8 and 9 after 1 h of incubation at 70 °C; however, at pH 6 and 7, after 30 min incubation, it started to lose its activity. Especially, the enzyme activity was reduced by half at pH 6, and almost 80% was lost at pH 7 after 1 h of incubation (Fig. 3d). In the case of pH stability, GvAbf exhibited better profile under weak acidic conditions than neutral and alkaline pH values. At optimum pH and temperature values, GvAbf retained 90% of activity after 1 h of incubation. α-l-Arabinofuranosidase from G. caldoxylolyticus TK4 kept its full activity at the optimum working conditions (pH 6.0 and at 75 °C after 12 h of incubation) [31]. In addition, α-l-Arabinofuranosidases from C. saccharolyticus and C. thermocellum have similar optimum temperature values, 80 and 82 °C, respectively [39, 40]. C. saccharolyticus has optimum pH 5.5 [39], which is nearly the same optimum working pH as GvAbf. Interestingly, α-l-Arabinofuranosidase from G. stearothermophilus T-6 has the same optimum working temperature [30] as GvAbf which correlates with the above mentioned sequence identity. α-l-Arabinofuranosidases from T. maritima and T. petrophila have different optimum temperature values as 90 and 70 °C and exhibited the optimum working pH as 7 and 6, respectively [41, 42]. Another α-l-Arabinofuranosidase from T. xylanilyticus shows 75 °C as optimum temperature. In summary, it was concluded that α-l-Arabinofuranosidases from gram positive bacteria have weak acid optimal Ph, while those from gram negative have slightly higher optimal pH.
Effects of pH and temperature on the activity and stability of the purified Gv α-l-Arabinofuranosidase. a Effect of pH, b temperature on the GvAbf activity. c pH, and d temperature stability of the GvAbf. The residual activity was monitored, and the maximum activity was defined as 100%. Values shown were the mean of the duplicate experiments
The activity of GvAbf was measured under standard assay conditions in presence of several metal ions and chemicals. Different effects on the GvAbf activity was observed in the presence of various ions and chemicals on the hydrolysis of pNP-α-l-arabinofuranoside. Effects of metal ions, chemicals and organic solvents on GvAbf activity were listed in Table 1. The enzyme activity was highly stimulated in the presence of 1 mM Mn2+, Co2+ and 1% EtOAc (v/v). Similar stimulating effect was observed on α-l-Arabinofuranosidase activity from Geobacillus Caldoxylolyticus TK4 with Mn2+ and Co2+ ions [31]. Similarly, 1 mM Mn2+ ions had small stimulating effect on the activity of α-l-Arabinofuranosidase activity from Bacillus stearothermophilus T-6 [30], endoarabinanase from Thermotoga thermarum [25] and α-L-Arabinofuranosidase from Paenibacillus sp. DG-22 [24]. However, no effect on activity of alpha-l-Arabinofuranosidase from Anoxybacillus kestanbolensis AC26Sari was detected in presence of 1 mM Mn2+, Co2+ [22]. 1 mM Ca2+, K+, Cu2+, Ni2+ and DTT had slight stimulating effect on the activity of enzyme. Addition of 1 mM Na+, Cd2+, Mg2+, EDTA and 1% BME was not significantly changed the enzyme activity. Since the addition of EDTA did not affect the enzyme activity significantly, this result suggests that no metals are needed for enzymatic activity [24, 30]. Furthermore, 1 mM Zn2+ has inhibitory effect on the enzyme activity displaying 57% residual activity. Similar results were obtained with α-l-Arabinofuranosidase from Paenibacillus sp. DG-22 [24]. Interestingly, Zn2+ ions had no detected effect on α-l-Arabinofuranosidase activity from G. stearothermophilus T-6 [30]. However, in the presence of Mn2+ ion as well as Cu2+, Zn2+, and Ni2+ had a significant decrease on alpha-l-Arabinofuranosidase activity from Thermotoga maritima Msb8 [41] and GH43 endoarabinanase from Thermotoga thermarum [25]. Most of the solvents tested; acetone, hexane, ethanol, methanol and acetonitrile did not affect the hydrolysis of the substrate significantly.
Kinetic parameters of GvAbf were determined with two different substrates; pNP-α-l-arabinofuranoside and sugar beet arabinan (Table 2). The GvAbf exhibited a simple Michaelis–Menten kinetics and Lineweaver–Burk plot showed a linear response over the tested concentration range in both cases. GvAbf has a higher affinity to pNP-α-l-arabinofuranoside and sugar beet arabinan compared to the other α-l-Arabinofuranosidases from GH51 superfamily with the exception of the one from G. caldoxylolyticus TK4 [31]. Regarding the Km values with sugar beet arabinan, GvAbf has about 200-fold higher affinity than α-l-Arabinofuranosidase from Thermotoga petrophila [43], showing the specificity of GvAbf to its natural substrate. Interestingly, α-l-Arabinofuranosidase from the host G. stearothermophilus had twofold higher Km value with pNP-α-l-arabinofuranoside [30], comparing to GvAbf.
To assure GvAbf hydrolysis activity on its substrate, the products of sugar beet arabinan as a results of treatment with GvAbf enzyme were analyzed via thin layer chromatography (TLC) (data not shown). TLC analysis indicated that arabinose was the only product to be released from sugar beet arabinan by the enzyme. Non treated arabinan with GvAbf did not give any product spots detected by TLC as expected. This result indicated that GvAbf enzyme is an exo-acting enzyme that hydrolyzes the nonreducing end of its substrate. The finding here was consistent with other previously reported Abfs hydrolysis activity [22, 26, 31].
Synergistic action of GvAbf in fruit juice enrichment
Polysaccharides and hemicellulosic components are responsible for turbidity and viscosity of the raw juices; therefore, carbohydrate and hemicellulose degrading enzymes have been exploited recently for extraction and enrichment of fruit juices in terms of yield, clarification and quality characteristics of juices [44]. Abfs are also known as accessory enzymes working with other xylanolytic enzymes together via an orchestral action. It is well know that Abf has a higher synergistic impact especially with xylanase rather than individual Abf action [4,5,6,7,8]. Importantly, Abf and other hemicellulases like xylanase play synergistically a promising role in the beverage industry [5, 45].
In the present study, the synergistic role of the GvAbf was investigated in fruit juice enrichments of apple, grape, peach and orange pulps. For this purpose, a commercially available xylanase enzyme was also used in our studies. The effect of xylanase has been previously demonstrated in many studies for fruit juice extraction and clarification process [17, 36, 46,47,48]. Therefore, the clarity level, extraction yield and the reducing sugar content of variety fruit juices were tested applying GvAbf and xylanase in separate and combined manner in this study.
Treatment with GvAbf itself led to increase of 10.93, 2.94, 10.16 and 7.27% in the yield of apple, grape, peach and orange juices, respectively, compared to control untreated juice (Table 3). Obviously, commercial xylanase displayed better yield compared to GvAbf alone. The best results were obtained with simultaneous treatment of both enzymes with 26.56, 30.88, 32.20 and 40.00% increase in the yield of apple, grape, peach and orange juices, respectively, compared to untreated juice as control. The increase in the yield may be correlated with the breakdown of all polymeric carbohydrates, hemicelluloses and starches of the pulp, thus increasing the yield of juice by enabling better processing [44]. Similar results have been achieved in presence of xylanase enzyme for increasing yield of fruit juices and the maximum apple juice yield is obtained as 15.35% in presence of xylanase from Streptomyces sp. [17]. So far, there is no any results has been reported with any Abfs in terms of increasing fruit juice yield in the literature.
The juice clarity, measured in terms of % transmittance (%T) at 660 nm, was also significantly affected by increase of 47.26, 25.99, 44.97 and 41.77 %T in apple, grape, peach and orange juice, respectively, in presence of both GvAbf and xylanase enzymes (Table 3). The efficiency of GvAbf in clarity of all treated fruit juices was very low (3.49 for grape, 10.88 for orange, 5.50% for peach and 14.36% for apple); however, it displayed additive effect with the xylanase enzyme together. The effect of xylanase itself was better compared to GvAbf treatment for the clarity of all tested juices in our studies. Several reports are available on the use of pectinase and other enzymes for clarification of fruit juice but literature citing the use of xylanases for the clarification of fruit juice is hardly available [17, 48] and there is no any study with Abfs for the same purpose so far.
The reducing sugar content in the all enzyme treated and extracted juices in our studies was also found to be more than control juices (Table 3). The maximum increase of reducing sugar level was observed in the case of grape juice among all GvAbf treated samples with an increase of 6.57% (apple was 3.10%, orange was 2.13% and peach was 0.91%). The effect of xylanase on peach juice displayed better increase in terms of reducing sugar level (41.17%) compared to untreated control and it was followed by grape (14.75%), orange (7.04%), and apple (9.80%) in our studies. Even though the highest reducing sugar level was observed in apple juice (1151.19 µg/ml), the maximum increase of reducing sugar level was achieved with peach juice (%51.10) in simultaneously treatment of GvAbf and xyalanse. The similar studies in terms of release in reducing sugar level were obtained with xylanase enzyme for the application of fruit juices [17, 36, 46]. Similarly, the release of reducing sugar contents of apple, grape and orange juices were improved by 17.21, 16.79, and 19.57%, respectively, with xylanase from Streptomyces sp. [17]. In another study, addition of xylanase from Geobacillus sp. TF16 to apple and orange juices increased the reducing sugar content 1.03 and 2.85-fold, respectively [46]. Xylanase from Sclerotinia sclerotiorum S2 fungus also improved the reducing sugar level of orange and apple juice 5 and 10 times, respectively after 24 h treatment [36]. Up to date, there is no any study related to reducing sugar level of fruit juices treated with any Abf enzyme.
Conclusions
In this study, for the first time, a thermostable α-l-Arabinofuranosidase (GvAbf) from previously identified G. vulcani GS90 by 16S rRNA was cloned in E. coli DH5α and expressed in E. coli BL21 (λDE). The biochemical properties were obtained in terms of utilization of this enzyme as a potential candidate in variety of industry. Well known synergistic role with xylanase enzymes also observed in the fruit juice extraction and clarification process. GvAbf obtained from G. vulcani GS90 could be remarkably helpful in facilitating industrial application as an accessory enzyme, especially in fruit juice enrichment processes.
References
Taiz L, Zeiger E (2010) Plant physiology, 3rd edn. Sinauer Associates, Sunderland
Kaji A (1984) l-Arabinosidases. Chem Biochem 42:383–394
Hoffmam ZB, Oliveira LC, Cota J, Alvarez TM, Diogo JA, de Oliveira Neto M, Citadini APS, Leite VBP, Squina FM, Murakami MT, Ruller R (2013) Characterization of a hexameric exo-acting GH51 α-l-Arabinofuranosidase from the mesophilic Bacillus subtilis. Mol Biotechnol 55:260–267
Hashimoto T, Nakata Y (2003) Synergistic degradation of arabinoxylan with alpha-l-Arabinofuranosidase, xylanase and alpha-xylosidase from soy sauce koji mold, Aspergillus oryzae, in high salt condition. J Biosci Bioeng 95:164–169
Raweesri P, Riangrungrojana P, Pinphanichakarn P (2008) α-l-Arabinofuranosidase from Streptomyces sp. PC22: purification, characterization and its synergistic action with xylanolytic enzymes in the degradation of xylan and agricultural residues. Biores Technol 99:8981–8986
Yang W, Bai Y, Yang P, Luo H, Huang H, Meng K, Shi P, Wang Y, Yao B (2015) A novel bifunctional GH51 exo-α-l-Arabinofuranosidase/endo-xylanase from Alicyclobacillus sp. A4 with significant biomass-degrading capacity. Biotechnol Biofuels 8:1–11
Park J-M, Jang M-U, Oh GW, Lee E-H, Kang J-H, Song Y-B, Han NS, Kim T-J (2015) Synergistic action modes of arabinan degradation by exo- and endo-arabinosyl hydrolases. J Microbiol Biotechnol 25:227–233
Bouraoui H, Desrousseaux M-L, Ioannou E, Alvira P, Manaï M, Rémond C, Dumon C, Fernandez‑Fuentes N, O’Donohue MJ (2016) The GH51 α-l-Arabinofuranosidase from Paenibacillus sp. THS1 is multifunctional, hydrolyzing main-chain and side-chain glycosidic bonds in heteroxylans. Biotechnol Biofuels 9:1–14
Aryan A-P, Wilson B, Strauss C-R, Williams P-J (1987) The properties of glycosidases of Vitis vinifera and a comparison of their β-glucosidase activity with that of exogenous enzymes: an assessment of possible applications in enology. Am J Enol Vitic 38:182–188
Saha B (2000) Alpha-l-Arabinofuranosidases: biochemistry, molecular biology and application in biotechnology. Biotechnol Adv 18:403–423
Campbell GL, Bedford MR (1992) Enzyme applications for monogastric feeds: a review. Can J Anim Sci 72:449–466
Viikari L, Kantelinen A, Sundquist J, Linko M (1994) Xylanases in bleaching from an idea to the industry. FEMS Microbiol Rev 13:335–350
Numan M, Bhosle N (2006) Alpha-l-Arabinofuranosidases: the potential applications in biotechnology. J Ind Microbiol Biotechnol 33:247–260
Himmel ME, Ding SY, Johnson DK, Adney WS, Nimlos MR, Brady JW, Foust TD (2007) Biomass recalcitrance: engineering plants and enzymes for biofuels production. Science 315:804–807
Nawel B, Saida B, Estelle C, Hakima H, Duchironb F (2011) Production and partial characterization of xylanase produced by Jonesia denitrificans isolated in Algerian soil. Process Biochem 46:519–525
Makkonen HP, Nakas JP (2005) Use of xylanase and Arabinofuranosidase for arabinose removal from unbleached kraft pulp. Biotechnol Lett 27:1675–1679
Adigüzel AO, Tunçer M (2016) Production, characterization and application of a xylanase from Streptomyces sp. AOA40 in fruit juice and bakery industries. J Food Biotechnol 30:189–218
Rosmine E, Sainjan NC, Silvester R, Alikkunju A, Varghese SA (2017) Statistical optimisation of xylanase production by estuarine Streptomyces sp. and its application in clarification of fruit juice. J Genet Eng Biotechnol 15:393–401
Lee RC, Hrmova M, Burton RA, Lahnstein J, Fincher GB (2003) Bifunctional family 3 glycoside hydrolases from barley with alpha-lArabinofuranosidase and alpha-d-xylosidase activity: characterization, primary structures and COOH-terminal. J Biol Chem 278:5377–5387
Rahman AK, Kato K, Kawai S, Takamizawa K (2003) Substrate specificity of the alpha-l-Arabinofuranosidase from Rhizomucor pusillus HHT-1. Carbohydr Res 338:1469–1476
Debeche T, Cummings N, Connerton I, Debeire P, O’Donohue MJ (2000) Genetic and biochemical characterization of a highly thermostable alpha-l-Arabinofuranosidase from Thermobacillus xylanilyticus. Appl Environ Microb 66:1734–1736
Canakci S, Kacagan M, Inan K, Belduz AO, Saha BC (2008) Cloning, purification, and characterization of a thermostable alpha-l-Arabinofuranosidase from Anoxybacillus kestanbolensis AC26Sari. Appl Microbiol Biotechnol 81:61–68
Morozkina EV, Slutskaya ES, Fedorova TV, Tugay TI, Golubeva LI, Koroleva OV (2010) Extremophilic microorganisms: biochemical adaptation and biotechnological application (review). Appl Biochem Microbiol 46:1–14
Lee SH, Lee YE (2014) Cloning, expression, and characterization of a thermostable GH51 α-l-Arabinofuranosidase from Paenibacillus sp. DG-22. J Microbiol Biotechnol 24:236–244
Shi H, Ding H, Huang Y, Wang L, Zhang Y, Li X, Wang F (2014) Expression and characterization of a GH43 endoarabinanase from Thermotoga thermarum. BMC Biotechnol 14:1–9
Ahmed S, Luis AS, Bras JLA, Ghosh A, Gautam S, Gupta MN et al (2013) A novel α-l-Arabinofuranosidase of family 43 glycoside hydrolase (Ct43Araf) from Clostridium thermocellum. PLoS One 8(9):e73575
Wang W, Mai-Gisondi G, Stogios PJ, Kaur A, Xu X, Cui H, Turunen O, Savchenko A, Master ER (2014) Elucidation of the molecular basis for arabinoxylan-debranching activity of a thermostable family GH62 α-l-Arabinofuranosidase from Streptomyces thermoviolaceus. Appl Environ Microbiol 80:5317–5329
Kaur A-P, Nocek B-P, Xu X, Lowden M-J, Leyva J-F, Stogios P-J, Cui H, Leo R-D, Powlowski J, Tsang A, Savchenko A (2015) Functional and structural diversity in GH62 α-l-Arabinofuranosidases from the thermophilic fungus Scytalidium thermophilum. Microb Biotechnol 8:419–433
Bezalel L, Shoham Y, Rosenberg E (1993) Characterization and delignification activity of a thermostable α-l-Arabinofuranosidase from Bacillus Stearothermophilus. Appl Microbiol Biotechnol 40:57–62
Gilead S, Shoham Y (1995) Purification and characterization of α-l-Arabinofuranosidase from Bacillus stearothermophilus T-6. Appl Environ Microbiol 61:170–174
Canakci S, Belduz AO, Saha BC, Yasar A, Ayaz FA, Yayli N (2007) Purification and characterization of a highly thermostable α-l-Arabinofuranosidase from Geobacillus Caldoxylolyticus Tk4. Appl Microbiol Biotechnol 75:813–820
Sürmeli Y (2013) Investigation of alkaline and thermal stability of alpha-l-Arabinofuranosidase produced by directed evolution. Unpublished master thesis, İzmir Institute of Technology, İzmir
Yavuz E, Gunes H, Harsa S, Yenidunya AF (2004) Identification of extracellular enzyme producing thermophilic bacilli from Balcova (Agamemnon) geothermal site by ITS rDNA RFLP. J Appl Microbiol 97:810–817
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–251
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 3:426–428
Olfa E, Mondher M, Issam S, Ferid L, Nejib MM (2007) Induction, properties and application of xylanase activity from Sclerotinia Sclerotiorum S2 fungus. J Food Biochem 31:96–107
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Shallom D, Belakhov V, Solomon D, Shoham G, Baasov T, Shoham Y (2002) Detailed kinetic analysis and identification of the nucleophile in alpha-l-Arabinofuranosidase from Geobacillus Stearothermophilus T-6, a family 51 glycoside hydrolase. J Biol Chem 277:43667–43673
Lim Y-R, Yoon R-Y, Seo E-S, Kim Y-S, Park C-S, Oh D-K (2010) Hydrolytic properties of a thermostable α-l-Arabinofuranosidase from Caldicellulosiruptor saccharolyticus. J Appl Microbiol 109:1188–1197
Taylor EJ, Smith NL, Turkenburg JP, D’Souza S, Gilbert HJ, Davies GJ (2006) Structural insight into the ligand specificity of a thermostable family 51 Arabinofuranosidase, Araf51, from Clostridium thermocellum. Biochem J 395:31–37
Miyazaki K (2005) Hyperthermophilic alpha-l-Arabinofuranosidase from Thermotoga maritima Msb8: molecular cloning, gene expression, and characterization of the recombinant protein. Extremophiles 9:399–406
dos Santos C, Squina F, Navarro A, Oldiges D, Leme A, Ruller R et al (2011) Functional and biophysical characterization of a hyperthermostable GH51 α-l-Arabinofuranosidase from Thermotoga petrophila. Biotech Lett 33:131–137
Squina FM, Santos CR, Ribeiro DA, Cota J, de Oliveira RR, Ruller R, Mort A, Murakami MT, Prade RA (2010) Substrate cleavage pattern, biophysical characterization and low-resolution structure of a novel hyperthermostable arabinanase from Thermotoga petrophila. Biochem Biophys Res Commun 399:505–511
Sharma HP, Patel H, Sugandha (2017) Enzymatic added extraction and clarification of fruit juices—a review. Crit Rev Food Sci Nutr 57:1215–1227
Huang D, Liu J, Qi Y, Yang K, Xu Y, Feng L (2017) Synergistic hydrolysis of xylan using novel xylanases, β-xylosidases, and an α-l-Arabinofuranosidase from Geobacillus thermodenitrificans NG80-2. Appl Microbiol Biotechnol 101:6023–6037
Cakmak U, Ertunga NS (2016) Gene cloning, expression, immobilization and characterization of endo-xylanase from Geobacillus sp. TF16 and investigation of its industrial applications. J Mol Catal B Enzym 133:S288–S298
Nagar S, Mittal A, Gupta VK (2012) Enzymatic clarification of fruit juices (apple, pineapple, and tomato) using purified Bacillus pumilus SV-85S xylanase. Biotechnol Bioprocess Eng 17:1165–1175
Bajaj BK, Manhas K (2012) Production and characterization of xylanase from Bacillus licheniformis P11 (C) with potential for fruit juice and bakery industry. Biocatal Agric Biotechnol 4:330–337
Acknowledgements
The authors would like to thank Biotechnology and Bioengineering Research Center at İzmir Institute of Technology for the facilities and technical support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
İlgü, H., Sürmeli, Y. & Şanlı-Mohamed, G. A thermophilic α-l-Arabinofuranosidase from Geobacillus vulcani GS90: heterologous expression, biochemical characterization, and its synergistic action in fruit juice enrichment. Eur Food Res Technol 244, 1627–1636 (2018). https://doi.org/10.1007/s00217-018-3075-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-018-3075-7