Abstract
Parkinson’s disease is a health-threatening neurodegenerative disease of the elderly with clinical manifestations of motor and non-motor deficits such as tremor palsy and loss of smell. Alpha-synuclein (α-Syn) is the pathological basis of PD, it can abnormally aggregate into insoluble forms such as oligomers, fibrils, and plaques, causing degeneration of nigrostriatal dopaminergic neurons in the substantia nigra in the patient’s brain and the formation of Lewy bodies (LBs) and Lewy neuritis (LN) inclusions. As a result, achieving α-Syn aggregate detection in the early stages of PD can effectively stop or delay the progression of the disease. In this paper, we provide a brief overview and analysis of the molecular structures and α-Syn in vivo and in vitro detection methods, such as mass spectrometry, antigen–antibody recognition, electrochemical sensors, and imaging techniques, intending to provide more technological support for detecting α-Syn early in the disease and intervening in the progression of Parkinson’s disease.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Parkinson’s disease (PD) has emerged as the second major neurodegenerative disease that threatens people’s health in recent years. Its clinical manifestations are mainly motor symptoms such as resting tremor, bradykinesia, tonicity, and non-motor symptoms including olfactory loss, sleep disturbance, and gastrointestinal complications, which have a significant impact on the patient’s daily life [1, 2]. Since the pathogenesis has not been completely grasped and no efficient treatments for PD, all current preventive and therapeutic measures are merely helpful to delay the PD progression. Therefore, it is essential to seek strategies for PD early diagnosis and treatment.
In 1985, the discovery of amyloid-beta (Aβ) motivated researchers to investigate the molecular pathogenesis of AD proteins, which were discovered as the key proteins causing neurodegenerative diseases, along with microtubule-associated protein tau (Tau) and α-Syn. α-Syn is the main component of intracellular fibrillar Lewy bodies (LBs) and Lewy neuritis (LN). It can trigger nigral dopaminergic neuron degeneration and motor symptoms (Fig. 1a). Misfolded α-Syn can form protein oligomers, fibrils, and neuronal deposits with rich β-sheet structures and accumulate in patients’ cerebral cortex and hippocampus, causing memory and motor deficits. However, identifying the above pathological process in the early stages of PD is difficult [3, 4]. Therefore, the early detection of misfolded α-Syn is essential to control PD. Compared with changes in dopaminergic or vesicular monoamine transporter protein 2 (VMAT2), misfolded α-Syn has been illustrated that is the preferential evidence of PD before motor symptoms emergence [5, 6]. Currently, there have been numerous studies on α-Syn conformational changes and the pathogenesis and early diagnostic techniques of PD. In this review, we will summarize and prospect the relevant in vivo and in vitro studies to provide potential solutions for the early diagnosis and treatment of PD via real-time tracking of α-Syn (Scheme 1).
Physiological functions, structures, and characterization of α-Syn
α-Syn is a 140 amino acid (AA) presynaptic terminal protein encoded by SCNA genes. It is a soluble monomer in the physiological state and participates in cell membrane interactions, synaptic function, vesicle transport, and dopamine synthesis, storage, and release in the central nervous system (CNS) [2, 7]. α-Syn consists of three structurally distinct physiological functional regions (Fig. 1): the N-terminal region (AA1-60) has mutation sites and contains four incomplete repetitive sequences that can participate in membrane binding interactions and promote the formation of β-sheet structures; the central NAC hydrophobic region (AA61-95) is highly involved in the formation of β-sheet structures and the production of insoluble LBs and LNs; the C-terminal region (AA96-140) is full of proline, which is highly acidic and can engage in post-translational modifications (PTMs) of proteins to regulate their function.
α-Syn is a pathogenic biomarker of neurodegenerative illnesses such as PD, multiple system atrophy (MSA), and dementia with Lewy bodies (DLBs) and accounts for barely 1% of total brain protein content [6, 8,9,10]. The mechanism of α-Syn aggregation in the brain has not been clarified, and when intracellular homeostasis is disrupted, its non-structural monomer and sub-stable α-helical tetrameric forms will oligomerize, and form toxic oligomers, fibrils, and deposits with different intermediate conformations through PTMs, which are difficult to distinguish from other amyloid proteins [3, 8, 11, 12]. Studies have revealed that the preformed α-Syn conformational changes followed an S-shaped aggregation growth mechanism during the early stage of fibrillization [13], and self-conjugation interactions of α-Syn oligomers in the late stage of aggregation would accelerate the formation of fibrils and large-sized aggregates [9, 14]. Thioflavin-T (ThT) [11] exhibits different binding sites and binding patterns when generating fibrils with two polycrystalline α-Syn mutants (A30P, A53T), which indicates that there are non-single binding sites on the surface of α-Syn fibrils and lead to their conformational changes [8] (Fig. 2). This shows that it is difficult to identify α-Syn from other amyloid proteins because of its low content and a high degree of conformational plasticity in the brain. Therefore, the early dynamic detection of α-Syn conformation and content in vivo is necessary for early diagnosis and treatment of PD.
Confocal microscopy images of α-Syn amyloid fibrils. Reprinted with permission from ref. [8]
Analytical methods commonly used to characterize α-Syn are mainly spectroscopy, microscopy, mass spectrometry (MS), electrophoresis, etc. Circular dichroism (CD) [15], fluorescence correlation spectroscopy (FCS) [16], Raman spectroscopy [17], atomic force microscopy (AFM) [18,19,20], and single-molecule total internal reflection fluorescence microscopy (TIRFM) [21] were used to monitor aggregation mechanism of recombinant α-Syn in vitro, allowing greater determination and sensitivity in samples such as cerebrospinal fluid (CSF) to obtain the α-Syn conformational changes and its secondary or tertiary structures. MS has high sensitivity and specificity for separating low molecular weight protein samples based on the mass-to-charge ratio upon vaporization and ionization. However, the method is less repeatable for protein separation with the same mass-to-charge ratio, poor hydrophobicity, and interrupted intermolecular interactions [22].
By using several MS methods, Rebecca’s group [23] has implemented a conformational analysis of α-Syn and apolipoprotein C-II (ApoC-II). Hydrogen–deuterium exchange mass spectrometry (HDX-MS), electrospray ionization mass spectrometry (ESI–MS), and ion mobility mass spectrometry (IM-MS) are sensitive to intrinsically disordered protein (IDPs) and can distinguish the disordered and structured proteins, confirm the disordered nature of α-Syn and ApoC-II, and reflect their conformational dynamics information. Payel et al. [24] detected different synaptic nuclear protein variants and quantified the α-Syn truncated at amino acid 119 and the acetylated N-terminal α-Syn from cingulate cortex and occipital cortex of control and PD patients by using liquid chromatography and mass spectrometry (LC–MS/MS) with differential solubilization, immunoprecipitation, and targeted proteomics approaches to analyze α-Syn. The results showed that the other α-Syn variants levels except for α-Syn103−119 (Ac-α-Syn1−6, α-Syn13−21, α-Syn35−43, α-Syn46−58, α-Syn61−80, and α-Syn81−96) in PD patient’s cingulate region and Ac-α-Syn1−6 in the soluble fraction were significantly higher than the controls, and whether acetylated α-Syn can be used as a potential biomarker for PD needs to be further investigated. Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) combined with Western blotting and size exclusion chromatography (SEC) can detect protein aggregates based on antigen–antibody reaction and molecular sieve effect [25]. Furthermore, applying an electrical potential to the capillary, the capillary electrophoresis (CE) technique can efficiently separate the amyloid proteins such as Aβ and overcome the effects of SDS and gel smears. CE needs to be further improved to increase the resolution of amyloid protein aggregates and has the potential for early oligomers detection like Aβ and α-Syn [26]. Online nucleic acid aptamer affinity solid-phase extraction capillary electrophoresis–mass spectrometry (AA-SPE-CE-MS) [27] can improve the shortcomings of traditional CE-MS, which has a small injection volume, easily affected by sample concentration, and realize the purification, separation, and identification of α-Syn in blood by specific binding of single-stranded DNA aptamer with low detection limit and sensitivity to acidic and alkaline environment, which will be used in the clinical monitoring of α-Syn in PD patient’s brains. Microfluidic capillary electrophoresis (MCE) [25] and affinity capillary electrophoresis (ACE) [26] can improve the migration rate of proteins by using binding agents (antibodies, aptamers, serum proteins, etc.), further improving protein separation with the advantages of lower sample consumption and higher automation potential.
In vitro approaches for the detection of α-Syn
Immunohistological results of autopsy brain sections can be used to diagnose and reflect the severity of PD [9, 28]; the method is often difficult to accept by patients’ families and is commonly characterized by sampling errors. Therefore, a more cost-effective sampling method to detect α-Syn is required. Braak et al. proposed a hypothesis [7, 29,30,31,32] that microorganisms of the gastrointestinal can conduct immunological and endocrine signaling through the gut-brain axis; it was suggested that α-Syn could spread from the enteric nervous system (ENS) to CNS when exposed to environmental and genetic damage. Studies showed that α-Syn has been found in the tissues of patients’ skin [33,34,35,36,37,38,39,40], gastrointestinal tract [41, 42], retina [43, 44], and body fluids such as blood [45, 46], CSF [47,48,49], saliva [50,51,52], and urine [53] (Fig. 3). In recent years, some analytical methods for detecting α-Syn and its conformation in the actual human biological samples have been developed, which will facilitate the early diagnosis and prevention of PD.
Detection of actual human biological samples
Immunofluorescence (IF) staining based on antigen–antibody recognition, enzyme-linked immunosorbent assay (ELISA), and electrochemical sensors are commonly used to detectα-Syn in human biological samples such as skin tissues and body fluids. Giorgia et al. [35] performed immunofluorescence analysis on skin tissue from PD patients’ ankle, thigh, and neck; the results revealed that α-Syn formed positive deposits with 5G4 antibodies in each tissue’s dermal nerves and the degree of neck skin degeneration increased with the duration of the disease. Sharon’s team [54] detected total α-Syn of 16.7 ± 5.9 ng/ml and 55.9 ± 10.3 ng/ml in CSF and saliva samples by using phospholipid ELISA, whereas the traditional sandwich ELISA recorded 1.2 ± 0.4 ng/ml in CSF samples and could not achieve α-Syn detection in saliva. This approach was more sensitive than sandwich ELISA. For most peptides and proteins, IF and ELISA have the advantage of rapid analysis speed, low radiation, and high specificity, but lack international standard dyeing, and are vulnerable to personnel operation skill, body gland secretion, type and quantity of protein, protease inhibitors, and category of kits, restricting their accurate detection of α-Syn in vitro. Currently, there are electrochemical sensors mainly to detect Aβ, and fewer available to detect α-Syn, such as polyethylene glycol gold surface electrode sensors [55] based on antigen–antibody recognition, graphene oxide sensors [56], nucleic acid aptamer sensors [57, 58], indium tin oxide electrode sensors [59], etc. [60,61,62] (Fig. 4), have lower detection limits and higher specificity and sensitivity for α-Syn in vitro and are simple, portable, and mass-produced. However, their further clinical applications are restricted by the variety of expensive electrode materials, complex structural modifications, and serious biological contamination.
Electrochemical biosensors for α-Syn detection. a Schematic illustration of graphene oxide–modified biosensor to detect α-Syn autoantibodies. Reprinted with permission from ref. [56]. b Schematic illustration of the electrochemical aptamer sensor to detect α-Syn oligomer. Reprinted with permission from ref. [57]. c Schematic representation of the indium tin oxide electrode sensors to detect α‐Syn. Reprinted with permission from ref [59]
Detection of recombinant α-Syn in vitro
α-Syn aggregates
Currently, there are many emerging techniques applied to detect α-Syn such as fluorescence imaging techniques and real-time quaking-induced conversion (RT-QuIC). Fluorescence imaging techniques [63,64,65] can better target the protein and generate strong fluorescence responses, as a potential technique for finding amyloid in vivo and in vitro without using any radionuclides, with lower injury, higher sensitivities, lower costs, detection limits, and faster elimination in vivo. ThT (Table 1) is a benzothiazole molecule based on the intramolecular charge transfer (TICT) mechanism to form a rigid molecular structure upon binding the β-sheet structure of amyloid proteins and generates fluorescence based on the energy released by the n-π* transition. Although the ThT method is the “gold standard” for detecting amyloid fibrils in vitro and a tool for recognizing their aggregation and restraint mechanisms [66, 67]. ThT is difficult to realize α-Syn detection because it lacks specificity and low sensitivity, has susceptibility to interference by sample background fluorescence signal and inability to penetrate the blood–brain barrier (BBB), and easily quenches after aggregation. Therefore, it is necessary to further develop other types of fluorescent probes to detect α-Syn aggregation.
Thomas M’s group presented four N-arylaminonaphthalene sulfonates (NAS) derivatives, 2,6-ANS, 2,6-TNS, bis-ANS, and bis-TNS, as universal probes for the detection of amyloid protein; bis-ANS and bis-TNS are the most sensitive probes to amyloid proteins. Compared with ThT (Kd = 14.9 M), the structure of NAS was more susceptible to α-Syn, with Kd values of 8.8, 11.7, 8.6, and 11.6 M, their charged properties and emission wavelength preventing them from being used as in vivo imaging probes for α-Syn [68]. Two fluorescence probes based on the benzothiazole structure, C15H15N3(C1) and C16H16N3O2(C2), can detect α-Syn at submicromolar concentrations. C1 and C2 do not interfere with α-syn aggregation and have lower detection limits and higher sensitivity than THT. They tend to bind α-Syn aggregates in SH-SY5Y cells with little cytotoxicity, so they are potential probes for detecting α-Syn [69]. Gorka et al. [70] found that the trochanteric thiazole molecules RB1 and RB2 have an extended π system, the molecular size of RB1 is between RB2 and ThT, and it has a higher affinity with α-Syn aggregate. Compared to ThT, the maximum absorption of RB1 combined with α-Syn aggregates was red-shifted at 76 nm, and the fluorescence intensity was increased by 112 times. So water-soluble RB1 could as a potential probe detect α-Syn aggregates in living cells, which can overcome strong background signals in living cells and stain the cytoplasm of HeLa and SH-SYY cells.
Needham’s group [71] created a new generation of probe ThX with methoxy and dimethylamino groups and PAP_1 based on the ThT. ThX contains a higher density of electron clouds surrounding the C (sp3)-N bond, restricted rotation, and improved lipophilicity. ThX is suitable to detect α-Syn aggregates in high-background biological samples such as plasma and cerebrospinal fluid, because it can recognize α-Syn oligomers that ThT is unable, and the binding affinity and brightness of wild reconstituted α-Syn aggregates are increased by 7 times and 5 times. Although the PAP_1 probe was 100 times less brighter than ThT, it had a greater binding affinity for α-Syn aggregates and could image single recombinant α-Syn aggregates in brain tissue [72, 73]. Real-time quaking-induced conversion (RT-QuIC) [62, 74,75,76] can combine with ThT to rapidly detect α-Syn with low concentrations in vitro. However, due to the influence of sample size, concentration, temperature of α-Syn, and other conditions, the sensitivity and specificity of this method in the clinical application need to be further verified.
Monomers, oligomers, and fibrils of α-Syn
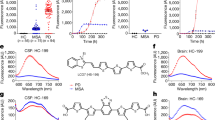
Compared with other conformations, α-Syn oligomers have a stronger neurological damaging effect on the brain and can cause neuronal injury and death. Therefore, it is essential to detect α-syn oligomers and inhibit production. Curcumin [77] may hasten the transition of α-Syn from oligomers to fibrils and decrease neurotoxicity by reducing the number of oligomers. Epigallocatechin gallate (EGCG) [78] can preferentially bind α-Syn fibrils and form them into novel amorphous and smaller aggregates. As a result, probes or inhibitors with curcumin, EGCG, and Anle138b [79] structures can intervene early in the α-Syn aggregation process. BE01, BE02, mBE01, and mBE02 [80] are four highly sensitive bifunctional fluorescence probes with higher fluorescence intensity than ThT (Fig. 5a) and simultaneously detect α-Syn and H2O2. The non-methylated structure of BE01 and BE02 binds α-Syn with lower bonding constants, allowing the detection of a single α-Syn fibril as shown in Fig. 5b. In addition, phenothiazine derivative 11b showed a high affinity for α-Syn fibrils and will be expected as a radioactive ligand to target α-Syn in vivo after further characterization [81].
a Synthetic scheme for the dyes of BE01, BE02, mBE01, and mBE02. b Schematic illustration of the fluorescence intensities of the BE, mBE, and ThT probes in PBS buffer, with H2O2, with α-Syn aggregates and with concomitant H2O2 and α-Syn aggregates. Reprinted with permission and adapted from ref. [80]
A few fluorescence probes with different phenyl, alkyl, and halogen substitutions and a large aromatic conjugated system can bind α-Syn monomers and oligomers, be used as its in vitro potential imaging probes, and accumulate experience for further development of in vivo imaging probe technology. By selecting target α-Syn oligomers and fibrils, respectively, the aromatic dyes pentamethyl anthocyanine SL-631 and trimethyl anthocyanine SH-299 [82] further enhance the detection limit of α-Syn fibrils. DCVJ probe [83] binds α-Syn oligomers with 2–4 times higher fluorescence intensity than that of bind monomers or fibrils. JC-1 carbocyanine [84] can distinguish α-Syn monomers and fibrils by emitting lights of different colors and has a lower detection limit for α-Syn monomers.
Additionally, aggregation-induced emission (AIE) probes and dyes [85, 86] were developed and applied for α-Syn detection. The methylpyridine and dimethylamine in the ASCP structure [87] improve the ability of electron-π conjugate expansion, share the ThT binding site, and have a larger Stokes shift (145 nm), which can replace ThT to research α-Syn fibrils growth in vitro. TPE-TPP [88] is a sensitive probe for identifying α-Syn monomers, oligomers, and fibrils with higher affinity, fluorescence intensity, and lower signal-to-noise ratio. It can compete with ThT on the binding site of α-Syn fibrils; however, its selectivity and affinity for other proteins are unidentified. Currently, the emerging fluorescence probes and RT-QuIC methods only achieved the detection of in vitro recombinant α-Syn; it is necessary to improve these methods to realize the detection of α-Syn in actual human samples and in vivo.
Compared with in vitro recombinant α-Syn fibrils, various α-Syn conformations in the human brain and actual human biological samples are significantly different in composition and cellular environment; thus, the recombinant α-Syn cannot completely simulate the in vivo aggregation of α-Syn. Therefore, it is necessary to develop in vivo detection techniques for α-Syn.
In vivo methods for the detection of α-Syn
At present, compared with in vitro detection techniques, radiographic positron emission tomography (PET), single photon emission computed tomography (SPECT), and non-radiographic magnetic resonance imaging (MRI) can achieve in situ detection of α-Syn in vivo. These methods accurately reflect abnormal metabolic changes of the brain and anatomical information to achieve early diagnosis and identification of brain diseases such as PD.
Radiative PET ligands for imaging α-Syn
PET and SPECT are less invasive and have higher specificity, sensitivity, and spatial resolution, allowing labeling biological samples such as enzymes or proteins by radionuclides (20 and 110 min) with short half-lives and high safety including 11C, 18F, 123I, or 99Tc and tracking imaging at the nanoscale [89, 90].
At present, only the US Food and Drug Administration (FDA)–approved PET probes [18F]Florbetapir [91], [18F]Florbetabeben [92, 93], and [18F]Flutemetamol [94,95,96] can be used to detect Aβ, while fewer PET ligands can target α-Syn, and have not been imaged in vivo. Although the ligands [11C]PiB [97], [18F]BF227 [98, 99], and [11C]-PBB3 [100] are selective for α-Syn recombinant fibrils in vitro, more easily bound to Aβ and Tau fibrils, which are highly abundant in human brain homogenates and have similar β-sheet structure rather than α-Syn fibrils [101]. Therefore, it is crucial to develop high-sensitivity PET ligands that target α-Syn to detect α-Syn fibrils in vivo at low concentrations. Studies demonstrated that the phenothiazine derivatives as [125I] SIL23[102], [11C]-SIL5, [11C]2a, and [18F]2b [103], indole diene derivatives [18F]WC-58a and [18F]46a[104, 105], and thiazole derivatives Anle253b [106] and [18F]DABTA-11 [12, 107, 108] were highly selective for in vitro reconstituted α-Syn fibrils. Among them, ligands [11C]-SIL5, [11C]2a and [18F]2b, Anle253b, and [18F] DABTA-11 can penetrate BBB of healthy cynomolgus monkeys or rats and achieve rapid brain elution, and these structures are anticipated to be used for in vivo α-Syn imaging after further optimization [12, 102]. However, ligands [18F]WC-58a, [18F]46a, and [18F]DABTA-11 are unable to participate in the imaging of α-Syn fibrils in human brain homogenates due to their lipophilicity. Currently, the drawbacks of existing ligands structures may be improved by enhancing the affinity with α-Syn fibrils and reducing their lipophilicity, in order to provide a possibility for PET ligands (Table 2) to target human α-Syn imaging and as a potential diagnostic tool for neurodegenerative diseases like PD.
Non-radiative MRI to image α-Syn
MRI can provide information about brain anatomy, function, and metabolism [116, 117] based on the longitudinal (T1) and transverse (T2) relaxation signals generated by magnetized water protons in brain tissues. For the detection of PD, MRI-based conventional methods are mainly used to identify the secondary PD caused by structural lesions in the brain. In addition, the development of diffusion-weighted imaging (DWI), susceptibility-weighted imaging (SWI), and other MRI imaging technologies has further improved the reliability of PD diagnosis in clinical practice [90, 118].
Khairnar et al. [119] used diffusion kurtosis imaging (DKI) with mean kurtosis (MK) as the assessment index to scan α-syn overexpressing TNWT-61 mice and the method overcame the environmental limitation of water quality subanisotropy to achieve the detection of mouse brain microstructural changes, which could be an effective clinical diagnosis tool for early PD. Studies found that 3-month-old mice showed increased MK values in striatal and thalamic regions with insignificant changes in substantia nigra, hippocampal regions, and sensorimotor cortex, whereas 6-month-old TNWT-61 mice showed increased MK values in substantia nigra regions and behavioral assessments showed some age dependence. This suggests that the α-syn accumulation may be manifested in higher-density dopaminergic nerve terminals and that the young mouse model lacks dopaminergic neuronal degeneration; LB and brain atrophy is only applicable to imaging of early prodromal symptoms in PD patients. In view of the role of the glymphatic system and the meningeal lymphatic system in excreting toxic macromolecular protein metabolites and maintaining the metabolic balance of the central nervous system, Xue et al. [120] used dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI) to evaluate the basal meningeal lymphatic flow in patients with idiopathic PD (IPD) or atypical PD (APD); the results showed that meningeal lymphatic vessels have an ability to clear α-Syn. The impaired function can delay meningeal lymphatic drainage and aggravate the pathological reaction of α-Syn, indicating that this method is helpful for the early differentiation between IPD and APD, and meningeal lymphatic vessels are expected to be key targets to delay the progression of PD. MRI is an important method for the diagnosis of early PD brain structural imaging, which can demonstrate the functional and structural changes of α-Syn pathological areas in the brain through image differences. However, MRI is an expensive instrument with drawbacks of standardized tissue quantification methods, diagnostic indicators, and prognostic treatment strategies; it needs to overcome the extremely low chemical shift dispersion of α-Syn and improve high resolution in 3D structure detection and is unable to achieve α-Syn in situ imaging.
Conclusions and perspectives
In this paper, we reviewed the associated methods to detect α-Syn in vivo and in vitro. Currently, traditional methods based on antigen–antibody reactions to detect human actual samples such as autopsy, tissues, and biological fluids are specific for α-Syn, but with highly invasive, lower sensitivity, and are difficult to detect different forms of α-Syn. Although in vitro analytical detection and in vivo MRI can obtain information about secondary and tertiary structures of α-Syn and structural pathologies in the brain with high resolution, and realize the size quantification and image, it is difficult to realize in situ α-Syn detection and widespread use for clinical patients because of lack standardized quantitative methods and indicators, low resolution, and environmental influence. The abovementioned in vivo and in vitro methods for α-Syn detection have made considerable progress in illustrating the pathogenesis of PD and the dynamics of α-Syn aggregation, but they still lack effective methods for α-Syn in vivo detection and intervention.
The improvement of PET ligands and fluorescence imaging probes are potential methods and research hotspots to realize α-Syn in situ detection, early diagnosis, and therapy of PD. In situ α-Syn with lower abundance, complex intracerebral environment, and PTMs lead to different forms and binding sites that are difficult to be detected compared with Aβ, Tau, and in vitro recombinant α-Syn models. At present, fluorescence probes that target α-Syn have slow development compared to PET ligands. Only a few fluorescence probes can target in vitro and intracellular recombination α-Syn but have not yet achieved α-Syn in vivo imaging due to poor selectivity, elution ability, and high lipophilicity. A few α-Syn PET ligands are still in the primary stage compared to Aβ and Tau ligands. Although PET ligands can target different forms of α-Syn in vitro and penetrate the BBB in rodent animal models, its limited emission wavelengths are unsuitable for human studies, and no clinically approved PET ligands for in vivo α-Syn. Therefore, fluorescence probes and PET ligands for α-Syn in vivo imaging should have the following characteristics: (1) small molecular weight, low lipophilicity and cytotoxicity, long fluorescence lifetime, and good elution capacity after penetrating the BBB by passive diffusion; (2) highly sensitive to α-Syn, high selectivity and affinity, and low detection limit; (3) suitable emission wavelength to achieve α-Syn in situ imaging. Therefore, in further development and application of α-Syn in situ imaging technology, it is necessary to evaluate the fidelity of the recombinant α-Syn models in vitro and simulate its aggregation environment in the brain, considering the influence of the difference distribution and concentration of the target proteins and non-target proteins. Using structural modification strategies like substitution and enhanced conjugation improves the abovementioned features of new fluorescence probes and PET ligands. Meanwhile, microscopy, MS, and computer molecular docking technology could be used to clarify the process of aggregation dynamics, detect α-Syn conformation and size, and predict the binding sites. The development of potential probes and ligands for targeting various α-Syn conformations in vitro is anticipated to further apply for α-Syn in situ imaging.
References
Benadiba M, Luurtsema G, Wichert-Ana L, Buchpigel CA, Busatto FG. New molecular targets for PET and SPECT imaging in neurodegenerative diseases. Braz J Psychiatry. 2012;34(2):125–48. https://doi.org/10.1016/j.rbp.2012.07.002.
O’ Hara DM, Kalia SK, Kalia LV. Methods for detecting toxic α-Synuclein species as a biomarker for Parkinson’s disease. Crit Rev Clin Lab Sci. 2020;57(5):291–307. https://doi.org/10.1080/10408363.2019.1711359.
Kundel F, Tosatto L, Whiten DR, Wirthensohn DC, Horrocks MH, Klenerman D. Shedding light on aberrant interactions-a review of modern tools for studying protein aggregates. FEBS J. 2018;285(19):3604–30. https://doi.org/10.1111/febs.14409.
Cao KJ, Yang J. Translational opportunities for amyloid-targeting fluorophores. Chem Commun (Camb). 2018;54(66):9107–18. https://doi.org/10.1039/C8CC03619E.
Han D, Zheng W, Wang X, Chen Z. Proteostasis of α-Synuclein and its role in the pathogenesis of Parkinson’s disease. Front Cell Neurosci. 2020;14(45). https://doi.org/10.3389/fncel.2020.00045.
Fricova D, Harsanyiova J, Kralova TA. Alpha-synuclein in the gastrointestinal tract as a potential biomarker for early detection of Parkinson’s disease. Int J Mol Sci. 2020;21(22):8666. https://doi.org/10.3390/ijms21228666.
Rietdijk CD, Perez-Pardo P, Garssen J, van Wezel RJ, Kraneveld AD. Exploring Braak’s hypothesis of Parkinson’s disease. Front Neurol. 2017; 8(37). https://doi.org/10.3389/fneur.2017.00037.
Sulatskaya AI, Rodina NP, Sulatsky MI, Povarova OI, Antifeeva IA, Kuznetsova IM, Turoverov KK. Investigation of α-Synuclein amyloid fibrils using the fluorescent probe thioflavin-T. Int J Mol Sci. 2018;19(9):2486. https://doi.org/10.3390/ijms19092486.
Ghosh S, Sakshi, Swain BC, Chakraborty R, Tripathy U, Chattopadhyay K. A novel tool to investigate the early and late stages of α-synuclein aggregation. ACS Chem Neurosci. 2020;11(11):1610–1619. https://doi.org/10.1021/acschemneuro.0c00068.
Granado N, Gómez-Benito M, García-Sanz P, Michel A, Dumoulin M, Moratalla R. Modeling Parkinson’s disease with the alpha-synuclein protein. Front Pharmacol. 2020;11:356. https://doi.org/10.3389/fphar.2020.00356.
Sidhu A, Vaneyck J, Blum C, Segers-Nolten I, Subramaniam V. Polymorph-specific distribution of binding sites determines thioflavin-T fluorescence intensity in α-synuclein fibrils. Amyloid. 2018;25(3):189–96. https://doi.org/10.1080/13506129.2018.1517736.
Uzuegbunam BC, Librizzi D, Hooshyar YB. PET radiopharmaceuticals for Alzheimer’s disease and Parkinson’s disease diagnosis, the current and future landscape. Molecules. 2020;25(4):977. https://doi.org/10.3390/molecules25040977.
Avidan-Shpalter C, Gazit E. The early stages of amyloid formation: biophysical and structural characterization of human calcitonin pre-fibrillar assemblies. Amyloid. 2006;13(4):216–25. https://doi.org/10.1080/13506120600960643.
Tavassoly O, Nokhrin S, Dmitriev OY, Lee JS. Cu (II) and dopamine bind to α-synuclein and cause large conformational changes. FEBS J. 2014;281(12):2738–53. https://doi.org/10.1111/febs.12817.
Rekas A, Lo V, Gadd GE, Cappai R, Yun SI. PAMAM dendrimers as potential agents against fibrillation of alpha-synuclein, a Parkinson’s disease-related protein. Macromol Biosci. 2009;9(3):230–8. https://doi.org/10.1002/mabi.200800242.
Li X, Dong C, Hoffmann M, Garen CR, Cortez LM, Petersen NO. Woodside MT. Early stages of aggregation of engineered α-synuclein monomers and oligomers in solution. Sci Rep. 2019;11;9(1):1734. https://doi.org/10.1038/s41598-018-37584-6.
Flynn JD, Jiang Z, Lee JC. Segmental 13C-labeling and raman microspectroscopy of α-synuclein amyloid formation. Angew Chem Int Ed Engl. 2018;57(52):17069–72. https://doi.org/10.1002/anie.201809865.
Canale C, Torre B, Ricci D, Braga PC. Recognizing and avoiding artifacts in atomic force microscopy imaging. Methods Mol Biol. 2011;736:31–43. https://doi.org/10.1007/978-1-61779-105-5_3.
McAllister C, Karymov MA, Kawano Y, Lushnikov AY, Mikheikin A, Uversky VN, Lyubchenko YL. Protein interactions and misfolding analyzed by AFM force spectroscopy. J Mol Biol. 2005;354(5):1028–42. https://doi.org/10.1016/j.jmb.2005.10.012.
Campioni S, Carret G, Jordens S, Nicoud L, Mezzenga R, Riek R. The presence of an air-water interface affects formation and elongation of α-Synuclein fibrils. J Am Chem Soc. 2014;136(7):2866–75. https://doi.org/10.1021/ja412105t.
Taylor CG, Meisl G, Horrocks MH, Zetterberg H, Knowles TPJ, Klenerman D. Extrinsic amyloid-binding dyes for detection of individual protein aggregates in solution. Anal Chem. 2018;90(17):10385–93. https://doi.org/10.1021/acs.analchem.8b02226.
Beveridge R, Phillips AS, Denbigh L, Saleem HM, MacPhee CE, Barran PE. Relating gas phase to solution conformations: lessons from disordered proteins. Proteomics. 2015;15(16):2872–83. https://doi.org/10.1002/pmic.201400605.
Bhattacharjee P, Öhrfelt A, Lashley T, Blennow K, Brinkmalm A, Zetterberg H. Mass spectrometric analysis of Lewy body-enriched α-Synuclein in Parkinson’s disease. J Proteome Res. 2019;18(5):2109–20. https://doi.org/10.1021/acs.jproteome.8b00982.
Prakash A, Rezai T, Krastins B, Sarracino D, Athanas M, Russo P, Zhang H, Tian Y, Li Y, Kulasingam V, Drabovich A, Smith CR, Batruch I, Oran PE, Fredolini C, Luchini A, Liotta L, Petricoin E, Diamandis EP, Chan DW, Nelson R, Lopez MF. Interlaboratory reproducibility of selective reaction monitoring assays using multiple upfront analyte enrichment strategies. J Proteome Res. 2012;11(8):3986–95. https://doi.org/10.1021/pr300014s.
Pryor NE, Moss MA, Hestekin CN. Unraveling the early events of amyloid-β protein (Aβ) aggregation: techniques for the determination of Aβ aggregate size. Int J Mol Sci. 2012;13(3):3038–72. https://doi.org/10.3390/ijms13033038.
Zhang C, Woolfork AG, Suh K, Ovbude S, Bi C, Elzoeiry M, Hage DS. Clinical and pharmaceutical applications of affinity ligands in capillary electrophoresis: a review. J Pharm Biomed Anal. 2020;177:112882. https://doi.org/10.1016/j.jpba.2019.112882.
Pero-Gascon R, Benavente F, Minic Z, Berezovski MV, Sanz-Nebot V. On-line aptamer affinity solid-phase extraction capillary electrophoresis-mass spectrometry for the analysis of blood α-Synuclein. Anal Chem. 2020;92(1):1525–33. https://doi.org/10.1021/acs.analchem.9b04802.
Tiwari S, Atluri V, Kaushik A, Yndart A, Nair M. Alzheimer’s disease: pathogenesis, diagnostics, and therapeutics. Int J Nanomedicine. 2019;14:5541–54. https://doi.org/10.2147/IJN.S200490.
Karanth S, Nelson PT, Katsumata Y, Kryscio RJ, Schmitt FA, Fardo DW, Cykowski MD, Jicha GA, Van Eldik LJ, Abner EL. Prevalence and clinical phenotype of quadruple misfolded proteins in older adults. JAMA Neurol. 2020;77(10):1299–307. https://doi.org/10.1001/jamaneurol.2020.1741.
PP Liu, Xie Y, Meng XY, Kang JS. History and progress of hypotheses and clinical trials for Alzheimer’s disease. Signal Transduct Target Ther. 2019;4:29. https://doi.org/10.1038/s41392-019-0063-8.
Ruffmann C, Parkkinen L. Gut feelings about α-synuclein in gastrointestinal biopsies: biomarker in the making? Mov Disord. 2016;31(2):193–202. https://doi.org/10.1002/mds.26480.
Ruffmann C, Bengoa-Vergniory N, Poggiolini I, Ritchie D, Hu MT. Alegre-Abarrategui J, Parkkinen L. Detection of alpha-synuclein conformational variants from gastro-intestinal biopsy tissue as a potential biomarker for Parkinson’s disease. Neuropathol Appl Neurobiol. 2018;44(7):722-736. https://doi.org/10.1111/nan.12486.
Hawkes CH, Del Tredici K, Braak H. Parkinson’s disease: the dual hit theory revisited. Ann N Y Acad Sci. 2009;1170:615–22. https://doi.org/10.1111/j.1749-6632.2009.04365.x.
Beach TG, Adler CH, Sue LI, Vedders L, Lue L, White Iii CL, Akiyama H, Caviness JN, Shill HA, Sabbagh MN, Walker DG. Arizona Parkinson’s Disease Consortium. Multi-organ distribution of phosphorylated alpha-synuclein histopathology in subjects with Lewy body disorders. Acta Neuropathol. 2010;119(6):689–702. https://doi.org/10.1007/s00401-010-0664-3.
Melli G, Vacchi E, Biemmi V, Galati S, Staedler C, Ambrosini R, Kaelin-Lang A. Cervical skin denervation associates with alpha-synuclein aggregates in Parkinson disease. Ann Clin Transl Neurol. 2018;5(11):1394–407. https://doi.org/10.1002/acn3.669.
Zange L, Noack C, Hahn K, Stenzel W, Lipp A. Phosphorylated α-synuclein in skin nerve fibres differentiates Parkinson’s disease from multiple system atrophy. Brain. 2015;138(8):2310–21. https://doi.org/10.1093/brain/awv138.
Kuzkina A, Schulmeyer L, Monoranu CM, Volkmann J, Sommer C, Doppler K. The aggregation state of α-synuclein deposits in dermal nerve fibers of patients with Parkinson’s disease resembles that in the brain. Parkinsonism Relat Disord. 2019;64:66–72. https://doi.org/10.1016/j.parkreldis.2019.03.003.
Spinelli KJ, Taylor JK, Osterberg VR, Churchill MJ, Pollock E, Moore C, Meshul CK, Unni VK. Presynaptic alpha-synuclein aggregation in a mouse model of Parkinson’s disease. J Neurosci. 2014;34(6):2037–50. https://doi.org/10.1523/JNEUROSCI.2581-13.2014.
Doppler Doppler K, Ebert S, Uçeyler N, Trenkwalder C, Ebentheuer J, Volkmann J, Sommer C. Cutaneous neuropathy in Parkinson’s disease: a window into brain pathology. Acta Neuropathol. 2014;128(1):99–109. https://doi.org/10.1007/s00401-014-1284-0.
Liu X, Yang J, Yuan Y, He Q, Gao Y, Jiang C, Li L, Xu Y. Optimization of the detection method for phosphorylated α-synuclein in Parkinson disease by skin biopsy. Front Neurol. 2020; 11: 569446. https://doi.org/10.3389/fneur.2020.569446.
Antunes L, Frasquilho S, Ostaszewski M, Weber J, Longhino L, Antony P, Baumuratov A, Buttini M, et al. Similar α-synuclein staining in the colon mucosa in patients with Parkinson’s disease and controls. Movement Disord. 2016;31(10):1567–70. https://doi.org/10.1002/mds.26702.
Gold A, Turkalp ZT, Munoz DG. Enteric alpha-synuclein expression is increased in Parkinson’s disease but not Alzheimer’s disease. Movement Disord. 2013;28(2):237–41. https://doi.org/10.1002/mds.25298.
Veys L, Vandenabeele M, Ortuno-Lizaran I, Baekelandt V, Cuenca N, Moons L, De Groef L. Retinal alpha-synuclein deposits in Parkinson’s disease patients and animal models. Acta Neuropathol. 2019;137(3):379–95. https://doi.org/10.1007/s00401-018-01956-z.
Leger F, Fernagut PO, Canron MH, Leoni S, Vital C, Tison F, Bezard E, Vital A. Protein aggregation in the aging retina. J Neuropathol Exp Neurol. 2011;70(1):63–8. https://doi.org/10.1097/NEN.0b013e31820376cc.
Nakai M, Fujita M, Waragai M, Sugama S, Wei J, Akatsu H, Ohtaka-Maruyama C, Okado H, et al. Expression of α-synuclein, a presynaptic protein implicated in Parkinson’s disease, in erythropoietic lineage. Biochem Bioph Res Co. 2007;358(1):104–10. https://doi.org/10.1016/j.bbrc.2007.04.108.
Abd ES, Grigoletto J, Poli M, Arosio P, Arkadir D, Sharon R. alpha-Synuclein in blood cells differentiates Parkinson’s disease from healthy controls. Ann Clin Transl Neurol. 2019;6(12):2426–36. https://doi.org/10.1002/acn3.50944.
Katayama T, Sawada J, Takahashi K, Yahara O. Cerebrospinal fluid biomarkers in Parkinson’s disease: a critical overview of the literature and meta-analyses. Brain Sci. 2020;10(7):466. https://doi.org/10.3390/brainsci10070466.
Ning H, Wu Q, Han D, Yao T, Wang J, Lu W, Lv S, Jia Q, et al. Baseline concentration of misfolded α-synuclein aggregates in cerebrospinal fluid predicts risk of cognitive decline in Parkinson’s disease. Neuropath Appl Neuro. 2018;45(4):398–409. https://doi.org/10.1111/nan.12524.
Camporesi E, Nilsson J, Brinkmalm A, Becker B, Ashton NJ, Blennow K, Zetterberg H. Fluid biomarkers for synaptic dysfunction and loss. Biomarker Insights. 2020;15:2138511671. https://doi.org/10.1177/1177271920950319.
Cao Z, Wu Y, Liu G, Jiang Y, Wang X, Wang Z, Feng T. α-Synuclein in salivary extracellular vesicles as a potential biomarker of Parkinson’s disease. Neurosci Lett. 2018;696:114–20. https://doi.org/10.1016/j.neulet.2018.12.030.
Vivacqua G, Suppa A, Mancinelli R, Belvisi D, Fabbrini A, Costanzo M, Formica A, Onori P, et al. Salivary alpha-synuclein in the diagnosis of Parkinson’s disease and progressive supranuclear palsy. Parkinsonism Relat D. 2019;63:143–8. https://doi.org/10.1016/j.parkreldis.2019.02.014.
Ashton NJ, Ide M, Zetterberg H, Blennow K. Salivary biomarkers for Alzheimer’s disease and related disorders. Neurol Ther. 2019;8(Suppl 2):83–94. https://doi.org/10.1007/s40120-019-00168-1.
Nam D, Lee J, Lee M, Kim J, Seol W, Son I, Ho DH. Detection and assessment of α-synuclein oligomers in the urine of Parkinson’s disease patients. J Parkinsons Dis. 2020;10(3):981–91. https://doi.org/10.3233/JPD-201983.
Abd-Elhadi S, Basora M, Vilas D, Tolosa E, Sharon R. Total α-synuclein levels in human blood cells, CSF, and saliva determined by a lipid-ELISA. Anal Bioanal Chem. 2016;408(27):7669–77. https://doi.org/10.1007/s00216-016-9863-7.
Bryan T, Luo X, Forsgren L, Morozova-Roche LA, Davis JJ. The robust electrochemical detection of a Parkinson’s disease marker in whole blood sera. Chem Sci (Cambridge). 2012;3(12):3468–73. https://doi.org/10.1039/c2sc21221h.
Xu Q, Cheng H, Lehr J, Patil AV, Davis JJ. Graphene oxide interfaces in serum based autoantibody quantification. Anal Chem. 2014;87(1):346–50. https://doi.org/10.1021/ac503890e.
Taghdisi SM, Danesh NM, Nameghi MA, Ramezani M, Alibolandi M, Hassanzadeh-Khayat M, Emrani AS, Abnous K. A novel electrochemical aptasensor based on nontarget-induced high accumulation of methylene blue on the surface of electrode for sensing of α-synuclein oligomer. Biosens Bioelectron. 2018;123:14–8. https://doi.org/10.1016/j.bios.2018.09.081.
Wu Q, Tan R, Mi X, Tu Y. Electrochemiluminescent aptamer-sensor for alpha synuclein oligomer based on a metal-organic framework. Analyst. 2020;145(6):2159–67. https://doi.org/10.1039/D0AN00169D.
Ge CY, Rahman MM, Zhang W, Lopa NS, Jin L, Yoon S, Jang H, Xu GR et al. An electrochemical immunosensor based on a self-assembled monolayer modified electrode for label-free detection of alpha-synuclein. Sensors (Basel). 2020;20(3). https://doi.org/10.3390/s20030617.
Hassan Q, Li S, Ferrag C, Kerman K. Electrochemical biosensors for the detection and study of α-synuclein related to Parkinson’s disease – a review. Anal Chim Acta. 2019;1089:32–9. https://doi.org/10.1016/j.aca.2019.09.013.
Chan T, Chow AM, Tang DWF, Li Q, Wang X, Brown IR, Kerman K. Interaction of baicalein and copper with α-synuclein: electrochemical approach to Parkinson’s disease. J Electroanal Chem. 2010;648(2):151–5. https://doi.org/10.1016/j.jelechem.2010.07.015.
Sonuc KM, Sezginturk MK. Cerebrospinal fluid levels of alpha-synuclein measured using a poly-glutamic acid-modified gold nanoparticle-doped disposable neuro-biosensor system. Analyst. 2019;144(2):611–21. https://doi.org/10.1039/c8an01279b.
Eberling JL, Dave KD, Frasier MA. α-Synuclein imaging: a critical need for Parkinson’s disease research. J Parkinsons Dis. 2013;3(4):565–7. https://doi.org/10.3233/JPD-130247.
Xu MM, Ren WM, Tang XC, Hu YH, Zhang HY. Advances in development of fluorescent probes for detecting amyloid-beta aggregates. Acta Pharmacol Sin. 2016;37(6):719–30. https://doi.org/10.1038/aps.2015.155.
Goldberg JM, Batjargal S, Chen BS, Petersson EJ. Thioamide quenching of fluorescent probes through photoinduced electron transfer: mechanistic studies and applications. J Am Chem Soc. 2013;135(49):18651–8. https://doi.org/10.1021/ja409709x.
Naiki H, Higuchi K, Hosokawa M, Takeda T. Fluorometric determination of amyloid fibrils in vitro using the fluorescent dye, thioflavin-T1. Anal Biochem. 1989;177(2):244–9. https://doi.org/10.1016/0003-2697(89)90046-8.
Biancalana M, Koide S. Molecular mechanism of thioflavin-T binding to amyloid fibrils. Bba-Bioenergetics. 2010;1804(7):1405–12. https://doi.org/10.1016/j.bbapap.2010.04.001.
Celej MS, Jares-Erijman EA, Jovin TM. Fluorescent N-arylaminonaphthalene sulfonate probes for amyloid aggregation of α-synuclein. Biophys J. 208;94(12):4867–4879. https://doi.org/10.1529/biophysj.107.125211.
Pravin N, Kumar R, Tripathi S, Kumar P, Mohite GM, Navalkar A, Panigrahi R, Singh N, et al. Benzimidazole-based fluorophores for the detection of amyloid fibrils with higher sensitivity than thioflavin-T. J Neurochem. 2021;156(6):1003–19. https://doi.org/10.1111/jnc.15138.
Gaur P, Galkin M, Kurochka A, Ghosh S, Yushchenko DA, Shvadchak VV. Fluorescent probe for selective imaging of α-synuclein fibrils in living cells. Acs Chem Neurosci. 2021;12(8):1293–8. https://doi.org/10.1021/acschemneuro.1c00090.
Needham LM, Weber J, Varela JA, Fyfe JWB, Do DT, Xu CK, Tutton L, Cliffe R, et al. ThX-a next-generation probe for the early detection of amyloid aggregates. Chem Sci. 2020;11(18):4578–83. https://doi.org/10.1039/C9SC04730A.
Gorka F, Daly S, Pearson CM, Bulovaite E, Zhang YP, Handa A, Grant SGN, Snaddon TN, et al. A comparative study of high-contrast fluorescence lifetime probes for imaging amyloid in tissue. J Phys Chem B. 2021;125(50):13710–7. https://doi.org/10.1021/acs.jpcb.1c07762.
Needham LM, Weber J, Pearson CM, Do DT, Gorka F, Lyu G, Bohndiek SE, Snaddon TN, Lee SF. A comparative photophysical study of structural modifications of thioflavin T-inspired fluorophores. J Phys Chem Lett. 2020;11(19):8406–16. https://doi.org/10.1021/acs.jpclett.0c01549.
Shi S, Mitteregger-Kretzschmar G, Giese A, Kretzschmar HA. Establishing quantitative real-time quaking-induced conversion (qRT-QuIC) for highly sensitive detection and quantification of PrPSc in prion-infected tissues. Acta Neuropathol Commun. 2013;1:44. https://doi.org/10.1186/2051-5960-1-44.
Bongianni M, Catalan M, Perra D, Fontana E, Janes F, Bertolotti C, et al. Olfactory swab sampling optimization for α-synuclein aggregate detection in patients with Parkinson’s disease. Transl Neurodegener. 2022;11(1):37. https://doi.org/10.1186/s40035-022-00311-3.
Bhumkar A, Magnan C, Lau D, Jun ESW, Dzamko N, Gambin Y, Sierecki E. Single-molecule counting coupled to rapid amplification enables detection of α-synuclein aggregates in cerebrospinal fluid of Parkinson’s disease patients. Angew Chem Int Ed Engl. 2021;60(21):11874–83. https://doi.org/10.1002/anie.202014898.
Singh PK, Kotia V, Ghosh D, Mohite GM, Kumar A, Maji SK. Curcumin modulates α-synuclein aggregation and toxicity. Acs Chem Neurosci. 2013;4(3):393–407. https://doi.org/10.1021/cn3001203.
Bieschke J, Russ J, Friedrich RP, Ehrnhoefer DE, Wobst H, Neugebauer K, Wanker EE. EGCG remodels mature α-synuclein and amyloid-β fibrils and reduces cellular toxicity. Proc Natl Acad Sci. 2010;107(17):7710–5. https://doi.org/10.1073/pnas.0910723107.
Wagner J, Ryazanov S, Leonov A, Levin J, Shi S, Schmidt F, Prix C, Pan-Montojo F, et al. Anle138b: a novel oligomer modulator for disease-modifying therapy of neurodegenerative diseases such as prion and Parkinson’s disease. Acta Neuropathol. 2013;125(6):795–813. https://doi.org/10.1007/s00401-013-1114-9.
Needham LM, Weber J, Fyfe JWB, Kabia OM, Do DT, Klimont E, Zhang Y, Rodrigues M, Dobson CM, Ghandi S, Bohndiek SE, Snaddon TN, Lee SF. Correction to bifunctional fluorescent probes for detection of amyloid aggregates and reactive oxygen species. R Soc Open Sci. 2018;5(2):171399. https://doi.org/10.1098/rsos.180308.
Yu L, Cui J, Padakanti PK, Engel L, Bagchi DP, Kotzbauer PT, Tu Z. Synthesis and in vitro evaluation of α-synuclein ligands. Bioorgan Med Chem. 2012;20(15):4625–34. https://doi.org/10.1016/j.bmc.2012.06.023.
Kovalska VB, Losytskyy MY, Tolmachev OI, Slominskii YL, Segers-Nolten GMJ, Subramaniam V, Yarmoluk SM. Tri- and pentamethine cyanine dyes for fluorescent detection of α-synuclein oligomeric aggregates. J Fluoresc. 2012;22(6):1441–8. https://doi.org/10.1007/s10895-012-1081-x.
Paslawski W, Andreasen M, Nielsen SB, Lorenzen N, Thomsen K, Kaspersen JD, Pedersen JS, Otzen DE. High stability and cooperative unfolding of α-synuclein oligomers. Biochemistry-US. 2014;53(39):6252–63. https://doi.org/10.1021/bi5007833.
Lee J, Lee I, Choe Y, Kang S, Kim HY, Gai W, Hahn J, Paik SR. Real-time analysis of amyloid fibril formation of α-synuclein using a fibrillation-state-specific fluorescent probe of JC-1. Biochem J. 2009;418(2):311–23. https://doi.org/10.1042/BJ20081572.
Wang Y, Qiu Y, Sun A, Xiong Y, Tan H, Shi Y, Yu P, Roy G, et al. Dual-functional AIE fluorescent probes for imaging β-amyloid plaques and lipid droplets. Anal Chim Acta. 2020;1133:109–18. https://doi.org/10.1016/j.aca.2020.07.073.
Mei J, Leung NLC, Kwok RTK, Lam JWY, Tang BZ. Aggregation-induced emission: together we shine, united we soar. Chem Rev. 2015;115(21):11718–940. https://doi.org/10.1021/acs.chemrev.5b00263.
Marzano NR, Wray KM, Johnston CL, Paudel BP, Hong Y, van Oijen A, Ecroyd H. An α-cyanostilbene derivative for the enhanced detection and imaging of amyloid fibril aggregates. Acs Chem Neurosci. 2020;11(24):4191–202. https://doi.org/10.1021/acschemneuro.0c00478.
Leung CW, Guo F, Hong Y, Zhao E, Kwok RT, Leung NL, Chen S, Vaikath NN, El-Agnaf OM, Tang Y, Gai WP, Tang BZ. Detection of oligomers and fibrils of α-synuclein by AIE-gen with strong fluorescence. ChemComm (Camb). 2015;51(10):1866–9. https://doi.org/10.1039/C4CC07911F.
Benadiba M, Luurtsema G, Wichert-Ana L, Buchpigel CA, Busatto FG. New molecular targets for PET and SPECT imaging in neurodegenerative diseases. Rev Bras Psiquiatr. 2012;34(S2):S125–36. https://doi.org/10.1016/j.rbp.2012.07.002.
Brooks DJ. Imaging familial and sporadic neurodegenerative disorders associated with Parkinsonism. Neurotherapeutics. 2021;18(2):753–71. https://doi.org/10.1007/s13311-020-00994-4.
Cortes-Blanco A, Prieto-Yerro C, Martinez-Lazaro R, Zamora J, Jiménez-Huete A, Haberkamp M, Pohly J, Enzmann H, Zinserling J, Strassmann V, Broich K. Florbetapir (18F) for brain amyloid positron emission tomography: highlights on the European marketing approval. Alzheimers Dement. 2014;10(5 Suppl):S395–9. https://doi.org/10.1016/j.jalz.2013.09.007.
Rominger A, Brendel M, Burgold S, Keppler K, Baumann K, Xiong G, et al. Longitudinal assessment of cerebral β-amyloid deposition in mice overexpressing Swedish mutant β-amyloid precursor protein using 18F florbetaben PET. J Nucl Med. 2013;54:1127–34. https://doi.org/10.2967/jnumed.112.114660.
Barthel H, Sabri O. Florbetaben to trace amyloid-β in the Alzheimer brain by means of PET. J Alzheimers Dis. 2011;26(Suppl 3):117–21. https://doi.org/10.3233/JAD-2011-0068.
Martínez G, Vernooij RW, Fuentes Padilla P, Zamora J, Flicker L, Bonfill Cosp X. 18F PET with flutemetamol for the early diagnosis of Alzheimer’s disease dementia and other dementias in people with mild cognitive impairment (MCI). Cochrane Database Syst Rev. 2017; 11(11):CD012884. https://doi.org/10.1002/14651858.CD012884.
Skovronsky D, Dyrks T, Holl G, Krause S, Friebe M, Lehman L, Lindemann S, Dinkelborg LM, Masters CL, Villemagne VL. Imaging of amyloid beta in Alzheimer’s disease with 18F-BAY94-9172, a novel PET tracer: proof of mechanism. Lancet Neurol. 2008;7(2):129–35. https://doi.org/10.1016/S1474-4422(08)70001-2.
Shah M, Seibyl J, Cartier A, Bhatt R, Catafau AM. Molecular imaging insights into neurodegeneration: focus on alpha-synuclein radiotracers. J Nucl Med. 2014;55(9):1397–400. https://doi.org/10.2967/jnumed.113.136515.
Klunk WE, Engler H, Nordberg A, Wang Y, Blomqvist G, Holt DP, et al. Imaging brain amyloid in Alzheimer’s disease with pittsburgh compound-B. Ann Neurol. 2004;55(3):306–19. https://doi.org/10.1002/ana.20009.
Fodero-Tavoletti MT, Mulligan RS, Okamura N, Furumoto S, Rowe CC, Kudo Y, Masters CL, Cappai R, et al. In vitro characterisation of BF227 binding to α-synuclein/Lewy bodies. Eur J Pharmacol. 2009;617(1–3):54–8. https://doi.org/10.1016/j.ejphar.2009.06.042.
Josephson L, Stratman N, Liu Y, Qian F, Liang SH, Vasdev N, Patel S. The binding of BF-227-like benzoxazoles to human α-synuclein and amyloid β peptide fibrils. Mol Imaging. 2018;17:296472781. https://doi.org/10.1177/1536012118796297.
Koga S, Ono M, Sahara N, Higuchi M, Dickson DW. Fluorescence and autoradiographic evaluation of tau PET ligand PBB3 to α-synuclein pathology. Mov Disord. 2017;32(6):884–92. https://doi.org/10.1002/mds.27013.
Fujishiro H, Imamura AY, Lin WL, Uchikado H, Mark MH, Golbe LI, Markopoulou K, Wszolek ZK, et al. Diversity of pathological features other than Lewy bodies in familial Parkinson’s disease due to SNCA mutations. Am J Neurodegener Dis. 2013;2(4):266-275.
Bagchi DP, Yu L, Perlmutter JS, Xu J, Mach RH, Tu Z, Kotzbauer PT. Binding of the radioligand SIL23 to α-synuclein fibrils in Parkinson disease brain tissue establishes feasibility and screening approaches for developing a Parkinson disease imaging agent. PLoS ONE. 2013;8(2): e55031. https://doi.org/10.1371/journal.pone.0055031.
Zhang X, Jin H, Padakanti P, Li J, Yang H, Fan J, Mach R, Kotzbauer P, et al. Radiosynthesis and in vivo evaluation of two PET radioligands for imaging α-synuclein. Appl Sci. 2014;4(1):66–78. https://doi.org/10.3390/app4010066.
Chu W, Zhou D, Gaba V, Liu J, Li S, Peng X, et al. Design, synthesis, and characterization of 3-(Benzylidene) indolin-2-one derivatives as ligands for α-synuclein fibrils. J Med Chem. 2015;58:6002–17. https://doi.org/10.1021/acs.jmedchem.5b00571.
Yue X, Dhavale DD, Li J, Luo Z, Liu J, Yang H, Mach RH, Kotzbauer PT, Tu Z. Design, synthesis, and in vitro evaluation of quinolinyl analogues for α-synuclein aggregation. Bioorg Med Chem Lett. 2018;28(6):1011–9. https://doi.org/10.1016/j.bmcl.2018.02.031.
Maurer A, Leonov A, Ryazanov S, Herfert K, Kuebler L, Buss S, Schmidt F, Weckbecker D, et al. 11C Radiolabeling of anle253b: a putative PET tracer for Parkinson’s disease that binds to α-synuclein fibrils in vitro and crosses the blood-brain barrier. ChemMedChem. 2020;15(5):411–5. https://doi.org/10.1002/cmdc.201900689.
Hooshyar Yousefi B, Shi K, Reder S, Reder S, Herz M, Braeuer M, et al. First in vivo imaging and in vitro studies of 18F-DABTA in rat model with E46K alpha synuclein mutation. Eur J Nucl Med Mol Imaging. USA: SPRINGER. 2017;44(Suppl 2):161.
Fanti S, Bonfiglioli R, Decristoforo C. Highlights of the 30th annual congress of the EANM, Vienna 2017: “Yes we can–make nuclear medicine great again.” Eur J Nucl Med Mol I. 2018;45(10):1781–94. https://doi.org/10.1007/s00259-018-4029-9.
Clark CM, Schneider JA, Bedell BJ, Beach TG, Bilker WB, Mintun MA, et al. Use of florbetapir-PET for imaging beta-amyloid pathology. JAMA. 2011;305:275–83. https://doi.org/10.1001/jama.2010.2008.
Sabri O, Seibyl J, Rowe C, Barthel H. Beta-amyloid imaging with florbetaben. Clin Transl Imaging. 2015;3(1):13–26. https://doi.org/10.1007/s40336-015-0102-6.
Snellman A, Rokka J, Lopez-Picon FR, Eskola O, Wilson I, Farrar G, et al. Pharmacokinetics of [18F] flutemetamol in wild-type rodents and its binding to beta amyloid deposits in a mouse model of Alzheimer’s disease. Eur J Nucl Med Mol Imaging. 2012;39:1784–95. https://doi.org/10.1007/s00259-012-2178-9.
Klunk WE, Lopresti BJ, Ikonomovic MD, Lefterov IM, Koldamova RP, Abrahamson EE, et al. Binding of the positron emission tomography tracer Pittsburgh compound-B reflects the amount of amyloid-beta in Alzheimer’s disease brain but not in transgenic mouse brain. J Neurosci. 2005;25:10598–606. https://doi.org/10.1523/JNEUROSCI.2990-05.2005.
Levigoureux E, Lancelot S, Bouillot C, Chauveau F, Verdurand M, Verchere J, et al. Binding of the PET radiotracer [18F]BF227 does not reflect the presence of alpha-synuclein aggregates in transgenic mice. Curr Alzheimer Res. 2014;11:955–60.
Perez-Soriano A, Arena JE, Dinelle K, Miao Q, McKenzie J, Neilson N, Puschmann A, Schaffer P, Shinotoh H, Smith-Forrester J, Shahinfard E, Vafai N, Wile D, Wszolek Z, Higuchi M, Sossi V, Stoessl AJ. PBB3 imaging in Parkinsonian disorders: evidence for binding to tau and other proteins. Mov Disord. 2017;32(7):1016–24. https://doi.org/10.1002/mds.27029.
Aboagye E, Kraeber-Bodéré F. Highlights lecture EANM 2016: “Embracing molecular imaging and multi-modal imaging: a smart move for nuclear medicine towards personalized medicine.” Eur J Nucl Med Mol Imaging. 2017;44:1559–74. https://doi.org/10.1007/s00259-017-3704-6.
Yousaf T, Dervenoulas G, Politis M. Advances in MRI methodology. Int Rev Neurobiol. 2018;141:31–76. https://doi.org/10.1016/bs.irn.2018.08.008.
Cousins O, Yousaf T, Wilson H, Pagano G, Politis M. Molecular imaging of dementia with Lewy bodies. Int Rev Neurobiol. 2019;144:59–93. https://doi.org/10.1016/bs.irn.2018.10.007.
Politis M. Neuroimaging in Parkinson disease: from research setting to clinical practice. Nat Rev Neurol. 2014;10(12):708–22. https://doi.org/10.1038/nrneurol.2014.205.
Khairnar A, Ruda-Kucerova J, Szabó N, Drazanova E, Arab A, Hutter-Paier B, Neddens J, Latta P, Starcuk ZJ, Rektorova I. Early and progressive microstructural brain changes in mice overexpressing human α-synuclein detected by diffusion kurtosis imaging. Brain Behav Immun. 2017;61:197–208. https://doi.org/10.1016/j.bbi.2016.11.027.
Ding XB, Wang XB, Xia DH, Liu H, Tian HY, Fu Y, Chen YK, Qin C, Wang JQ, Xiang Z, Zhang ZX, Cao QC, Wang W, Li JY, Wu E, Tang BS, Ma MM, Teng JF, Wang XJ. Impaired meningeal lymphatic drainage in patients with idiopathic Parkinson’s disease. Nat Med. 2021;27(3):411–8. https://doi.org/10.1038/s41591-020-01198-1.
Acknowledgements
We thank Prof. Yanjun Zhang, Prof. Pengwei Zhuang, and Associate Prof. Hong Guo for their valuable comments and suggestions on this manuscript.
Funding
This study has been partially funded by the Tianjin Science and Technology Planning Project (21JCQNJC01350) and co-funded by the Tianjin University of Traditional Chinese Medicine Scientific Research Support Project and Tianjin University of Traditional Chinese Medicine twelfth College Students Science and Technology Innovation Fund (ZR19). The funders had no role in the study design or decision to publish this article.
Author information
Authors and Affiliations
Contributions
Dr. Mei-ling Chen and Dr. Yu-bo Li conceived the idea. Rui Wang, Jing-ya Li, Chan-lian Li, and Jun-miao Liu completed the work of literature retrieval. Dr. Shun-chao Pang and Rui Wang provided written input on the scientific content of drafts. Dr. Mei-ling Chen, Rui Wang, and Yu-ming Wang revised the manuscript and approved the final draft for submission. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
The authors mentioned have participated in this article.
Consent for publication
The authors approved the publication of this article.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, R., Pang, Sc., Li, Jy. et al. A review of the current research on in vivo and in vitro detection for alpha-synuclein: a biomarker of Parkinson’s disease. Anal Bioanal Chem 415, 1589–1605 (2023). https://doi.org/10.1007/s00216-023-04520-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-023-04520-1