Abstract
A novel strip test system combining immunomagnetic separation with lateral flow immunoassay (LFIA) was established for the accurate detection of Listeria monocytogenes. In this system, a pair of matched monoclonal antibodies was used to construct a sandwich immunoassay, in which superparamagnetic particles were coupled with one of the antibodies as a labeled antibody to capture the target bacteria, while the other antibody was immobilized on the detection zone. After a 20-min reaction, the strips were analyzed by a novel instrument which could detect the magnetic signal of the immunocomplex in a magnetic field. Sensitivity evaluation showed that the limit of detection (LOD) of the superparamagnetic LFIA system for L. monocytogenes was 104 CFU/mL, which was at least one log lower than conventional LFIA. No cross-reaction was observed when Salmonella, Escherichia coli O157:H7, or three types of harmless Listeria strains were tested. Further evaluation with actual food samples indicated that the superparamagnetic LFIA system showed 100 % concordance with real-time PCR. Therefore, this novel superparamagnetic LFIA system could be used as a rapid, sensitive, and specific method for the detection of L. monocytogenes.

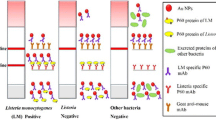
Schematic representation for the superparamagnetic LFIA system. Cartoon schematic showing the structure of the LFIA strip (a), negative result schematic when there was no L. monocytogenes in the sample (b), positive result schematic when there was L. monocytogenes in the sample (c)
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Listeria monocytogenes (L. monocytogenes, LM) is a Gram-positive microbe and the only species in the genus of Listeria which is pathogenic to human beings. Pathogenic infection by L. monocytogenes can lead to listeriosis, a deadly bacterial infection which has high hospitalization and fatality rates for newborns, pregnant women, the elderly, and immune-compromised individuals [1, 2]. With high tolerance to different growth conditions including high concentrations of salt and low pH levels, L. monocytogenes can easily survive in raw foods and ready-to-eat foods, which pose great potential threats to food safety and public health [3]. LM’s ability to grow in refrigerators at 4 °C makes it even more dangerous for human health [3–5].
For the sake of food supervision and control, rapid detection methods for L. monocytogenes have been widely reported since they save the time which traditional colony culture used to take [6]. Biochemical assays, molecular approaches, and immunoassays are considered as three major categories of methodologies for the rapid detection of target bacteria [7]. However, biochemical assays require large-scale equipment and expensive reagents which makes them not suitable for extensive use. Molecular approaches which rely on polymerase chain reaction are widely applied, but they also need well-equipped facilities and well-trained laboratory technicians to reduce the false-positive results. Immunoassays, which take full advantage of the binding effects between antigen and antibody, are sensitive and reproducible and are more convenient compared to genetic approaches since only a few simple procedures are needed.
Lateral flow immunoassay is one of the immunoassays that are widely applied in on-site detection. It provides a simple strip test which uses a nanoparticle-labeled antibody probe to quickly detect the sample without any expensive equipment and skilled analysts [8, 9]. So far, colloidal gold is one of the most frequently used nanoparticles because of its physical stability and low cost. However, it can only enable a semiquantitative analysis based on visual observation without concrete data [10]. To overcome this disadvantage, a lateral flow immunoassay using superparamagnetic beads was developed in this study, with magnetic beads substituting for colloidal gold to establish a detection method integrating immunomagnetic separation and immunoassay. Superparamagnetic particles possess stable physical properties and uniform size that will not aggregate or react with other reagents or materials after being labeled. With the help of magnetic assay reader, an ideal device to measure the quantity of magnetic tracers, a fast screening kit of L. monocytogenes in food samples can be developed. The sensitivity and specificity of the system were investigated in the current study. Practical food samples were also used to evaluate the accuracy of the system.
Material and methods
Chemical reagents, apparatus, and other materials
Sodium phosphate dibasic (Na2HPO4, 99 %), sodium phosphate monobasic monohydrate (NaH2PO4·H2O, 98–102.0 %), Triton X-100, and bovine serum albumin (BSA) were purchased from Shanghai Sangon Ltd. (Shanghai, China). 2-(N-Morpholino) ethanesulfonic acid (MES) was purchased from Sigma-Aldrich. 1-Ethyl-3-[3-dimethylaminopropyl]carbodiimide hydrochloride (EDC) and N-hydroxysulfosuccinimide (Sulfo-NHS) were purchased from Thermo Fisher Scientific Inc. (Rockford,, IL, USA). Goat anti-mouse IgG antibody was purchased from Arista Biologicals Inc. (PA, USA). Monoclonal antibodies against L. monocytogenes (LZH1 and LZF7) were purchased from Abcam (Cambridge, UK). Superparamagnetic particles (140 nm) were obtained from Key Laboratory for Special Functional Materials, Henan University (Henan, China). Nitrocellulose membrane, the conjugate pad, and the absorbent pad were purchased from Whatman Corporation (Clifton, NJ, USA). XYZ Dispensing System and CM4000 Guillotine Cutter were purchased from BioDot Inc. (Irvine, CA, USA). The MAR Assay Development System (magnetic assay reader, MAR) was donated by Magna Biosciences LLC (CA, USA). Listeria enrichment broth (LEB) was bought from OXOID Company (Basingstoke, UK). 7500 Real-time PCR equipment was purchased from ABI Company (Foster City, CA, USA). Several types of bacteria including L. monocytogenes (CMCC54002), Escherichia coli O157:H7 (ATCC11775), Salmonella (ATCC35640), Listeria ivanovii (ATCC19119), Listeria innocua (isolated by Guangdong Institute of Microbiology, Guangzhou), Listeria welshimeri, Listeria seeligeri (ATCC35967), and Listeria grayi (isolated by Guangdong Institute of Microbiology, Guangzhou) were obtained from Shenzhen Entry-Exit Inspection and Quarantine Bureau.
Conjugation of superparamagnetic particles with the antibody against L. monocytogenes
The carboxyl-functionalized superparamagnetic particles were conjugated with the LM antibody (LZH1) based on the method and protocol of Bangs Laboratories, Inc. [11]. To form an amine-reactive sulfo-NHS ester, 2.5 mg of the particles were mixed with 2.1 mM NHS and 4.2 mM EDC in MES-buffered saline at pH 5.0. After washing and magnetic separation, the particles were dispersed in 50 mM borate buffer (pH = 8.5). After the addition of 0.1 mg of LM antibody, the solution was incubated for 3 h at room temperature until a stable amide bond formed between the antibody and the magnetic particles. Residual active coupling sites were blocked by 10 % BSA solution at 37 °C for 30 min. The conjugated magnetic particles prepared via this protocol were stable for approximately 12 months when stored at 4 °C.
Preparation of the superparamagnetic LFIA strips
LZF7 monoclonal antibodies against L. monocytogenes were diluted in 10 mM sodium phosphate buffer to a concentration of 2 mg/mL and striped onto the nitrocellulose membrane as the test line. Goat anti-mouse IgG antibodies were diluted in 10 mM sodium phosphate buffer and striped at 1 mg/mL onto the nitrocellulose membrane as the control line. Both diluted antibodies were simultaneously dispensed onto the membrane using the XYZ Dispensing System. The distance between the test line and control line is 5 mm. The striped nitrocellulose membranes were dried at 37 °C for 4 h in a vacuum oven. The magnetic bead-labeled antibodies were dispensed at a ratio of 1 ml/strip onto the conjugate pad by XYZ Dispensing System. The sample pad was saturated with 20 mM sodium phosphate buffer containing BSA (1 %, w/v) and Triton X-100 (0.5 %, w/v). Both the pads were dried at 37 °C for 3 h in a vacuum oven. The superparamagnetic lateral flow immunoassay (LFIA) system was assembled in its standard configuration as shown in Fig. 1. The prepared strip was cut into individual 5 mm using the CM4000 Guillotine Cutter and was incorporated into a plastic board subsequently to form the testing kit.
Schematic representation for the superparamagnetic LFIA system. A Cartoon schematic showing the structure of the LFIA strip (a), negative result schematic when there was no L. monocytogenes in the sample (b), positive result schematic when there was L. monocytogenes in the sample (c). B An image of the magnetic assay reader
Analytical procedure
The standard strain of L. monocytogenes was activated and enriched by culture at 30 ± 1 °C for 24 h. After washing by 10 mM phosphate buffer solution (PBS) for three times and centrifugation at 8500 rpm for 15 min, 0.3 % formalin was added to the bacteria pellet for 24 h to inactivate them. Fifty microliters of inactivated L. monocytogenes was then added onto the sample port of the strip, and the captured magnetic signals were read by MAR after a 20-min reaction. The magnetic signal is expressed as the relative magnetic units (RMU) and is shown to be directly proportional to the amount of magnetic particles on the strip [12, 13]. The ratio between magnetic signals of test line (RMUtest) and those of the control line (RMUcontrol) was calculated according to the formula below. Fifty negative samples were detected by LFIA strip method; the average value was calculated, and two times of this value is defined as the cutoff value, that is 0.015. A relative signal rate of 0.015 or above was defined as positive, while a rate less than 0.015 was negative.
Determination of sensitivity and specificity
Sensitivity of the LFIA strip was determined by serial-diluted inactivated L. monocytogenes. Briefly, tenfold-diluted L. monocytogenes (101, 102, 103, 104, 105, 106, 107, and 108 CFU/mL) were added onto the superparamagnetic LFIA strips separately, and the signals were detected by MAR. The limit of detection (LOD) was then calculated accordingly. The specificity of the assay was examined using pathogenic bacteria such as E. coli O157:H7 and Salmonella as well as nonpathogenic Listeria species including L. ivanovii, L. innocua, L. welshimeri, L. seeligeri, and L. grayi. Each type of live organism was diluted to 108 CFU/mL with PBS. Fifty microliters of bacteria solution of each kind was then added to the superparamagnetic LFIA strip, followed by MAR detecting. The limit of detection was defined as the minimal concentration whose corresponding relative signal rate was the minimum value exceeding the cutoff value.
Practical food sample tests
Seventy-three food samples, including two positive control samples and one negative, were detected using the LFIA strip. We conduct the bacterial enrichment according to China national standard on detecting L. monocytogenes (GB478930-2010) [14]; that is, 25 g food sample was randomly selected and added to 225 mL LEB for enrichment, incubating at 30 ± 1. After incubation for 24 h, 0.1 mL primary enrichment solution was added to 10 mL LEB enrichment broth at 30 ± 1 for 24 h. Solution after twice enrichments was applied to real-time PCR and LFIA detection. After the enrichment step, each enriched solution was detected by the superparamagnetic LFIA strip and the results were recorded by MAR equipment. In this study, real-time PCR assay was conducted in parallel to assess the accuracy of the superparamagnetic LFIA strip. A pair of specific primers (forward primer 5′-TGC AAG TCC TAA GAC GCCA-3′, reverse primer 5′-CAC TGC ATC TCC GTG GTA TAC TAA-3′) and a probe (FAM-CGC CGA TTT CAT CCG CGT GTT TCT TTT CGG CG-Dabcyl) for detecting L. monocytogenes were used in real-time PCR.
Results and discussion
Performance and sensitivity of superparamagnetic LFIA
After adding the suspension culture of L. monocytogenes, the immunomagnetic beads sprayed on the prepared magnetic LFIA strips started to migrate along the strip, leaving brown straight lines on both the test line and the control line on the strip. With the help of the magnetic assay reader, quantitative data was obtained within 20 min.
As shown in Fig. 2 and Table S1 in the Electronic Supplementary Material (ESM), L. monocytogenes with a concentration of 1 × 104 CFU/mL or higher could be detected by the superparamagnetic LFIA method as their relative signal rate was higher than the threshold value (0.015), implying that the LOD of this method was 1 × 104 CFU/mL. When the concentration of L. monocytogenes decreased to less than 1 × 104 CFU/mL, the signal became too weak to be detected. Compared to the complicated methods related to nucleic acid or biosensors, lateral flow immunoassays are more convenient. In previous studies, the LODs of conventional lateral flow immunoassays are approximately from 105 to 106 CFU/mL [15, 16]. According to the current regulation (GB29921-2013) [17], the concentration limit of L. monocytogenes in food is 0 CFU/mL in China. Though the limit of detection of 104 CFU/mL is kind of higher than the limit established by the current regulation, our assay could obtain 10 times lower LODs compared to conventional lateral flow immunoassays. Since only 50 μL sample solution was needed in each strip test, it could be determined that the minimum detectable concentration of the target bacteria was 500 CFU/test. When the concentration decreases to less than 500 CFU/test, a pre-enrichment procedure could be carried out to enable analysis of low concentration samples as conventional lateral flow immunoassays [18–20]. Moreover, it should be noted that it only took 20 min to complete the superparamagnetic LFIA test, which was faster than previous reports [16, 18, 21]. Considering the rapidness and specificity of the current assay, it still could serve as an ideal tool during the preliminary screening of food.
Sensitivity of superparamagnetic LFIA strip on detecting Listeria monocytogenes. A The relative signal rate (T/C) was in positive correlation with L. monocytogenes ranging from 103 to 108 CFU/mL. B In the detection range, linear correlation between the logit transformation value of relative signal rate and the concentration of L. monocytogenes
Specificity test
In our study, different types of microorganism were subjected to the tests to evaluate the specificity of the superparamagnetic LFIA method. Results shown in Fig. 3 and ESM Table S2 suggested that the matched monoclonal antibodies against L. monocytogenes could distinguish L. monocytogenes from other pathogenic bacteria such as Salmonella and E. coli O157:H7. As high concentration (1 × 108 CFU/mL) of each bacteria species were used for the detection, although there were some weak cross-reaction effects for the detection of nonpathogenic Listeria spp. (L. ivanovii and L. seeligeri), superparamagnetic LFIA still possessed high specificity.
Advantages of the assay
Besides high sensitivity and specificity, another advantage of this method was the easy operation, making it a promising candidate for preliminary screening kit. Only two steps, adding bacterium solution and checking the result presented by the equipment, were required to perform the screening, eliminating the needs of professional laboratory technicians, and making it possible to high-throughput screening. PCR-based methods were proved to be high-efficiency detection methods for bacteria with excellent specificity [22, 23]; however, they were highly dependent on efficient DNA isolation and limited by its accuracy in detecting live bacteria [24, 25]. For antibody-based detection methods, multiple steps were usually required [18]. For example, ELISA, as a golden standard, required sample processing before analysis and several washing steps during the tests, which increased the complexity of the experimental procedure and affected the detection accuracy. The deficiencies mentioned above were avoided in the superparamagnetic LFIA described herein.
Practical food sample evaluation
Seventy-three samples from different food sources, including chicken, fish, flammulina velutipes, etc., were tested by superparamagnetic LFIA to evaluate the feasibility of this method in food safety monitoring. Real-time PCR was conducted in parallel as a reference method to increase the creditability of the results. Table 1 and ESM Table S3 showed that the results obtained via either superparamagnetic LFIA or real-time PCR were 100 % matched, suggesting that superparamagnetic LFIA harbored high accuracy on LM detection. As a preliminary screening method, superparamagnetic LFIA was able to analyze food samples rapidly with a high accuracy, which would play an important role on pathogenic bacteria detecting, especially for entry-exit inspection and quarantine. It also offered a less time-consuming protocol for food monitoring departments, without purchasing expensive equipment such as fluorescence-based real-time PCR systems. In this study, we managed to establish the correlation curve between the concentration of L. monocytogenes and the detected signal value in a certain range. However, we can only quantify the after-enrich L. monocytogenes concentration; we cannot calculate the correction factor due to the enrichment [14] at this moment. Further investigations are needed to obtain the quantitative results.
Conclusion
In the current study, a novel detecting method which combined the advantages of immunomagnetic enrichment techniques and semiquantitative lateral flow immunoassays was established for the detection of L. monocytogenes. The assay could concentrate the target antigen and rapidly analyze the sample through two simple steps and give the result within 20 min. The sensitivity and specificity was shown to be quite good. In addition, the accuracy was proved to be as high as that of real-time PCR. We believe that the superparamagnetic LFIA described here would play vital role in food safety monitoring by rapid, sensitive, specific, high-throughput, and accurate detection of L. monocytogenes.
References
Vazquez-Boland JA, Kuhn M, Berche P, Chakraborty T, Dominguez-Bernal G et al (2001) Listeria pathogenesis and molecular virulence determinants. Clin Microbiol Rev 14:584–640
McLauchlin J (1990) Human listeriosis in Britain, 1967–85, a summary of 722 cases. 2. Listeriosis in non-pregnant individuals, a changing pattern of infection and seasonal incidence. Epidemiol Infect 104:191–201
Donnelly CW (2001) Listeria monocytogenes: a continuing challenge. Nutr Rev 59:183–194
Uyttendaele M, De Troy P, Debevere J (1999) Incidence of Listeria monocytogenes in different types of meat products on the Belgian retail market. Int J Food Microbiol 53:75–80
Johansson T (1998) Enhanced detection and enumeration of Listeria monocytogenes from foodstuffs and food-processing environments. Int J Food Microbiol 40:77–85
Galikowska E, Kunikowska D, Tokarska-Pietrzak E, Dziadziuszko H, Los JM et al (2011) Specific detection of Salmonella enterica and Escherichia coli strains by using ELISA with bacteriophages as recognition agents. Eur J Clin Microbiol Infect Dis 30:1067–1073
Garrec N, Dilasser F, Pourcher AM, Perelle S, Fach P (2003) Comparison of a cultural method with ListerScreen plus Rapid'L.mono or PCR-ELISA methods for the enumeration of L. monocytogenes in naturally contaminated sewage sludge. J Microbiol Methods 55:763–773
Shiu CM, Wang JJ, Yu FY (2010) Sensitive enzyme-linked immunosorbent assay and rapid one-step immunochromatographic strip for fumonisin B1 in grain-based food and feed samples. J Sci Food Agric 90:1020–1026
Pattarawarapan M, Nangola S, Cressey TR, Tayapiwatana C (2007) Development of a one-step immunochromatographic strip test for the rapid detection of nevirapine (NVP), a commonly used antiretroviral drug for the treatment of HIV/AIDS. Talanta 71:462–470
Liu X, Xiang JJ, Tang Y, Zhang XL, Fu QQ et al (2012) Colloidal gold nanoparticle probe-based immunochromatographic assay for the rapid detection of chromium ions in water and serum samples. Anal Chim Acta 745:99–105
Bangs Laboratories i (2002) TechNote 205
Yuan H, Yan F, Ma L, Wu F, Zhuang J et al (2011) Carboxyl-functionalized superparamagnetic Fe3O4/poly(St-co-MPS)/SiO2 composite particles for rapid and sensitive immunoassay. J Nanosci Nanotechnol 11:2232–2236
Workman S, Wells SK, Pau CP, Owen SM, Dong XF et al (2009) Rapid detection of HIV-1 p24 antigen using magnetic immuno-chromatography (MICT). J Virol Methods 160:14–21
(2010) National Food Safety Standard. Food microbiological examination: Listeria monocytogenes. 4789.30
Noguera P, Posthuma-Trumpie GA, van Tuil M, van der Wal FJ, de Boer A et al (2011) Carbon nanoparticles in lateral flow methods to detect genes encoding virulence factors of Shiga toxin-producing Escherichia coli. Anal Bioanal Chem 399:831–838
Kitao T, Miyoshi-Akiyama T, Shimada K, Tanaka M, Narahara K et al (2010) Development of an immunochromatographic assay for the rapid detection of AAC(6')-Iae-producing multidrug-resistant Pseudomonas aeruginosa. J Antimicrob Chemother 65:1382–1386
(2013) National food safety standard. Limitation of pathogenic bacteria in food. 29921
Kim H-S, Cho I-H, Seo S-M, Jeon J-W, Paek S-H (2012) In situ immuno-magnetic concentration-based biosensor systems for the rapid detection of Listeria monocytogenes. Mater Sci Eng C 32:160–166
Stevens KA, Jaykus LA (2004) Bacterial separation and concentration from complex sample matrices: a review. Crit Rev Microbiol 30:7–24
Sharma H, Mutharasan R (2013) Rapid and sensitive immunodetection of Listeria monocytogenes in milk using a novel piezoelectric cantilever sensor. Biosens Bioelectron 45:158–162
Shim WB, Choi JG, Kim JY, Yang ZY, Lee KH et al (2008) Enhanced rapidity for qualitative detection of Listeria monocytogenes using an enzyme-linked immunosorbent assay and immunochromatography strip test combined with immunomagnetic bead separation. J Food Prot 71:781–789
Liu H-B, Ramalingam N, Jiang Y, Dai C-C, Hui KM et al (2009) Rapid distribution of a liquid column into a matrix of nanoliter wells for parallel real-time quantitative PCR. Sensors Actuators B Chem 135:671–677
Ramalingam N, Liu H-B, Dai C-C, Jiang Y, Wang H et al (2009) Real-time PCR array chip with capillary-driven sample loading and reactor sealing for point-of-care applications. Biomed Microdevices 11:1007–1020
Jaradat Z, Schutze G, Bhunia A (2002) Genetic homogeneity among Listeria monocytogenes strains from infected patients and meat products from two geographic locations determined by phenotyping, ribotyping and PCR analysis of virulence genes. Int J Food Microbiol 76:1–10
Shu B, Zhang C, Xing D (2013) Highly sensitive identification of foodborne pathogenic Listeria monocytogenes using single-phase continuous-flow nested PCR microfluidics with on-line fluorescence detection. Microfluid Nanofluid 15:161–172
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 46 kb)
Rights and permissions
About this article
Cite this article
Shi, L., Wu, F., Wen, Y. et al. A novel method to detect Listeria monocytogenes via superparamagnetic lateral flow immunoassay. Anal Bioanal Chem 407, 529–535 (2015). https://doi.org/10.1007/s00216-014-8276-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-014-8276-8