Abstract
Coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has affected the world’s health systems for more than two years. This disease causes a high mortality rate followed by cytokine storm–induced oxidative stress and acute respiratory distress syndrome (ARDS). Therefore, many drugs have been considered with emphasis on their anti-inflammatory and antioxidant effects in controlling the consequences of SARS-CoV-2 infection. Icariin is a major bioactive pharmaceutical compound derived from Epimedium plants, which is known due to its anti-inflammatory and antioxidant effects. Additionally, the protective effects of icariin have been studied in different pathologies through modulating intracellular pathways. In addition to the potential effect of this compound on inflammation and oxidative stress caused by SARS-CoV-2 infection, it appears to interfere with intracellular pathways involved in viral entry into the cell. Therefore, this paper aims to review the molecular mechanisms of anti-inflammatory and antioxidant properties of icariin, and hypothesizes its potential to inhibit SARS-CoV-2 entry into host cells through modulating the intracellular pathways.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
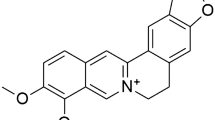
The coronavirus disease 2019 (COVID-19) epidemic originated in Wuhan, China, and it has now spread worldwide. COVID-19 is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), a new member of the coronavirus family. Despite the development of vaccines, there is no definitive cure for reducing the mortality rate caused by it. Icariin (C33H40O15) is the most abundant and the main phytochemical (secondary metabolite) of the ancient Chinese medicinal plant, Epimedium prenylflavonoids (EP), used for its potent anti-inflammatory and antioxidant properties for centuries (Li et al. 2022). It exhibits wide positive effects on bone, cardiovascular, neurologic, lung, and liver against different pathologies (Zeng et al. 2022). Table 1 summarizes the anti-inflammatory and anti-oxidative effects of icariin in experimental studies. Although no study has been conducted to investigate the effect of this compound on COVID-19, its molecular mechanisms indicate its possible effect on various aspects of the pathophysiology of this disease. Therefore, the aim of this review is to hypothesize the therapeutic potential of icariin on molecular factors involved in SARS-CoV-2 infection.
Anti-inflammatory properties of icariin
Cytokine storm is known as one of the main causes of mortality followed by SARS-CoV-2 infection (Ghasemnejad-Berenji 2021; Jiang et al. 2022). Elevated levels of different pro-inflammatory cytokines such as IL (interleukin)-1, IL-6, and tumor necrosis factor α (TNF-α) have been detected in COVID-19 patients associated with acute respiratory distress syndrome (ARDS) (Jiang et al. 2022). Accordingly, anti-inflammatory agents are an important part of treatment strategies against SARS-CoV-2 infection. One of the most well-known features of icariin is its anti-inflammatory effects, which have been studied in different models. Here, the mechanism of anti-inflammatory effects of this compound is investigated.
Angiotensin II (Ang II) is the main effector molecule of the renin-angiotensin system which is thought to be involved in inducing inflammatory responses in COVID-19 patients. Decreased ACE2 levels on the cell surface followed by its endocytosis contribute to increasing Ang II, as shown in COVID-19 patients (Khezri 2021). Elevated Ang II level has been associated with different inflammatory pathologies. One of the main targets of Ang II is angiotensin type 1 receptor (AT1R), which its activation leads to induces inflammatory responses (Phillips and Kagiyama 2002). The main downstream effector of AT1R followed by Ang II binding is the PI3K/AKT signaling pathway which activates inflammatory factors such as NF-κB (El-Shoura et al. 2018). Regardless of the PI3K/AKT/NF-κB pathway activation by Ang II, it has been shown that ORF7a protein of SARS-CoV-2 activates NF-κB leading to induce the expression of pro-inflammatory cytokines such as IL-1β, IL-1α, IL-6, IL-10, IL-8, and TNF-α (Su et al. 2021). In this case, there are numerous studies indicating the effect of icariin on the activity of mentioned factors. For instance, it has been reported that icariin suppresses the destructive effects of Ang II on human umbilical vein endothelial cells (Wang et al. 2008), cardiomyocytes (Zhou et al. 2014), and cerebrovascular remodeling (Dong et al. 2019). On the other hand, it has been reported that icariin suppresses cigarette smoke–induced TNF-α expression through NF-κB inhibition (Li et al. 2014). Also, it has been indicated that icariin has protective effects against lipopolysaccharide-stimulated macrophages through NF-κB suppression leading to inhibit TNF-α expression (Chen et al. 2010). Additionally, protective effects of icariin in a rat model of cerebral ischemia–reperfusion injury have been shown to be mediated by peroxisome proliferator–activated receptors-γ (c-γ) activation and NF-κB suppression (Xiong et al. 2016).
The other main intracellular factor involved in inflammatory responses is the signal transducer and activator of transcription (STAT), which its over-activity has been detected in the kidney of COVID-19 patients (Salem et al. 2021). Also, it has been proposed that elevated level of leptin in COVID-19 patients is associated with STAT3 and AKT over-activity and pro-inflammatory cytokine expression (Wang et al. 2021). On the other hand, STAT over-activity may be involved in Th17 response in COVID-19 patients (Martonik et al. 2021). However, the inhibitory effect of icariin on STAT activity has been shown in different models. For instance, icariin has been shown to suppress Th1/Th17 responses through suppression of STAT1 and STAT3 activation leading to inhibit inflammatory responses in an animal model of colitis (Tao et al. 2013). In addition, it has been indicated that STAT3 inhibition by icariin leads to regulating Th17 activity and alleviates rheumatoid arthritis in a murine model (Chi et al. 2014). There are several studies indicating the effect of icariin on Th17 function without emphasizing the STAT activity. In this regard, it has been shown that icariin regulates Th17/Treg function in murine airways leading to reduce the expression of IL-6 and TGF-β (Wei et al. 2015).
Neutrophil infiltration to the lungs during SARS-CoV-2 infection has been shown to be closely related to lung injury (Wang et al. 2020a). Elevated expression of several adhesion molecules involved in neutrophil recruitment such as intercellular adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule 1 (VCAM-1) has been detected in COVID-19 patients (Tong et al. 2020; Spadaro et al. 2021). Regarding the effect of icariin on neutrophil infiltration during inflammatory processes, it has been shown that icariin decreases neutrophil infiltration into the lung followed by lipopolysaccharide-induced acute inflammation in rats via NF-κB suppression (Xu et al. 2010). On the other hand, it has been demonstrated that icariin suppresses ICAM-1 expression via NF-κB inhibition leading to suppression of neutrophil recruitment and inhibiting lung injury in mice affected with cobra venom factor (Guo et al. 2018). In another study, it has been demonstrated that treatment of keratinocytes with icariin followed by TNF-α/IFN-γ-induced inflammation leads to inhibit the expression of IL-8, IL-1β, and ICAM-1 performing an anti-inflammatory effect (Kong et al. 2015). Additionally, icariin has been reported to suppress ICAM-1 and VCAM-1 expression and exhibits a protective effect against high glucose-induced inflammation in human umbilical vein endothelial cells (Sun et al. 2019).
One of the main consequences of cytokine storm is fibrosis in different organs especially the lungs (Grillo et al. 2021). In COVID-19 patients, Ang II-AT1R axis and elevated inflammatory cytokines are represented as the main cause of lung fibrosis (Brosnahan et al. 2020). In this case, it can be re-referred to the PI3K/AKT singling pathway, as it has been shown that Ang II-AT1R axis induces activation of this pathway leading to lung fibrosis induction (Hu et al. 2018). However, it has been demonstrated that icariin augments bleomycin-induced pulmonary fibrosis in rats by targeting the NF-κB pathway (Du et al. 2021). Based on this evidence, it can be said that negative regulation of the PI3K/AKT pathway by icariin may lead to suppression of lung fibrosis in COVID-19 patients and further studies in this regard may be constructive.
The other consequence of high levels of Ang II and cytokine storm in COVID-19 patients is blood coagulation which is closely related to the severity of the disease (Han et al. 2020; Khezri and Ghasemnejad-Berenji 2021). Interestingly, it has been reported that platelets isolated from COVID-19 patients are over-activated due to over-activity of the PI3K/AKT signaling pathway (Pelzl et al. 2021). Since different studies have been conducted to suppress blood coagulation through inhibition of this pathway (Khezri et al. 2022), designing similar studies in order to clarifying the effect of icariin on platelet activity in COVID-19 patients can also be considered. However, previous studies have indicated the anti-platelet activity of icariin without emphasizing on mentioned factors. For instance, it has been demonstrated that icariin improves the imbalance between plasminogen activator inhibitor-1 and tissue-type plasminogen activator activities, suppresses platelet activity, and inhibits blood coagulation in rabbits fed a high-cholesterol diet (Zhang et al. 2013; Irfan et al. 2021). In addition, inhibitory effects of icariin on platelet activation have been reported in spontaneously hypertensive rats (Li et al. 2021).
Antioxidant effects of icariin
Disturbing the balance between free radicals’ generation and the ability of cells to their elimination leads to oxidative stress induction (Pizzino et al. 2017). Reactive oxygen species (ROS) play a central role in alterations in pulmonary and red blood cell activity and contribute to hypoxic respiratory failure in severe cases of COVID-19 (Laforge et al. 2020). Since oxidative stress plays a crucial role in pathogenesis of respiratory viruses, especially SARS-CoV-2, therapeutic interventions to modify oxidative stress represent a rational approach for the treatment of lower respiratory tract infections (Karkhanei et al. 2021). However, there are several intracellular factors which can be examined here. Nuclear factor erythroid 2–related transcription factor (Nrf2) is one of the main transcription factors of antioxidant enzymes which its activation exhibits protective effects against different pathologies (Liu et al. 2019). Therefore, therapeutic interventions to activate it in SARS-CoV-2 infection have been highly regarded as a treatment option (Cuadrado et al. 2020). Regarding the Nrf2-activating effects of icariin, it has been shown that icariin augments cigarette smoke–induced oxidative stress in lung human epithelial cells through Nrf2 activation, increased glutathione levels, and suppression of ROS generation (Wu et al. 2014). Additionally, protective effects of icariin on hypoxia/reoxygenation-induced oxidative stress have been shown to be mediated by Nrf2-heme oxygenase 1 (HO-1) activation in cardiomyocytes (Sun et al. 2021). Also, it has been reported that icariin inhibits carrageenan-induced acute inflammation and oxidative stress in rats through increasing the Nrf2 expression and reducing NF-κB expression (El-Shitany and Eid 2019).
Hypothesis of possible effect of icariin on SARS-CoV-2 entry to the host cells
In order to study the effect of a compound on COVID-19, it is necessary to first explain its potential ability to suppress entry of the SARS-CoV-2 into the host cell. So far, four main factors are presented mediating the SARS-CoV-2 entry to host cells, including angiotensin-converting enzyme 2 (ACE2), transmembrane Serine Protease 2 (TMPRSS2), furin, and cluster of differentiation 147 (CD147).
ACE2 is an enzymatic receptor on cell surface which has been introduced as the main SARS-CoV-2 receptor because of its high expression on respiratory system cells (Jiang et al. 2022). In addition to the respiratory system, high expression of ACE2 has been shown in different other organs, including the intestine, skin, and testes (reviewed by Khezri et al. (2021)). Regarding the mechanism of SARS-CoV-2 entry to host cells through ACE2, it has been shown that clathrin-mediated endocytosis is involved (Bayati et al. 2021). In a closer inspection, it has been indicated that phosphorylation of AP2M1 which encodes the μ2 subunit of AP2 complex, an adapter protein complex for clathrin, is involved in clathrin-mediated SARS-CoV-2 endocytosis (Wang et al. 2020b). One of the main regulators of the clathrin-mediated endocytosis is the phosphatidylinositol 3‑kinase (PI3K)/AKT signaling pathway which is involved in entry of different viruses that use clathrin-mediated endocytosis to infect host cells (reviewed by Khezri et al. (2021)). Interestingly, the PI3K/AKT signaling pathway suppression has been shown to suppress SARS-CoV-2 entry to host cells (Sun et al. 2021). In a closer inspection, existing evidence suggests that the association between the PI3K/AKT signaling pathway and clathrin-mediated endocytosis of SARS-CoV-2 may be mediated by AP2M1. In this regard, it has been shown that activation of the PI3K/AKT signaling pathway in acute myeloid leukemia stem cells contributes to induce AP2M1 expression (Yu et al. 2021). In addition, it has been reported that sunitinib, a PI3K/AKT inhibitor, reduces SARS-CoV-2 infection via inhibiting AP2M1 phosphorylation (Wang et al. 2020b). Based on this evidence, it can be said that the effect of a compound on the activity of this pathway can affect the entry of SARS-CoV-2 into the cell. It is clearly understood that icariin suppresses the PI3K/AKT signaling pathway. In this case, it has been shown that icariin augments lipopolysaccharide-induced inflammation in rat lung tissue through inhibition of the PI3K/AKT signaling pathway (Xu et al. 2010). In addition, it has been shown that icariin induces apoptosis of human lung adenocarcinoma cells through suppression of the PI3K/AKT pathway (Wu et al. 2019). This data suggests the probable inhibitory effect of icariin on SARS-CoV-2 through suppression of clathrin-mediated endocytosis by inhibition of the PI3K/AKT/AP2M1 pathway.
Another aspect of the study of ACE2 in COVID-19 is related to its shedding which contributes to form soluble ACE2 (sACE2) (Zoufaly et al. 2020). It is clearly understood that increased sACE2 levels suppress SARS-CoV-2 infectivity through binding to its Spike protein (Zoufaly et al. 2020). One of the main enzymes involved in ACE2 shedding is a disintegrin and metalloprotease 17 (ADAM17) which its role in suppression of SARS-CoV-2 infectivity has also been studied (Jiang et al. 2022). In this regard, although there is no evidence indicating the effect of icariin on ADAM17 expression and activity, its effect on pathways involved in ADAM17 expression can be examined. Mitogen-activated protein kinase (MAPK) is an intracellular factor which regulates ADAM17 expression positively and is involved in ACE2 shedding by mediating of ADAM17 (de Queiroz et al. 2020). On the other hand, it has been shown that icariin induces mesenchymal stem cell differentiation via MAPK activation (Wu et al. 2015). Therefore, studies can be designed to investigate the effect of icariin on the MAPK/ADAM17 pathway, ACE2 shedding, and subsequently suppression of virus infectivity.
TMPRSS2 is involved in SARS-CoV-2 fusion into the cell (Matsuyama et al. 2020). Extensive expression of TMPRSS2 in various tissues and cells introduces it as a candidate for therapeutic interventions to prevent the SARS-CoV-2 entry and replication in the host cell. Regarding the association between icariin and TMPRSS2, in a computational study, it has been shown that icariin has a strong binding affinity to TMPRSS2 which introduces it as a potential inhibitor of SARS-CoV-2 entry to host cells (Chikhale et al. 2020).
CD147 is the other known receptor for SARS-CoV-2 to enter the host cells (Ulrich and Pillat 2020). It seems that this receptor is involved in SARS-CoV-2 infectivity in cardiomyocytes as it has been reported that melatonin augments myocardial injury caused by SARS-CoV-2 through CD147 (Loh 2020). In this case, it has been indicated that icariin suppresses CD147 expression and exhibits a protective role against apoptosis in myocardial cells and it can also be considered in the case of SARS-CoV-2 infection (Shi et al. 2018).
In addition to the mentioned factors, furin and different forms of cathepsins including cathepsins B, L, and K are involved in SARS-CoV-2 Spike protein cleavage leading to its fusion to the cell (Bollavaram et al. 2021). There is no data indicating the effect of icariin on furin activity, but it serves as a cathepsin K inhibitor in different tissues. For instance, it has been shown that icariin inhibits bone degradation in an animal model of arthritis through suppression of cathepsin K (Sun et al. 2013).
Although there is not any study on the effects of icariin on the mentioned factors, in an in-silico study, it has been reported that icariin could interact with the catalytic residues of the RBD of the spike glycoprotein (Tyr505, Asn501, Ser494, Gln493, and Leu455). In addition, it was shown that icariin extensively interacts with the active amino acid residues of cathepsin B (Istifli et al. 2021).
Collectively, this data introduces icariin as a strong candidate to investigate its effects on SARS-CoV-2 infectivity through different mechanisms and designing studies in this case can be constructive.
Icariin in clinical trials
Double-blinded, placebo-controlled randomized trial studies used the purified extract of EP to examine its safety, pharmacokinetics in healthy people, and its effect on osteoporosis in post-menopausal women. The results showed no substance-related adverse effects, and the drug was well tolerated. The serum levels of EP metabolites were measured, while icariside I and icariin serum levels were undetectable (Teo et al. 2019; Yong et al. 2021). Additionally, treating late menopausal osteopenia women (lumbar spine bone densitometry T score of − 2 to − 2.5) with daily ingestion of icariin in a 24-month randomized trial showed significant improvement in bone loss prevention (Zhang et al. 2007). We found no evidence of icariin application for viral diseases in humans, while in animal studies, icariin showed survival effects for duck viral hepatitis in ducklings (Xiong et al. 2014). Collectively, these studies suggest that purified icariin can be evaluated in the design of clinical trials to evaluate its various benefits in COVID-19 patients.
Conclusion
Based on the existing evidence, icariin has been approved to be an anti-inflammatory and antioxidant agent in different pathologies. These effects have been associated with its effects on different inflammatory factors (i.e., NF-κB and STATs) which contribute to inducing the expression of pro-inflammatory cytokines, the most important of which are TNF-α, IL-1, and IL-6. These factors are involved in the induction of fibrosis in various organs, and the effect of icariin on their expression has been shown to inhibit organ fibrosis in various models. Since the role of the mentioned factors in the pathophysiology of COVID-19 has been shown, the positive effects of icariin in this disease can be considered. On the other hand, the interplay between icariin and intracellular pathways involved in the entry of SARS-CoV-2 into the host cells has been investigated in non-COVID-19 models, which offers hypotheses for the beneficial effects of this compound on the disease. However, it seems that designing studies to investigate these effects in SARS-CoV-2 infection can be considered as a treatment option. Figure 1 depicts the molecular mechanisms of icariin interfering with SARS-CoV-2 infection.
Data availability
Not applicable.
References
Bayati A, Kumar R, Francis V, McPherson PS (2021) SARS-CoV-2 infects cells after viral entry via clathrin-mediated endocytosis. Journal of Biological Chemistry 296.
Bollavaram K, Leeman TH, Lee MW, Kulkarni A, Upshaw SG, Yang J, Song H, Platt MO (2021) Multiple sites on SARS-CoV-2 spike protein are susceptible to proteolysis by cathepsins B, K, L, S, and V. Protein Sci. https://doi.org/10.1002/pro.4073
Brosnahan SB, Jonkman AH, Kugler MC, Munger JS, Kaufman DA (2020) COVID-19 and respiratory system disorders: current knowledge, future clinical and translational research questions. Arterioscler Thromb Vasc Biol 40:2586–2597. https://doi.org/10.1161/ATVBAHA.120.314515
Chen S-R, Xu X-Z, Wang Y-H, Chen J-W, Xu S-W, Gu L-Q, Liu P-Q (2010) Icariin derivative inhibits inflammation through suppression of p38 mitogen-activated protein kinase and nuclear factor-κb pathways. Biol Pharm Bull 33:1307–1313. https://doi.org/10.1248/bpb.33.1307
Chi L, Gao W, Shu X, Lu X (2014) A natural flavonoid glucoside, icariin, regulates Th17 and alleviates rheumatoid arthritis in a murine model. Mediators of Inflammation 2014.https://doi.org/10.1155/2014/392062
Chikhale RV, Gupta VK, Eldesoky GE, Wabaidur SM, Patil SA, Islam MA (2020) Identification of potential anti-TMPRSS2 natural products through homology modelling, virtual screening and molecular dynamics simulation studies. Journal of Biomolecular Structure and Dynamics: 1-16.https://doi.org/10.1080/07391102.2020.1798813
Cuadrado A, Pajares M, Benito C, Jiménez-Villegas J, Escoll M, Fernández-Ginés R, Yagüe AJG, Lastra D, Manda G, Rojo AI (2020) Can activation of NRF2 be a strategy against COVID-19? Trends Pharmacol Sci 41:598–610. https://doi.org/10.1016/j.tips.2020.07.003
de Queiroz TM, Lakkappa N, Lazartigues E (2020) ADAM17-mediated shedding of inflammatory cytokines in hypertension. Front Pharmacol 11:1154. https://doi.org/10.3389/fphar.2020.01154
Dong H, Ming S, Fang J, Li Y, Liu L (2019) Icariin ameliorates angiotensin II-induced cerebrovascular remodeling by inhibiting Nox2-containing NADPH oxidase activation. Hum Cell 32:22–30. https://doi.org/10.1007/s13577-018-0220-3
Du W, Tang Z, Yang F, Liu X, Dong J (2021) Icariin attenuates bleomycin-induced pulmonary fibrosis by targeting Hippo/YAP pathway. Biomed Pharmacother 143:112152. https://doi.org/10.1016/j.biopha.2021.112152
El-Shitany NA, Eid BG (2019) Icariin modulates carrageenan-induced acute inflammation through HO-1/Nrf2 and NF-kB signaling pathways. Biomed Pharmacother 120:109567. https://doi.org/10.1016/j.biopha.2019.109567
El-Shoura EA, Messiha BA, Sharkawi SM, Hemeida RA (2018) Perindopril ameliorates lipopolysaccharide-induced brain injury through modulation of angiotensin-II/angiotensin-1- and related signaling pathways. Eur J Pharmacol 834:305–317. https://doi.org/10.1016/j.ejphar.2018.07.046
Ghasemnejad-Berenji M (2021) Immunomodulatory and anti-inflammatory potential of crocin in COVID-19 treatment. J Food Biochem 45:e13718. https://doi.org/10.1111/jfbc.13718
Grillo F, Barisione E, Ball L, Mastracci L, Fiocca R (2021) Lung fibrosis: an undervalued finding in COVID-19 pathological series. Lancet Infect Dis 21:e72. https://doi.org/10.1016/S1473-3099(20)30582-X
Guo J, Jiao L, Zhang L, Yang Y, Zhu F-j, Min L, Sun Q-y (2018) Effects of icariin on acute lung injury in mice induced by activation of the complement alternative pathway. Chin Pharmacol Bull 34:988–993
Han H, Yang L, Liu R, Liu F, Wu K-l, Li J, Liu X-H, Zhu C-l (2020) Prominent changes in blood coagulation of patients with SARS-CoV-2 infection. Clin Chem Lab Med (CCLM) 58:1116–1120. https://doi.org/10.1515/cclm-2020-0188
Hu H, Hu S, Xu S, Gao Y, Zeng F, Shui H (2018) miR-29b regulates Ang II-induced EMT of rat renal tubular epithelial cells via targeting PI3K/AKT signaling pathway. Int J Mol Med 42:453–460. https://doi.org/10.3892/ijmm.2018.3579
Irfan M, Kwon T-H, Lee D-H, Hong S-B, Oh J-W, Kim S-D, Rhee MH (2021) Antiplatelet and antithrombotic effects of Epimedium koreanum Nakai. Evidence-Based Complementary and Alternative Medicine 2021.
Istifli ES, Tepe AŞ, Netz PA, Sarikürkcü C, Kilic IH, Tepe B (2021) Determination of the interaction between the receptor binding domain of 2019-nCoV spike protein, TMPRSS2, cathepsin B and cathepsin L, and glycosidic and aglycon forms of some flavonols. Turk J Biol 45:484. https://doi.org/10.3906/biy-2104-51
Jiang Y, Rubin L, Peng T, Liu L, Xing X, Lazarovici P, Zheng W (2022) Cytokine storm in COVID-19: from viral infection to immune responses, diagnosis and therapy. Int J Biol Sci 18:459. https://doi.org/10.7150/ijbs.59272
Karkhanei B, Ghane ET, Mehri F (2021) Evaluation of oxidative stress level: total antioxidant capacity, total oxidant status and glutathione activity in patients with COVID-19. New Microbes New Infect 42:100897. https://doi.org/10.1016/j.nmni.2021.100897
Khezri MR (2021) PI3K/AKT signaling pathway: a possible target for adjuvant therapy in COVID-19. Hum Cell 34:700–701. https://doi.org/10.1007/s13577-021-00484-5
Khezri MR, Ghasemnejad-Berenji M (2021) Neurological effects of elevated levels of angiotensin II in COVID-19 patients. Hum Cell 34:1941–1942. https://doi.org/10.1007/s13577-021-00605-0
Khezri MR, Varzandeh R, Ghasemnejad-Berenji M (2022) The probable role and therapeutic potential of the PI3K/AKT signaling pathway in SARS-CoV-2 induced coagulopathy. Cell Mol Biol Lett 27:1–10. https://doi.org/10.1186/s11658-022-00308-w
Khezri MR, Zolbanin NM, Ghasemnejad-Berenji M, Jafari R (2021) Azithromycin: immunomodulatory and antiviral properties for SARS-CoV-2 infection. European journal of pharmacology: 174191.https://doi.org/10.1016/j.ejphar.2021.174191
Kong L, Liu J, Wang J, Luo Q, Zhang H, Liu B, Xu F, Pang Q, Liu Y, Dong J (2015) Icariin inhibits TNF-α/IFN-γ induced inflammatory response via inhibition of the substance P and p38-MAPK signaling pathway in human keratinocytes. Int Immunopharmacol 29:401–407. https://doi.org/10.1016/j.intimp.2015.10.023
Laforge M, Elbim C, Frère C, Hémadi M, Massaad C, Nuss P, Benoliel J-J, Becker C (2020) Tissue damage from neutrophil-induced oxidative stress in COVID-19. Nat Rev Immunol 20:515–516. https://doi.org/10.1038/s41577-020-0407-1
Li L, Sun J, Xu C, Zhang H, Wu J, Liu B, Dong J (2014) Icariin ameliorates cigarette smoke induced inflammatory responses via suppression of NF-κB and modulation of GR in vivo and in vitro. PLoS ONE 9:e102345. https://doi.org/10.1371/journal.pone.0102345
Li X, Yang HF, Chen Y, Pei LJ, Jiang R (2021) Effect of the icariin on endothelial microparticles, endothelial progenitor cells, platelets, and erectile function in spontaneously hypertensive rats. Andrology. https://doi.org/10.1111/andr.13127
Li J, Xia T, Liu J, Chen F, Chen H, Zhuo Y, Wu W (2022) Molecular mechanism by which icariin regulates osteogenic signaling pathways in the treatment of steroid-induced avascular necrosis of the femoral head. Chin J Tissue Eng Res 26:780
Liu XJ, Lv YF, Cui WZ, Li Y, Liu Y, Xue YT, Dong F (2021) Icariin inhibits hypoxia/reoxygenation-induced ferroptosis of cardiomyocytes via regulation of the Nrf2/HO-1 signaling pathway. FEBS Open Bio 11:2966–2976. https://doi.org/10.1002/2211-5463.13276
Liu Q, Gao Y, Ci X (2019) Role of Nrf2 and its activators in respiratory diseases. Oxidative medicine and cellular longevity 2019.https://doi.org/10.1155/2019/7090534
Loh D (2020) The potential of melatonin in the prevention and attenuation of oxidative hemolysis and myocardial injury from cd147 SARS-CoV-2 spike protein receptor binding. Melatonin Res 3:380–416. https://doi.org/10.32794/mr11250069
Martonik D, Parfieniuk-Kowerda A, Rogalska M, Flisiak R (2021) The role of Th17 response in COVID-19. Cells 10:1550. https://doi.org/10.3390/cells10061550
Matsuyama S, Nao N, Shirato K, Kawase M, Saito S, Takayama I, Nagata N, Sekizuka T, Katoh H, Kato F (2020) Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells. Proc Natl Acad Sci 117:7001–7003. https://doi.org/10.1073/pnas.2002589117
Pelzl L, Singh A, Funk J, Witzemann A, Zlamal J, Marini I, Weich K, Abou-Khalel W, Hammer S, Uzun G (2021) Platelet activation via PI3K/AKT signaling pathway in COVID-19. Research and Practice in Thrombosis and Haemostasis.
Phillips MI, Kagiyama S (2002) Angiotensin II as a pro-inflammatory mediator. Curr opin investig drugs (London, England: 2000) 3:569–577
Pizzino G, Irrera N, Cucinotta M, Pallio G, Mannino F, Arcoraci V, Squadrito F, Altavilla D, Bitto A (2017) Oxidative stress: harms and benefits for human health. Oxidative medicine and cellular longevity 2017.https://doi.org/10.1155/2017/8416763
Salem F, Li XZ, Hindi J, Casablanca NM, Zhong F, El Jamal SM, Haroon Al Rasheed MR, Li L, Lee K, Chan L (2021) Activation of STAT3 signaling pathway in the kidney of COVID-19 patients. Journal of nephrology: 1-9.https://doi.org/10.1007/s40620-021-01173-0
Shi Y, Yan W, Lin Q, Wang W (2018) Icariin influences cardiac remodeling following myocardial infarction by regulating the CD147/MMP-9 pathway. J Int Med Res 46:2371–2385. https://doi.org/10.1177/0300060518762060
Spadaro S, Fogagnolo A, Campo G, Zucchetti O, Verri M, Ottaviani I, Tunstall T, Grasso S, Scaramuzzo V, Murgolo F (2021) Markers of endothelial and epithelial pulmonary injury in mechanically ventilated COVID-19 ICU patients. Crit Care 25:1–9. https://doi.org/10.1186/s13054-021-03499-4
Su C-M, Wang L, Yoo D (2021) Activation of NF-κB and induction of proinflammatory cytokine expressions mediated by ORF7a protein of SARS-CoV-2. Sci Rep 11:1–12. https://doi.org/10.1038/s41598-021-92941-2
Sun P, Liu Y, Deng X, Yu C, Dai N, Yuan X, Chen L, Yu S, Si W, Wang X (2013) An inhibitor of cathepsin K, icariin suppresses cartilage and bone degradation in mice of collagen-induced arthritis. Phytomedicine 20:975–979. https://doi.org/10.1016/j.phymed.2013.04.019
Sun S, Liu L, Tian X, Guo Y, Cao Y, Mei Y, Wang C (2019) Icariin attenuates high glucose-induced apoptosis, oxidative stress, and inflammation in human umbilical venous endothelial cells. Planta Med 85:473–482. https://doi.org/10.1055/a-0837-0975
Sun F, Mu C, Kwok HF, Xu J, Wu Y, Liu W, Sabatier J-M, Annweiler C, Li X, Cao Z (2021) Capivasertib restricts SARS-CoV-2 cellular entry: a potential clinical application for COVID-19. Int J Biol Sci 17:2348. https://doi.org/10.7150/ijbs.57810
Tao F, Qian C, Guo W, Luo Q, Xu Q, Sun Y (2013) Inhibition of Th1/Th17 responses via suppression of STAT1 and STAT3 activation contributes to the amelioration of murine experimental colitis by a natural flavonoid glucoside icariin. Biochem Pharmacol 85:798–807. https://doi.org/10.1016/j.bcp.2012.12.002
Teo YL, Cheong WF, Cazenave-Gassiot A, Ji S, Logan S, Lee ZXK, Li J, Seng KY, Lee LS-U, Yong EL (2019) Pharmacokinetics of prenylflavonoids following oral ingestion of standardized epimedium extract in humans. Planta Med 85:347–355. https://doi.org/10.1055/a-0806-7673
Tong M, Jiang Y, Xia D, Xiong Y, Zheng Q, Chen F, Zou L, Xiao W, Zhu Y (2020) Elevated expression of serum endothelial cell adhesion molecules in COVID-19 patients. J Infect Dis 222:894–898. https://doi.org/10.1093/infdis/jiaa349
Ulrich H, Pillat MM (2020) CD147 as a target for COVID-19 treatment: suggested effects of azithromycin and stem cell engagement. Stem Cell Rev Rep 16:434–440. https://doi.org/10.1007/s12015-020-09976-7
Wang Q-J, Pan Z-W, Wang Y, Yang J, Jia Y, Kong L-Y (2008) Protective effects of icariin on human umbilical vein endothelial cell injured by angiotensin II. J Chin Pharm Sci 17:16
Wang J, Li Q, Yin Y, Zhang Y, Cao Y, Lin X, Huang L, Hoffmann D, Lu M, Qiu Y (2020) Excessive neutrophils and neutrophil extracellular traps in COVID-19. Front Immunol 11:2063. https://doi.org/10.3389/fimmu.2020.02063
Wang P-G, Tang D-J, Hua Z, Wang Z, An J (2020) Sunitinib reduces the infection of SARS-CoV, MERS-CoV and SARS-CoV-2 partially by inhibiting AP2M1 phosphorylation. Cell Discovery 6:1–5. https://doi.org/10.1038/s41421-020-00217-2
Wang J, Xu Y, Zhang X, Wang S, Peng Z, Guo J, Jiang H, Liu J, Xie Y, Wang J (2021) Leptin correlates with monocytes activation and severe condition in COVID-19 patients. J Leukoc Biol. https://doi.org/10.1002/JLB.5HI1020-704R
Wei Y, Liu B, Sun J, Lv Y, Luo Q, Liu F, Dong J (2015) Regulation of Th17/Treg function contributes to the attenuation of chronic airway inflammation by icariin in ovalbumin-induced murine asthma model. Immunobiology 220:789–797. https://doi.org/10.1016/j.imbio.2014.12.015
Wu J, Xu H, Wong PF, Xia S, Xu J, Dong J (2014) Icaritin attenuates cigarette smoke-mediated oxidative stress in human lung epithelial cells via activation of PI3K-AKT and Nrf2 signaling. Food Chem Toxicol 64:307–313. https://doi.org/10.1016/j.fct.2013.12.006
Wu Y, Xia L, Zhou Y, Xu Y, Jiang X (2015) Icariin induces osteogenic differentiation of bone mesenchymal stem cells in a MAPK-dependent manner. Cell Prolif 48:375–384. https://doi.org/10.1111/cpr.12185
Wu X, Kong W, Qi X, Wang S, Chen Y, Zhao Z, Wang W, Lin X, Lai J, Yu Z (2019) Icariin induces apoptosis of human lung adenocarcinoma cells by activating the mitochondrial apoptotic pathway. Life Sci 239:116879. https://doi.org/10.1016/j.lfs.2019.116879
Xiong W, Chen Y, Wang Y, Liu J (2014) Roles of the antioxidant properties of icariin and its phosphorylated derivative in the protection against duck virus hepatitis. BMC Vet Res 10:1–9. https://doi.org/10.1186/s12917-014-0226-3
Xiong D, Deng Y, Huang B, Yin C, Liu B, Shi J, Gong Q (2016) Icariin attenuates cerebral ischemia–reperfusion injury through inhibition of inflammatory response mediated by NF-κB, PPARα and PPARγ in rats. Int Immunopharmacol 30:157–162. https://doi.org/10.1016/j.intimp.2015.11.035
Xu C-Q, Liu B-J, Wu J-F, Xu Y-C, Duan X-H, Cao Y-X, Dong J-C (2010) Icariin attenuates LPS-induced acute inflammatory responses: involvement of PI3K/Akt and NF-κB signaling pathway. Eur J Pharmacol 642:146–153. https://doi.org/10.1016/j.ejphar.2010.05.012
Yong E-L, Cheong WF, Huang Z, Thu WPP, Cazenave-Gassiot A, Seng KY, Logan S (2021) Randomized, double-blind, placebo-controlled trial to examine the safety, pharmacokinetics and effects of Epimedium prenylflavonoids, on bone specific alkaline phosphatase and the osteoclast adaptor protein TRAF6 in post-menopausal women. Phytomedicine 91:153680. https://doi.org/10.1016/j.phymed.2021.153680
Yu D-H, Chen C, Liu X-P, Yao J, Li S, Ruan X-L (2021) Dysregulation of miR-138-5p/RPS6KA1-AP2M1 is associated with poor prognosis in AML. Front Cell Dev Biol 9:270. https://doi.org/10.3389/fcell.2021.641629
Zeng Y, Xiong Y, Yang T, Wang Y, Zeng J, Zhou S, Luo Y, Li L (2022) Icariin and its metabolites as potential protective phytochemicals against cardiovascular disease: from effects to molecular mechanisms. Biomed Pharmacother 147:112642. https://doi.org/10.1016/j.biopha.2022.112642
Zhang G, Qin L, Shi Y (2007) Epimedium-derived phytoestrogen flavonoids exert beneficial effect on preventing bone loss in late postmenopausal women: a 24-month randomized, double-blind and placebo-controlled trial. J Bone Miner Res 22:1072–1079. https://doi.org/10.1359/jbmr.070405
Zhang W-p, Bai X-j, Zheng X-p, Xie X-l, Yuan Z-y (2013) Icariin attenuates the enhanced prothrombotic state in atherosclerotic rabbits independently of its lipid-lowering effects. Planta Med 79:731–736. https://doi.org/10.1055/s-0032-1328551
Zhou H, Yuan Y, Liu Y, Deng W, Zong J, Bian ZY, Dai J, Tang QZ (2014) Icariin attenuates angiotensin II-induced hypertrophy and apoptosis in H9c2 cardiomyocytes by inhibiting reactive oxygen species-dependent JNK and p38 pathways. Exp Ther Med 7:1116–1122. https://doi.org/10.3892/etm.2014.1598
Zoufaly A, Poglitsch M, Aberle JH, Hoepler W, Seitz T, Traugott M, Grieb A, Pawelka E, Laferl H, Wenisch C (2020) Human recombinant soluble ACE2 in severe COVID-19. Lancet Respir Med 8:1154–1158. https://doi.org/10.1016/S2213-2600(20)30418-5
Author information
Authors and Affiliations
Contributions
Mohammad Rafi Khezri: design and writing manuscript and design figure. Donya Moloodsouri, Fereshteh Nazari Khanamiri, and Tooba Mohammadi: writing. Morteza Ghasemnejad-Berenji: revision of the manuscript. All authors read and approved the manuscript and all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Yes.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khezri, M.R., Nazari-Khanamiri, F., Mohammadi, T. et al. Potential effects of icariin, the Epimedium-derived bioactive compound in the treatment of COVID‐19: a hypothesis. Naunyn-Schmiedeberg's Arch Pharmacol 395, 1019–1027 (2022). https://doi.org/10.1007/s00210-022-02262-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-022-02262-y