Abstract
Numerous studies reveal that metabolism dysfunction contributes to the development of pathological cardiac hypertrophy. While the abnormal lipid and glucose utilization in cardiomyocytes responding to hypertrophic stimuli have been extensively studied, the alteration and implication of glutaminolysis are rarely discussed. In the present work, we provide the first evidence that glutamate dehydrogenase (GDH), an enzyme that catalyzes conversion of glutamate into ɑ-ketoglutarate (AKG), participates in isoprenaline (ISO)-induced cardiac hypertrophy through activating mammalian target of rapamycin (mTOR) signaling. The expression and activity of GDH were enhanced in cultured cardiomyocytes and rat hearts following ISO treatment. Overexpression of GDH, but not its enzymatically inactive mutant, provoked cardiac hypertrophy. In contrast, GDH knockdown could relieve ISO-triggered hypertrophic responses. The intracellular AKG level was elevated by ISO or GDH overexpression, which led to increased phosphorylation of mTOR and downstream effector ribosomal protein S6 kinase (S6K). Exogenous supplement of AKG also resulted in mTOR activation and cardiomyocyte hypertrophy. However, incubation with rapamycin, an mTOR inhibitor, attenuated hypertrophic responses in cardiomyocytes. Furthermore, GDH silencing protected rats from ISO-induced cardiac hypertrophy. These findings give a further insight into the role of GDH in cardiac hypertrophy and suggest it as a potential target for hypertrophy-related cardiomyopathy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pathological cardiac hypertrophy happens when the heart undergoes pressure or capacity overload and is typically manifested by enhanced protein synthesis, enlargement of cardiomyocyte size, and thickened ventricular walls. Although it initially acts as a compensatory response to maintain output, prolonged hypertrophy will impair cardiac function and eventually progress to heart failure (Tham et al., 2015). Since the heart is an organ of high energy consumption and must continually generate ATP at a rapid rate to sustain contractile, it is of importance to pay close attention to metabolism and energy production process in cardiac diseases including the development of hypertrophy (Lopaschuk et al., 2010). A variety of energy substrates, such as fatty acids, carbohydrates (glucose and lactate), ketone bodies, and amino acids, are known to contribute differently to meet the energy demand of the heart (Karwi et al., 2018). During recent years, it has been recognized that abnormal fatty acid metabolism and glucose utilization occur in hypertrophic cardiomyocytes and facilitate the onset of heart failure (Bernardo et al., 2010). However, the potential roles of amino acids metabolism, especially for glutamine, are rarely discussed.
Glutamine is the most abundant amino acid in human blood. Increased glutamine uptake elevates the cellular glutamate pool that is required for the production of non-essential amino acids, and thus supports protein synthesis (Karlstaedt et al., 2018). Glutamate dehydrogenase (GDH) is one of the key enzymes involved in glutaminolysis. It catalyzes the removal of hydrogen from glutamate to form α-ketoglutarate (AKG). GDH regulates not only amino acid metabolism and ATP production, but also ammonia elimination, insulin secretion, and neurotransmitter recycling, thereby playing important roles in cancers, hyperinsulinism/hyperammonemia syndrome, and neurodegenerative diseases (Mathioudakis et al., 2019; Drews et al., 2020). Nevertheless, the implication of GDH in cardiovascular system remains to be elucidated.
In mammalian cells, AKG, which is the product of GDH, enters into the tricarboxylic acid (TCA) cycle, providing carbon and nitrogen sources for growth (Fan et al., 2013). Besides, AKG has been recognized to participate in various epigenetic modifications. The change of AKG/succinate ratio affects histone H3 lysine 27 trimethylation (H3K27me3) and ten-eleven translocation (TET)-dependent DNA demethylation (Carey et al., 2015). AKG also plays a potential role in modulating signaling transduction. It has been proven that enhanced glutaminolysis or a cell-permeable AKG analog stimulates lysosomal translocation and activation of mammalian target of rapamycin (mTOR) cascade (Duran et al., 2012). These facts indicate that manipulation of GDH will exercise a profound influence on cell metabolism and function via affecting AKG production. In this study, we reveal that GDH promotes isoprenaline (ISO)-induced cardiac hypertrophy in an enzyme activity-dependent way. Its pro-hypertrophic effect can be attributed to increased AKG level and subsequent activation of mTOR signaling pathway. Therefore, GDH may present a potential new target for therapeutic intervention of hypertrophy-related cardiac abnormalities.
Materials and methods
Cell culture and transfection
Neonatal rat cardiomyocytes (NRCMs) were isolated from 1- to 3-day-old Sprague–Dawley (SD) rats as described previously (Yu et al., 2013). Cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM, Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (FBS, Gibco) and 0.1 mM 5-bromodeoxyuridine (BrdU, Thermo Fisher Scientific, Rockford, IL, USA) and maintained at 37 ℃ with 5% CO2. The H9c2 rat ventricular cell line was purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China) and cultured under the same conditions. For RNA interference and plasmid transfection, cells were transfected using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA) with reduced serum medium (Opti-MEM, Gibco) following the manufacturer’s instruction. Small interfering RNA (siRNA) against GDH (siGDH) and relevant negative control (NC) siRNA were obtained from GenePharma (Shanghai, China). Plasmid expressing wild-type GDH was purchased from Sangon Biotech (Shanghai, China). Plasmid expressing catalytically inactive mutant of GDH was constructed using Fast mutagenesis kit from TransGen Biotech (Beijing, China).
Animal models
All animal experiments were conducted following the Guide for the Care and Use of Laboratory Animals (NIH Publication No. 85–23, revised 1996) and were approved by the Research Ethics Committee of Sun Yat-Sen University. SD rats (male, 180–220 g, SPF grade, Certification No. 44008500025186) were provided by the Experimental Animal Center of Sun Yat-Sen University. The animals were housed in individually ventilated cages with 12-h daylight/dark cycles at 21–23 ℃ and fed with standard laboratory chow and water ad libitum. To downregulate GDH expression in vivo, the rats were intramyocardially injected with adeno-associated virus encoding GDH shRNA (AAV-shGDH) or negative control shRNA (AAV-shNC) purchased from Hanbio Biotechnology (Shanghai, China). The serotype of virus was AAV9 and the sequences of shGDH and shNC are listed in Supplementary Tab. S1. The schematic diagram of shGDH vector is shown in Supplementary Fig. S1. After 2 weeks, ISO (1.2 mg kg–1 day–1, Sigma-Aldrich, St. Louis, MO, USA) was administrated for 7 consecutive days by subcutaneous injection to induce cardiac hypertrophy.
Echocardiography and histological analysis
Rats were anaesthetized with 3% (v/v) isoflurane, and two-dimensional-guided M-mode echocardiography was performed with a Technos MPX ultrasound system (Esaote, Genoa, Italy) as described in our previous report (Zhou et al., 2006). Cardiac function indexes, including the heart rate (HR), ejection fraction (EF), fractional shortening (FS), and left ventricular posterior wall thicknesses at end-diastole and end-systole (LVPW-d and LVPW-s), were measured. Afterward, all animals were sacrificed. The hearts were quickly removed and weighed. Cardiac tissues were fixed in 4% paraformaldehyde, and then embedded in paraffin. For morphometric measurements, the histological cross-sections were stained respectively with hematoxylin–eosin (H&E), picric-Sirius red (PSR), and wheat germ agglutinin (WGA). The rest of the tissue samples were stored at − 80 ℃ for further detection.
RNA extraction and qRT-PCR
Total RNA was isolated from NRCMs or myocardial tissues by TRIzol Reagent (Takara Biotechnology, Dalian, China) according to the manufacturer’s instructions. After that, 5 μg of total RNA was used as template to synthesize cDNA with RevertAid First-Strand cDNA Synthesis Kit (Thermo Fisher Scientific). The expression levels of target mRNAs were determined using SYBR Green Realtime PCR Master Mix (Toyobo Life Science, Osaka, Japan) with LightCycler480 II (Roche, Basel, Switzerland). Rat-specific primers for GDH, atrial natriuretic factor (ANF), myosin heavy chain β (β-MHC), and β-Actin were provided by Sangon Biotech as listed in Supplementary Tab. S2. The relative expression level was determined by 2–ΔΔCt method and normalized to β-actin expression.
Western blot
Proteins in NRCMs or myocardial tissues were extracted with RIPA lysis buffer (Beyotime Biotechnology, Nantong, China) supplemented with protease inhibitors and phosphatase inhibitors on ice. Cleared cell lysates were obtained by centrifugation at 12,000 g for 15 min at 4 ℃. After quantification using BCA Protein Assay Kit (Thermo Fisher Scientific), 30 μg of the lysates was boiled for 5 min and submitted to Western blot analysis according to our previous report (Ding et al., 2021). Lysates were separated by 10% SDS-PAGE for the analysis of GDH, ANF, and β-MHC, and equal amounts of protein from the same sample were submitted to 8% SDS-PAGE for detection of p-S6K, S6K, p-mTOR, and mTOR. Proteins were transferred to PVDF membranes (Millipore, Billerica, MA, USA) and blocked with 5% skimmed milk at room temperature for 1 h. After washed and labeled, the membranes were cut into thin strips according to the positions of molecular weight markers and incubated with related primary antibodies at 4 ℃ for 16 h. Protein levels were detected with Image Quant LAS 4000 mini (GE Healthcare, Waukesha, WI, USA). The intensities of bands were quantified using Quantity One software (Bio-Rad, California, USA). Primary and secondary antibodies used for Western blot analysis are listed in Supplementary Tab. S3. GAPDH served as an internal reference.
Measurement of cell surface area
NRCMs seeded in 48-well plate were washed with PBS and fixed in 4% paraformaldehyde for 15 min. Cells were then permeabilized in 0.03% Triton X-100 for 15 min. After incubation with 0.1% rhodamine phalloidin (Invitrogen) for 1 h, cells were washed with PBS and further stained with 4′,6-diamidino-2-phenylindole (DAPI, Cell Signaling Technology, Danvers, MA, USA) for 10 min. Cell surface area was determined with High Content Screening System (Thermo Fisher Scientific), and 25 randomly selected fields were chosen for analysis by a built-in image analysis software.
Measurement of GDH activity
GDH activity was measured with a coupled enzyme assay kit (Sigma-Aldrich) according to the manufacturer’s instruction. NRCMs (1 × 106) or myocardial tissues (50 mg) were homogenized in 200 μL of ice-cold lysate buffer and centrifuged to remove insoluble materials. Glutamate was consumed by GDH to generate nicotinamide adenine dinucleotide (NADH), which subsequently reacted with a probe to generate a colorimetric product. The absorbance was measured at 450 nm. GDH activity was calculated as NADH amount divided by reaction time.
Determination of AKG level
NRCMs or myocardial tissues were homogenized in ice-cold lysate buffer and centrifuged to remove insoluble materials. AKG level was determined with a ɑ-ketoglutarate assay kit (Sigma-Aldrich) according to the manufacturer’s instruction. Briefly, the lysates were mixed with a reaction mixture and incubated for 30 min at 37 ℃. Fluorescence intensity (λex = 535 nm and λem = 587 nm) was measured, and AKG amount was calculated with parallel standard curve. Data were normalized to total protein content of the lysates.
Statistical analyses
The data are expressed as the mean ± standard error of the mean (SEM). Unpaired student’s t test was used to determine the differences between two groups. The statistical analyses among multiple groups were performed with one-way ANOVA with Bonferroni post hoc test. Statistical significance was defined as P < 0.05.
Results
GDH participates in ISO-induced cardiomyocyte hypertrophy
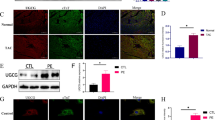
ISO, as a nonselective β-adrenergic receptor agonist, has been widely used to stimulate cardiac hypertrophy both in vitro and in vivo (Osadchii, 2007). In this study, we observed that the mRNA and protein levels of hypertrophic biomarkers ANF and β-MHC were elevated in a time-dependent manner following incubation with 10 μM ISO in cultured NRCMs (Fig. 1a, b). ISO treatment also led to the enlargement of cell surface area (Fig. 1c). Meanwhile, GDH expression was upregulated by ISO, indicating its possible involvement in the pathogenesis of hypertrophy. To verify this point, GDH was knocked down by transfection with siRNA in NRCMs. As shown in Fig. 1d–f, the increase in the expression of hypertrophic biomarkers and cell surface area could be obviously attenuated by GDH depletion. These evidences suggested that GDH might act as a contributing factor to ISO-provoked cardiomyocyte hypertrophy.
GDH expression is elevated by ISO, and knockdown of GDH attenuates ISO-induced cardiomyocyte hypertrophy. a–c NRCMs were treated with 10 μM ISO for the indicated time points. The expression of GDH, ANF and β-MHC was determined by qRT-PCR and Western blot respectively. The cell surface area was measured by rhodamine-phalloidin staining. d–f NRCMs were transfected with GDH siRNA (siGDH) or negative control (NC) for 48 h, and further stimulated by 10 μM ISO for 24 h. The levels of GDH and hypertrophic markers were detected, and the cell surface area was measured. Scale bar: 100 μm. The data are presented as the mean ± SEM. *P < 0.05; **P < 0.01 vs. the control (CON) or NC group. #P < 0.05; ##P < 0.01 vs. the NC + ISO group. n = 4
The pro-hypertrophic effect of GDH depends on its enzyme activity
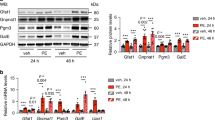
GDH is a key enzyme that catalyzes the oxidative deamination of glutamate. In NRCMs, ISO treatment (10 μM for 24 h) resulted in an obvious elevation of GDH activity (Fig. 2a). Furthermore, NRCMs were transfected with plasmids encoding wild-type GDH or its dehydrogenase-defective counterpart (GDH-R443S) that the arginine at 443 was replaced by serine (Fig. 2b). Overexpression of wild-type GDH, but not GDH-R443S, could increase GDH activity in NRCMs (Fig. 2c), which was in line with previous studies (Plaitakis et al., 2003; Mastorodemos et al., 2005). In addition, we found that only with enzyme activity could GDH induce significant hypertrophic responses, as manifested by increased mRNA and protein expression of ANF and β-MHC (Fig. 2d, e), as well as the enlargement of cell surface area (Fig. 2f).
Overexpression of GDH provokes hypertrophic responses in NRCMs. a GDH activity was measured in cell lysates of NRCMs following treatment with 10 μM ISO for 24 h. b Diagram of a catalytically inactive mutant of GDH with the arginine at 443 replaced by serine (GDH-R443S). c NRCMs were transfected with plasmids encoding wild-type GDH (GDH-WT) or GDH-R443S for 48 h. GDH enzyme activity measured. d, e The mRNA and protein levels of GDH and hypertrophic markers were determined. f The cell surface area of NRCMs was measured. Scale bar: 100 μm. The data are presented as the mean ± SEM. *P < 0.05; **P < 0.01 vs. the control (CON) or vector group. ns no statistical difference. n = 4
Elevation of AKG level provokes cardiomyocyte hypertrophy
AKG produced anaplerotically from glutamate by GDH is a rate-determining intermediate in TCA cycle and plays crucial roles in cellular energy metabolism (Wu et al., 2016). Besides, it is a potential biomarker for myocardial energy expenditure, associating with clinical severity and short-term outcome of chronic heart failure (Chen et al., 2014a). Therefore, the intracellular level of AKG was further measured. ISO incubation promoted AKG content in NRCMs, which could be attenuated by GDH knockdown (Fig. 3a). Overexpression of wild-type GDH also facilitated AKG production, while its inactive mutant GDH-R443S showed no influence (Fig. 3b). To validate the participation of AKG in the pathogenesis of cardiac hypertrophy, NRCMs were incubated with AKG for 24 h, and the hypertrophic responses were detected. Exogenous addition of AKG resulted in a concentration-dependent elevation of intracellular AKG level (Supplementary Fig. S2). The expression of ANF and β-MHC was upregulated along with increased concentrations of AKG (Fig. 3c, d). And AKG provoked the enlargement of cardiomyocytes (Fig. 3e). Moreover, AKG supplement could exacerbate the hypertrophic responses triggered by ISO (Fig. 3f–h). The above results indicated the involvement of AKG in ISO-induced cardiac hypertrophy.
Increased AKG production stimulates cardiomyocyte hypertrophy. a AKG level was measured in NRCMs with or without GDH siRNA (siGDH) transfection following ISO stimulation (10 μM for 24 h). b Wild-type GDH (GDH-WT) and its dehydrogenase-defective counterpart (GDH-R443S) were overexpressed in NRCMs. The intracellular AKG content was subsequently determined. c–e NRCMs were incubated with different concentrations of AKG for 24 h. The hypertrophic responses of cells were detected by monitoring the hypertrophic markers expression and cell surface area. f–h NRCMs were incubated with 80 μM AKG and 5 μM ISO for 24 h. The levels of hypertrophic markers and cell surface area were measured respectively. Scale bar: 100 μm. The data are presented as the mean ± SEM. *P < 0.05; **P < 0.01 vs. the control (CON) or vector group. ##P < 0.01 vs. the ISO group. ns no statistical difference. n = 4
GDH mediates cardiac hypertrophy via activation of mTOR signaling
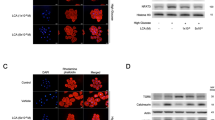
It has been well demonstrated that mTOR signaling cascade is responsible for enhanced protein synthesis and hypertrophic growth of cardiomyocytes (Wang and Proud, 2006). Excessive glutaminolysis or a cell-permeable AKG analog is reported to stimulate lysosomal translocation and activation of mTOR pathway (Duran et al., 2012). To explore the association of GDH-mediated hypertrophy with mTOR signaling, the phosphorylation states of mTOR and the downstream effector S6K were determined in NRCMs. Our data showed that ISO treatment led to elevated levels of phosphorylated mTOR and S6K, accompanying with increased ANF and β-MHC expression, indicating the activation of mTOR signaling during cardiomyocyte hypertrophy (Fig. 4a). Similar effects were observed following overexpression of wild-type GDH or supplement of AKG (Fig. 4b, c). In contrast, GDH knockdown attenuated ISO-induced mTOR pathway activation and increase in ANF and β-MHC levels (Fig. 4d). Moreover, treatment with rapamycin, an mTOR inhibitor, disrupted ISO or GDH overexpression–induced hypertrophic marker expression (Fig. 4e). Rapamycin also suppressed mTOR activation and hypertrophic responses stimulated by AKG (Fig. 4f). Additionally, AKG supplement ameliorated the inhibitive effects of GDH knockdown on hypertrophy and mTOR activation, which could be rescued by rapamycin (Fig. 4g). These facts indicated the participation of GDH in the development of cardiac hypertrophy, and the elevation of AKG level and subsequent activation of mTOR signaling might play an important role in this process.
The activation of mTOR signaling is involved in the pro-hypertrophic effects of GDH and AKG. a–c The phosphorylated states of mTOR and its downstream effector S6K were determined in NRCMs with ISO treatment, overexpression of wild-type GDH (GDH-WT) and inactive mutant (GDH-R443S), or AKG supplement respectively. d The effects of GDH siRNA (siGDH) transfection on ISO-induced mTOR and S6K phosphorylation were measured in NRCMs. e NRCMs were subjected to ISO stimulation or GDH overexpression in the presence of mTOR inhibitor rapamycin (Rapa, 100 nM). f NRCMs were incubated with indicated concentrations of AKG with or without rapamycin. g NRCMs were treated with ISO, siGDH, AKG and rapamycin. The levels of mTOR and S6K phosphorylation were measured, and the expression of hypertrophic markers was simultaneously detected. The data are presented as the mean ± SEM. *P < 0.05, **P < 0.01 vs. the control (CON) or non-treatment group. #P < 0.05, ##P < 0.01, vs. the ISO group. $P < 0.05, $$P < 0.01, vs. the GDH or AKG 160 μM group. ※P < 0.05, ※※P < 0.01, vs. the ISO + siGDH group. &&P < 0.01, vs. the ISO + siGDH + AKG group. n = 4
GDH knockdown protects rats from ISO-induced cardiac hypertrophy
To investigate the possibility that targeting GDH could be utilized to counteract cardiac hypertrophy in vivo, SD rats were submitted to intramyocardial injection with adeno-associated virus encoding GDH shRNA. After 2 weeks, ISO (1.2 mg kg–1 day–1) was administrated subcutaneously for 7 days. The hearts of animals received ISO were obviously enlarged (Fig. 5a). Histological examination by H&E and PSR staining revealed cardiomyocyte disarrangement and onset of myocardial fibrosis (Fig. 5b, c). Enlargement of cell surface area was visualized by staining with WGA, which can specifically bind to glycoprotein on the myocardial cell membrane (Fig. 5d). The heart weight-to-body weight (HW/BW) ratio was increased responding to ISO injection (Fig. 5f). Neither ISO nor GDH shRNA significantly affected the heart rate (Fig. 5g). Moreover, echocardiography indicated that the left ventricular posterior wall thickness, fractional shortening, and ejection fraction were elevated after ISO treatment (Fig. 5e, h–k). These ISO-triggered pathological abnormities were all relieved in rats receiving AAV-shGDH. The enzyme activity of GDH and AKG level in left ventricle cardiac tissues was increased by ISO, which could be attenuated following GDH depletion (Fig. 5l, m). GDH knockdown also reversed the elevation of ANF and β-MHC expression and activation of mTOR pathway (Fig. 5n, o). Taken together, our results suggested that GDH-AKG-mTOR axis contributed to cardiac hypertrophy, which might represent a potential therapeutic target.
Knockdown of GDH alleviates ISO-induced cardiac hypertrophy in rats. SD rats were intramyocardially injected with adeno-associated virus encoding GDH shGDH (AAV-shGDH) or negative control shRNA (AAV-shNC). The animals further received subcutaneous ISO infusion (1.2 mg kg–1 day–1) for 7 days. a–e The hypertrophic changes of rat hearts were observed by gross morphologic examination (scale bar: 5 mm), H&E staining (scale bar: 5 mm), PSR staining (scale bar: 5 mm), WGA staining (scale bar: 200 μm), and echocardiography. f The heart weight to body weight (HW/BW) ratio was calculated. g The heart rate was calculated. h–k Echocardiographic parameters including left ventricular posterior wall thickness at end-diastole (LVPW-d) and end-systole (LVPW-s), ejection fraction (EF), and fraction shortening (FS) were measured. l, m GDH enzyme activity and AKG level in left ventricle tissues were detected. n, o The levels of GDH, hypertrophic markers, phosphorylated mTOR and S6K were determined. The data are presented as mean ± SEM, **P < 0.01 vs. the AAV-shNC group. ##P < 0.01 vs. the AAV-shNC + ISO group. n = 6
Discussion
The hypertrophied and failing myocardium is characterized by abnormities in energy and substrate metabolism, including alterations in high-energy-phosphate content, mitochondrial function, and increased dependence on glucose for energy supply (van Bilsen et al., 2004). Cardiomyocytes have low ATP content and high rate of ATP hydrolysis. Responding to enhanced workload, the substrate utilization switches from fatty acid to glucose as a compensatory protective mechanism, since ATP generation from glucose requires less oxygen (Bernardo et al., 2010; Lopaschuk et al., 2010; Tham et al., 2015). Besides, the metabolisms of other substrates, such as amino acid, lactate, and ketone bodies, are also profoundly changed (Karwi et al., 2018). For example, suppression of branched-chain amino acid catabolic gene expression along with concomitant tissue accumulation of α-keto acids has been identified as a signature of metabolic reprogramming in mouse failing hearts (Sun et al., 2016). The alteration of the pyruvate-lactate axis by monocarboxylic acid transporter 4 interference is a fundamental and early feature of cardiac hypertrophy and failure (Cluntun et al., 2021). Moreover, the activation of glutaminolysis, which is an anaplerotic pathway incorporating AKG derived from glutamine into TCA cycle, occurs in pulmonary hypertension and ischemic heart (Piao et al., 2013; Watanabe et al., 2020). Thus, it has been proposed that the interference of metabolic processes may exhibit benefits in the treatment of cardiovascular diseases (Bernardo et al., 2010). In the present work, we revealed that GDH, a key enzyme of glutaminolysis, participated in ISO-induced pathological cardiac hypertrophy. It gives a further insight into the functions of GDH and may help to better understand the mechanisms of metabolic modulation underlying cardiac hypertrophy.
GDH conducts the conversion from glutamate to AKG, thereby participating in intracellular metabolic processes that facilitate cell growth and proliferation (Zdzisinska et al., 2017). Our data showed that the pro-hypertrophic effect of GDH was dependent on its enzyme activity. Overexpression of GDH-R443S, a catalytical inactive mutant (Plaitakis et al., 2003; Mastorodemos et al., 2005), could not induce cardiac hypertrophy. Glutamine, the enzymatic substrate of GDH, is the most abundant amino acid in the blood and the only amino acid extracted by myocardium in net amounts (Vanky et al., 2006). It not only provides energy for the survival of tumor cells, but also helps normal cells to maintain intracellular redox balance and promotes the formation of various biological macromolecules (Karlstaedt et al., 2018; Durante, 2019). Previous studies have demonstrated that glutamine can mitigate numerous risk factors for cardiovascular diseases, such as hypertension, hyperlipidemia, glucose intolerance, obesity, and diabetes. However, excessive shunting of glutamine to the TCA cycle, that is mediated by GDH, will precipitate aberrant angiogenic responses and the development of pulmonary arterial hypertension (Durante, 2019). The upregulation of GDH can be found in multiple cancer cell lines (Wang et al., 2009; Jin et al., 2015, 2018). Overactivation of GDH facilitates tumor growth, invasion, and migration, while pharmacological blockade of GDH restricts oncogenesis (Son et al., 2013; Yang et al., 2014). GDH is also significantly upregulated during hypoxia/reoxygenation (H/R) in renal tubular epithelial cells, and silencing GDH attenuates the effect of miR-30a-5p inhibitor on H/R-stimulated oxidative damage and apoptosis (He et al., 2020). The abnormal activated GDH results in an increase in ATP content and thus closure of KATP channel on the plasma membrane of β-cells, leading to the depolarization of the membrane potential, which finally drives the release of insulin (Stanley et al., 1998). Nevertheless, GDH is merely discussed in cardiovascular diseases and the association of GDH with cardiac hypertrophy remains to be elucidated. In our study, ISO stimulation resulted in cardiac hypertrophy, accompanying with significant upregulation of GDH expression and activity. Overexpression of GDH was sufficient to provoke hypertrophic responses in cardiomyocytes, while GDH knockdown could alleviate hypertrophy both in vivo and in vitro. These findings suggest that suppression of GDH may offer a new approach for the treatment of cardiac hypertrophy and relevant myocardium injuries. However, it is still necessary to further validate the pro-hypertrophic role of GDH overexpression in animals. And so far, little is known about the long-term effects of GDH inhibition, especially the influences on cardiovascular system. Considering the important role of GDH in glutaminolysis and energy metabolism, close attention should be paid to the potential benefits and risks underlying chronic suppression of GDH.
As the product of GDH, AKG guides the carbon skeleton into the TCA cycle and changes the intracellular metabolism pattern (Carobbio et al., 2009; Karaca et al., 2018; Luczkowska et al., 2020). In the TCA cycle, AKG originates from isocitrate by oxidative decarboxylation catalyzed by isocitrate dehydrogenase and is further converted by AKG dehydrogenase to succinyl-CoA and NADH (Zdzisinska et al., 2017). Incorporation of AKG into the TCA cycle replenishes the metabolic intermediates to produce ATP and supports the biosynthetic demands for nicotinamide adenine dinucleotide phosphate (NADP+) during rapid cell growth (Piao et al., 2013). Previous reports have revealed that AKG possesses various beneficial effects. Dietary AKG supplement promotes a longer, healthier life in mice, associating with decreased levels of systemic inflammatory cytokines (Asadi Shahmirzadi et al., 2020). It also stabilizes immune system homeostasis, thereby extending lifespan and delaying age-related diseases (Wu et al., 2016). Moreover, AKG increases the bone mass of aged rodents, attenuates senescence-associated bone loss, and accelerates bone regeneration (Wang et al., 2020). Administration of AKG increases amino acids pool that is necessary for synthesis of type I collagen, and thus has a positive role in bone quality (Zdzisinska et al., 2017). In addition, AKG can exert anti-cancer activity. It inhibits HIF-1α binding activity in hepatoma, resulting in downregulation of VEGF expression and suppression of angiogenesis (Matsumoto et al., 2006). AKG also alleviates proliferation of colon adenocarcinoma cells via influencing the expression of cell cycle genes (Rzeski et al., 2012). Nevertheless, the influence of AKG on oncogenesis is still controversial. The demand for AKG in tumor cells is well known, and maintaining the homeostasis of AKG may be necessary for tumor cells to sustain rapid proliferation and to survive in adverse environments (Jin et al., 2015). Currently, the information on the potential roles of AKG in cardiovascular system is relatively limited. In vascular endothelial cells, AKG addition promotes cell proliferation after glutamine deprivation (Kim et al., 2017). AKG is suggested to be a biomarker of chronic heart failure, since its level is elevated in the failing hearts of patients and positive relevant to myocardial energy expenditure (Dunn et al., 2007; Chen et al., 2014a). In ISO-treated NRCMs and rat heart tissues, we observed significant upregulation of GDH expression and subsequent increase in intracellular AKG level. Forced overexpression of wild-type GDH, but not its mutant counterpart GDH-R443S, could elevate the intracellular level of AKG. Furthermore, exogenous addition of AKG led to increased expression of hypertrophic markers and cell surface area. These evidences suggest that the enhanced AKG production responding to ISO stimulation and GDH activation may contribute to the hypertrophic growth of cardiomyocytes.
More than functioning as an energy donor, AKG also regulates epigenetic processes and cellular signaling as an obligatory co-substrate and a signaling molecule (Zdzisinska et al., 2017). AKG is essential for AKG-dependent dioxygenases to catalyze hydroxylation reactions and modulate substrates including proteins, nucleic acids, lipids, and metabolic intermediates, such as the hypoxia-inducible factor (HIF) prolyl hydroxylases, which helps to mediate the degradation of HIF (Zdzisinska et al., 2017). Additionally, AKG affects the functioning of ten–eleven translocation hydroxylases (TETs) that involved in DNA demethylation and influences epigenetic modifications of chromatin (Yang et al., 2016). AKG is also reported to be an activator of mTOR pathway. In tumor cells, AKG analog stimulates GTP loading of RagB, thereby mediating lysosomal translocation and subsequent mTOR activation to promote cell size enlargement and autophagy inhibition (Duran et al., 2012). Numerous studies have demonstrated that mTOR plays a central role in regulating cell metabolism, growth, and survival. mTOR promotes anabolic processes such as protein, nucleotide, and lipid synthesis, whereas it inhibits catabolic pathways, such as autophagy so that contributes to development of adaptive cardiac hypertrophy in response to hypertrophic stimuli (Sciarretta et al., 2018). S6K is one of the earliest identified and best characterized substrates of mTOR. It can be directly phosphorylated and activated by mTOR to promote translation (Sciarretta et al., 2018). Our studies revealed that ISO treatment led to increased phosphorylation of mTOR and S6K, which was in line with previous reports showing the mobilization of mTOR/S6K cascade by β-adrenergic stimulation (Gao et al., 2017; Morishige et al., 2019; Wang et al., 2019). More importantly, we found that both GDH overexpression and AKG addition provoked mTOR signaling, while GDH knockdown alleviated ISO-induced mTOR activation, indicating the involvement of mTOR pathway in GDH-mediated cardiac hypertrophy. To further confirm this point, NRCMs were treated with rapamycin, a well-established inhibitor of mTOR. Rapamycin forms a complex with the immunophilin FK-506-binding protein 12 which then restricts mTOR activity (Yang et al., 2013). The suppression of mTOR pathway by rapamycin has been proved to attenuate pathological hypertrophy and reverse cardiac dysfunction induced by pressure overload or β-adrenergic receptor stimulation (Colombo et al., 2003; McMullen et al., 2004; Hasumi et al., 2014). In ISO-induced cardiac hypertrophic cardiomyocytes, rapamycin helps to preserve structure integrity and function of mitochondria, block proinflammatory responses, and contribute to the maintenance of energy homeostasis (Chen et al., 2014b). Our results showed that rapamycin treatment ameliorated mTOR activation and cardiomyocyte hypertrophy induced by ISO or GDH overexpression. In addition, the inhibitive effects of GDH knockdown on hypertrophy and mTOR activation were compromised at the presence of AKG, which could still be rescued by rapamycin. Thus, it may be speculated that the modulation of mTOR signaling via GDH-mediated AKG production is involved in the pathogenesis of cardiac hypertrophy.
In summary, our present study revealed the involvement of GDH in the development of ISO-induced cardiac hypertrophy. Excessive GDH expression increased intracellular level of AKG, which further activated mTOR signaling and contributed to cardiac hypertrophy (Fig. 6). The protective effects of GDH knockdown suggest a therapeutic potential for cardiac hypertrophy and heart failure by targeting GDH.
Schematic of the role of GDH in ISO-induced cardiac hypertrophy. In response to ISO stimulation, the expression and activity of GDH are elevated. GDH increases the intracellular levels of AKG, which further activates mTOR signaling, leading to the development of cardiac hypertrophy. The pro-hypertrophic effect of GDH can be alleviated by mTOR inhibitor rapamycin
Data availability
The data and materials used during the current study are available from the corresponding author on reasonable request.
References
Asadi Shahmirzadi A, Edgar D, Liao CY, Hsu YM, Lucanic M, Asadi Shahmirzadi A, Wiley CD, Gan G, Kim DE, Kasler HG, Kuehnemann C, Kaplowitz B, Bhaumik D, Riley RR, Kennedy BK, Lithgow GJ (2020) Alpha-ketoglutarate, an endogenous metabolite, extends lifespan and compresses morbidity in aging mice. Cell Metab 32:447-456 e446
Bernardo BC, Weeks KL, Pretorius L, McMullen JR (2010) Molecular distinction between physiological and pathological cardiac hypertrophy: experimental findings and therapeutic strategies. Pharmacol Ther 128:191–227
Carey BW, Finley LW, Cross JR, Allis CD, Thompson CB (2015) Intracellular alpha-ketoglutarate maintains the pluripotency of embryonic stem cells. Nature 518:413–416
Carobbio S, Frigerio F, Rubi B, Vetterli L, Bloksgaard M, Gjinovci A, Pournourmohammadi S, Herrera PL, Reith W, Mandrup S, Maechler P (2009) Deletion of glutamate dehydrogenase in beta-cells abolishes part of the insulin secretory response not required for glucose homeostasis. J Biol Chem 284:921–929
Chen PA, Xu ZH, Huang YL, Luo Y, Zhu DJ, Wang P, Du ZY, Yang Y, Wu DH, Lai WY, Ren H, Xu DL (2014a) Increased serum 2-oxoglutarate associated with high myocardial energy expenditure and poor prognosis in chronic heart failure patients. Biochim Biophys Acta 1842:2120–2125
Chen X, Zeng S, Zou J, Chen Y, Yue Z, Gao Y, Zhang L, Cao W, Liu P (2014b) Rapamycin attenuated cardiac hypertrophy induced by isoproterenol and maintained energy homeostasis via inhibiting NF-kappaB activation. Mediators Inflamm 2014:868753
Cluntun AA, Badolia R, Lettlova S, Parnell KM, Shankar TS, Diakos NA, Olson KA, Taleb I, Tatum SM, Berg JA, Cunningham CN, Van Ry T, Bott AJ, Krokidi AT, Fogarty S, Skedros S, Swiatek WI, Yu X, Luo B, Merx S, Navankasattusas S, Cox JE, Ducker GS, Holland WL, McKellar SH, Rutter J, Drakos SG (2021) The pyruvate-lactate axis modulates cardiac hypertrophy and heart failure. Cell Metab 33:629-648 e610
Colombo F, Gosselin H, El-Helou V, Calderone A (2003) Beta-adrenergic receptor-mediated DNA synthesis in neonatal rat cardiac fibroblasts proceeds via a phosphatidylinositol 3-kinase dependent pathway refractory to the antiproliferative action of cyclic AMP. J Cell Physiol 195:322–330
Ding YQ, Zhang YH, Lu J, Li B, Yu WJ, Yue ZB, Hu YH, Wang PX, Li JY, Cai SD, Ye JT, Liu PQ (2021) MicroRNA-214 contributes to Ang II-induced cardiac hypertrophy by targeting SIRT3 to provoke mitochondrial malfunction. Acta Pharmacol Sin 42:1422–1436
Drews L, Zimmermann M, Westhoff P, Brilhaus D, Poss RE, Bergmann L, Wiek C, Brenneisen P, Piekorz RP, Mettler-Altmann T, Weber APM, Reichert AS (2020) Ammonia inhibits energy metabolism in astrocytes in a rapid and glutamate dehydrogenase 2-dependent manner. Dis Model Mech 13:047134
Dunn WB, Broadhurst DI, Deepak SM, Buch MH, McDowell G, Spasic I, Ellis DI, Brooks N, Kell DB, Neyses L (2007) Serum metabolomics reveals many novel metabolic markers of heart failure, including pseudouridine and 2-oxoglutarate. Metabolomics 3:413–426
Duran RV, Oppliger W, Robitaille AM, Heiserich L, Skendaj R, Gottlieb E, Hall MN (2012) Glutaminolysis activates Rag-mTORC1 signaling. Mol Cell 47:349–358
Durante W (2019) The emerging role of l-glutamine in cardiovascular health and disease. Nutrients 11:2092
Fan J, Kamphorst JJ, Mathew R, Chung MK, White E, Shlomi T, Rabinowitz JD (2013) Glutamine-driven oxidative phosphorylation is a major ATP source in transformed mammalian cells in both normoxia and hypoxia. Mol Syst Biol 9:712
Gao L, Guo Y, Liu X, Shang D, Du Y (2017) KLF15 protects against isoproterenol-induced cardiac hypertrophy via regulation of cell death and inhibition of Akt/mTOR signaling. Biochem Biophys Res Commun 487:22–27
Hasumi Y, Baba M, Hasumi H, Huang Y, Lang M, Reindorf R, Oh HB, Sciarretta S, Nagashima K, Haines DC, Schneider MD, Adelstein RS, Schmidt LS, Sadoshima J, Marston Linehan W (2014) Folliculin (Flcn) inactivation leads to murine cardiac hypertrophy through mTORC1 deregulation. Hum Mol Genet 23:5706–5719
He Y, Lang X, Cheng D, Zhang T, Yang Z, Xiong R (2020) miR30a5p inhibits hypoxia/reoxygenationinduced oxidative stress and apoptosis in HK2 renal tubular epithelial cells by targeting glutamate dehydrogenase 1 (GLUD1). Oncol Rep 44:1539–1549
Jin L, Chun J, Pan C, Kumar A, Zhang G, Ha Y, Li D, Alesi GN, Kang Y, Zhou L, Yu WM, Magliocca KR, Khuri FR, Qu CK, Metallo C, Owonikoko TK, Kang S (2018) The PLAG1-GDH1 axis promotes Anoikis resistance and tumor metastasis through CamKK2-AMPK signaling in LKB1-deficient lung cancer. Mol Cell 69:87-99 e87
Jin L, Li D, Alesi GN, Fan J, Kang HB, Lu Z, Boggon TJ, Jin P, Yi H, Wright ER, Duong D, Seyfried NT, Egnatchik R, DeBerardinis RJ, Magliocca KR, He C, Arellano ML, Khoury HJ, Shin DM, Khuri FR, Kang S (2015) Glutamate dehydrogenase 1 signals through antioxidant glutathione peroxidase 1 to regulate redox homeostasis and tumor growth. Cancer Cell 27:257–270
Karaca M, Martin-Levilain J, Grimaldi M, Li L, Dizin E, Emre Y, Maechler P (2018) Liver glutamate dehydrogenase controls whole-body energy partitioning through amino acid-derived gluconeogenesis and ammonia homeostasis. Diabetes 67:1949–1961
Karlstaedt A, Schiffer W, Taegtmeyer H (2018) Actionable metabolic pathways in heart failure and cancer-lessons from cancer cell metabolism. Front Cardiovasc Med 5:71
Karwi QG, Uddin GM, Ho KL, Lopaschuk GD (2018) Loss of metabolic flexibility in the failing heart. Front Cardiovasc Med 5:68
Kim B, Li J, Jang C, Arany Z (2017) Glutamine fuels proliferation but not migration of endothelial cells. EMBO J 36:2321–2333
Lopaschuk GD, Ussher JR, Folmes CD, Jaswal JS, Stanley WC (2010) Myocardial fatty acid metabolism in health and disease. Physiol Rev 90:207–258
Luczkowska K, Stekelenburg C, Sloan-Bena F, Ranza E, Gastaldi G, Schwitzgebel V, Maechler P (2020) Hyperinsulinism associated with GLUD1 mutation: allosteric regulation and functional characterization of p.G446V glutamate dehydrogenase. Hum Genomics 14:9
Mastorodemos V, Zaganas I, Spanaki C, Bessa M, Plaitakis A (2005) Molecular basis of human glutamate dehydrogenase regulation under changing energy demands. J Neurosci Res 79:65–73
Mathioudakis L, Bourbouli M, Daklada E, Kargatzi S, Michaelidou K, Zaganas I (2019) Localization of human glutamate dehydrogenases provides insights into their metabolic role and their involvement in disease processes. Neurochem Res 44:170–187
Matsumoto K, Imagawa S, Obara N, Suzuki N, Takahashi S, Nagasawa T, Yamamoto M (2006) 2-Oxoglutarate downregulates expression of vascular endothelial growth factor and erythropoietin through decreasing hypoxia-inducible factor-1alpha and inhibits angiogenesis. J Cell Physiol 209:333–340
McMullen JR, Sherwood MC, Tarnavski O, Zhang L, Dorfman AL, Shioi T, Izumo S (2004) Inhibition of mTOR signaling with rapamycin regresses established cardiac hypertrophy induced by pressure overload. Circulation 109:3050–3055
Morishige S, Takahashi-Yanaga F, Ishikane S, Arioka M, Igawa K, Kuroo A, Tomooka K, Shiose A, Sasaguri T (2019) 2,5-Dimethylcelecoxib prevents isoprenaline-induced cardiomyocyte hypertrophy and cardiac fibroblast activation by inhibiting Akt-mediated GSK-3 phosphorylation. Biochem Pharmacol 168:82–90
Osadchii OE (2007) Cardiac hypertrophy induced by sustained beta-adrenoreceptor activation: pathophysiological aspects. Heart Fail Rev 12:66–86
Piao L, Fang YH, Parikh K, Ryan JJ, Toth PT, Archer SL (2013) Cardiac glutaminolysis: a maladaptive cancer metabolism pathway in the right ventricle in pulmonary hypertension. J Mol Med (berl) 91:1185–1197
Plaitakis A, Spanaki C, Mastorodemos V, Zaganas I (2003) Study of structure–function relationships in human glutamate dehydrogenases reveals novel molecular mechanisms for the regulation of the nerve tissue-specific (GLUD2) isoenzyme. Neurochem Int 43:401–410
Rzeski W, Walczak K, Juszczak M, Langner E, Pozarowski P, Kandefer-Szerszen M, Pierzynowski SG (2012) Alpha-ketoglutarate (AKG) inhibits proliferation of colon adenocarcinoma cells in normoxic conditions. Scand J Gastroenterol 47:565–571
Sciarretta S, Forte M, Frati G, Sadoshima J (2018) New insights into the role of mTOR signaling in the cardiovascular system. Circ Res 122:489–505
Son J, Lyssiotis CA, Ying H, Wang X, Hua S, Ligorio M, Perera RM, Ferrone CR, Mullarky E, Shyh-Chang N, Kang Y, Fleming JB, Bardeesy N, Asara JM, Haigis MC, DePinho RA, Cantley LC, Kimmelman AC (2013) Glutamine supports pancreatic cancer growth through a KRAS-regulated metabolic pathway. Nature 496:101–105
Stanley CA, Lieu YK, Hsu BY, Burlina AB, Greenberg CR, Hopwood NJ, Perlman K, Rich BH, Zammarchi E, Poncz M (1998) Hyperinsulinism and hyperammonemia in infants with regulatory mutations of the glutamate dehydrogenase gene. N Engl J Med 338:1352–1357
Sun H, Olson KC, Gao C, Prosdocimo DA, Zhou M, Wang Z, Jeyaraj D, Youn JY, Ren S, Liu Y, Rau CD, Shah S, Ilkayeva O, Gui WJ, William NS, Wynn RM, Newgard CB, Cai H, Xiao X, Chuang DT, Schulze PC, Lynch C, Jain MK, Wang Y (2016) Catabolic defect of branched-chain amino acids promotes heart failure. Circulation 133:2038–2049
Tham YK, Bernardo BC, Ooi JY, Weeks KL, McMullen JR (2015) Pathophysiology of cardiac hypertrophy and heart failure: signaling pathways and novel therapeutic targets. Arch Toxicol 89:1401–1438
van Bilsen M, Smeets PJ, Gilde AJ, van der Vusse GJ (2004) Metabolic remodelling of the failing heart: the cardiac burn-out syndrome? Cardiovasc Res 61:218–226
Vanky FB, Hakanson E, Jorfeldt L, Svedjeholm R (2006) Does glutamate influence myocardial and peripheral tissue metabolism after aortic valve replacement for aortic stenosis? Clin Nutr 25:913–922
Wang JW, Peng SY, Li JT, Wang Y, Zhang ZP, Cheng Y, Cheng DQ, Weng WH, Wu XS, Fei XZ, Quan ZW, Li JY, Li SG, Liu YB (2009) Identification of metastasis-associated proteins involved in gallbladder carcinoma metastasis by proteomic analysis and functional exploration of chloride intracellular channel 1. Cancer Lett 281:71–81
Wang L, Yuan D, Zheng J, Wu X, Wang J, Liu X, He Y, Zhang C, Liu C, Wang T, Zhou Z (2019) Chikusetsu saponin IVa attenuates isoprenaline-induced myocardial fibrosis in mice through activation autophagy mediated by AMPK/mTOR/ULK1 signaling. Phytomedicine 58:152764
Wang X, Proud CG (2006) The mTOR pathway in the control of protein synthesis. Physiology (Bethesda) 21:362–369
Wang Y, Deng P, Liu Y, Wu Y, Chen Y, Guo Y, Zhang S, Zheng X, Zhou L, Liu W, Li Q, Lin W, Qi X, Ou G, Wang C, Yuan Q (2020) Alpha-ketoglutarate ameliorates age-related osteoporosis via regulating histone methylations. Nat Commun 11:5596
Watanabe K, Nagao M, Toh R, Irino Y, Shinohara M, Iino T, Yoshikawa S, Tanaka H, Satomi-Kobayashi S, Ishida T, Hirata KI (2020) Critical role of glutamine metabolism in cardiomyocytes under oxidative stress. Biochem Biophys Res Commun 534:687–693
Wu N, Yang M, Gaur U, Xu H, Yao Y, Li D (2016) Alpha-ketoglutarate: physiological functions and applications. Biomol Ther (Seoul) 24:1–8
Yang C, Ko B, Hensley CT, Jiang L, Wasti AT, Kim J, Sudderth J, Calvaruso MA, Lumata L, Mitsche M, Rutter J, Merritt ME, DeBerardinis RJ (2014) Glutamine oxidation maintains the TCA cycle and cell survival during impaired mitochondrial pyruvate transport. Mol Cell 56:414–424
Yang H, Rudge DG, Koos JD, Vaidialingam B, Yang HJ, Pavletich NP (2013) mTOR kinase structure, mechanism and regulation. Nature 497:217–223
Yang Q, Liang X, Sun X, Zhang L, Fu X, Rogers CJ, Berim A, Zhang S, Wang S, Wang B, Foretz M, Viollet B, Gang DR, Rodgers BD, Zhu MJ, Du M (2016) AMPK/alpha-ketoglutarate axis dynamically mediates DNA demethylation in the prdm16 promoter and brown adipogenesis. Cell Metab 24:542–554
Yu SS, Cai Y, Ye JT, Pi RB, Chen SR, Liu PQ, Shen XY, Ji Y (2013) Sirtuin 6 protects cardiomyocytes from hypertrophy in vitro via inhibition of NF-kappaB-dependent transcriptional activity. Br J Pharmacol 168:117–128
Zdzisinska B, Zurek A, Kandefer-Szerszen M (2017) Alpha-ketoglutarate as a molecule with pleiotropic activity: well-known and novel possibilities of therapeutic use. Arch Immunol Ther Exp (Warsz) 65:21–36
Zhou SG, Zhou SF, Huang HQ, Chen JW, Huang M, Liu PQ (2006) Proteomic analysis of hypertrophied myocardial protein patterns in renovascularly hypertensive and spontaneously hypertensive rats. J Proteome Res 5:2901–2908
Funding
This research was supported by grants from the National Natural Science Foundation of China (82070268), the Local Innovative and Research Teams Project of Guangdong Pearl River Talents Program (2017BT01Y093), the National Engineering and Technology Research Center for New Drug Druggability Evaluation (Seed Program of Guangdong Province, 2017B090903004), and the Guangdong Basic and Applied Basic Research Foundation (2020A1515011512).
Author information
Authors and Affiliations
Contributions
Liu PQ and Ye JT conceived and designed the research. Lin ZR conducted experiments. Li ZZ, Cao YJ, and Yu WJ contributed to new reagents or analytical tools. Lin ZR analyzed data. Lin ZR wrote the manuscript. All the authors read and approved the manuscript, and all data were generated in-house and that no paper mill was used.
Corresponding authors
Ethics declarations
Ethics approval
The animal experiments protocol has been reviewed and approved by the Institutional Animal Care and Use Committee (IACUC), Sun Yat-Sen University (Approval No. SYSU-IACUC-2021–000282).
Consent to participate
All the authors consent for participation.
Consent for publication
All the authors consent for publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1
(DOCX 154 KB)
Supplementary file2
(XLSX 12 KB)
Supplementary file3
(XLSX 35.5 MB)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, ZR., Li, ZZ., Cao, YJ. et al. GDH promotes isoprenaline-induced cardiac hypertrophy by activating mTOR signaling via elevation of α-ketoglutarate level. Naunyn-Schmiedeberg's Arch Pharmacol 395, 1373–1385 (2022). https://doi.org/10.1007/s00210-022-02252-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-022-02252-0