Abstract
Matrix metalloproteinases (MMPs) are a group of endopeptidases that degrade the extracellular matrix and are responsible for many physiological and pathological processes. We aim to review the MMP inhibition from a clinical perspective and its possible therapeutic use in the future. MMPs play a role in various neurodegenerative and cerebrovascular diseases such as large artery atherosclerosis and ischemic stroke; for example, MMPs increase blood–brain barrier permeability favoring neuroinflammation. Synthetic MMPs inhibitors have been tested mostly in oncological trials and failed to demonstrate efficacy; some of them were discontinued because of the severe adverse reactions. Tetracyclines, in submicrobial doses, act as an MMP inhibitor, although tetracyclines have not yet been proven effective in several neurological conditions in which they were tested against placebo; it is uncertain whether there may be a use for tetracyclines in cerebrovascular disease, as a neuroprotective agent or in dolichoectasia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Matrix metalloproteinases (MMPs) are zinc-dependent endopeptidases known for their capacity to proteolyze elements of the extracellular matrix (ECM), such as collagen, proteoglycans, laminin, elastin, and fibronectin (Allan et al. 1995). ECM comprises up to 20% of the brain volume (Nicholson and Rice 1986) and is an essential part of the central nervous system (CNS) (Rosenberg 2017). There are three principal compartments of the brain ECM: the basal lamina, the perineuronal nets, and the neural interstitial matrix (Lau et al. 2013). The basal lamina acts as a boundary between endothelial cells and CNS parenchyma (Lau et al. 2013) and is one of the major sites of action of the MMPs (Rosenberg 2017). The MMPs also act at the cell surface, activating growth factors, death receptors, and other signaling molecules (Rosenberg 2009).
There are at least 26 MMPs (Adhikari et al. 2017) divided into collagenases, gelatinases, stromelysins, and membrane-type MMPs (Rosenberg 2009). Collagenases degrade collagen, the main component of bone and cartilage, gelatinases degrade molecules in the basal lamina around capillaries to facilitate angiogenesis and neurogenesis, and stromelysins (MMP-3, MMP-10, MMP-11, and MMP-7) degrade components of the ECM. Membrane-type MMPs play a role in the activation of other proteases and growth factors (Rosenberg 2009). Matrilysin (MMP-7) degrades ECM elements such as collagen and laminin (Fanjul-Fernández et al. 2010).
MMPs are regulated at several levels, and one of these includes secreted proteins called tissue inhibitors of metalloproteinases (TIMPs) (Mastroianni and Liuzzi 2007). TIMPs are small glycoproteins that bind to MMPs, forming non-covalent complexes. These complexes inhibit the activity of the MMPs (Mastroianni and Liuzzi 2007; Castro et al. 2011). Four TIMPs have been described (TIMP-1, TIMP-2, TIMP-3, and TIMP-4). TIMPs are not specific, but they show some preference; i.e., TIMP-1 mainly inhibits MMP-9, whereas TIMP-2 inhibits MMP-2, etc. (Mastroianni and Liuzzi 2007; Castro et al. 2011; Rosenberg 2009). Under physiological conditions, there is a balance between MMPs and TIMPs. Inappropriate production of MMPs and/or TIMPs may lead to pathological conditions such as inflammation, disrupt wound healing, or promote invasion of cancer cells or contiguous spreading of infectious diseases (Mastroianni and Liuzzi 2007; Gutierrez et al. 2016). While MMPs are present in pathological processes such as neuroinflammation, they also play an important role in angiogenesis, bone growth, wound healing, and tissue regeneration (Mignatti and Rifkin 1996; Jabłońska-Trypuć et al. 2016).
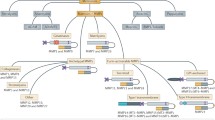
The MMPs that most relate to brain processes are MMP-2, MMP-3, MMP-9, MMP-10, and MMP-14 (Rosenberg 2009). In the CNS, MMPs are absent or present in undetectable concentrations (Behl et al. 2021). MMP-2 is also present in healthy brain and in cerebrospinal fluid (Rosenberg 2009). MMP-3 is essential for neuroplasticity and neuroregeneration (Lech et al. 2019).
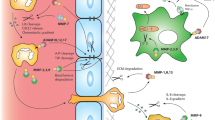
In pathological conditions, MMPs may cause increased blood–brain barrier (BBB) permeability by acting on the basal lamina and tight junctions in endothelial cells, resulting in the final common pathway of acute neuroinflammatory damage (Rosenberg 2009, 2017). MMPs are also involved in neuronal death, hypoxia/ischemia lesions, and demyelinization (Behl et al. 2021; Rosenberg 2009) (Table 1).
In hypoxic-ischemic lesions, MMP-2, MMP-3, and MMP-9 increase the permeability of the blood–brain barrier with a consequent greater risk of hemorrhagic transformation (Rosenberg 2009).
Synthetic MMPs inhibitors have been created, and their study has been primarily in oncological clinical studies. However, all showed neutral or even negative results. One of the biggest problems and concerns is adverse reactions, as MMPs are also essential for cell function.
In addition to their antimicrobial effect, tetracyclines are potent inhibitors of MMPs due to their antiapoptotic activity, among other functions (Griffin et al. 2010; Jabłońska-Trypuć et al. 2016).
Methodology
We performed a search in PubMed with the following MeSH terms: “Matrix Metalloproteinase Inhibitors,” “stroke,” “dementia,” “intracranial aneurysm,” “aortic aneurysm,” “multiple sclerosis,” “clinical study,” “randomized controlled trial,” and “humans.”
Pharmacology
During the last decades, pharmaceutical companies have developed several synthetic MMP inhibitors (Tables 2 and 3). Due to the lack of efficacy or serious adverse effects, these drugs have been withdrawn in phase II or III studies. Only doxycycline has FDA approval for periodontitis.
Minocycline, a second-generation tetracycline, readily crosses the blood–brain barrier (Macdonald et al. 1973) and has neuroinflammatory and antiapoptotic actions, decreasing microglial activity and inhibiting the activity of MMPs so that it could play a role in the prevention of cell death (Stirling et al. 2005; Cankaya et al. 2019).
Because of the ability of tetracyclines to inhibit collagenolysis and angiogenesis, they have been used in an attempt to correct excessive collagen breakdown and possibly impact favorably early stages of the inflammatory cascade (Rosenberg 2009). Nonetheless, the use of MMP inhibitors has been associated with adverse side effects, and there is a theoretical concern that broad-spectrum MMP inhibitors that may block important functions such as remodeling the ECM may cause joint stiffness that limits their use (Rosenberg 2009). Photosensitivity and hyperpigmentation have also been described with broad-spectrum MMP inhibitors (Nakasujja et al. 2013; Frenzel et al. 2008). In a study comparing minocycline vs placebo in patients with HIV-associated cognitive impairment, 4% of the patients taking minocycline developed grade 2 hyperpigmentation, and 5% of the patients developed a grade 4 rash (Nakasujja et al. 2013). Furthermore, in a study with minocycline in AD, of those randomized to 400 mg or 200 mg minocycline daily, only 29% and 72% of participants completed the study, respectively. The reasons for discontinuing the treatment were dizziness and gastrointestinal and dermatologic adverse effects (Howard et al. 2020).
Cerebrovascular disease and related disorders
Acute ischemic stroke
MMP activation leads to BBB disruption (Rosenberg 2009). Some meta-analyses have shown that MMP-9 (− 1562 C/T) gene polymorphism is associated with the risk of acute ischemic stroke (AIS) (Misra et al. 2018; He et al. 2017), as well as MMP-12 (− 1082 A/G) gene polymorphism.
This association between MMP-9 (1562 C/T) gene polymorphism seems to be more marked in the Asian population (Jiang et al. 2020; Misra et al. 2018).
There is evidence too that that serum levels of MMP-8 and myeloperoxidase (MPO acts as an activator of MMP-8(Saari et al. 1990)) are higher in patients with AIS than in controls (Palm et al. 2018). Furthermore, MMP-8 serum levels are lower in strokes due to small vessel disease compared to strokes due to cardioembolism or large vessel disease, although this difference may relate to variations in stroke size (Palm et al. 2018).
Because MMPs disrupt the BBB, MMP inhibition has been investigated as a therapeutic intervention in AIS. In rats with occlusion of the middle cerebral artery, treatment with minocycline showed a significant decrease in the expression of MMP-2 and MMP-9 in the brain compared to controls. Importantly, this study also demonstrated that the MMP inhibitory effect of minocycline did not affect the fibrinolytic activity of intravenous tissue plasminogen activator (tPA) (Machado et al. 2009). However, MMP inhibition with BB-1101 (a broad-spectrum MMP inhibitor) did not reduce the infarct size in rats after 48 h, and in fact, it led to neurological deterioration (Sood et al. 2008).
Three RCTs compared minocycline 200 mg versus placebo for 5 days orally administrated in patients with an AIS with onset between 6 and 24 h. The three studies showed a lower National Institutes of Health Stroke Scale (NIHSS) at 90 days after randomization (Padma Srivastava et al. 2012; Lampl et al. 2007; Amiri-Nikpour et al. 2015). Additionally, in two of these studies, the Barthel Index was higher, and the modified Rankin Score (mRS) was lower in the minocycline group (Padma Srivastava et al. 2012; Lampl et al. 2007). No differences were seen in hemorrhagic transformation, recurrent stroke, or myocardial infarct (Padma Srivastava et al. 2012; Lampl et al. 2007). Interestingly, a subgroup analysis by sex showed that NIHSS was lower in men who received minocycline compared with women (Amiri-Nikpour et al. 2015). Despite this early success, not all studies have reproduced the beneficial effect of MMP inhibition in the acute stroke setting. Another study using minocycline 100 mg administered intravenously within 24 h of stroke onset versus placebo did not show efficacy in terms of functional independence at day 90(Kohler et al. 2013). A meta-analysis of seven RCTs (including the four mentioned above plus two including hemorrhagic strokes and one including both ischemic and hemorrhagic strokes) using minocycline versus placebo confirmed that minocycline was associated with greater functional independence and motor performance (measured by mRS, Barthel Index, and NIHSS) compared to placebo in patients with AIS, but not in patients with hemorrhagic stroke (Malhotra et al. 2018). Larger studies are needed to verify the effectiveness of minocycline in the management of AIS, especially in combination with mechanical thrombectomy.
MMPs play a role in all stages of atherosclerotic disease. In brain larger arteries with advanced atherosclerotic disease, MMPs promote plaque rupture (Fig. 1) (Huang et al. 2017; Hunter et al. 2018; Myasoedova et al. 2018). A study of autopsied cerebral arteries in HIV + patients versus HIV-negative controls found a greater imbalance of MMPs and TIMPs (greater staining of MMP-2, MMP-3, and MMP-9 as well as less staining of TIMP-1 and TIMP-2) in HIV-positive patients and with a cell count of < 200 cel/ul. This imbalance suggests a greater elastolytic activity, which suggests greater vulnerability of the plaques to endothelial injury and thrombosis, which could lead to a higher incidence of AIS in patients with HIV (Hunter et al. 2018). A future therapeutic option may be to consider MMP inhibition in vulnerable or high-risk plaques to stabilize the plaque and decrease the short-term risk of artery-to-artery embolism or in situ thrombosis. It is of interest to know if the inhibition of MMPs by tetracyclines would have an effect on hemorrhagic transformation after an AIS.
Vascular dementia
Patients with multi-infarct dementia or dementia in the setting of small vessel disease have higher CSF concentrations of MMP-9 compared with non-demented controls or with patients with Alzheimer’s dementia (Adair et al. 2004). Another study showed increased levels of MMP-10 in CSF in patients with subcortical vascular disease and patients with Alzheimer’s dementia compared to controls (Bjerke et al. 2011). Similarly, high brain MMP-3 (Rosenberg et al. 2001) but decreased plasma levels of TIMP-1 (Lorenzl et al. 2008) has been associated with vascular dementia compared to controls. Whether MMP regulation may be a therapeutic target for vascular dementia remains unclear.
Dilatative arteriopathies
Dolichoectasia
Dolichoectasia is a common cause of stroke and carries a high risk of mortality (Gutierrez et al. 2011). In a study of autopsied brains, our group demonstrated that large artery dolichoectasia was associated with higher MMP-9 and MMP-2 expression and lower TIMP-2 (Gutierrez et al. 2016). In another study, levels of serum MMP-2 were higher in the presence of a cervical carotid kink or tortuosity than in their absence (Arslan et al. 2016). Nonetheless, in another study, intracranial dolichoectasia was associated with lower serum levels of MMP-3, but not MMP-2 or MMP-9(Pico et al. 2010). In this study, there was a negative correlation between basilar artery diameter and MMP-3 serum levels. It is unknown whether MMP-modifying therapies may play a role in dolichoectasia.
Intracranial saccular aneurysms
Inflammation plays a role in aneurysm formation and rupture. Inflammation leads to a release of MMPs that degrade and remodel the ECM (Hashimoto et al. 2005). ECM degradation leads to weakness of the vessel wall and rupture (Hashimoto et al. 2005). A feasibility study in patients with brain arteriovenous malformations or intracranial aneurysms randomized participants to doxycycline or minocycline (200 mg/day) up to 2 years; only 10 of 26 patients completed the 2 years of treatment. Treatment-related adverse events occurred in 13 of 26 patients. The minocycline group tended to have more side effects, but no statistically significant (Frenzel et al. 2008). Although the authors recommended a more comprehensive study to assess the risk of bleeding in cerebral vascular malformations with minocycline or doxycycline, no more clinical trials are known. Further RCTs are required to confirm the findings of this preliminary study.
Abdominal aortic aneurysm (AAA)
MMP inhibition has been tested among people with AAA and may inform of possible therapeutic role for MMP inhibition in brain dilatative arteriopathies. For example, doxycycline is safe in patients and reduces the growth rates of abdominal aortic aneurysms (Baxter et al. 2002) and, when administered for 2 weeks, alters aortic wall neutrophil and cytotoxic T cells content in patients undergoing elective AAA repair (Lindeman et al. 2009). In patients randomized to doxycycline 100 mg twice daily or placebo for 6 months after endovascular aneurysm repair showed that participants who received doxycycline had lower MMP-9 plasma levels at 6 months compared to baseline. However, there was no significant effect of doxycycline in aneurysm size (Hackmann et al. 2008). Furthermore, another study of small AAAs in which participants were randomized to doxycycline 100 mg versus placebo for 18 months was stopped early because an interim analysis showed an unexpected increase in aneurysm growth in the doxycycline group (Meijer et al. 2013). Similarly, another RCT of 254 patients investigated doxycycline 100 mg twice daily for 24 months aimed at growth reduction of small AAAs. At the 24-month follow-up mark, doxycycline did not significantly reduce the aneurysm growth (Baxter et al. 2020). The maximum transverse diameter (mean change) after 2 years was 0.36 cm in both groups.
Neurodegenerative disorders
Alzheimer’s disease
There is evidence that MMPs play a role in Alzheimer’s disease (AD). MMP-9 is elevated in persons with AD, and TIMP-1 and MMP-9 have been proposed as CSF-markers, in addition to T-Tau, P-Tau181, and Aβ1-42 (Bjerke et al. 2011). It has been shown that MMP-14 is negatively correlated with amyloid precursor protein expression in the hippocampus of persons with AZ (Cheng et al. 2018).
Because tetracyclines have an in vitro anti-amyloidogenic activity inhibiting the formation of ß-amyloid aggregates (Forloni et al. 2001), there have been studies focusing on MMP inhibition in AD. A multicenter RCT explored if 24-month treatment with 200 mg or 400 mg minocycline vs placebo could modify cognitive and functional decline in patients with mild AD. There were a considerable number of treatment withdrawals; however, the treatment with minocycline did not show any significant difference in delaying the progress of cognitive or functional impairment (Howard et al. 2020). In a pilot study, 101 individuals with mild AD were randomized to orally intake of doxycycline 200 mg daily plus rifampin 300 mg daily for 3 months or to placebo. The outcome was defined as a change in the standardized Alzheimer’s Disease Assessment Scale-cognitive subscale (ADAScog) at 6 and 12 months. There was significantly less worsening in the ADAScog at 6 months in the doxycycline/rifampin group but no significant differences at 12 months, however (Loeb et al. 2004). Another multicenter RCT of individuals with mild to moderate AD randomized 406 participants to four groups: doxycycline 100 twice daily plus rifampin 300 mg daily, doxycycline 100 mg twice daily plus placebo, rifampin 300 mg plus placebo, and placebo-doxycycline plus placebo-rifampin. The study failed to demonstrate the benefit of doxycycline with or without rifampin (Molloy et al. 2013). It is uncertain whether risk stratification among people with AD and evidence of dysregulated MMPs may respond better to doxycycline.
Huntington’s disease
Huntington’s disease (HD) is an autosomal neurodegenerative disorder characterized by a progressive movement disorder associated with psychiatric and cognitive decline. It has been shown that MMP-10 cleaves huntingtin, and decrease of MMP-10 reduces cell death in HD (Miller et al. 2010). Studies in mice showed a beneficial effect of minocycline delaying progression disease (Stack et al. 2006). A pilot study with 14 patients with genetically confirmed HD received minocycline 100 mg daily for 24 months. After 1 year, three patients were lost to follow-up, and 11 patients were psychiatrically, neurologically, and neuropsychologically evaluated. In the remaining 11 participants, neurological, neuropsychological, and psychiatric scoring remained stable or improved, but the motor symptoms did not improve (Bonelli et al. 2004). Subsequently, the Huntington Study Group-DOMINO conducted an RCT using a futility design. The null hypothesis was that minocycline would reduce the mean decline in the Total Functional Capacity (TFC) Score from baseline to 18 months by at least 25% compared to placebo and a historical database from past clinical trials in HD. One hundred fourteen participants were randomized to minocycline 100 mg twice daily or placebo. Mean TFC Score decline in the minocycline group was greater than the pre-specified 25% but not statistically significant (Investigators, The Huntington Study Group DOMINO 2010). Investigators concluded that it would be futile to proceed with another clinical trial using minocycline involving more participants.
Creutzfeldt-Jakob disease
A phase 2 RCT tested the efficacy and safety of doxycycline 100 mg daily in the treatment of Creutzfeldt-Jakob disease (CJD). The primary outcome was survival from the time of randomization to death. After an interim analysis of 121 patients, the trial was stopped because of the lack of superiority of doxycycline compared with placebo (Haïk et al. 2014). Another study explored the use of doxycycline 100 mg. The study consisted of two parts. In the first part, a double-blinded RCT, seven participants in early stages of CJD in the treatment group vs five controls with placebo were enrolled. The median survival time was greater in the doxycycline group versus controls (95 vs 33 days) but not statistically significant (HR 0.481, 95% CI 0.238–2.966) (Varges et al. 2017). The second part was an observational study with patients who were treated compassionately with doxycycline vs a matched treatment-naive control and showed a modest although statistically significant increase in survival time (unadjusted median survival time for doxycycline group was 346 days versus 208 days for the control group, HR 0.607, 95% CI 0.372–0.989). The results of both studies were combined and showed a slight increase in survival time in the doxycycline group (p = 0.049, HR 0.63, 95% CI 0.402–0.999) (Varges et al. 2017). More clinical trials in early-stage CJD are needed to define whether MMP inhibition should be routinely recommended in people with CJD.
Parkinson’s disease
MMP-3 is involved in microglia activation, thus playing an important role in neuronal degeneration in Parkinson’s disease (Choi et al. 2008).
As a potent inhibitor of microglial activation, minocycline was used in a phase 2 clinical trial to test the safety and progression of the disease in patients with a recent diagnosis of Parkinson’s disease that did not yet require symptomatic treatment. Participants were randomized to placebo, creatine (nutritional supplement), or minocycline (200 mg/day). Although minocycline was not declared futile, no reduction in disease progression was shown (Ninds Net-Pd Investigators 2006). An extension of the study with a follow-up in an additional 6 months (18 months in total) also showed no effect on the progression of Parkinson’s disease (Ninds Net-Pd Investigators 2008). Authors recommended a larger phase 3 study.
Neuroimmunology
Multiple sclerosis
Exacerbation of acute multiple sclerosis (MS) causes an increase in MMP-9 concentrations in CSF (Gijbels et al. 1992). Treatment with high-dose prednisolone, which restores BBB integrity, lowers MMP concentrations in CSF (Rosenberg et al. 1996). Furthermore, minocycline reduces the gadolinium-enhancing lesions in MRI (Metz et al. 2004). Two studies tested minocycline 100 mg twice daily for 6 months and doxycycline for 4 months, respectively, in patients with relapsing–remitting MS. Both studies showed a reduction in contrast-enhancing lesions on MRI (Zabad et al. 2007; Minagar et al. 2008). Another pilot study evaluated minocycline 100 mg twice daily in 10 patients with relapsing–remitting MS for 36 months and found a reduction in the mean total number of contrast-enhanced lesions during the first 6 months of treatment compared with the 3 months run-in period (0.22 vs 1.38); the mean total number of contrast-enhanced lesions dropped until 0.09 during the last 30 months treatment extension (Zhang et al. 2008). Another study compared glatiramer acetate plus minocycline versus glatiramer acetate plus placebo in 44 relapsing–remitting MS patients for a total of 9 months and showed a trend toward greater reduction in the total number of contrast-enhancing lesions on T1-weighted images and the number of confirmed relapses (Metz et al. 2009). Another RCT combined interferon-β-1a with minocycline 100 mg twice daily vs interferon-β-1a with placebo for 96 weeks in relapsing–remitting MS, 149 patients were randomized to the intervention group, and the primary efficacy endpoint (time to first qualifying relapse) was similar for both groups, showing that minocycline added to interferon-β-1a had no significant benefit (Sørensen et al. 2016).
HIV-associated neurocognitive disorders
Human immunodeficiency virus (HIV) affects directly the CNS and causes various neurological syndromes, including HIV-associated neurocognitive disorders (HAND) (McArthur et al. 2010). MMP-1 and MMP-9 are increased during primary HIV infection, and imbalance between MMPs and TIMPs contributes to progression of HIV-associated disorders (Mastroianni and Liuzzi 2007). Minocycline is a potent inhibitor of gelatinases (Paemen et al. 1996) and has been tested in human and animal HIV models of HAND. In an animal study, pigtailed macaques were intravenously inoculated with simian immunodeficiency virus; five of them were treated with minocycline (4 mg/kg per day), and six received no treatment. The macaques were sacrificed on day 84. In the untreated macaques, three developed moderate encephalitis, two severe, and one developed no encephalitis; in the group treated with minocycline, three out of five macaques did not develop encephalitis, and the other two macaques developed only mild encephalitis (Zink et al. 2005). Furthermore, minocycline suppressed viral load in the brain and decreased the expression of CNS inflammatory markers. In another study in humans, 107 HIV + individuals on antiretrovirals with cognitive impairment received either minocycline 100 mg or placebo orally twice daily for 24 weeks. A baseline neuropsychological test was performed and at 12 and 24 weeks. In the primary analysis, the study did not show a benefit of minocycline on HAND (Sacktor et al. 2011). As a secondary analysis, CSF of 21 individuals out of 107 included (13 of the placebo group and 8 of minocycline group) was studied for markers of oxidative stress, neuronal injury, and inflammation. In the minocycline group, there was a reduction in ceramides (indicator of oxidative stress); however, there were no statistically significant changes in protein carbonyl, CSF neurofilament heavy chain, neurotransmitters, or markers of inflammation such tumor necrosis factor alfa, interleukin-6, interleukin-8, and hepatocyte growth factor (Sacktor et al. 2014). A second RCT in Uganda enrolled 73 people with HIV (90% women) not on antiretrovirals to test whether minocycline had an effect on neurocognitive performance. In this study, minocycline failed to demonstrate a benefit in neurocognitive performance and provided class II evidence that minocycline 100 mg twice a day for 24 weeks does not improve HAND (Nakasujja et al. 2013).
Conclusions
Broad-spectrum and selective MMP inhibitors have been studied as therapeutic strategies in neurological and vascular conditions, but the record of effectiveness and safety is mixed. Adverse effects to MMP inhibitors are time-dependent, mostly seen in medium- and long-term clinical trials. Based on this, a more promising option may be to focus on short-term MMP inhibition. Such scenario may include AIS in which short-term MMP inhibition is hypothesized to prevent BBB disruption and vasogenic edema, therefore reducing the risk of hemorrhagic transformation or stabilizing vulnerable atherosclerotic plaques prone to rupture. In cases of intracranial dolichoectasia, MMP inhibition should be further studied, given the poor prognosis and lack of alternative specific treatments. Tetracyclines are well-known and cost-effective drugs, and their use could be easily implemented in clinical practice if proven effective in RCT.
Expert opinion
Tetracyclines are MMP inhibitors that have been studied for several decades and have not yet reached a primary clinical application as MMP inhibitors. In cerebrovascular disease, specifically in ischemic stroke, a positive trend has been seen in terms of functional status at 90 days post-stroke with the use of tetracyclines, but further replication of these results is needed. MMP inhibition has been explored as a neuroprotection strategy in people with ischemic stroke, but no significant results have been achieved. Because these trials were conducted before the thrombectomy era, it would be reasonable to reevaluate whether tetracyclines may play a role as neuroprotective therapies in conjunction with mechanical thrombectomy and thrombolysis.
Dilatative arteriopathy is a disease without specific treatment and with devastating consequences. Increased MMP expression in the arterial wall has been postulated to be a driving force in the progression of this disease. Therefore, MMP inhibition with tetracyclines or other specific compounds could be a plausible therapeutic target to be studied. The use of tetracyclines in other arteriopathies such as saccular aneurysms or arteriovenous malformations remains uncertain.
There is promising data regarding MMP inhibitors’ use for patients with Creutzfeldt-Jakob disease, as doxycycline relates to discreet survival benefits. Further studies using MMP inhibition in earlier stages of the disease are advisable.
One point of concern is the tolerability of tetracyclines and their adverse effects. Gastrointestinal effects are common and lead to poor compliance. Synthetic MMP inhibitors have also been associated with carcinogenicity. We believe that shorter exposures to MMP inhibitors may be able to overcome these side effects associated with chronic use.
Although the track record of MMP inhibition as a therapeutic modality has mixed results and only a few clinical indications, several ongoing trials are studying the use of MMP inhibition or modification in various conditions (as of May 2021, 96 protocols related to “metalloproteinases” were listed as active in clinicaltrial.gov) (see Table 3. In neurology, some studies include minocycline as a neuroprotective agent in AIS or to decrease the risk of cardiac or cerebral vascular events (Table 4) or minocycline use to delay the progression of multiple sclerosis.
We believe that essential aspects to consider when planning future therapeutic interventions based on MMP inhibition are the stages of the diseases in which the medications are used and the duration of exposure given side effects. Early use of MMP inhibition may have a more significant impact on disease progression as opposed to late stages, which tend to have a more relentless disease progression.
Tetracyclines are potent MMP inhibitors with good bioavailability in the central nervous system, and despite several therapy failures in neurological diseases, we consider that further studies are needed to evaluate whether these medications have a therapeutic role in neurological diseases.
Data availability
Not applicable.
References
Adair JC, Charlie J, Dencoff JE, Kaye JA, Quinn JF, Camicioli RM, Stetler-Stevenson WG, Rosenberg GA (2004) Measurement of gelatinase B (MMP-9) in the cerebrospinal fluid of patients with vascular dementia and Alzheimer disease. Stroke 35:e159–e162
Adhikari N, Mukherjee A, Saha A, Jha T (2017) Arylsulfonamides and selectivity of matrix metalloproteinase-2: an overview. Eur J Med Chem 129:72–109
Allan JA, Docherty AJ, Barker PJ, Huskisson NS, Reynolds JJ, Murphy G (1995) Binding of gelatinases A and B to type-I collagen and other matrix components. Biochemical Journal 309(Pt 1):299–306
Amiri-Nikpour MR, Nazarbaghi S, Hamdi-Holasou M, Rezaei Y (2015) An open-label evaluator-blinded clinical study of minocycline neuroprotection in ischemic stroke: gender-dependent effect. Acta Neurol Scand 131:45–50
Arslan Y, Arslan İB, Pekçevik Y, Şener U, Köse Ş, Zorlu Y (2016) Matrix metalloproteinase levels in cervical and intracranial carotid dolichoarteriopathies. J Stroke Cerebrovas Dis 25:2153–8
Baxter BT, Matsumura J, Curci JA, McBride R, Larson L, Blackwelder W, Lam D, Wijesinha M, Terrin M, N-TA3CT Investigators (2020) Effect of doxycycline on aneurysm growth among patients with small infrarenal abdominal aortic aneurysms: a randomized clinical trial. JAMA 323:2029–2038. https://doi.org/10.1001/jama.2020.5230
Baxter BT, Pearce WH, Waltke EA, Littooy FN, Hallett JW Jr, Kent KC, Upchurch GR Jr, Chaikof EL, Mills JL, Fleckten B, Longo GM, Lee JK, Thompson RW (2002) Prolonged administration of doxycycline in patients with small asymptomatic abdominal aortic aneurysms: report of a prospective (phase II) multicenter study. J Vasc Surg 36:1–12
Behl T, Kaur G, Sehgal A, Bhardwaj S, Singh S, Buhas C, Judea-Pusta C, Uivarosan D, Munteanu MA, Bungau S (2021) ‘Multifaceted role of matrix metalloproteinases in neurodegenerative diseases: pathophysiological and therapeutic perspectives’. Int J Mol Sci, 22
Bjerke M, Zetterberg H, Edman Å, Blennow K, Wallin A, Andreasson U (2011) Cerebrospinal fluid matrix metalloproteinases and tissue inhibitor of metalloproteinases in combination with subcortical and cortical biomarkers in vascular dementia and Alzheimer’s disease. J Alzheimers Dis 27:665–676
Bonelli RM, Hödl AK, Hofmann P, Kapfhammer HP (2004) Neuroprotection in Huntington’s disease: a 2-year study on minocycline. Int Clin Psychopharmacol 19:337–342
Cankaya S, Cankaya B, Kilic U, Kilic E, Yulug B (2019) The therapeutic role of minocycline in Parkinson’s disease. Drugs Context 8:212553
Castro MM, Kandasamy AD, Youssef N, Schulz R (2011) Matrix metalloproteinase inhibitor properties of tetracyclines: therapeutic potential in cardiovascular diseases. Pharmacol Res 64:551–560
Cheng J, Liu HP, Lee CC, Chen MY, Lin WY, Tsai FJ (2018) Matrix metalloproteinase 14 modulates diabetes and Alzheimer’s disease cross-talk: a meta-analysis. Neurol Sci 39:267–274
Choi DH, Kim EM, Son HJ, Joh TH, Kim YS, Kim D, Flint Beal M, Hwang O (2008) A novel intracellular role of matrix metalloproteinase-3 during apoptosis of dopaminergic cells. J Neurochem 106:405–415
Fanjul-Fernández M, Folgueras AR, Cabrera S, López-Otín C (2010) Matrix metalloproteinases: evolution, gene regulation and functional analysis in mouse models. Biochem Biophys Acta 1803:3–19
Forloni G, Colombo L, Girola L, Tagliavini F, Salmona M (2001) Anti-amyloidogenic activity of tetracyclines: studies in vitro. FEBS Lett 487:404–407
Frenzel T, Lee CZ, Kim H, Quinnine NJ, Hashimoto T, Lawton MT, Guglielmo BJ, McCulloch CE, Young WL (2008) Feasibility of minocycline and doxycycline use as potential vasculostatic therapy for brain vascular malformations: pilot study of adverse events and tolerance. Cerebrovasc Dis 25:157–163
Gijbels K, Masure S, Carton H, Opdenakker G (1992) Gelatinase in the cerebrospinal fluid of patients with multiple sclerosis and other inflammatory neurological disorders. J Neuroimmunol 41:29–34
Griffin MO, Fricovsky E, Ceballos G, Villarreal F (2010) ‘Tetracyclines: a pleitropic family of compounds with promising therapeutic properties Review of the Literature.’ Am J Physiol Cell Physiol 299:C539–C548
Gutierrez J, Menshawy K, Goldman J, Dwork AJ, Elkind MS, Marshall RS, Morgello S (2016) Metalloproteinases and brain arterial remodeling among individuals with and those without HIV infection. J Infect Dis 214:1329–1335
Gutierrez J, Sacco RL, Wright CB (2011) Dolichoectasia-an evolving arterial disease. Nat Rev Neurol 7:41–50
Hackmann AE, Rubin BG, Sanchez LA, Geraghty PA, Thompson RW, Curci JA (2008) A randomized, placebo-controlled trial of doxycycline after endoluminal aneurysm repair. J Vasc Surg 48:519–26 (discussion 26)
Haïk S, Marcon G, Mallet A, Tettamanti M, Welaratne A, Giaccone G, Azimi S, Pietrini V, Fabreguettes JR, Imperiale D, Cesaro P, Buffa C, Aucan C, Lucca U, Peckeu L, Suardi S, Tranchant C, Zerr I, Houillier C, Redaelli V, Vespignani H, Campanella A, Sellal F, Krasnianski A, Seilhean D, Heinemann U, Sedel F, Canovi M, Gobbi M, Di Fede G, Laplanche JL, Pocchiari M, Salmona M, Forloni G, Brandel JP, Tagliavini F (2014) Doxycycline in Creutzfeldt-Jakob disease: a phase 2, randomised, double-blind, placebo-controlled trial. Lancet Neurol 13:150–158
Hashimoto T, Matsumoto MM, Li JF, Lawton MT, Young WL (2005) Suppression of MMP-9 by doxycycline in brain arteriovenous malformations. BMC Neurol 5:1
He T, Wang J, Wang XL, Deng WS, Sun P (2017) Association between the matrix metalloproteinase-9 rs3918242 polymorphism and ischemic stroke susceptibility: a meta-analysis. J Stroke Cerebrovasc Dis 26:1136–1143
Howard R, Zubko O, Bradley R, Harper E, Pank L, O’Brien J, Fox C, Tabet N, Livingston G, Bentham P, McShane R, Burns A, Ritchie C, Reeves S, Lovestone S, Ballard C, Noble W, Nilforooshan R, Wilcock G, Gray R (2020) Minocycline at 2 different dosages vs placebo for patients with mild alzheimer disease: a randomized clinical trial. JAMA Neurol 77:164–174
Huang X, Ye Q, Zhang Z, Huang X, Zhu Z, Chen Y, Li J, Chen S, Xia N, Mao X, Han L, Ye Z (2017) Association of matrix metalloproteinase-3 gene 5A/6A polymorphism with the recurrence of ischemic stroke: a prospective observational study. Brain Res 1674:55–61
Hunter MD, Shenoy A, Dwork A, Elkind MSV, Marshall R, Mohr JP, Morgello S, Gutierrez J (2018) Brain vascular intima vulnerability among HIV-positive and negative individuals. AIDS 32:2209–2216
Investigators, The Huntington Study Group DOMINO (2010) A futility study of minocycline in Huntington’s disease. Mov Disord 25:2219–2224
Jabłońska-Trypuć A, Matejczyk M, Rosochacki S (2016) Matrix metalloproteinases (MMPs), the main extracellular matrix (ECM) enzymes in collagen degradation, as a target for anticancer drugs. J Enzyme Inhib Med Chem 31:177–183
Jiang Y, Liu H, Wang Y, Shi X, Shao Y, Xu Z (2020) Meta-analysis of matrix metalloproteinase (MMP)-9 C1562T polymorphism and susceptibility to ischemic stroke in the Chinese population. J Int Med Res 48:300060520926427
Kohler E, Prentice DA, Bates TR, Hankey GJ, Claxton A, van Heerden J, Blacker D (2013) Intravenous minocycline in acute stroke. Stroke 44:2493–2499
Lampl Y, Boaz M, Gilad R, Lorberboym M, Dabby R, Rapoport A, Anca-Hershkowitz M, Sadeh M (2007) Minocycline treatment in acute stroke: an open-label, evaluator-blinded study. Neurology 69:1404–1410
Lau LW, Cua R, Keough MB, Haylock-Jacobs S, Yong VW (2013) Pathophysiology of the brain extracellular matrix: a new target for remyelination. Nat Rev Neurosci 14:722–729
Lech AM, Wiera G, Mozrzymas JW (2019) Matrix metalloproteinase-3 in brain physiology and neurodegeneration. Adv Clin Exp Med 28:1717–1722
Lindeman JH, Abdul-Hussien H, van Bockel JH, Wolterbeek R, Kleemann R (2009) Clinical trial of doxycycline for matrix metalloproteinase-9 inhibition in patients with an abdominal aneurysm: doxycycline selectively depletes aortic wall neutrophils and cytotoxic T cells. Circulation 119:2209–2216
Loeb MB, Molloy DW, Smieja M, Standish T, Goldsmith CH, Mahony J, Smith S, Borrie M, Decoteau E, Davidson W, McDougall A, Gnarpe J, O’DONNell M, Chernesky M (2004) A randomized, controlled trial of doxycycline and rifampin for patients with Alzheimer’s disease. J Am Geriatr Soc 52:381–7
Lorenzl S, Buerger K, Hampel H, Beal MF (2008) Profiles of matrix metalloproteinases and their inhibitors in plasma of patients with dementia. Int Psychogeriatr 20:67–76
Macdonald H, Kelly RG, Allen ES, Noble JF, Kanegis LA (1973) Pharmacokinetic studies on minocycline in man. Clin Pharmacol Ther 14:852–861
Machado LS, Sazonova IY, Kozak A, Wiley DC, El-Remessy AB, Ergul A, Hess DC, Waller JL, Fagan SC (2009) Minocycline and tissue-type plasminogen activator for stroke. Stroke 40:3028–3033
Malhotra K, Chang JJ, Khunger A, Blacker D, Switzer JA, Goyal N, Hernandez AV, Pasupuleti V, Alexandrov AV, Tsivgoulis G (2018) Minocycline for acute stroke treatment: a systematic review and meta-analysis of randomized clinical trials. J Neurol 265:1871–1879
Mastroianni CM, Liuzzi GM (2007) Matrix metalloproteinase dysregulation in HIV infection: implications for therapeutic strategies. Trends Mol Med 13:449–459
McArthur JC, Steiner J, Sacktor N, Nath A (2010) Human immunodeficiency virus-associated neurocognitive disorders: mind the gap. Ann Neurol 67:699–714
Meijer CA, Stijnen T, Wasser MN, Hamming JF, van Bockel JH, Lindeman JH (2013) Doxycycline for stabilization of abdominal aortic aneurysms: a randomized trial. Ann Intern Med 159:815–823
Metz LM, Li D, Traboulsee A, Myles ML, Duquette P, Godin J, Constantin M, Yong VW (2009) Glatiramer acetate in combination with minocycline in patients with relapsing–remitting multiple sclerosis: results of a Canadian, multicenter, double-blind, placebo-controlled trial. Mult Scler 15:1183–1194
Metz LM, Zhang Y, Yeung M, Patry DG, Bell RB, Stoian CA, Yong VW, Patten SB, Duquette P, Antel JP, Mitchell JR (2004) Minocycline reduces gadolinium-enhancing magnetic resonance imaging lesions in multiple sclerosis. Ann Neurol 55:756
Mignatti P, Rifkin DB (1996) Plasminogen activators and matrix metalloproteinases in angiogenesis. Enzyme Protein 49:117–137
Miller JP, Holcomb J, Al-Ramahi I, de Haro M, Gafni J, Zhang N, Kim E, Sanhueza M, Torcassi C, Kwak S, Botas J, Hughes RE, Ellerby LM (2010) Matrix metalloproteinases are modifiers of huntingtin proteolysis and toxicity in Huntington’s disease. Neuron 67:199–212
Minagar A, Alexander JS, Schwendimann RN, Kelley RE, Gonzalez-Toledo E, Jimenez JJ, Mauro L, Jy W, Smith SJ (2008) Combination therapy with interferon beta-1a and doxycycline in multiple sclerosis: an open-label trial. Arch Neurol 65:199–204
Misra S, Talwar P, Kumar A, Kumar P, Sagar R, Vibha D, Pandit AK, Gulati A, Kushwaha S, Prasad K (2018) Association between matrix metalloproteinase family gene polymorphisms and risk of ischemic stroke: a systematic review and meta-analysis of 29 studies. Gene 672:180–194
Molloy DW, Standish TI, Zhou Q, Guyatt G (2013) A multicenter, blinded, randomized, factorial controlled trial of doxycycline and rifampin for treatment of Alzheimer’s disease: the DARAD trial. Int J Geriatr Psychiatry 28:463–470
Myasoedova VA, Chistiakov DA, Grechko AV, Orekhov AN (2018) Matrix metalloproteinases in pro-atherosclerotic arterial remodeling. J Mol Cell Cardiol 123:159–167
Nakasujja N, Miyahara S, Evans S, Lee A, Musisi S, Katabira E, Robertson K, Ronald A, Clifford DB, Sacktor N (2013) Randomized trial of minocycline in the treatment of HIV-associated cognitive impairment. Neurology 80:196–202
Nicholson C, Rice ME (1986) The migration of substances in the neuronal microenvironmenta. Ann N Y Acad Sci 481:55–68
Ninds Net-Pd Investigators (2006) A randomized, double-blind, futility clinical trial of creatine and minocycline in early Parkinson disease. Neurology 66:664–671
Ninds Net-Pd Investigators (2008) A pilot clinical trial of creatine and minocycline in early Parkinson disease: 18-month results. Clin Neuropharmacol 31:141–150
Padma Srivastava MV, Bhasin A, Bhatia R, Garg A, Gaikwad S, Prasad K, Singh MB, Tripathi M (2012) Efficacy of minocycline in acute ischemic stroke: a single-blinded, placebo-controlled trial. Neurol India 60:23–28
Paemen L, Martens E, Norga K, Masure S, Roets E, Hoogmartens J, Opdenakker G (1996) The gelatinase inhibitory activity of tetracyclines and chemically modified tetracycline analogues as measured by a novel microtiter assay for inhibitors. Biochem Pharmacol 52:105–111
Palm F, Pussinen PJ, Safer A, Tervahartiala T, Sorsa T, Urbanek C, Becher H, Grau AJ (2018) Serum matrix metalloproteinase-8, tissue inhibitor of metalloproteinase and myeloperoxidase in ischemic stroke. Atherosclerosis 271:9–14
Pico F, Jacob MP, Labreuche J, Soufir N, Touboul PJ, Benessiano J, Cambien F, Grandchamp B, Michel JB, Amarenco P (2010) Matrix metalloproteinase-3 and intracranial arterial dolichoectasia. Ann Neurol 67:508–515
Rosenberg GA (2009) Matrix metalloproteinases and their multiple roles in neurodegenerative diseases. Lancet Neurol 8:205–216
Rosenberg GA (2017) ‘Chapter 61 - Matrix Metalloproteinases and Extracellular Matrix in the Central Nervous System.’ in Louis R. Caplan, José Biller, Megan C. Leary, Eng H. Lo, Ajith J. Thomas, Midori Yenari and John H. Zhang (eds.), Primer on Cerebrovascular Diseases (Second Edition) (Academic Press: San Diego)
Rosenberg GA, Dencoff JE, Correa N Jr, Reiners M, Ford CC (1996) Effect of steroids on CSF matrix metalloproteinases in multiple sclerosis: relation to blood-brain barrier injury. Neurology 46:1626–1632
Rosenberg GA, Sullivan N, Esiri MM (2001) White matter damage is associated with matrix metalloproteinases in vascular dementia. Stroke 32:1162–1168
Saari H, Suomalainen K, Lindy O, Konttinen YT, Sorsa T (1990) Activation of latent human neutrophil collagenase by reactive oxygen species and serine proteases. Biochem Biophys Res Commun 171:979–987
Sacktor N, Miyahara S, Deng L, Evans S, Schifitto G, Cohen BA, Paul R, Robertson K, Jarocki B, Scarsi K, Coombs RW, Zink MC, Nath A, Smith E, Ellis RJ, Singer E, Weihe J, McCarthy S, Hosey L, Clifford DB (2011) Minocycline treatment for HIV-associated cognitive impairment: results from a randomized trial. Neurology 77:1135–1142
Sacktor N, Miyahara S, Evans S, Schifitto G, Cohen B, Haughey N, Drewes JL, Graham D, Zink MC, Anderson C, Nath A, Pardo CA, McCarthy S, Hosey L, Clifford D (2014) Impact of minocycline on cerebrospinal fluid markers of oxidative stress, neuronal injury, and inflammation in HIV-seropositive individuals with cognitive impairment. J Neurovirol 20:620–626
Sood RR, Taheri S, Candelario-Jalil E, Estrada EY, Rosenberg GA (2008) Early beneficial effect of matrix metalloproteinase inhibition on blood-brain barrier permeability as measured by magnetic resonance imaging countered by impaired long-term recovery after stroke in rat brain. J Cereb Blood Flow Metab 28:431–438
Sørensen PS, Sellebjerg F, Lycke J, Färkkilä M, Créange A, Lund CG, Schluep M, Frederiksen JL, Stenager E, Pfleger C, Garde E, Kinnunen E, Marhardt K (2016) Minocycline added to subcutaneous interferon β-1a in multiple sclerosis: randomized RECYCLINE study. Eur J Neurol 23:861–870
Stack EC, Smith KM, Ryu H, Cormier K, Chen M, Hagerty SW, Del Signore SJ, Cudkowicz ME, Friedlander RM, Ferrante RJ (2006) Combination therapy using minocycline and coenzyme Q10 in R6/2 transgenic Huntington’s disease mice. Biochem Biophys Acta 1762:373–380
Stirling DP, Koochesfahani KM, Steeves JD, Tetzlaff W (2005) Minocycline as a neuroprotective agent. Neuroscientist 11:308–322
Varges D, Manthey H, Heinemann U, Ponto C, Schmitz M, Schulz-Schaeffer WJ, Krasnianski A, Breithaupt M, Fincke F, Kramer K, Friede T, Zerr I (2017) Doxycycline in early CJD: a double-blinded randomised phase II and observational study. J Neurol Neurosurg Psychiatry 88:119–125
Wang XX, Tan MS, Yu JT, Tan L (2014) Matrix metalloproteinases and their multiple roles in Alzheimer’s disease. Biomed Res Int 2014:908636
Zabad RK, Metz LM, Todoruk TR, Zhang Y, Mitchell JR, Yeung M, Patry DG, Bell RB, Yong VW (2007) The clinical response to minocycline in multiple sclerosis is accompanied by beneficial immune changes: a pilot study. Mult Scler 13:517–526
Zhang Y, Metz LM, Yong VW, Bell RB, Yeung M, Patry DG, Mitchell JR (2008) Pilot study of minocycline in relapsing-remitting multiple sclerosis. Can J Neurol Sci 35:185–191
Zink MC, Uhrlaub J, DeWitt J, Voelker T, Bullock B, Mankowski J, Tarwater P, Clements J, Barber S (2005) Neuroprotective and anti-human immunodeficiency virus activity of minocycline. JAMA 293:2003–2011
Author information
Authors and Affiliations
Contributions
JG conceived and designed this review article and revised the manuscript. ELN collected and extracted data and wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
The authors declare consent to publish this article.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lopez-Navarro, E.R., Gutierrez, J. Metalloproteinases and their inhibitors in neurological disease. Naunyn-Schmiedeberg's Arch Pharmacol 395, 27–38 (2022). https://doi.org/10.1007/s00210-021-02188-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-021-02188-x