Abstract
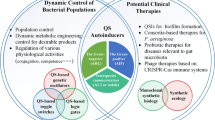
Bacteria utilize small signal molecules to monitor population densities. Bacteria arrange gene regulation in a method called Quorum Sensing (QS). The most widespread signalling molecules are N-Acyl Homoserine Lactones (AHLs/HSLs) for Gram-negative bacteria communities. QS plays significant role in the organizing of the bacterial gene that adapts to harsh environmental conditions for bacteria. It is involved in the arrangement of duties, such as biofilm formation occurrence, virulence activity of bacteria, production of antibiotics, plasmid conjugal transfer incident, pigmentation phenomenon and production of exopolysaccharide (EPS). QS obviously impacts on human health, agriculture and environment. AHL-related QS researches have been extensively studied and understood in depth for cell to cell intercommunication channel in Gram-negative bacteria. It is understood that AHL-based QS research has been extensively studied for cell-to-cell communication in Gram-negative bacteria; hence, a comprehensive study of AHLs, which are bacterial signal molecules, is required. The purpose of this review is to examine the effects of QS-mediated AHLs in many areas by looking at them from a different perspectives, such as clinic samples, food industry, aquatic life and wastewater treatment system.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Use of antibiotics and their resistance, troubles in treatment infectious diseases and Quorum sensing
Various strategies have been developed to combat pathogenic bacteria, one of the most important problems of our time. However, the progress of resistance to antibiotics makes it trouble to fight infectious diseases. This makes it necessary to discover alternative treatment methods to classical treatment ones. The discovery of the Quorum Sensing (QS) mechanism, which enables the perception of the environment in microorganisms, has gained a new perspective in the treatment of diseases (De Kievit and Iglewski 2000; Miller and Bassler 2002; Waters and Bassler 2005). By identifying the signal molecules included in the QS system of the infectious microorganism, it may be possible to separate the microorganism from fragments by initiation steps in the host at different points of binding, propagation, communication (sensing) and propagation cycle infection. This will increase the success of infection treatment.
Today, bacteria with antibiotic resistance are a serious problem worldwide. Antibiotics utilized in the treatment and recovery of infections attract attention as one of the substantial discoveries of the last century. This success against infectious agents has been overshadowed by the widespread and improper usage of antibiotics, and in consequence of the bacteria becoming resistant to antibiotics in the recent years has led to a problem that threatens public health. (Dcosta et al. 2011). Microorganisms have developed different resistance mechanisms against antibiotics to which they are sensitive, and this situation has expanded their virulence properties (Davies and Department 2010).
Many bacterial cell–cell communication signal molecules have been found out recently, mainly, acyl homoserine lactone (AHL or HSL), hydroxyl-palmitic acid methyl ester (PAME), cyclic thiolactone (AIP), furanosylborate (AI-2) and methyl dodecenoic acid (DSF) (Derome et al. 2016). These molecules affect bacterial toxicity. Among these QS signals, AHLs are the most prominent cell–cell communication signals. The QS phenomenon is not unique to prokaryotic organisms; unicellular eukaryotic fungus pathogens also regulate their biological functions using QS signals (Albuquerque and Casadevall 2012). This communication system controls the activation of more than one mechanism in microorganisms. These bacteria develop as multicellular groups or biofilms on all living surfaces (Liu et al. 2016). In short, "QS" provides some advantages to prokaryotic microorganisms, such as the complex cell structures in eukaryotes.
In the report of the World Health Organization, it is stated that more than 60% of hospital-acquired infections have been given rise to by antibiotic-resistant bacteria (Ducel and Fabry 2002; Wilson et al. 2016). It is important to identify the agents involved in QS event for controlling diseases caused by these bacteria and to develop new treatments (Defoirdt 2018). With the usage of antibiotics in the 1930s, a unique era began for the treatment process of bacterial infections. As a result, it gave rise to a significant dropping in the number of deaths in bacterial origin infections. Nevertheless, the therapeutic methods are limited, and resistant bacteria are emerging and spreading. Naturally, the treatment of some infections has become increasingly difficult (Cámara et al. 2002).
Among the strains with the highest antibiotic resistance are Gram-negative species, including Pseudomonas aeruginosa, Escherichia coli, Klebsiella pneumoniae (WHO and World Health Organization 2017). Owing to multidrug resistance that develops in Gram-negative bacteria, untreatable infections may emerge recently. Most of beta-lactam antibiotics, quinolones, aminoglycosides, trimethoprim–sulfometaxozole are the basic antibiotics used in Gram-negative bacterial infections (Yekani et al. 2018). Because of the frequent use of third-generation antibiotics in prophylaxis and empirical treatment and not being included in limited antibiotic applications, the ratio of resistance to antibiotics in this group has increased (Tamma et al. 2011, 2014, 2019). Carbapenems are the most effective broad-spectrum antibiotics against Gram-negative bacilli. However, due to carbapenem resistance that develops in Gram negatives, treatment options are restricted, prolonging hospital stay and bringing along many problems, such as increased treatment costs and patient mortality (Ghafur 2014; Erdönmez et al. 2017). For example, this situation requires the addition of options, such as tigecycline and colistin to the treatment. Thus, these multidrug resistant organisms are difficult to treat and new agents are urgently needed. Nevertheless, there is no argument that this requirement could be met for foreseeable future. Gene expressions important in bacterial virulence are regulated by the QS mechanism. This situation reveals the importance of the QS system.
N-acyl homoserine lactones mediated QS
Although high living organisms use sounds and words to communicate with each other, bacteria provide this function with some communication molecules. These communication molecules are also referred to as autoinducers because of the regulation of the metabolism of the cells and other cells in which they are produced. This event is expressed as "Quorum Sensing" or environmental sensing or bacterial communication (Kravchenko et al. 2006; Khajanchi et al. 2011; Chang et al. 2014). QS is a standard mechanism utilized by the Gram-negative bacteria species and operated.
The LuxI/LuxR protein mechanism manages this kind of communication channel depicted in Fig. 1 (De Kievit and Iglewski 2000; Schauder and Bassler 2001; Turan et al. 2017). QS period begins by a LuxI protein via producing an acylated homoserin-lactone molecule. The concentration density of the produced autoinducer intensifies to an enough degree where a cognate LuxR protein perceives it and directs all dependent target genes (Schauder and Bassler 2001). Namely, LuxI-like protein is an autoinductor synthase that catalyzes the formation of a specific AHL. The AHL molecule diffuses freely across the cell membrane at high cell density. The LuxR is a transcriptional regulator system which binds to the diffusing AHL and prompts the transcription of its target genes (Li and Tian 2012).
QS is described as a communication mechanism by which a single microorganism releases the signal molecule into the medium and senses it by the population, ensuring that gene expression takes place in a coordinated manner taking into account gene expression of an entire population (Watson et al. 2002; Kawaguchi et al. 2008; Schikora et al. 2011). QS takes part an major role in the organizing of the bacterial gene that adapts to harsh environmental conditions for bacteria. It is also involved in the regulation of missions, such as formation of biofilm occurrence, bacterial virulence activity, production of antibiotic, plasmid conjugal transfer incident, pigmentation phenomenon and production of exopolysaccharide (EPS). QS directly affects on human health, environment and agriculture an overall(Turan et al. 2017). The defense process of an organism can cope with many pathogenic microbial kinds involved in virulence by QS. The coordinated invasion in the host system happens when the bacterial population density achieves sufficiently high values. The appropriate method to alleviate bacterial infections may be the control of QS signals. Bacterial actions are not advantageous only when expressed by a bacterium, but may be useful to bacteria when expressed on a group basis (Kumari et al. 2008).
Many plant, animal and human pathogens compose virulence event by bacterial cell-to-cell communications through signalling molecules. The signal molecules released by Gram-negative bacteria communicate with the coordination of gene expression depending on the intensity of the bacterial population (Cataldi et al. 2007; Pinto et al. 2007; Soares and Ahmer 2011; Hou et al. 2017; Habimana et al. 2018). Bacteria communities control the existence of other bacteria by occurring and reacting to specific chemical signalling agents. There are more than one chemical signalling molecule for the communication of bacteria and these molecules are involved in the creation of the effect resulting from changes in the microenvironment by communicating both within and between species (Miller and Bassler 2002; Davis et al. 2010). It is observed that there is a difference in antibiotic resistance between the species isolated from clinical samples and the species isolated from the environment of the Gram-negative bacteria groups, such as P. aeruginosa, E. coli and K. pneumoniae (Cepas et al. 2019). Among the reasons for this difference, changes in microorganism metabolism due to the difference in signal molecules generated by the QS system are considered.
Many various AHL signals have been obviously identified for the last decade. The structure and concentration of AHL is extremely substantial for AHL-based QS experiments. The length of the acyl chain generally ranges from 4 to 18 carbons with increasing two carbon numbers, such as C4, C6 and C8 (Huang et al. 2016). The chemical structures with general structure of lactones are shown in Fig. 2. As seen, a lactone ring is composed of amide and carbonyl groups and one hydrocarbon chain. Although the signal molecules of N-acyl homoserine lactones belong to Gram-negative bacteria, oligopeptides to Gram-positive bacteria (Kumari et al. 2008).
Determining the antibiotic susceptibility of P. aeruginosa, E. coli and K. pneumoniae species isolated from various clinics and showing different resistance profiles, investigating which signal molecules are effective in infection and elucidating the relationship between the signal molecules used and resistance profiles are among the main goals in preventing the problem of resistance. It should also be aimed to investigate which signal molecules play a role in resistance to various antibiotic groups.
AHL-dependent QS researches have been comprehensively studied and understood in depth for cell-to-cell connection and communication skills in Gram-negative bacteria. The method works with three basic rules:
-
(i)
AHL signals emitted to the environment;
-
(ii)
an AHL synthase protein for generating the AHL signal;
-
(iii)
the regulatory protein that senses and replies to the concentration of AHL signals (Shrout and Nerenberg 2012; Lade et al. 2014).
Screening for AHL production from bacterial strains is typically based on the bacteriological monitoring systems consisting of a phenotypic response activated via an AHL receptor protein (Ravn et al. 2001). The best studied QS system is the LuxR–LuxI system in Gram-negative bacteria with widespread antibiotic resistance, and the signal molecule is AHL. 60% of all microbial infections are caused by biofilm, the regulation mechanism of this source is done by the quorum sensing system (Jefferson 2004). The number of known QS systems is increasing day by day as various bacterial species are examined. By determining the amount and type of AHL, it will be easier to remove infections or contaminated environments from the mentioned microorganisms. The detection of these AHLs in environments should not always be considered negative. For example, in a study where both nitrification and denitrification efficiencies of activated sludge systems were increased by the addition of C6-HSL and C8-HSL, it was stated that the investigation of methods to improve certain AHL-producing bacterial species would provide a potential strategy for regulation and optimization in activated sludge systems (Yan et al. 2020).
Effect, result and identification of AHLs in different areas
Diagnosing of AHLs on clinic samples
It is thought that the illumination of the bacterial communication system known as the perception of the environment or the majority, which is called the "QS" system in microorganisms, will increase the chance of success of antibacterial therapy. Therefore, the functioning of all virulence factors associated with this system will be enlightened, and its place in treatment will be revealed. It can be explained the effect of these virulence factors as follows; Due to this system, bacteria act like multicellular eukaryotic organisms and gain a number of advantages that bacteria cannot achieve alone. Many bacterial behaviors are controlled by environmental perception mechanisms. Bacteria can distinguish between low or high population densities in their environment with the signal molecules perceived by them, and thus, controlling gene expression at the population degree in response to the alteration in the number of cells in the environment.
QS is known as the language of bacteria (Turan et al. 2017). AHL, the main signalling molecules of Gram-negative bacteria can express pathogenic agents, and the QS system can take an important role in the detection of infection. AHL is significant in the organizing of bacterial virulence genes. It can also interact with eukaryotic cells, decrease an immune reply that facilitates infection (Jiang et al. 2016). Inhibition of this system, which has been found to be effective in virulence, is a new approach in the treatment of many infections. In the formation of this inhibition, as in the action mechanisms of antibiotics, either the production of the signal molecule will be inhibited, the signal molecule will be destroyed or blocked or other bacteria will be prevented from sensing the signal molecules.
AHL based QS has also been shown to cause chronic level bacterial infections cases by P. aeruginosa, B. cepacia, which cause lung illness in patients with Cystic Fibrosis (CF) (Middleton et al. 2002; Chambers et al. 2005). QS has been represented to be managing for the formation of biofilms in both inanimate surfaces and lung tissue (Wu et al. 2000; Kjelleberg and Molin 2002; Middleton et al. 2002). Furthermore, QS acts in the pathogenesis of some gastrointestinal (GI) diseases. QS has been shown to be effective in intestinal colonization by pathogens and has been extensively proven in the regulation of virulence genes on pathogens (Sperandio et al. 1999; Surette et al. 1999; Falcão et al. 2004; Sircili et al. 2004; Kaper and Sperandio 2005; Kendall and Sperandio 2007).
Some studies have been conducted for the detection of AHL in some people based on the clinical samples. One of these studies have displayed the presence of bacterial AHL signalling molecules in sputum experimental samples of CF patients infected with Pseudomonas aeruginosa or Burkholderia cepacia. AHLs were found to check up on the expression of bacterial virulence genes. It has also been shown to be effective in triggering the occurrence of antibiotic-resistant biofilms and host chemokine releases. In this context, there are observations and evidence that different forms of inhibition of the bacterial communication system can exert antibacterial effects.
For detection of AHLs, the test samples of bronchoalveolar lavage (BAL) were experimentally determined by means of dichloromethane-extracted supernatants by utilizing the bioluminescence dependent AHL reporter plasmid pSB1075. Under these conditions, it was s observed that AHLs (C10–C14) generated light (Ward et al. 2003). These AHLs are the signalling molecules of Pseudomonas aeruginosa. The presence of these species of bacteria is an extensive reason of inflammation formation of the ear canal (otitis externa) in dogs. Up to this research, there has no news on finding of AHLs in the clinical samples of veterinary-related fields. Samples from ear canals with otitis externa, and samples from healthy dogs were used for controlled experiments. It enables the quantification of AHL kinds in clinical and non-clinical samples by the procedure applied. Also, a time depending diminish in AHL intensity was observed for the treated dogs. Liquid chromatography–mass spectrometry/mass spectrometry (LC–MS/MS) analytical method was conducted for defining AHLs opted P. aeruginosa. As a result, C4-HSL, C6-HSL and 3OC12-HSL were determined in experimental clinical samples(Kušar et al. 2016). P. aeruginosa, a Gram-negative bacteria, is the predominant pathogen that can infect humans and affect CF patients at certain age periods. P. aeruginosa infection has been reported in 60% of people with CF in the age of 16 and older in the UK. Wen et al., has been used P. aeruginosa AHL detection circuit to analyse CF patient sputum samples applied in an E. coli-based cell-free system. It was demonstared the presence of 3OC12-HSL from P. aeruginosa-infected respiratory samples (Wen et al. 2017). Middleton et al. have detected direct AHL in sputum samples taken from CF patients colonized with P. aeruginosa and B. cepacia. The bacterial cell detection was performed to diagnose the presence of AHL kinds. High resolution MS coupled LC was taken advantage for identification tests of AHL kinds (Middleton et al. 2002). The AHLs identified in sputum samples taken from CF patients were found as C6-HSL, 3OC12-DL-HSL. Wu et al. infected the lung tissues of mice by P. aeruginosa and studied experimentally (Wu et al. 2000). Bacterial detection system was used to identify AHL in in vivo experiments of these infected lung tissues. In addition, AHLs have been observed in severe pathological cases. Thus, the connection between P. aeruginosa and QS pathogenicity status was confirmed. Sokol et al. used a bacterial cell detection method to define AHLs in the lung homogenates of B. kenocepacia-infected mice (Sokol et al. 2003). Similarly, it was demonstrated the act of QS in B. kenocepakia infections. In another study, Jacobi et al. have studied both liver and spleen homogenates of Y. enterocolitica serotype O8 infected mice, which are immensely pathogenic in mice, both in vitro and in vivo (Jacobi et al. 2003) and used a bioluminescence bacterial cell detection system to detect AHLs. In this study, the relationship between QS-based AHL relationship was confirmed. The resistance of bacteria to antibiotics has been shown in Fig. 3.
The resistance of bacteria to antibiotics; a Bacteria medium, b Destruction of some bacteria with the addition of antibiotics in the environment, c Communication of bacteria not destruction in the environment with the help of signal molecules, d After signalling, bacteria provide resistance to antibiotics
The importance of AHLs in food industry
The formation of persistent biofilms containing pathogens is one of the big problems in the food processing industry, as surface contamination in contact with food can have significant public health impacts (Jamuna Bai 2016). Compounds found in food matrixes and storage conditions can affect the production of AHLs (Medina-Martinez et al. 2006). Pseudomonas spp., P. fluorescens, P. putida and P. fragi are the proteinaceous dominant spoilage microbiota for aerobically cooled beef (Jay et al. 2003), raw milk (Kraft 1992), fish (Tryfinopoulou et al. 2002), chicken (Arnaut-Rollier et al. 1999) and protein products (Gennari and Dragotto 1992; Ternström et al. 1993). Other some spoilage organisms in aerobic cold manner stored raw foods are kinds of Aeromonas, Shewanella, Moraxella, Psychrobacter and Acinetobacter and psychrotrophic members of the Enterobacteriaceae, such as Hafnia alvei, Serratia spp. and Enterobacter spp (Borch et al. 1996; Stanbridge and Davies 1998; Liu et al. 2006). Medina et al. conducted a study to make a decision on AHL production of A. hydrophila and A. caviae strains and to appraise the effect of environmental reasons on AHL generation. It was presented that the Aeromonas test strains could compose AHLs (especially C4-HSL) with a different of status of temperature effect, pH, NaCl concentrations usually were come across in foods linked with this microorganism when increasing enough numbers (population density) was accessed (Medina-Martinez et al. 2006).
Meat is one of the most consumed food products and has a very short shelf life. In one study, the samples of beef were investigated for microbial relation and the existence of AHLs during the storage studies. Isolates were monitorized for AHLs formation, and opted spices were followed by QS inhibitory (QSI) activity. The impact of added spice process on AHLs manufacturing of Y. enterocolitica was indicated by quantitation via high-performance thin layer chromatography (HP-TLC). The results demonstrated that microbial relation of beef mostly happens in the presence of Enterobacteriaceae and lactic acid bacteria (LAB). Outcomes showed the asset of AHLs in beef meat during the spoilage event. It was also observed that the spice decreased the formation of AHLs by reducing the spoilage rate. (Gopu and Shetty 2016). In another study, AHLs were diagnosed from five commercially genarated vacuum-packed meat samples. It was showed that compounds stimulating QS mechanisms are prevalent in spoiling vacuum pack meat within scope of study (Bruhn et al. 2004). The schematic representation of bacterial AHL production, uptake of signal molecules by the bacterial receptor and gene expression is shown in Fig. 4.
Effect of AHL molecules on aquatic life
The seafood sector is among the fastest developing food production industry worldwide. Pathogens causing infectious diseases can cause great economic damage to the aquaculture industry. Aeromonas spp. and Vibrio spp. are the most common aquatic pathogens that kill shrimps, fish and molluscs (Niu et al. 2014; de la Fuente et al. 2015; Zhao et al. 2015; Baker-Austin and Oliver 2018). Bacteria attack these living organisms by QS-originated pathogenicity. Conventionally, bacterial infections are cured using antimicrobials. Even if there are numerous remedy methods which are accepted useful in aquaculture, the studies requiring methods that can minimize commercial economic losses on an industrial scale should be carried out (Kalia et al. 2019). Most of farmers also apply antibiotics in a prophylactic strategy, even when pathogens do not become apparent(Holmström et al. 2003). This application has concluded in the progress of (multiple) antibiotic resistance (Cabello 2006) which does antibiotic treatments impotent in inspection of diseases (Karunasagar et al. 1994; Defoirdt et al. 2011). In one of the studies, a cell sensor was developed and used to measure the QS signalling molecule. It is aimed to monitor the bacteria responsible for the degradation of freshwater fish production in real time. A cell sensor was developed and used to measure the QS signalling molecule. A modified carbon electrode is used to increase accuracy. Electrochemical impedance spectroscopy has been used to save the cell impedance signal affected by Pseudomonas aeruginosa and has been determined 3OC12-HSL molecules. As a result of this work, it was concluded that it could serve as an effective and innovative approach in monitoring deterioration in freshwater fish production (Jiang et al. 2018). In another study, the presence of AHLs in marine bacterial strains collected from heavily contaminated surfaces in the Gulf of Santa Marta (Colombia) has been proven. The detection and identification process of AHLs were carried out by operating the microbial biosensor Escherichia coli (pSB401) and GC–MS, HPLC–MS experiments. All isolated marine strains have been found to have QS systems mediated by C4-HSL or C6-HSL, and in some cases both (Cuadrado-Silva et al. 2013). AHLs act a crucial role in Gram-negative bacterial communication channel by supporting the constitution of biofilms and extracellular polymeric substances (EPS). Wang et al. developed a stable and sensitive procedure for detection of AHL. To obtain AHLs and detect concentration level of AHLs in wastewater treatment biofilms ultrasonic extraction, an Oasis hydrophilic–lipophilic-balanced (HLB) sorbent system in column format assembled with ultra-performance liquid chromatography linked to tandem mass spectrometry (UPLC-MS/MS) was used. This method is ideal and easy to follow process for quantitative experiments of AHLs in wastewater treatment biofilms (Wang et al. 2017).
Sturgeon is fresh water-culture fish in China, and it has high susceptibility to spoilage. In order to identify QS signals acting on sturgeon, it was checked whether QS was involved in spoilage. Control of production of AHLs and specific spoilages were performed on sturgeon stored at 4 °C. At the end of study by thin layer chromatography (TLC), high-performance liquid chromatographytriple quadrupole tandem mass spectrometry (HPLC-TQMS) and high-performance liquid chromatography–quadrupole time-of-flight mass spectrometry (HPLC/qTOF-MS), it was assigned that the AHLs generated by A. veronii were C6-HSL, C8-HSL, 3-oxo-C8-HSL and 3-OH-C8-HSL. This study brought to light that QS system was probably mediated in the regulation of sturgeon spoilage. First, it was declared that the spoilage caused by Aeromonas was from C8-HSL and 3-OH-C8-HSL production (Gui et al. 2018).
Industrial significance of AHL molecules and their effect for wastewater treatment system
AHLs utilized by Gram-negative bacteria communities are one of the best defining signal molecules that play an important role in organizing virulence factor secretion, manufacturing of exoenzyme and QS originated biofilm formation (Ma et al. 2018). The cooling water procedures are applied to eliminate heat constituted in different industries. Biological contamination with microorganism accumulation on wet surfaces is one of the major trouble in cooling water systems. Biological contamination of these systems gives rise to blockage of condenser pipes and heat exchanger pipes. Okutsu et al. examined the event between QS and biofouling in the cooling water system. These researchers took 192 bacterial strains from the five cooling water systems and monitored for AHL formation. AHLs extract samples were made a decision as Aeromonas hydrophila, Lysobacter sp., Methylobacterium oryzae and Bosea massiliensis. AHLs formed by B. Massiliensis as follows: C6-HSL, 3-oxo-C6-HSL and 3-oxo-C8-HSL. AHLs occurred by Lysobacter sp. were assigned as C10-HSL and 3-oxo-C10-HSL. Samples were defined with liquid chromatography–mass spectrometry (LC–MS) (Okutsu et al. 2016). The formation of AHLs in membrane bioreactors remeding wastewater has paid much attention as AHLs have been presented to play critical role in biofouling (Huang et al. 2020). AHL-mediated QS mechanisms are responsible for occurrence of biofilm, swarming motility (Labbate et al. 2004) and the formation of microbial by-products, which ensure architectural assistance for biofilms in membrane bioreactor (Tan et al. 2014). C6-HSL, C8-HSL, 3-oxo-C8-HSL, C10-HSL and C12-HSL are universally diagnosed in membrane bioreactor wastewater treatment systems (Yeon et al. 2009; Oh et al. 2012; Kim et al. 2013; Yu et al. 2016a, b, 2019; Liu et al. 2019).
Lately, QS has known significant acts in biologically wastewater treatment operations. Honda et al. studied to research the variety of AHL molecules both biological wastewater treatment procedure and their places (Honda et al. 2019). They tested and measured 10 AHLs in activated sludge samples at wastewater treatment factories. For this aim, these researchers were used Fourier transform mass spectrometry (FTMS) and bioassay on TLC. Determination of AHL via LC-FTMS was found much more responsive than TLC experiments. The sludge samples were gathered at seven wastewater treatment plants in Japan and Thailand, which were selected to check against different process kinds containing a pilot-scale membrane bioreactor procedure. As a result, they identified C4-HSL, C7-HSL and C8-HSL kinds of AHL in the samples (Honda et al. 2019). Increasing in cell density after communication of bacteria among themselves is shown in Fig. 5.
Prevention of production of quorum sensing molecules
Tatedo et al. observed in their study on P.aeruginosa isolates that azithromycin affects gene regions responsible for bacterial communication, thereby reducing signal molecules and preventing elastase gene expression. It has also been found that a molecule called triclosan inhibits an enzyme responsible for signal molecule synthesis (Tateda et al. 1996).
Degradation of N-acyl homoserine lactone signal molecule or inhibition
Degradation of the synthesized N-acyl homoserine lactone molecules may be another target. This mechanism is provided by either enzymes or some inhibitors. The signal molecules of Gram negatives can be destroyed by AHL lactonase and AHL acylase. The signal molecules of Gram positives can be destroyed by some inhibitors. It has been reported that phenolic inhibitors “closantel” and “RWJ-49815” disrupt protein metabolism by inhibiting histidine kinase (Zhang and Dong 2004).
Prevention of uptake of the lactone signal molecule of N-acyl homoserine
Blocking the synthesis of signal receptor proteins or reducing the binding of these proteins to the receptor by AHL analogs can also be applied as a separate mechanism to inhibit the reception of the majority sense signal. It has been reported that the furanone compounds produced by Delisea pulchra, a red macro algae and stored in its vesicles, are structurally linked to the LuxR protein, which is an AHL signal analogue, leading to the release of AHL and thus inhibiting the bacterial environment detection system of V. fisheri (Rasmussen and Givskov 2006).
Conclusion
Researches conducted in the past years have been provided an important perception of cell-to-cell communication and cooperation systems. AHLs in the QS systems are bacterial signal molecules. One of the most significant conditions for defining the QS system is the presence of AHLs in the environment. That's why it is extremely important to get to know AHLs. Many bacterial events can be overcome by understanding the QS system.
The recent researches have suggested that knowing the density of the QS can offer many advantages. The purpose of this review is to emphasize the importance of QS-based AHLs as QS affects health, agriculture and environment. Having knowledge of QS-mediated AHL systems are extremely critical for future studies in many areas, such as health, food, agriculture and industry. Thus, we are of the opinion that this review will attract attention in related areas.
References
Albuquerque P, Casadevall A (2012) Quorum sensing in fungi a review. Med Mycol 50:337–345
Arnaut-Rollier I, De Zutter L, Van Hoof J (1999) Identities of the Pseudomonas spp. in flora from chilled chicken. Int J Food Microbiol 48:87–96
Baker-Austin C, Oliver JD (2018) Vibrio vulnificus: new insights into a deadly opportunistic pathogen. Environ Microbiol 20:423–430
Borch E, Kant-Muermans M-L, Blixt Y (1996) Bacterial spoilage of meat and cured meat products. Int J Food Microbiol 33:103–120
Bruhn JB, Christensen AB, Flodgaard LR et al (2004) Presence of acylated homoserine lactones (AHLs) and AHL-producing bacteria in meat and potential role of AHL in spoilage of meat. Appl Environ Microbiol 70:4293–4302
Cabello FC (2006) Heavy use of prophylactic antibiotics in aquaculture: a growing problem for human and animal health and for the environment. Environ Microbiol 8:1137–1144
Cámara M, Williams P, Hardman A (2002) Controlling infection by tuning in and turning down the volume of bacterial small-talk. Lancet Infect Dis 2:667–676
Cataldi TRI, Bianco G, Palazzo L, Quaranta V (2007) Occurrence of N-acyl-l-homoserine lactones in extracts of some Gram-negative bacteria evaluated by gas chromatography-mass spectrometry. Anal Biochem. https://doi.org/10.1016/j.ab.2006.11.037
Cepas V, López Y, Muñoz E et al (2019) Relationship between biofilm formation and antimicrobial resistance in gram-negative bacteria. Microb Drug Resist. https://doi.org/10.1089/mdr.2018.0027
Chambers CE, Visser MB, Schwab U, Sokol PA (2005) Identification of N-acylhomoserine lactones in mucopurulent respiratory secretions from cystic fibrosis patients. FEMS Microbiol Lett. https://doi.org/10.1016/j.femsle.2005.01.055
Chang CY, Krishnan T, Wang H et al (2014) Non-antibiotic quorum sensing inhibitors acting against N-acyl homoserine lactone synthase as druggable target. Sci Rep. https://doi.org/10.1038/srep07245
Cuadrado-Silva CT, Castellanos L, Arévalo-Ferro C, Osorno OE (2013) Detection of quorum sensing systems of bacteria isolated from fouled marine organisms. Biochem Syst Ecol. https://doi.org/10.1016/j.bse.2012.09.010
Davies JD and Department D (2010) Origins and evolution of antibiotic resistance Julian. Microbiol Mol Biol Rev. https://doi.org/10.1128/MMBR.00016-10
Davis BM, Jensen R, Williams P, O’Shea P (2010) The interaction of N-acylhomoserine lactone quorum sensing signaling molecules with biological membranes: implications for inter-kingdom signaling. PLoS ONE. https://doi.org/10.1371/journal.pone.0013522
Dcosta VM, King CE, Kalan L et al (2011) Antibiotic resistance is ancient. Nature 2:2
De Kievit TR, Iglewski BH (2000) Bacterial quorum sensing in pathogenic relationships. Infect Immun 68:4839–4849
de la Fuente M, Miranda CD, Jopia P et al (2015) Growth inhibition of bacterial fish pathogens and quorum-sensing blocking by bacteria recovered from Chilean salmonid farms. J Aquat Anim Health. https://doi.org/10.1080/08997659.2014.1001534
Defoirdt T (2018) Quorum-sensing systems as targets for antivirulence therapy. Trends Microbiol 26:313–328
Defoirdt T, Thanh LD, Van Delsen B et al (2011) N-acylhomoserine lactone-degrading Bacillus strains isolated from aquaculture animals. Aquaculture. https://doi.org/10.1016/j.aquaculture.2010.11.046
Derome N, Gauthier J, Boutin S, Llewellyn M (2016) Bacterial opportunistic pathogens of fish
Ducel G, Fabry J (2002) Prevention of hospital-acquired infections: A practical guide, 2nd edn. World Health Organization, Geneva
Erdönmez D, Rad AY, Aksöz N (2017) Quorum sensing molecules production by nosocomial and soil isolates Acinetobacter baumannii. Arch Microbiol. https://doi.org/10.1007/s00203-017-1408-8
Falcão JP, Sharp F, Sperandio V (2004) Cell-to-cell signaling in intestinal pathogens. Curr Issues Intest Microbiol
Gennari M, Dragotto F (1992) A study of the incidence of different fluorescent Pseudomonas species and biovars in the microflora of fresh and spoiled meat and fish, raw milk, cheese, soil and water. J Appl Bacteriol 72:281–288
Ghafur A (2014) Emergence of pan-drug resistance amongst gram negative bacteria! The First case series from India. J Microbiol Infect Dis. https://doi.org/10.5799/ahinjs.02.2014.03.0145
Gopu V, Shetty PH (2016) Regulation of acylated homoserine lactones (AHLs) in beef by spice marination. J Food Sci Technol 53:2686–2694
Gui M, Liu L, Wu R et al (2018) Detection of new quorum sensing N-acyl homoserine lactones from aeromonas veronii. Front Microbiol. https://doi.org/10.3389/fmicb.2018.01712
Habimana JD, Ji J, Pi F, et al (2018) A class-specific artificial receptor-based on molecularly imprinted polymer-coated quantum dot centers for the detection of signaling molecules, N-acyl-homoserine lactones present in gram-negative bacteria. Analytica Chimica Acta doi: https://doi.org/10.1016/j.aca.2018.05.018
Holmström K, Gräslund S, Wahlström A et al (2003) Antibiotic use in shrimp farming and implications for environmental impacts and human health. Int J Food Sci Technol. https://doi.org/10.1046/j.1365-2621.2003.00671.x
Honda R, Phan PT, Tobino T et al (2019) Diversity of N-acyl homoserine lactones in activated sludge detected by Fourier transform mass spectrometry. NPJ Clean Water. https://doi.org/10.1038/s41545-019-0035-0
Hou HM, Zhu YL, Wang JY et al (2017) Characteristics of N-Acylhomoserine lactones produced by Hafnia alvei H4 isolated from spoiled instant sea cucumber. Sensors (switzerland). https://doi.org/10.3390/s17040772
Huang J, Shi Y, Zeng G et al (2016) Acyl-homoserine lactone-based quorum sensing and quorum quenching hold promise to determine the performance of biological wastewater treatments: An overview. Chemosphere 157:137–151
Huang S, Zhang H, Ng TCA et al (2020) Analysis of N-Acy-L-homoserine lactones (AHLs) in wastewater treatment systems using SPE-LLE with LC-MS/MS. Water Res 177:115756
Jacobi CA, Bach A, Eberl L et al (2003) Detection of N-(3-oxohexanoyl)-l-homoserine lactone in mice infected with yersinia enterocolitica serotype O8. Infect Immun. https://doi.org/10.1128/IAI.71.11.6624-6626.2003
Jamuna Bai A (2016) Effect of small chain N acyl homoserine lactone quorum sensing signals on biofilms of food-borne pathogens. J Food Sci Technol 53:3609
Jay JM, Vilai JP, Hughes ME (2003) Profile and activity of the bacterial biota of ground beef held from freshness to spoilage at 5–7 C. Int J Food Microbiol 81:105–111
Jefferson KK (2004) What drives bacteria to produce a biofilm?—Jefferson - 2006 - FEMS Microbiology Letters - Wiley Online Library. FEMS Microbiol. Lett.
Jiang H, Jiang D, Shao J, Sun X (2016) Magnetic molecularly imprinted polymer nanoparticles based electrochemical sensor for the measurement of Gram-negative bacterial quorum signaling molecules (N-acyl-homoserine-lactones). Biosens Bioelectron. https://doi.org/10.1016/j.bios.2015.07.045
Jiang D, Liu Y, Jiang H et al (2018) A novel screen-printed mast cell-based electrochemical sensor for detecting spoilage bacterial quorum signaling molecules (N-acyl-homoserine-lactones) in freshwater fish. Biosens Bioelectron. https://doi.org/10.1016/j.bios.2017.11.040
Kalia VC, Patel SKS, Kang YC, Lee JK (2019) Quorum sensing inhibitors as antipathogens: biotechnological applications. Biotechnol Adv 37:68–90
Kaper JB, Sperandio V (2005) Bacterial cell-to-cell signaling in the gastrointestinal tract. Infect Immun 2:3197–3209
Karunasagar I, Pai R, Malathi GR, Karunasagar I (1994) Mass mortality of Penaeus monodon larvae due to antibiotic-resistant Vibrio harveyi infection. Aquaculture. https://doi.org/10.1016/0044-8486(94)90309-3
Kawaguchi T, Yung PC, Norman RS, Decho AW (2008) Rapid screening of quorum-sensing signal N-acyl homoserine lactones by an in vitro cell-free assay. Appl Environ Microbiol. https://doi.org/10.1128/AEM.02869-07
Kendall MM, Sperandio V (2007) Quorum sensing by enteric pathogens. Curr Opin Gastroenterol 23:10–15
Khajanchi BK, Kirtley ML, Brackman SM, Chopra AK (2011) Immunomodulatory and protective roles of quorum-sensing signaling molecules N-Acyl homoserine lactones during infection of mice with Aeromonas hydrophila. Infect Immun. https://doi.org/10.1128/IAI.00096-11
Kim S-R, Oh H-S, Jo S-J et al (2013) Biofouling control with bead-entrapped quorum quenching bacteria in membrane bioreactors: physical and biological effects. Environ Sci Technol 47:836–842
Kjelleberg S, Molin S (2002) Is there a role for quorum sensing signals in bacterial biofilms? Curr Opin Microbiol 5:254–258
Kraft AA (1992) Spoilage of dairy products, vegetables, fruits and other foods. Psychrotrophic Bacteria Foods Dis Spoil 2:113–120
Kravchenko VV, Kaufmann GF, Mathison JC et al (2006) N-(3-oxo-acyl)homoserine lactones signal cell activation through a mechanism distinct from the canonical pathogen-associated molecular pattern recognition receptor pathways. J Biol Chem. https://doi.org/10.1074/jbc.M606613200
Kumari A, Pasini P, Daunert S (2008) Detection of bacterial quorum sensing N-acyl homoserine lactones in clinical samples. Anal Bioanal Chem 5:1619–1627
Kušar D, Šrimpf K, Isaković P et al (2016) Determination of N-acylhomoserine lactones of Pseudomonas aeruginosa in clinical samples from dogs with otitis externa. BMC Vet Res. https://doi.org/10.1186/s12917-016-0843-0
Labbate M, Queck SY, Koh KS et al (2004) Quorum sensing-controlled biofilm development in Serratia liquefaciens MG1. J Bacteriol 186:692–698
Lade H, Paul D, Kweon JH (2014) Quorum quenching mediated approaches for control of membrane biofouling. Int J Biol Sci 2014:1–25
Li YH, Tian X (2012) Quorum sensing and bacterial social interactions in biofilms. Sensors 12:2519–2538
Liu M, Gray JM, Griffiths MW (2006) Occurrence of proteolytic activity and N-acyl-homoserine lactone signals in the spoilage of aerobically chill-stored proteinaceous raw foods. J Food Prot 69:2729–2737
Liu S, Gunawan C, Barraud N et al (2016) Understanding, monitoring, and controlling biofilm growth in drinking water distribution systems. Environ Sci Technol 50:8954–8976
Liu J, Eng CY, Ho JS et al (2019) Quorum quenching in anaerobic membrane bioreactor for fouling control. Water Res 156:159–167
Ma H, Wang X, Zhang Y et al (2018) The diversity, distribution and function of N-acyl-homoserine lactone (AHL) in industrial anaerobic granular sludge. Biores Technol. https://doi.org/10.1016/j.biortech.2017.09.043
Medina-Martinez MS, Uyttendaele M, Demolder V, Debevere J (2006) Influence of food system conditions on N-acyl-l-homoserine lactones production by Aeromonas spp. Int J Food Microbiol 112:244–252
Middleton B, Rodgers HC, Cámara M et al (2002) Direct detection of N-acylhomoserine lactones in cystic fibrosis sputum. FEMS Microbiol Lett. https://doi.org/10.1016/S0378-1097(01)00556-0
Miller MB, Bassler BL (2002) Quorum sensing in bacteria. Annu Rev Microbiol. https://doi.org/10.1146/annurev.micro.55.1.165
Niu Y, Defoirdt T, Baruah K et al (2014) Bacillus sp LT3 improves the survival of gnotobiotic brine shrimp (Artemia franciscana) larvae challenged with Vibrio campbellii by enhancing the innate immune response and by decreasing the activity of shrimp-associated vibrios. Vet Microbiol. https://doi.org/10.1016/j.vetmic.2014.08.007
Oh H-S, Yeon K-M, Yang C-S et al (2012) Control of membrane biofouling in MBR for wastewater treatment by quorum quenching bacteria encapsulated in microporous membrane. Environ Sci Technol 46:4877–4884
Okutsu N, Morohoshi T, Xie X et al (2016) Characterization of N-acylhomoserine lactones produced by bacteria isolated from industrial cooling water systems. Sensors (switzerland). https://doi.org/10.3390/s16010044
Pinto UM, de Souza VE, Martins ML, Vanetti MCD (2007) Detection of acylated homoserine lactones in gram-negative proteolytic psychrotrophic bacteria isolated from cooled raw milk. Food Control 18:1322–1327. https://doi.org/10.1016/j.foodcont.2006.09.005
Rasmussen TB, Givskov M (2006) Quorum-sensing inhibitors as anti-pathogenic drugs. Int J Med Microbiol 296:149–161
Ravn L, Christensen AB, Molin S et al (2001) Methods for detecting acylated homoserine lactones produced by Gram-negative bacteria and their application in studies of AHL-production kinetics. J Microbiol Methods. https://doi.org/10.1016/S0167-7012(01)00217-2
Schauder S, Bassler BL (2001) The languages of bacteria. Gen Dev 15:1468–1480
Schikora A, Schenk ST, Stein E et al (2011) N-acyl-homoserine lactone confers resistance toward biotrophic and hemibiotrophic pathogens via altered activation of AtMPK6. Plant Physiol. https://doi.org/10.1104/pp.111.180604
Shrout JD, Nerenberg R (2012) Monitoring bacterial twitter: Does quorum sensing determine the behavior of water and wastewater treatment biofilms? Environ Sci Technol 46:1995–2005
Sircili MP, Walters M, Trabulsi LR, Sperandio V (2004) Modulation of enteropathogenic Escherichia coli virulence by quorum sensing. Infect Immun 72:2329–2337. https://doi.org/10.1128/IAI.72.4.2329-2337.2004
Soares JA, Ahmer BMM (2011) Detection of acyl-homoserine lactones by Escherichia and Salmonella. Curr Opin Microbiol 14:188–193
Sokol PA, Sajjan U, Visser MB et al (2003) The CepIR quorum-sensing system contributes to the virulence of Burkholderia cenocepacia respiratory infections. Microbiology. https://doi.org/10.1099/mic.0.26540-0
Sperandio V, Mellies JL, Nguyen W et al (1999) Quorum sensing controls expression of the type III secretion gene transcription and protein secretion in enterohemorrhagic and enteropathogenic Escherichia coli. Proc Natl Acad Sci USA 96:15196–15201. https://doi.org/10.1073/pnas.96.26.15196
Stanbridge LH, Davies AR (1998) The microbiology of chill-stored meat. Microbiol Meat Poultry 174–219
Surette MG, Miller MB, Bassler BL (1999) Quorum sensing in Escherichia coli, Salmonella typhimurium, and Vibrio harveyi: A new family of genes responsible for autoinducer production. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.96.4.1639
Tamma PD, Putcha N, Suh YD et al (2011) Does prolonged β-lactam infusions improve clinical outcomes compared to intermittent infusions? A meta-analysis and systematic review of randomized, controlled trials. BMC Infect Dis. https://doi.org/10.1186/1471-2334-11-181
Tamma PD, Holmes A, Ashley ED (2014) Antimicrobial stewardship: another focus for patient safety? Curr Opin Infect Dis 27:348–355
Tamma PD, Miller MA, Cosgrove SE (2019) Rethinking how antibiotics are prescribed. JAMA. https://doi.org/10.1001/jama.2018.19509
Tan CH, Koh KS, Xie C et al (2014) The role of quorum sensing signalling in EPS production and the assembly of a sludge community into aerobic granules. ISME J 8:1186–1197
Tateda K, Ishii Y, Matsumoto T et al (1996) Direct evidence for antipseudomonal activity of macrolides: Exposure- dependent bactericidal activity and inhibition of protein synthesis by erythromycin, clarithromycin, and azithromycin. Antimicrob Agents Chemother. https://doi.org/10.1128/aac.40.10.2271
Ternström A, Lindberg A-M, Molin G (1993) Classification of the spoilage flora of raw and pasteurized bovine milk, with special reference to Pseudomonas and Bacillus. J Appl Bacteriol 75:25–34
Tryfinopoulou P, Tsakalidou E, Nychas G-J (2002) Characterization of Pseudomonas spp. associated with spoilage of gilt-head sea bream stored under various conditions. Appl Environ Microbiol 68:65–72
Turan NB, Chormey DS, Büyükpınar Ç et al (2017) Quorum sensing: little talks for an effective bacterial coordination. TrAC Trends Anal Chem 91:1–11
Wang J, Ding L, Li K et al (2017) Development of an extraction method and LC–MS analysis for N-acylated-L-homoserine lactones (AHLs) in wastewater treatment biofilms. J Chromatogr B. https://doi.org/10.1016/j.jchromb.2016.11.029
Ward C, Cámara M, Forrest I et al (2003) Preliminary findings of quorum signal molecules in clinically stable lung allograft recipients. Thorax 58:444–446. https://doi.org/10.1136/thorax.58.5.444
Waters CM, Bassler BL (2005) QUORUM SENSING: cell-to-cell communication in bacteria. Annu Rev Cell Dev Biol. https://doi.org/10.1146/annurev.cellbio.21.012704.131001
Watson WT, Minogue TD, Val DL et al (2002) Structural basis and specificity of acyl-homoserine mactone signal production in bacterial quorum sensing. Mol Cell. https://doi.org/10.1016/S1097-2765(02)00480-X
Wen KY, Cameron L, Chappell J, et al (2017) A cell-free biosensor for detecting quorum sensing molecules in P. aeruginosa-infected respiratory samples. ACS Synthetic Biology. doi: https://doi.org/10.1021/acssynbio.7b00219
WHO , World Health Organization (2017) Antibacterial agents in clinical development: an analysis of the antibacterial clinical development pipeline, including tuberculosis (WHO/EMP/IAU/2017.11)
Wilson APR, Livermore DM, Otter JA et al (2016) Prevention and control of multi-drug-resistant Gram-negative bacteria: Recommendations from a Joint Working Party. J Hospital Infect 92:S1–S44
Wu H, Song Z, Hentzer M et al (2000) Detection of N-acylhomoserine lactones in lung tissues of mice infected with Pseudomonas aeruginosa. Microbiology. https://doi.org/10.1099/00221287-146-10-2481
Yan X, Zheng S, Huo Z et al (2020) Effects of exogenous N-acyl-homoserine lactones on nutrient removal, sludge properties and microbial community structures during activated sludge process. Chemosphere. https://doi.org/10.1016/j.chemosphere.2020.126945
Yekani M, Baghi HB, Sefidan FY et al (2018) The rates of quinolone, trimethoprim/sulfamethoxazole and aminoglycoside resistance among Enterobacteriaceae isolated from urinary tract infections in Azerbaijan, Iran. GMS Hyg Infect Control. https://doi.org/10.3205/dgkh000313
Yeon KM, Cheong WS, Oh HS et al (2009) Quorum sensing: a new biofouling control paradigm in a membrane bioreactor for advanced wastewater treatment. Environ Sci Technol. https://doi.org/10.1021/es8019275
Yu H, Liang H, Qu F et al (2016a) Biofouling control by biostimulation of quorum-quenching bacteria in a membrane bioreactor for wastewater treatment. Biotechnol Bioeng 113:2624–2632
Yu H, Xu G, Qu F et al (2016b) Effect of solid retention time on membrane fouling in membrane bioreactor: from the perspective of quorum sensing and quorum quenching. Appl Microbiol Biotechnol 100:7887–7897
Yu H, Lee K, Zhang X, Choo K-H (2019) Core-shell structured quorum quenching beads for more sustainable anti-biofouling in membrane bioreactors. Water Res 150:321–329
Zhang LH, Dong YH (2004) Quorum sensing and signal interference: diverse implications. Mol Microbiol 53:1563–1571
Zhao J, Chen M, Quan CS, Fan SD (2015) Mechanisms of quorum sensing and strategies for quorum sensing disruption in aquaculture pathogens. J Fish Dis. https://doi.org/10.1111/jfd.12299
Acknowledgements
The authors are thankful for grants of the Scientific and Technological Research Council of Turkey (Grant Number: 119Z184) and Aksaray University Scientific Research Projects Coordination (Grant Number: 2018/060). Because the information obtained for preparation of this paper was gathered during these projects studies.
Author information
Authors and Affiliations
Contributions
ÖA: conceptualization, writing review and editing. DE: conceptualization, writing review and editing. BÖA: conceptualization, writing review and editing. MO: conceptualization, writing review and editing. All the authors agreed to submit the manuscript.
Corresponding author
Ethics declarations
Conflict of interest statement
Nothing declared.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Acet, Ö., Erdönmez, D., Acet, B.Ö. et al. N-acyl homoserine lactone molecules assisted quorum sensing: effects consequences and monitoring of bacteria talking in real life. Arch Microbiol 203, 3739–3749 (2021). https://doi.org/10.1007/s00203-021-02381-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-021-02381-9