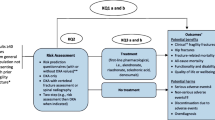
Abstract
Summary
This scoping review described the use, effectiveness, and cost-effectiveness of clinical fracture-risk assessment tools to prevent future osteoporotic fractures among older adults. Results show that the screening was not superior in preventing all osteoporosis-related fractures to usual care. However, it positively influenced participants’ perspectives on osteoporosis, may have reduced hip fractures, and seemed cost-effective.
Purpose
We aim to provide a synopsis of the evidence about the use of clinical fracture-risk assessment tools to influence health outcomes, including reducing future osteoporotic fractures and their cost-effectiveness.
Methods
We followed the guidelines of Arksey and O’Malley and their modifications. A comprehensive search strategy was created to search CINAHL, Medline, and Embase databases until June 29, 2021, with no restrictions. We critically appraised the quality of all included studies.
Results
Fourteen studies were included in the review after screening 2484 titles and 68 full-text articles. Four randomized controlled trials investigated the effectiveness of clinical fracture-risk assessment tools in reducing all fractures among older women. Using those assessment tools did not show a statistically significant reduction in osteoporotic fracture risk compared to usual care; however, additional analyses of two of these trials showed a trend toward reducing hip fractures, and the results might be clinically significant. Four studies tested the impact of screening programs on other health outcomes, and participants reported positive results. Eight simulation studies estimated the cost-effectiveness of using these tools to screen for fractures, with the majority showing significant potential savings.
Conclusion
According to the available evidence to date, using clinical fracture-risk assessment screening tools was not more effective than usual care in preventing all osteoporosis-related fractures. However, using those screening tools positively influenced women’s perspectives on osteoporosis, may have reduced hip fracture risk, and could potentially be cost-effective. This is a relatively new research area where additional studies are needed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis is a bone disease identified by a decrease in bone mineral density resulting from an imbalance between bone formation and bone reabsorption [1]. Osteoporosis places significant health and economic burden on society worldwide. Globally, 18.3% (> 200 million) of people have osteoporosis [2, 3]. In Canada alone, an estimated 2 million individuals are affected by osteoporosis, and at least 1 in 3 women and 1 in 5 men will break a bone due to osteoporosis in their lifetime [4]. One of the most serious osteoporotic fractures is those of the hip. Hip fractures lead to the most morbidity compared to other fracture sites, with mortality rates reported to be 30% in the first year alone after a hip fracture [5]. The current gold standard for diagnosing osteoporosis is measuring bone mineral density (BMD) via dual-energy X-ray absorptiometry (DXA, previously DEXA). Despite its ability to measure bone density accurately and conveniently, DXA has a low sensitivity for predicting osteoporotic fractures, and it is a costly procedure [6]. This low sensitivity led to the development of several assessment tools that use a wide variety of clinical factors to determine fracture risk. These tools include the Fracture Risk Assessment Tool (FRAX), the Canadian Association of Radiologists and Osteoporosis Canada (CAROC) tool, the Garvan institute bone fracture risk calculator (GARVAN), and the Simple Calculated Osteoporosis Risk Estimation (SCORE). Among these tools, the FRAX appears to be the most cited in the literature and the most widely used tool to predict fracture risk [7]. The FRAX uses clinical risk factors to calculate the absolute 10-year risk of hip fracture or other major osteoporosis-related fractures. Examples of clinical risk factors include body mass index (BMI), alcohol intake, and other secondary conditions, such as rheumatoid arthritis. Although not necessary, BMD can also be included in the FRAX calculation.
The ability of the FRAX to predict osteoporotic fractures has been validated in several places in the world, including North America, through several large-scale studies [8, 9]. After identifying those at risk, healthcare providers could then follow up with more in-depth assessments and implement treatment protocols to increase the health outcomes of those at risk for an osteoporotic fracture. However, despite the predictive value of the FRAX, we do not know whether implementing risk assessment tools can translate into clinically significant outcomes, such as reducing osteoporotic fractures or saving costs, and there is seemingly less research dedicated to determining this. To address this research gap, the primary objective of our study was to review the evidence on the effectiveness of using clinical fracture-risk assessment tools in reducing osteoporotic fractures and/or influencing other health outcomes. A secondary objective was to summarize any evidence of the cost-effectiveness of using these clinical tools during investigations in fracture prevention.
Methods
A scoping review directed by the guidelines of Arksey and O’Malley (2005) and its modifications [10] was carried out to systematically search the peer-reviewed literature. The scoping review methodology was selected to provide an overview of any available evidence on our research question. The updated PRISMA reporting guidelines for scoping reviews were followed [11].
Inclusion and exclusion criteria
We included primary studies relating to fracture risk assessment using a validated screening tool in preventing osteoporosis-related fractures or any other patient health outcomes in all settings and populations. All validated clinical risk assessment tools were included, such as FRAX [12], GARVAN [13], and CAROC [14]. Both qualitative and quantitative studies that contributed relevant information were included. Within our search criteria, we excluded articles older than the year 2000 because the validated clinical risk assessment tools were created after this date, as well as conference proceedings, articles without data, and dissertations. Screening studies not focusing on the value of validated clinical risk assessment tools have been excluded.
Search strategy
With the assistance of a health librarian, a list of combinations of keywords and medical sub-headings was created, and three major databases: Medline, Embase, and CINAHL, were searched until June 29, 2021. In addition, we scanned the references of potential studies to identify any additional relevant articles that did not appear in our initial search. A post hoc search of Medline was done to extend the search until November 06, 2022, to ensure no additional relevant articles were published but no additional articles were included as a result of this additional step. The full search strategy and a list of the keywords used in the Embase search can be found in Appendix 1.
Screening of articles
All abstracts were independently reviewed by two different members of the research team. Any disagreements over inclusion were resolved through consensus and, where necessary, discussion with a third member of the review team. Following the abstract review, this process was replicated to complete the full-article review.
Data extraction and synthesis
Data extraction forms were validated by all members of the research team and were pilot tested in three studies for feasibility and comprehensiveness. Minor adjustments were made until a consensus was reached among all team members. Two reviewers independently reviewed full-text and extracted data into Microsoft Excel. The data included general information such as study design, countries, outcome measures, and details on results, conclusion, limitations, and implications. Narrative data synthesis was undertaken, and a meta-analysis was not deemed appropriate due to the nature of this review and the data included.
The quality of each study was independently appraised by two different team members using appropriate assessment tools. Specifically, the risk of bias in included randomized controlled trials (RCT)s was assessed using the PEDro scale (Appendix 2). Similarly, the Critical Appraisal Skills Program (CASP) tool was used to evaluate the validity of the qualitative studies (Appendix 3), and the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) was used to assess the quality of reporting published health economic evaluations (Appendix 4).
Results
Our literature searches identified 766 articles from Medline, 2220 articles from Embase, and 523 articles from CINAHL, totaling 3509 articles. After removing duplicates, there was a total of 2484 articles. When reviewing the abstracts, 64 articles were selected for a full-text review. Four additional articles were identified from manually searching relevant reference lists and added to the full-text review. Following the full-text review of the 68 articles, 32 were excluded because they focused on the predictive ability of the tools rather than investigating clinical outcomes such as fracture prevention, and 22 were excluded due to lacking primary data, such as review studies and conference proceedings. Thus, a total of 14 articles were included in the review. Figure 1 presents the PRISMA flow chart of article inclusion.
Study attributes and characteristics of sample
Table 1 outlines the general information of the 14 included articles, which all were published between 2005 and 2020. Most of the studies were conducted in the USA, and several were published in the Osteoporosis International journal. Except for three studies that used the SCORE [15] and Osteoporosis Self-assessment Tool (OST) [1] to assess clinical fracture risk, respectively, 11 studies used the FRAX as a clinical fracture-risk assessment tool.
In terms of the primary outcome of interest, three RCT studies examined the effectiveness of implementing FRAX in reducing future osteoporotic fractures [16,17,18]. One additional trial compared the influence of the SCORE-based screening strategy with other two strategies on osteoporosis treatment initiation, risk-related behaviors, knowledge, and incidence of osteoporosis fractures[19]. Additionally, one of the trials also investigated the impact of using FRAX on anxiety and quality of life [16].
Two qualitative studies were included; one study evaluated women’s experiences and perspectives on screening for osteoporosis [20]. The other qualitative study examined whether osteoporosis screening by FRAX would affect women’s decision-making on modifying bone health risk factors and treatment of osteoporosis [21].
The remaining eight studies provided health economic analyses to investigate the value for money of using different fracture risk screening strategies [1, 15, 22,23,24,25,26,27].
Primary outcomes
Fracture prevention
Four trials used clinical fracture-risk assessment tools (FRAX and SCORE) to screen and prevent osteoporosis-related fractures among older women. They determined that including these tools in the osteoporosis screening process may not yield significant health outcomes, such as reducing fractures, but is an important step in the comprehensive screening process [16,17,18,19] (Table 2).
Specifically, three studies utilized the FRAX to determine the 10-year probability of sustaining an osteoporotic fracture and measured the proportion of participants who had at least one osteoporosis-related fracture during follow-up. All three studies found that using FRAX in osteoporosis risk selection or in treatment thresholds did not reduce the incidence of all osteoporosis-related fractures compared to usual care. The FRAX-based screening program did, however, significantly reduce the incidence of hip fractures (p = 0.002) in the Shepstone study, which was a secondary outcome [16]. Rubin et al., based on the per-protocol analyses, found that the screening group with FRAX followed by DXA had a significant reduction of all fractures as compared to the control group when only FRAX > 15% [17].
The findings of these pragmatic RCTs might have been compromised by potential selection bias, medication nonadherence in the screening groups, as well as higher than expected uptake of screening and medication in the control groups. For instance, in Merlijin et al., the participants in the screening sub-group were younger and healthier than the control participants since those who were of higher age, cigarette and alcohol consumers, and with more comorbidities were either not interested in DXA (12%) or dropped out (17%) in the screening sub-group. They also reported that 31% of the participants with a treatment indication in the screening group did not start taking the prescribed medication by the end of first year.
In terms of the increased uptake in the control group, Shepstone et al. reported that 24% of the screening group received at least one prescription for osteoporosis medication versus 16% of participants in the control group. In Merjilin et al., overall, 20.7% of participants in the screening group reported having antiosteoporosis medication versus 5.3% of the usual care group. Rubin et al. reported that 25% of women in the control group had a DXA scan, compared to 48% in the screening group, and 18% of women in the control group received osteoporosis medication versus 23% in the screening group.
The screening processes were slightly different among the three studies. In Shepstone et al.’s study, the 10-year age-specific hip fracture probability calculated by the FRAX risk algorithm was used to decide whether to recommend a BMD assessment through a dual-energy DXA scan [16]. The 10-year hip fracture probability was then recalculated based on the FRAX-BMD scores. Participants were categorized into age-specific low or high-risk fracture groups using the FRAX-BMD scores. Finally, all participants and their general practitioners (GPs) were informed of the screening results by letters. The participants with a high fracture risk were advised to make an appointment with G.P.s to discuss treatment options. In Rubin et al.’s study, participants with a 10-year probability of major osteoporotic fractures over or equal to 15% were offered a DXA scan. Then participants and their G.P.s received a letter with the results after the DXA and treatment recommendations. Still, the final decision was with the G.P. In Merlijn et al.’s study, the screening included FRAX, DXA, vertebral fracture assessment (VFA), falls, and blood tests. Based on screening results, the participants with a high risk of major osteoporotic fracture were indicated for a personalized anti-osteoporosis medication treatment [18].
Additionally, one trial compared fracture occurrence and other outcomes among women in three screening groups, namely, universal BMD testing, SCORE-based screening, and osteoporotic fracture risk factors-based screening group (no comparison with usual care) [19]. They found that women in the SCORE-based screening group had a significantly higher fracture rate in follow-up compared with the universal BMD testing group.
Overall, all studies concluded that using FRAX or SCORE might not significantly reduce the rate of fractures. Nonetheless, screening tools identify women with a high risk of fracture as well as women who will benefit from additional fracture risk screening.
Additional clinical and health outcomes
The impact of the clinical osteoporosis screening programs on other outcomes such as health perspectives, anxiety, and quality of life, was explored in four studies included in our review [16, 19,20,21]. Supplementary to investigating fracture prevention, the RCT completed by Shepstone et al. also studied the impact of the screening process on the anxiety and quality of life of the women screened [16]. Shepstone et al. (2017) did not find that the screening program improved participants’ quality of life as measured by the EuroQol-5D and the Short Form 12 Health Survey measure and did not reduce anxiety levels measured by the State-Trait-Anxiety Index [16]. LaCroix et al. found that women in the universal BMD testing group were more likely to discuss osteoporosis with healthcare providers and had better knowledge about osteoporosis risk factors than women in the SCORE-based screening and the osteoporotic fracture risk factors-based screening groups [19].
The remaining two studies were qualitative studies that sought to investigate the perspectives and experiences of the women who underwent fracture risk screening programs [20, 21]. The results of the two qualitative studies indicated that fracture risk screening was a positive experience for older women with no reported adverse effects. Rothman et al. found that screening could be used to reassure individuals about their health status and help them acquire information they would not have known otherwise [20]. Similarly, Dunniway et al. found that undergoing a risk assessment for osteoporosis can help motivate women to change their health behaviors and improve health habits [21]. After receiving an abnormal result (either being diagnosed with osteoporosis or receiving a FRAX score that met the threshold for treatment), women demonstrated a 59% increase in calcium intake and a 94% increase in vitamin D intake over a 3-month period.
Secondary outcome: cost-effectiveness and economic evaluation
Eight simulation studies in our review sought to determine the cost-effectiveness of various osteoporotic screening strategies. Except for one study that focused on both men and women, seven focused solely on women in the simulated analysis. With respect to data analysis, three studies used only a Markov model, two studies used both Markov and decision tree model, one study used a decision tree model alone, one study used a discrete-event simulation model, and one used an individual-level state-transition model. In terms of study perspective, five studies reported using a payer’s perspective, one used a national health system perspective, and two adopted a societal perspective with respect to costs. Overall, almost all studies found that different osteoporotic screening strategies using a clinical fracture risk assessment tool were cost-effective in comparison to the no-screening alternative (see Table 3 for features and main findings of the economic evaluation studies).
Nayak et al. examined the cost-effectiveness of three screen strategies compared to a control group of women aged 55 years old and older. The screening strategies include (1) DXA alone, (2) the calcaneal quantitative ultrasonography (QUS) before DXA, and (3) the SCORE tool before DXA [15]. They found that all screening strategies improved quality-adjusted life-years (QALYs) in American women and were cheaper than no screening (screening initiated at age of 65 years and older). They also found that comparing screening initiation ages from 55 to 80, screening at the age of 55 with DXA − 2.5 and rescreening every 5 years was the most cost-effective with an Incremental Cost-Effectiveness Ratio under $ 50,000 per QALY.
Ito and Leslie’s study evaluated the health and economic impact of various fracture prevention strategies for women in rural areas in Manitoba, Canada, with limited access to DXA. The fracture prevention strategies were (1) watchful waiting strategy (no DXA screening and pharmacotherapy after a fracture), (2) FRAX-BMD-based strategy (DXA screening, FRAX-BMD screening and pharmacotherapy), and (3) FRAX-based strategy (FRAX screening and pharmacotherapy) [22]. The results indicated that the watchful waiting strategy was the most costly and less effective than other strategies. Furthermore, for women traveling fewer than 25 miles, the FRAX-BMD-based strategy was preferred as the travel cost was moderate, whereas for individuals who had to travel more than 25 miles to receive a DXA scan, FRAX without DXA followed by a pharmacotherapy proved to be the superior screening strategy in terms of cost.
Walter et al.’s study evaluated the cost-effectiveness of implementing the (1) micro RNAs-based osteoporotic fracture risk assessment (osteomiRtm test), (2) the FRAX, (3) DXA, and (4) no screening strategies in a cohort of Austrian women aged 50 and over [23]. The osteomiRtm is a novel tool for assessing fracture risks based on serum microRNA profiles. Their findings revealed that assessment and monitoring using the osteomiRtm test reduced the incidence of fracture compared with the other comparison groups using FRAX, DXA, or no screening and is more cost-effective than both the FRAX and no screening.
In Soini et al.’s study, (1) the two Proposed Osteoporosis Management (POMs) screening models (FRAX followed by point-of-care pulse-echo ultrasound [PEUS] and DXA and fracture prevention treatment [FPT] as needed) and (2) the Conventional Osteoporosis Management (COM) screening model (FRAX followed by DXA and FPT if needed) were economically evaluated among older women in Finland [26]. They found that POMs resulted in considerable savings as well as similar QALY gain when compared with COM.
Su et al. compared the utility and cost-effectiveness of three osteoporotic fracture screening strategies, including (1) DXA, (2) FRAX with DXA, and (3) QUS with DXA for hip fracture prevention with no screening in a cohort of older Chinese men and women (aged 65 or over) in Hong Kong [27]. They found that all three strategies led to more QALYs than no screening. No screening was more expensive than any other strategy in men at any age and in women aged 70 years or over. Additionally, they found that pre-screening with FRAX followed by DXA for women aged between 65 and 75 years old was the most cost-effective in the high-risk group.
Chandran et al. sought to determine the FRAX-based intervention thresholds (ITs) at which the therapeutic intervention with generic alendronate becomes cost-effective among Singaporean women over 50 years of age [24]. The use of generic alendronate was shown to reduce healthcare costs at the major osteoporotic fracture (MOF) intervention thresholds for women over 65 years. In comparison, hip fracture intervention thresholds were cost-effective between 60 and 65 years, assuming full adherence. Furthermore, they found that administering alendronate was only cost-effective for women above 50 years if the MOF and hip fracture intervention thresholds were 14% and 3.5%, respectively.
Finally, contradicting findings were presented in Kingkaew et al.’s study [1]. They analyzed the cost-effectiveness of three strategies in screening and treating osteoporosis among postmenopausal women in Thailand, namely (1) no screening, (2) DXA screening, and (3) Osteoporosis Self-assessment Tool (OST) with sequential DXA screening. Findings showed that both DXA and OST with sequential DXA screening and treatment strategies were not cost-effective compared to no screening. However, they pointed out that using OST in conjunction with DXA was less expensive than using DXA alone for the younger age group (< 60 years old).
Critical appraisal (divided by design)
The quality assessment of the four included RCTs revealed that trials are of good or fair internal validity as they scored 5/10 in LaCroix et al. [19], 7/10 in Merlijin et al.’s study [18], 6/10 in Shepstone et al. [16], and 5/10 in Rubin et al. [17] on the PEDro scale. Although these studies demonstrated significant strengths, all studies did not conceal allocation, and there was no evidence of study therapists being blinded, as seen in Appendix 2.
The quality of the two qualitative articles was assessed using the CASP tool, and both were determined to be high-quality articles by the assessors, as seen in Appendix 3.
The quality appraisal of the cost analysis studies was overall high. The CHEERS checklist reporting compliance score (a higher score is better) was: 24/28 in three studies, 25/28 in three studies, 26/28 in one study, and 22/28 in one study. The least reported CHEERS checklist items were item #4 (health economic analysis plan), which was reported in two studies, item #21 (approach to engagement with patients and others affected by the study) reported in two studies, and item #25 (effect of engagement with patients and others affected by the study), which was reported in no study (Appendix 4).
Discussion
This scoping review examined whether the clinical fracture risk assessment tools are effective in improving patient outcomes, reducing future fractures and producing other clinically significant outcomes. Among the 14 included studies, four trial studies looked at incident fracture prevention as the primary outcome, and overall, there was insufficient evidence about their value. However, some evidence indicated that questionnaire-based screening is promising in reducing hip fractures in older women, as shown in Rubin et al.’s and Shepstone et al.’s studies when followed with a DXA scan. Additionally, four articles explored other health outcomes, and two of them suggested that FRAX screening can positively change women’s perspectives and health behaviors on osteoporosis treatments and fracture risk modifications without causing an increase in anxiety levels. Finally, eight simulated studies investigated whether using osteoporotic fracture risk screenings and subsequent treatments is cost-effective. Except for one study, all studies suggested that different osteoporotic fracture risk screening strategies were cost-effective and led to increased QALYs in comparison to no screening.
Although the results of our study did not find statistically significant evidence to indicate that using the FRAX is effective at preventing osteoporosis-related fractures at all fracture sites, several factors perhaps diluted the intervention effect. First, given the nature of these pragmatic trials, we cannot rule out the contamination effect in the control group. Simply screening with FRAX might increase awareness of osteoporotic fracture risks. Rubin et al. found that 25% of women in the control group had a DXA scan, compared to 48% in the screening group, and 18% of women in the control group received osteoporosis medication versus 23% in the screening group [17]. Second, compliance with the treatment in the intervention group might limit the intervention effects in these trials. It was reported in the Merlijin et al. study that 31% of the participants with a treatment indication in the intervention group did not even start taking the prescribed medication, and medication adherence reduced over time [18]. Therefore, both increased chances of treatment uptake in the control group and decreased treatment compliance over time could have reduced the intervention’s effectiveness. Vertebral fractures also were not investigated independently in the included studies, which might have contributed to the nonsignificant results.
In Shepstone et al.’ study, the 10-year risk of hip fracture rather than the risk of all osteoporotic fractures was used [16]. Using the hip fracture risk as the screening approach would be more sensitive to predicting and, therefore, better at preventing hip fractures, rather than fractures at other sites. The per-protocol analysis in Rubin et al.’ study yielded a significant hip fracture reduction in the screening sub-group who had DXA scan compared to participants in the control group with FRAX ≥ 15% [17]. However, this finding might have potential selection bias. The participants in the screening sub-group were younger and healthier than the control participants since those of higher age, cigarette and alcohol consumers, and with more comorbidities were either not interested in DXA (12%) or dropped out (17%) in the screening group. Despite the bias, the positive findings from these studies might be clinically relevant. As compared to all osteoporosis-related fractures, those of the hip are the most severe. In addition, to the high mortality rates, research demonstrates that older adults who survive a hip fracture have high rates of disability, are more likely to be admitted to a nursing home and have a poor quality of life [5, 28, 29]. Therefore, steps taken to prevent hip fractures can positively impact those at risk and the healthcare system overall. The research also suggests that using FRAX scores to identify women at risk and subsequently completing a DXA scan may lead to greater fracture prevention compared to using FRAX alone. However, only women who met a certain FRAX score threshold received a DXA scan. Therefore, due to these implications of the 2-step study design, it is challenging to discern whether the greater fracture prevention was due to the utilization of DXA screening or the greater susceptibility of the women.
Four articles explored other health outcomes. The results suggest that screening tools may positively influence women’s health behaviors by encouraging them to obtain information about osteoporosis [20]. Similarly, for individuals who may be at risk of or who have been diagnosed with osteoporosis, abnormal test results can also increase their awareness of the disease [30]. Based on these results, it can be inferred that when women know their personal risk level paired with an understanding of the associated implications and treatment options, they will be more adept at avoiding the negative consequences of the disease. In the article investigating the quality of life, the positive effects of screening were not reflected, however, the study only utilized simple objective measures [16]. Thus, it is possible that the women did notice positive changes in their lives that were not captured by the tools used. The same article also demonstrated that screening does not increase anxiety, which suggests no negative impact of screening. Overall, there is minimal research investigating the impacts of screening on health outcomes, but the research suggests that screening can positively influence health with minimal to no adverse effects.
Various simulation studies have explored the cost-effectiveness of using risk assessment tools compared to more costly methods, such as DXA or X-ray, to assess fracture risk. Although there is insufficient or weak evidence on the use of clinical fracture risk assessment tools such as FRAX, reviewed evidence suggests that using these tools as part of screening strategies is cost-effective compared to no screening. In our included studies, reviewed evidence indicated that in conjunction with osteoporosis medications, different screening strategies with or without a questionnaire-based pre-screening significantly reduced fracture risk-related costs compared with no screening at all. However, the cost-effectiveness of different strategies varied widely under different circumstances. An Incremental Cost-Effectiveness Ratio threshold and age can impact the cost-effectiveness of screening strategies. Nayak et al. found that pre-screened by SCORE followed a DXA − 2.5 cost less than DXA screening alone at ages between 55 to 65, assuming Incremental Cost-Effectiveness Ratios less than $20,000 per QALY, while DXA − 2.5 alone with rescreening every 5 years were most cost-effective at ages 60 and over assuming a willingness-to-pay $ 100,000 per QALY [15]. Furthermore, the geographic proximity of DXA might be another factor that influences the cost-effectiveness of screening strategies. Due to the travel burden, Ito et al.’s study showed that the FRAX without DXA could be more cost-effective than DXA alone for women with a travel distance ≥ 25 miles, while it was the opposite for women with a travel distance < 25 miles [22]. Of importance to note is that these cost studies used fracture reduction rates data from drug studies and not pragmatic trials of screening tools, and this might have contributed to the high savings estimated in these studies.
The use of FRAX and DXA is the gold standard for fracture risk assessment and is most frequently investigated in the literature for osteoporosis management [7, 31]. One limitation of using DXA is that it is rarely available in many primary care facilities due to cost. Some novel osteoporosis diagnosis tools, such as PEUS and serum microRNA (osteomiR™), seem promising alternatives. Soini and colleagues found that by incorporating PEUS in a conventional screening strategy (FRAX followed DXA), osteoporosis management costs were reduced significantly [26]. This might be because accessing the DXA is expensive, especially in remote areas considering the traveling costs. The cost-utility of osteomiR™ in Walter’s study revealed that compared with using DXA alone or with FRAX alone, osteomiR™ led to increased QALYs and reduced incidence of fractures [23]. They noted that due to the insufficient accuracy of DXA for identifying high fracture risks, osteomiR™ should be included in the standard care to increase the diagnostic performance of osteoporotic fracture risks.
Implications on the healthcare system
This research will serve as foundational knowledge as the global population continues to age, and the number of fractures is projected to increase significantly [32]. A 2015–2016 report from Osteoporosis Canada states that it costs the Canadian Healthcare system approximately 2.3 billion dollars a year to treat osteoporosis and the resulting fractures [33]. Older women are at a higher risk than men for hip fractures; thus, it is appropriate that research to date focused on older women. Each hip fracture alone costs about $21,000 within the first year after hospitalization, and those costs more than double per year if the patient needs to be institutionalized [33]. Therefore, if more fractures could be prevented and at a lower overall cost to the healthcare system by using these tools, there is a significant benefit to increasing the utilization and research surrounding these tools.
Future research directions
Overall, more high-quality pragmatic experimental studies, such as RCTs, are needed to investigate the effectiveness of using clinical risk assessment tools to produce clinically significant outcomes. Specifically, additional research should compare the effectiveness of utilizing the FRAX compared to DXA screening for women with the same fracture risk level and follow participants for longer periods. In addition, much of the current research is focused on FRAX. Therefore, there is also a need for additional research to explore the effectiveness of using the different types of assessment tools (e.g. CAROC, GARVAN). Exploring various tools will allow researchers to determine if they are as effective as the FRAX at preventing fractures or which tool has the most significant impact on fracture prevention. Vertebral fractures should be included as an independent outcome in future studies. Once more trials are available on this topic, a meta-analysis can help pool results from several studies to provide a better conclusion. Finally, future trials should include a cost-analysis component, or simulated cost-analysis studies should use rates from pragmatic trials to improve the cost-saving estimation.
Strengths and limitations
This review has many strengths. First, to the best of our knowledge, this is the first review to summarize evidence on the effectiveness of clinical fracture-risk assessment tools in preventing fractures and producing other clinical outcomes. Second, a comprehensive search strategy was created with the help of an information specialist to ensure a valid strategy was employed. Third, at least two independent reviewers were included throughout the process to bolster the rigor of the results. Finally, we have critically appraised all included studies and found that the majority of included studies have a high level of internal validity (less sources of bias). A limitation of this review is that only studies completed in English were included, so relevant studies in other languages may have been excluded. Lastly, like all literature reviews, our results are limited by the published research available. With more data available, the results might change.
Conclusion
While previous research has been focused on validating clinical risk assessment tools, there has been significantly less research investigating whether utilizing these tools produces tangible clinical outcomes, including fracture prevention. Although based on limited studies, this scoping review suggested that screening with osteoporosis clinical risk assessment tools was not more effective than usual care in preventing the incidence of all osteoporotic fractures. This finding might have resulted from suboptimal participation and adherence in the intervention groups and higher than expected screening and osteoporosis medication uptake in the control groups. However, screening tools could play a role in osteoporotic hip fracture prevention. Also, we found that using these tools positively influenced women’s perspectives on osteoporosis and subsequent lifestyle choices. Our review also suggests that including these tools in the screening has the potential to be a cost-effective approach to preventing osteoporosis-related fractures, especially hip fractures.
References
Kingkaew P, Maleewong U, Ngarmukos C, Teerawattananon Y (2012) Evidence to inform decision makers in Thailand: a cost-effectiveness analysis of screening and treatment strategies for postmenopausal osteoporosis. Value Health 15(1 Suppl):S20-28
Salari N, Ghasemi H, Mohammadi L, Behzadi MH, Rabieenia E, Shohaimi S, Mohammadi M (2021) The global prevalence of osteoporosis in the world: a comprehensive systematic review and meta-analysis. J Orthopaedic Surgery and Research 16(1):1–20
Sozen T, Ozisik L, Basaran NC (2017) An overview and management of osteoporosis. Eur J Rheumatol 4(1):46–56
Government of Canada (2021) Osteoporosis and related fractures in Canada. City.
Leibson CL, Tosteson ANA, Gabriel SE, Ransom JE, Melton LJ (2002) Mortality, disability, and nursing home use for persons with and without hip fracture: a population-based study. J Am Geriatr Soc 50(10):1644–1650
Schuit SC, van der Klift M, Weel AE, de Laet CE, Burger H, Seeman E, Hofman A, Uitterlinden AG, van Leeuwen JP, Pols HA (2004) Fracture incidence and association with bone mineral density in elderly men and women: the Rotterdam Study. Bone 34(1):195–202
Kanis JA, Johansson H, Oden A, Cooper C, McCloskey EV (2014) Epidemiology and Quality of Life Working Group of, I. O. F. Worldwide uptake of FRAX. Arch Osteoporos 9(1):166
Fraser LA, Langsetmo L, Berger C, Ioannidis G, Goltzman D, Adachi JD, Papaioannou A, Josse R, Kovacs CS, Olszynski WP, Towheed T, Hanley DA, Kaiser SM, Prior J, Jamal S, Kreiger N, Brown JP, Johansson H, Oden A, McCloskey E, Kanis JA, Leslie WD, G CaMos Research (2011) Fracture prediction and calibration of a Canadian FRAX(R) tool: a population-based report from CaMos. Osteoporos Int 22(3):829–837
Leslie WD, Lix LM, Johansson H, Oden A, McCloskey E, Kanis JA, P Manitoba Bone Density (2010) Independent clinical validation of a Canadian FRAX tool: fracture prediction and model calibration. J Bone Miner Res 25(11):2350–2358
Levac D, Colquhoun H, O’Brien KK (2010) Scoping studies: advancing the methodology. Implement Sci 5(1):69
Tricco AC, Lillie E, Zarin W, O’Brien KK, Colquhoun H, Levac D, Moher D, Peters MDJ, Horsley T, Weeks L, Hempel S, Akl EA, Chang C, McGowan J, Stewart L, Hartling L, Aldcroft A, Wilson MG, Garritty C, Lewin S, Godfrey CM, Macdonald MT, Langlois EV, Soares-Weiser K, Moriarty J, Clifford T, Tuncalp O, Straus SE (2018) PRISMA Extension for Scoping Reviews (PRISMA-ScR): checklist and explanation. Ann Intern Med 169(7):467–473
Kanis JA, Hans D, Cooper C, Baim S, Bilezikian JP, Binkley N, Cauley JA, Compston JE, Dawson-Hughes B, El-Hajj Fuleihan G, Johansson H, Leslie WD, Lewiecki EM, Luckey M, Oden A, Papapoulos SE, Poiana C, Rizzoli R, Wahl DA, McCloskey EV, FI Task Force of the (2011) Interpretation and use of FRAX in clinical practice. Osteoporos Int 22(9):2395–2411
Garvan Institute of Medical Research (2008) Bone Fracture Risk Calculator. City.
Siminoski K, Leslie WD, Frame H, Hodsman A, Josse RG, Khan A, Lentle BC, Levesque J, Lyons DJ, Tarulli G, Brown JP (2007) Recommendations for bone mineral density reporting in Canada: a shift to absolute fracture risk assessment. J Clin Densitom 10(2):120–123
Nayak S, Roberts MS, Greenspan SL (2011) Cost-effectiveness of different screening strategies for osteoporosis in postmenopausal women. Ann Intern Med 155(11):751–761
Shepstone L, Lenaghan E, Cooper C, Clarke S, Fong-Soe-Khioe R, Fordham R, Gittoes N, Harvey I, Harvey N, Heawood A, Holland R, Howe A, Kanis J, Marshall T, O’Neill T, Peters T, Redmond N, Torgerson D, Turner D, McCloskey E, SS Team (2018) Screening in the community to reduce fractures in older women (SCOOP): a randomised controlled trial. Lancet 391(10122):741–747
Rubin KH, Rothmann MJ, Holmberg T, Hoiberg M, Moller S, Barkmann R, Gluer CC, Hermann AP, Bech M, Gram J, Brixen K (2018) Effectiveness of a two-step population-based osteoporosis screening program using FRAX: the randomized Risk-stratified Osteoporosis Strategy Evaluation (ROSE) study. Osteoporos Int 29(3):567–578
Merlijn T, Swart KM, van Schoor NM, Heymans MW, van der Zwaard BC, van der Heijden AA, Rutters F, Lips P, van der Horst HE, Niemeijer C, Netelenbos JC, Elders PJ (2019) The effect of a screening and treatment program for the prevention of fractures in older women: a randomized pragmatic trial. J Bone Miner Res 34(11):1993–2000
Lacroix AZ, Buist DS, Brenneman SK, Abbott TA 3rd (2005) Evaluation of three population-based strategies for fracture prevention: results of the osteoporosis population-based risk assessment (OPRA) trial. Med Care 43(3):293–302
Rothmann MJ, Huniche L, Ammentorp J, Barkmann R, Gluer CC, Hermann AP (2014) Women’s perspectives and experiences on screening for osteoporosis (Risk-stratified Osteoporosis Strategy Evaluation, ROSE). Arch Osteoporos 9(1):192
Dunniway DL, Camune B, Baldwin K, Crane JK (2012) FRAX(R) counseling for bone health behavior change in women 50 years of age and older. J Am Acad Nurse Pract 24(6):382–389
Ito K, Leslie WD (2015) Cost-effectiveness of fracture prevention in rural women with limited access to dual-energy X-ray absorptiometry. Osteoporos Int 26(8):2111–2119
Walter E, Dellago H, Grillari J, Dimai HP, Hack M (2018) Cost-utility analysis of fracture risk assessment using microRNAs compared with standard tools and no monitoring in the Austrian female population. Bone 108:44–54
Chandran M, Ganesan G, Tan KB, Reginster JY, Hiligsmann M (2021) Cost-effectiveness of FRAX(R)-based intervention thresholds for management of osteoporosis in Singaporean women. Osteoporos Int 32(1):133–144
Martin-Sanchez M, Comas M, Posso M, Louro J, Domingo L, Tebe C, Castells X, Espallargues M (2019) Cost-Effectiveness of the screening for the primary prevention of fragility hip fracture in spain using FRAX((R)). Calcif Tissue Int 105(3):263–270
Soini E, Riekkinen O, Kroger H, Mankinen P, Hallinen T, Karjalainen JP (2018) Cost-effectiveness of pulse-echo ultrasonometry in osteoporosis management. Clinicoecon Outcomes Res 10:279–292
Su Y, Lai FTT, Yip BHK, Leung JCS, Kwok TCY (2018) Cost-effectiveness of osteoporosis screening strategies for hip fracture prevention in older Chinese people: a decision tree modeling study in the Mr. OS and Ms. OS cohort in Hong Kong. Osteoporos Int 29(8):1793–1805
Papaioannou A, Kennedy CC, Ioannidis G, Sawka A, Hopman WM, Pickard L, Brown JP, Josse RG, Kaiser S, Anastassiades T, Goltzman D, Papadimitropoulos M, Tenenhouse A, Prior JC, Olszynski WP, Adachi JD, G CaMos Study (2009) The impact of incident fractures on health-related quality of life: 5 years of data from the Canadian Multicentre Osteoporosis Study. Osteoporos Int 20(5):703–714
Auais M, Morin SN, Finch L, Ahmed S, Mayo N (2018) Toward a meaningful definition of recovery after hip fracture: comparing two definitions for community-dwelling older adults. Arch Phys Med Rehabil 99(6):1108–1115
Denkinger MD, Lukas A, Nikolaus T, Hauer K (2015) Factors associated with fear of falling and associated activity restriction in community-dwelling older adults: a systematic review. Am J Geriatr Psychiatry 23(1):72–86
Lewiecki EM (2005) Clinical applications of bone density testing for osteoporosis. Minerva Med 96(5):317–330
Osteoporosis Canada (2015) Make the FIRST break the LAST with Fracture Liasion Services. City.
Osteoporosis Canada (2016) Better Bone Health: 2015–2016 annual report. City.
Acknowledgements
We would like to acknowledge the support of Ms. Paola Durando MLS, AHIP, Health Sciences Librarian, Queen's University Library, who helped with creating and performing the search strategy. We would like to also thank Yuan Chen and Tiffany Wing Lam for helping in screening articles.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
Not applicable
Conflicts of interest
None.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix 1. Search Strategies for three databases
Medline | |
|---|---|
Search terms | Results |
1 frax.mp | 1484 |
2 garvan.mp | 65 |
3 qfracture.mp | 42 |
4 caroc.mp | 22 |
5 (fracture* adj5 risk assessment*).mp | 1639 |
6 risk assessment*.mp | 327,007 |
7 Risk Assessment/ | 283,558 |
8 1 or 2 or 3 or 4 or 5 or 6 or 7 | 327,557 |
9 osteoporosis/ or osteopSearchorosis, postmenopausal/ | 57,951 |
10 osteoporosis.mp | 91,742 |
11 osteoporotic fracture*.mp | 11,997 |
12 fragility fracture*.mp | 4084 |
13 hip fractures/ or femoral neck fractures/ | 25,509 |
14 hip fracture*.mp | 24,547 |
15 or/9–14 | 120,913 |
16 8 and 15 | 5482 |
17 limit 16 to humans | 5070 |
18 limit 17 to yr = “2000–current” | 4864 |
19 limit 18 to dt = 20,180,701–20,210,629 | 762 |
20 fracture risk scale*.mp | 6 |
21 19 or 20 | 766 |
Embase | |
|---|---|
Search terms | Results |
1 frax.mp | 4017 |
2 garvan.mp | 154 |
3 qfracture.mp | 110 |
4 caroc.mp | 46 |
5 (fracture* adj5 risk assessment*).mp | 3185 |
6 risk assessment*.mp | 651,218 |
7 risk assessment/ | 619,253 |
8 1 or 2 or 3 or 4 or 5 or 6 or 7 | 653,210 |
9 exp osteoporosis/ | 143,291 |
10 osteoporosis.mp | 170,261 |
11 osteoporotic fracture*.mp | 13,188 |
12 fragility fracture*.mp | 22,012 |
13 exp hip fracture/ | 42,826 |
14 hip fracture*.mp | 37,422 |
15 or/9–14 | 208,790 |
16 8 and 15 | 16,518 |
17 limit 16 to human | 15,890 |
18 limit 17 to yr = “2000–current” | 15,084 |
19 limit 18 to dc = 20,180,701–20,210,629 | 4114 |
20 fracture risk scale*.mp | 8 |
21 19 or 20 | 4118 |
22 limit 21 to (books or chapter or conference abstract or conference paper or “conference review” or editorial) | 1898 |
23 21 not 22 | 2220 |
CINAHL | |||
|---|---|---|---|
Search ID# | Search terms | Search options | Results |
S16 | S14 OR S15 | Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 497 |
S15 | “fracture risk scale*” | Limiters—Published Date: 20,180,701–20,210,731; Human Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 1 |
S14 | S8 AND S13 | Limiters—Published Date: 20,180,701–20,210,731; Human Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 497 |
S13 | S9 OR S10 OR S11 OR S12 | Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 33,588 |
S12 | (MH “Hip Fractures”) | Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 10,843 |
S11 | “fragility fracture*” | Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 1,400 |
S10 | (MH “Osteoporotic Fractures”) | Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 629 |
S9 | (MH “Osteoporosis + ”) | Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 23,589 |
S8 | S1 OR S2 OR S3 OR S4 OR S5 OR S6 OR S7 | Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 124,629 |
S7 | “risk assessment” | Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 124,231 |
S6 | (MH “Risk Assessment”) | Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 116,820 |
S5 | “fracture risk assessment*” | Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 376 |
S4 | “caroc” | Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 6 |
S3 | “qfracture” | Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 17 |
S2 | “garvan” | Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 199 |
S1 | “frax” | Expanders—Apply related words; Apply equivalent subjects Search modes—Boolean/Phrase | 523 |
Appendix 2. PEDro Scales completed for the RCT studies
PEDro Criteria | LaCroix, 2005 | Shepstone, 2017 | Rubin, 2017 | Merlijn, 2019 |
|---|---|---|---|---|
1. Eligibility criteria were specified | ✔ | ✔ | ✔ | ✔ |
2. Subjects were randomly allocated to groups (in a crossover study, subjects were randomly allocated an order in which treatments were received) | 1 | 1 | 1 | 1 |
3. Allocation was concealed | 0 | 0 | 0 | 0 |
4. The groups were similar at baseline regarding the most important prognostic indicators | 0 | 1 | 1 | 1 |
5. There was blinding of all subjects | 1 | 0 | 0 | 0 |
6. There was blinding of all therapists who administered the therapy | 0 | 0 | 0 | 0 |
7. There was blinding of all assessors who measured at least one key outcome | 0 | 0 | 0 | 1 |
8. Measures of at least one key outcome were obtained from more than 85% of the subjects initially allocated to groups | 1 | 1 | 0 | 1 |
9. All subjects for whom outcome measures were available received the treatment or control condition as allocated or, where this was not the case, data for at least one key outcome was analyzed by “intention to treat” | 1 | 1 | 1 | 1 |
10. The results of between-group statistical comparisons are reported for at least one key outcome | 1 | 1 | 1 | 1 |
11. The study provides both point measures and measures of variability for at least one key outcome | 0 | 1 | 1 | 1 |
Overall PEDro Score | 5/10 | 6/10 | 5/10 | 7/10 |
Appendix 3. CASP tool for qualitative studies
CASP tool | Dunniway,2010 | Rothman,2014 |
|---|---|---|
Section A: Are the results valid | ||
1. Was there a clear statement of the aims of the research? | Yes | Yes |
2. Is a qualitative methodology appropriate | Yes | Yes |
3. Was the research design appropriate to address the aims of the research? | Yes | Yes |
4. Was the recruitment strategy appropriate to the aims of the research? | Yes | Yes |
5. Was the data collected in a way that addressed the research issue? | Yes | Yes |
6. Has the relationship between researcher and participants been adequately considered | No | Unclear |
Section B: What are the results? | ||
7. Have ethical issues been taken into consideration? | Unclear | Yes |
8. Was the data analysis sufficiently rigorous? | Yes | Yes |
9. Is there a clear statement of findings? | Yes | Yes |
Section C: Will the results help locally? | ||
10. How valuable is the research | Page 7 | Page 8 |
Appendix 4. CHEERS for economical evaluations
CHEERS 2022 Checklist | Studies | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
Item # | Items | Guidance for Reposting | Nayak, 2011 | Kingkaew, 2011 | Ito,2015 | Walter, 2017 | Soini, 2018 | Su, 2018 | Martin-Sanchez, 2019 | Chandra, 2020 |
1 | Title | Identify the study as an economic evaluation and specify the interventions being compared | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
2 | Abstract | Provide a structured summary that highlights context, key methods, results, and alternative analyses | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
3 | Background and objectives | Give the context for the study, the study question, and its practical relevance for decision-making in policy or practice | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
4 | Health economic analysis plan | Indicate whether a health economic analysis plan was developed and where available | Not Reported | Not Reported | Not Reported | Reported | Reported | Not Reported | Not Reported | Not Reported |
5 | Study population | Describe characteristics of the study population (such as age range, demographics, socioeconomic, or clinical characteristics) | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
6 | Setting and location | Provide relevant contextual information that may influence findings | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
7 | Comparators | Describe the interventions or strategies being compared and why chosen | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
8 | Perspective | State the perspective(s) adopted by the study and why chosen | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
9 | Time horizon | State the time horizon for the study and why appropriate | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
10 | Discount rate | Report the discount rate(s) and reason chosen | Reported | Reported | Reported | Reported | Reported | Not Reported | Reported | Reported |
11 | Selection of outcomes | Describe what outcomes were used as the measure(s) of benefit(s) and harm(s) | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
12 | Measurement of outcomes | Describe how outcomes used to capture benefit(s) and harm(s) were measured | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
13 | Valuation of outcomes | Describe the population and methods used to measure and value outcomes | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
14 | Measurement and valuation of resources and costs | Describe how costs were valued | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
15 | Currency, price date, and conversion | Report the dates of the estimated resource quantities and unit costs, plus the currency and year of conversion | Reported | Reported | Reported | Reported | Reported | Not Reported | Reported | Reported |
16 | Rationale and description of model | If modeling is used, describe in detail and why used. Report if the model is publicly available and where it can be accessed | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
17 | Analytics and assumptions | Describe any methods for analyzing or statistically transforming data, any extrapolation methods, and approaches for validating any model used | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
18 | Characterizing heterogeneity | Describe any methods used for estimating how the results of the study vary for sub-groups | Reported | Not Reported | Not Reported | Not Reported | Not Reported | Reported | Reported | Reported |
19 | Characterizing distributional effects | Describe how impacts are distributed across different individuals or adjustments made to reflect priority populations | Reported | Not Reported | Not Reported | Reported | Not Reported | Reported | Reported | Reported |
20 | Characterizing uncertainty | Describe methods to characterize any sources of uncertainty in the analysis | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
21 | Approach to engagement with patients and others affected by the study | Describe any approaches to engage patients or service recipients, the general public, communities, or stakeholders (e.g., clinicians or payers) in the design of the study | Not Reported | Reported | Not Reported | Not Reported | Not Reported | Not Reported | Reported | Not Reported |
22 | Study parameters | Report all analytic inputs (e.g., values, ranges, references) including uncertainty or distributional assumptions | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
23 | Summary of main results | Report the mean values for the main categories of costs and outcomes of interest and summarize them in the most appropriate overall measure | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
24 | Effect of uncertainty | Describe how uncertainty about analytic judgments, inputs, or projections affect findings. Report the effect of choice of discount rate and time horizon, if applicable | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
25 | Effect of engagement with patients and others affected by the study | Report on any difference patient/service recipient, general public, community, or stakeholder involvement made to the approach or findings of the study | Not Reported | Not Reported | Not Reported | Not Reported | Not Reported | Not Reported | Not Reported | Not Reported |
26 | Study findings, limitations, generalizability, and current knowledge | Report key findings, limitations, ethical or equity considerations not captured, and how these could impact patients, policy, or practice | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
27 | Source of funding | Describe how the study was funded and any role of the funder in the identification, design, conduct, and reporting of the analysis | Reported | Reported | Not Reported | Reported | Reported | Reported | Reported | Reported |
28 | Conflicts of interest | Report authors conflicts of interest according to journal or International Committee of Medical Journal Editors requirements | Reported | Reported | Reported | Reported | Reported | Reported | Reported | Reported |
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Auais, M., Angermann, H., Grubb, M. et al. The effectiveness and cost-effectiveness of clinical fracture-risk assessment tools in reducing future osteoporotic fractures among older adults: a structured scoping review. Osteoporos Int 34, 823–840 (2023). https://doi.org/10.1007/s00198-022-06659-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-022-06659-6