Abstract
Summary
This population-based cohort study with a 3-year follow-up revealed that the annual incidence rates of vertebral fracture (VF) and severe VF (sVF) were 5.9%/year and 1.7%/year, respectively. The presence of mild VF at the baseline was a significant risk factor for incident sVF in participants without prevalent sVF.
Introduction
This study aimed to estimate the incidence of morphometric vertebral fracture (VF) and severe VF (sVF) in men and women and clarify whether the presence of a mild VF (mVF) increases the risk of incident sVF.
Methods
Data from the population-based cohort study, entitled the Research on Osteoarthritis/Osteoporosis Against Disability (ROAD) study, were analyzed. In total, 1190 participants aged ≥ 40 years (mean age, 65.0 ± 11.2) years completed whole-spine lateral radiography both at the third (2012–2013, baseline) and fourth surveys performed 3 years later (2015–2016, follow-up). VF was defined using Genant’s semi-quantitative (SQ) method: VF as SQ ≥ 1, mVF as SQ = 1, and sVF as SQ ≥ 2. Cumulative incidence of VF and sVF was estimated. Multivariate logistic regression analyses were performed to evaluate risk factors for incident sVF.
Results
The baseline prevalence of mVF and sVF were 16.8% and 6.0%, respectively. The annual incidence rates of VF and sVF were 5.9%/year and 1.7%/year, respectively. The annual incidence rates of sVF in participants without prevalent VF, with prevalent mVF, and with prevalent sVF were 0.6%/year, 3.8%/year, and 11.7%/year (p < 0.001), respectively. Multivariate logistic regression analyses in participants without prevalent sVF showed that the adjusted odds ratios for incident sVF were 4.12 [95% confident interval 1.85–9.16] and 4.53 [1.49–13.77] if the number of prevalent mVF at the baseline was 1 and ≥ 2, respectively.
Conclusions
The annual incidence rates of VF and sVF were 5.9%/year and 1.7%/year, respectively. The presence of prevalent mVF was an independent risk factor for incident sVF.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Vertebral fracture (VF) is the most common among osteoporotic fractures [1]. It is associated with long-continued back pain, impaired function, and decreased health-related quality of life (HRQoL) [2,3,4]. Furthermore, several studies have reported higher mortality associated with VF [5, 6]. Therefore, preventing VF is imperative for improving life expectancy and maintaining the HRQoL of older people. The first step in preventing VF is to understand its precise epidemiology. However, only about one third of all vertebral deformities noted on radiographs come to medical attention [7]. Hence, research at medical institutions alone do not provide sufficient information; surveying the general population with radiographs is mandatory to understand its epidemiology. Given this complexity, studies on the epidemiology of VF are still limited worldwide. While different prevalences of VF among different ethnic groups have been reported [8], reports on the incidence of VF in Japan are even more limited [9, 10]. Furthermore, these reports were based on studies performed in the mid-1990s, leaving a gap of more than 20 years to date. During the last 20 years, Japanese life expectancy has increased by approximately 5 years [11], probably owing to the escalation of knowledge on health care and the improvement in public health policy. Hence, some improvement has occurred in the epidemiology of VF. We conducted the present study to update the knowledge on the incidence of VF in Japan.
Additionally, risk factors for incident severe VF (sVF) were analyzed, featuring the presence of mild VF (mVF). Prevalent morphometrical VF has been reported as a strong risk factor for incident VF [9, 10, 12, 13]; however, mVF has not been studied sufficiently in the perspective of risk factors for incident sVF. A few studies have reported mVF as a risk factor for incident VF in women [14,15,16], but to the best of our knowledge, no reports exist on mVF as a risk factor in men. Furthermore, mVF has been reported to have low clinical relevance in cross-sectional studies [17, 18]. Thus, the clinical significance of mVF remains unclear. Therefore, using a nationwide, population-based cohort including both men and women, the Research on Osteoarthritis/Osteoporosis Against Disability (ROAD) study, we aimed to estimate the incidence of morphometric VF and sVF in men and women and to analyze whether mVF is a risk factor for sVF.
Methods
Participants
The ROAD study, which commenced in 2005, is a prospective study designed to establish epidemiologic indices for evaluating bone and joint diseases. It consists of population-based cohorts in three communities in Japan: a mountainous region in Hidakagawa, Wakayama; a coastal region in Taiji, Wakayama; and an urban region in Itabashi, Tokyo. Details of the study have been described previously [19, 20]; therefore, only a summary is provided here. In the present study, we investigated the third and fourth surveys of the ROAD study from the mountainous and coastal regions, describing the cohorts in the two regions.
The third survey of the ROAD study, the baseline of the present study, was performed in 2012–2013. Invitation letters to the third survey were distributed to the inhabitants whose names were listed in the former two ROAD study surveys. In addition to former participants, inhabitants aged ≥ 40 years who were willing to participate in the ROAD study were included. New participants were recruited through public relations papers issued by local governments, without any restrictions other than age ≥ 40 years. Throughout the study, the inclusion criteria were the ability to (1) walk to the clinic where the survey was performed, (2) provide self-reported data, and (3) understand and sign an informed consent form. There were no specific exclusion criteria.
Whole-spine radiography was performed from the third survey of the ROAD study onward (2012–2013). Thus, 1486 inhabitants (483 men and 1003 women) participated in the third survey and underwent whole-spine radiography. In the fourth survey of the ROAD study performed in 2015–2016, 3 years after the third survey (follow-up of the present study), we attempted to trace and review these 1486 participants from the third survey. All of them were invited to attend the fourth survey and undergo repeat radiography using the identical method. The present study cohort included all participants aged ≥ 40 years who participated in both the third and fourth surveys and underwent whole-spine radiography.
The study was approved by the ethics committees of the University of Tokyo (nos. 1264 and 1326) and Tokyo Metropolitan Institute of Gerontology (no. 5). Written informed consent was provided by all participants.
Questionnaire, interview, and anthropometric measurements
Participants completed a 400-item interviewer-administered questionnaire that assessed lifestyle characteristics, such as occupation, smoking habits, alcohol consumption, family history, medical history, physical activity, reproductive variables, and HRQoL. Current smokers were defined as those who smoked, regardless of the number of pack-years; never- and ex-smokers were classified as non-smoking. Current habitual alcohol consumption was defined as habitual alcohol consumption at least once a week regardless of the amount; never- and ex-drinkers were classified as non-drinking. Steroidal use was defined as a history of > 3 months of steroidal use. Anthropometric measurements included height and weight, and body mass index (BMI) was calculated (weight [kg]/height2 [m2]). Medical information on participants’ systemic, local, and mental status was obtained by experienced orthopedists.
Radiographic assessment
Stand-up lateral radiographs of the whole spine were obtained for each participant by licensed radiography technicians using a 40-inch film. Films were positioned so that the bones from C2 to the proximal femur were in the range. All radiographs were evaluated for the presence and severity of VF by a spine surgeon (C.H.) using Genant’s semi-quantitative (SQ) method [21]. All visible vertebrae from T4 to the most caudal vertebra were assessed for SQ, where each vertebra was graded from 0 to 3 (0, normal; 1, mildly deformed; 2, moderately deformed; 3, severely deformed). SQ is not a pure quantitative method and does not require morphometrical measurement. Vertebral deformities that did not represent fractures were excluded, such as Schmorl’s nodes, Scheuermann disease, and short vertebral height [22, 23]. Vertebrae with poor visibility or image quality were not graded and were excluded from the analysis. Vertebrae with SQ ≥ 1 were defined as VF, vertebrae with SQ = 1 were defined as mild VF (mVF), and vertebrae with SQ ≥ 2 were defined as severe VF (sVF).
To investigate the intra- and inter-observer reliability of SQ grading, 50 randomly selected radiographs were graded by the same reader (C.H.) at an interval of > 2 weeks as well as by another orthopedist (T.I.) who was blinded to the scores assigned by C.H. The percentage of agreement and kappa statistics were 98.1% and 0.70 for intra-observer and 98.0% and 0.62 for inter-observer reliabilities, respectively [17].
Bone mineral density measurement
Bone mineral density (BMD) was measured at the lumbar spine (L2–L4) and proximal femur using dual-energy X-ray absorptiometry (Hologic Discovery; Hologic, Waltham, MA). To maintain measurement quality, the same equipment was used and the same spine phantom was scanned daily to monitor the machine’s performance in the study population in different regions. BMD of the phantom was adjusted to 1.032 ± 0.016 g/cm2 (± 1.5%) in all examinations. In addition, the same physician (N.Y.) examined all participants to prevent observer variability. Osteoporosis was diagnosed when either the lumbar (L-BMD) or femoral neck (FN-BMD) BMD was ≥ 2.5 standard deviations (SD) below the mean level for a young adult reference population, according to the World Health Organization (WHO) definitions [24]. To define osteoporosis, the BMD values of the reference male and female populations were used for the male and female participants, respectively. The cut-off values for osteoporosis were 0.714 g/cm2 for L-BMD in both men and women and 0.546 g/cm2 in men and 0.515 g/cm2 in women for FN-BMD [25].
Estimation of cumulative incidence of VF and sVF
For this study, we defined two outcomes. First, a participant was defined as having an incident VF if at least one vertebra which was not VF (SQ = 0) at baseline was diagnosed as VF (SQ ≥ 1) at the follow-up. Second, a participant was defined as having an incident sVF if at least one vertebra which was not sVF (SQ ≤ 1) at baseline was diagnosed as sVF (SQ ≥ 2) at the follow-up.
Cumulative incidence was calculated using the results of diagnoses with these definitions. Cumulative incidence was simply defined as the ratio of incident cases to the at-risk population at the beginning of the observation period. In this study, cumulative incidence over 3 years was thus detected as the number of incident cases divided by the number of the at-risk population at baseline. Because the cohort of the present study comprised participants with radiographs at both baseline and follow-up, the calculated indices were those of survivors.
Statistical analysis
Descriptive statistics were determined and presented as means and SDs or frequencies and percentages, unless otherwise specified. Differences in proportions were examined using the chi-square test. When the expected cell size was < 5, Fisher’s exact test was used. Differences in continuous values between two groups were examined using Student’s unpaired t tests. To determine the risk factors for incident sVF, univariate logistic regression analyses were performed with incident sVF as an objective variable. Odds ratios (ORs) and 95% confidence intervals (95% CIs) were calculated. As we could not confirm the assumption of linearity for BMI, it was transformed into a four-level categorical value: < 18.5, ≥ 18.5, < 25.0 (reference), ≥ 25.0, < 27.5, and ≥ 27.5. Furthermore, to determine independent risk factors for incident sVF in participants without prevalent sVF, multivariate logistic regression analysis was performed with incident sVF as an objective variable and with significant risk factors in the univariate logistic regression analysis model as independent variables. All statistical analyses were performed using STATA statistical software, version 15.1 (STATA, College Station, TX). All p values < 0.05 were considered to indicate significance.
Results
Of the 1486 participants in the third survey (baseline of the present study), 1320 participants participated in the fourth survey 3 years later (follow-up of the present study). Among them, 157 (11.9%) did not undergo plain radiography and were thus excluded. Reasons for dropouts were as follows: 32 (2.2%) had died by the time of the follow-up, 40 (2.7%) did not join the follow-up due to bad health, 72 (4.8%) declined the invitation to the follow-up, three (0.2%) had moved away, and 19 (1.3%) did not participate for other reasons.
Thus, 1190 participants (80.1% of the baseline participants) (384 men and 806 women) underwent plain radiography at both baseline and follow-up; these comprised the present study’s cohort. Their mean ± SD age at baseline was 65.0 ± 11.2 years. Baseline characteristics of the cohort are shown in Table 1. Men were significantly, although slightly, older than women. BMI was significantly higher in men than in women. Both L-BMD and FN-BMD were significantly higher in men than in women; thus, the proportion of having osteoporosis according to the WHO criteria was significantly higher in women than in men. While the prevalence of VF and mVF at baseline was significantly higher in men than in women, the prevalence of sVF at baseline was higher in women, which was in accordance with our previous report [17].
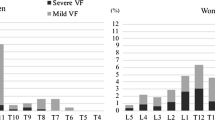
Table 2 shows the number and cumulative incidence in 3 years of VF and sVF in (i) the overall population and (ii) subgroups classified by the baseline status: (A) participants without prevalent VF, (B) participants with prevalent mVF but without prevalent sVF, and (C) participants with prevalent sVF. The annual rates of incident VF and sVF were 5.9%/year (7.6%/year in men and 5.2%/year in women, p = 0.003) and 1.7%/year (1.0%/year in men and 2.0%/year in women, p = 0.04), respectively. The incidence of both VF and sVF tended to increase sequentially according to the baseline status: (A) without prevalent VF, (B) with prevalent mVF, and (C) with prevalent sVF.
As the incidence of VF and sVF in participants with prevalent sVF (C) was significantly higher than that in participants without prevalent sVF (A + B) (16.9%/year vs. 5.2%/year for VF, p < 0.001, and 11.7%/year vs. 1.0%/year for sVF, p < 0.001), we examined characteristics by dividing the cohort into two subgroups: A + B and C. Figure 1 shows the incidence of VF and sVF in each subgroup and age stratum (40–49 years, 50–59 years, 60–69 years, 70–79 years, and ≥ 80 years). The incidence of VF and sVF tended to increase with age in both male and female participants without prevalent sVF (A + B) (for a 5-year increase: VF, OR 1.25 [95% CI 1.11–1.41] in men and OR 1.49 [95% CI 1.32–1.69] in women; sVF, OR 1.51 [95% CI 1.06–2.16] in men and OR 2.03 [95% CI 1.55–2.66] in women). By contrast, in participants with prevalent sVF (C), the incidence of VF and sVF was not age-dependent either in men or in women: VF, OR 1.31 [95% CI 0.70–2.46] in men and OR 1.29 [95% CI 0.93–1.80] in women; sVF, OR 1.28 [95% CI 0.57–2.87] in men and OR 1.30 [95% CI 0.91–1.86].
Then, we focused on incident sVF in evaluating risk factors since mVF has been reported to have low clinical relevance [17, 18]. Table 3 shows the baseline risk factors for incident sVF in participants without prevalent sVF (A + B) and participants with prevalent sVF (C). Univariate logistic regression analyses in subgroup A + B revealed that the age, FN-BMD, and presence of mVF at baseline were associated with incident sVF, whereas female sex, lower BMI (< 18.5 kg/m2), and lower L-BMD were not. Multiple mVFs at baseline had a higher OR for incident sVF than single mVF at baseline. In subgroup C, as all participants had at least one mVF at baseline, the OR was calculated for multiple mVFs at baseline in comparison with single mVF at baseline, which was 6.75 [95% CI 1.60–28.49].
Since the number of incident sVF in subgroup C was relatively small (N = 25), we performed multivariate logistic regression analyses in subgroup A + B to define independent risk factors for incident sVF (Table 4). Factors with p values < 0.05 in the univariate analyses were included in the multivariate model. Because the nature of osteoporosis has been reported to be different in men and women [26, 27], the analyses were performed in the whole cohort first and then in men and women, separately. The age and the presence of mVF at baseline (either single or multiple) were independent risk factors for incident sVF. Multiple mVFs at baseline showed a slightly higher OR than single mVF at baseline. In men-only analysis, the presence of single or multiple mVFs at baseline remained as a significant risk factor, whereas in women-only analysis, age and the presence of multiple mVFs at baseline remained significant. In another multivariate model where the number of prevalent mVF of 1 and ≥ 2 were united in one variable, as “the presence of prevalent mVF,” and two other variables (age and FN-BMD) were also included as independent variables, the adjusted OR of the presence of prevalent mVF for incident sVF was 4.22 [95% CI 2.03–8.80].
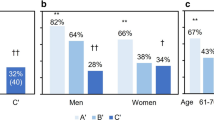
To evaluate the effect of baseline status on the incidence of sVF, we further divided subgroup B into two subgroups: (B1) participants with single mVF but without sVF at baseline and (B2) participants with multiple mVFs but without sVF at baseline. The cumulative incidence of sVF in 3 years was calculated according to the baseline status and sex (Fig. 2). In comparison with participants without prevalent sVF (A + B1 + B2), participants with prevalent sVF (C) were significantly more susceptible to incident sVF (men 23.1% vs. 2.4%, p = 0.006; women 37.9% vs. 3.5%, p < 0.001). When comparing subgroup B (B1 + B2) with subgroup A, participants with at least one mVF at baseline (B1 + B2) were more susceptible to incident sVF than participants without any VF (A) (men 8.75% vs. 0.7%, p < 0.001; women 14.1% vs. 2.2%, p < 0.001). In the comparison of B1 and B2, participants with single mVF (B1) tended to have slightly higher incidence of sVF than those with multiple mVFs (B2) in men with non-significant difference (9.4% vs. 7.4%, p = 1.00), whereas the tendency reversed in women (12.7% vs. 20.0%, p = 0.43).
Cumulative incidence of severe vertebral fractures (sVF, Genant’s semi-quantitative grading SQ ≥ 2) according to the baseline status in men and women. Subgroups were divided according to the baseline status: A participants without any vertebral fracture, B1 participants with single mild VF (mVF) but without sVF, B2 Participants with multiple mVFs but without sVF, and C participants with sVF
Discussion
The present study evaluated the cumulative incidence of VF and sVF over 3 years using radiographic diagnoses after two radiographic examinations of the whole spine in the lateral view of population-based subjects. In this cohort analysis, we estimated that the cumulative incidence rates of VF and sVF were 5.9%/year (7.6%/year in men and 5.2%/year in women) for VF and 1.7%/year (1.0%/year in men and 2.0%/year in women) for sVF. We also found that older age and the presence of mVF at baseline were independent risk factors for incident sVF in participants without sVF at baseline.
Compared with studies on the prevalence of VF, studies on the incidence of VF are limited. One reason for this is the fact that VF is not always symptomatic, and longitudinal population surveys are required to clarify the incidence of VF. However, few such longitudinal observational studies with high participation rates have been reported, representing the principal strength of this study. Population-based studies reporting the incidence of VF have been performed in Western countries such as the USA [28], the Netherlands [13], and Spain [29] and in a multicenter study involving 19 European countries [30]. The reported incidence of VF varies from 0.5%/year to 1.5%/year, reflecting differences in ethnicities, mean ages of the participants, and criteria used to define VF. Van der Klift et al. and Felsenberg et al. used the McCloskey–Kanis criteria, which is a quantitative method and needs 3SD reduction in the vertebral height to define VF, to report the incidence of VF. The reported rates of incident VF were 5.9/1000 person years (PY) in men and 14.7/1000 PY in women [13] and 5.7/1000 PY in men and 10.7/1000PY in women [30]. Compared with their finding, we reported higher incidence even for sVF (1.0%/year in men and 2.0%/year in women). The McCloskey–Kanis criteria might be stricter than Genant SQ ≥ 2.
Kanterwicz et al. used modified Genant’s SQ method to define sVF in their 4-year study including 2493 Spanish women [29]. The reported rates of incident sVF were approximately 1.38%/year in participants without prevalent sVF and 7.7%/year in participants with prevalent sVF. Compared with that study, we reported a comparable rate of incident sVF (1.2%/year) in women without prevalent sVF (A + B) and a higher rate of incident sVF (12.6%/year) in women with prevalent sVF (C), indicating that prevalent sVF might be a stronger risk factor for incident sVF in Japanese women than in Spanish women.
A few studies have reported the incidence of VF in Japan; however, none of them used Genant’s SQ method to define incident VF [10, 31, 32]. Owing to the discordance in the definitions of VF, direct comparisons of the reported incidence between past studies and the present study might not be appropriate. However, the reported rates are similar to the incidence of sVF in our study (2.2%/year by Fujiwara et al. [32] and 1.7%/year by Kadowaki et al. [10] compared with our 1.7%/year).
To confirm whether the participants of the present study were representative of the Japanese population, we compared anthropometric measurements and the frequency of smoking and alcohol consumption between the participants and the general Japanese population [33]. Details of the comparisons are presented in Supplementary Tables 1 and 2. Briefly, the overall BMI of participants was basically comparable to that of the broader Japanese population; however, the participants had healthier lifestyles than the general Japanese population in terms of smoking habits. Given that the results obtained from the present study applied to the total age-sex distribution derived from the Japanese census in 2015 [34], it can be assumed that 4,200,000 Japanese people (2,200,000 men and 2,000,000 women) aged ≥ 40 years would sustain incident VF annually and that 1,180,000 people (280,000 men and 900,000 women) aged ≥ 40 years would sustain incident sVF annually.
Most past studies reporting the incidence of VF did not distinguish the baseline status in terms of the presence of prevalent sVF. However, as prevalent sVF has been reported to be a strong risk factor for incident VF [9, 10, 12, 13], we analyzed participants without prevalent sVF (A + B) and those with prevalent sVF (C) separately. The present study revealed that incident VF and sVF were age-dependent in patients without prevalent sVF, whereas they were not in patients with prevalent sVF. Figure 1 shows that the highest incidence of sVF in subgroup C was in participants in their 70 s, and participants aged ≥ 80 years showed a lower incidence than participants in their 70 s. However, the results might be under bias since the present study is based on survivors’ analyses; participants aged ≥ 80 years with prevalent and incident sVF, who were supposed to have the shortest life expectancy and most impaired ADL, might have not joined the follow-up survey because of death or poor health.
The present study also revealed that prevalent mVF as well as sVF are risk factors for incident sVF. Many studies have reported prevalent sVF as a strong risk factor for the incident sVF [9, 35,36,37]. The present study confirmed the same tendency in both men and women as shown in Fig. 2, comparing subgroup C with subgroup A + B1 + B2. By contrast, literature regarding prevalent mVF as a risk factor for incident sVF is scarce, especially in men. Roux et al. reported that the presence of mVF increased the risk of new VF 1.79-fold (95% CI 1.31–2.45) in post-menopausal women [14]. Melton et al. reported that the presence of a 20% reduction in any vertebral heights, which corresponds to prevalent mVF, in terms of vertebral height reduction, in the present study, increased the risk of new VF 2.2-fold (95% CI 1.3–3.8) in post-menopausal women [16]. Our study revealed that it increased 4.22-fold (95% CI 2.03–8.80) compared with those without prevalent mVF. The results hold in the whole-cohort analysis with adjustment for sex and in men-only and women-only analyses. There supposed to be some associations among factors included in the model: older age, lower BMD, and presence of mVF might be associated with one another. However, the mean of variance inflation factors was 7.0, indicating that this model is acceptable. In addition, multiple mVFs were a stronger risk factor for sVF with an adjusted OR of 4.53 than single mVF with OR of 4.12. As indicated by past reports [38] and the present study, interventions are required for people with prevalent sVF, a severely deformed vertebra, to prevent subsequent VF. Similarly, attention should be paid to people with mVF, a mildly deformed vertebra. At the very least, known risk factors for osteoporotic fractures such as a clinical history of fracture, smoking habit, and alcohol consumption need to be evaluated.
This study has several limitations. First, the present study cohort may not be truly representative of the general Japanese population. Specifically, the proportion of smokers was quite low compared with the general population in men and women (Suppl. Table 2); thus, the incidence of VF and sVF in the present study might be underestimated because smoking is a risk factor for osteoporotic fracture [39]. Second, those who had died before the follow-up and those who were in poor health did not participate in the follow-up study, which might have caused an underestimation of the incidence of VF. To minimize the effect of dropouts, we intend to continue studying VF through the ROAD surveys and will analyze the incidence using a person-year method. Third, the proportion of patients who underwent osteoporosis treatment at baseline was low (two men and 68 women, 8.4% of the total cohort), whereas seven men and 110 women were diagnosed with osteoporosis through the WHO definition, leaving 94 (80%) participants with untreated osteoporosis. More aggressive osteoporosis treatment might have resulted in a protective effect on incident VF. Fourth, the incidence of VF varies according to its definition; differences between a quantitative method and a qualitative method have been reported [40, 41]. In addition, VF diagnosis were based on morphometry, regardless of clinical symptoms. Some deformities, especially mild ones, might not be true fractures but degenerative changes, although we strained to include fractures alone by recognizing end-plate fractures [22]. Finally, a limited number of factors were included in the multivariate logistic regression model; factors that have been reported as risks, such as low BMI, smoking habit, alcohol consumption, and steroidal use, were not adjusted for. The number of incident sVF in participants without prevalent sVF was 35, which allowed three or fewer variables to be included in the logistic regression model to avoid overfitting. As our objective was to evaluate whether mVF was a risk factor for incident sVF but not to survey all potential risk factors, we decided to only include those factors that were significant in the univariate model. The models analyzing men and women separately may be overfitting. Furthermore, because the ROAD study is still ongoing, we can and will analyze risk factors in a more detailed way with the accumulation of study periods and a larger number of participants with incident sVF.
Despite these limitations, we estimated that the annual cumulative incidence rates of VF and sVF in Japan were 5.9%/year (7.6%/year in men and 5.2%/year in women) and 1.7%/year (1.0%/year in men and 2.0%/year in women), respectively. We also revealed the incidence of VF and sVF classified by age stratum and sex. Further, we clarified that the presence of mVF was an independent risk factor for incident sVF in participants without prevalent sVF at baseline, with multiple mVFs giving higher OR than single mVF. These findings will contribute to the effective prevention of VF and sVF.
Data Availability
Not applicable.
Code availability
Not applicable.
References
Riggs BL, Melton LJ 3rd (1995) The worldwide problem of osteoporosis: insights afforded by epidemiology. Bone 17:505s–511s
Suzuki N, Ogikubo O, Hansson T (2009) The prognosis for pain, disability, activities of daily living and quality of life after an acute osteoporotic vertebral body fracture: its relation to fracture level, type of fracture and grade of fracture deformation. Eur Spine J 18:77–88
Cockerill W, Lunt M, Silman AJ et al (2004) Health-related quality of life and radiographic vertebral fracture. Osteoporos Int 15:113–119
Oleksik A, Lips P, Dawson A, Minshall ME, Shen W, Cooper C, Kanis J (2000) Health-related quality of life in postmenopausal women with low BMD with or without prevalent vertebral fractures. J Bone Miner Res 15:1384–1392
Johnell O, Kanis JA, Odén A, Sernbo I, Redlund-Johnell I, Petterson C, De Laet C, Jönsson B (2004) Mortality after osteoporotic fractures. Osteoporos Int 15:38–42
Hasserius R, Karlsson MK, Nilsson BE, Redlund-Johnell I, Johnell O, Study EVO (2003) Prevalent vertebral deformities predict increased mortality and increased fracture rate in both men and women: a 10-year population-based study of 598 individuals from the Swedish cohort in the European Vertebral Osteoporosis Study. Osteoporos Int 14:61–68
Cooper C, Atkinson EJ, O’Fallon WM, Melton LJ (1992) Incidence of clinically diagnosed vertebral fractures: a population-based study in Rochester, Minnesota, 1985–1989. J Bone Miner Res 7:221–227
Ross PD, Fujiwara S, Huang C, Davis JW, Epstein RS, Wasnich RD, Kodama K, Melton LJ (1995) Vertebral fracture prevalence in women in Hiroshima compared to Caucasians or Japanese in the US. Int J Epidemiol 24:1171–1177
Fujiwara S, Hamaya E, Goto W, Masunari N, Furukawa K, Fukunaga M, Nakamura T, Miyauchi A, Chen P (2011) Vertebral fracture status and the World Health Organization risk factors for predicting osteoporotic fracture risk in Japan. Bone 49:520–525
Kadowaki E, Tamaki J, Iki M, Sato Y, Chiba Y, Kajita E, Kagamimori S, Kagawa Y, Yoneshima H (2010) Prevalent vertebral deformity independently increases incident vertebral fracture risk in middle-aged and elderly Japanese women: the Japanese Population-based Osteoporosis (JPOS) Cohort Study. Osteoporos Int 21:1513–1522
Bank TW (2020) Life expectancy at birth, total (years) - Japan. https://data.worldbank.org/indicator/SP.DYN.LE00.IN?locations=JP. Accessed 20 Aug 2020
Chen P, Krege JH, Adachi JD et al (2009) Vertebral fracture status and the World Health Organization risk factors for predicting osteoporotic fracture risk. J Bone Miner Res 24:495–502
Van der Klift M, De Laet CE, McCloskey EV, Hofman A, Pols HA (2002) The incidence of vertebral fractures in men and women: the Rotterdam Study. J Bone Miner Res 17:1051–1056
Roux C, Fechtenbaum J, Kolta S, Briot K, Girard M (2007) Mild prevalent and incident vertebral fractures are risk factors for new fractures. Osteoporos Int 18:1617–1624
Kanterewicz E, Puigoriol E, Rodríguez Cros JR, Peris P (2019) Prevalent vertebral fractures and minor vertebral deformities analyzed by vertebral fracture assessment (VFA) increases the risk of incident fractures in postmenopausal women: the FRODOS study. Osteoporos Int 30:2141–2149
Melton LJ, Wenger DE, Atkinson EJ, Achenbach SJ, Berquist TH, Riggs BL, Jiang G, Eastell R (2006) Influence of baseline deformity definition on subsequent vertebral fracture risk in postmenopausal women. Osteoporos Int 17:978–985
Horii C, Asai Y, Iidaka T et al (2019) Differences in prevalence and associated factors between mild and severe vertebral fractures in Japanese men and women: the third survey of the ROAD study. J Bone Miner Metab 37:844–853
Szulc P, Munoz F, Marchand F, Delmas PD (2001) Semiquantitative evaluation of prevalent vertebral deformities in men and their relationship with osteoporosis: the MINOS study. Osteoporos Int 12:302–310
Yoshimura N, Muraki S, Oka H et al (2009) Prevalence of knee osteoarthritis, lumbar spondylosis, and osteoporosis in Japanese men and women: the research on osteoarthritis/osteoporosis against disability study. J Bone Miner Metab 27:620–628
Yoshimura N, Muraki S, Oka H, Kawaguchi H, Nakamura K, Akune T (2010) Cohort profile: research on Osteoarthritis/Osteoporosis Against Disability study. Int J Epidemiol 39:988–995
Genant HK, Wu CY, van Kuijk C, Nevitt MC (1993) Vertebral fracture assessment using a semiquantitative technique. J Bone Miner Res 8:1137–1148
Lentle B, Koromani F, Brown JP et al (2019) The Radiology of Osteoporotic Vertebral Fractures Revisited. J Bone Miner Res 34:409–418
Wáng YXJ, Deng M, He LC, Che-Nordin N, Santiago FR (2018) Osteoporotic vertebral endplate and cortex fractures: a pictorial review. J Orthop Translat 15:35–49
Group WS (2007) Assessment of osteoporosis at the primary health care level. Technical Report. Accessed 17 Aug. 2020
Orimo H, Hayashi Y, Fukunaga M et al (2001) Diagnostic criteria for primary osteoporosis: year 2000 revision. J Bone Miner Metab 19:331–337
van der Klift M, de Laet CD, Pols HA (2005) Assessment of fracture risk: who should be treated for osteoporosis? Best Pract Res Clin Rheumatol 19:937–950
Bonnick SL (2006) Osteoporosis in men and women. Clin Cornerstone 8:28–39
Samelson EJ, Hannan MT, Zhang Y, Genant HK, Felson DT, Kiel DP (2006) Incidence and risk factors for vertebral fracture in women and men: 25-year follow-up results from the population-based Framingham study. J Bone Miner Res 21:1207–1214
Kanterewicz E, Puigoriol E, Rodriguez Cros JR, Peris P (2019) Prevalent vertebral fractures and minor vertebral deformities analyzed by vertebral fracture assessment (VFA) increases the risk of incident fractures in postmenopausal women: the FRODOS study. Osteoporos Int 30:2141–2149
Felsenberg D, Silman AJ, Lunt M et al (2002) Incidence of vertebral fracture in europe: results from the European Prospective Osteoporosis Study (EPOS). J Bone Miner Res 17:716–724
Yoshimura N, Kinoshita H, Oka H, Muraki S, Mabuchi A, Kawaguchi H, Nakamura K (2006) Cumulative incidence and changes in the prevalence of vertebral fractures in a rural Japanese community: a 10-year follow-up of the Miyama cohort. Arch Osteoporos 1:43–49
Fujiwara S, Kasagi F, Masunari N, Naito K, Suzuki G, Fukunaga M (2003) Fracture prediction from bone mineral density in Japanese men and women. J Bone Miner Res 18:1547–1553
Ministry of Health LaW (2012) The report of National Health and Nutrition Survey 2012. https://www.mhlw.go.jp/bunya/kenkou/eiyou/h24-houkoku.html.
Statistics Breau of Japan (2015) Population Census/Basic Complete Tabulation on Population and Households Japan. https://www.e-stat.go.jp/en/stat-search/files?page=1&layout=datalist&toukei=00200521&tstat=000001080615&cycle=0&tclass1=000001089055&tclass2=000001089056&stat_infid=000031473212. Accessed 20 Aug 2020
Kanis JA, Oden A, Johnell O et al (2007) The use of clinical risk factors enhances the performance of BMD in the prediction of hip and osteoporotic fractures in men and women. Osteoporos Int 18:1033–1046
Black DM, Arden NK, Palermo L, Pearson J, Cummings SR (1999) Prevalent vertebral deformities predict hip fractures and new vertebral deformities but not wrist fractures. Study of Osteoporotic Fractures Research Group. J Bone Miner Res 14:821–828
Melton LJ, Atkinson EJ, Cooper C, O’Fallon WM, Riggs BL (1999) Vertebral fractures predict subsequent fractures. Osteoporos Int 10:214–221
Kendler DL, Bauer DC, Davison KS et al (2016) Vertebral Fractures: Clinical Importance and Management. Am J Med 129:221.e221–210
Thorin MH, Wihlborg A, Åkesson K, Gerdhem P (2016) Smoking, smoking cessation, and fracture risk in elderly women followed for 10 years. Osteoporos Int 27:249–255
Oei L, Koromani F, Breda SJ et al (2018) Osteoporotic vertebral fracture prevalence varies widely between qualitative and quantitative radiological assessment methods: the rotterdam study. J Bone Miner Res 33:560–568
Lentle BC, Hg Oei E, Goltzman D et al (2018) Vertebral fractures and morphometric deformities. J Bone Miner Res 33:1544–1545
Ministry of Health, Labour and Welfare. The report of National Health and Nutrition Survey 2012. https://www.mhlw.go.jp/bunya/kenkou/eiyou/h24-houkoku.html. Accessed 8 Feb 2021
Acknowledgements
The authors wish to thank Dr. Naoki Hirabayashi of Kawakami Clinic, Hidakagawa Town; Mrs. Tomoko Takijiri, Mrs. Rie Takiguchi, Mrs. Kyoko Maeda, Ms. Ikuyo Ueyama, Mrs. Michiko Mori, Mrs. Hisayo Sugimoto, and other members of the public office in Hidakagawa Town; and Mrs. Tamako Tsutsumi, Mrs. Kanami Maeda, Mrs. Megumi Takino, Mrs. Shuko Okada, Mrs. Kazuyo Setoh, Mrs. Chise Ryouno, Mrs. Miki Shimosaki, Mrs. Chika Yamaguchi, Mrs. Yuki Shimoji, and other members of the public office in Taiji Town for their assistance in locating and scheduling participants for examinations. We would also like to thank Mrs. Kyoko Hattori, Mrs. Toki Sakurai, Mrs. Saeko Sahara, and Mr. Noriyuki Oe for their assistance with data reduction and administration.
Funding
This work was supported by the Grant-in-Aid funding from the Ministry of Health, Labour and Welfare: H17-Men-eki-009 (Director, Kozo Nakamura), H20-Choujyu-009 (Director, Noriko Yoshimura), H23-Choujyu-002 (Director, Toru Akune), H25-Choujyu-007 (Director, Noriko Yoshimura), and H25-Nanchitou (Men)-005 (Director, Sakae Tanaka). The study was also supported by the Scientific Research grants B26293139, B23390172, and B20390182 and Challenging Exploratory Research grants 15K15219 and 24659317 to Noriko Yoshimura; Scientific Research grants B23390356 and C20591774 and Challenging Exploratory Research grants 23659580 to Shigeyuki Muraki; Challenging Exploratory Research grants 24659666 and 21659349 and Young Scientists A18689031 to Hiroyuki Oka; Scientific Research grants B26293329, B23390357, and C20591737 and Challenging Exploratory Research grants 25670293 to Toru Akune; and by the Collaborating Research with NSF from the Ministry of Education, Culture, Sports, Science and Technology in Japan 08033011–00262 (Director, Noriko Yoshimura). The study was partly supported by grants from the Japan Agency for Medical Research and Development (17gk0210007h0003, Director, Sakae Tanaka). Further, the study was partly supported by grants from the Japan Osteoporosis Society (Noriko Yoshimura, Shigeyuki Muraki, Hiroyuki Oka, and Toru Akune), Japan Osteoporosis Foundation (2015, Noriko Yoshimura, and 2018, Chiaki Horii), The Nakatomi Foundation (2020, Chiaki Horii), research aids from the Japanese Orthopaedic Association (JOA-Subsidized Science Project Research 2006–1 and 2010–2, Director, Hiroshi Kawaguchi; and 2014–1, Director, Kozo Nakamura), Japanese Society for Musculoskeletal Medicine (2015, Director, Shigeyuki Muraki; and 2017, Director, Noriko Yoshimura), Mitsui Sumitomo Insurance Welfare Foundation (2016, Director, Noriko Yoshimura), and Japan Dairy Association (2017, Director, Noriko Yoshimura).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by the ethics committees of the University of Tokyo (nos. 1264 and 1326) and the Tokyo Metropolitan Institute of Gerontology (no. 5). Written informed consent was provided by all participants.
Consent for publication
Not applicable.
Conflicts of interest
None.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Horii, C., Iidaka, T., Muraki, S. et al. The cumulative incidence of and risk factors for morphometric severe vertebral fractures in Japanese men and women: the ROAD study third and fourth surveys. Osteoporos Int 33, 889–899 (2022). https://doi.org/10.1007/s00198-021-06143-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-021-06143-7