Abstract
Summary
Bone mineral density (BMD) is significantly decreased after gastrectomy in patients with gastric cancer. Calcium malabsorption, secondary hyperparathyroidism, and dominant bone resorption appear to contribute to bone loss in these patients. Patients should undergo early surveillance and nutritional or pharmacologic intensive interventions for bone health.
Purpose
Survivorship care, including bone health, has become an important issue in gastric cancer. We performed a meta-analysis of the available observational studies to determine whether and how osteoporosis risk is increased after gastrectomy in patients with gastric cancer.
Methods
A total of 1204 patients (802 men) from 19 cohort studies were included. We evaluated the prevalence of osteoporosis in postgastrectomy patients, comparing the incidence according to the type of gastrectomy and sex. Additionally, we evaluated changes in bone mineral density (BMD) and bone metabolism-related markers pre- to postoperatively and between patients who underwent gastrectomy and matched controls. Proportion meta-analysis was performed and pooled odds ratios (ORs) were calculated.
Results
The pooled incidence estimate was 36% [95% confidence interval (CI), 32–40]. The incidence of osteoporosis was significantly higher in women than in men (OR = 1.90, p < 0.001) but was similar between partial and total gastrectomy groups (OR = 0.983, p = 0.939). BMD was significantly decreased, and calcium, phosphorous, and parathyroid hormone levels were significantly increased in patients after gastrectomy compared to those before gastrectomy. BMD and calcium and 25OH-vitamin D levels were significantly decreased, and parathyroid hormone and 1,25OH-vitamin D levels were significantly increased in the gastrectomy group compared to that in the control group.
Conclusion
We found that BMD is significantly decreased after gastrectomy in patients with gastric cancer. Vitamin D deficiency and secondary hyperparathyroidism are suggested to be common mechanism underlying BMD impairment. After resection, patients should undergo long-term nutritional and bone health surveillance, in addition to their oncological follow-up.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Although it presents a geographic distribution, gastric cancer is the fourth most common type of cancer worldwide [1]. With early detection and curative treatment, such as gastrectomy, the survival rate of early gastric cancer is approximately 90%, especially in Korea and Japan [2]. Thus, over the last several decades, the number of gastric cancer survivors has increased, and survivorship care concerns, including bone health, have become an important issue [3, 4].
Osteoporosis is a known chronic complication after gastrectomy in patients with gastric cancer [5]. Although the exact pathogenesis of osteoporosis in gastric cancer is unknown, old age, malabsorption, and the cancer itself and its treatment are independent risk factors for osteoporosis. Poor status of vitamin D and calcium results in secondary hyperparathyroidism, which increases the rate of bone loss. Chemotherapy can also affect bone metabolism, as it does in patients with other cancers; however, the evidence is limited. Although several studies examined the incidence or prevalence of osteoporosis after gastrectomy in patients with gastric cancer, the results varied widely. In addition, because of variations in the measurement method and bone site evaluated, relevant systematic reviews and meta-analyses are rare.
Considering the increased number of gastric cancer survivors [3, 4], it is important to determine whether and how patients with gastric cancer are at increased risk of osteoporosis after gastrectomy. Therefore, we performed a systematic review and meta-analysis of the available observational studies, evaluating the associations among gastrectomy and osteoporosis risk, mechanism, and characteristics, including bone mineral density (BMD) changes after surgery.
Materials and methods
Search methods for the identification of studies
This meta-analysis was conducted according to the updated guidelines of the Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P).[6] Two researchers (blinded by the authors) independently searched MEDLINE (PubMed), EMBASE, and Cochrane Library databases in September 2018. An overview of the search strategy is presented in Supplementary 1. Articles that met the selection criteria (not limited to randomized studies, including prospective and case-control studies) were included in the meta-analysis. We attempted to obtain complete data for the analysis by contacting the authors of articles with insufficient or missing data.
Study selection criteria
Studies were screened and selected by all investigators on the basis of a priori criteria. Inclusion criteria were as follows: (1) the study was a comparative or single-cohort study that investigated the medical records of patients with gastric cancer who underwent partial or total gastrectomy, and (2) at least one of the following main clinical outcomes was reported: the incidence of osteoporosis, changes in BMD, or changes in bone metabolism related markers. Partial gastrectomy included Billroth I and Billroth II operations, and total gastrectomy included the Roux-en-Y operation. Bone metabolism-related markers included parathyroid hormone (PTH), 25-monohydroxy vitamin D (25(OH)D), 1,25-dihydroxy vitamin D(1,25(OH)D), serum calcium, and phosphorus.
Exclusion criteria were as follows: (1) the study accessing patients with bariatric surgery or gastric ulcers; (2) the study used national registry data; (3) the study accessing patients with a prior history of osteoporosis treatment (bisphosphonate, selective estrogen receptor modulator, or vitamin D) that could influence the natural course of bone metabolism after gastrectomy; and (4) the article was a review, expert opinion, case report, or basic science study.
Outcome measures and data extraction
The primary outcome for the meta-analysis was the prevalence of osteoporosis in postgastrectomy patients. In each study, osteoporosis is defined as T-scores ≤ −2.5 at either the femoral neck or spine according to the World Health Organization definition [7]. The risk of osteoporosis was also evaluated according to the type of gastrectomy (partial or total) and sex (male or female). The secondary outcomes of this study were changes in BMD and bone metabolism-related markers, which were compared pre- and postoperatively in patients who underwent gastrectomy and between patients who underwent gastrectomy and matched controls.
For every eligible study, the following data were extracted and entered into a spreadsheet by two reviewers: the family name of the first author, year of publication, country, number of patients, type of surgery, mean years after the index operation, sample characteristics (age, sex ratio, body mass index), and changes in BMD (g/cm2) and bone metabolism-related markers. When bone mineral density was measured at various sites (femoral neck, lumbar or total), we included data from the sites where the highest values were measured because these should best reflect the change over time and to have a consistent approach.
Quality assessment and publication bias
Two of the authors (blinded by the authors) independently evaluated the quality of all studies, using the Newcastle–Ottawa Scales. This tool comprises three parameters: selection, comparability, and outcome. Each parameter consists of subcategorized items; selection has a maximum of four stars; comparability has a maximum of two stars; and exposure or outcome has a maximum of three stars. We assessed the presence of publication bias using Begg’s funnel plot [8] and Egger’s test [9].
Statistical analysis
The primary analysis involved a proportion meta-analysis of the data from all relevant studies that reported the incidence of osteoporosis. For subgroup analyses (binary outcomes), the effect sizes were calculated as odds ratios (ORs), and the studies were weighted according to the number of included patients.
In the secondary analyses (continuous outcomes), effect sizes were calculated as Hedges adjusted g weighted mean difference (WMD), which represents the magnitude of the difference between the comparative groups, for each outcome [10]. The WMD was computed separately for all available control and treatment groups in each study. All types of effect sizes were presented along with their 95% credible intervals (CIs).
A fixed effects or random effects model was used to quantify the pooled effect size of the included studies, depending on the heterogeneity of the data. Heterogeneity between comparable studies was tested using chi square (χ2) and I2 tests; P > 0.1 and I2 < 50%, respectively, were used as established criteria to determine statistical heterogeneity. All analyses were performed using Stata software (version 14.0; Stata Corporation, College Station, TX, USA). This study was exempted from institutional review board review since it did not involve any human subjects.
Results
Description of the included studies
The primary search of the databases yielded 973 records. After duplicates were removed, 413 articles were screened by title and abstract. As a result, 77 full-text articles were selected and reviewed for eligibility. A total of 19 studies were finally included in the systematic review (Supplementary 2). The studies identified for the meta-analysis involved 1204 participants, with men comprising 802 (66.6%) of the sample population. Among the 19 studies, 7 articles reported the incidence of osteoporosis, 5 studies reported the pre-/postgastrectomy changes in patient outcomes, and 7 included direct comparisons of patient and healthy control group.
Incidence of osteoporosis
Seven studies, involving a total of 634 patients, reported data on the incidence of osteoporosis (Table 1) [11,12,13,14,15,16,17]. The pooled prevalence estimate was 36% [95% confidence interval (95% CI) 32–40]. Additionally, the pooled incidence was 26% (95% CI 21–31) in male patients and 55% (95% CI 44–65) in female patients and was 34% (95% CI 29–39) in the partial gastrectomy group and 34% (95% CI 25–43) in the total gastrectomy group. The incidence of osteoporosis was higher in women than in men, and the pooled estimate of the crude OR for osteoporosis was 1.90 (95% CI 1.391–2.593, p < 0.001) (Fig. 1a). In contrast, the osteoporosis rate of the partial gastrectomy group was similar to that of the total gastrectomy group (OR = 0.983 95% CI 0.638–1.515, p = 0.939) (Fig. 1b).
Changes in BMD and serum factors between pre- and postgastrectomy patients
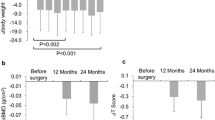
Five studies, involving a total of 125 patients, reported data on BMD and bone metabolism-related marker levels in pre- and postgastrectomy patients (Table 2) [18,19,20,21,22]. BMD (pooled WMD = −0.070; 95% CI−0.112–−0.028; p = 0.001) and 1,25(OH)D levels (pooled WMD = −6.14; 95% CI−6.72–−5.55; p < 0.001) were significantly decreased in the gastrectomy state compared to those before gastrectomy. In contrast, PTH (pooled WMD = 12.29; 95% CI 1.59–22.99; p < 0.001), calcium (pooled WMD = 0.15; 95% CI 0.11–0.19; p < 0.001), and phosphorus levels (pooled WMD = 0.31; 95% CI 0.23–0.40; p < 0.001) were significantly increased in the gastrectomy state compared to those before gastrectomy. However, pre- and postoperative levels of 25(OH)D (pooled WMD = 3.16; 95% CI−4.34–10.66; p = 0.409) did not differ significantly (Fig. 2).
Forest plots of the changes in bone mineral density (BMD) and bone metabolism-related markers in pre- and postgastrectomy patients: (a) BMD measured by dual-energy X-ray absorptiometry; (b) 25 hydroxy vitamin D; (c) 1,25 dihydroxyvitamin D; (d) parathyroid hormone; (e) serum calcium; and (f) serum phosphorus. Effect sizes are indicated as Hedges’ g weighted mean differences and 95% confidence interval (CI)
Changes in BMD and serum factors between postgastrectomy patients and matched healthy controls
Seven studies, involving a total of 445 patients, reported data on BMD and bone-related levels in patients and matched controls (Table 3) [23,24,25,26,27,28,29]. BMD (pooled WMD = −0.094; 95% CI –0.169–−0.019; p = 0.014), 25(OH)D (pooled WMD = −4.12; 95% CI −.77–−1.46; p = 0.002), and serum calcium levels (pooled SMD = −0.27; 95% CI −0.45–−0.09; p = 0.004) were significantly lower in the gastrectomy group compared to those in the healthy control group. In contrast, PTH (pooled WMD = 1.794; 95% CI 0.38–3.21; p = 0.013) and 1,25(OH)D levels (pooled WMD = −11.73; 95% CI 5.27–18.19; p < 0.001) were significantly higher in the gastrectomy group compared to those in the healthy control group. However, the serum phosphorus level (pooled WMD = −0.08; 95% CI −0.61–0.48; p = 0.767) did not differ significantly between patients and controls (Fig. 3).
Forest plots of the changes in bone mineral density (BMD) and bone metabolism-related markers in postgastrectomy patients and matched controls as outcome variables: (a) BMD measured by dual-energy X-ray absorptiometry; (b) 25 hydroxy vitamin D; (c) 1,25 dihydroxyvitamin D; (d) parathyroid hormone; (e) serum calcium; and (f) serum phosphorus. Effect sizes are indicated as Hedges’ g weighted mean differences and 95% CI
Quality assessment and publication bias
In terms of the methodological quality, the mean value of the awarded stars was 6.4 (12 studies had 6 stars, 7 studies had 7 stars; Supplementary 3). The Begg’s funnel plot was not asymmetrical, and the p-values for bias were not significant for all outcomes (Supplementary 4).
Discussion
In the present meta-analysis, we found that the prevalence of osteoporosis is greater in gastric cancer survivors than in the general population. In the Korea National Health and Nutrition Examination Survey (KNHANES), the prevalence of osteoporosis was reported as 21.8% in persons 50 years old or older, 7.8% in men, and 34.9% in women [30]. In the United States, the NHANES 2005–2006 study reported the prevalence of osteoporosis at femur as 10.0 and 2.0% in women and men, respectively [31]. In the present study, the prevalence was 36.0% among all patients (mean age of the study range from 54 to 68 years) and was 26% in male patients and 55% in female patients. The present study revealed that male patients, in particular, have a much higher incidence of osteoporosis than that in the general population. Female patients also had a higher incidence of osteoporosis than that in the general population but to a smaller extent.
Although the risk of osteoporosis was higher in female patients than in male patients, the OR of osteoporosis in female patients was 1.90, which is much smaller than that in the general population [32]. Even a previous study reported a higher incidence of osteoporotic fractures in male patients with gastric cancer than in female patients with gastric cancer [15]. Compared to that in the general male population, male patients with gastric cancer have a relatively higher risk of osteoporosis. This may be related to the greater frequency of gastric cancer in men than in women [33], and the greater frequency of smoking in men than in women, which is a well-known risk factor for osteoporosis [15, 34]. In addition, because there is bone loss after gastrectomy in both sexes, clinicians should consider screening for osteoporosis in this population of both men and women. Another pathogenesis, other than postmenopausal bone loss, may be associated with osteoporosis in postgastrectomy patients. As the type of gastrectomy operation was not associated with osteoporosis in the present study, the risk of osteoporosis may be associated with the gastrectomy itself, rather than the surgical method. These results are in line with the findings presented in studies for examining BMD after bariatric surgery [35,36,37,38].
In the present study, BMD was significantly decreased after gastrectomy, both cross-sectionally and longitudinally (Figs. 2a and 3a). Moreover, we confirmed that serum calcium and PTH levels were significantly increased after gastrectomy (Fig. 2e). These findings suggest that osteoporosis after gastrectomy in patients with gastric cancer is due to secondary hyperparathyroidism; that could be explained by decreased intestinal calcium absorption and alterations in calcium homeostasis, causing a greater bone resorption than formation. These mechanisms in postgastrectomy patients could provide evidence for administration of dietary calcium and bone antiresorptive agents.
Furthermore, serum 25(OH)D levels were significantly lower, while 1,25(OH)D levels were significantly higher in the gastrectomy group compared to the healthy control group. In postgastrectomy patients, intestinal calcium absorption is decreased [39, 40] which leads to increased PTH levels and maintained serum calcium level with bone resorption. Elevated 1,25(OH)D levels may be explained by elevated l-a-hydroxylase activity in the kidney, mediated by increased PTH levels and hypophosphatemia [41]. Baek et al. reported that patients with a higher postoperative PTH level exhibited a more pronounced decrease in BMD [18]. Similarly, another study found that postgastrectomy patients with fractures have significant increased serum calcium levels compared to those in patients without fractures by 1-year postoperatively [15]. In contrast, our meta-analysis revealed that 1,25(OH)D levels were significantly decreased in gastrectomy state compared to those before gastrectomy, which might be explained by severe 25(OH)D levels deficiency without vitamin D supplement after gastrectomy. Nevertheless, results of vitamin D status are inconsistent and complex as vitamin D-binding protein and vitamin D supplementation were not studied equally in all researches and, therefore, should be interpreted cautiously.
The recommendations for follow-up schedules, treatment, and prevention for osteoporosis after gastrectomy are not well defined in the current guidelines due to a lack of evidence. The American Gastroenterological Association (AGA) recommends dual-energy X-ray absorptiometry (DXA) evaluation in patients with a low level of evidence for at least 10 years postgastrectomy [5] and in patients with any of the following conditions: low-trauma fracture history, postmenopausal female or male over 50 years of age, and hypogonadism. The present meta-analysis and other studies have demonstrated that osteoporosis and fractures occur in the relatively early phase (within at least 5 years) after gastrectomy [11, 12, 15,16,17, 42]. In addition, changes in serum markers or hormones, indicating bone remodeling imbalance, occur early, within 1 year after gastrectomy [18]. Therefore, a much earlier assessment of BMD and serum markers, including calcium, phosphorus, 25(OH)D, PTH, and bone markers, should be performed postoperatively. In addition, similar to the development of FRAX to predict fracture risk in the general population, a new scoring system and risk factor stratification are needed for gastric cancer survivors with gastrectomy.
The present study has several limitations. First, the current analysis was based on a very small cohort. Due to a lack of concern about survivorship care in past decades, only a very few rigorously performed trials were executed, until recently. To overcome this limitation, we performed a meta-analysis as an alternative method. Second, osteoporosis is associated with many factors, and the studies did not adjust for all covariables, such as menopausal status in women, or calcium supplement. Finally, we could not directly compare the risk of osteoporosis in gastric cancer survivors to that in general population with age sex matched. Moreover, the age-specific risk of osteoporosis in gastric cancer survivors, especially in younger patients, comparing to that in general population can be the interesting topic in future study.
Conclusions
In conclusion, osteoporosis is a common and clinically significant problem after gastrectomy in patients with gastric cancer. Calcium malabsorption, secondary hyperparathyroidism, and dominant bone resorption appear to be associated with the bone loss in these patients. Patients should undergo early surveillance and nutritional or pharmacologic intensive interventions for bone health, in addition to their oncological follow-up. Future large, high-quality, population-based observational studies are needed to establish standardized guidelines with recommendations for osteoporosis prevention and treatment in gastric cancer survivors.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424
Jung K-W, Won Y-J, Kong H-J, Oh C-M, Shin A, Lee J-S (2013) Survival of Korean adult cancer patients by stage at diagnosis, 2006-2010: national cancer registry study. Cancer Res Treat 45(3):162–171
Shapiro CL (2018) Cancer Survivorship. N Engl J Med 379(25):2438–2450
Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH, Stein KD, Alteri R, Jemal A (2016) Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin 66(4):271–289
Bernstein CN, Leslie WD, Leboff MS (2003) AGA technical review on osteoporosis in gastrointestinal diseases. Gastroenterology 124(3):795–841
Shamseer L, Moher D, Clarke M, Ghersi D, Liberati A, Petticrew M, Shekelle P, Stewart LA, Group P-P (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: elaboration and explanation. BMJ 350:g7647
Kanis JA on behalf of the World Health Organization Scientific Group (2007) Assessment of osteoporosis at the primary health-care level. Technical report. Sheffield: World Health Organization Collaborating Centre for Metabolic Bone Diseases, University of Sheffield
Begg CB, Mazumdar M (1994) Operating characteristics of a rank correlation test for publication bias. Biometrics 50(4):1088–1101
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315(7109):629–634
Becker BJ (1988) Synthesizing standardized mean-change measures. Br J Math Stat Psychol 41:257–278
Climent M, Pera M, Aymar I, Ramón JM, Grande L, Nogués X (2018) Bone health in long-term gastric cancer survivors: a prospective study of high-dose vitamin D supplementation using an easy administration scheme. J Bone Miner Metab 36(4):462–469
Glatzle J, Piert M, Meile T, Besenthal I, Schafer JF, Konigsrainer A, Zittel TT (2005) Prevalence of vertebral alterations and the effects of calcium and vitamin D supplementation on calcium metabolism and bone mineral density after gastrectomy. Br J Surg 92(5):579–585
Krogsgaard MR, Frolich A, Lund B, Lund B (1995) Long-term changes in bone mass after partial gastrectomy in a well-defined population and its relation to tobacco and alcohol consumption. World J Surg 19(6):867–871
Lim JS, Lee JI (2011) Prevalence, pathophysiology, screening and management of osteoporosis in gastric cancer patients. J Gastric Cancer 11(1):7–15
Oh HJ, Lim CH, Yoon BH, Yoon SB, Baeg MK, Kim WC, Cho YK, Park JM, Choi MG, Yoo HM, Song KY, Jeon HM, Park CH (2017) Fracture after gastrectomy for gastric cancer: A long-term follow-up observational study. Eur J Cancer 72:28–36
Seo GH, Kang HY, Choe EK (2018) Osteoporosis and fracture after gastrectomy for stomach cancer: A nationwide claims study. Medicine (Baltimore) 97(17):e0532
Yoo SH, Lee JA, Kang SY, Kim YS, Sunwoo S, Kim BS, Yook JH (2018) Risk of osteoporosis after gastrectomy in long-term gastric cancer survivors. Gastric Cancer 21(4):720–727
Baek KH, Jeon HM, Lee SS, Lim DJ, Oh KW, Lee WY, Rhee EJ, Han JH, Cha BY, Lee KW, Son HY, Kang SK, Kang MI (2008) Short-term changes in bone and mineral metabolism following gastrectomy in gastric cancer patients. Bone 42(1):61–67
Koufuji K, Takeda J, Aoyagi K, Yano S, Murakami N, Hori H, Terasaki Y, Shirouz K (1999) A study on metabolic bone disease before and after distal gastrectomy in gastric cancer patients by dual X-ray mineral absorptiometry measurements and markers of bone metabolism. Jpn J Gastroenterol Surg 32(12):2631–2636
Liedman B, Bosaeus I, Mellström D, Lundell L (1997) Osteoporosis after total gastrectomy: results of a prospective, clinical study. Scand J Gastroenterol 32(11):1090–1095
Wetscher G, Redmond E, Watfah C, Perdikis G, Gadenstatter M, Pointner R (1994) Bone disorders following total gastrectomy. Dig Dis Sci 39(12):2511–2515
Noh HM, Yoo JH, Jeong JY, Park YS (2018) Bone mineral density after treatment for gastric cancer: endoscopic treatment versus gastrectomy. Medicine (Baltimore) 97(1):e9582
Heiskanen JT, Kroger H, Paakkonen M, Parviainen MT, Lamberg-Allardt C, Alhava E (2001) Bone mineral metabolism after total gastrectomy. Bone 28(1):123–127
Klein KB, Orwoll ES, Lieberman DA, Meier DE, McClung MR, Parfitt AM (1987) Metabolic bone disease in asymptomatic men after partial gastrectomy with Billroth II anastomosis. Gastroenterology 92(3):608–616
Kwon SJ, Hahm JS, Cho YJ, Ahn Y, Shin DI (2000) The influence of gastrectomy on the change of bone metabolism and bone density. Korean J Intern Med 15(1):25–31
Resch H, Pietschmann P, Pernecker B, Krexner E, Willvonseder R (1992) The influence of partial gastrectomy on biochemical parameters of bone metabolism and bone density. Clin Investig 70(5):426–429
Bisballe S, Eriksen EF, Melsen F, Mosekilde L, Sorensen OH, Hessov I (1991) Osteopenia and osteomalacia after gastrectomy: interrelations between biochemical markers of bone remodelling, vitamin D metabolites, and bone histomorphometry. Gut 32(11):1303–1307
Imawari M, Kozawa K, Akanuma Y, Koizumi S, Itakura H, Kosaka K (1980) Serum 25-hydroxyvitamin D and vitamin D-binding protein levels and mineral metabolism after partial and total gastrectomy. Gastroenterology 79(2):255–258
Inoue K, Shiomi K, Higashide S, Kan N, Nio Y, Tobe T, Shigeno C, Konishi J, Okumura H, Yamamuro T et al (1992) Metabolic bone disease following gastrectomy: assessment by dual energy X-ray absorptiometry. Br J Surg 79(4):321–324
Park EJ, Joo IW, Jang M-J, Kim YT, Oh K, Oh HJ (2014) Prevalence of osteoporosis in the Korean population based on Korea National Health and Nutrition Examination Survey (KNHANES), 2008-2011. Yonsei Med J 55(4):1049–1057
Looker AC, Melton LJ, Harris TB, Borrud LG, Shepherd JA (2010) Prevalence and trends in low femur bone density among older US adults: NHANES 2005–2006 compared with NHANES III. J Bone Miner Res 25(1):64–71
Melton LJ 3rd, Chrischilles EA, Cooper C, Lane AW, Riggs BL (1992) Perspective. How many women have osteoporosis? J Bone Miner Res 7(9):1005–1010
Roder DM (2002) The epidemiology of gastric cancer. Gastric Cancer 5(1):5–11
Compston J, Cooper A, Cooper C, Gittoes N, Gregson C, Harvey N, Hope S, Kanis J, McCloskey E, Poole KE (2017) UK clinical guideline for the prevention and treatment of osteoporosis. Arch Osteoporos 12(1):43
Blom-Hogestol IK, Hewitt S, Chahal-Kummen M, Brunborg C, Gulseth HL, Kristinsson JA, Eriksen EF, Mala T (2019) Bone metabolism, bone mineral density and low-energy fractures 10 years after Roux-en-Y gastric bypass. Bone 127:436–445
Bredella MA, Greenblatt LB, Eajazi A, Torriani M, Yu EW (2017) Effects of Roux-en-Y gastric bypass and sleeve gastrectomy on bone mineral density and marrow adipose tissue. Bone 95:85–90
Geoffroy M, Charlot-Lambrecht I, Chrusciel J, Gaubil-Kaladjian I, Diaz-Cives A, Eschard JP, Salmon JH (2019) Impact of bariatric surgery on bone mineral density: observational study of 110 patients followed up in a specialized center for the treatment of obesity in France. Obes Surg 29(6):1765–1772
Luhrs AR, Davalos G, Lerebours R, Yoo J, Park C, Tabone L, Omotosho P, Torquati A, Portenier D, Guerron AD (2019) Determining changes in bone metabolism after bariatric surgery in postmenopausal women. Surg Endosc
Deller DJ (1966) Radiocalcium absorption after partial gastrectomy. Am J Dig Dis 11(1):10–19
Fischermann K, Harly S, Worning H, Zacho A (1967) Pancreatic function and the absorption of fat, iron, vitamin B12, and calcium after total gastrectomy for gastric cancer. Gut 8(3):260–266
Lips P (2006) Vitamin D physiology. Prog Biophys Mol Biol 92(1):4–8
Cuerda C, Camblor M, Bretón I, Velasco C, Parón L, Hervás E, Muñoz-Calero A, García-Peris P (2007) Cirugía gástrica como factor de riesgo nutricional. Nutr Hosp 22(3):330–336
Funding
This study was funded by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea [grant number: HI18C0284].
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
This meta-analysis summarizes previously published data and does not include new human data or tissue that requires ethical approval and consent. The authors assume that the studies reviewed were conducted after ethical approval and consent and in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Oh, H., Yoon, BH., Ha, YC. et al. The change of bone mineral density and bone metabolism after gastrectomy for gastric cancer: a meta-analysis. Osteoporos Int 31, 267–275 (2020). https://doi.org/10.1007/s00198-019-05220-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-019-05220-2