Abstract
Introduction and hypothesis
Most of the literature on pelvic organ prolapse (POP) has been generated from postmenopausal patients in high-income countries. In the Democratic Republic of the Congo (DRC), a significant proportion of patients who present for surgical management of POP are premenopausal. Little is known about the impact of POP on pelvic floor symptoms in this population. The objective was to describe pelvic floor symptoms and sexual function among premenopausal patients presenting for POP surgery in DRC.
Methods
We performed a prospective cohort study of symptomatic premenopausal patients undergoing fertility-sparing POP surgery at a large referral hospital in the DRC. Pelvic floor symptoms were evaluated with the Pelvic Floor Distress Inventory Questionnaire and sexual function with the Pelvic organ prolapse/urinary Incontinence Sexual Questionnaire. Data are presented as means with standard deviations or counts with percentages.
Results
A total of 107 patients were recruited between April 2019 and December 2021. All had either stage III (95.3%) or stage IV (4.7%) prolapse. Ages were 34.2 ± 6.7 years; 78.5% were married. A majority of patients experienced low abdominal pain (82.2%), heaviness or dullness (95.3%), and bulging or protrusion of the prolapse (92.5%). Almost two-thirds of patients reported no longer being sexually active, and 80% stated that they were not sexually active because of POP. Of the 37 sexually active patients (34.6%), nearly all reported significant sexual impairment because of the prolapse, with only 4 reporting no sexual impairment.
Conclusions
This study represents one of the largest prospective series of patients with premenopausal POP. Our results highlight the severity of pelvic floor symptoms and the negative effects on sexual function among this patient population with POP.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Most of the available literature on the impact of pelvic organ prolapse (POP) on quality of life comes from high-income countries, where parity and age are the usual risk factors [1, 2]. In low-/middle-income countries, however, high parity rates, heavy daily physical work, and poor nutrition are additional contributing factors for POP [3,4,5,6,7,8]. Countries in sub-Saharan Africa have much higher fertility rates (lifetime births per woman) than many other parts of the world, emphasizing the need for further research into POP in this region [9]. Although the burden of POP in sub-Saharan Africa is not precisely known, a recent meta-analysis concluded that the prevalence of POP in developing countries is about 15% overall [10]. A study at Panzi Hospital, a large tertiary referral center in Eastern Democratic Republic of the Congo (DRC), reported that 25% of prolapse patients who presented for surgery were premenopausal, highlighting the problem of early age of onset in this region [11].
The Pelvic Floor Distress Inventory Questionnaire (PFDI-20) and the Pelvic organ prolapse/urinary Incontinence Sexual Questionnaire (PISQ-12) are validated questionnaires that were created in English, and have been translated into and re-validated in French [12, 13]. The PFDI-20 evaluates the impact of pelvic floor disorders (PFDs) on the patient’s overall quality of life, whereas the PISQ-12 focuses on sexual function among patients with POP and/or urinary incontinence, including evaluations of physical, emotional, and partner-related components [14, 15]. In this study, we administered these questionnaires to a cohort of symptomatic pre-menopausal patients presenting for POP surgery at Panzi Hospital in Bukavu, DRC. Our aim was to characterize the pelvic floor symptoms and sexual function associated with advanced POP in this understudied younger population of patients.
Materials and methods
We conducted a prospective, descriptive study of all symptomatic premenopausal patients seeking surgical treatment for POP at Panzi Hospital between April 2019 and December 2021. We have previously described the approach at our institution for integrated care for patients with POP [11]. Briefly, patients are referred to our hospital from throughout the Eastern DRC region and neighboring countries. Patients receive a consultation with a gynecologist, who confirms the presence of POP and assesses its severity using the Pelvic Organ Quantification system. The gynecologist reviews the diagnosis with the patient and discusses treatment options. In addition, because almost 40% of women in our region have experienced prior sexual violence [16], every patient who presents to Panzi Hospital for gynecological care receives an evaluation by a social worker or a psychologist and may undergo treatment for their trauma. Ethical approval for this study was obtained from the Democratic Republic of the Congo Ministry of Public Health National Health Ethics Committee (Ref # 148PM2020).
Premenopausal patients seeking treatment for symptomatic prolapse, confirmed on examination, and electing to undergo surgical repair, were included in this study after informed consent was obtained. All patients underwent fertility-sparing repair with a hysteropexy surgery.
As part of standard clinical care at Panzi Hospital, pelvic floor symptoms and the impact of POP on sexual function were evaluated with the French-language versions of the PFDI-20 and PISQ-12 respectively [12, 13]. Because of low levels of literacy in our patient population, the questionnaires were administered by a trained interviewer fluent in both French and each patient’s native language. Demographic data were extracted from patient intake forms. Data are presented as means with standard deviations, or counts with percentages.
Results
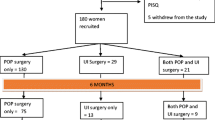
A total of 107 premenopausal women with stage III/IV POP consented to this study between April 2019 and December 2021. Age at presentation to Panzi Hospital was 34.2 ± 6.7 years. Of this cohort, 102 patients (95.3%) had stage III prolapse, and the remaining 5 (4.7%) had stage IV prolapse (Table 1). Owing to travel distances, typically by foot, a hospital stay of 1 week, and local referral patterns, the vast majority of patients present with POP stage ≥ III. All 107 patients had multicompartment prolapse (Table 2). Most presented more than 2 years after the start of prolapse symptoms (n = 66, 61.7%); a minority waited 6 years or more (n = 14, 13.1%; Table 1). Regarding key underlying contributors to early-onset POP, multiparity was nearly universal, and 42% were grand multiparous women. Eighty-eight (82.2%) were farmers, implying heavy daily physical work; and 31 (29.0%) had a body mass index (BMI) < 20 kg/m2. All patients underwent fertility-sparing surgery with either a sacrospinous or uterosacral hysteropexy (Table 1).
Mean PFDI-20 score was 109.8 ± 27.6 (Table 3). The most common symptoms reported were lower abdominal pain (n = 88, 82.2%), heaviness or dullness (n = 102, 95.3%), bulging sensation (n = 99, 92.5%), and pain or discomfort in the lower abdomen or genital region (n = 87, 81.3%). Over half (n = 63, 58.9%) had to splint to defecate, and over two-thirds (n = 74, 69.2%) had flatus incontinence. Nearly half (n = 52, 48.6%) had stress urinary incontinence, and 15% reported being bothered “moderately” or “quite a bit.” Over half (n = 66, 61.7%) reported difficulty emptying completely with voiding, and 47 (43.9%) had to splint to void.
All 107 patients also completed the PISQ-12. Only 37 patients (34.6%) reported being sexually active. Of the 84 patients who were married (78.5%), only 35 (58.3%) reported being sexually active. Most of the women who were not sexually active (n = 70, 65.4%) reported that their sexual inactivity was due to POP (n = 56, 80.0%). Overall, 52 (48.6%) reported usually or always having a negative emotional reaction during intercourse, and 66 (61.7%) reported avoiding sexual activity because of bulge symptoms (Table 4).
Among the 37 women (34.6%) who reported being sexually active, most reported significant sexual impairment owing to the prolapse, with only 4 women (10.8%) reporting that they were not affected by their prolapse. Twenty (54.1%) reported that they were never/seldom sexually excited and 21 (56.8%) were never/seldom satisfied with their sex lives. The overall PISQ-12 scores among sexually active and sexually inactive groups were nearly identical.
Discussion
Our findings highlight the devastating effects associated with POP, including impaired sexual function, among young women in the DRC. This is one of the largest studies among premenopausal patients with POP.
There are a variety of contributing factors in the development of POP, including age, parity, heavy daily physical work, and poor nutrition [1,2,3,4,5,6,7,8]. Specifically, increasing parity has been shown to play a particularly important role in the pathophysiology of POP [17]. In our study, patients presented at a mean age of 34 years old, with most having symptoms for 2–5 years. Many of our patients had underlying risk factors for developing POP, as 42% were grand multiparous women, 80% were farmers, and 29% had a BMI < 20 kg/m2.
Previous studies have highlighted the stigma surrounding POP. Owing to a combination of a lack of money, limited access to conventional health care and the associated stigma, many patients in this region turn to traditional healers for help [3, 18]. In our study, most of the patients presented within 5 years of prolapse symptoms (n = 93, 86.9%). This figure compares favorably with previously published data from sub-Saharan Africa. In a small study by Gjerde et al. in Ethiopia, about half of the 24 women interviewed reported that they had had POP for 10 years or longer before seeking specialized gynecological care [19]. To decrease the stigma associated with this common issue and to encourage earlier presentation for treatment, Panzi Hospital implemented several outreach programs to the surrounding communities, including rural areas, to educate the population about POP and the availability of services. These outreach programs include mobile clinics that travel to rural areas in Eastern DRC, along with social workers who go to churches and meet with local leaders [11].
In Gjerde et al.’s qualitative study, the women also reported significant discomfort from their POP, affecting the ability to complete activities of daily living, including household chores, childcare, and gathering and carrying water and firewood [19]. Wusu-Ansah and Opare-Addo administered the PFDI-20 and the pelvic floor impact questionnaire to women in rural Ghana to determine the prevalence of POP and impact on women in that region. Twenty-one women in their cohort had POP, with 81% of these women reporting POP symptoms, but only 62% were bothered by their symptoms [20]. All the patients in our cohort were symptomatic when they presented for surgery, and 80% of them were farmers. Because their daily activities typically require heavy and prolonged physical exertion, these pelvic floor disabilities may impair their ability to provide for their families.
Pelvic organ prolapse has also been associated with impaired sexual health, including decreased libido, decreased frequency of sexual encounters, and difficulty achieving orgasms, as well as marital difficulties [21,22,23,24,25,26]. Gjerde et al. interviewed 24 women in Ethiopia with stage II to IV POP and described the women’s fear of consequences from disclosing their condition to their friends and families, including husbands [19]. Many women in this study chose to disclose the condition to their husbands only when it became more serious, with 4 of these women (16.7%) reporting that their husbands divorced them because of the prolapse. A study in Nepal found that 74% of their participants with POP had difficulty with sexual activity, with a mean age in their study of 54 years [27]. That study reported that 24% of the husbands re-married after their wives disclosed their condition. For our young premenopausal cohort, almost 80% were married and only 34.5% were sexually active. Over half of our cohort reported that they were never or seldom satisfied with their sex lives. Of the women who were not sexually active, 80% reported that this was because of their POP.
Of note, in the Eastern region of DRC, where Panzi Hospital is located, a 2010 study reported that almost 40% of women had experienced sexual violence [16]. Female genital mutilation (FGM) is relatively rare in our region, and some reports from the greater DRC suggest prevalence rates of ~ 5% [28]. All the patients in our study, sexually active and not sexually active, filled out the PISQ-12 questionnaires, and 48.6% always or usually had negative emotional reactions during intercourse. Some of their negative reactions could be attributed to discomfort and/or urinary or fecal incontinence from POP. Nevertheless, we acknowledge that the PISQ-12 does not survey for sexual violence, and so our data cannot assess the role of this factor in our patients’ sexual inactivity or negative emotions during sex.
The strengths of this study include its relatively large sample size of premenopausal patients, for which there is a scarcity of published data. This study was also strengthened by using validated questionnaires that gathered data about both PFD symptoms and sexual function. A weakness of this study is that the cohort was a highly self-selected group, in that each patient had chosen to travel to the hospital for care. Additionally, even though all patients responded completely to the two validated surveys, one of the weaknesses of this study is that the questionnaires were administered orally owing to low literacy rates. Similar to previous studies among populations with low literacy, questionnaires were administered during interviews with trained clinical staff [29], who were aware that the patients were presenting for POP evaluation, but to limit confirmation bias, they were unaware of their physical examination findings.
Overall, our findings underscore the severity of the effect of pelvic floor symptoms on quality of life and sexual health among this young patient population in DRC. Our findings emphasize the importance of outreach, so that potentially earlier and more widely available treatments can be a priority for women with POP.
Data Availability
The data that support the findings of this study are available from the corresponding author, AW, upon reasonable request.
References
Hendrix SL, Clark A, Nygaard I, Aragaki A, Barnabei V, McTiernan A. Pelvic organ prolapse in the Women’s Health Initiative: gravity and gravidity. Am J Obstet Gynecol. 2002;186:1160–1166. https://doi.org/10.1067/mob.2002.123819.
Nygaard I, Bradley C, Brandt D. Pelvic organ prolapse in older women: prevalence and risk factors. Obstet Gynecol. 2004;104:489–97. https://doi.org/10.1097/01.AOG.0000136100.10818.d8.
Walker GJA, Gunasekera P. Pelvic organ prolapse and incontinence in developing countries: review of prevalence and risk factors. Int Urogynecol J. 2011;22:127–35. https://doi.org/10.1007/s00192-010-1215-0.
Scherf C, Morison L, Fiander A, Ekpo G, Walraven G. Epidemiology of pelvic organ prolapse in rural Gambia, West Africa. BJOG. 2002;109:431–6. https://doi.org/10.1111/j.1471-0528.2002.01109.x.
Brækken IH, Majida M, Ellström Engh M, Holme IM, Bø K. Pelvic floor function is independently associated with pelvic organ prolapse. BJOG. 2009;116:1706–14. https://doi.org/10.1111/j.1471-0528.2009.02379.x.
Woodman PJ, Swift SE, O’Boyle AL, et al. Prevalence of severe pelvic organ prolapse in relation to job description and socioeconomic status: a multicenter cross-sectional study. Int Urogynecol J. 2006;17:340–5. https://doi.org/10.1007/s00192-005-0009-2.
Nygaard IE, Shaw JM. Physical activity and the pelvic floor. Am J Obstet Gynecol. 2016;214:164–71. https://doi.org/10.1016/j.ajog.2015.08.067.
Belayneh T, Gebeyehu A, Adefris M, Rortveit G, Awoke T. Pelvic organ prolapse in Northwest Ethiopia: a population-based study. Int Urogynecol J. 2020;31:1873–81. https://doi.org/10.1007/s00192-019-04196-1.
World Fertility Patterns 2015 – Data Booklet (ST/ESA/ SER. A/370). United Nations, Department of Economic and Social Affairs, Population Division. 2015.
Islam RM, Oldroyd J, Rana J, Romero L, Karim MN. Prevalence of symptomatic pelvic floor disorders in community-dwelling women in low and middle-income countries: a systematic review and meta-analysis. Int Urogynecol J. 2019;30:2001–11. https://doi.org/10.1007/s00192-019-03992-z.
Ntakwinja M, Borazjani A, Vodusek Z, Tambwe AM, Mukwege D. Surgical management of pelvic organ prolapse in a high-volume resource-limited setting. Int J Gynecol Obstet. 2022;156:145–50. https://doi.org/10.1002/ijgo.13684.
Fatton B, Letouzey V, Lagrange E, Mares P, Jacquetin B, de Tayrac R. Validation linguistique en français de la version courte du questionnaire sur la sexualité (PISQ-12) chez les patientes présentant un prolapsus génital et/ou une incontinence urinaire. J Gynecol Obstet Biol Reprod. 2009;38:662–7. https://doi.org/10.1016/j.jgyn.2009.02.008.
De Tayrac R, Deval B, Fernandez H, Marès P. Development of a linguistically validated French version of two short-form, condition-specific quality of life questionnaires for women with pelvic floor disorders (PFDI-20 and PFIQ-7). J Gynecol Obstet Biol Reprod. 2007;36:738–48. https://doi.org/10.1016/j.jgyn.2007.08.002.
Rogers RG, Coates KW, Kammerer-Doak D, Khalsa S, Qualls C. A short form of the Pelvic Organ Prolapse/Urinary Incontinence Sexual Questionnaire (PISQ-12). Int Urogynecol J. 2003;14:164–8. https://doi.org/10.1007/s00192-003-1063-2.
Rogers RG, Kammerer-Doak D, Villarreal A, Coates K, Qualls C. A new instrument to measure sexual function in women with urinary incontinence or pelvic organ prolapse. Am J Obstet Gynecol. 2001;184:552–8. https://doi.org/10.1067/mob.2001.111100.
Johnson K, Scott J, Rughita B, et al. Association of sexual violence and human rights violations with physical and mental health in territories of the Eastern Democratic Republic of the Congo. JAMA. 2010;304:553–62. https://doi.org/10.1001/jama.2010.1086.
Larsudd-Kåverud J, Gyhagen J, Åkervall S, Molin M, Milsom I, Wagg A, Gyhagen M. The influence of pregnancy, parity, and mode of delivery on urinary incontinence and prolapse surgery—a national register study. Am J Obstet Gynecol. 2023;228:61.e1–e13. https://doi.org/10.1016/j.ajog.2022.07.035.
Gjerde JL, Rortveit G, Adefris M, Mekonnen H, Belayneh T, Blystad A. The lucky ones get cured: health care seeking among women with pelvic organ prolapse in Amhara Region, Ethiopia. PLoS One 2018;13:e0207651. https://doi.org/10.1371/journal.pone.0207651.
Gjerde JL, Rortveit G, Muleta M, Adefris M, Blystad A. Living with pelvic organ prolapse: voices of women from Amhara region, Ethiopia. Int Urogynecol J. 2017;28:361–6. https://doi.org/10.1007/s00192-016-3077-6.
Wusu-Ansah OK, Opare-Addo HS. Pelvic organ prolapse in rural Ghana. Int J Gynecol Obstet. 2008;103:121–4. https://doi.org/10.1016/j.ijgo.2008.06.014.
Ellerkmann RM, Cundiff GW, Melick CF, Nihira MA, Leffler K, Bent AE. Correlation of symptoms with location and severity of pelvic organ prolapse. Am J Obstet Gynecol. 2001;185:1332–8. https://doi.org/10.1067/mob.2001.119078.
Barber MD, Visco AG, Wyman JF, Fantl JA, Bump RC. Sexual function in women with urinary incontinence and pelvic organ prolapse. Obstet Gynecol. 2002;99:281–9. https://doi.org/10.1016/S0029-7844(01)01727-6.
Handa VL, Cundiff G, Chang HH, Helzlsouer KJ. Female sexual function and pelvic floor disorders. Obstet Gynecol. 2008;111:1045–52. https://doi.org/10.1097/AOG.0b013e31816bbe85.
Weber AM, Walters MD, Schover LR, Mitchinson A. Sexual function in women with uterovaginal prolapse and urinary incontinence. Obstet Gynecol. 1995;85:483–7. https://doi.org/10.1016/0029-7844(94)00434-F.
Rogers GR, Villarreal A, Kammerer-Doak D, Qualls C. Sexual function in women with and without urinary incontinence and/or pelvic organ prolapse. Int Urogynecol J. 2001;12:361–5. https://doi.org/10.1007/s001920170012.
Özel B, White T, Urwitz-Lane R, Minaglia S. The impact of pelvic organ prolapse on sexual function in women with urinary incontinence. Int Urogynecol J. 2006;17:14–7. https://doi.org/10.1007/s00192-005-1327-0.
Shrestha B, Onta S, Choulagai B, et al. Women’s experiences and health care-seeking practices in relation to uterine prolapse in a hill district of Nepal. BMC Womens Health 2014;14:20. https://doi.org/10.1186/1472-6874-14-20.
Rahman A, Toubia N. Female genital mutilation: a guide to laws and policies worldwide. Zed Books, London; 2000.
Goba GK, Legesse AY, Zelelow YB, et al. Reliability and validity of the Tigrigna version of the Pelvic Floor Distress Inventory-Short Form 20 (PFDI-20) and Pelvic Floor Impact Questionnaire-7 (PFIQ-7). Int Urogynecol J. 2019;30:65–70. https://doi.org/10.1007/s00192-018-3583-9.
Author information
Authors and Affiliations
Contributions
M. Ntakwinja: project development, data collection, manuscript writing; A. Werth: data analysis, manuscript writing; A. Borazjani: data analysis, manuscript writing; C. Iglesia: manuscript writing; K.J. Williams: manuscript writing; D. Mukwege: project development, manuscript editing.
Corresponding author
Ethics declarations
Conflicts of interest
M. Ntakwinja: none; A. Werth: none; A. Borazjani: patents related to treatments and diagnostics for pelvic floor disorders, consultant and equity shareholder in Cosm Medical Corp; C. Iglesia: SGS executive board, NICHD PFDN pelvic floor disorders network advisory board chair, US FDA OBGYN Devices Panel special government employee, medical advisory board member for the Patty Brisben Foundation, Editorial board for Urogynecology journal and OBGManagement journal; K. Williams: none; D. Mukwege: none.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ntakwinja, M., Werth, A., Borazjani, A. et al. Pelvic floor symptoms among premenopausal women with pelvic organ prolapse in the Democratic Republic of the Congo. Int Urogynecol J 35, 103–108 (2024). https://doi.org/10.1007/s00192-023-05670-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-023-05670-7