Abstract
Introduction and hypothesis
The differential impact of specific pelvic organ prolapse (POP) surgery on sexual activity and function is unknown. Our primary aim was to analyse sexual inactivity and function in women with symptomatic advanced stages of POP and the changes incurred after laparoscopic or vaginal mesh surgery.
Methods
We performed a secondary analysis of sexual outcomes of a previously published randomised controlled trial comparing laparoscopic sacrocolpopexy/cervicopexy (LSC-Cx) and anterior vaginal mesh (AVM) in 120 women (60/group) with symptomatic anterior POP stage ≥ 3 and apical ≥ 2. Sexual activity and function were assessed preoperatively and 1 and 2 years postoperatively using the Pelvic Organ Prolapse/Incontinence Sexual Questionnaire-IUGA-Revised (PISQ-IR).
Results
Sexual activity was recovered in 42.9% of non-sexually active (NSA) women 1 year postoperatively, mainly in women with higher preoperative POP-related subscale scores of the PISQ-IR, which indicated a negative preoperative sexuality by POP. Recovery of sexual activity was greater after LSC-Cx, albeit not significantly (2 years: 35.5% AVM vs. 45% LSC-Cx). Among sexually active (SA) women preoperatively remaining SA postoperatively, the difference in the mean PISQ-IR summary score significantly improved [mean baseline difference – 2 years; all: 0.3 (95% CI 0.1 to 0.5) p = 0.001; AVM 0.19 (95% CI –0.1 to −0.5) p > 0.05; LSC-Cx 0.37 (95% CI 0.1 to 0.7) p = 0.003]. Preoperative dyspareunia was significantly reduced after LSC-Cx (baseline: 24.6%, 2 years: 9.8%, p = 0.0448), but not after AVM (baseline: 20.7%, 2 years: 18.2%, p = 0.7385).
Conclusions
Most women reported improved sexual activity and function 2 years after LSC-Cx or AVM, mainly because of enhanced POP-related subscales in both NSA and SA women. Recovery of sexual activity and improved sexual function were greater after LSC-Cx compared to AVM, likely related to less postoperative dyspareunia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The sexuality of women with pelvic organ prolapse (POP) is underexplored, especially in those with POP with a high risk of recurrence (≥ stage 3) [1]. In addition, the impact of POP surgery on sexual activity and function is controversial in the literature [2], with some studies reporting improvement [3,4,5,6], while others showed no differences [7, 8] or worse sexuality [9] after surgery. These different results may be the consequence of the great variety of surgical techniques (vaginal, abdominal, with or without mesh, with or without anti-incontinence surgery) and the diversity of the measuring instruments used.
Although different randomised controlled trials have assessed dyspareunia after POP surgery with or without mesh, not all used validated questionnaires on sexual function or evaluated sexual activity pre- and postoperatively [1]. Therefore, the ICI 2017 recommends the use of validated questionnaires to measure sexual function in women before and after prolapse surgery and to report sexual activity and dyspareunia rates pre- and post-intervention in all patients [1] to determine the positive and negative effects of different surgical procedures on female sexuality. With this knowledge we can provide advice to women according to their sex life at the time of treatment planning and their expectations after surgery.
The PISQ-IR (Pelvic Organ Prolapse Questionnaire/Urinary Incontinence Sexual Function Questionnaire-IUGA Revised) is a validated evaluation tool which can be used clinically as well as in research for the assessment of sexuality in women with female pelvic floor disorders [10]. It is the only condition-specific questionnaire to assess both sexually active and sexually inactive women.
In this context, the objective of this analysis was to describe sexual activity and function preoperatively with PISQ-IR and longitudinally over a 2-year follow-up period in women who required POP surgery with mesh [11]. In women with advanced stages of POP (≥ stage 3 anterior vaginal wall descent, ≥ stage 2 apical descent), we hypothesised that POP impaired sexual activity and function, and we also hypothesised that postoperative female sexuality will significantly improve but will differ between women undergoing laparoscopic sacrocolpopexy/cervicopexy (LSC-Cx) versus anterior vaginal mesh (AVM) repair.
Materials and methods
The present study was a planned secondary analysis of the sexual activity and function outcomes of a prospective randomised controlled trial (RCT) performed from January 2011 to March 2017 [11]. We included women with symptomatic POP requiring surgery (primary or recurrence), with anterior vaginal wall descent (≥ stage 3) and apical descent (≥ stage 2). Patients were randomly allocated into two groups according to the type of POP surgery with mesh: AVM (Elevate® Anterior and Apical; AMS, Minnetonka, MN, USA) or LSC-Cx, using a computer-generated randomisation list by order of inclusion.
The exclusion criteria included women under 21 years of age, with comorbidity or at high anaesthetic risk requiring a particular approach, the inability to comprehend questionnaires or attend follow-up visits, previous sacrocolpopexy or a vaginal mesh procedure and a history of pelvic radiotherapy.
The study protocol was approved by the Ethics Committee of our institution (number: 2010/5989), and the trial was registered at ClinicalTrials.gov (Identifier: NCT01097200). Written informed consent was obtained from all patients.
The primary aims and detailed methodology of this randomised prospective study have been described elsewhere [11].
Patients were visited preoperatively and 1 and 2 years postoperatively by two urogynaecologists not related to the surgery. In all the visits, the patients completed the same validated self-administered Spanish versions of the following questionnaires for urinary, anal and POP symptoms: the International Consultation on Incontinence Questionnaire-Short Form (ICIQ-UI-SF) [12] with scores ranging from 0 to 21 points and the Pelvic Floor Distress Inventory-20 (PFDI-20) [13], which has a global score ranging from 0 to 300. Sexual activity and function were evaluated with the PISQ-IR, which is a condition-specific questionnaire validated in Spanish [10], including 14 items and 6 subscales addressed to sexually active (SA) patients and 5 items and 4 subscales for non-sexually active (NSA) patients (Appendix 1). The scores were calculated with the mean calculation method. Higher scores in SA patients indicate better sexual function, while in NSA patients, these indicate a greater impact on sexual function. Global sexual function was analysed according to the single summary score of PISQ-IR only available for SA women [14]. To date, a clinically meaningful difference for the PISQ-IR summary score has not been quantified. However, a cutoff of 2.68 for the PISQ-IR summary score allowed sexual dysfunction to be diagnosed in SA women with pelvic floor disorders with a sensitivity of 90% and a specificity of 71% [15]. In the present analysis, dyspareunia was considered for both SA and NSA women: in SA women, when the patient reported “sometimes”, “usually” or “always” to PISQ-IR question 11: “How often do you feel pain during sexual intercourse?” or coital activity avoidance due to pain. In NSA women, dyspareunia was considered when the patient reported pain as a cause of inactivity.
The medical history and physical examination of all the patients were obtained including the POP-Q classification, modified Oxford scale and classification of the ICS-IUGA [16].
Sample size calculation
The sample size of 120 patients (60/arm) was calculated according to the main variable (anatomical success), as described by Bataller et al. [11]. In the present study, we analysed the results of sexual activity and function pre- and postoperatively using the PISQ-IR. The validation studies of the PISQ-IR do not define the minimal important difference; therefore, the adequacy of the sample was evaluated according to the magnitude of the differences to be detected. Although this was a secondary analysis, the sample allowed the detection of medium changes (Cohen), with differences equivalent to standard deviations of 0.53, with a β error of 0.2 and an α error of 0.05, in the PISQ-IR scores for each subgroup.
Statistical analysis
The results are presented as absolute and relative frequencies for categorical variables and as mean and standard deviations for the continuous variables. Fisher’s exact test was used for the comparative analysis of groups for categorical variables. The continuous variables were analysed with Student’s t-test if the variables followed normal distribution or the Mann-Whitney U test if the variables did not follow normal distribution. The normality of the distribution of the variables was evaluated with the Kolmogorov-Smirnov test. Comparisons between pre- and postoperative data in Table 3 were analysed in a paired t-test for the continuous variables and chi-square for proportions. The correlations were evaluated with Spearman’s rank correlation coefficient (rho). The level of significance was set at p < 0.05. All the analyses were performed with the SPSS software package (19.0 version, SPSS Inc., Chicago, IL).
Results
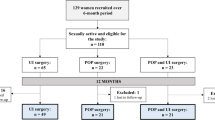
The anatomical results, demographic data, intra-surgical variables and complications 1 year postoperatively were published in a previous article [11]. Figure 1 shows a CONSORT flow diagram of the study. Table 1 shows the baseline characteristics of the 120 women included in the study according to sexual activity or not, with no statistically significant differences between surgical approaches [11]. Concomitant diseases (hypertension, diabetes, neurological disorders or mental health disorders) were analysed, showing no differences between the two approaches or between SA and NSA women.
Sexual activity
The sexual activity at baseline and follow-up, according to question 1 of the PISQ-IR, is shown in Fig. 2. Missing preoperative patient-reported sexual outcomes were low (5 women did not answer the PISQ-IR) and the follow-up rate at 1 and 2 years was high (95.8% and 88.3%, respectively).
The rate of NSA women at baseline was 50.4%. Figure 3 shows the reasons for not being SA at baseline and at follow-up. Among the women who were NSA preoperatively, those who became SA 1 year postoperatively differed from those who remained NSA. Women in whom sexual activity recovered showed statistically significantly higher mean scores in the NSA-condition specific (2.2; SD 0.7; 95% CI 1.9–2.5 vs. 1.6; SD 0.7; 95% CI 1.4–1.8), the mean NSA-global quality rating (2.9; SD 0.7; 95% CI 2.6–3.2 vs. 2.1; SD 1.1; 95% CI 1.7–2.5) and the mean NSA-condition impact subscales (2.7; SD 0.8; 95% CI 2.4–3 vs. 1.8; SD 1; 95% CI 1.5–2.1), with no differences between the two surgical approaches.
Figure 4 shows the responses in regard to fear of urine or stool incontinence and/or POP causing the avoidance/restriction of sexual activity. No differences in responses were observed between the surgical approaches (data not shown).
Finally, the mean score of the PISQ-IR subscales for NSA women, divided according to the AVM or LSC-Cx approach, are shown in Table 2. We observed a statistically significant improvement in the mean PISQ-IR scores in two subscales: condition-specific reasons for not being active and condition impact on sexual quality, without differences between approaches.
Sexual function
On the other hand, the sexual function of the SA women was also analysed in the present study by the mean score of the PISQ-IR subscales (Table 2). In SA women, there was a statistically significant improvement in the mean PISQ-IR score in three subscales: global quality rating of sexual quality, assessment of condition-specific impacts on activity and condition-specific impact on sexual quality, without differences between approaches.
The mean PISQ-IR summary score for SA women preoperatively (N = 57) was 3.09 (SD0.49; 95% CI 2.9–3.1). Significant improvement was observed in the mean values at 1 (N = 74) (3.26; SD 0.4; 95% CI 3.2–3.4) and 2 years of follow-up (N = 66) (3.27; SD 0.34; 95% CI 3.2–3.4), with no differences between the surgical approaches. The rate of preoperative female sexual dysfunction (FSD) was 29.8% (N = 17), with no statistically significant differences on comparing LSC-Cx (23.5%) to AVM (39.1%). Among women with preoperative FSD, one continued to have FSD and ten showed improvement 2 years postoperatively (the remaining 6 women became NSA).
To assess the evolution of global sexual function after surgery with the PISQ-IR summary score, we focused on SA women at baseline who maintained sexual activity during follow-up (Table 3). Among these women (N = 42), the PISQ-IR summary score 2 years postoperatively improved in 32 (76.2%) (79.2% LSC-Cx vs. 72.2% AVM), while 1 woman (2.4%) had the same score pre- and postoperatively (4.1% LSC-Cx), and 9 women (21.4%) scored lower (16.7% LSC-Cx vs. 27.8% AVM). Moreover, the overall FSD rate among these 42 women was 4.8% (N = 2). In addition, at 2 years postoperatively, nine women also became NSA for different reasons: POP- or surgery-related (33.3%) and not related to surgery or POP (66.6%), such as no interest or partner health issues. Analysis of the FSD rate and the rate of women who ceased sexual activity comparing LSC-Cx to AVM was not performed because of the small sample size at the 2-year follow-up in these subgroups.
Finally, dyspareunia was analysed separately, comparing LSC-Cx to AVM pre- and postoperatively (Fig. 5). Preoperative dyspareunia was significantly reduced after LSC-Cx but not after AVM.
Discussion
The present study showed POP to have an important impact on sexual activity and function in a cohort of women with POP with a high risk of recurrence (≥ stage 3). Both outcomes showed significant postoperative improvement, which was maintained at 2 years of follow-up. The positive effect of POP correction only affected the POP-related subscales in both NSA and SA women. However, the global quality rating of sexual quality improved exclusively in SA women. Resolution of preoperative dyspareunia was statistically significant at 2 years of follow-up following the LSC-Cx approach but not after AVM. The recovery of sexual activity and the improvement of sexual function tended to be greater after LSC-Cx compared to AVM, but no statistically significant differences were found between the two approaches in relation to the overall sexual activity and function scores.
In an attempt to infer how our results confirm or contrast with published literature, there were three main obstacles. First, the populations studied in the literature differ; that is, most of the studies on female sexuality after POP surgery included women with POP stage 2 and very few [11, 17] focused on a population with a high risk of recurrence (≥ stage 3). The second obstacle was the variety of surgical approaches described, with only a few studies [6, 11, 18, 19] comparing vaginal and abdominal mesh and analysing sexual outcomes, essentially focused on anatomical outcomes and dyspareunia. Lastly, only two studies [4, 20] used the PISQ-IR to assess sexual activity and function after POP surgery. In contrast, most of the studies [3, 6, 17, 21,22,23] used the PISQ-12 as a validated condition-specific questionnaire, but this questionnaire does not allow assessment of NSA patients. Moreover, some studies used generic sexual function questionnaires [18, 19], which do not assess changes in sexual health specifically caused by POP, and others used non-validated questionnaires.
Preoperative sexual activity
In our population, half of the women with POP with a high risk of recurrence were NSA. The main reason for their sexual inactivity was POP and/or bladder/bowel problems. Nearly two-thirds of NSA women reported avoidance of sexual activity (5% a little; 26% some; 34.5% a lot) due to fear of vaginal bulging and/or leaking urine and/or stool. These results are in contrast with a multicentre prospective cohort study in women with advanced POP awaiting surgery [4]. This study found “no interest” as the first reason for sexual inactivity and “no partner” as a second, which were the second and the third reasons, respectively, in the present study. Moreover, this multicentre study described a lower rate of NSA women who avoided sexual activity because of fear of vaginal bulging and/or leaking urine and/or stool (34%). These discrepancies may be explained by several factors, particularly different study populations. The NSA women in the above-mentioned study were older (mean age: 69.26 years) compared with our sample (mean age: 63.3 years). In our study, 72.4% of NSA women had a partner compared to only 59.1% in the multicentre study. In addition, they also included women with POP stage 2 (16.9% of NSA), whereas only women with ≥ stage 3 were included in our study.
Preoperative sexual function
Among SA women at baseline, nearly one-third were identified as having sexual function impairment. Furthermore, three-fifths reported some proportion of restriction of sexual activity (14% a little, 14% some, 30% a lot) due to fear of vaginal bulging and/or leaking of urine and/or stool. This indicates that POP with a high risk of recurrence may cause sexual inactivity as well as sexual dysfunction. This was consistent with a qualitative study by Roos et al. with semi-structured interviews with partnered SA women scheduled to undergo pelvic floor surgery [24]. Among women planning to undergo POP surgery, 12% evaluated their sex life positively with no POP-related impact. However, 29% of women gave a negative evaluation of their sex life, and 59% described an overall good sex life but with negative aspects. The most common sexual concerns due to POP were related to body image issues, discomfort or obstruction by POP and mental distraction because of POP. Similar to this study, a recent qualitative study [25], based on focus groups of women seeking treatment for POP (stage 2–4), reported comparable results. Women frequently described the impact of POP on sexual function, mainly due to two limitations. First, sexual activities exacerbated their physical discomfort related to POP: “It was uncomfortable for me. It was painful.” Second, the presence of POP negatively affected their body image and caused a decrease in desire: “Affects your sex life because you are always thinking about it.” This study added a conceptual framework for important patient outcomes for POP, which confirmed improvement in sexual function as the third outcome ranked: “I want to be intimate without feeling embarrassed.”
Postoperative sexual activity
Overall, in our sample, women reported a significant increase in sexual activity rates after surgery with mesh. Other studies have shown different results. Two RCTs and one prospective cohort study found no changes in sexual activity after different POP surgery approaches: native tissue apical repair [3], robot-assisted abdominal mesh for apical POP [21] or vaginal mesh for anterior POP [17]. Only the last study [17] focused on women with POP ≥ stage 3, although the patients were older (67 ± 9 years) than those in our sample. In addition, a recent meta-analysis [22] of RCTs on vaginal mesh repair versus native tissue repair for POP described a slight overall decrease in sexual activity after both surgical approaches. However, the findings of this meta-analysis should be interpreted with caution because of potential biases due to several limitations of the studies reviewed.
In our sample, there was a decrease, albeit not statistically significant, in sexual activity on comparing the first- and second-year follow-up. Furthermore, the rate of sexual activity was still significantly better than before surgery. Moreover, at the 2-year follow-up, less than one-fourth of NSA women reported avoidance of sexual activity (2.5% a little; 10% some; 2.5% a lot) due to fear of vaginal bulging and/or leaking of urine and/or stool, regardless of the type of surgery, representing a significant reduction of sexual avoidance after POP correction, similar to other studies [3, 21, 23]. In addition, in regard to the PISQ-IR subscales, this positive impact of surgical treatment on NSA women was also seen in POP-related subscales, which significantly improved postoperatively.
In the present study, sexual activity was postoperatively recovered in four out of nine NSA women at 1 year. However, women reporting recovery of sexual activity after surgery were mainly those who preoperatively described a higher negative effect of POP on their sexuality. In addition, the recovery of sexual activity was greater after LSC-Cx than after AVM, albeit not significantly (2-year follow-up: 45% vs. 35.5%, respectively). In a prospective multicentre cohort study [18] comparing laparoscopic sacral hysteropexy to vaginal mesh hysteropexy, more women reported sexual activity 1 year after an abdominal approach (abdominal approach: 48% at baseline vs. 72% at 1 year; vaginal approach: 44% at baseline vs. 44% at 1 year; p = 0.03). These differences between the two approaches may be explained by two factors: first, the baseline differences between groups, with the vaginal group being older and with a greater number of postmenopausal women, and second, the lack of randomisation, that is, patients/surgeons may have been more likely to accept/recommend vaginal mesh for women less likely to be SA in the future.
Postoperative sexual function
In our sample, an overall improvement was observed regarding sexual function after surgery with abdominal or vaginal mesh in women with a high risk of recurrence. Postoperative improvement of sexual function not only affects the specific condition subscales but also the overall quality of life. Similar results were observed in a previous prospective cohort study [6] of patients undergoing abdominal (97% with mesh) and vaginal repairs (66% with mesh). The authors found that PISQ-12 scores improved significantly and in a similar manner in both the abdominal and vaginal approaches at all time points (p = 0.0001).
In addition, global FSD rates reduced after 2 years of follow-up in the present study and at least half of the women with FSD at baseline improved. Furthermore, restriction of sexual activity due to fear of vaginal bulging and/or urine and/or stool leakage only affected 6% of the SA women (0% a little; 4.5% some, 1.5% a lot), demonstrating a relevant decrease after POP correction. This improvement in sexual function was also found in the studies by Zanten and Ko [21, 23], both of which compared PISQ-12 scores after robotic sacrocolpopexy and laparoscopic sacrocervicopexy. Nevertheless, their analysis of the PISQ-12 domains only showed a statistically significantly difference in sexual dysfunction due to vaginal bulging. One limitation of these two studies was the low response rate (76.4% in Zanten et al. and 57.3% in Ko et al.).
Finally, in relation to the impact of the different surgical approaches on sexual function, Gutman et al. [18] found similar improvements in sexual function in the Female Sexual Function Index, comparing laparoscopic sacral hysteropexy to vaginal mesh hysteropexy after adjusting for age, parity, POPQ, C and Bp, menopausal status and baseline score. Nevertheless, the domain of satisfaction showed greater improvement in women who chose to undergo the laparoscopic compared to the vaginal approach. However, the specific impact due to improvement of POP symptoms was unknown, as they used a generic sexual health questionnaire instead of a condition-specific questionnaire. This instrument was also used in a multicentre randomised controlled trial [19] comparing LSC-Cx and AVM. There were no differences in sexual function between groups (p = 0.248) among SA women at 1 year of follow-up, but the rate of dyspareunia or apareunia was significantly higher after the vaginal than laparoscopic approach (30% versus 14%, respectively, p = 0031). This was also confirmed by our results. Despite similar anatomical and symptom correction after both approaches [11], pre- and postoperative dyspareunia rates after AVM were similar, whereas preoperative dyspareunia was significantly reduced after LSC-Cx. Vaginal mesh surgery may affect vaginal sensitivity because of scars on the vaginal walls, shortening of the vagina, retraction and fibrosis or mesh exposure [11, 18, 19, 26, 27], which could explain the differences between the two types of surgery.
Strengths and limitations
The main strengths of our study lie in the use of the condition-specific PISQ-IR questionnaire to assess not only sexual function but also sexual activity. To our knowledge, this is the first RCT comparing these two surgical techniques focused on a subgroup of patients with a high risk of recurrence (≥ stage 3), with minimal heterogeneity of the group to analyse female sexuality. Other strengths of the present study include the long-term follow-up, a lower than expected loss to follow-up rate in an RCT as well as a higher rate of response related to sexual data compared to other studies [21, 23].
One limitation for replication of the present study is that the AMS Elevate® system is no longer commercially available, although other vaginal meshes have similar characteristics. We also acknowledge that although the 1- and 2-year follow-up rates of 95.8% and 88.3% were high, this study may be insufficiently powered to identify statistically significant differences in sexual function measures between groups. A further limitation for sexual evaluation may be that qualitative assessment associated with the condition-specific questionnaire was not performed.
The implications of these new data
Despite the use of POP surgery worldwide, data on sexual activity and function are scarce, especially after mesh surgery. The results of this study provide better knowledge of sexual activity and function in women with POP with a high risk of recurrence. To the best of our knowledge, it is the first study in the literature using the PISQ-IR to assess female sexuality after POP mesh surgery via an abdominal and vaginal approach. Our data provide information to properly advise women according to their sex life at the time of planning the treatment, as we found that women who reported sexual inactivity or sexual function impairment due to POP may expect improvement after surgery. However, other dimensions (partner-related, desire, orgasm, etc.) as a cause of inactivity or FSD may not change after surgery.
At present, there is great controversy regarding the use of mesh in POP surgery; therefore, this surgical approach is very limited worldwide, especially the vaginal approach. It is, therefore, important to extend the use of the PISQ-IR to explore the impact of other surgical approaches that are currently performed on female sexuality. It is likely that with extrapolation of our results to other surgical approaches similar results would be found, but this should be confirmed in future studies.
Conclusions
In summary, after vaginal or abdominal mesh surgery to treat patients with POP with a high risk of recurrence, most women reported improved female sexuality at 2 years of follow-up. However, the positive impact after POP correction was mainly due to enhanced POP-related subscales in both NSA and SA women. The recovery of sexual activity and the improvement of sexual function tended to be greater after LSC-Cx than after AVM, which may be related to the resolution of preoperative dyspareunia. Considering these findings, an abdominal surgical approach should be considered in women with POP undergoing mesh surgery who wish to maintain or recover sexual activity and better preserve female sexuality. Further prospective RCTs with larger sample sizes and a longer follow-up are needed to confirm our findings.
Abbreviations
- POP:
-
Pelvic organ prolapse
- PISQ-IR:
-
Pelvic Organ Prolapse Questionnaire/Urinary Incontinence Sexual Function Questionnaire-IUGA Revised
- RCT:
-
Randomised controlled trial
- AVM:
-
Anterior vaginal mesh
- LSC-Cx:
-
Laparoscopic sacrocolpopexy/cervicopexy
- ICIQ-UI-SF:
-
International Consultation on Incontinence Questionnaire-Short Form
- PFDI-20:
-
Pelvic Floor Distress Inventory-20
- SA:
-
Sexually active
- NSA:
-
Non-sexually active
- FSD:
-
Female sexual dysfunction
References
Maher C, Baessler K, Barber M, Cheon C, Consten E, Cooper K, et al (2016) Pelvic organ prolapse surgery. In: Incontinence Abrams P, Cardozo L, Wagg A, Wein A, editors. International Consultation on Incontinence, 6th edition, Tokyo; pp 1855–1991. ISBN: 978–0–9569607-3-3.
Ghielmetti T, Kuhn P, Dreher EF, Kuhn A. Gynaecological operations: do they improve sexual life? Eur J ObstetGynecolReprodBiol. 2006;129(2):104–10. https://doi.org/10.1016/j.ejogrb.2006.05.026.
Lukacz ES, Warren LK, Richter HE, et al. Quality of life and sexual function 2 years after vaginal surgery for prolapse. ObstetGynecol. 2016;127(6):1071–9. https://doi.org/10.1097/AOG.0000000000001442.
Anglès-Acedo S, Ros-Cerro C, Espuña-Pons M, Valero-Fernandez EM. Sexual activity and function of women with severe pelvic organ prolapse subjected to a classical vaginal surgery. A multicentre study. ActasUrológicasEspañolas (English Edition). 2019;43(7):389–95. https://doi.org/10.1016/j.acuro.2019.02.003.
Pan K, Cao L, Ryan NA, et al. Laparoscopic sacral hysteropexy versus laparoscopic sacrocolpopexy with hysterectomy for pelvic organ prolapse. Int Urogynecol J. 2016;27:93. https://doi.org/10.1007/s00192-015-2775-9.
Gupta P, Payne J, Killinger KA, et al. Analysis of changes in sexual function in women undergoing pelvic organ prolapse repair with abdominal or vaginal approaches. Int Urogynecol J. 2016;27:1919. https://doi.org/10.1007/s00192-016-3066-9.
Bartuzi A, Futyma K, Kulik-Rechberger B, Skorupski P, Rechberger T. Transvaginal Prolift(®) mesh surgery due to advanced pelvic organ prolapse does not impair female sexual function: a prospective study. Eur J ObstetGynecolReprodBiol. 2012;165(2):295–8. https://doi.org/10.1016/j.ejogrb.2012.07.011.
Occhino J. A, Trabuco E. C, Heisler C. A, Klingele C. J, Gebhart J. B (2011) Changes in vaginal anatomy and sexual function after vaginal surgery. Int Urogynecol J 22:799–804. https://doi.org/10.1007/s00192-011-1386-3.
Milani R, Salvatore S, Soligo M, Pifarotti P, Meschia M, Cortese M. Functional and anatomical outcome of anterior and posterior vaginal prolapse repair with prolene mesh. BJOG. 2005;112(1):107–11. https://doi.org/10.1111/j.1471-0528.2004.00332.x.
Mestre M, Lleberia J, Pubill J, Espuna-Pons M. Spanish version of the pelvic organ prolapse/urinary incontinence sexual questionnaire IUGA-revised (PISQ-IR): transcultural validation. Int Urogynecol J. 2017;28:1865–73. https://doi.org/10.1007/s00192-017-3312-9.
Bataller E, Ros C, Anglès S, et al. Anatomical outcomes 1 year after pelvic organ prolapse surgery in patients with and without a uterus at a high risk of recurrence: a randomised controlled trial comparing laparoscopic sacrocolpopexy/cervicopexy and anterior vaginal mesh. Int Urogynecol J. 2019;30:545. https://doi.org/10.1007/s00192-018-3702-7.
Espuña Pons M, Rebollo Álvarez P, Puig Clota M. Validation of the Spanish version of the international consultation on incontinence questionnaire-short form. A questionnaire for assessing the urinary incontinence. Med Clin. 2004;122:288–92. https://doi.org/10.1016/s0025-7753(04)74212-8.
Sanchez-Sanchez B, Torres-Lacomba M, Yuste-Sanchez MJ, et al. Cultural adaptation and validation of the pelvic floor distress inventory short form (PFDI-20) and pelvic floor impact questionnaire short form (PFIQ-7) Spanish versions. Eur J ObstetGynecolReprodBiol. 2013;170:281–5. https://doi.org/10.1016/j.ejogrb.2013.07.006.
Constantine ML, Pauls RN, Rogers RR, Rockwood TH. Validation of a single summary score for the prolapse/incontinence sexual questionnaire–IUGA revised (PISQ-IR). Int Urogynecol J. 2017;28(12):1901–7. https://doi.org/10.1007/s00192-017-3373-9.
Grzybowska ME, Futyma K, Wydra D. Identification of the pelvic organ prolapse/incontinence sexual questionnaire—IUGA revised (PISQ-IR) Cutoff scores for impaired sexual function in women with pelvic floor disorders. J Clin Med. 2020;9:13. https://doi.org/10.3390/jcm9010013.
Haylen BT, Maher CF, Barber MD, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic organ prolapse (POP). Int Urogynecol J. 2016;27(2):165–94. https://doi.org/10.1007/s00192-015-2932-1.
de Tayrac R, Brouziyne M, Priou G, et al. Transvaginal repair of stage III-IV cystocele using a lightweight mesh: safety and 36-month outcome. Int Urogynecol J. 2015;26:1147. https://doi.org/10.1007/s00192-015-2659-z.
Gutman RE, Rardin CR, Sokol E, et al. Vaginal and laparoscopic mesh hysteropexy for uterovaginal prolapse: a parallel cohort study. Am J ObstetGynecol. 2017;216(1):38.e1–38.e11. https://doi.org/10.1016/j.ajog.2016.08.035.
Lucot JP, Cosson M, Bader G, et al. Safety of vaginal mesh surgery versus laparoscopic mesh Sacropexy for cystocele repair: results of the prosthetic pelvic floor repair (PROSPERE) randomized controlled trial. Eur Urol. 2018;74(2):167–76. https://doi.org/10.1016/j.eururo.2018.01.044.
Pilka R, Gágyor D, Študentová M, Neubert D, Dzvinčuk P. Laparoscopic and robotic sacropexy: retrospective review of learning curve experiences and follow-up. CeskaGynekol. 2017;82(4):261–7.
van Zanten F, Brem C, Lenters E, et al. Sexual function after robot-assisted prolapse surgery: a prospective study. Int Urogynecol J. 2018;29:905–12. https://doi.org/10.1007/s00192-018-3645-z.
Liao SC, Huang WC, Su TH, et al. Changes in female sexual function after vaginal mesh repair versus native tissue repair for pelvic organ prolapse: a meta-analysis of randomized controlled trials. J Sex Med. 2019;16:633–9. https://doi.org/10.1016/j.jsxm.2019.02.016.
Ko YC, Yoo EH, Han GH, Kim YM. Comparison of sexual function between sacrocolpopexy and sacrocervicopexy. ObstetGynecol Sci. 2017;60(2):207–12. https://doi.org/10.5468/ogs.2017.60.2.207.
Roos AM, Thakar R, Sultan AH, Burger CW, Paulus AT. Pelvic floor dysfunction: women's sexual concerns unraveled. J Sex Med. 2014;11:743–52. https://doi.org/10.1111/jsm.12070.
Sung VW, Rogers RG, Barber MD, Clark MA. Conceptual framework for patient-important treatment outcomes for pelvic organ prolapse. NeurourolUrodyn. 2014;33(4):414–9. https://doi.org/10.1002/nau.22397.
Lakeman MM, Laan E, Roovers JP. The effects of prolapse surgery on vaginal wall sensibility, vaginal vasocongestion, and sexual function: a prospective single Centre study. NeurourolUrodyn. 2014;33(8):1217–24. https://doi.org/10.1002/nau.22491.
Maher C, Feiner B, Baessler K, Christmann-Schmid C, Haya N, Marjoribanks J. Transvaginal mesh or grafts compared with native tissue repair for vaginal prolapse. The Cochrane data base of systematic reviews. 2016;2:CD012079. https://doi.org/10.1002/14651858.CD012079.
Acknowledgements
We thank Dr. Lluís Aragonès for his contribution to the statistical analyses.
Funding
The Elevate® Anterior and Apical kits were donated by AMS as a grant for the study without any other support or supervision.
Author information
Authors and Affiliations
Contributions
S. Anglès-Acedo: data collection, data analysis, manuscript writing, manuscript editing.
C. Ros-Cerro: project development, data analysis, manuscript editing.
S. Escura-Sancho: data collection.
M.J. Palau-Pascual: data collection.
E. Bataller-Sánchez: project development.
M. Espuña-Pons: project development, manuscript editing.
F. Carmona-Herrera: project development.
Corresponding author
Ethics declarations
Financial disclaimers/conflict of interest
Dr. Bataller was a consultant for AMS (Astora Women’s Health). The remaining authors claim no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Conference Presentations
This study was presented as an oral presentation in the following congresses:
(1) 12th Annual Congress Euroepean Urogynaecological Association, Tel Aviv, 16–18 October 2019
(2) 22nd Annual Congress of the European Society for Sexual Medicine, Prague, 23–25 January 2020. Best Presentation Award in the topic of Reconstructive Surgery.
This study was presented as a poster in the following meeting:
(3) 35 Edición Nacional Formación SEGO (Sociedad Española de Ginecología y Obstetrícia), Málaga, 25–28 June 2019
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Anglès-Acedo, S., Ros-Cerro, C., Escura-Sancho, S. et al. Sexual activity and function in women with advanced stages of pelvic organ prolapse, before and after laparoscopic or vaginal mesh surgery. Int Urogynecol J 32, 1157–1168 (2021). https://doi.org/10.1007/s00192-020-04406-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-020-04406-1