Abstract
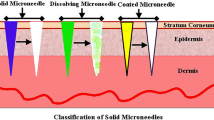
In this work, solid silicon (Si) microneedles with a higher aspect ratio and sharper tips are fabricated. Tetramethylammonium Hydroxide (TMAH) etching factors have been optimized and used to fabricate long and tapered microneedles. The needles thus fabricated are found to be suitable for transdermal drug delivery applications. The optimized etching factors include varying the concentration of TMAH, etching time, etching rates, temperature, and window size of optical mask. It is found that by increasing the window size, the etch rates in both vertical and lateral directions increase extensively. However, on increasing the temperature beyond 90 °C, etching becomes rapid and uncontrollable. In order to obtain microneedles with high aspect ratio, sample placement in the glass boat of TMAH setup and TMAH concentration should be manipulated to attain a higher etch rate in vertical direction compared to the lateral one. Solid silicon microneedles with an average height of 158 μm, base width of 110.5 μm, aspect ratio of 1.43, tip angle of 19.4° and tip diameter of 0.40 μm are successfully fabricated. A microhardness value of 44.4 (HRC) was obtained for the fabricated Si microneedles. This is 52.2 times higher than the skin Ultimate Tensile Strength (UTS), which makes insertion of microneedles through the skin safer and easier without any breakage.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Prausnitz MR (2004) Microneedles for transdermal drug delivery. Adv Drug Deliv Rev 56:581–587
Prausnitz MR, Langer R (2008) Transdermal drug delivery. Nat Biotechnol 26:1261–1268
Guy RH, Hadgraft J (2003) Transdermal Drug Delivery. Marcel Dekker, New York
Williams A (2003) Transdermal and topical drug delivery. Pharmaceutical Press, London
Prausnitz MR, Mitragotri S, Langer R (2004) Current status and future potential of transdermal drug delivery. Nat Rev Drug Discov 3:115–124
Henry S, McAllister DV, Allen MG, Prausnitz MR (1998) Microfabricated microneedles: a novel approach to transdermal drug delivery. J Pharm Sci 87:922–925
Lin L, Pisano AP (1999) Silicon-processed microneedles. IEEE J Microelectromech Syst 8:78–84
Tao SL, Desai TA (2003) Microfabricated drug delivery systems: from particles to pores. Adv Drug Deliv Rev 55:315–328
McAllister DV, Wang PM, Davis SP, Park JH, Canatella PJ, Allen MG, Prausnitz MR (2003) Microfabricated needles for transdermal delivery of macromolecules and nanoparticles: fabrication methods and transport studies. Proc Natl Acad Sci U S A 100:13755–13760
Van der maaden K, Sekerdag E, Schipper P, Kersten G, Jiskoot W, Bouwstra J (2015) Layer-by-layer assembly of inactivated poliovirus and N-Trimethyl chitosan on pH-sensitive microneedles for dermal vaccination. Langmuir 31:8654–8660
Quan FS, Kim YC, Song JM, Hwang HS, Compans RW, Prausnitz MR (2013) Long-term protective immunity from an influenza virus-like particle vaccine administered with a microneedle patch. Clin Vaccine Immunol 20:1433–1439
Van der maaden K, Jiskoot W, Bouwstra J (2012) Microneedle technologies for (trans) dermal drug and vaccine delivery. J Control Release 161:645–655
Cormier M, Johnson B, Ameri M, Nyam K, Libiran L, Zhang DD, Daddona P (2004) Transdermal delivery of desmopressin using a coated microneedle array patch system. J Control Release 97:503–511
Park JH, Allen MG, Prausnitz MR (2005) Biodegradable polymer microneedles: fabrication, mechanics and transdermal drug delivery. J Control Release 104:51–66
Martanto W, Moore JS, Kashlan O, Kamath R, Wang PM, O’Neal JM, Prausnitz MR (2006) Microinfusion using hollow microneedles. Pharm Res 23:104–113
Bariya SH, Gohel MC, Mehta TA, Sharma OP (2012) Microneedles: an emerging transdermal drug delivery system. J Pharm Pharmacol 64:11–29
Van der maaden K, Luttge R, Vos PJ, Bouwstra J, Kersten G, Ploemen I (2015) Microneedle-based drug and vaccine delivery via nanoporous microneedle arrays. Drug Deliv and Transl Res 5:397–406
McAllister DV, Allen MG, Prausnitz MR (2000) Microfabricated microneedles for gene and drug delivery. Annu Rev Biomed Eng 2:289–313
Donnelly RF, Singh TRR, Woolfson AD (2010) Microneedle-based drug delivery systems: microfabrication, drug delivery, and safety. Drug Deliv 17:187–207
Kim YC, Park JH, Prausnitz MR (2012) Microneedles for drug and vaccine delivery. Adv Drug Deliv Rev 64:1547–1568
Pradeep Narayanan S, Raghavan S (2016) Transdermal drug delivery with tiny (micro) needles—an overview. South Asian Journal of Research in Engineering Science and Technology 1:378–387
Gill HS, Prausnitz MR (2007) Coated microneedles for transdermal delivery. J Control Release 117:227–237
Vrdoljak A, McGrath MG, Carey JB, Draper SJ, Hill AVS, O’Mahony C, Crean AM, Moore AC (2012) Coated microneedle arrays for transcutaneous delivery of live virus vaccines. J Control Release 159:34–42
Hegde NR, Kaveri SV, Bayry J (2011) Recent advances in the administration of vaccines for infectious diseases: microneedles as painless delivery devices for mass vaccination. Drug Discov Today 16:1061–1068
Cha KJ, Kim T, Park SJ, Kim DS (2014) Simple and cost-effective fabrication of solid biodegradable polymer microneedle arrays with adjustable aspect ratio for transdermal drug delivery using acupuncture microneedles. J Micromech Microeng 24:115015–115022
Hong X, Wei L, Wu F, Wu Z, Chen L, Liu Z, Yuan W (2013) Dissolving and biodegradable microneedle technologies for transdermal sustained delivery of drug and vaccine. Drug Des Deve Ther 7:945–952
LaVan DA, McGuire T, Langer R (2003) Small-scale systems for in vivo drug delivery. Nat Biotechnol 21:1184–1191
Hashmi S, Ling P, Hashmi G, Reed M, Gaugler R, Timmer W (1995) Genetic transformation of nematodes using arrays of micromechanical piercing structures. BioTechniques 19:766–770
Kaushik S, Hord AH, Denson DD, McAllister DV, Smitra S, Allen MG, Prausnitz MR (2001) Lack of pain associated with Microfabricated microneedles. Anesth Analg 92:502–504
Indermun S, Luttge R, Choonara YE, Kumar P, Toit LCD, Modi G, Pillay V (2014) Current advances in the fabrication of microneedles for transdermal delivery. J Control Release 185:130–138
Mikszta JA, Alarcon JB, Brittingham JM, Sutter DE, Pettis RJ, Harvey NG (2002) Improved genetic immunization via micromechanical disruption of skin-barrier function and targeted epidermal delivery. Nat Med 8:415–419
Martanto W, Davis SP, Holiday NR, Wang J, Gill HS, Prausnitz MR (2004) Transdermal delivery of insulin using microneedles in vivo. Pharm Res 21:947–952
Park JH, Choi SO, Seo S, Choy YB, Prausnitz MR (2010) A microneedle roller for transdermal drug delivery. Eur J Pharm Biopharm 76:282–289
Hamzah AA, Aziz NA, Majlis BY, Yunas J, Dee CF, Bais B (2012) Optimization of HNA etching parameters to produce high aspect ratio solid silicon microneedles. J Micromech Microeng 22:095017 (10pp)
Li WZ, Huo MR, Zhou JP, Zhou YQ, Hao BH, Liu T, Zhang Y (2010) Super-short solid silicon microneedles for transdermal drug delivery applications. Int J Pharm 389:122–129
Gill HS, Denson DD, Burris BA, Prausnitz MR (2008) Effect of microneedle design on pain in human subjects. Clin J Pain 24:585–594
Ji J, Tay FEH, Miao J, Iliescu C (2006) Microfabricated microneedle with porous tip for drug delivery. J Micromech Microeng 16:958–964
Wilke N, Hibert C, O’Brien J, Morrissey A (2005) Silicon microneedle electrode array with temperature monitoring for electroporation. Sens Actuators A Phys 123–124:319–325
Hanein Y, Schabmueller CGJ, Holman G, Lucke P, Denton DD, Bohringer KF (2003) High-aspect ratio submicrometer needles for intracellular applications. J Micromech Microeng 13:S91–S95
Lee JW, Park JH, Prausnitz MR (2008) Dissolving microneedles for transdermal drug delivery. Biomaterials 29:2113–2124
Chu LY, Choi SO, Prausnitz MR (2010) Fabrication of dissolving polymer microneedles for controlled drug encapsulation and delivery: bubble and pedestal microneedle designs. J Pharm Sci 99:4228–4238
Sullivan SP, Koutsonanos DG, Martin MDP, Lee JW, Zarnitsyn V, Choi SO, Murthy N, Compans RW, Skountouz I, Prausnitz MR (2010) Dissolving polymer microneedle patches for influenza vaccination. Nat Med 16:915–920
Migalska K, Morrow DIJ, Garland MJ, Thakur R, Woolfson AD, Donelly RF (2011) Laser-engineered dissolving microneedle arrays for transdermal macromolecular drug delivery. Pharm Res 28:1919–1930
Garland MJ, Salvador EC, Migalska K, Woolfson AD, Donelly RF (2012) Dissolving polymeric microneedle arrays for electrically assisted transdermal drug delivery. J Control Release 159:52–59
McCrudden MTC, Alkilani AZ, McCrudden CM, McAlister E, McCarthy HO, Woolfson AD, Donelly RF (2014) Design and physicochemical characterisation of novel dissolving polymeric microneedle arrays for transdermal delivery of high dose, low molecular weight drugs. J Control Release 180:71–80
Lahiji SF, Dangol M, Jung H (2015) A patchless dissolving microneedle delivery system enabling rapid and efficient transdermal drug delivery. Sci Rep 5:7914 (1–7)
Mistilis MJ, Joyce JC, Esser ES, Skountouz I, Compans RW, Bommarius AS, Prausnitz MR (2016) Long-term stability of influenza vaccine in a dissolving microneedle patch. Drug Deliv and Transl Res. doi:10.1007/s13346-016-0282-2
Chen YT, Hsu CC, Tsai CH, Kang SW (2010) Fabrication of microneedles. J Mar Sci Tech Taiw 18:243–248
Music S, Vincekovic NF, Sekovanic L (2011) Precipitation of amorphous SiO2 particles and their properties. Braz J Chem Eng 28:89–94
Matsui Y, Adachi S (2012) Optical properties of porous silicon layers formed by Electroless photovoltaic etching. ECS J Solid State Sci Technol 1:R80–R85
Oliver WC, Pharr GM (1992) An improved technique for determining hardness and elastic modulus using load and displacement sensing indentation experiments. J Mater Res 7:1564–1583
Sakharova NA, Fernandes JV, Antunes JM, Oliveira MC (2009) Comparison between Berkovich, Vickers and conical indentation tests: a three-dimensional numerical simulation study. Int J Solids Struct 46:1095–1104
Gallagher AJ, Anniadh AN, Bruyere K, Otténio M, Xie H, Gilchrist MD (2012) Dynamic tensile properties of human skin. IRCOBI Conference 59:494–502
Metrics M https://mdmetric.com/tech/hardnessconversion.html. Accessed 18 August 2016
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pradeep Narayanan, S., Raghavan, S. Solid silicon microneedles for drug delivery applications. Int J Adv Manuf Technol 93, 407–422 (2017). https://doi.org/10.1007/s00170-016-9698-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00170-016-9698-6