Abstract
Metazoan parasites and their hosts experience the environmental influence of the seasonal presence of Western and Eastern subzones with different oceanographic conditions on the Yucatan continental shelf (YS). In addition, natural seeps and oil transport in the area lead to the presence of hydrocarbons. We hypothesized that the parasite infrapopulations and infracommunities of Haemulon aurolineatum will respond to environmental variability related with ongoing oceanographic subzone conditions of the YS. Spatial and multivariate statistical analyses were used to test this hypothesis. For 17 sampling sites along the YS, 55 parasite morphospecies were recovered from 146 fishes. There were significant differences in the number of parasite species between Western and Eastern subzones, but not in the number of parasite individuals. Spatial autocorrelation on environmental variables as a consequence of the Yucatan current was found. Overall, the parasite metrics suggested a region with a good ecosystem health condition naturally influenced by oceanographic processes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
The Yucatan continental shelf (YS) is considered to sustain a very productive fishery industry. In addition, there are plans of oil extraction in the region due to its hydrocarbon reserves (CNH 2015). Since marine parasites are sensitive to oil spills (Centeno-Chalé et al. 2015; Vidal-Martínez et al. 2014) and other anthropogenic disturbances, it is useful to count with accurate baseline environmental information on the current (undisturbed) ecosystem health conditions of the YS. This information could be helpful to compare the ecosystem health conditions under environmental alterations such as those produced by habitat fragmentation, climate change, overfishing, or oil spills if extraction activities are performed. Marine parasites are useful bioindicators of the ecosystem health condition because they are exposed to pollution as much as free-living organisms (Vidal-Martínez et al. 2019). Due to their complex life-cycles, marine parasites respond to either natural (e.g., hurricanes) (Aguirre-Macedo et al. 2011) or anthropogenic (e.g., contaminants) environmental alterations (Sures et al. 2017). Parasite community studies as bioindicators for the Southern Gulf of Mexico have been mainly focused on marine benthic flatfishes (Centeno-Chalé et al. 2015; Vidal-Martínez et al. 2014, 2019). Thus, it is unknown how parasitic communities of pelagic fish respond to natural or anthropogenic environmental changes. The tomtate grunt (Haemulon aurolineatum) is a pelagic fish found along the Gulf of Mexico (GoM) (Chávez-López and Morán-Silva 2019), and in the YS it is one of the most abundant species (Vega-Cendejas and de Santillan 2019) with potential economic interest (Vallarta-Zárate et al. 2018).
The YS presents a great richness and abundance of marine species due to the masses of deep-water (cold and nutrient-rich), which pass from the Caribbean Sea to the GoM (Pech-Pool et al. 2010). These water masses produce seasonal upwelling events caused by the friction between the Yucatan current and the shallow topography of the continental shelf off the coast of Cabo Catoche (Reyes-Mendoza et al. 2015). During upwelling (spring and summer), the YS presents two zones with different oceanographic conditions: an eastern subzone with high biological productivity due to the mixing of nutrient rich water masses, and a western subzone influenced by fresh water draining into the coast from underground springs (Herrera-Silveira et al. 2013). During fall and winter there is no evidence of these oceanographic differences due to a low or absent upwelling caused by northerly wind events (Reyes-Mendoza et al. 2015). Thus, oceanographic conditions homogenize the YS which in turn would produce spatial autocorrelation (with environmental variables having similar values across a relatively large study area, i.e., 10,000 km2). Clearly, it is necessary to determine which is the relative contribution of spatial autocorrelation as a proxy of the influence of oceanographic conditions together with the contribution of the prevailing environmental conditions. As part of a larger research project on the ecosystem health condition of the GoM, we obtained spatial information of the parasite infracommunities of the tomtate grunt (Haemulon aurolineatum) along the YS during summer, when the upwelling would be stronger as suggested by Reyes-Mendoza et al. (2015). Using this data set, we hypothesized that the metazoan parasite infracommunities of the tomtate grunt would differ in species composition and relative abundance between subzones during an upwelling event on the YS. Furthermore, we hypothesized that the spatial autocorrelation between sampling points would affect the values of the physicochemical variables and contaminants present in the environment, which in turn would affect the metrics of the infrapopulations and infracommunities of parasites of the tomtate grunt. Therefore, the aims of this study were: i) to determine whether the parasite infracommunity metrics of the tomtate grunt exhibit significance differences among subzones in the YS, and ii) determine if there are statistical associations between spatial and environmental variables and the parasite infrapopulation and infracommunity metrics of the tomtate grunt.
Materials and Methods
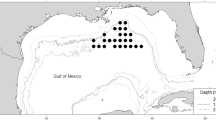
In June 2018, 20 sites were sampled along the YS between 15 and 40 m depths, aboard a coastal fishing vessel (Fig. 1) covering an area of 11,369 km2. However, fish were captured only in 17 sites. All the remaining analyses in this contribution were based on these 17 sites. At each site, 16 variables were measured, including biological (organisms), oceanographic (physicochemical), and pollutant (heavy metals and hydrocarbons) variables (Table S1). Zooplanktonic organisms were captured with a Bongo net (60 cm in diameter, 333 µ filter mesh) at a speed between 1–2 knots. The fish were caught with hooks and lines, establishing a maximum catch of 10 specimens per site. These specimens were measured (cm) and weighed (g), isolated in plastic bags and maintained at − 4 °C for transportation to CINVESTAV Merida. All parasitological procedures followed those recommended by Vidal-Martínez et al. (2002). The parasite infrapopulations per sampling site were described using prevalence (as the percentage of infected host) and mean abundance (average of individuals of a parasite species in a sample of examined hosts) of each parasite species (Bush et al. 1997). Physicochemical variables (dissolved oxygen, temperature, salinity, chlorophyll-a and hydrogen potential) were collected with a CTD device. Heavy metals (Cd, Ni, Pb, V) and hydrocarbon (Polycyclic Aromatic Hydrocarbons-PAHs) concentrations were obtained by the Geochemistry Laboratory from fish muscle tissue, water, and sediment following the methodology of Dótor et al. (2021) and Árcega-Cabrera et al. (2021). Briefly, 0.5 g of muscle tissue for heavy metals and 0.5–5.0 g for PAHs were required. Water samples were obtained using plastic Niskin bottles set up as a rosette and 1L was used for each contaminant (heavy metal and PAHs). Sediment samples were collected using a Hessler Sandia MK-III box corer and 0.5 g were used for heavy metal analysis and 1 g for PAHs. Heavy metal quantification was carried out using an ICP-MS model iCAP Q Thermo Scientific. Precision and accuracy of the analysis were determined with certified reference materials (CRM) PACS-3 and MESS-4 for trace metals (National Research Council of Canada). For PAHs quantification, samples were added with a subrogated solution of deuterated PAHs (1–3 dimethyl-2 nitrobenzene, acenaphthene δ10, phenanthrene δ10, pyrene δ10, triphenyl phosphate, chrysene δ10 and perylene δ10 from UltraScientific). Also, the o-terphenyl (Supelco) was used as an internal standard in all analyses quantified by gas chromatography using a Clarus 500 Perkin Elmer gas chromatographer.
To test the first hypothesis, all the captured fish were grouped in two subzones: West-YS and East-YS according to the subzones proposed by Herrera-Silveira et al. (2013). The number of parasite individuals of all species per individual host (raw infracommunity data) in each sampled station were transformed by fourth square root. A Bray–Curtis similarity matrix was built, and analysis of similarities (ANOSIM) was performed. ANOSIM was used to test the hypothesis of no differences in the infracommunities’ relative abundance between subzones. Similarity Percentage (SIMPER) analysis was used to determine which species contributed the most to the observed patterns of infracommunity similarity. These statistical analyses were performed with Primer v.7 software. To determine the possible statistical relationship between the abundance of each parasite species and the number of species and individuals of metazoan parasites as dependent variables, with the independent variables represented by the environmental variables and spatial autocorrelation between sampling points (Borcard et al. 1992), we used the methods proposed by Bauman et al. (2018). First, we built a spatial neighborhood using Gabriel´s graph as a frame (Fig. S1). Second, we determined the potential presence of spatial autocorrelation in the independent variables, using the Moran´s index random test (Thioulouse et al. 2018). If spatial autocorrelation is present, then spatial weighting matrices constituted by spatial predictors called Moran Eigenvector Maps (MEMs) are produced. MEMs allow the separation of the variance explained by spatial autocorrelation from that explained by the environmental variability. Finally, redundancy analysis (RDA) and variance partitioning (Perez-Neto et al. 2006) were used to determine the amount of explained variance by spatial autocorrelation and environmental variables with respect to the relationship between these independent variables and the parasite metrics (number of individuals of each parasite species, and number of parasite species and individuals, all transformed to Hellinger distance). All spatial and multivariate analyses were performed in R (R Development Core Team, http://www.R-project.com) using the packages ade4, adespatial, adegraphics, spdep, maptools and vegan, modifying the script proposed by Dray (2021). This script has been included in Table S2 and ran using the data of Table S3. The significance in all statistical analyses was established as α < 0.05 unless otherwise stated.
Results and Discussion
A total of 146 tomtate grunts from the 17 sampled sites were captured and examined for both parasites and contaminants. The total fish length range was 15.9–25 cm (21.32 ± 1.84) and no significant differences were found in fish length between subzones (t(144, 0.05) = 0.85, P = 0.40). A total of 22,432 metazoan parasite individuals were quantified, identifying 55 morphospecies (Table S4). The values of the oceanographic variables, and the concentrations of cadmium (Cd), Nickel (Ni), Lead (Pb), Vanadium (V) and Polycyclic Aromatic Hydrocarbons (PAHs) in muscle tissue of fish, water, and sediment are presented in Table S1. The ANOSIM analysis determined numerical differences in the similarity of the infracommunities between Eastern and Western YS subzones (R = 0.13, P = 0.01). In addition, SIMPER analysis identified the species that contributed to 90% of the total number of individuals, showing that the species of the Eastern subzone were a subset of that of the Western subzone (Table 1). Therefore, the differences between subzones can be attributed to the rare species in the metazoan parasite infracommunities of the Western subzone. This pattern of a higher number of species in Western subzone with respect to the Eastern subzone was corroborated by rarefaction curves (Fig. S2). The presence of a smaller number of parasite species in the Eastern subzone could be attributed to the incapacity of the rare parasite species to disperse successfully and complete their life cycles due to the current velocity conditions of the Yucatan current and the littoral Yucatan current. Further support for this interpretation comes from Lara-Hernández et al. (2019), who observed that transport of Panulirus argus larvae towards the Caribbean Sea is caused by the Caribbean current, while the littoral Yucatan current transports them into the Gulf of Mexico.
Regarding the statistical association between environmental variables with parasite community metrics, we found a high level of spatial autocorrelation in all the environmental variables (Table S5) suggesting two things. First, the values of the environmental variables are not independent between sampling points, which can be attributed to the effect of the Yucatan currents, homogenizing the whole study area by transporting nutrients, heavy metals, hydrocarbons, and organisms from Eastern to Western YS. Second, since environmental variables had a component of environmental autocorrelation, it was necessary to separate this component to determine its relative contribution as well as that of the environmental variables themselves to the explained variance of the relationship of these predictors with the parasite metrics. Therefore, we calculated MEMs for all the combinations of geographical distances among sampling points, retaining only the 5 MEMs explaining the highest amount of variance (Table S6). These 5 MEMs together with the environmental variables were used as independent variables in a RDA analysis, with the parasite metrics as dependent variables. The RDA analysis was significant (Fig. 2; P < 0.01; 99 permutations). The variation partitioning analysis (Fig. S3) showed that the environmental variables by themselves were not significant predictors of the parasite metrics (P = 0.06), producing only 15% of the explained variance. However, by combining the variance explained by the MEMs (29%) and that of the environmental variables, the analysis became significant (P < 0.01) with a 44% of explained variance for the relationship between environment and the parasite metrics (Table S7). The most relevant dependent variables were the number of parasite species and the abundance of the monogenean Haliotrematoides spp. The number of parasite species was different between subzones, with the Western subzone having 13 additional species compared with Eastern subzone (Table S4). This suggests that current speed in the Western zone is not as restrictive for transmission as it is in the Eastern subzone. In the case of Haliotrematoides spp., this species was responsible for the lack of significant differences in the number of parasites between subzones due to its high abundance. The most relevant predictors were MEMs 10, 11, and 51, salinity, water temperature, and chlorophyll a (Fig. 2). These predictors are in line with the fact that currently there are no oil extraction activities in the YS. Therefore, the environmental conditions in the study zone are considered good for the completion of the life cycles of the parasites living there. Similar results were obtained by Vidal-Martínez et al (2019) for the metazoan parasite communities of the dusky flounder Syacium papillosum along the YS. Several other variables related to heavy metals and hydrocarbons were apparently relevant in the RDA analysis. However, it is important to remember that the environmental variables only accounted for 7% of the explained variance. The persistence of these variables in our analysis can be explained by the presence of natural seeps at various points within the YS (Love et al. 2013), which leads to the presence of hydrocarbons. In regard to Cadmium and Vanadium, Árcega-Cabrera et al. (2021) suggested that these heavy metals are most likely transported by the Yucatan current and by underground freshwater currents into the YS. Still, a large amount of unexplained variance remains (56%). We speculate that part of this variance should be related to finer oceanographic processes occurring along the YS, and to demographic stochasticity affecting the probability of completion of the parasite life cycles, especially in the Eastern subzone of the YS.
Redundancy analysis of the environmental and spatial variables (MEMs) as independent variables and the infracommunity metrics of the metazoan parasites of the tomtate grunt Haemulon aurolineatum as dependent variables. The RDA for all sampling sites accounted for 35% of the total variance and was highly significant for the first and all four canonical axes (P < 0.01; 99 permutations) for the 17 sampling sites. The acronyms were as follows: Do-Dollfusentis chandleri; Dy-Dasyrhynchus sp.; Go-Gorgorhynchus sp.; Ha-Haliotrematoides spp.; Le-Lecithochirium musculus; St2-Stephanostomum sp. 2; St4-Stephanostomum sp. 4; Pc-Procamallanus sp.; Py-Prosorhynchus sp.; spp-Total number of species per fish; indiv-Total number of parasite individuals collected per fish; Temp-Sea water temperature at 10 m depth (°C); Sal-Salinity (UPS); Chl.a-Chlorophyll a; FCd-Cadmium in tomtate grunt; FPb-Lead in tomtate grunt; FV-Vanadium in tomtate grunt; FNi-Nickel in tomtate grunt; FPAHS-Total concentrations of polyaromatic hydrocarbons in tomtate grunt; WCd-Cadmium in water; WPb-Lead in water; WNi-Nickel in water; WV-Vanadium in water; WPAHs-Total concentrations of polyaromatic hydrocarbons in water; WALIF-Total of aliphatic hydrocarbons in water; SPAHs-Total concentrations of polyaromatic hydrocarbons in sediment
In conclusion, our first hypothesis that the parasite infracommunities of the tomtate grunt would respond to seasonal oceanographic differences between subzones in the YS was partially accepted, since only differences in the number of species were found. Our second hypothesis that spatial autocorrelation and environmental variability would affect the parasite metrics was accepted. It was found that spatial autocorrelation produced by the Yucatan currents flowing from Eastern to Western subzones produced strong spatial structure in the environmental variables. The results obtained suggest that the large influence of oceanographic currents and the apparently small influence of environmental variables (including contaminants) are what could be expected in a relatively pristine zone such as the YS. These results establish baseline data that will be useful not only in the possibility of oil spills related to future oil extraction activities in the region, but also to understanding how fish and their parasites respond to the natural environmental conditions of the YS. Thus, we consider it necessary to maintain a permanent monitoring program of the ecosystem health of the YS to determine the influence of these oceanographic processes on the metrics of both fish and parasite communities through time.
References
Aguirre-Macedo ML, Vidal-Martínez VM, Lafferty KD (2011) Trematode communities in snails can indicate impact and recovery from hurricanes in a tropical coastal lagoon. Int J Parasitol 41:1403–1408. https://doi.org/10.1016/j.ijpara.2011.10.002
Arcega-Cabrera F, Gold-Bouchot G, Lamas-Cosío E et al (2021) Spatial and temporal variations of vanadium and cadmium in surface water from the Yucatan shelf. Bull Environ Contam Toxicol. https://doi.org/10.1007/s00128-021-03234-3
Bauman D, Drouet T, Fortin MJ, Dray S (2018) Optimizing the choice of a spatial weighting matrix in eigenvector-based methods. Ecology 99:2159–2166
Borcard D, Legendre P, Drapeau P (1992) Partialling out the spatial component of ecological variation. Ecology 73:1045–1055
Bush AO, Lafferty KD, Lotz JM et al (1997) Parasitology meets ecology on its own terms: Margolis et al. revisited. J Parasitol 83:575–583
Centeno-Chalé OA, Aguirre-Macedo ML, Gold-Bouchot G et al (2015) Effects of oil spill related chemical pollution on helminth parasites in Mexican flounder Cyclopsetta chittendeni from the Campeche sound, Gulf of Mexico. Ecotoxicol Environ Safe 119:162–169. https://doi.org/10.1016/j.ecoenv.2015.04.030
Chávez-López R, Morán-Silva Á (2019) Revisión de la composición de especies de peces capturadas incidentalmente en la pesquería de camarón en el Golfo de México. Ciencia Pesquera 27:65–82
CNH Comisión Nacional de Hidrocarburos (2015) Plan quinquenal de licitaciones para la exploración y extracción de hidrocarburos. https://www.gob.mx/cnh. Accessed 28 Apr 2021
Dótor-Almazán A, Gold-Bouchot G, Lamas-Cosío E et al (2021) Spatial and temporal distribution of trace metals in shallow marine sediments of the Yucatan shelf, Gulf of Mexico. Bull Environ Contam Toxicol. https://doi.org/10.1007/s00128-021-03170-2
Dray S (2021) Moran’s Eigenvector Maps and related methods for the spatial multiscale analysis of ecological data. https://cran.r-project.org/web/packages/adespatial/vignettes/tutorial.html. Accessed 28 May 2021
Herrera-Silveira JA, Comín FA, Capurro-Filograso L (2013) Landscape, land use, and management in the Coastal zone of Yucatan Peninsula. In: Day JW, Yañez-Arancibia A (eds) Gulf of Mexico origin, waters, and biota, vol 4. Ecosistem-based management. Texas A & M University Press, Texas, pp 225–242
Lara-Hernández JA, Zavala-Hidalgo J, Sanvicente-Añorve L (2019) Connectivity and larval dispersal pathways of Panulirus argus in the Gulf of Mexico: a numerical study. J Sea Res 155:101814. https://doi.org/10.1016/j.seares.2019.101814
Love MS, Baldera A, Yeng C et al (2013) The Gulf of Mexico ecosystem. Ocean Conservancy, New Orleans
Pech-Pool D, Mascaró-Miquelajáuregui M, Simoes N et al (2010) Ambientes marinos. In: Durán R, Méndez M (eds) Biodiversidad y desarrollo humano en Yucatán. CICY, PPD-FMAM, CONABIO, SEDUMA, Yucatán, pp 21–23
Peres-Neto PR, Legendre P, Dray S, Borcard D (2006) Variation partitioning of species data matrices: estimation and comparison of fractions. Ecology 87:2614–2625
Reyes-Mendoza O, Mariño-Tapia I, Herrera-Silveira J et al (2015) The effects of wind on upwelling off Cabo Catoche. J Coast Res 319:638–650. https://doi.org/10.2112/JCOASTRES-D-15-00043.1
Sures B, Nachev M, Selbach C et al (2017) Parasite responses to pollution: what we know and where we go in ‘Environmental Parasitology.’ Parasite Vector. https://doi.org/10.1186/s13071-017-2001-3
Thioulouse J, Dray S, Dufour AB et al (2018) Multivariate analysis of ecological data in ade4. Springer, New York
Vallarta-Zárate JRF, Martínez-Magaña VH, Huidobro-Campos L et al (2018) Región plataforma continental de Yucatán: acústica pesquera, batimetría oceanografía y biología. INAPESCA, Mexico
Vega-Cendejas ME, de Santillana MH (2019) Demersal fish assemblages and their diversity on the Yucatan continental shelf: Gulf of Mexico. Reg Stud Mar Sci 29:100640. https://doi.org/10.1016/j.rsma.2019.100640
Vidal-Martínez VM, Aguirre-Macedo ML, Scholz T et al (2002) Atlas of the helminth parasites of cichlid fish of Mexico, 1st edn. Instuto Politécnico Nacional, México
Vidal-Martínez VM, Centeno-Chalé OA, Torres-Irineo E et al (2014) The metazoan parasite communities of the shoal flounder (Syacium gunteri) as bioindicators of chemical contamination in the southern Gulf of Mexico. Parasite Vector. https://doi.org/10.1186/s13071-014-0541-3
Vidal-Martínez VM, Velázquez-Abunader I, Centeno-Chalé OA et al (2019) Metazoan parasite infracommunities of the dusky flounder (Syacium papillosum) as bioindicators of environmental conditions in the continental shelf of the Yucatan Peninsula, Mexico. Parasite Vectors 12:18. https://doi.org/10.1186/s13071-019-3524-6
Acknowledgements
This article is part of the Master thesis of JGGT at CINVESTAV Mérida-Unit. We thank the National Council for Science and Technology CONACYT for provided scholarship (No.637098). We are grateful to the staff of the Aquatic Pathology Laboratory, especially Arturo Centeno-Chále, Francisco Puc-Itzá, Clara Vivas-Rodríguez, Nadia Herrera-Castillo, Efraín Sarabia-Eb, and Víctor Ceja of Geochemistry Laboratory, all from CINVESTAV Mérida. JGGT thanks Dr. Frank Ocaña-Borrego and MSc Zita Arriaga-Piñon for their help with the statistical analyses, and Mr Ehecatl Vidal-Aguirre and Dr. Lee Couch (University of New Mexico) for their help with English language. This research was financially supported by the Mexican Ministry of Energy through the Hydrocarbon Found, project 201441. This is a contribution of the Gulf of Mexico Research Consortium (CIGoM). We acknowledge PEMEX´S specific request to the Hydrocarbon Found to address the environmental effects of oil spills in the Gulf of Mexico.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
García-Teh, J.G., Vidal-Martínez, V.M., Mariño-Tapia, I. et al. Metazoan Parasite Infracommunities of the Tomtate Grunt (Haemulon aurolineatum) as Potential Bioindicators of Environmental Conditions in the Yucatan Continental Shelf, Mexico. Bull Environ Contam Toxicol 108, 49–54 (2022). https://doi.org/10.1007/s00128-021-03305-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-021-03305-5