Abstract
A simple, rapid and sensitive spectrofluorimetric method was developed for the determination of di-syston, ethion and phorate in environmental water samples. The procedure is based on the oxidation of these pesticides with cerium (IV) to produce cerium (III), and its fluorescence was monitored at 368 ± 3 nm after excitation at 257 ± 3 nm. The variables effecting oxidation of each pesticide were studied and optimized. Under the experimental conditions used, the calibration graphs were linear over the range 0.2–15, 0.1–13, 0.1–13 ng mL−1 for di-syston, ethion and phorate, respectively. The limit of detection and quantification were in the range 0.034–0.096 and 0.112–0.316 ng mL−1, respectively. Intra- and inter-day assay precisions, expressed as the relative standard deviation (RSD), were lower than 5.2 % and 6.7 %, respectively. Good recoveries in the range 86 %–108 % were obtained for spiked water samples. The proposed method was applied to the determination of studied pesticides in environmental water samples.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Organothiophosphates (OTPs) with a thiophosphoryl (P=S) functional group constitute a broad class of widely used OPPs insecticides. These compounds are used frequently in agricultural lands worldwide and has resulted highly toxic residuals in crops, livestock, and poultry products which has further led to their migration into underground aquifers (Parham and Saeed 2015). These compounds are highly toxic to human health and are powerful inhibitors of acetylcholinesterase (AChE) enzyme, causing accumulation of acetylcholine at nerve endings in the peripheral or central nervous system (Parham and Saeed 2015; Khaled et al. 2014).
The utilization of Organophosphorus pesticides (OPPs) pesticides is favored to the usage of other pesticides because of their ability to degrade more readily in the environment (Lambropoulou and Albanis 2001; Schellin et al. 2004). OPPs have been found in groundwaters, surface waters, lagoons and drinking water in varying concentrations and hence, there is an increasing demand for developing methods for the determination of such contaminants in food and environmental analysis (Schellin et al. 2004). The European Union (EU) allows a maximum concentration of 0.1 µg L−1 of each individual pesticide and 0.5 µg L−1 of the sum of pesticides in environmental and drinking water (Chen and Huang 2006; Berijani et al. 2006; Fu et al. 2009). Due to this legal limit, a simple, efficient and sensitive method to evaluate and monitor these compounds, at trace levels, in environmental matrices is required.
Di-syston (O,O-diethyl S-2-ethylthioethyl phosphorodithioate), ethion ( O,O,O′,O′-tetraethyl S,S′-methylene bis(phosphorodithioate) and phorate ( O,O-diethyl S-ethylthiomethyl phosphorodithioate), Fig. 1, are OTP insecticides. The identification and quantification of these pesticides in the environment samples are generally based on chromatographic methods, such as gas chromatography (GC) or high-performance liquid chromatography (HPLC) coupled with mass spectroscopy (MS), i.e. GC–MS (Lambropoulou and Albanis 2001; Schellin et al. 2004; Chen and Huang 2006; Pereira dos Anjos and de Andrade 2014; Anagnostopoulos and Miliadis 2013) and HPLC–MS (Anagnostopoulos and Miliadis 2013; Narayan Sinh et al. 2011), or other detectors (Berijani et al. 2006; Samadi et al. 2012; He et al. 2009). Sensors (Khaled et al. 2014; Jiang et al. 2008), spectrophotometric (Mathew et al. 2007; Jafari 2006) and electrochemical (Qiu et al. 2007) methods have also been used for determination of these pesticides. These chromatographic methods are very sensitive and reliable, but they are time-consuming and expensive. Moreover, they can only be performed by highly trained technicians, are not convenient for on-site or in-field detection and require highly specialized facilities with complex sample pre-treatment steps (Khaled et al. 2014; de Armasa et al. 2002). Therefore, it is priority to develop simple, sensitive and rapid analytical methodologies as practical alternatives to these robust and efficient chromatographic methods. Fluorescence spectrometry is a sensitive, selective and relatively low cost method for the quantitative analysis of pesticides and other pollutants (Aaron and Coly 2000; Vega Morales et al. 2010; Bavili Tabrizi et al. 2014). Due to these features we propose here a simple, sensitive and inexpensive spectrofluorimetric method for the determination of di-syston, ethion and phorate in environmental water samples.
Materials and Methods
A Shimadzu RF-5301 PC spectrofluorophotometer, equipped with a 150 W Xenon lamp and 1.00 cm quartz cells, was used for the fluorescence measurements. Both excitation and emission slits were adjusted to 3 nm and the sensitivity adjusted to low. A thermostated water bath (636; Friedberg/Hessen, Germany) was used throughout the work.
Di-syston, ethion and phorate were purchased from PolyScience Corporation (Chemical Division, Analytical Standards, Arlington Heights, Illinois 60004). All solvents and chemicals including sulfuric acid, methanol and Ce(IV)-sulfate-tetrahydrat were obtained from E. Merck (Darmstadt, Germany).
A stock standard solution of each pesticide at a concentration of 200 μg mL−1 was prepared by diluting an appropriate volume of 1 % (m/v) solution of each pesticide to 25 mL with methanol. These solutions were stored under dark conditions in refrigerator when not in use. Working standard solutions were obtained daily by appropriately diluting this stock solution with ultrapure water. The Ce(IV) solution at concentration of 0.1 mol L−1 was prepared in 2.0 mol L−1 sulfuric acid and was kept at 4°C for 2 weeks. Also, a 2.0 mol L−1 H2SO4 solution was prepared. All other reagents were of analytical reagent grade (E. Merck). The water used for sample preparation was de-ionized and purified using a Milli-Q system (Advantage A 10, Millipore, France).
For calibration an aliquot of sample solution containing di-syston, ethion and phorate in the range 0.2–15, 0.1–13 and 0.1–13 ng mL−1, respectively, were transferred into 15-mL calibrated centrifuge tubes. This was followed by addition of 0.3 mL of 2.0 mol L−1 sulfuric acid and 0.04 mL of 0.1 mol L−1 Ce(IV) solution. The volume of the obtained solutions were completed to 10 mL with ultra-pure water and the resultant solutions were equilibrated at 95°C for 16, 25 and 10 min in the case of di-syston, ethion and phorate, respectively. Then, solutions were cooled to room temperature and the fluorescence intensity of each solution was measured at 368 ± 3 nm while excited at 257 ± 3 nm against reagent’s blank.
Real water samples were obtained from different districts of Azerbaijan-e-sharghi. Tap water samples were taken from our lab in Tabriz and a spring water sample came from Marand. River water samples were collected from Aras River (Jolfa). Bottled mineral water samples were obtained from local markets in Tabriz. All samples were collected randomly and filtered through 0.45 μm filter paper before use in order to remove suspended solids. The filtered water samples were stored at 4°C until analysis. Some interfering cations can be removed prior to analysis by using cation-exchange resin. For this purpose, 100 mL of each water sample was treated with 2.0 g of strong cation-exchange resin in batch mode for 30 min. Then, aliquots of 1.0 mL of upper solutions were subjected to the spectrofluorimetric analysis.
Results and Discussion
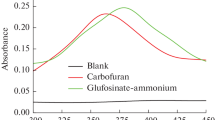
Ce(IV) is a well-known oxidizing agent. It can be easily reduced to Ce(III) that shows a characteristic fluorescence in sulfuric acid medium. Ce(III) is usually more fluorescent than the oxidation products and therefore the measurement of its fluorescence can be used as a very sensitive method for determination of certain drugs by monitoring the fluorescence of the formed Ce(III) (Bavili Tabrizi 2006, 2007a, b). But, the literature survey revealed that this system rarely or has not been used for the analysis of these pesticides. Thus, in the present work the studied pesticides were oxidized by Ce(IV) in sulfuric acid medium and the fluorescence intensity of the produced Ce(III) was monitored. The excitation and emission spectra for di-syston-Ce(IV) system have been given in Fig. 2. Similar spectra were obtained for ethion and phorate systems.
The effect of Ce(IV) concentration on the fluorescence intensities was evaluated in the range 1.0–10 (×10−4) mol L−1. In Fig. 3, it was shown that Ce(IV) at concentration of 4.0–6.0 × 10−4 mol L−1 led to the maximum and constant signals for studied pesticides. At concentrations lower than this range the fluorescence intensity dropped due to insufficient Ce(IV) for oxidation. On the other hand, higher concentrations of Ce(IV) were reported to probably quench the fluorescence thus decreasing the detected intensity (Bavili Tabrizi 2006, 2007a, b). An aliquot of 40 µL of 0.1 mol L−1 Ce(IV) was used for the oxidation of pesticides in the rest of work.
The effect of Ce(IV) concentration on the analytical signals, 10.0 ng mL−1 of each pesticide was used; other conditions have been mentioned in the text and Fig. 1
The effect of sulfuric acid concentration on the fluorescence intensities was depicted in Fig. 4. It was observed that the fluorescence intensity was rapidly increasing up to sulfuric acid concentration of 0.05 mol L−1, and then remained approximately constant up to 0.5 mol L−1. Hence, an aliquot of 0.3 mL of 2.0 mol L−1 sulfuric acid (final concentration of 0.06 mol L−1) was taken as optimum for other experiments.
The effect of sulfuric acid concentration on the analytical signals, 10.0 ng mL−1 of each pesticide was used; other conditions have been mentioned in the text and Fig. 1
It was found that the heating at high temperatures for several minutes was necessary for the completion of the reaction. Thus, oxidation reactions were carried out at different temperatures (ranging from 25 to 100°C) in a thermostated water bath, for periods ranging from 10 to 100 min. The results indicated that an equilibration time of 16, 25 and 10 min at 95°C was adequate to obtain maximum fluorescence intensities in the case of di-syston, ethion and phorate, respectively.
The linear concentration range, RSD, limit of detection (LOD) and quantification (LOQ) for studied pesticides are summarized in Table 1. The LOD and LOQ were defined as 3Sb/m and 10Sb/m, respectively, where Sb is the standard deviation of the blank measurements and m is the slope of the calibration line. The precision of the method was determined by analyzing eight samples on the same day (intra-day precision) or three samples on different days (inter-day precision), and expressed as RSD. The intra-day precision was lower than 5.2 % and the inter–day precision was lower than 6.7 %. The results have been summarized in Table 2.
Compared with references which use GC or HPLC–MS for determination of these analytes, the proposed method does not require high investment and maintenance costs of the instruments. More importantly, our LOD is comparable to or even better than some of these methods which use very sensitive detection techniques (Lambropoulou and Albanis 2001; Parham and Saeed 2015; Schellin et al. 2004; Berijani et al. 2006; Pereira dos Anjos and de Andrade 2014; Samadi et al. 2012; You et al. 2013).
For interference study the influence of frequently encountered species were studied by analyzing sample solutions containing 5.0 ng mL−1 of ethion with different amounts of possible interfering. The tolerance limit was taken as the concentration causing an error less than 8 % in the determination of the pesticide. The tolerance limits for the foreign ions studied are shown in Table 3. The results showed that the most of the ions tested did not interfere with their concentrations up to at least 500-fold excess related to the pesticide. As can be seen, the main interference in the determination of pesticide is due to Cu2+. According to the literature some interfering cations can be removed prior to analysis by using cation-exchange resins or by precipitation in alkaline medium and filtration (Pons et al. 2008). Thus, a batch cation-exchange method was used before the analysis.
For the application of the method, an aliquot of 1.0 mL of each treated water sample was transferred to clean centrifuge tubes and spiked with pesticide at the three test concentrations and then analyzed following the optimized procedure. The recoveries ranged from 86 % to 108 %, as shown in Table 4, and were satisfactory. Typical spectra for di-syston are shown in Fig. 1. No additional peaks caused by interfering compounds were observed at the emission wavelength that was used. Therefore, the similarities in the excitation and emission spectra found in each of the sample types and the reasonable recoveries that were found indicated that there were no significant matrix effects on the proposed method. Also, the UV absorption spectra were recorded for the blank sample and standard solution of each pesticide. The results indicated that the reagents and analytes showed no or negligible absorption at the excitation and emission wavelengths of Ce(III). Finally, the proposed method was successfully applied to the analysis of studied pesticides in environmental water samples and the results are presented in Table 4. EU regulations for drinking water quality set a limit in concentration of 0.5 μg L−1 for the sum of all pesticides and 0.1 μg L−1 for each compound (Lambropoulou et al. 2000). On the other hand, based on US Environmental Protection Agency reports, the maximum modeled concentration of ethion, di-syston and phorate in surface water were 25, 39 and 53.2 ng mL−1 (US EPA), respectively. Thus, our results were lower than these specified levels.
Conclusions
This report describes a validated spectrofluorimetric method for the assay of di-syston, ethion and phorate in environmental water samples. Method validation using spiked real samples demonstrated that the method is capable of detecting trace pesticides with adequate trueness and precision. In addition, sensitivity of the method is enough for the determination of pesticides in variety of environmental samples. In addition, the obtained LODs and LOQs are comparable or better than those of other methods which use high sophisticated and expensive GC or HPLC–MS instruments. From the economic point of view, the proposed method is simple, rapid, sensitive and inexpensive, and thus seems a good alternative to previously reported methods.
References
Aaron J-J, Coly A (2000) Luminescence methods in pesticide analysis. Applications to the environment. Analusis 28:699–709
Anagnostopoulos C, Miliadis GE (2013) Development and validation of an easy multiresidue method for the determination of multiclass pesticide residues using GC–MS/MS and LC–MS/MS in olive oil and olives C. Talanta 112:1–10
Bavili Tabrizi A (2006) A simple spectrofluorimetric method for determination of mefenamic acid in pharmaceutical preparation and urine. Bull Korean Chem Soc 27:1199–1202
Bavili Tabrizi A (2007a) A new spectrofluorimetric method for determination of nifedipine in pharmaceutical formulations. Chem Anal (Warsaw) 52:635–643
Bavili Tabrizi A (2007b) A simple spectrofluorimetric method for determination of piroxicam and propranolol in pharmaceutical preparations. J Food Drug Anal 15:242–248
Bavili Tabrizi A, Rashidi MR, Ostadi H (2014) A nanoparticle-based solid-phase extraction procedure followed by spectrofluorimetry to determine carbaryl in different water samples. J Braz Chem Soc 25:709–715
Berijani S, Assadi Y, Anbia M, Milani Hosseini M-R, Elham A (2006) Dispersive liquid–liquid microextraction combined with gas chromatography-flame photometric detection very simple, rapid and sensitive method for the determination of organophosphorus pesticides in water. J Chromatogr A 1123:1–9
Chen P-S, Huang S-D (2006) Determination of ethoprop, diazinon, disulfoton and fenthion using dynamic hollow fiber-protected liquid-phase microextraction coupled with gas chromatography–mass spectrometry. Talanta 69:669–675
De Armasa G, Miró M, Estela JM, Cerdà V (2002) Simultaneous determination of fluorophores with overlapped spectra by sequential injection analysis coupled to variable angle scanning fluorescence spectrometry and multivariate linear regression algorithms. Anal Chim Acta 471:173–186
Fu LL, Xiujuan HJ, Zhao X, Wang H, Wang X (2009) Application of dispersive liquid–liquid microextraction for the analysis of triazophos and carbaryl pesticides in water and fruit juice samples Lingyan. Anal Chim Acta 632:289–295
He L, He L, Xie H, Wang C, Jiang X, Lu K (2009) Ionic liquid-based dispersive liquid–liquid microextraction followed high-performance liquid chromatography for the determination of organophosphorus pesticides in water sample. Anal Chim Acta 655:52–59
Jafari MT (2006) Determination and identification of malathion, ethion and dichlorovos using ion mobility spectrometry. Talanta 69:1054–1058
Jiang X, Li D, Xu X, Ying Y, Li Y, Ye Z, Wang J (2008) Immunosensors for detection of pesticide residues. Biosens Bioelectron 23:1577–1587
Khaled E, Kamel MS, Hassan HN, Abdel-Gawad H, Aboul-Enein HY (2014) Performance of a portable biosensor for the analysis of ethion residues. Talanta 119:467–472
Lambropoulou DA, Albanis TA (2001) Optimization of headspace solid-phase microextraction conditions for the determination of organophosphorus insecticides in natural waters. J Chromatogr A 922:243–255
Lambropoulou D, Sakellarides T, Albanis T (2000) Determination of organophosphorus insecticides in natural waters using SPE-disks and SPME followed by GC/FTD and GC/MS. Fresenius J Anal Chem 368:616–623
Mathew SB, Pillai AK, Gupta VK (2007) A rapid spectrophotometric assay of some organophosphorus pesticide residues in vegetable samples. Spectrochim Acta A 67:1430–1432
Narayan Sinh S, Vasudev K, Vardhana Rao Vishnu M, Odetokun M (2011) Quantification of organophosphate insecticides in drinking water in urban areas using lyophilization and high-performance liquid chromatography–electrospray ionization-mass spectrometry techniques. Int J Mass Spectrom 300:12–20
Parham H, Saeed S (2015) Resonance Rayleigh scattering method for determination of ethion using silver nanoparticles as probe. Talanta 31:570–576
Pereira dos Anjos J, de Andrade JB (2014) Determination of nineteen pesticides residues (organophosphates, organochlorine, pyrethroids, carbamate, thiocarbamate and strobilurin) in coconut water by SDME/GC–MS. Microchem J 112:119–126
Pons C, Santos JLM, Lima JLFC, Forteza R, Víctor V (2008) Multi-pumping flow system for the determination of nitrite and nitrate in water samples. Microchim Acta 161:73–79
Qiu P, Ni Y-N, Kokot S (2007) Determination of pesticide ethion by linear sweep stripping voltammetry. Chem Res Chin Univ 23:14–17
Samadi S, Sereshti H, Assadi Y (2012) Ultra-preconcentration and determination of thirteen organophosphorus pesticides in water samples using solid-phase extraction followed by dispersive liquid–liquid microextraction and gas chromatography with flame photometric detection. J Chromatogr A 1219:61–65
Schellin M, Hauser B, Popp P (2004) Determination of organophosphorus pesticides using membrane-assisted solvent extraction combined with large volume injection–gaschromatography–mass spectrometric detection. J Chromatogr A 1040:251–258
US Environmental Protection Agency (US EPA). http://www.epa.gov/pesticides/
Vega Morales T, Montesdeoca Esponda S, Santana Rodriguez JJ, Efremova Aaron S, Aaron J-J (2010) Luminescence methods for study and determination of pollutants in the environment. Maced J Chem Chem Eng 29:1–42
You X, Xing Z, Liu F, Jiang N (2013) Air-assisted liquid–liquid microextraction used for the rapid determination of organophosphorus pesticides in juice samples. J Chromatogr A 1311:41–47
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflict of interest.
Rights and permissions
About this article
Cite this article
Bavili Tabrizi, A., Abdollahi, A. Determination of Organothiophosphate Insecticides in Environmental Water Samples by a Very Simple and Sensitive Spectrofluorimetric Method. Bull Environ Contam Toxicol 95, 536–541 (2015). https://doi.org/10.1007/s00128-015-1612-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-015-1612-7