Abstract
Purpose
Thoracic endovascular aortic repair (TEVAR) for traumatic thoracic aortic injury (TTAI) reports short-term benefits. However, long-term durability and the need of reintervention remain unclear. Here, we determined mid-term outcome of TEVAR for TTAI and investigated the influence of the length of proximal landing zone on aorta.
Methods
Between October 2009 and February 2018, 69 patients diagnosed TTAI and 42 included patients underwent TEVAR. Patients were divided into two groups by the length of proximal landing zone; ≤ 20 mm and > 20 mm. The primary endpoint was success and survival rate, and the secondary endpoint was the increase of aorta size and the need of reintervention.
Results
The mean follow-up period was 47.9 ± 29.6 months and 100% success rate. No endoleaks or additional reinterventions during the follow-up period. The cumulative survival of all-cause death was 90.5 ± 2.3%, 85.7 ± 4.1% and 61.7 ± 8.4% at 1, 5 and 7 years, respectively. There was no statistically significant difference in the increase of aorta size due to the length of the proximal landing zone (p = 0.65).
Conclusion
In selective TTAI patients for TEVAR, the length of proximal landing zone did not considerably influence the aorta size or needed further reintervention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Traumatic thoracic aortic injury (TTAI) is a devastating injury. Prehospital mortality occurs in more than 75% of patients and up to 50% of those arriving at the hospital alive may die within the first 24 h after injury [1]. Thoracic endovascular aortic repair (TEVAR), first described by Dake et al. [2], is widely used for the management of TTAI including hybrid surgery. Regardless of the short-term benefits of reduced morbidity and mortality compared with those of open surgery, the long-term durability of TEVAR and the need for secondary aortic reintervention following the procedure still remain an area of interest that should be clarified. Most TTAIs occur within 20 mm of the origin of the left subclavian artery (LSA) [3]. In emergency situations, it presents a considerable challenge to follow the recommended 20-mm proximal landing zone to perform traumatic TEVAR. In addition, we should consider that TTAI patients’ aorta were mostly previously normal pathophysiology, comparing with other aortic aneurysmal diseases requiring TEVAR [4]. Therefore, there are some doubts whether to follow the 20 mm length of proximal landing zone or to reassess the required length of landing zone.
The aim of the present study was to analyze the mid-term outcomes of a single-center experience with TEVAR for TTAI. The primary endpoint was the success rate and survival rate of TEVAR for TTAI, and the secondary endpoint was the increase of aortic size and the need of reintervention related to the length of proximal landing zone.
Methods
Patients
Between October 2009 and February 2018, 69 patients were diagnosed with TTAI at Kyungpook National University Hospital. We excluded 27 patients with traumatic ascending aortic injury and descending aortic injury but did not undergo stent-graft placement due to irreversible brain injury, who rejected operation, and with aortic rupture before entering the operating room. 42 patients underwent TEVAR for TTAI and were included in this study to evaluate the clinical result. Pre-operative characteristics, procedural details, and post-operative outcomes were analyzed.
Patients were categorized into two groups by the length of proximal landing zone (> 20 mm and ≤ 20 mm) to compare the changes of the aortic size and need for reintervention. The institutional review board (IRB) of Kyungpook National University Hospital approved this retrospective study and no informed consent was required (IRB approval No. 2018-09-004).
Trauma protocol and operative strategies
Trauma patients are evaluated by our hospital protocol, including computed tomography (CT) scans of the head, chest, and abdomen, and further specific radiological examinations whenever necessary. The patients were evaluated by multidisciplinary trauma team, and the injury severity score (ISS) was assessed [5]. The urgency of surgery was determined considering the aortic injury grade, presence of associated injury, mentality, and predicted performance.
Pre-operative CT scans were reviewed to measure the bilateral vertebral artery size for assessing the subclavian steal syndrome with a policy of selective delayed subclavian artery revascularization. All TEVAR procedures were performed via the transfemoral approach. All procedures were performed with the patient under general anesthesia, and cerebrospinal fluid drainage was not performed for any patient. Anticoagulation with heparin was prescribed at a dose of 3000–5000 units. Patients with associated injuries who had a high risk of bleeding, such as intracranial hemorrhage or solid organ injury, received a less dose of heparin.
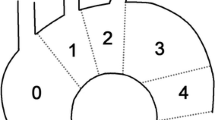
TEVAR was performed using S&G SEAL Thoracic Stent-Grafts (S&G Biotech, Seongnam, Korea) and Valiant Thoracic Stent-Grafts (Medtronic Vascular, Santa Rosa, CA, USA). For patients with a healthy native aorta, stent-graft oversizing was 5–10%, and excessive oversizing was considered over 20%. We attempted to apply the distal stent-graft on a native aorta, without exceeding the 10–11th thoracic vertebrae. The proximal landing zones in the aortic arch were classified as 0–4 according to Ishimaru’s classification [6]. Combined operations were performed according to the severity of injury at other sites. Follow-up CT scans were performed before discharge, at 6 and 12 months after operation, and annually thereafter to exclude complications such as endoleak, stent migration, or pseudoaneurysm.
Definition
TTAI grade was classified as I–IV according to injury severity using CT scans. Grade I is characterized by an intimal tear, with no involvement of the media and no contour abnormalities to the outside surface of the aorta. Grade II represents an injury that extends to the media, such as an intramural hematoma or dissection, with the presence of an external contour abnormality. Grade III indicates an aortic pseudoaneurysm, and grade IV indicates free rupture [7].
Emergency TEVAR was defined when a trauma patient was taken directly to the operating room from the emergency department for emergency repair, and urgent TEVAR was defined as repair performed within 24–48 h after admission.
Primary success was defined as the complete exclusion of the primary target lesion without any additional interventions. Aortic reintervention was defined as the need for any surgical or endovascular interventions following the initial TEVAR procedure during follow-up. An endoleak was defined as radiological evidence of blood flow outside the stent graft according to published guidelines [8]. Increase of the proximal aortic size was defined as a difference between the maximum proximal aortic size on final follow-up CT scans and pre-operative maximum proximal aortic diameter.
Statistical analysis
Categorical variables are expressed as the numbers and percentages of patients. Continuous variables are expressed as the mean ± standard deviation or the median [interquartile range (IQR)]. Statistical analysis was performed to compare the groups using Pearson’s chi-square or Fisher’s exact test. Survival status, including overall survival rates, and freedom from aortic-related event were assessed using Kaplan–Meier survival curves. Statistical analyses were performed using SPSS 20 software version (IBM, Armonk, NY).
Results
Patient characteristics
The baseline characteristics of 42 patients who underwent TEVAR for TTAI are shown in Table 1. The mean follow-up period was 47.9 ± 29.6 months. The baseline characteristics of 42 patients who underwent TEVAR are shown in Table 1. The median age of the patients was 47 (range 19–86) years, and 30 (71.4%) patients were males. The mean Glasgow Coma Scale (GCS) score was 13.2 ± 3.4 and the mean ISS score was 28.6 ± 8.9.
The mechanism of injury and details of aortic injury are described in Table 2. The most common mechanism of injury was automobile accidents, followed by auto-pedestrian collisions, and motorcycle accidents. Emergency TEVAR was performed for 26 (61.9%) patients, and a combined operation was performed for 21 (50.0%) patients.
The procedural details are shown in Table 3. The procedural success rate was 100% (42/42). No patient underwent or converted to open surgical aortic repair after the introduction of TEVAR, and there were no endoleaks or additional reintervention during follow-up. Zone 1 TEVAR was performed for one (2.4%) patient whose debranching of the left common carotid artery and LSA to the ascending aorta was accomplished through median sternotomy. Primary endoleaks were found in two patients, who required the insertion of a balloon angioplasty and additional stent insertion, respectively, and TEVAR was terminated. LSA revascularization was performed prior to TEVAR through median sternotomy only for Zone 1 patient.
Post-operative outcomes are shown in Table 4. There were additional aorta-related complications (two cases). One patient suffered distal malperfusion due to extended aortic injury and required additional peripheral stents insertion after TEVAR. The other patient developed rhabdomyolysis due to severe aorta rupture needing massive hydration and dialysis. The procedure-related neurological complications were not recorded; however, permanent neurologic deficit due to pre-operative poor mentality occurred in five patients. Cases of left-arm claudication or other complications related to the coverage of the LSA were not present, and spinal cord ischemia was not observed. In-hospital mortality was observed in 4 (9.5%) patients.
Survival
The overall mortality was 16.7% (7/42), and aorta-related mortality was 2.4% (1/42) during follow-up. At 1, 5, and 7 years, the cumulative survival of all-cause death was 90.5 ± 2.3%, 85.7 ± 4.1%, and 61.7 ± 8.4%, respectively. Kaplan–Meier curves of freedom from aortic-related event, cumulative survival of aorta-related death and cumulative survival of all-cause death are shown in Figs. 1, 2, and 3.
Changes in the proximal aortic size according to the proximal landing zone length
We analyzed the proximal aortic size according to the proximal landing zone length in 41 patients except for 1 patient who underwent Zone 1 TEVAR (Fig. 4). A total of 17 (47.5%) patients had proximal landing zone length of ≤ 20 mm; among them, 4 (9.5%) patients had a landing zone length of 10–15 mm from the stent covering zone to the initial tear site. There were no endoleaks or additional reintervention according to the proximal landing zone length during follow-up.
When we had comparison of the proximal aortic size increase of > 0% according to the proximal landing zone (≤ 20 mm and > 20 mm), there was no statistically significant difference between the two groups (p = 0.65). In addition, the proximal aortic size increase of ≤ 0% was not different between proximal landing zone length of > 20 mm and ≤ 20 mm (p = 0.90) (Table 5).
When comparing the aortic size and aortic diameter increase according to the zone, there was no difference between zone 2, zone 3, and zone 4 TEVAR. The details of these results are shown in Table 6.
Discussion
TTAI is a life-threatening emergency. Thoracic aortic injury occurs in 2% of patients with blunt thoracic trauma and is the second most common cause of death among trauma patients, exceeded only by intracranial hemorrhage [9,10,11]. TTAI is frequently related to a sudden deceleration in motor vehicle accidents, and the injury most commonly occurs at fixed points of the descending aorta. More than 80% of TTAIs are located in the region of the aortic isthmus and typically within 20 mm of the ligamentum arteriosum of the descending thoracic aorta [3] [12].
The injury is most likely the result of multiple simultaneous forces acting on the aorta during blunt impact [12]. These forces include movement of the sternum posteriorly with compression of the aorta onto the spine, a sudden increase in hydrostatic forces within the aorta, and the deceleration stress on the aorta with shearing and torsion of the descending aorta, which remains focally fixed by the ligamentum arteriosum [4, 12, 13].
Since TEVAR was first introduced in the 1990s [2], it has generally been recognized as an alternative to open surgery for descending aortic pathologies. Furthermore, the recent use of the hybrid approach has contributed to a wider acceptance of TEVAR. However, common post-operative adverse events associated with TEVAR, such as endoleak, stent-graft migration, and retrograde-type aortic dissection, which require reintervention for better prognosis, are regarded as its weaknesses [14].
Our study showed that the procedural success rate was 100%, and no patient underwent open surgical aortic repair after the introduction of TEVAR. There were no endoleaks or additional reinterventions during follow-up. Similarly, previous studies have reported favorable results [9, 15,16,17,18]. Ehrlichet al. [18] reported that TEVAR with 41 patients of TTAI was technically successful in all cases (100%), and the overall in-hospital mortality rate was 2.4% with a mean 13 months of follow-up. Lachat et al. [17] reported that the immediate technical success rate was 100%, and there were no post-procedure complications in all but one patient, who died 12 h post-operatively (8% mortality). There was no intervention-related morbidity or mortality during the mean follow-up of 17 months. In addition, Steuer et al. [9] reported that after a median follow-up of 36 months (range 10–98 months), among 17 patients, in-hospital mortality was 24% (4/17), and 3 patients required reintervention (18%), (each patient only once; 1 for a type I endoleak, and 2 for pseudocoarctation secondary to stent-graft infolding). Two of them were treated endovascularly, and one had a stent-graft explantation.
In our study, the overall mortality and aorta-related mortality were 16.7% (7/42) and 2.4% (1/42), respectively, during follow-up, and in-hospital mortality was observed for 4 (9.5%) patients. Of the four patients with in-hospital mortality, three patients died from pulmonary complication, and one patient died from multi-organ failure due to distal organ malperfusion by extended TTAI. The mean ISS score of these patients was 38.5 and the mean GCS score was 7.5. Among them, three patients had brain hemorrhage. Aorta-unrelated late deaths secondary to unknown causes occurred in three patients.
TEVAR should be dependent on the type of underlying pathology. Patients with TTAI are usually young or middle-aged without any preexisting aortic diseases. There are two main differences between TTAI and aneurysmal aortic disease. In particular, TTAI patients are young patients with a normal-sized aorta. Therefore, the aortic diameter in TTAI is narrower than that in aneurysmal disease [19]. Devices with a smaller diameter are required for these younger patients with TTAI whose aortas have not begun to dilate [18]. In addition, the aorta in TTAI has a more acutely angulated aortic arch [20, 21]. When the curvature of the aortic arch is more angulated, delivery, deployment, and apposition of the stent graft would be more difficult, which is representative of a bird-beak configuration [20]. If combined with excessive oversizing on a healthy, stiff aortic wall, this could lead to a predisposition to device failure and stent-graft collapse [22, 23].
Despite the various benefits of TEVAR for TTAI, the long-term results of TEVAR remain a major issue. If trauma patients are young, a longer follow-up is necessary to assess the long-term durability of the devices and the adaptability of the stent grafts to possible degenerative changes in the thoracic aorta with increasing age [9, 18]. Therefore, a multi-institutional prospective, randomized trial is required for TTAI patients. In addition, the most important anatomic characteristic of any lesion following TAVAR is the presence of an adequate proximal neck. The aortic isthmus is usually extremely close to the LSA and sometimes the lesion in contiguity or with a limited distance from the vessel. Proximal apposition is an important factor for long-term stability and patients with associated problems often require reintervention [18]. In addition, LSA coverage is required for up to 40% of patients [24, 25].
The issue of coverage of the LSA during endografting remains controversial [25, 26]. Some initial concerns are distal arm ischemia and possible vertebrobasilar insufficiency related to posterior brain circulation. Therefore, pre-operative evaluation of the vertebral arteries, vertebrobasilar system, and spinal arterial supply can allow selective revascularization [4]. If revascularization is performed, the brachial plexus and phrenic nerve may be injured [25].
Classically, for successful stent-graft placement, it is recommended that the proximal landing zone is located at least 20 mm away from the primary target lesion and that stent graft is angled appropriately in the aortic arch. However, the 20-mm landing zone is standard for a normal proximal aorta in the treatment of aneurysm disease. In the setting of TTAI of commonly in young people with previously normal aorta in emergency states, we analyzed the proximal aortic size according to the proximal landing zone length. Our results showed that when a short proximal landing zone was used, there were no endoleaks or reintervention and the aortic size increase was not significant different compared with that in the group with > 20 mm proximal landing zone length. Although it is not applicable to different aortic pathologies, this result suggests that TTAI has a unique and different pathology. In TTAI, the injury is focal and often not even circumferential. Indeed, the TTAI often completely resolves with the repair of the intimal tear after TEVAR [4]. Therefore, this report suggests that the placement of the stent graft for coverage of this lesion may not require strict adherence to this recommended proximal landing zone length of 20 mm, and providing adequate coverage for sealing the intimal tear may be sufficient for repair. In addition, stent-graft oversizing and aortic pathology are important factors that could affect the increase in aorta size. Therefore, prospective, randomized trial is required.
Our study demonstrated favorable mid-term outcomes after TEVAR compared with the results of previous studies. However, our study has several limitations. First, this was a single-center retrospective study that included a small number of patient, thus, a possible selection bias might have affected the results. Second, the follow-up duration was relatively short. Third, the lack of commercial availability and our own stock of stent grafts during emergency necessitated oversizing in several cases. Finally, the indication for TEVAR is “broad” and TEVAR was also performed for Grade I aortic injury, which have affected our results.
In conclusion, survival after TEVAR for TTAI was satisfactory in mid-term result for selected patients. In addition, a short proximal landing zone may be considered as acceptable for selective trauma patients (relatively young people with previously normal aorta). As applicability is still limited to the aortic pathology of a trauma population, further studies are required with large, randomized population.
References
Fabian TC, Richardson JD, Croce MA, Smith JS Jr, Rodman G Jr, Kearney PA, et al. Prospective study of blunt aortic injury: multicenter trial of the American Association for the surgery of trauma. J Trauma. 1997;42(3):374–80 (discussion 80-3).
Dake MD, Miller DC, Semba CP, Mitchell RS, Walker PJ, Liddell RP. Transluminal placement of endovascular stent-grafts for the treatment of descending thoracic aortic aneurysms. N Engl J Med. 1994;331(26):1729–34.
Borsa JJ, Hoffer EK, Karmy-Jones R, Fontaine AB, Bloch RD, Yoon JK, et al. Angiographic description of blunt traumatic injuries to the thoracic aorta with specific relevance to endograft repair. J Endovasc Ther. 2002;9(Suppl 2):II84–91.
Brinster DR. Endovascular repair of blunt thoracic aortic injuries. Semin Thorac Cardiovasc Surg. 2009;21(4):393–8.
Baker SP, O’Neill B, Haddon W Jr, Long WB. The injury severity score: a method for describing patients with multiple injuries and evaluating emergency care. J Trauma. 1974;14(3):187–96.
Mitchell RS, Ishimaru S, Ehrlich MP, Iwase T, Lauterjung L, Shimono T, et al. First International Summit on thoracic aortic endografting: roundtable on thoracic aortic dissection as an indication for endografting. J Endovasc Ther. 2002;9(Suppl 2):98–105.
Azizzadeh A, Keyhani K, Miller CC 3rd, Coogan SM, Safi HJ, Estrera AL. Blunt traumatic aortic injury: initial experience with endovascular repair. J Vasc Surg. 2009;49(6):1403–8.
Fillinger MF, Greenberg RK, McKinsey JF, Chaikof EL, Society for Vascular Surgery Ad Hoc Committee on TRS. Reporting standards for thoracic endovascular aortic repair (TEVAR). J Vasc Surg. 2010;52(4):1022–33 (33 e15).
Steuer J, Wanhainen A, Thelin S, Nyman R, Eriksson MO, Bjorck M. Outcome of endovascular treatment of traumatic aortic transection. J Vasc Surg. 2012;56(4):973–8.
Clancy TV, Gary Maxwell J, Covington DL, Brinker CC, Blackman D. A statewide analysis of level I and II trauma centers for patients with major injuries. J Trauma. 2001;51(2):346–51.
Ungar TC, Wolf SJ, Haukoos JS, Dyer DS, Moore EE. Derivation of a clinical decision rule to exclude thoracic aortic imaging in patients with blunt chest trauma after motor vehicle collisions. J Trauma. 2006;61(5):1150–5.
Pretre R, Chilcott M. Blunt trauma to the heart and great vessels. N Engl J Med. 1997;336(9):626–32.
Neschis DG, Scalea TM, Flinn WR, Griffith BP. Blunt aortic injury. N Engl J Med. 2008;359(16):1708–16.
Zhang L, Zhao Z, Chen Y, Sun Y, Bao J, Jing Z, et al. Reintervention after endovascular repair for aortic dissection: a systematic review and meta-analysis. J Thorac Cardiovasc Surg. 2016;152(5):1279–88 (e3).
Peterson BG, Matsumura JS, Morasch MD, West MA, Eskandari MK. Percutaneous endovascular repair of blunt thoracic aortic transection. J Trauma. 2005;59(5):1062–5.
Fattori R, Russo V, Lovato L, Di Bartolomeo R. Optimal management of traumatic aortic injury. Eur J Vasc Endovasc Surg. 2009;37(1):8–14.
Lachat M, Pfammatter T, Witzke H, Bernard E, Wolfensberger U, Kunzli A, et al. Acute traumatic aortic rupture: early stent-graft repair. Eur J Cardiothorac Surg. 2002;21(6):959–63.
Ehrlich MP, Rousseau H, Heijman R, Piquet P, Beregi JP, Nienaber CA, et al. Early outcome of endovascular treatment of acute traumatic aortic injuries: the talent thoracic retrospective registry. Ann Thorac Surg. 2009;88(4):1258–63.
Coady MA, Ikonomidis JS, Cheung AT, Matsumoto AH, Dake MD, Chaikof EL, et al. Surgical management of descending thoracic aortic disease: open and endovascular approaches: a scientific statement from the American Heart Association. Circulation. 2010;121(25):2780–804.
Garcia Reyes ME, Goncalves Martins G, Fernandez Valenzuela V, Dominguez Gonzalez JM, Maeso Lebrun J, Bellmunt Montoya S. Long-term outcomes of thoracic endovascular aortic repair focused on bird beak and oversizing in blunt traumatic thoracic aortic injury. Ann Vasc Surg. 2018;50:140–7.
Alberta HB, Takayama T, Smits TC, Wendorff BB, Cambria RP, Farber MA, et al. Aortic arch morphology and aortic length in patients with dissection, traumatic, and aneurysmal disease. Eur J Vasc Endovasc Surg. 2015;50(6):754–60.
Kasirajan K, Dake MD, Lumsden A, Bavaria J, Makaroun MS. Incidence and outcomes after infolding or collapse of thoracic stent grafts. J Vasc Surg. 2012;55(3):652–8 (discussion 8).
Spiliotopoulos K, Kokotsakis J, Argiriou M, Dedeilias P, Farsaris D, Diamantis T, et al. Endovascular repair for blunt thoracic aortic injury: 11-year outcomes and postoperative surveillance experience. J Thorac Cardiovasc Surg. 2014;148(6):2956–61.
Azizzadeh A, Ray HM, Dubose JJ, Charlton-Ouw KM, Miller CC, Coogan SM, et al. Outcomes of endovascular repair for patients with blunt traumatic aortic injury. J Trauma Acute Care Surg. 2014;76(2):510–6.
McBride CL, Dubose JJ, Miller CC 3rd, Perlick AP, Charlton-Ouw KM, Estrera AL, et al. Intentional left subclavian artery coverage during thoracic endovascular aortic repair for traumatic aortic injury. J Vasc Surg. 2015;61(1):73–9.
Antonello M, Menegolo M, Maturi C, Dall’antonia A, Lepidi S, Frigo AC, et al. Intentional coverage of the left subclavian artery during endovascular repair of traumatic descending thoracic aortic transection. J Vasc Surg. 2013;57(3):684–90 (e1).
Acknowledgements
We certify that this report is our own work and all sources of information used in this report have been fully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Son, SA., Jung, H., Cho, J.Y. et al. Mid-term outcomes of endovascular repair for traumatic thoracic aortic injury: a single-center experience. Eur J Trauma Emerg Surg 45, 965–972 (2019). https://doi.org/10.1007/s00068-019-01166-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00068-019-01166-6