Abstract
Purpose
Although the current guidelines recommend bridging thrombolysis (BT) therapy, which is intravenous thrombolysis (IVT) followed by endovascular thrombectomy (EVT), for patients with acute ischemic stroke from large vessel occlusion (AIS-LVO), the effectiveness and safety of IVT remain controversial. We performed a meta-analysis to demonstrate the non-inferiority of direct EVT alone (DEVT) compared to BT for the efficacy and safety in patients with AIS-LVO who were eligible for IVT.
Methods
The literature was searched in big databases between 1 January 1990 and 1 April 2021. The search included both randomized clinical trials (RCTs) and nonrandomized studies (NRSs) that compared DEVT with BT for patients with AIS-LVO who were eligible for IVT (time from stroke onset ≤ 4.5 h). Only NRSs with good intergroup variable matching were included in the study. Outcomes measured included 90-day functional independence, mortality, symptomatic intracranial hemorrhage (sICH), and successful recanalization. The noninferiority margin for risk difference was set at 5% from the literature review.
Results
Three RCTs (n = 1094) and four NRSs (n = 1366) were included in the meta-analysis. There were 1227 patients (49.9%) in the DEVT group and 1233 patients (50.1%) in the BT group. A statistically significant noninferiority of DEVT compared to BT was concluded in 90-day functional independence, mortality and successful reperfusion. Even in the sICH rate, DEVT group showed a superiority (risk difference, −2%; 95% confidence interval, −4 to −0.002%).
Conclusion
Evidence from RCTs and observational NRSs supports the use of DEVT (without IVT) as the first choice for treatment of patients with AIS-LVO within a time span of 4.5 h or less from stroke onset.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The current guidelines recommend bridging thrombolysis (BT) therapy, which is intravenous thrombolysis (IVT) followed by endovascular thrombectomy (EVT), for patients with acute ischemic stroke from large vessel occlusion (AIS-LVO) [1]. Although it has been proven that IVT is a reperfusion strategy with substantial efficacy, the existence of inherent unfavorable effects cannot be ignored. Implementation of IVT may delay initiation of EVT and induce worse clinical outcomes [2, 3] and IVT can result in distal migration due to thrombus fragmentation. In addition, various studies have reported increased symptomatic intracranial hemorrhage (sICH) rates in addition to higher costs of procedures [4].
To verify the effectiveness of IVT, various research studies that compared direct EVT alone (DEVT) with BT have been published and have shown conflicting results. Over several years, the investigators of several systematic reviews found that there was no significant difference in outcomes between the two reperfusion strategies [5, 6]. In contrast, the results of other meta-analyses suggested that BT had better outcomes than DEVT [7, 8]; however, those studies were likely to have a high risk of selection bias owing to a comparison of two patient groups with different eligibilities for IVT, such as stroke etiology, comorbidities, and time window from stroke onset. To minimize the aforementioned selection bias, a recent meta-analysis of patients who were only eligible for IVT found that DEVT may provide similar safety and efficacy compared with BT [9]; however, the meta-analysis did not include any randomized clinical trials (RCTs).
Therefore, we performed a meta-analysis to conclude the noninferiority of DEVT compared to BT for the efficacy and safety in patients with AIS-LVO who were eligible for IVT (with time from stroke onset ≤ 4.5 h), using both RCTs and nonrandomized studies (NRSs). Our study was designed to maximize patient matching.
Material and Methods
This research-level meta-analysis was guided and presented in a manner that adheres to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [10]. Because the data supporting this study were derived from published literature, no institutional review board approval was required. The supplementary material describes the guidelines in detail (Supplementary Table S1).
Search Strategy and Eligibility Criteria
Two investigators (K.M.J. and H.H.C.) conducted a systematic search of the PubMed, EMBASE, and Cochrane Library Central Register of Controlled Trials databases for relevant articles, which included RCTs and NRSs, between 1 January 1990, and 1 April 2021 The predefined search strategy was established in accordance with the Population, Intervention, Comparison, and Outcome (PICO) format using a combination of terms supplemented with keywords or MeSH terms (Supplementary Table S2). Additional manual screening was performed if necessary for additional candidate articles found during full-text review. The search strategy placed no restrictions on the publication language, study period, or sample size. All search results were transferred to EndNote software (version X9, Clarivate Analytics, Philadelphia, PA, USA) and electronically removed duplications. After this process, we reviewed only titles with abstracts, and we excluded studies that were not relevant to this subject or were unpublished studies, conference abstracts, letters, or case reports.
The articles identified by the full-text review were subjected to the following inclusion criteria: (1) patients aged 18 years or older and diagnosed with AIS-LVO; (2) homogeneous cohorts consisting of only patients who were eligible for IVT, including a time window from stroke onset of less than or equal to 4.5 h; (3) RCTs and NRSs that conducted patient matching using propensity scores; (4) a comparison of clinical and radiologic outcomes for patients who had undergone DEVT vs. BT; (5) a presentation of outcomes with regard to modified Rankin Scale (mRS) of 0–2 at the 90-day mark for mortality, sICH, and successful recanalization rates by groups and (6) EVT procedures, including all methods such as stent retriever, direct aspiration technique, and intra-arterial thrombolysis.
Studies were excluded if they (1) enrolled patients who were not eligible for IVT but were assigned to the DEVT group, especially with a time window from time to stroke onset of greater than 4.5 h and (2) reported results of additionally performed subgroup analyses in RCTs not originally designed as DEVT vs. BT groups. Two investigators (K.M.J., and H.H.C.) independently screened the titles and abstracts, identified duplicates, reviewed full-text articles, and determined their eligibility. Consensus was established through discussion with the corresponding author (Y.D.C.) in cases of a discrepancy between decisions.
Data Extraction and Outcomes
A standardized extraction sheet was used to extract the baseline and outcome characteristics of each included study. The data were extracted by two investigators (K.M.J. and H.H.C.) and cross-checked by a corresponding investigator (Y.D.C.). The primary outcome was defined as the rate of patients who achieved functional independence at 90 days from stroke onset, corresponding to a mRS of less than or equal to 2 (90-day functional independence). Secondary outcomes were (1) all-cause mortality within 90 days of stroke onset (mortality); (2) the incidence rates of sICH as defined by the Heidelberg bleeding classification [11] and the European Cooperative Acute Stroke Study II criteria [12] and (3) the rates of successful recanalization according to a thrombolysis in cerebral infarction grade of greater than or equal to 2b/3 (successful recanalization).
Quality Assessment
The quality assessment of the included studies was presented in a manner adherent to Cochrane Collaboration’s tool for assessing the risk of bias, version 2.0 (RoB 2.0) for RCTs and the Review of the Development of the Risk Of Bias In Nonrandomized Studies of Interventions (ROBINS-I), which included detailed guidance for the review of meta-analyses. The following five domains of the RoB 2.0 were assessed for individual trials: randomization process, intended intervention, missing data, outcome measurement, and result selection. The risk of bias was evaluated with the following three categories: low, some concerns, and high, and was assigned an overall risk by the algorithm. The checklist with seven domains in ROBINS‑I was evaluated for NRS: confounding, participant selection, intervention classification, intended intervention, missing data, outcome measurement, and result selection. After a comprehensive evaluation of the judgment of each domain, the overall risk was specified according to the algorithm. The “traffic light” plots of the domain-level judgments for each result were presented in a graphical format [13].
Statistical Analysis
All analyses in this study were performed using the Review Manager (RevMan), version 5.3 (The Cochrane Collaboration, Oxford, UK), and Stata/SE, version 15.0 (Stata Corp LP; College Station, TX, USA), software using the metan, metabias, metareg and metafunnel commands. Risk difference (RD) with 95% confidence intervals (CI) from each study were combined to estimate the pooled RD with 95% CIs for the primary and secondary outcomes. The Mantel-Haenszel random-effects model was used to comparing the DEVT with BT (the RD and 95% CIs). All 2‑sided p-values < 0.05 were considered to indicate statistical significance. Combined analysis was performed individually for the primary and secondary outcomes, and each subgroup analysis was presented by dividing it into RCTs and NRSs.
The noninferiority margin was determined based on a prior, large scale systematic review, meta-analysis and RCTs [14, 15]. The margin was set at 5%, as per a large survey of stroke experts to establish the minimally clinically important difference for stroke treatment [16]. Based on the noninferiority margin of 5%, DEVT was concluded to be noninferior to BT for an outcome when the lower 95% CI bound for treatment outcome was above the margin of −5% or the upper 95% CI bound for adverse effect was below the margin of 5%. In addition, when the 95% CI interval did not cross 0, DEVT was concluded to be superior to BT.
Statistical heterogeneity was estimated using the I2 statistic, which measures the degree of inconsistency across studies in a meta-analysis. An I2 value less than 25%, less than 50%, less than 75%, and greater than 75% was considered to indicate low, moderate, high, and severe heterogeneity, respectively. Publication bias in each study was investigated by visually estimating its asymmetry within the plots of RDs against error or variance of the study (funnel plots). If asymmetry was suspected, the Begg rank correlation test and Egger regression test were conducted; 2‑tailed p-values < 0.05 were considered to indicate potential bias.
To confirm the results and make them comparative, sensitivity analysis was additionally performed by omitting any individual studies that showed statistically significant differences from the pooled analysis for mortality, sICH, and successful recanalization. After the subgroup analysis, we consequently calculated and presented the pooled RDs with 95% CIs for the remaining studies.
Results
Study Selection and Baseline Characteristics
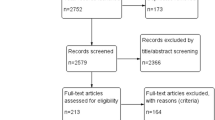
As the literature search flow diagram presented in Fig. 1 indicates, the search strategy yielded 110 articles after removal of duplicates. After retrieving 39 articles for full-text review, 7 studies (3 RCTs [17,18,19] and 4 NRSs [20,21,22,23]) met the eligibility criteria and were included in the meta-analysis.
The baseline characteristics of these seven studies are presented in Table 1. The 7 studies contained a total of 2460 patients with AIS who underwent DEVT or BT; we compared the outcomes of 90-day functional independence, mortality, sICH, and successful recanalization among these patients. Only one study was designed within a single institutional setting, and the remaining six had multicenter settings. Most studies included LVO of the anterior circulation, whereas a posterior circulation stroke involving the basilar artery and posterior cerebral artery was included in only one study.
A total of 1227 patients (49.9%) in the DEVT group and 1233 patients (50.1%) in the BT group were consequently enrolled for quantitative analysis. All included studies only enrolled patients who presented within 4.5 h of stroke onset and had no contraindications to IVT. Alteplase was administered at a dose of 0.6–0.9 mg/kg in the BT group; the mechanical thrombectomy was performed using stent-retrievers or catheter aspiration or combination method. Endovascular treatment strategies with details of individual studies are presented in supplementary table S3.
Quality Assessment and Publication bias of Included Studies
Individual risk of bias of the enrolled studies was evaluated using RoB 2.0 suitable for RCTs and ROBINS‑I for NRSs. Among the enrolled 3 RCTs, all of the ROB 2.0 domains were judged to have a low risk of bias except for one trial. The Suzuki trial could not completely rule out the possibility that the carers delivering the intervention would recognize the exposure group during the trial. Due to the limitations of the inherent characteristics of an NRS, the results evaluated through the ROBINS‑I tool showed an overall higher level of risk of bias compared with the RoB for RCTs. Domain 1 referred to confounding bias, although it was not possible to completely control the confounder owing to the properties of an NRS; therefore, it was judged that there was no serious residual confounding effect by minimizing the expected risk through propensity score matching. In addition, because it is almost impossible to achieve complete assessor blindness to intervention in the outcome measurement domain, all studies were judged as a moderate risk of bias. The authors did not exclude individual studies with moderate risk of bias because it was assumed that their individual results would not alter the overall results of the combined analysis. Traffic light plots and summary graphs are each presented for RoB 2.0 and ROBINS‑I in Fig. 2a,b, respectively.
There was no considerable visual asymmetry in the funnel plots for 90-day functional independence and sICH, whereas minor or moderate asymmetry was observed for mortality and successful recanalization (Supplementary Fig. 1). Although the presence of plot asymmetry was detected, the Begg and Egger test revealed p > 0.05, which indicated that no significant publication bias was detected.
90-day Functional Independence
The combined analysis of the RCTs and NRSs revealed a 90-day functional independence overall rate of 43.5% (1055/2423). The DEVT and BT groups achieved this primary end point at the following rates: 44.4% (537/1209) and 42.7% (518/1214), respectively. Combined result from the random-effect model showed the RD for 90-day functional independence was 2% (95% CI, −2 to 6%), which was concluded to be a statistically significant non-inferiority of DEVT to BT. Subgroup analysis of both RCTs and NRSs demonstrated relatively similar results which the DEVT group presented a non-inferiority compared to BT in 90-day functional independence (RCT RD, 2%; 95% CI, −4 to 7% and NRS RD 2%; 95% CI, −4 to 7%). Furthermore, I2 was found to be 0%, indicating low heterogeneity between the enrolled studies in the combined and subgroup analysis (Fig. 3).
Forest plot presenting the association of direct endovascular thrombectomy alone (DEVT), compared with bridging thrombolysis (BT), with 90-day functional independence (defined as modified Rankin Scale of 0–2) in all included studies stratified by study design (RCT vs. NRS). Tests for heterogeneity are described using I2 and P value. RCT randomized controlled trial, NRS non-randomized study
Mortality and sICH
In the combined analysis, the mortality rate was 18.8% (455/2423), and the rates of the DEVT and BT groups were 17.8% (215/1209) and 19.8% (240/1214), respectively. The integrated results indicated the RD for mortality was −2% (95% CI, −6% to 2%). Subgroup analysis showed that the RD was RCT −1% (95% CI −5% to 3%) and NRS −5% (95% CI, −14% to 4%), respectively (Fig. 4a).
Forest plot presenting the association of direct endovascular thrombectomy alone (DEVT), compared with bridging thrombolysis (BT) in all included studies stratified by study design (RCT versus NRS) with a mortality, b symptomatic intracranial hemorrhage, and c successful recanalization (defined as thrombolysis in cerebral infarction grade ≥ 2B). Tests for heterogeneity are described using I2 and P value. RCT randomized controlled trial, NRS non-randomized study
The pooled results for sICH showed that DEVT presented a superiority. The overall sICH rates were DEVT 6.3% (76/1210), BT 8.5% (103/1210), RD −2% (95% CI, −4% to −0.002%). Subgroup analysis indicated the RD of RCT −2% (95% CI, −5% to 1%) and NRS −1% (95% CI, −4% to 2%) (Fig. 4b). Meanwhile, there were no substantial heterogeneities between the included studies in the combined analysis for mortality and sICH.
Successful Recanalization
In the combined analysis, 85.7% (2078/2424) of patients achieved successful recanalization. Among the specific groups, 85.2% (1027/1206) of patients achieved recanalization in the DEVT group compared with 86.3% (1051/1218) of patients in the BT group. The combined analysis indicated that DEVT was non-inferior for the successful recanalization compared to BT. The pooled results indicated the total RD 0% (95% CI, −4% to 4%), RCT RD −3% (−8% to 1%) and NRS RD 3% (−5% to 10%) (Fig. 4c).
Sensitivity Analysis
Although none of the studies enrolled for the 90-day functional independence test showed biased results, further analyses were required for mortality, sICH, and successful recanalization. After eliminating eccentric studies, the combined analysis showed that DEVT was consistently non-inferior to BT for mortality, sICH, and successful recanalization (RD, −1%; 95% CI, −4% to 2%; RD, −1%; 95% CI, −3% to 1%; and RD, −3%; 95% CI, −4% to 0%, respectively) (Supplementary Fig. 2). Even in sub-analysis except for one study involving posterior circulation stroke, non-inferiority was maintained in the 90-day functional independence test. (RD, 2%; 95% CI, −3% to 6%) (Supplementary Fig. 3).
Discussion
Although the current guidelines recommend BT for patients with AIS-LVO, the effectiveness and safety of IVT using alteplase have been controversial in the current EVT era. In this meta-analysis, a statistically significant non-inferiority of DEVT compared to BT was concluded in 90-day functional independence, mortality and successful reperfusion in patients with AIS-LVO who were eligible for IVT (time from stroke onset ≤ 4.5 h). Even in the sICH rate, DEVT group showed a superiority, where the additional cost associated with IVT can also be avoided. The results of this meta-analysis that incorporated three recently published RCTs and four patient-matched NRSs suggest that DEVT alone offers clinically non-inferior effectiveness compared with BT in patients with AIS-LVO who were eligible for both reperfusion strategies.
Various previous meta-analyses that have compared clinical outcomes between DEVT and BT have been reported. Similar to our findings, some systematic reviews indicated no significant differences in clinical outcomes between DEVT and BT in patients with AIS-LVO [5, 6]. Although these reviews included both unadjusted or nonrandomized studies, results revealed that DEVT had equal clinical effectiveness to BT. Liu et al. [5] reported that DEVT was associated with a lower risk of intracranial hemorrhage compared with BT.
On the contrary, Mistry et al. [7] conducted a pooled analysis of 13 studies, including the Highly Effective Reperfusion Evaluated in Multiple Endovascular Stroke Trials (HERMES) collaboration [24]. Results of the integrated analysis suggested that patients with BT had better 90-day functional outcomes, lower mortality, higher recanalization rate, and equal odds of sICH compared with DEVT; however, as the possibility of selection bias could exist, those findings should be interpreted with caution. These discrepancies noted among several meta-analyses could partially be due to two major limitations: a difference in study design and the confounding effect of IVT eligibility. Because those RCTs [25,26,27,28,29] were originally designed as IVT alone vs. BT, studies with participants repurposed and reclassified through post hoc or subgroup analysis were likely to be subject to substantial confounding bias.
Indeed, IVT eligibility has been presumed to be a confounder of these contradictory findings [21]. It has been known that patients who are ineligible for IVT are more likely to have underlying factors predicting a relatively worse clinical outcome. For example, these patients may have received treatment beyond the 4.5‑h window from stroke onset, or they may have pre-existing comorbidities, such as cardiac disease or prior stroke [9, 30]. In particular, the association of time delay with poorer outcomes was demonstrated in measures of probability of functional independence [3]. Patients with ischemic stroke and past history of atrial fibrillation have relatively worse clinical outcomes than those without comorbidities [31].
In this respect, in an effort to minimize risk of bias or confounding effects, recent meta-analyses have attempted to obtain comparable baseline profiles by conducting a subgroup analysis suitable for both DEVT and BT through adjustments for IVT eligibility [5, 9]. The results of these studies were generally consistent with our findings for both the primary and secondary outcomes. In particular, Kaesmacher et al. [9] suggested that an RCT format is necessary because IVT ineligibility itself can be a confounder that causes poor technical and clinical success. In this respect our updated meta-analysis included three recently published RCTs with relatively substantial patient samples with various clinical variables and IVT eligibility that were adequately controlled; results showed that DEVT alone could offer similar non-inferior effectiveness and safety compared with BT.
Various studies have reported that IVT increases the risk of ICH [1]. Furthermore, sICH was reported to be associated with a worse 90-day functional independence and higher mortality. Our study reinforced these results because DEVT was found to have a statistically lower sICH rate compared with IVT in patients with AIS. Therefore, it would be estimated that DEVT alone has advantages in terms of 90-day clinical outcome and mortality of patients. In addition, IVT was known as a sole factor that induced clot migration and retriever inaccessibility [4, 32]. These periprocedural findings distinctly observed in the patients treated with BT would make it difficult to achieve a first-pass effect or ultimately have a lower score on the thrombolysis in cerebral infarction (TICI) grading scale. Of note, given that a higher first-pass effect and TICI grade indicate better outcomes [33, 34], it may be consequently deduced that patients treated with BT would have worse outcomes than those treated with DEVT alone.
Shortening of the time from stroke onset has been proven to be an essential factor associated with better outcomes in patients with AIS-LVO. Given this consideration, these patients should receive direct transport to a DEVT-capable stroke center equipped with an angiography suite to reduce the time delay before implementation of DEVT [35]. Meanwhile, because delaying the door-to-puncture time in BT has been proved through many published studies [36], we investigated patient outcomes with or without IVT in the current meta-analysis. Our results suggest a non-inferior effectiveness of DEVT compared with BT. These findings may provide the rationale for conducting related studies in the future. Although it would be challenging to apply these results to all patients with AIS-LVO, the specific strategy with DEVT could be an alternative option to current treatment strategies for AIS-LVO, including IVT alone and BT, based on individual patient clinical profiles.
Several limitations were inherent to our study. First, the protocols (i.e. thrombectomy procedure) of the studies included in this analysis were not completely consistent. Although all EVT procedures such as stent retriever, catheter aspiration, and intra-arterial thrombolysis were included, we could not analyze the confounding effect of individual procedural differences. In particular, with the exception of three RCTs, in most of the NRSs, the procedure was selected at the discretion of the clinician. Therefore, a corresponding potential bias could exist, and cautious consideration might be required when interpreting these results or changing the treatment strategy for patients with AIS-LVO. Second, we were unable to perform meta-regression analysis on the variable of time delay, which is an important factor in the judgment of patient eligibility for IVT. This is because the results of subgroup analyses that used time interval as an independent variable could not be found in the enrolled studies. In the future, individual studies on the effect of time delay on the comparison of DEVT and BT will be required. Third, due to the inherent limitations of NRSs, the variables identified or not identified in individual studies may represent potential confounding factors that cannot be excluded despite propensity score matching. A larger volume meta-analysis including additional ongoing RCTs will be needed to improve study quality and reliability and to ensure generalization of DEVT to all patients with AIS-LVO who are eligible to receive DEVT, BT, or both. Finally, this study could not demonstrate non-inferiority for the most stringent margin of 1.3% [37]. The current consensus for selection of the margin recommends the strictest figure should be applied; however, the discrepancies between the scientific and the “real-world” applicable margin would be substantially challenging for conducting the non-inferior analysis. In fact, even in the previously published meta-analyses, the margin of 1.3% has not demonstrated the non-inferiority [14, 15]. Moreover, our study met all of the fairly stringent non-inferiority margin of 5% assessed for key efficacy outcomes. It represents a strong evidence base for the non-inferiority of DEVT compared to BT.
Conclusion
The current meta-analysis of 7 studies, including 2460 patients with AIS-LVO who were eligible for treatment with both DEVT and BT, compared efficacy and safety between the two reperfusion strategies. The results of this analysis demonstrated that DEVT alone management of AIS revealed non-inferior effectiveness in 90-day functional independence, mortality, and recanalization outcomes compared with BT and may result in a significantly lower incidence of sICH. Although it is recognized that all patients with AIS-LVO could not be subject to this reperfusion strategy randomly, our results contribute to the hierarchy of evidence for DEVT and reveal various potential management options, including IVT alone, BT, and DEVT, depending on the individual characteristics of each patient with AIS-LVO.
References
Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, Biller J, Brown M, Demaerschalk BM, Hoh B, Jauch EC, Kidwell CS, Leslie-Mazwi TM, Ovbiagele B, Scott PA, Sheth KN, Southerland AM, Summers DV, Tirschwell DL. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2019;50:e344-418. Erratum in: Stroke. 2019;50:e440-1.
Ribo M, Boned S, Rubiera M, Tomasello A, Coscojuela P, Hernández D, Pagola J, Juega J, Rodriguez N, Muchada M, Rodriguez-Luna D, Molina CA. Direct transfer to angiosuite to reduce door-to-puncture time in thrombectomy for acute stroke. J Neurointerv Surg. 2018;10:221-4.
Saver JL, Goyal M, van der Lugt A, Menon BK, Majoie CB, Dippel DW, Campbell BC, Nogueira RG, Demchuk AM, Tomasello A, Cardona P, Devlin TG, Frei DF, du Mesnil de Rochemont R, Berkhemer OA, Jovin TG, Siddiqui AH, van Zwam WH, Davis SM, Castaño C, Sapkota BL, Fransen PS, Molina C, van Oostenbrugge RJ, Chamorro Á, Lingsma H, Silver FL, Donnan GA, Shuaib A, Brown S, Stouch B, Mitchell PJ, Davalos A, Roos YB, Hill MD; HERMES Collaborators. Time to Treatment With Endovascular Thrombectomy and Outcomes From Ischemic Stroke: A Meta-analysis. JAMA. 2016;316:1279-88.
Kaesmacher J, Boeckh-Behrens T, Simon S, Maegerlein C, Kleine JF, Zimmer C, Schirmer L, Poppert H, Huber T. Risk of Thrombus Fragmentation during Endovascular Stroke Treatment. AJNR Am J Neuroradiol. 2017;38:991-8.
Liu M, Li G. Is Direct Endovascular Treatment as an Alternative of Bridging Therapy in Acute Stroke Patients with Large Vessel Occlusion? J Stroke Cerebrovasc Dis. 2019;28:531-41.
Phan K, Dmytriw AA, Maingard J, Asadi H, Griessenauer CJ, Ng W, Kewagamang K, Mobbs RJ, Moore JM, Ogilvy CS, Thomas AJ. Endovascular Thrombectomy Alone versus Combined with Intravenous Thrombolysis. World Neurosurg. 2017;108:850-8.e2.
Mistry EA, Mistry AM, Nakawah MO, Chitale RV, James RF, Volpi JJ, Fusco MR. Mechanical Thrombectomy Outcomes With and Without Intravenous Thrombolysis in Stroke Patients: A Meta-Analysis. Stroke. 2017;48:2450-6.
Pan X, Liu G, Wu B, Liu X, Fang Y. Comparative efficacy and safety of bridging strategies with direct mechanical thrombectomy in large vessel occlusion: A systematic review and meta-analysis. Medicine (Baltimore). 2019;98:e14956.
Kaesmacher J, Mordasini P, Arnold M, López-Cancio E, Cerdá N, Boeckh-Behrens T, Kleine JF, Goyal M, Hill MD, Pereira VM, Saver JL, Gralla J, Fischer U. Direct mechanical thrombectomy in tPA-ineligible and -eligible patients versus the bridging approach: a meta-analysis. J Neurointerv Surg. 2019;11:20-7.
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6:e1000100.
von Kummer R, Broderick JP, Campbell BC, Demchuk A, Goyal M, Hill MD, Treurniet KM, Majoie CB, Marquering HA, Mazya MV, San Román L, Saver JL, Strbian D, Whiteley W, Hacke W. The Heidelberg Bleeding Classification: Classification of Bleeding Events After Ischemic Stroke and Reperfusion Therapy. Stroke. 2015;46:2981-6.
Hacke W, Kaste M, Fieschi C, von Kummer R, Davalos A, Meier D, Larrue V, Bluhmki E, Davis S, Donnan G, Schneider D, Diez-Tejedor E, Trouillas P. Randomised double-blind placebo-controlled trial of thrombolytic therapy with intravenous alteplase in acute ischaemic stroke (ECASS II). Second European-Australasian Acute Stroke Study Investigators. Lancet. 1998;352:1245-51.
McGuinness LA, Higgins JPT. Risk-of-bias VISualization (robvis): An R package and Shiny web app for visualizing risk-of-bias assessments. Res Synth Methods. 2021;12:55-61.
Lin CH, Saver JL, Ovbiagele B, Huang WY, Lee M. Endovascular thrombectomy without versus with intravenous thrombolysis in acute ischemic stroke: a non-inferiority meta-analysis of randomized clinical trials. J Neurointerv Surg. 2021; https://doi.org/10.1136/neurintsurg-2021-017667. Epub ahead of print.
Burgos AM, Saver JL. Evidence that Tenecteplase Is Noninferior to Alteplase for Acute Ischemic Stroke: Meta-Analysis of 5 Randomized Trials. Stroke. 2019;50:2156-62.
Fisher M, Albers GW, Donnan GA, Furlan AJ, Grotta JC, Kidwell CS, Sacco RL, Wechsler LR; Stroke Therapy Academic Industry Roundtable IV. Enhancing the development and approval of acute stroke therapies: Stroke Therapy Academic Industry roundtable. Stroke. 2005;36:1808-13.
Suzuki K, Matsumaru Y, Takeuchi M, Morimoto M, Kanazawa R, Takayama Y, Kamiya Y, Shigeta K, Okubo S, Hayakawa M, Ishii N, Koguchi Y, Takigawa T, Inoue M, Naito H, Ota T, Hirano T, Kato N, Ueda T, Iguchi Y, Akaji K, Tsuruta W, Miki K, Fujimoto S, Higashida T, Iwasaki M, Aoki J, Nishiyama Y, Otsuka T, Kimura K; SKIP Study Investigators. Effect of Mechanical Thrombectomy Without vs With Intravenous Thrombolysis on Functional Outcome Among Patients With Acute Ischemic Stroke: The SKIP Randomized Clinical Trial. JAMA. 2021;325:244-253. Erratum in: JAMA. 2021;325:1795.
Yang P, Zhang Y, Zhang L, Zhang Y, Treurniet KM, Chen W, Peng Y, Han H, Wang J, Wang S, Yin C, Liu S, Wang P, Fang Q, Shi H, Yang J, Wen C, Li C, Jiang C, Sun J, Yue X, Lou M, Zhang M, Shu H, Sun D, Liang H, Li T, Guo F, Ke K, Yuan H, Wang G, Yang W, Shi H, Li T, Li Z, Xing P, Zhang P, Zhou Y, Wang H, Xu Y, Huang Q, Wu T, Zhao R, Li Q, Fang Y, Wang L, Lu J, Li Y, Fu J, Zhong X, Wang Y, Wang L, Goyal M, Dippel DWJ, Hong B, Deng B, Roos YBWEM, Majoie CBLM, Liu J; DIRECT-MT Investigators. Endovascular Thrombectomy with or without Intravenous Alteplase in Acute Stroke. N Engl J Med. 2020;382:1981-93.
Zi W, Qiu Z, Li F, Sang H, Wu D, Luo W, Liu S, Yuan J, Song J, Shi Z, Huang W, Zhang M, Liu W, Guo Z, Qiu T, Shi Q, Zhou P, Wang L, Fu X, Liu S, Yang S, Zhang S, Zhou Z, Huang X, Wang Y, Luo J, Bai Y, Zhang M, Wu Y, Zeng G, Wan Y, Wen C, Wen H, Ling W, Chen Z, Peng M, Ai Z, Guo F, Li H, Guo J, Guan H, Wang Z, Liu Y, Pu J, Wang Z, Liu H, Chen L, Huang J, Yang G, Gong Z, Shuai J, Nogueira RG, Yang Q; DEVT Trial Investigators. Effect of Endovascular Treatment Alone vs Intravenous Alteplase Plus Endovascular Treatment on Functional Independence in Patients With Acute Ischemic Stroke: The DEVT Randomized Clinical Trial. JAMA. 2021;325:234-43.
Bellwald S, Weber R, Dobrocky T, Nordmeyer H, Jung S, Hadisurya J, Mordasini P, Mono ML, Stracke CP, Sarikaya H, Bernasconi C, Berger K, Arnold M, Chapot R, Gralla J, Fischer U. Direct Mechanical Intervention Versus Bridging Therapy in Stroke Patients Eligible for Intravenous Thrombolysis: A Pooled Analysis of 2 Registries. Stroke. 2017;48:3282-8.
Broeg-Morvay A, Mordasini P, Bernasconi C, Bühlmann M, Pult F, Arnold M, Schroth G, Jung S, Mattle HP, Gralla J, Fischer U. Direct Mechanical Intervention Versus Combined Intravenous and Mechanical Intervention in Large Artery Anterior Circulation Stroke: A Matched-Pairs Analysis. Stroke. 2016;47:1037-44.
Tong X, Wang Y, Fiehler J, Bauer CT, Jia B, Zhang X, Huo X, Luo G, Wang A, Pan Y, Ma N, Gao F, Mo D, Song L, Sun X, Liu L, Deng Y, Li X, Wang B, Ma G, Wang Y, Ren Z, Miao Z; ANGEL-ACT Study Group. Thrombectomy Versus Combined Thrombolysis and Thrombectomy in Patients With Acute Stroke: A Matched-Control Study. Stroke. 2021;52:1589-600.
Wang H, Zi W, Hao Y, Yang D, Shi Z, Lin M, Wang S, Liu W, Wang Z, Liu X, Guo F, Liu Y, Xu G, Xiong Y, Liu X; ACTUAL Investigators. Direct endovascular treatment: an alternative for bridging therapy in anterior circulation large-vessel occlusion stroke. Eur J Neurol. 2017;24:935-43.
Goyal M, Menon BK, van Zwam WH, Dippel DW, Mitchell PJ, Demchuk AM, Dávalos A, Majoie CB, van der Lugt A, de Miquel MA, Donnan GA, Roos YB, Bonafe A, Jahan R, Diener HC, van den Berg LA, Levy EI, Berkhemer OA, Pereira VM, Rempel J, Millán M, Davis SM, Roy D, Thornton J, Román LS, Ribó M, Beumer D, Stouch B, Brown S, Campbell BC, van Oostenbrugge RJ, Saver JL, Hill MD, Jovin TG; HERMES collaborators. Endovascular thrombectomy after large-vessel ischaemic stroke: a meta-analysis of individual patient data from five randomised trials. Lancet. 2016;387:1723-31.
Berkhemer OA, Fransen PS, Beumer D, van den Berg LA, Lingsma HF, Yoo AJ, Schonewille WJ, Vos JA, Nederkoorn PJ, Wermer MJ, van Walderveen MA, Staals J, Hofmeijer J, van Oostayen JA, Lycklama à Nijeholt GJ, Boiten J, Brouwer PA, Emmer BJ, de Bruijn SF, van Dijk LC, Kappelle LJ, Lo RH, van Dijk EJ, de Vries J, de Kort PL, van Rooij WJ, van den Berg JS, van Hasselt BA, Aerden LA, Dallinga RJ, Visser MC, Bot JC, Vroomen PC, Eshghi O, Schreuder TH, Heijboer RJ, Keizer K, Tielbeek AV, den Hertog HM, Gerrits DG, van den Berg-Vos RM, Karas GB, Steyerberg EW, Flach HZ, Marquering HA, Sprengers ME, Jenniskens SF, Beenen LF, van den Berg R, Koudstaal PJ, van Zwam WH, Roos YB, van der Lugt A, van Oostenbrugge RJ, Majoie CB, Dippel DW; MR CLEAN Investigators. A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med. 2015;372:11-20. Erratum in: N Engl J Med. 2015;372:394.
Campbell BC, Mitchell PJ, Kleinig TJ, Dewey HM, Churilov L, Yassi N, Yan B, Dowling RJ, Parsons MW, Oxley TJ, Wu TY, Brooks M, Simpson MA, Miteff F, Levi CR, Krause M, Harrington TJ, Faulder KC, Steinfort BS, Priglinger M, Ang T, Scroop R, Barber PA, McGuinness B, Wijeratne T, Phan TG, Chong W, Chandra RV, Bladin CF, Badve M, Rice H, de Villiers L, Ma H, Desmond PM, Donnan GA, Davis SM; EXTEND-IA Investigators. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med. 2015;372:1009-18.
Goyal M, Demchuk AM, Menon BK, Eesa M, Rempel JL, Thornton J, Roy D, Jovin TG, Willinsky RA, Sapkota BL, Dowlatshahi D, Frei DF, Kamal NR, Montanera WJ, Poppe AY, Ryckborst KJ, Silver FL, Shuaib A, Tampieri D, Williams D, Bang OY, Baxter BW, Burns PA, Choe H, Heo JH, Holmstedt CA, Jankowitz B, Kelly M, Linares G, Mandzia JL, Shankar J, Sohn SI, Swartz RH, Barber PA, Coutts SB, Smith EE, Morrish WF, Weill A, Subramaniam S, Mitha AP, Wong JH, Lowerison MW, Sajobi TT, Hill MD; ESCAPE Trial Investigators. Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med. 2015;372:1019-30.
Jovin TG, Chamorro A, Cobo E, de Miquel MA, Molina CA, Rovira A, San Román L, Serena J, Abilleira S, Ribó M, Millán M, Urra X, Cardona P, López-Cancio E, Tomasello A, Castaño C, Blasco J, Aja L, Dorado L, Quesada H, Rubiera M, Hernandez-Pérez M, Goyal M, Demchuk AM, von Kummer R, Gallofré M, Dávalos A; REVASCAT Trial Investigators. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N Engl J Med. 2015;372:2296-306.
Saver JL, Goyal M, Bonafe A, Diener HC, Levy EI, Pereira VM, Albers GW, Cognard C, Cohen DJ, Hacke W, Jansen O, Jovin TG, Mattle HP, Nogueira RG, Siddiqui AH, Yavagal DR, Baxter BW, Devlin TG, Lopes DK, Reddy VK, du Mesnil de Rochemont R, Singer OC, Jahan R; SWIFT PRIME Investigators. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N Engl J Med. 2015;372:2285-95.
Vidale S, Romoli M, Consoli D, Agostoni EC. Bridging versus Direct Mechanical Thrombectomy in Acute Ischemic Stroke: A Subgroup Pooled Meta-Analysis for Time of Intervention, Eligibility, and Study Design. Cerebrovasc Dis. 2020;49:223-32.
Arnold M, Halpern M, Meier N, Fischer U, Haefeli T, Kappeler L, Brekenfeld C, Mattle HP, Nedeltchev K. Age-dependent differences in demographics, risk factors, co-morbidity, etiology, management, and clinical outcome of acute ischemic stroke. J Neurol. 2008;255:1503-7.
Ren Y, Churilov L, Mitchell P, Dowling R, Bush S, Yan B. Clot Migration Is Associated With Intravenous Thrombolysis in the Setting of Acute Ischemic Stroke. Stroke. 2018;49:3060-2.
Bai X, Zhang X, Yang W, Zhang Y, Wang T, Xu R, Wang Y, Li L, Feng Y, Yang K, Wang X, Song H, Ma Q, Jiao L. Influence of first-pass effect on recanalization outcomes in the era of mechanical thrombectomy: a systemic review and meta-analysis. Neuroradiology. 2021;63:795-807.
Jang KM, Nam TK, Ko MJ, Choi HH, Kwon JT, Park SW, Byun JS. Thrombolysis in Cerebral Infarction Grade 2C or 3 Represents a Better Outcome than 2B for Endovascular Thrombectomy in Acute Ischemic Stroke: A Network Meta-Analysis. World Neurosurg. 2020;136:e419-39.
Mendez B, Requena M, Aires A, Martins N, Boned S, Rubiera M, Tomasello A, Coscojuela P, Muchada M, Rodríguez-Luna D, Rodríguez-Villatoro N, Juega J, Pagola J, Molina CA, Ribó M. Direct Transfer to Angio-Suite to Reduce Workflow Times and Increase Favorable Clinical Outcome. Stroke. 2018;49:2723-7.
Atchaneeyasakul K, Desai S, Malhotra K, Dolia J, Shah K, Saver JL, Jovin T, Jadhav AP. Intravenous tPA Delays Door-To-Puncture Time in Acute Ischemic Stroke with Large Vessel Occlusion. J Stroke Cerebrovasc Dis. 2021;30:105732.
Cranston JS, Kaplan BD, Saver JL. Minimal Clinically Important Difference for Safe and Simple Novel Acute Ischemic Stroke Therapies. Stroke. 2017;48:2946-51.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
K.M. Jang, H.H. Choi, M.-j. Jang and Y.D. Cho declare that they have no competing interests.
Supplementary Information
62_2021_1116_MOESM1_ESM.docx
Supplementary Table S1. Checklist of items to include when reporting a systematic review or meta-analysis (PRISMA guideline).
62_2021_1116_MOESM2_ESM.tif
Supplementary Table S2. Search strategy in accordance with the Population, Intervention, Comparison and Outcome (PICO) format.
62_2021_1116_MOESM4_ESM.tif
Supplementary Fig. 1 Funnel plots for publication bias in (A) 90-day functional independence (defined as modified Rankin Scale scores of 0–2), (B) mortality, (C) symptomatic intracerebral hemorrhage, and (D) successful recanalization
62_2021_1116_MOESM5_ESM.tif
Supplementary Fig. 2 Sensitivity analysis for the association of direct endovascular thrombectomy alone (DEVT) compared with bridging thrombolysis (BT) with (A) mortality excluding the Broeg-Morvay trial, (B) symptomatic intracranial hemorrhage excluding the Tong trial, and (C) successful recanalization excluding the Wang trial. Tests for heterogeneity are described using I2 and P value. RCT randomized clinical trial, NRS nonrandomized study
62_2021_1116_MOESM6_ESM.tif
Supplementary Fig. 3 Sensitivity analysis for the association of direct endovascular thrombectomy alone (DEVT) compared with bridging thrombolysis (BT) with 90-day functional independence excluding study involving posterior circulation stroke. Tests for heterogeneity are described using I2 and P value. RCT randomized clinical trial, NRS nonrandomized study
Rights and permissions
About this article
Cite this article
Jang, K.M., Choi, H.H., Jang, Mj. et al. Direct Endovascular Thrombectomy Alone vs. Bridging Thrombolysis for Patients with Acute Ischemic Stroke. Clin Neuroradiol 32, 603–613 (2022). https://doi.org/10.1007/s00062-021-01116-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00062-021-01116-z