Abstract
Background
The use of intravenous thrombolysis (IVT) before mechanical thrombectomy (MT) for acute ischemic stroke due to large vessel occlusion (AIS-LVO) is a debatable subject in the field of neuro-interventional surgery. We conducted this systematic review and meta-analysis to synthesize evidence from published studies on the outcomes of IVT + MT compared with MT alone in AIS-LVO patients.
Methods
We searched PubMed, Scopus, Web of Science, and Cochrane Central Register of Controlled Trials from inception to January 2022 for relevant clinical trials and observational studies. Eligible studies were identified, and all relevant outcomes were pooled in the meta-analysis DerSimonian-Liard random-effects model.
Results
Forty-nine studies, with a total of 36,123 patients, were included in this meta-analysis. IVT + MT was significantly superior to MT alone in terms of successful recanalization (RR 1.06, 95% CI 1.03 to 1.09), mortality (RR 0.75, 95% CI 0.68–0.82), favorable functional outcome (RR 1.21, 95% CI 1.13 to 1.29), and complete recanalization (RR 1.06, 95% CI 1.00 to 1.11). There were no significant differences between the two groups in terms of improvement of the National Institute of Health Stroke Scale (NIHSS) score at 24 h or at discharge (p > 0.05). Complications including symptomatic intracranial hemorrhage, symptomatic intracerebral hemorrhage (sICH), procedure-related complications, and parenchymal hematoma were comparable between the two groups (p > 0.05).
Conclusion
For AIS-LVO, IVT + MT is associated with slightly better rates of survival, successful and complete recanalization, and favorable functional outcome as compared with MT alone. Further clinical trials are needed to corroborate such benefits of bridging IVT.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The efficacy of intravenous thrombolysis (IVT) has been previously studied in acute ischemic stroke (AIS) patients when given in the strict time window and in the absence of contraindications [1]. However, the efficacy of IVT is limited in AIS patients with large vessel occlusion (AIS-LVO), with only 6–30% of patients achieving successful recanalization [2]. Moreover, IVT might increase the risk of secondary hemorrhage, which limits its applicability in this patient population [3, 4]. On the other hand, mechanical thrombectomy (MT) has provided higher successful recanalization rates and has, therefore, become the standard of care for patients with AIS-LVO who meet the eligibility criteria for endovascular treatment (EVT) [5, 6].
Given the differences between IVT and MT in procedures, time window, and canalization mechanism, investigators sought to investigate whether the use of IVT prior to MT (known as the bridging therapy) provides greater benefit than MT alone. It has been advocated that IVT prior to MT might shorten the time of successful MT by changing the nature of the blood clot, making it more amenable to mechanical intervention, and dissolving the residual thrombotic material [7, 8]. On the other hand, the administration of IVT in combination with MT might increase the risk of intracranial hemorrhage.
According to the American Stroke Association [6], eligible AIS-LVO patients can still receive IVT and undergo MT. Several studies, both randomized clinical trials (RCTs) and observational studies, have compared MT alone with MT + IVT in patients with AIS-LVO, meeting the criteria for both treatment modalities. However, the results of published studies are inconsistent and inconclusive in terms of functional outcome, secondary intracranial hemorrhage, and mortality [8,9,10,11,12,13].
To date, experts and clinical practice guidelines agree that IVT + MT is statistically superior to MT alone; however, there are some doubts about whether this superiority is significantly sufficient to advocate the use of IVT + MT over MT alone for AIS-LVO. There is a lack of class I evidence on the associated benefits and risks of combining IVT and MT in AIS-LVO compared with MT alone. Therefore, we conducted this systematic review and meta-analysis to synthesize evidence from all published studies on the outcomes of combined bridging therapy and MT compared with MT alone in terms of successful recanalization, complete recanalization, mortality, functional independence, NIH stroke score (NIHSS), and complications.
Methods
We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement guidelines during this systematic review and meta-analysis [14]. The methods were done in strict accordance with the Cochrane Handbook of Systematic Reviews and Meta-analysis of Interventions (version 5.1.0).
Eligibility criteria
We included all studies satisfying the following criteria:
-
1.
Population: studies on patients with AIS-LVO
-
2.
Intervention: studies where the experimental or exposed group received IVT + MT
-
3.
Comparator: studies where the control group received MT alone
-
4.
Outcome: we included studies reporting at least one of the following outcomes: mortality rates, successful recanalization, complete recanalization, favorable mRS at 90 days, NIHSS score at baseline, 24 h after the stroke event and/or at discharge from the hospital, onset-to-groin time, procedural time, and complications.
-
5.
Study design: studies with comparative designs, whether RCTs or observational studies comparing the outcomes of MT alone with IVT + MT.
We excluded studies that were not in English and studies on either MT alone or MT combined with IVT without direct comparison between both arms.
Literature search
We performed a comprehensive literature search of four electronic databases (PubMed, Scopus, Web of Science, Cochrane CENTRAL) from inception until 1 January 2022 using this search query (“Mechanical thrombectomy” OR “Endovascular thrombectomy”) AND (Intravenous thrombolysis” OR Alteplase) AND (“Acute Ischemic Stroke” OR AIS). All duplicates were removed, and all references of the included studies were screened manually for any eligible studies.
Screening of the literature search results
Retrieved results from the literature search were screened in two steps. In the first step, the title and abstracts of all articles were screened for eligibility. Then, the full-text articles of eligible abstracts were retrieved and screened for eligibility.
Data extraction
Data were extracted to a uniform data extraction sheet. The extracted data included (1) characteristics of the included studies, (2) characteristics of the population of included studies, (3) risk of bias domains, and (4) outcome measures.
Outcome measures
In the present meta-analysis, we considered the following outcome measures:
Mortality
Defined as the proportion of patients who died; it is represented as the risk ratio (RR) between the two groups.
Successful recanalization
Defined as the proportion of patients with Thrombolysis in Cerebral Infarction (mTICI) grades 2b-3; it is represented as the RR between the two groups.
Complete recanalization
Defined as the proportion of patients with modified Thrombolysis in Cerebral Infarction (mTICI) grade 3; it is represented as the RR between the two groups.
Functional independence or favorable functional outcome
It is defined as achieving modified Rankin Scale (mRS) score of 0–2; it is represented as the RR between the two groups.
NIHSS (change from baseline)
The NIH stroke score will be presented as the mean difference between the two study groups from baseline to endpoint.
Symptomatic intracerebral hemorrhage
The incidence of symptomatic intracerebral hemorrhage (sICH) will be expressed as the RR between the two groups.
Symptomatic intracranial hemorrhage
The incidence of symptomatic intracranial hemorrhage will be expressed as the RR between the two groups.
Parenchymal hematoma
The incidence of parenchymal hematoma will be expressed as the RR between the two groups.
Procedure-related complications
The incidence of procedure-related complications will be expressed as the RR between the two groups.
Onset-to-groin time (in minutes)
It is defined as the time from onset of stroke to groin puncture; it will be represented as the mean difference between the two study groups.
Procedural time (in minutes)
It is defined as the time from groin puncture to recanalization; it will be represented as the mean difference between the two study groups.
Synthesis of results
For outcomes that constitute continuous data, the mean difference (MD) between the two groups from the baseline to the endpoint, with its standard deviation (SD), were pooled in the DerSimonian-Laird random-effect model. In the case of studies reporting data in multiple time points, we considered the last endpoint for the primary analysis. For outcomes that constitute dichotomous data, the frequency of events and the total number of patients in each group were pooled as relative risk between the two groups in the DerSimonian-Laird random-effect model. All statistical analyses were done by Review Manager software (RevMan, version 5.4) for macOS, StataMP version 17 for macOS, and Open Meta[analyst] for Microsoft Windows.
Heterogeneity assessment
Statistical heterogeneity among studies was evaluated by the Chi-square test (Cochrane Q test). Then, the chi-square statistic, Cochrane Q, was used to calculate the I-squared according to the equation: I2 = \(\left(\frac{Q-df}{Q}\right)\times 100\mathrm{\%}\). A Chi-square P value less than 0.1 was considered as significant heterogeneity. I-square values ≥ 50% were indicative of high heterogeneity.
Risk of bias across studies
Two authors independently assessed the quality of included clinical trials in strict accordance with the Cochrane handbook of systematic reviews of interventions 5.1.0 (updated March 2011). We used the quality assessment table provided in (part 2, Chapter 8.5) the same book. For the observational studies, we used New Castle Ottawa Scale (NOS). Any discrepancies between the two assessors were resolved through discussion and including a third assessor. To explore the publication bias across studies, we constructed funnel plots to present the relationship between effect size and standard error. Two methods assessed evidence of publication bias; (1) Egger’s regression test and (2) the Begg and Mazumdar rank correlation test (Kendall’s tau).
Certainty assessment
We conducted a certainty assessment through sensitivity analysis (also called leave-one-out meta-analysis) to test the evidence’s robustness. For every outcome in the meta-analysis, we ran sensitivity analysis in multiple scenarios, excluding one study in each scenario to make sure the overall effect size was not dependent on any single study.
Meta-regression analysis
To test whether the study outcomes were dependent on the onset-to-groin time, we conducted meta-regression analysis models where the effect estimates and the corresponding (standard errors) were plotted against the onset-to-groin time (on the X-axis). The regression coefficient was calculated an omnibus P value of < 0.05 was considered for statistical significance. The meta-regression analysis was done by the Open Meta[Analyst] software of Oxford University’s Center of Evidence-based Medicine.
Results
Literature search results and study selection
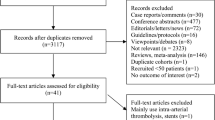
Our literature search process retrieved 2752 records. Following titles and abstract screening, 213 articles were eligible for full-text screening. From these 213 studies, 49 studies were included in the meta-analysis. Also, the references of the included studies were manually searched, and no further articles were included. The flow chart of the study selection process is shown in the PRISMA flow diagram in (Fig. 1).
Study characteristics
The population of the studies was homogenous; all studies enrolled 36,123 patients with AIS-LVO undergoing MT. Four studies were RCTs, while forty-five studies were observational studies. The characteristics of the included studies are summarized in Table 1, while summary and baseline characteristics of populations of these studies are shown in Table 2.
Risk of bias within studies
The quality of included studies ranged from moderate to high quality according to the Cochrane Risk of Bias Assessment tool for the RCTs and the Newcastle Ottawa Scale for the observational studies. The risk of bias summary is shown in Fig. 2 A and B, while the detailed risk of bias in every study is available in Figs. 1S and 2S in the supplementary file.
Mortality
The pooled RR of mortality favoured the IVT + MT group who had significantly fewer mortality events compared with the MT alone group (16.5% vs. 19.7%; RR 0.75, 95% CI [0.68 to 0.82], p < 0.0001, Fig. 3). The pooled studies were not homogenous (p = 0.02; I2 = 36%). Subgroup analysis of mortality according to the study design showed that the observational studies (0.72, 95% CI [0.65 to 0.80]) but not the RCTs (0.94, 95% CI [0.75 to 1.17]) had statistically significant pooled RR in favor of the IVT + MT group.
Complete and successful recanalization
The pooled RR of successful recanalization favoured the IVT + MT group over MT alone (77.7% vs. 75.9%; RR 1.06, 95% CI [1.03 to 1.09], p = 0.0007, Fig. 3). The pooled RR of complete recanalization favoured the IVT + MT group over MT alone (RR 1.06, 95% CI [1.01 to 1.11], p = 0.02, Fig. 3). Subgroup analysis of successful recanalization according to the study design showed that the observational studies (1.07, 95% CI [1.04 to 1.10]) but not the RCTs (1.04, 95% CI [1.00 to 1.09]) had statistically significant pooled RR in favor of the IVT + MT group.
Favorable functional outcome (or functional independence)
The pooled RR of favorable functional outcome favoured the IVT + MT group (45.2% vs. 39%; RR 1.21, 95% CI [1.13 to 1.29], p = 0.00001, Fig. 3). The pooled studies were not homogenous (p = 0.0001; I2 = 52%). Subgroup analysis of favorable functional outcome according to the study design showed that the observational studies (1.25, 95% CI [1.17 to 1.34]) but not the RCTs (0.99, 95% CI [0.89 to 1.09]) had statistically significant pooled RR in favor of the IVT + MT group.
Improvement in NIHSS score
The overall mean difference (MD) of improvement in the NIHSS score from baseline did not favor either groups (MD − 0.34, 95% CI [− 0.80 to 0.11]). Subgroup analysis, according to the time point at which NIHSS score improvement was assessed, did not show any difference between the two groups either at 24 h (MD − 0.45, 95% CI [− 1.04 to 0.13]) or at discharge (MD − 0.70, 95% CI [− 1.51 to 0.11], Fig. 4). The pooled studies were homogenous (p = 0.0005, I2 = 65%). Subgroup analysis of the improvement in NIHSS score according to the study design showed that the observational studies (MD − 0.87, 95% CI [− 1.83 to 0.09]) but not the RCTs (MD − 0.15, 95% CI [− 1.71 to 1.41]) had statistically significant pooled MD in favor of the IVT + MT group (Fig. 4).
Complications
Symptomatic intracerebral hemorrhage
The pooled RR of sICH did not favor either of groups (RR 1.00, 95% CI [0.74 to 1.36], p = 0.99, Fig. 5). The pooled studies were homogenous (p = 0.95, I2 = 0%).
Symptomatic intracranial hemorrhage
The pooled RR of symptomatic intracranial hemorrhage did not favor either of groups (RR 0.88, 95% CI [0.70 to 1.10], p = 0.27, Fig. 5). The pooled studies were homogenous (p = 0.20, I2 = 21%).
Any intracranial hemorrhage
The pooled RR of symptomatic intracranial hemorrhage did not favor either of groups (RR 1.13, 95% CI [0.97 to 1.32], p = 0.11, Fig. 5). The pooled studies were homogenous (p = 0.12, I2 = 32%).
Parenchymal hematoma
The pooled RR of parenchymal hematoma did not favor either of groups (RR 1.13, 95% CI [0.82 to 1.56], p = 0.46, Fig. 5). The pooled studies were homogenous (p = 0.13, I2 = 35%).
Procedure-related complications
The pooled RR of procedure-related complications did not favor either of groups (RR 1.13, 95% CI [0.82 to 1.55], p = 0.46, Fig. 5). The pooled studies were homogenous (p = 0.12, I2 = 45%).
Time variables
The overall mean difference (MD) of the onset-to-groin time did not favor either of groups (MD − 9.11 min, 95% CI [− 26.28 to 8.06], p = 0.30). The overall mean difference (MD) of the procedural time did not favor either of the two groups (MD − 2.40 min, 95% CI [− 8.44 to 3.64], p = 0.44; forest plots are available in the supplementary file).
Results of the subgroup analysis
We conducted subgroup analysis for the main outcomes according to the study design (RCTs only vs. observational studies vs. all studies). Observational studies showed an advantage for the IVT + MT over MT alone in terms of mortality, successful recanalization (mTICI 2b-3), and 90-day favorable outcome (0–2), but RCTs did not show the same advantage (Table 3).
Results of the meta-regression analysis
We ran multiple meta-regression analysis models to test whether the type of intervention or the onset-to-groin time could significantly influence the effect estimates. Bridging therapy was a significant predictor of less mortality (ß − 0.073; p = 0.003) and more successful recanalization (ß 0.099; p = 0.002) when compared with MT alone as the reference category in the regression model (Table 4). On the other hand, the onset-to-groin time was significantly associated with symptomatic intracranial hemorrhage (ß 0.001; p = 0.017) or any intracranial hemorrhage (ß 0.001, p = 0.026, Fig. 6).
Discussion
Summary of the main findings
The present meta-analysis provides evidence (class 1) that for patients with AIS-LVO, IVT + MT is superior to direct MT alone in terms of the rates of mortality, successful recanalization, complete recanalization, and the favorable functional outcome. Notably, our meta-analysis found that there were no significant differences between the two groups in terms of onset-to-groin time, procedural time, NIHSS scores (at 24 h and discharge), or complications including both hemorrhagic and procedure-related complications.
In terms of the improvement in NIHSS score, the subgroup analysis according to the study design showed consistent findings between RCTs and non-RCTs. However, in terms of mortality, successful recanalization (mTICI 2b-3), and 90-day favorable outcome, observational studies favoured IVT + MT over MT alone while RCTs showed no difference between the two groups (Table 3). RCTs addressing the comparisons between both treatment options are scarce; therefore, most of the evidence synthesized in this systematic review and meta-analysis is built on observational data which might be susceptible to confounding bias. Nonetheless, until further large, better-designed RCTs are conducted, the present meta-analysis provides the most comprehensive, up-to-date, evidence-based guidance on the comparison of MT + IVT versus MT alone in AIS patients. The outcomes of this meta-analysis span most of the clinically important outcomes for decision making including mortality, complete successful recanalization, functional independence on the mRS, NIHSS score, onset-to-groin time, procedural time, and procedure-related complications.
Explanation of the study findings
A few physiological mechanisms have been proposed to explain why MT + IVT might be superior to MT alone. On the one hand, it is suggested that IVT might provide synergistic effects by preparing the microvascular environment before MT. Furthermore, IVT might shorten the procedural time and reduce catheterization attempts [8]. On the other hand, IVT might facilitate the lysis of distal emboli resulting from thrombus fragmentation. Therefore, it might reduce the risk of procedural complications [15, 16].
Agreement and disagreement with previous studies
The first RCT on MT + IVT versus direct MT alone showed that direct MT was not inferior in terms of the functional outcome [15]. Additionally, the bridging therapy did not significantly reduce the numbers or the size of peripheral emboli compared with direct MT [7]. In another RCT by Broeg-Morvay et al. [7], MT + IVT did not provide additional benefits compared with MT alone in patients with no contraindications for IVT. Leker et al. [16] showed in their pilot study that patients who received IVT + MT needed fewer attempts during the EVT, while the study by Balodis et al. [8] showed that recanalization was achieved by the first pass in > 50% of cases within both groups without a significant difference.
A meta-analysis by Mistry et al. [17] examined 13 retrospective studies and found superior benefits of using bridging therapy in combination with IVT over direct MT in terms of mRS score and mortality. A summary of the results of our meta-analysis in comparison with previous meta-analyses is shown in Table 5.
Significance of the work
This study expands the literature by providing class I evidence that bridging therapy with IVT in combination with MT is superior to the direct MT without IVT for patients with AIS-LVO. A few individual studies and previous meta-analyses revealed that direct MT alone is not inferior to IVT + MT [1,2,3]. Moreover, some of these prior studies suggested that the combination of bridging IVT and MT might be associated with an increased risk of hemorrhagic complications [4]. However, the present meta-analysis showed a significant difference in favor of the bridging therapy. In this study, we analyzed data from 49 studies (4 RCTs and 45 non-RCTs) with a total of 36,123 patients, making it, to the best of our knowledge, the largest and most comprehensive meta-analysis addressing this comparison to date.
Strength points and limitations
This study has several strong points including: (1) We ran comprehensive research on multiple databases finding and including more studies than previously published meta-analyses, (2) We ran multiple sensitivity analyses to test the counterfactual, (3) We conducted all steps according to the guidelines of Cochrane Handbook of Systematic Reviews and Meta-analysis of interventions, (4) This manuscript is reported according to the PRISMA statement, and (5) For all outcomes, we stratified the effect estimates by the study design into RCTs only, non-RCT studies only, and all. This allowed us to synthesize statistically robust evidence without compromising the overall quality of the review.
Nonetheless, this study has a few limitations, including (1) some studies applied pre-selection criteria for assigning patients to the bridging therapy or the direct MT groups; such pre-selection might provide an opportunity to confounding variables to influence the study outcomes. (2) Some studies included only patients for whom IVT was not contraindicated which narrows the external validity of the results; this comparison and results are not applicable in patients for whom IVT is contraindicated since direct MT will be the first-line intervention for this subgroup of patients. (3) This meta-analysis, and also the previous meta-analyses, did not take into consideration the impact of the EVT device on such a comparison. This limitation stems from the lack of data about the used EVT technique in the included patients.
Conclusion
For patients with AIS-LVO, IVT + MT is associated with slightly better rates of survival, successful and complete recanalization, and favorable independence as compared with the direct MT alone. The cost-effectiveness of the bridging therapy combined with MT compared with the direct MT alone should be evaluated in future studies to guide decision-making in the clinical setting. Additionally, since most of the available data are from observational studies, further clinical trials are needed to provide more robust evidence on this comparison.
Abbreviations
- AIS:
-
Acute ischemic stroke
- AIS-LVO:
-
Acute ischemic stroke due to large vessel occlusion
- CI:
-
Confidence interval
- EVT:
-
Endovascular therapy
- IVT:
-
Intravenous thrombolysis
- LVO:
-
Large vessel occlusion
- MD:
-
Mean difference
- MT:
-
Mechanical thrombectomy
- mTICI:
-
Modified Thrombolysis in Cerebral Infarction grade
- NIHSS:
-
National Institute of Health Stroke Score
- RCT:
-
Randomized-controlled trials
- RR:
-
Risk Ratio
- IV:
-
Inverse variance (meta-analysis method)
- M-H:
-
Mantel-Haenszel (meta-analysis method)
References
Tsivgoulis G, Safouris A, Alexandrov AV (2015) Safety of intravenous thrombolysis for acute ischemic stroke in specific conditions. Expert Opin Drug Saf 14:845–864. https://doi.org/10.1517/14740338.2015.1032242
Seners P, Turc G, Maïer B et al (2016) Incidence and predictors of early recanalization after intravenous thrombolysis: a systematic review and meta-analysis. Stroke 47:2409–2412. https://doi.org/10.1161/STROKEAHA.116.014181
Whiteley WN, Emberson J, Lees KR et al (2016) Risk of intracerebral haemorrhage with alteplase after acute ischaemic stroke: a secondary analysis of an individual patient data meta-analysis. Lancet Neurol 15:925–933. https://doi.org/10.1016/S1474-4422(16)30076-X
Bhatia R, Hill MD, Shobha N et al (2010) Low rates of acute recanalization with intravenous recombinant tissue plasminogen activator in ischemic stroke: real-world experience and a call for action. Stroke 41:2254–2258. https://doi.org/10.1161/STROKEAHA.110.592535
Campbell BCV, Mitchell PJ, Kleinig TJ et al (2015) Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med 372:1009–1018. https://doi.org/10.1056/NEJMoa1414792
Powers WJ, Rabinstein AA, Ackerson T et al (2019) Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke. Stroke 50:e344–e418
Broeg-Morvay A, Mordasini P, Bernasconi C et al (2016) Direct mechanical intervention versus combined intravenous and mechanical intervention in large artery anterior circulation stroke: a matched-pairs analysis. Stroke 47:1037–1044. https://doi.org/10.1161/STROKEAHA.115.011134
Balodis A, Radzina M, Miglane E et al (2019) Endovascular thrombectomy in anterior circulation stroke and clinical value of bridging with intravenous thrombolysis. Acta Radiol 60:308–314. https://doi.org/10.1177/0284185118780897
Jian Y, Zhao L, Jia B et al (2021) Direct versus bridging mechanical thrombectomy in elderly patients with acute large vessel occlusion: a multicenter cohort study. Clin Interv Aging 16:1265–1274. https://doi.org/10.2147/CIA.S313171
Casetta I, Pracucci G, Saletti A et al (2019) Combined intravenous and endovascular treatment versus primary mechanical thrombectomy. The Italian Registry of Endovascular Treatment in Acute Stroke. Int J stroke Off J Int Stroke Soc 14:898–907. https://doi.org/10.1177/1747493019851279
Chalos V, LeCouffe NE, Uyttenboogaart M et al (2019) Endovascular treatment with or without prior intravenous alteplase for acute ischemic stroke. J Am Heart Assoc 8:e011592. https://doi.org/10.1161/JAHA.118.011592
Merlino G, Sponza M, Petralia B et al (2017) Short and long-term outcomes after combined intravenous thrombolysis and mechanical thrombectomy versus direct mechanical thrombectomy: a prospective single-center study. J Thromb Thrombolysis 44:203–209. https://doi.org/10.1007/s11239-017-1527-8
Minnerup J, Wersching H, Teuber A et al (2016) Outcome after thrombectomy and intravenous thrombolysis in patients with acute ischemic stroke: a prospective observational study. Stroke 47:1584–1592. https://doi.org/10.1161/STROKEAHA.116.012619
Liberati A, Altman DG, Tetzlaff J et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 339:b2700–b2700. https://doi.org/10.1136/bmj.b2700
Yang P, Zhang Y, Zhang L et al (2020) Endovascular thrombectomy with or without intravenous alteplase in acute stroke. N Engl J Med 382:1981–1993. https://doi.org/10.1056/NEJMoa2001123
Leker RR, Pikis S, Gomori JM, Cohen JE (2015) Is bridging necessary? A pilot study of bridging versus primary stentriever-based endovascular reperfusion in large anterior circulation strokes. J stroke Cerebrovasc Dis Off J Natl Stroke Assoc 24:1163–1167. https://doi.org/10.1016/j.jstrokecerebrovasdis.2015.01.008
Mistry EA, Mistry AM, Nakawah MO et al (2017) Mechanical thrombectomy outcomes with and without intravenous thrombolysis in stroke patients: a meta-analysis. Stroke 48:2450–2456. https://doi.org/10.1161/STROKEAHA.117.017320
Machado M, Alves M, Fior A et al (2021) Functional outcome after mechanical thrombectomy with or without previous thrombolysis. J stroke Cerebrovasc Dis Off J Natl Stroke Assoc 30:105495. https://doi.org/10.1016/j.jstrokecerebrovasdis.2020.105495
Maier IL, Behme D, Schnieder M et al (2017) Bridging-therapy with intravenous recombinant tissue plasminogen activator improves functional outcome in patients with endovascular treatment in acute stroke. J Neurol Sci 372:300–304. https://doi.org/10.1016/j.jns.2016.12.001
Di Maria F, Mazighi M, Kyheng M et al (2018) Intravenous thrombolysis prior to mechanical thrombectomy in acute ischemic stroke: silver bullet or useless bystander? J stroke 20:385–393. https://doi.org/10.5853/jos.2018.01543
Pienimäki J-P, Ollikainen J, Sillanpää N, Protto S (2021) In-hospital intravenous thrombolysis offers no benefit in mechanical thrombectomy in optimized tertiary stroke center setting. Cardiovasc Intervent Radiol 44:580–586. https://doi.org/10.1007/s00270-020-02727-8
Rossi R, Fitzgerald S, Molina S et al (2021) The administration of rtPA before mechanical thrombectomy in acute ischemic stroke patients is associated with a significant reduction of the retrieved clot area but it does not influence revascularization outcome. J Thromb Thrombolysis 51:545–551. https://doi.org/10.1007/s11239-020-02279-1
Sallustio F, Koch G, Alemseged F et al (2018) Effect of mechanical thrombectomy alone or in combination with intravenous thrombolysis for acute ischemic stroke. J Neurol 265:2875–2880. https://doi.org/10.1007/s00415-018-9073-7
Suzuki K, Matsumaru Y, Takeuchi M et al (2021) Effect of mechanical thrombectomy without vs with intravenous thrombolysis on functional outcome among patients with acute ischemic stroke: The SKIP Randomized Clinical Trial. JAMA 325:244–253. https://doi.org/10.1001/jama.2020.23522
Tajima Y, Hayasaka M, Ebihara K et al (2019) Effectiveness of low-dose intravenous tissue plasminogen activator before stent retriever or aspiration mechanical thrombectomy. J Vasc Interv Radiol 30:134–140. https://doi.org/10.1016/j.jvir.2018.11.005
Wang H, Zi W, Hao Y et al (2017) Direct endovascular treatment: an alternative for bridging therapy in anterior circulation large-vessel occlusion stroke. Eur J Neurol 24:935–943. https://doi.org/10.1111/ene.13311
Weber R, Nordmeyer H, Hadisurya J et al (2017) Comparison of outcome and interventional complication rate in patients with acute stroke treated with mechanical thrombectomy with and without bridging thrombolysis. J Neurointerv Surg 9:229–233. https://doi.org/10.1136/neurintsurg-2015-012236
Wee C-K, McAuliffe W, Phatouros CC et al (2017) Outcomes of endovascular thrombectomy with and without thrombolysis for acute large artery ischaemic stroke at a tertiary stroke centre. Cerebrovasc Dis Extra 7:95–102. https://doi.org/10.1159/000470855
Yi T-Y, Chen W-H, Wu Y-M et al (2018) Adjuvant intra-arterial rt-PA injection at the initially deployed solitaire stent enhances the efficacy of mechanical thrombectomy in acute ischemic stroke. J Neurol Sci 386:69–73. https://doi.org/10.1016/j.jns.2018.01.012
Zi W, Qiu Z, Li F et al (2021) Effect of endovascular treatment alone vs intravenous alteplase plus endovascular treatment on functional independence in patients with acute ischemic stroke: the DEVT randomized clinical trial. JAMA 325:234–243. https://doi.org/10.1001/jama.2020.23523
Al-Khaled M, Brüning T, Gottwald C et al (2018) Comparing outcome and recanalization results in patients with anterior circulation stroke following endovascular treatment with and without a treatment with rt-PA: A single-center study. Brain Behav 8:e00974. https://doi.org/10.1002/brb3.974
Behme D, Kabbasch C, Kowoll A et al (2016) Intravenous thrombolysis facilitates successful recanalization with stent-retriever mechanical thrombectomy in middle cerebral artery occlusions. J stroke Cerebrovasc Dis Off J Natl Stroke Assoc 25:954–959. https://doi.org/10.1016/j.jstrokecerebrovasdis.2016.01.007
Broocks G, Meyer L, Kabiri R et al (2021) Impact of intravenous alteplase on sub-angiographic emboli in high-resolution diffusion-weighted imaging following successful thrombectomy. Eur Radiol 31:8228–8235. https://doi.org/10.1007/s00330-021-07980-0
Charbonnier G, Bonnet L, Bouamra B et al (2020) Does intravenous thrombolysis influence the time of recanalization and success of mechanical thrombectomy during the acute phase of cerebral infarction? Cerebrovasc Dis Extra 10:28–35. https://doi.org/10.1159/000507119
Choi JH, Im SH, Lee KJ et al (2018) Comparison of outcomes after mechanical thrombectomy alone or combined with intravenous thrombolysis and mechanical thrombectomy for patients with acute ischemic stroke due to large vessel occlusion. World Neurosurg 114:e165–e172. https://doi.org/10.1016/j.wneu.2018.02.126
Gamba M, Gilberti N, Premi E et al (2019) Intravenous fibrinolysis plus endovascular thrombectomy versus direct endovascular thrombectomy for anterior circulation acute ischemic stroke: clinical and infarct volume results. BMC Neurol 19:103. https://doi.org/10.1186/s12883-019-1341-3
Guedin P, Larcher A, Decroix J-P et al (2015) Prior IV thrombolysis facilitates mechanical thrombectomy in acute ischemic stroke. J stroke Cerebrovasc Dis Off J Natl Stroke Assoc 24:952–957. https://doi.org/10.1016/j.jstrokecerebrovasdis.2014.12.015
Guimarães Rocha M, Carvalho A, Rodrigues M et al (2019) Primary thrombectomy versus combined mechanical thrombectomy and intravenous thrombolysis in large vessel occlusion acute ischemic Stroke. J stroke Cerebrovasc Dis Off J Natl Stroke Assoc 28:627–631. https://doi.org/10.1016/j.jstrokecerebrovasdis.2018.11.002
Heiferman DM, Li DD, Pecoraro NC et al (2017) Intra-arterial alteplase thrombolysis during mechanical thrombectomy for acute ischemic stroke. J stroke Cerebrovasc Dis Off J Natl Stroke Assoc 26:3004–3008. https://doi.org/10.1016/j.jstrokecerebrovasdis.2017.07.031
Imbarrato G, Bentley J, Gordhan A (2018) Clinical outcomes of endovascular thrombectomy in tissue plasminogen activator versus non-tissue plasminogen activator patients at primary stroke care centers. J Neurosci Rural Pract 9:240–244. https://doi.org/10.4103/jnrp.jnrp_497_17
Kaesmacher J, Kleine JF (2018) Bridging therapy with i. v. rtPA in MCA occlusion prior to endovascular thrombectomy: a Double-Edged Sword? Clin Neuroradiol 28:81–89. https://doi.org/10.1007/s00062-016-0533-0
Abilleira S, Ribera A, Cardona P et al (2017) Outcomes after direct thrombectomy or combined intravenous and endovascular treatment are not different. Stroke 48:375–378. https://doi.org/10.1161/STROKEAHA.116.015857
Bhatia R, Shobha N, Menon BK et al (2014) Combined full-dose IV and endovascular thrombolysis in acute ischaemic stroke. Int J stroke Off J Int Stroke Soc 9:974–979. https://doi.org/10.1111/j.1747-4949.2012.00890.x
Dávalos A, Pereira VM, Chapot R et al (2012) Retrospective multicenter study of Solitaire FR for revascularization in the treatment of acute ischemic stroke. Stroke 43:2699–2705. https://doi.org/10.1161/STROKEAHA.112.663328
Kass-Hout T, Kass-Hout O, Mokin M et al (2014) Is bridging with intravenous thrombolysis of any benefit in endovascular therapy for acute ischemic stroke? World Neurosurg 82:e453–e458. https://doi.org/10.1016/j.wneu.2013.01.097
Pfefferkorn T, Holtmannspötter M, Patzig M et al (2012) Preceding intravenous thrombolysis facilitates endovascular mechanical recanalization in large intracranial artery occlusion. Int J stroke Off J Int Stroke Soc 7:14–18. https://doi.org/10.1111/j.1747-4949.2011.00639.x
Rai AT, Boo S, Buseman C et al (2018) Intravenous thrombolysis before endovascular therapy for large vessel strokes can lead to significantly higher hospital costs without improving outcomes. J Neurointerv Surg 10:17–21. https://doi.org/10.1136/neurintsurg-2016-012830
Gong L, Zheng X, Feng L et al (2019) Bridging therapy versus direct mechanical thrombectomy in patients with acute ischemic stroke due to middle cerebral artery occlusion: a clinical-histological analysis of retrieved thrombi. Cell Transplant 28:684–690. https://doi.org/10.1177/0963689718823206
Alonso de Leciñana M, Martínez-Sánchez P, García-Pastor A et al (2017) Mechanical thrombectomy in patients with medical contraindications for intravenous thrombolysis: a prospective observational study. J Neurointerv Surg 9:1041–1046. https://doi.org/10.1136/neurintsurg-2016-012727
Bourcier R, Alexandre P-L, Eugène F et al (2018) Is bridging therapy still required in stroke due to carotid artery terminus occlusions? J Neurointerv Surg 10:625–628. https://doi.org/10.1136/neurintsurg-2017-013398
Ferrigno M, Bricout N, Leys D et al (2018) Intravenous recombinant tissue-type plasminogen activator: influence on outcome in anterior circulation ischemic stroke treated by mechanical thrombectomy. Stroke 49:1377–1385. https://doi.org/10.1161/STROKEAHA.118.020490
Maingard J, Shvarts Y, Motyer R et al (2019) Outcomes of endovascular thrombectomy with and without bridging thrombolysis for acute large vessel occlusion ischaemic stroke. Intern Med J 49:345–351. https://doi.org/10.1111/imj.14069
Park H-K, Chung J-W, Hong J-H et al (2017) Preceding intravenous thrombolysis in patients receiving endovascular therapy. Cerebrovasc Dis 44:51–58. https://doi.org/10.1159/000471492
LeCouffe NE, Kappelhof M, Treurniet KM et al (2021) A randomized trial of intravenous alteplase before endovascular treatment for stroke. N Engl J Med 385:1833–1844. https://doi.org/10.1056/NEJMoa2107727
Goyal N, Tsivgoulis G, Frei D et al (2018) Comparative safety and efficacy of combined IVT and MT with direct MT in large vessel occlusion. Neurology 90:e1274–e1282. https://doi.org/10.1212/WNL.0000000000005299
Tong X, Wang Y, Fiehler J et al (2021) Thrombectomy versus combined thrombolysis and thrombectomy in patients with acute stroke: a matched-control study. Stroke 52:1589–1600. https://doi.org/10.1161/STROKEAHA.120.031599
Kandregula S, Savardekar AR, Sharma P et al (2021) Direct thrombectomy versus bridging thrombolysis with mechanical thrombectomy in middle cerebral artery stroke: a real-world analysis through National Inpatient Sample data. Neurosurg Focus 51:E4. https://doi.org/10.3171/2021.4.FOCUS21132
Podlasek A, Dhillon PS, Butt W et al (2021) Direct mechanical thrombectomy without intravenous thrombolysis versus bridging therapy for acute ischemic stroke: A meta-analysis of randomized controlled trials. Int J stroke Off J Int Stroke Soc 16:621–631. https://doi.org/10.1177/17474930211021353
Fan L, Zang L, Liu X et al (2021) Outcomes of mechanical thrombectomy with pre-intravenous thrombolysis: a systematic review and meta-analysis. J Neurol 268:2420–2428. https://doi.org/10.1007/s00415-020-09778-4
Vidale S, Romoli M, Consoli D, Agostoni EC (2020) Bridging versus direct mechanical thrombectomy in acute ischemic stroke: a subgroup pooled meta-analysis for time of intervention, eligibility, and study design. Cerebrovasc Dis 49:223–232. https://doi.org/10.1159/000507844
Katsanos AH, Malhotra K, Goyal N et al (2019) Intravenous thrombolysis prior to mechanical thrombectomy in large vessel occlusions. Ann Neurol 86:395–406. https://doi.org/10.1002/ana.25544
Phan K, Dmytriw AA, Maingard J et al (2017) Endovascular thrombectomy alone versus combined with intravenous thrombolysis. World Neurosurg 108:850-858.e2. https://doi.org/10.1016/j.wneu.2017.08.040
Tsivgoulis G, Katsanos AH, Mavridis D et al (2016) Mechanical Thrombectomy improves functional outcomes independent of pretreatment with intravenous thrombolysis. Stroke 47:1661–1664. https://doi.org/10.1161/STROKEAHA.116.013097
Acknowledgements
None.
Funding
None. This study is not funded by a third party.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Hazem S. Ghaith and Mohamed Elfil have equal contributions as first authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ghaith, H.S., Elfil, M., Gabra, M.D. et al. Intravenous thrombolysis before mechanical thrombectomy for acute ischemic stroke due to large vessel occlusion; should we cross that bridge? A systematic review and meta-analysis of 36,123 patients. Neurol Sci 43, 6243–6269 (2022). https://doi.org/10.1007/s10072-022-06283-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-022-06283-6