Abstract
Viral diseases are the cause of many global epidemics, leading to deaths, affecting the quality of life of populations, and impairing public health. The limitations in the treatment of viral diseases and the constant resistance to conventional antiviral treatments encourage researchers to discover new compounds. In this perspective, this literature review presents isolated molecules and extracts of natural products capable of inhibiting the activity of the nonstructural protein that acts as the RNA-dependent RNA polymerase. The literature review presented natural compounds with the potential to be tested as alternative medicines or used in the development of synthetic drugs to prevent the replication of RNA viruses, such as COVID-19, hepatitis C, and dengue viruses, among others. Natural products are known to exhibit remarkable activities in mitigation of different viral diseases, in addition, they help to decrease the aggravation of infections. Consequently, reducing hospitalization time and deaths.
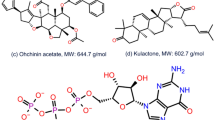
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The RNA-dependent RNA polymerase (RdRp) is described in the literature as a promising target for the development of antiviral drugs [1]. The RdRp enzyme is part of the viral replication-transcription complex, and a specific inhibitor of this enzyme could prevent the complex from functioning properly, consequently interrupting the protein synthesis and virus multiplication. Among the positive aspects of RdRp is the fact that it has no homology with host cell proteins. Therefore, inhibitors developed for this protein may be more selective and, in turn, have few side effects and no toxicity in human cells [1, 2]. The SARS-CoV-2 virus genome has a positive-sense single-stranded RNA, which contains the orf1ab gene that encodes nonstructural proteins (NSPs) (replication-transcription complex) as well as genes that encode structural proteins. The NSPs 1 to 16 are encoded by the orf1ab gene, and nsp12 is one of the enzymes responsible for the duplication of the genome that acts as the RNA-dependent RNA polymerase (RdRp) [3]. HCV, from the Flaviviridae family, also has a positive-sense single-stranded RNA responsible for encoding a polyprotein. This polyprotein is sequentially cleaved by host and virus proteases into mature proteins such as central, structural, ion channel, and nonstructural proteins (NSPs), named p7, NS2, NS3, NS4A, NS4B, NS5A, and NS5B [4, 5]. The NS5B enzyme is the most studied target for the development of HCV therapies [6]. The drug therapy commonly includes pegylated interferon (PEG-IFN) combined with the nucleoside analog ribavirin and direct-acting antiviral (DAA) therapy [7]. DAAs are drugs that target nonstructural proteins, especially the NS5B polymerase. DENV, from the Flaviviridae family, has four serotypes responsible for the same symptoms [8]. This virus is composed of a positive-sense single-stranded RNA molecule with an open reading frame. This structure is surrounded by untranslated regions that play structural and functional roles in the formation of RNAs (genome replication and transcription) [9]. NS5, which acts as the RdRp enzyme, is responsible for the replication of DENV and is common to its four serotypes. This makes this protein the target of choice for the development of drugs against DENV [8].
Therefore, the objective of this study was to review the literature on isolated substances and/or extracts of natural compounds that function as viral RdRp inhibitors since these compounds may have promising antiviral activities and serve as a basis for future research, such as in the development of drugs. Compounds that were reported to have this potential but did not present a half-maximal inhibitory concentration capable of inhibiting 50% of the enzyme (IC50) were excluded from this review, as well as potential inhibitors selected only with in silico data. For this purpose, several online databases like Medline, Scopus, Web of Science, Embase, Lilacs and Scielo were used with the following terms in different combinations as medicinal plant, plant extract, bioactive, phytochemical, bioactive, natural product, antiviral agents, RNA polymerase, RNA replicase inhibitor.
Viral diseases
Viruses are threats to public health because they can affect the physical, social, economic, and mental health of populations worldwide. The phenomenon of zoonotic spillover, climate change, and the increase in international travel are some of the factors that are directly related to the increase in the spread of viral diseases [10]. Since the first outbreak of the Severe Acute Respiratory Syndrome (SARS-CoV) in late 2002, and of the Middle East Respiratory Syndrome (MERS-CoV) in 2012, the need to develop effective therapies and vaccines has become evident. This context was intensified in December 2019, when the first cases of SARS-CoV-2 contamination were reported in China; over the next few months, the virus quickly spread to several countries and became a public health emergency [11]. The novel coronavirus (SARS-CoV-2) pandemic was declared by the World Health Organization (WHO) on March 11, 2020, and as of August 23, 2022, more than 596 million cases and 6.455.500 deaths from COVID-19 had been recorded worldwide [12]. Although several vaccines have been developed, there is currently no therapy available to treat COVID-19. Thus, an antiviral drug capable of inhibiting SARS-CoV-2 replication is needed, since the disease is now responsible for millions of deaths and is still affecting the routine and economy of the world population.
The hepatitis C virus (HCV) is also included in the statistics of the viruses that most affect global health, with the aggravating factor that the infection develops gradually, originating diseases such as liver fibrosis, cirrhosis, and hepatocellular carcinoma [5]. Anti-HCV antibodies are produced during the acute phase of infection; however, HCV can evade them, rendering the host immunity system ineffective. These immune events have not been fully elucidated [7]. This condition, alongside treatment interruption, contributes to approximately 71 million people presenting chronic hepatitis C worldwide [7, 13]. To date, there are no HCV vaccines [14].
Dengue (DENV) is the most prominent mosquito-borne viral disease and falls into the category of public health problems in tropical and subtropical countries. Dengue has existed for several decades; however, its occurrence increased 30-fold in the last 50 years. To prevent cases, it is necessary to fight the proliferation of the dengue mosquito, since there is no available chemotherapy so far [8] and the tetravalent dengue vaccine (Dengvaxia®) [15] is restricted to people who have already had contact with the virus [13]. The rapid antiviral drug resistance and the lack of preventive vaccines motivate the search for new substances; therefore, there is an increasing number of studies on natural compounds [4, 16, 17].
Molecular structures of isolated compounds with the potential to inhibit RdRp-HCV and RdRp-DENV
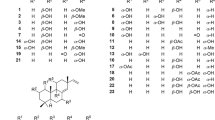
The substances in a plant extract promote a synergistic effect, which can contribute to the inhibition of multi-site enzymes. RdRp-HCV (NS5B), for instance, has five different small-molecule binding sites, which shows that there is great benefit in using a natural product extract, whether combined with other drugs or not [18]. In addition to synergism from several compounds against the same target, the antiviral action can result from the synergism of different molecules that act on different targets but with the same purpose. An extract can have several molecules that present different mechanisms of action, including blocking the entry of the virus into the cell, blocking enzyme synthesis, and inhibiting viral replication [19, 20]. However, it is necessary to isolate the compounds that constitute extracts, even if the purified form has no synergistic effect. Fractionation and purification allow us to characterize an active molecule and evaluate its activity and/or potential in different experimental models [18]. In Fig. 1 we present the molecular structures and names of the isolated compounds mentioned in the studies that were included in this review. Further on, some inherent characteristics of isolated compounds will be discussed. These features make it easier to understand the nature of the interactions between isolated compounds and the viral polymerase (RdRp), in addition to trying to indicate promising compounds that can be explored in the future.
Compound 26 (IC50: 7.4) was isolated from the extract of Tripterygium hypoglaucum. In a study that compared the inhibitory activity of compound 26 with that of other compounds isolated from the same species on the hepatitis C virus RNA-dependent RNA polymerase (RdRp-HCV), the researchers correlated the presence of structural elements of compound 26 with the inhibitory activity; the structural elements include the presence of a carboxylic acid in the E-ring and the aldehyde and hydroxyl groups in the A-ring (Fig. 1) [6]. Based on the results from an assay on the polymerase inhibitory activity of mono- and dialkylated flavanones isolated from the bark extract of Cryptocarya chartacea, [21] observed both more and less active components and correlated the presence of arylheptanoid side chains at C-6 and C-8 in compounds 21 and 18 with the inhibitory activity [21]. Important data on the structures of the carneic acids (compounds 15, 16, 14, 12, 13) were listed in a polymerase bioassay, in which the authors suggested some relevant features for the inhibitory activity on dengue virus RNA-dependent RNA polymerase (RdRp-DENV). Among these features are the presence of the 1-methypropenyl unit in position C-15 (Fig. 1) and the lower inhibitory potential of the compounds when this subunit is oxidized at C-18. They further listed the β-OH group at C-9 in compound 15 as likely responsible for the activity of this compound on the polymerase [22]. The literature indicates that some molecules selected from the Dacrydium balansae species did not show the same potential as isolated biflavonoids (compounds 28, 36, 7, 29), thus suggesting, through the comparison of chemical structures, the importance of the dimeric nature of biflavonoids (Fig. 1). Furthermore, the comparison between isolated biflavonoids led the authors to hypothesize that 4′-methoxylation decreases the potency of some of biflavonoids [8].
Compound 40 strongly inhibited RdRp (NS5B) activity (Table 1). [18] found that, in a cell culture system, 10 μM of this compound was able to inhibit more than 80% of NS5B expression [18]. In the same study, the authors also found that compounds 40 and 30 (Fig. 1) showed structural similarities and synergistic effect when inhibiting the polymerase activity [18]. Aside from being present in Eclipta Alba, compound 40 is also the main metabolite of the Eclipta prostrata species [23]. The literature reports that this compound can inhibit the secretion of interleukins and suppress the transcription of inflammatory genes [24]. The following biological effects were also cited: antifibrotic, anti-free radical, and antioxidant [23].
Compound 7, isolated from the Dacrydium balansae species, inhibits the inflammasome, which is an adaptor protein involved in inflammatory signaling. This protein complex detects pathogenic microorganisms, causing the release of inflammatory cytokines such as interleukin-1β and interleukin-18 [25]. The T. hypoglaucum species, in turn, is described to have traditional use in the treatment of several diseases, including inflammatory processes [26]. In 2018, researchers were able to isolate a diterpenoid (19-O-β-D-glucopyranosyl-labda-8), nine triterpenes, and compound 26 from this species [26] All these isolated metabolites showed anti-inflammatory and immunosuppressive activities. These data are also relevant to COVID-19, since the rapid stimulation of the immune system response triggers the activation of specific receptors that cause the release of many pro-inflammatory cytokines [27]. Interestingly, Brazilian researchers investigated possible mechanisms responsible for the SARS-CoV-2 invasion of the central nervous system, suggesting that the hyperactivation of the P2X7R receptor is the cause of the “cytokine storm”. This condition is an aggravating factor of the SARS-CoV-2 infection since the patient may develop psychiatric disorders, such as depression, and neurodegenerative diseases [28]. These data reinforce the urgent need for drugs capable of modulating the main substances (cytokines and chemokines) of the inflammatory process associated with COVID-19 before high concentrations of these substances cross the blood-brain barrier. Compound 30 and flavonoids such as fisetin, kaempferol, myricetin, astragalin, and rutin also inhibit the expression and synthesis of these cytokines [27]. According to the literature, curcumin is a natural compound responsible for altering structural proteins and stopping SARS-CoV-2 from entering the cell; it can also act in the mediating process of inflammatory cytokines [29]. Therefore, the aforementioned compounds could contribute to the development of antiviral therapies for RNA virus infections not only by inhibiting viral replication but also minimize secondary conditions related to the pathophysiology of viral infections. In previous studies, compounds 31 and 38 (Table 1) were observed to inhibit HCV RdRp. The authors suggested that either one of them may have multiple binding sites on NS5B and reported that these chemical compounds do not act as competitive inhibitors. All these data are extremely important because they indicate compounds with high inhibitory capacity that directly interfere with replication processes and may be good low-toxicity alternatives [30]. In the literature, several natural compounds have also been mentioned, capable of interfering with HCV replication and/or blocking the entry of HCV by binding to structural and nonstructural proteins [4].
In 2016, a phytochemical analysis of the Diospyros glans species disclosed the presence of compound 39, lupane- and ursane-type triterpenoids (Table 1) [31]. This finding marked the beginning of the discovery of an array of compounds capable of inhibiting the dengue virus replication. Later, in another study, compounds 5, 27, 4, 9, 10, and11 (known lupane-type triterpenoids) (Table 1) were isolated from the crude extract of Dyospyros carbonaria bark. The results were promising for both the crude extract of D. carbonaria and the 6 isolated compounds. Therefore, the researchers also analyzed the 38 endophytic fungi that were colonizing this species and, for the first time, compound 10 was isolated from an endophyte (Phomopsis sp. SNB-LAP1-7-32) [15]. The preliminary analysis of the endophyte Phomopsis sp. (SNB-LAP1-7-32) prompted further investigation of the extract and, this time, a total of 13 new carneic acids were obtained, among which, compounds 15, 16, 14, 18, and 13 showed significant activity (Table 1) [22]. Data from the literature show that several natural compounds such as curcumin, baicalein, delphinidin, naringenin, quercetin, and quinine were already related to DENV inhibitory activity, but few showed the potential to inhibit RdRp according to the IC50 value [32].
This review lists isolated compounds reported in the literature that showed the potential to inhibit the RdRp enzyme. Further details about these inhibitors are presented in Table 1.
The isolated compounds that showed lower concentrations when inhibiting the RdRp enzyme were grouped in Fig. 2.
Isolated compounds and groups of plant metabolites
Classes of secondary metabolites such as flavonoids, coumarins, terpenoids, essential oils, alkaloids, polysaccharides, and proteins are currently under testing for a wide range of antiviral activities [35].
A study reports that several natural compounds of the flavonoid class showed antiviral activities not only against SARS-CoV-2, but also against other RNA virusesFootnote 1 such as influenza A and B, human immunodeficiency 1 (HIV-1), and hepatitis C viruses [27]. Most of the isolated compounds included in this review with IC50 values below 5 μM belonged to the flavonoid and triterpenoid classes (Fig. 3), such as compounds 19, 21, 20, 18 (designated mono- and dialkylated flavanones), which showed an IC50 between 1.8 and 4.2 μM (Table 1) [21]. The biflavonoids isolated from D. balansae exhibited specific inhibitory activity against RdRp-DENV [8]. Compounds 29, 36, and 7 were isolated from the D. balansae species and, according to the literature, compounds 36 and 7 were also found in Garcinia subelliptica, described as an important source of polyphenols [36]. Compound 29, which is also present in Ginkgo biloba leaves, has shown anti-inflammatory effects [37] and the ability to change pre-mRNA splicing in vitro and in cellulo, biological effects that were also reported for compound 1 [38].
Triterpenoids comprise the largest class of secondary metabolites, with over 20,000 identified structures found in various medicinal plants. This is directly related to the fact that these secondary metabolites have a wide range of pharmacological activities, including antiviral effects [39]. Compound 31 is a known example of a triterpenoid used in several countries to treat liver-related diseases and diabetes, and also as a wound-healing agent [39]. Studies report that plants such as Lantana camara and Lisgustrum lucidum, like others from the Oleaceae family, are rich sources of compound 31and are used as alternative therapies. The traditional indication for this natural compound also includes alternative treatment for various chronic diseases [37]. Thus, this compound may be a valuable alternative to prevent viral infection, since it showed a significant IC50, and to ameliorate the complications or onset of chronic diseases associated with the initial viral infection. Another triterpenoid cited in this review is compound 10, which is isolated from birch (Betula sp.) and also found in many other species. This compound has a high potential to inhibit RdRp activity and is known mainly for its antiviral and antitumor effects [40]. Figure 3 shows the isolated compounds with IC50 values below 5.0 µM and the group of metabolites to which they belong.
Extracts and fractions with the potential to inhibit RdRp
Research shows that several plant species have anti-HCV potential, such as methanolic extracts from Ajuba parviflora, Ajuba bracteosa, Barberis lycium, and Citrus lemon [31]. One of the species included in this review was E. Alba, a plant used in traditional African medicine whose extract inhibited the enzymatic activity of RdRp in vitro (Table 2). From its extracts, researchers isolated phenolic compounds, alkaloids, and terpenoids [41]. Using the Western Blotting and RT-PCR techniques, researchers confirmed a decrease in NS5B and NS5A protein expression. They verified over 95% inhibition when the cells were incubated with the extract at a concentration of 130 μg/mL for 48 h [18].
Phyllanthus amarus is described in the literature as a valuable natural source and is used in different countries around the world; in South America, it is known as “stone breaker” or “shatter stone”. This plant has several pharmacological activities, including antiviral properties, and is used in traditional medicine for hepatitis A and B [42]. Looking at the traditional use of this plant, the literature has explored and found evidence of the anti-HCV effect of P. amarus, according to the IC50 values (between 5-10 µg/mL) of its extracts and fractions [20]. The same study compared extracts from different parts of P. amarus and found that the leaf extract (commonly used in homemade preparations) had the lowest concentration, capable of inhibiting only 50% of the target in question.
Regarding species popularly used in Yunnan, China, Polygonum cymosum showed a promising result, being able to inhibit 50% of the RdRp-mediated RNA synthesis at a concentration of 5.2 µg/mL (IC50) for the aqueous fraction (Table 2). Favorable results were also reported for the Ceratostigma willmottianum(methanolic extract), Dioscorea cirrhosa(methanolic extract), and Daucus maritimus (buthanolic fraction) extracts, which showed IC50 at concentrations of 3.1, 0.6, and 1.0 µg/mL, respectively (Table 2) [6]. The butanolic fraction of D. maritimus was most effective in inhibiting the RdRp of West Nile virus (WNV), but it was also effective in inhibiting the HCV protein (Table 2). In assays with the aqueous extract of D. cirrhosa (IC50 = 0.6 µg/mL), researchers found that this extract was able to increase cell viability (myocardial H9c2 cells) and decrease the content of reactive oxygen species when exposed to hydrogen peroxide. This result demonstrated the potential of this extract to protect the cell during oxidative stress [43], which is high during long-term viral infections. Restoring the balance depends on endogenous (albumin, urea, reduced glutathione) and exogenous (polyphenols, carotenoids, etc.) antioxidant compounds. Natural compounds with antioxidant properties may help reduce endothelial damage caused by oxidative stress. These compounds with antioxidant activity may be used to treat other diseases, such as COVID-19, since they act in the modulation of the immune system, a key spot to decrease complications caused by viral infections [44].
The Diospyros genus encompasses some species, out of a total of 350, that are known to have antiviral properties against the human immunodeficiency virus, human norovirus, H3N2, H5N3 influenza virus, and herpes simplex-1 virus. Several studies were conducted with different species of this genus. A phylogenetic analysis from a dendrogram curiously showed that extracts from different species but from the same geographical area had phytochemical similarities [31]. Research on the D. carbonaria species in 2016 and 2020 has resulted in several promising studies. Extracts from both D. carbonaria bark (44% inhibition at 5 μg/mL) [15] and Phomopsis sp. culture (60% inhibition at 10 µg/mL) [22] were able to inhibit the replication of the dengue virus. Several studies on medicinal plant extracts with antiviral potential against DENV appeared in the literature, but few of them focused specifically on the RdRp enzyme. The acetone extracts of Pavetta tomentosa and Asian Tarenna demonstrated pupicidal potency against Aedes aegypti, and P. tomentosa also had an effect against dengue viral cell line [45].
This review lists natural extracts, including one from the fungal endophyte Phomopsis sp. SNB-LAP1-7-32, which showed the potential to inhibit the RdRp enzyme as per the determined IC50 values. The extracts in a concentration up to 20 µg/mL are presented in Table 2; Table 3 lists the remaining extracts (>20 µg/mL).
The species that showed the lowest concentrations of the extract when inhibiting the RdRp enzyme are presented in Fig. 4.
Conclusions
The lack of specific pharmacological agents is a limitation in the treatment of viral diseases. This review compiles natural compounds that can be tested as alternative therapies or used as prototypes to develop synthetic drugs that prevent viral replication. The data shows the potential these compounds from natural products have against several diseases, either by directly acting to inhibit the RdRp enzyme or by reducing secondary conditions related to the pathophysiology of viral infections. By gathering these data, we intend for this study to contribute with new insights about the structure-activity relationship of these molecules.
Notes
In fact, the inhibition of replication and transcription processes is a mechanism of action common to molecules capable of interfering with the activity of RdRp, present in RNA viruses. Flavonoids such as kaempterol, fisetin, hyperoside, afzelin, biorobin, myricitrin, astragalin, quercetin, quercetin-3-O-glycoside and quercitrin showed high affinity for RdRp using molecular docking.
Abbreviations
- COVID-19 :
-
Coronavirus disease 2019
- DAA :
-
Direct-acting antiviral
- DENV :
-
Dengue
- HCV :
-
Hepatitis C virus
- HIV-1 :
-
Human immunodeficiency 1
- MERS-CoV :
-
Middle East Respiratory Syndrome
- NSPs :
-
Nonstructural proteins
- PEG-IFN :
-
Pegylated interferon
- RdRp :
-
RNA-dependent RNA polymerase
- RdRp-DENV :
-
dengue virus RNA-dependent RNA polymerase
- RdRp-HCV :
-
hepatitis C virus RNA-dependent RNA polymerase
- SARS-CoV :
-
Severe Acute Respiratory Syndrome
- WNV :
-
West Nile virus
References
Christy MP, Uekusa Y, Gerwick L, Gerwick WH. Natural products with potential to treat RNA virus pathogens including SARS-CoV-2. J Nat Prod. 2021;84:161–82. https://doi.org/10.1021/acs.jnatprod.0c00968.
Zhu W, Chen CZ, Gorshkov K, Xu M, Lo DC, Zheng W. RNA-dependent RNA polymerase as a target for COVID-19 drug discovery. SLAS Discov. 2020;25:1141–51. https://doi.org/10.1177/2472555220942123.
Narayanan N, Nair DT. Ritonavir may inhibit exoribonuclease activity of nsp14 from the SARS-CoV-2 virus and potentiate the activity of chain terminating drugs. Int J Biol Macromol. 2021;168:272–8. https://doi.org/10.1016/j.ijbiomac.2020.12.038.
El-Tantawy WH, Temraz A. Natural products for the management of the hepatitis C virus: a biochemical review. Arch Physiol Biochem. 2020;126:116–28. https://doi.org/10.1080/13813455.2018.1498902.
Cosset FL, Mialon C, Boson B, Granier C, Denolly S. HCV interplay with lipoproteins: Inside or outside the cells. Viruses. 2020;12:434. https://doi.org/10.3390/v12040434.
Jo M, Nakamura N, Kakiuchi N, Komatsu K, Qui MH, Shimotohno K, et al. Inhibitory effect of Yunnan traditional medicines on hepatitis C viral polymerase. J Nat Med. 2006;60:217–24. https://doi.org/10.1007/s11418-006-0041-7.
Casey JL, Feld JJ, MacParland SA. Restoration of HCV-specific immune responses with antiviral therapy: a case for DAA treatment in acute HCV infection. Cells. 2019;8:317. https://doi.org/10.3390/cells8040317.
Coulerie P, Eydoux C, Hnawia E, Stuhl L, MacIuk A, Lebouvier N, et al. Biflavonoids of Dacrydium balansae with potent inhibitory activity on dengue 2 NS5 polymerase. Planta Med. 2012;78:672–7. https://doi.org/10.1055/s-0031-1298355.
Cannalire R, Ki Chan KW, Burali MS, Gwee CP, Wang S, Astolfi A, et al. Pyridobenzothiazolones exert potent anti-dengue activity by hampering multiple functions of NS5 polymerase. ACS Med Chem Lett. 2020;11:773–82. https://doi.org/10.1021/acsmedchemlett.9b00619.
Artika IM, Wiyatno A, Ma’roef CN. Pathogenic viruses: molecular detection and characterization. Infect Genet Evol. 2020;81:104215. https://doi.org/10.1016/j.meegid.2020.104215.
Chen Y, Liu Q, Guo D. Emerging coronaviruses: genome structure, replication, and pathogenesis. J Med Virol. 2020;92:418–23. https://doi.org/10.1002/jmv.25681.
COVID-19 Map—Johns Hopkins Coronavirus Resource Center. 2021. Im Internet. https://coronavirus.jhu.edu/map.html.
WHO | World Health Organization. 2021. Im Internet: https://www.who.int/.
Sedeño-Monge V, Laguna-Meraz S, Santos-López G, Panduro A, Sosa-Jurado F, Jose-Abrego A, et al. A comprehensive update of the status of hepatitis C virus (HCV) infection in Mexico-a systematic review and meta-analysis (2008–2019). Ann Hepatol. 2021;20:100292. https://doi.org/10.1016/j.aohep.2020.100292.
Peyrat LA, Eparvier V, Eydoux C, Guillemot JC, Litaudon M, Stien D. Betulinic acid, the first Lupane-type triterpenoid isolated from both a Phomopsis sp. and Its host plant diospyros carbonaria Benoist. Chem Biodivers. 2017;14:e1600171. https://doi.org/10.1002/cbdv.201600171.
Tseliou M, Pirintsos SA, Lionis C, Castanas E, Sourvinos G. Antiviral effect of an essential oil combination derived from three aromatic plants (Coridothymus capitatus (L.) Rchb. f., Origanum dictamnus L. and Salvia fruticosa Mill.) against viruses causing infections of the upper respiratory tract. J Herb Med. 2019;17–18:100288. https://doi.org/10.1016/j.hermed.2019.100288.
Tahir ul Qamar M, Alqahtani SM, Alamri MA, Chen LL. Structural basis of SARS-CoV-2 3CLpro and anti-COVID-19 drug discovery from medicinal plants. J Pharm Anal. 2020;10:313–9. https://doi.org/10.1016/j.jpha.2020.03.009.
Manvar D, Mishra M, Kumar S, Pandey VN. Identification and evaluation of anti Hepatitis C virus phytochemicals from Eclipta alba. J Ethnopharmacol. 2012;144:545–54. https://doi.org/10.1016/j.jep.2012.09.036.
Ramalingam S, Karupannan S, Padmanaban P, Vijayan S, Sheriff K, Palani G, et al. Anti-dengue activity of Andrographis paniculata extracts and quantification of dengue viral inhibition by SYBR green reverse transcription polymerase chain reaction. AYU. 2018;39:87. https://doi.org/10.4103/ayu.ayu_144_17.
Ravikumar YS, Ray U, Nandhitha M, Perween A, Raja Naika H, Khanna N, et al. Inhibition of hepatitis C virus replication by herbal extract: Phyllanthus amarus as potent natural source. Virus Res. 2011;158:89–97. https://doi.org/10.1016/j.virusres.2011.03.014.
Allard PM, Dau ETH, Eydoux C, Guillemot JC, Dumontet V, Poullain C, et al. Alkylated flavanones from the bark of cryptocarya chartacea as dengue virus NS5 polymerase inhibitors. J Nat Prod. 2011;74:2446–53. https://doi.org/10.1021/np200715v.
Peyrat LA, Eparvier V, Eydoux C, Guillemot JC, Litaudon M, Stien D. Carneic Acids from an Endophytic Phomopsis sp. as Dengue Virus Polymerase Inhibitors. J Nat Prod. 2020;83:2330–6. https://doi.org/10.1021/acs.jnatprod.9b01169.
Zhu MM, Wang L, Yang D, Li C, Pang S-T, Li X-H, et al. Wedelolactone alleviates doxorubicin-induced inflammation and oxidative stress damage of podocytes by IκK/IκB/NF-κB pathway. Biomed Pharmacother. 2019;117:109088. https://doi.org/10.1016/j.biopha.2019.109088.
Pan H, Lin Y, Dou J, Fu Z, Yao Y, Ye S, et al. Wedelolactone facilitates Ser/Thr phosphorylation of NLRP3 dependent on PKA signalling to block inflammasome activation and pyroptosis. Cell Prolif. 2020;53:e12868. https://doi.org/10.1111/cpr.12868.
Rong S, Wan D, Fan Y, Liu S, Sun K, Huo J, et al. Amentoflavone affects epileptogenesis and exerts neuroprotective effects by inhibiting NLRP3 inflammasome. Front Pharm. 2019;10:856. https://doi.org/10.3389/fphar.2019.00856.
Zhao Q, Li HM, Chen XQ, Li RT, Liu D. Terpenoids from Tripterygium hypoglaucum and Their Anti-Inflammatory Activity. Chem Nat Compd. 2018;54:471–4. https://doi.org/10.1007/s10600-018-2381-4.
Mouffouk C, Mouffouk S, Mouffouk S, Hambaba L, Haba H. Flavonols as potential antiviral drugs targeting SARS-CoV-2 proteases (3CLpro and PLpro), spike protein, RNA-dependent RNA polymerase (RdRp) and angiotensin-converting enzyme II receptor (ACE2). Eur J Pharmacol. 2021;891:173759. https://doi.org/10.1016/j.ejphar.2020.173759.
Ribeiro DE, Oliveira-Giacomelli Á, Glaser T, Arnaud-Sampaio VF, Andrejew R, Dieckmann L. et al. Hyperactivation of P2X7 receptors as a culprit of COVID-19 neuropathology. Mol Psychiatry. 2021;26:1044–59. https://doi.org/10.1038/s41380-020-00965-3.
Zahedipour F, Hosseini SA, Sathyapalan T, Majeed M, Jamialahmadi T, Al-Rasadi K. et al. Potential effects of curcumin in the treatment of COVID-19 infection. Phyther Res. 2020;34:2911–20. https://doi.org/10.1002/ptr.6738.
Kong L, Li S, Liao Q, Zhang Y, Sun R, Zhu X, et al. Oleanolic acid and ursolic acid: Novel hepatitis C virus antivirals that inhibit NS5B activity. Antivir Res. 2013;98:44–53. https://doi.org/10.1016/j.antiviral.2013.02.003.
Peyrat LA, Eparvier V, Eydoux C, Guillemot JC, Stien D, Litaudon M. Chemical diversity and antiviral potential in the pantropical Diospyros genus. Fitoterapia. 2016;112:9–15. https://doi.org/10.1016/j.fitote.2016.04.017.
Goh VSL, Mok CK, Chu JJH. Antiviral natural products for arbovirus infections. Molecules. 2020;25:2796. https://doi.org/10.3390/molecules25122796.
Reddy BU, Mullick R, Kumar A, Sharma G, Bag P, Roy CL. et al. A natural small molecule inhibitor corilagin blocks HCV replication and modulates oxidative stress to reduce liver damage. Antivir Res. 2018;150:47–59. https://doi.org/10.1016/j.antiviral.2017.12.004.
Kim JW, Park SJ, Lim JH, Yang JW, Shin JC, Lee SW, et al. Triterpenoid saponins isolated from Platycodon grandiflorum inhibit Hepatitis C virus replication. Evid Based Complement Altern Med. 2013;2013. https://doi.org/10.1155/2013/560417.
Bekut M, Brkić S, Kladar N, Dragović G, Gavarić N, Božin B. Potential of selected Lamiaceae plants in anti(retro) viraltherapy. Pharm Res. 2018;133:301–14. https://doi.org/10.1016/j.phrs.2017.12.016.
Ito T, Yokota R, Watarai T, Mori K, Oyama M, Nagasawa H, et al. Isolation of six isoprenylated biflavonoids from the leaves of Garcinia subelliptica. Chem Pharm Bull. 2013;61:551–8. https://doi.org/10.1248/cpb.c12-01057.
Tsalikis J, Abdel-Nour M, Farahvash A, Sorbara MT, Poon S, Philpott DJ, et al. Isoginkgetin, a Natural Biflavonoid Proteasome Inhibitor, Sensitizes Cancer Cells to Apoptosis via Disruption of Lysosomal Homeostasis and Impaired Protein Clearance. Mol Cell Biol. 2019;39. https://doi.org/10.1128/mcb.00489-18.
Pawellek A, Ryder U, Tammsalu T, King LJ, Kreinin H, Ly T, et al. Characterisation of the biflavonoid hinokiflavone as a pre-mrna splicing modulator that inhibits senp. Elife. 2017;6. https://doi.org/10.7554/eLife.27402.
Salvador JAR, Leal AS, Valdeira AS, Gonçalves BMF, Alho DPS, Figueiredo SAC, et al. Oleanane-, ursane-, and quinone methide friedelane-type triterpenoid derivatives: recent advances in cancer treatment. Eur J Med Chem. 2017;142:95–130. https://doi.org/10.1016/j.ejmech.2017.07.013.
Ríos JL, Máñez S. New pharmacological opportunities for betulinic acid. Planta Med. 2018;84:8–19. https://doi.org/10.1055/s-0043-123472.
Guenné S, Ouattara N, Ouédraogo N, Ciobica A, Hilou A, Kiendrebeógo M. Phytochemistry and neuroprotective effects of Eclipta alba (L.) Hassk. J Complement Integr Med. 2020;17. https://doi.org/10.1515/jcim-2019-0026.
Patel JR, Tripathi P, Sharma V, Chauhan NS, Dixit VK. Phyllanthus amarus: ethnomedicinal uses, phytochemistry and pharmacology: a review. J Ethnopharmacol. 2011;138:286–313. https://doi.org/10.1016/j.jep.2011.09.040.
Liu C, Pan J, Wang A, Lan Y, Li Y, Lu Y. et al. Efficacy of water fraction from Dioscorea cirrhosa on oxidative stress and apoptosis in H9c2 cardiomyocytes induced by H2O2. J Tradit Chin Med. 2021;41:51–8. https://doi.org/10.19852/j.cnki.jtcm.2021.01.007.
Iddir M, Brito A, Dingeo G, Del Campo SSF, Samouda H, La Frano MR, et al. Strengthening the immune system and reducing inflammation and oxidative stress through diet and nutrition: considerations during the covid-19 crisis. Nutrients. 2020;12:1562. https://doi.org/10.3390/nu12061562.
Pratheeba T, Taranath V, Sai Gopal DVR, Natarajan D. Antidengue potential of leaf extracts of Pavetta tomentosa and Tarenna asiatica (Rubiaceae) against dengue virus and its vector Aedes aegypti (Diptera: Culicidae). Heliyon. 2019;5. https://doi.org/10.1016/j.heliyon.2019.e02732.
Miladi S, Abid N, Debarnôt C, Damak M, Canard B, Aouni M, et al. In vitro antiviral activities of extracts derived from Daucus maritimus seeds. Nat Prod Res. 2012;26:1027–32. https://doi.org/10.1080/14786419.2010.550263.
Acknowledgements
This study was supported by CAPES (Coordenação de Aperfeiçoamento de Pessoal de Ensino Superior, Brazil)—Funding code 001 and CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico, Brazil). The authors would like to thank the Academic Publishing Advisory Center (Centro de Assessoria de Publicação Acadêmica, CAPA—www.capa.ufpr.br) of the Federal University of Paraná (UFPR) for assistance with English language translation and developmental editing. Thanks to Center National de la recherché Scientifique (CNRS-France) and to the University of Poitiers-France for support.
Author contributions
DRBL: Conceptualization, methodology, research, writing-original draft, writing-revision and editing, visualization, project administration, acquisition of financing. SPC: Research, original draft writing, acquisition of funding. KMM: Software, resources, data curation, acquisition of funding. FCMB: Formal analysis, acquisition of financing. EdBS: Formal analysis, acquisition of financing. FBM: Software, visualization, acquisition of financing. DPM: Conceptualization, acquisition of financing. JdFGD: Conceptualization, acquisition of financing. OGM: Conceptualization, acquisition of financing. MDM: Conceptualization, supervision, acquisition of financing.
Funding
DRBL, SPC, FBM, FCMB, and EdBS received funding from CAPES (Coordenação de Aperfeiçoamento de Pessoal de Ensino Superior, Brazil), KMM received funding from Center National de la recherche Scientifique (CNRS-France), DPM and JdFGD received funding from Universidade Federal do Paraná (UFPR), CAPES (Coordenação de Aperfeiçoamento de Pessoal de Ensino Superior, Brazil) and OGM and MDM received funding from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Universidade Federal do Paraná (UFPR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Leite, D.R.B., Mantovani, K.M., Cordeiro, S.P. et al. RNA-dependent RNA polymerase (RdRp) natural antiviral inhibitors: a review. Med Chem Res 31, 2089–2102 (2022). https://doi.org/10.1007/s00044-022-02963-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-022-02963-2