Abstract
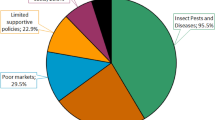
Weeds were a major constraint to food crop production in sub-Saharan Africa (SSA) much more before soil poverty and drought became a problem. Among the weeds infesting pearl millet fields, Cyperus spp. and Digitaria horizontalis are the dominant species in terms of occurrence frequency and emerged plant density and are difficult to control. The depressive effect of weeds on pearl millet yield is compounded by the presence of parasitic species. Buchnera hispida, Striga asiatica, and Striga hermonthica are the main parasitic weeds of pearl millet, of which S. hermonthica is the most damaging and widespread. Control options involve cultural and herbicidal techniques. Some control methods, including cropping systems (crop rotation and intercropping), were recommended for S. hermonthica management. Compared to other Striga hosts, pearl millet has benefited from little research into the development of resistant varieties. Few control options to weed/Striga in pearl millet farming system have been designed, more research is needed to identify innovative weed control strategies in a participatory approach to conservation agriculture. The priority research needs would be to highlight (1) developed herbicide-resistant weed species, the existence of S. hermonthica races in SSA; (2) develop pearl millet varieties resistant to Striga ecotypes; (3) the effect resulted in pearl millet roots × soil microorganisms and nutrients interactions from the rhizosphere level on Striga infection; and (4) Striga severity and aggressiveness induced by climate change.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
15.1 Introduction
Globally, infestation by weed flora and parasitic plants is one of the prominent sources of major agricultural crop loss, causing about $95 billion each year to farmers worldwide (Chaudhary et al. 2018). In Sub-Saharan Africa (SSA), accounting for 50% of the world’s agricultural land, only 1% of that land is suitable for long-term cultivation, attributable to the lack of fertile soil and poor land management practices (Edgerton 2009). While the data on the economic loss due to weed flora is still lacking, infestation by Striga spp. alone has been estimated to exceed $10 billion annually, and heavily infested fields with both weeds and Striga being abandoned by farmers (Gressel et al. 2004; Hearne 2009; Atera et al. 2012). These staggering losses come at a time where pressure on pearl millet, a staple food and fodder crop, production is further aggravated by the ever-increasing population, depleting natural resources, and climate change (Padgham 2009; Lybbert and Sumner 2010). In addition, the farming systems in which pearl millet is produced are characterized by the dominance of smallholder farmers, responsible for the production of over 80% of the locally consumed food, while the majority of these farmers are resource-deficient and have very limited access to loans for on-farm investments (Fan and Rue 2020). Thus, pearl millet has been postulated to play a vital role in ensuring food, nutritional and economic security in SSA. Unfortunately, its production is declining due to various constraints, of which the main factors include agricultural pests such as weed flora and especially parasitic plants, affecting not only the potential production capacity but also the quality of the harvested product.
Within recent years, noticeable efforts have been made to lower many of these barriers. As a result, a wide range of control strategies, including manual, mechanical, cultural, chemical, biological, and genetic, have been deployed either individually or in an integrated manner (Haussmann et al. 2000). Although these methods have provided capacity to farmers to diminish the impact of the weeds on pearl millet production, their success is still limited as the tremendous weed seed bank problem has not been adequately addressed (Kountche et al. 2016).
To realize the full potential of pearl millet, allowing the crop to meet unprecedented challenges would require a transition to sustainable farming practices that support agroecological intensification-based cropping system and environmental health as well as accelerated development and dissemination of innovative approaches that will improve pearl millet resilience to the weed flora and Striga-infection. This is an urgent task for food and nutritional security, given the importance of pearl millet in human diets in SSA.
This chapter aims to provide an overview of the strategies implemented so far to control weed flora and Striga spp. infestation in pearl millet production systems in Africa, with the prospect of highlighting what should be addressed in future research to ensure sustainable control of these invasive weeds.
15.2 Weed Management in Pearl Millet Production Systems in Sub-Saharan Africa
Several factors contribute to the heavy weed infestation in pearl millet fields. Indeed, the installation of the cropping season is spread over a long period with sporadic rains favorable to the development of weeds endowed with genotypic and phenotypic plasticity but which do not allow producers to carry out their sowing. This situation makes seedbed preparation operations more difficult for African farmers and gives a competitive advantage to weeds for the use of environmental resources such as soil nutrients and water compared to pearl millet plants. Mechanical or manual ridging, which is effective and recommended to control weeds without environmental pollution before the first sowing, is not commonly practiced in pearl millet fields. Apart from mowing shrubs and perennial grasses, the majority of pearl millet farmers do no-till seeding. As a result, pearl millet seedlings emerge in an overgrown field with weeds, leading to strong competition for nutrients (Fig. 15.1). In seedbed preparation, soil scraping or shallow plowing is usually done in millet fields. In favor of low soil moisture or regular rainfall, these cultural practices act on weediness as transplanting or a multiplication of the stands of ordinary or perennial grasses.
15.2.1 Weed Flora of Pearl Millet Cropping Systems
Similar to other major agricultural crops, weeds exert strong pressure on pearl millet plants to the point of smothering them in terms of high density coupled with high vegetative development. Farmers fear some weeds because of their survival and adaptability capability. They are considered major weeds whose capacity of nuisance in terms of high plant density, soil covering, and high cost of pearl millet production is significant (Table 15.1). The critical period of pearl millet-weed competition covered 15–42 days after the sowing (Dubey et al. 2023). In pearl millet, the literature about weed flora research is very limited, making the writing of this section difficult and justifying the lack of sufficient references.
15.2.2 Grain Yield Losses Attributed to Weeds
Notably, weeds compete with crops for nutrients, soil moisture, sunlight, and space when limiting, resulting in reduced yields, lower grain quality, and increased production costs (Chaudhary et al. 2018). Weed infestation leads to a significant yield reduction (Ahanchede and Gasquez 1995). Marnotte (1995) pointed out that in Sudan-Sahelian Africa, even before soil poverty was a constraint to farming, weeds were the major obstacle to good yields. Crops are increasingly invaded by weeds, and their density increases with each crop cycle. According to Koch et al. (1982), Deat and Bockel (1986), weeds are one of the major causes of crop failure in developing countries. Pearl millet production, including stalk biomass and grain yield, can be affected by weed infection (Fig. 15.1), but no African statistical data has been documented. In other places, uncontrolled weed infestation reduces pearl millet yield between 16% and 94%, depending on crop cultivars, nature and intensity of weeds, spacing, duration of weeds infestation, management practices, and environmental conditions (Mishra et al. 2018). In Sahelian countries, crop losses of millet are aggravated by the existence of parasitic weeds. Thus, pearl millet yield is reduced, and production can only be increased by increasing the cultivated area (Ahanchede and Gasquez 1995) or, more often, by abandoning heavily infested fields in favor of new land (Déat et al. 1980). Nowadays, these palliative alternatives are compromised by the lack of land due to demographic pressure.
15.2.3 Control Options
As pearl millet is predominantly grown in rain-fed conditions in Africa, weeds deprive the crop of vital nutrients and moisture, affecting the yield accordingly. Because of wider row spacing and slow initial growth in pearl millet, weeds are more problematic during the juvenile crop growth period (Chaudhary et al. 2018). Therefore, early control is required to overcome weed incidence on millet growth and productivity. The concept of the critical period of weed competition, during which weeds have the greatest effect on crop growth, requires the implementation of a weeding operation within this period. Overall, weed management in pearl millet fields in Africa involves mainly manual, cultural, mechanical, and herbicidal techniques.
15.2.3.1 Manual Weeding
In the traditional farming systems, hand pulling and hoe weeding are the two most common practices used in pearl millet fields to control the weeds. These manual interventions were performed from the 2–3 leaf stage to the tillering stage. It is important to stress that both practices are time and energy-consuming, requiring much manpower. This might certainly explain the lack of success.
15.2.3.2 Mechanical Weeding
The most practical mechanical weeding includes the mounding and the ridging using animal attraction that can be asinine, oxen, or horse, depending on the locality. The mounding can be practiced from seedbed establishment to crop flowering while the ridging is implemented, especially from the millet flowering phase. Ridges can be tied to hold water, prevent run-off, and promote plant water use (Mason et al. 2015). Alternatively, tillage options available include shallow cultivation with a harrow (tines), ridging and mounding, tied ridging, and localized tillage to form micro-catchments termed “Zaï” (Fatondji et al. 2001; Nicou and Charreau 1985). As indirect weed control options, these practices can potentially increase water infiltration of early season rains and have little effect on crop root growth, with yield increases from 0% to 15% (Nicou and Charreau 1985).
15.2.3.3 Chemical Control
Over the last decades, weed infestation has worsened as a consequence of nutrient deficiencies in the soil, poor land management, labor unavailability, and to changes in cultivation habits as a sign of modernization of agriculture in Africa. Agrochemical-based weed control is therefore being extensively used in pearl millet production systems in Africa as a weed control strategy. Herbicidal weeding is carried out from seedbed preparation to the whole of the vegetative cycle of millet. However, despite its impressive results in weed dissemination, the major problem is that the majority of pearl millet producers use non-homologous herbicides and have not received adequate, if not any, training in herbicide storage, preparation prior to and application. Furthermore, herbicides are used throughout the cropping season, leading to a detrimental effect on the soil microbial and weed flora, seriously and negatively affecting the organic status of cultivated soils. Since crop yield is reduced under weed infestation, there is a need for developing promising and sustainable solutions to the dual challenges of achieving food security, while supporting a healthy environment.
15.3 Parasitic Weeds Management in Pearl Millet Farming Systems in SSA
In pearl millet production systems, three parasitic plants, including Buchnera hispida Buch. Ham ex D. Don, Striga asiatica (L.) Kuntze, and Striga hermonthica (Del.) Benth. (Orobanchaceae Ex Scrophulariaceae) are considered the most serious agricultural pests of economic importance (Tank et al. 2006). B. hispida, a facultative hemiparasite (Fig. 15.2), is generally found in isolated individuals throughout the cultivated area, especially on pearl millet plants. The parasite is widespread in tropical and subtropical areas of Africa, expanding from West through Central to East Africa, and occurs in more than 30 African countries. Moreover, the potential incidence of B. hispida on cereal crops, including pearl millet in the tropics, has been discussed by Iwoke and Okonkwo (1974).
Besides, S. hermonthica and S. asiatica, obligate root parasites, represent the two most economically important species in tropical areas of Africa (Haussmann et al. 2004; Parker 2012), attacking and irreversibly impacting production of monocotyledonous crops (Parker and Riches 1993). S. asiatica has been reported to attack pearl millet in Eastern Africa, especially in Ethiopia and Kenya, and very limited occurrences have been observed in Western Africa, such as Burkina Faso (Obilana and Ramaiah 1992). In S. hermonthica, no investigation has been initiated to highlight the extent of the parasitism of S. asiatica on pearl millet and other cereals.
15.3.1 Striga Distribution and Economic Incidence
Owing to its unparalleled ability to adapt to diverse climatic and environmental conditions, its high fecundity, and the longevity of the seed reserve in infested soils, Striga has emerged as the major and persistent biotic threat to major staple food, feed, and fodder crops (Pennisi 2010, 2015; Kountche et al. 2016). S. hermonthica appears to be a highly out-crossing species, thus, it is expected to show greater diversity within a population than in related autogamous species (Hamrick 1982; Koyama 2000). This mode of pollination has contributed to the genetic variation in S. hermonthica plants and also restricted the geographical distribution of this species depending on the availability of pollinators (Berner et al. 1997; Mohamed et al. 2007). S. hermonthica is a notorious root hemiparasite on pearl millet in eight West African countries (Nigeria, Ghana, Burkina Faso, Niger, Tchad, Mali, Senegal, and Mauritania); in five East African countries (Sudan, Ethiopia, Yemen, Kenya, and Uganda); and in three South African countries (Angola, Tanzania, and Mozambique) (Obilana and Ramaiah 1992; Parker 2012). However, Striga infection on pearl millet (Fig. 15.3) is insignificant in East Central and Southern Africa (Gressel et al. 2004).
In the various farming systems of sub-Saharan Africa, Striga prevents farmers from achieving the expected grain and fodder yields of pearl millet, sorghum, rice, and maize; hence aggravating food, nutritional and economic insecurity of already resource-deprived smallholder farmers. Striga parasitism inflicts serious damage ranging from few percent (10–31%) to complete crop failure (100%) depending on the crop cultivar, degree of infestation, rainfall pattern, and soil degradation (Atera et al. 2012; Gressel et al. 2004; Wilson et al. 2000). The Striga problem is often associated with low economic resources, low soil fertility, marginal environments with continued crop monoculture, and newly infested areas regrettably due to various human and agricultural activities (Oswald 2005; Rodenburg et al. 2005; Parker 2009). From the economic perspective, Striga infestation has been estimated to be USD 7 to 10 billion loss annually in cereal production systems (Gressel et al. 2004; Hearne 2009; Westwood et al. 2012). Unfortunately, specific statistics about losses due to Striga are not documented for pearl millet and are often combined with those of other cereals such as sorghum and/or maize (Gressel et al. 2004). For subsistence farmers in the arid and semi-arid regions of SSA, however, the highly undesirable consequences of these losses are a return to the top of the cycle, creating a worsening downward spiral and compromising a better horizon. A sustainable Striga control is thus fundamental to ensuring sustainable development and securing food, nutritional and economic security for millions of rural families in Striga-prone regions.
15.3.2 Farmers’ Knowledge and Approaches Towards Striga Management
When dealing with a complex problem such as Striga, research efforts for guiding our responses to such threat have also employed a participatory appraisal of the parasite. This includes farmer surveys carried out in Mali (Hoffmann et al. 1997; Tom V.M. unpublished data), in Burkina Faso (Traoré and Yonli 1999; Rouamba et al. 2021), in Nigeria (Emechebe et al. 2004), and in Kenya (Atera et al. 2012) to get insights into farmers perception on Striga problem and on ground knowledge of endogenous Striga control methods used for coping with the parasite. It appears that some of the smallholders knew about Striga plants, but the majority are unaware of how it reproduces and are generally unable to differentiate Striga species (Hoffmann et al. 1997). Water runoff, animal dung, and wind have been reported as the main factors worsening Striga occurrence. Furthermore, the increased incidence and severity of Striga damage were attributed to the declining soil fertility and the continuous monoculture cropping of host crops. Indigenous Striga control strategies have been inventoried in Western Africa (Table 15.2) and the most widely used were hoe weeding and hand pulling, application of organic manure, crop rotations, and intercropping (Emechebe et al. 2004). These cultural practices have also been recommended by research, but unfortunately, they are not applied according to scientific standards. Indeed, the doses of fertilizers and the modes of association/rotation of crops are not appropriate in Striga controlling. Certain practices, namely, Sorghum—pearl millet rotation, may seem odd for research of bounty on board, while the existence of physiological Striga strains could justify it.
15.3.3 Conventional Strategies Towards Striga Control
The witchweed Striga has long been a devastating agricultural parasitic plant, jeopardizing production of major cereal crops, including pearl millet, and prompting research over the years that aimed at developing management strategies. As a result of decades of remarkable Striga research efforts, different approaches have been developed and deployed for combatting Striga (Haussmann et al. 2000; Teka 2014; Kountche et al. 2016), targeting different impacts on the parasite lifecycle such as the reduction of the seed bank, limitation of seed production and reduction/prevention of seed dissemination to uninfected fields (Haussmann et al. 2000). Notably, control practices that affect germination and attachment of the parasite seed to the host are expected to be more effective as they can prevent parasitism before the host plant is irreversibly damaged and contribute to parasite seed bank reduction.
15.3.3.1 Cultural Methods
As the most traditional practice, cultural methods include hand weeding, tillage, and planting methods, improved soil fertility, cowpea intercropping with cereal, and rotation of cash/trap crops with cereals (Haussmann et al. 2000; Kuchinda et al. 2003; Hess and Dodo 2004; Goldwasser and Rodenburg 2013). Rotation of sesame (Sesamum indicum L.) with pearl millet and/or in association has been reported as a natural suicide germination strategy of Striga seeds (Hess and Dodo 2004). Despite its high potential to significantly contribute to Striga seed bank reduction, this strategy has, however, received little interest. Investigating new sesame production strategies could provide a sustainable alternative to enhance staple food crops’ resilience to Striga. The rotation of cereals like pearl millet with false hosts such as soybean (Glycine max (L.) Merr), cotton (Gossipium hisurtum L.), and voandzou (Voandzeia subterranea L.), which stimulate Striga seed germination but do not allow its fixation (Parkinson et al. 1987), has been recommended to farmers as the technique allows the reduction of the Striga seed bank in infested-soil. Only after several years of implementation can the degree of Striga infestation be reduced to a non-damaging level. However, the lack of cultivable land does not allow farmers to rotate cereals with legumes that are not part of their staple diet. In addition, genotypic differences exist between varieties within the same false-host species, implying that research should recommend legume varieties resistant to S. gesnerioides (Willd.) Vatke and exhibiting suicidal germination potential to S. hermonthica seeds (Traore et al. 2011).
Certain fertilization techniques have been shown to be of value in the control of Striga. Nitrogen fertilizers applied at high rates reduce Striga-related production losses by increasing the vigor of the host crop (Parker 1984; Kim and Adetimirin 1997). Cechin and Press (1993) reported that nitrogen fertilizers affect host exudation, while Pieterse (1991) emphasized that they inhibit the radicle elongation of S. hermonthica. However, the high cost of nitrogen fertilizers makes their use difficult on the farm.
An integrated management system called the “Push–Pull” technology (PPT) has been developed by the International Centre of Insect Physiology and Ecology (ICIPE) to control Striga hermonthica and insect pests (Khan et al. 2008). This technology involved intercropping cereal with a repellent crop Desmodium (Desmodium uncinatum Jacq.) (push), and planting an attractive trap crop, Napier grass (Pennisetum purpureum Schumach or Brachiaria) (pull), as a border crop around this inter-crop. S. hermonthica is controlled by Desmodium and induces abortive germination of Striga seeds that fail to develop and attach onto the hosts’ roots (Fig. 15.4) (Khan et al. 2008; Tsanuo et al. 2003). PPT was disseminated in Eastern Africa (Kenya, Mozambique, Uganda, Rwanda, Zimbabwe) and introduced during 2018–2019 in three West African countries (Burkina Faso, Ghana, and Senegal). PPT has limited success in the Sahelian regions, the major pearl millet producing areas in Africa. Because of one cropping season a year in the Sahel, farmers must plant both weed species every year, whereas in the PPT concept, they are perennial, so their planting is done once for years of farming. In the Sahel, during the whole rain-off season (7–8 months), Desmodium plants thus cannot be alive, and Brachiaria shoots will be fed by animals in raving. Because temperature and rainfall are the key drivers in Sahelian areas in determining suitable habitat niches of Desmodium, research should investigate local weeds to select those that could play similar roles.
15.3.3.2 Chemical Control
The concerns over the Striga problem have also necessitated the deployment of herbicides, aiming to mitigate the parasite infection and impact on major agricultural cereal crops. It is noteworthy that chemical control has been reported to have a high impact rate. Two post-emergence herbicides Triclopyr, 2,4-D and Triclopyr +2,4-D, were applied to Striga hermonthica affecting sorghum in Burkina Faso. Herbicide applications using Triclopyr or 2,4-D (at 1 L ha−1 on 70 and on 85 days after sowing (DAS)) and Triclopyr +2,4-D (at 0.5 L ha−1 on 70 and on 85 DAS) significantly reduced the number of emerged Striga, Striga flowering and seed formation (Traore et al. 2000). However, these results are not systematically transposable to pearl millet crops. Indeed, Clopiramid, 2,4-D, dicamba, picloram, and prosulfuron, belonging to Auxin-mimic and acetolactate inhibitor class herbicides, were evaluated by Dembele et al. (2005) in sorghum and pearl millet growth in Mali as seed priming agents. Their results showed that none of the herbicides used consistently reduced S. hermonthica on pearl millet, whereas their reducing effect on the purple witchweed on sorghum was significant. The most promising herbicide for pearl millet may be dicamba, which showed a slight and non-significant reduction in Striga densities in the field (Dembele et al. 2005). So far, the herbicides used for parasitic weeds include, for example, glyphosate, imidazolinones, glufosinate, 2,4-D, and dicamba (Eplee and Norris 1987; Aly 2007). However, it is important to stress that the output of chemical herbicides application can take various forms, including shifts in weed flora, and disturbed environmental and human health, owing to their hazardous effects. Mounting evidence highlights the many challenges posed by chemical herbicides that even the cost of Striga control and limitations of other control methods now necessitate further development of innovative alternative strategies.
15.3.3.3 Biological Control
Deploying the natural enemies, especially insects, bacterial and fungal antagonists, has long been considered as a potential alternative strategy to suppress Striga infestation. Ultimately, insect parasitoids have been reported on S. hermonthica plants in Eastern Africa (Greathead and Milner 1971), Nigeria (Williams and Caswell 1959), and Burkina Faso (Traore et al. 1996). However, most of these insects are polyphagous, among which we distinguish crop pests such as Spodoptera spp. and Helicoverpa armigera (Greathead 1984). Besides, bacteria have been isolated in West Africa from the rhizosphere of sorghum and maize infested by S. hermonthica. Two races (L1 and L2) of the bacterium Azospirillum brasilense from Mali (Bouillant et al. 1997), 15 isolates of the bacteria Pseudomonas fluorescens and P. putida from Nigeria (Ahonsi et al. 2002) significantly inhibited in in vitro germination of S. hermonthica seeds. Two other species of bacteria, namely Bacillus subtilis Cohn and Pontoena agglomerans [Enterobacter agglomerans (Beijerinck) comb. Nov], were isolated from S. hermonthica plants in Sudan (Abbasher et al. 1996). Moreover, the biological control of Striga through the use of the pathogenic fungus, Fusarium oxysporium as a mycoherbicide, has been recommended to farmers (Marley et al. 1999, 2005; Elzein and Kroschel 2004; Yonli et al. 2006; Zahran et al. 2008). To our knowledge, this control option has not been practically or extensively deployed in farmers’ fields. For the effective use of natural enemies of S. hermonthica identified by the research programs (insects, bacteria, fungi), no bio-herbicide was proposed to the end-users.
15.3.3.4 Genetic Control
As a key strategy towards Striga management, genetic control through the deployment of resistant varieties has gained a marked interest since the approach is believed to offer the most cost-effective and sustainable control of the pernicious weed (Ejeta 2007; Haussmann et al. 2000; Hearne 2009; Wilson et al. 2000; Yoder and Scholes 2010). However, Striga resistance in pearl millet has been much more elusive than in other cereals, such as sorghum and rice. Resistance of 274 Pennisetum glaucum subsp. monodii accessions were evaluated, and four accessions, including PS 202, PS 637, PS 639, and PS 727, were identified to be resistant to Striga, providing useful sources of Striga resistance for improving cultivated pearl millet in West Africa (Wilson et al. 2000; Wilson et al. 2004). Hence, improving Striga resistance in cultivated pearl millet has historically been challenging due to the limited genetic diversity for Striga resistance, lack of knowledge of resistance mechanisms and their molecular genetic basis, and in-field phenotyping constraints (Kountche et al. 2016). Nevertheless, significant progress has been made toward generating resistant varieties during the last two decades. Research conducted by the International Crops Research Institute for the Semi-Arid Tropics (ICRISAT) and its collaborators resulted in the identification of donor sources in cultivated pearl millet, and the development through a field-based phenotypic recurrent selection of the first Striga-resistant varieties (Kountche et al. 2013). Future efforts need to focus on unraveling the pearl millet-Striga interplay, thereby harnessing the as-yet largely untapped genetic potential of existing pearl millet germplasm.
15.3.3.5 Integrated Striga Management (ISM)
Although farmers have been provided with a wide range of control options, the fact remains that none of these methods has proved to effectively subdue the Striga problem (Joel 2000; Oswald 2005). It has been widely agreed that an integrated approach, incorporating a variety of strategies in a wise way, could provide the most comprehensive and sustainable way to deal with Striga (Ejeta 2007; Marley et al. 2004; Elzein et al. 2008; Kanampiu et al. 2003). Nevertheless, it has become evident that the massive and long-lived Striga seed bank accumulated over the years has hindered the efficiency of the conventional strategies applied individually or in an integrated manner, leading to the never-ending Striga problem (Kountche et al. 2016). Integrated Striga management (ISM) has been promoted in several African countries by ICRISAT, the International Institute for Tropical Agriculture (IITA), along with the technical assistance of national agricultural research scientists (NARS) using the Farmer Field School (FFS), a participatory agricultural extension approach, based on “learning by discovery” (Van de Fliert 1993). The FFS learning process builds on existing knowledge and enables farmers to evaluate new and existing technologies in their own fields and to adapt new technologies to their conditions and means. FFS has been upgraded to the cluster-based farmer field school (CBFFS) system developed by IITA and NARS in West Africa (Nathaniels 2005) to perform integrated Striga and soil fertility management (ISSFM). ISSFM is a cropping system approach that is not focused on a single technology and involves (1) a sound knowledge of Striga biology and control, (2) combinations of multiple Striga control options, (3) adaptation of control techniques to local conditions, and (4) a long-term Striga reduction and soil fertility improvement.
15.3.4 Emerging Strategies Toward Ending with Striga Problem
Given the evident and limited success of current methods to effectively contain the parasite invasion, a paradigm shift in how the Striga problem is approached scientifically and in development terms is required to ensure sustainable and rational management of the parasite. This is possible only when the tremendous Striga seed bank, is significantly, if not utterly, depleted to reach the least prejudicial level to host crop production. Towards ending the pernicious Striga problem, efforts are now being directed in harnessing the genetic potential for steady resistance and exploring the seed germination dependency on host-released phytohormones, aiming to develop an environmentally friendly alternative Striga management package.
Interestingly, the past decades have seen marked developments in plant phenomics and omics—genomic, transcriptomic, proteomic, and metabolomic—approaches. An opportunity lies ahead to dissect complex, quantitative traits when both genotype and phenotype can be assessed at a high level of detail. This is especially true for Striga research in pearl millet, for which forward genetics studies have yielded little progress in our understanding of the genetic layout of the traits. Since the molecular mechanisms underlining Striga resistance are yet to be elucidated, combining different omics approaches will help in dissecting pearl millet genes associated with Striga resistance traits, targeting both pre- and post-attachment resistance factors. This, in turn, will foster an in-depth understanding of the link between genotype and phenotype. A direct outcome will certainly be the identification, and mapping of several quantitative trait loci (QTLs) linked to resistance genes and their flanking markers, which will pave the way for pyramiding the identified QTLs/genes into and fast-tracking the development of locally adapted farmers-preferred elite varieties.
Besides optimizing the genetic potential, the suicidal germination approach has recently received considerable attention in the fight against the witchweed Striga. Although the approach is not new (Eplee 1975), the strategy has emerged as one of the most powerful means to deplete the accumulated seed bank, owing to its ability to induce destructive parasite seed germination in the host absence (Kountche et al. 2016; Samejima et al. 2016; Zwanenburg et al. 2016; Kountche et al. 2019). It does not only contribute towards lowering Striga infestation, leading to improved host crop productivity, but has the potential to revolutionize Striga management. Furthermore, there have been ongoing discussions about how the recent developments in strigolactone (SL) research can be further harnessed to develop new strategies for the sustainable control of the parasite. An integrated approach combining genetic resistance and suicidal germination technologies is likely to increase the rate and efficiency and provide the next generation of cost-effective and environmentally friendly alternative Striga control options for the well-being of resource-limited smallholders in SSA.
15.4 Gaps and Future Research Needs
Considering the unprecedented challenges on food security with the persistent weeds problem, the ever-increasing global population, depleting natural resources, and climate change, harnessing each and every single potential means of major crop improvement and tackling all potential causes are the need of the hour. In light of their striking and damage on major staple crops production, one should ask the question about what would be one of the best steps to ending noxious weed flora and witchweed Striga problems in infested fields.
While the genetic potential of pearl millet is yet largely to be exploited, deployment of the new and amazing gene editing technology, such as the Clustered Regularly Interspaced Short Palindromic Repeats/CRISPR-associated (CRISPR/Cas9) system, could potentially help in improving the efficiency of conventional breeding. As more information on the molecular mechanisms underlying host plant–Striga interaction becomes available, it is feasible to manipulate host plant genomes by disrupting genes that contribute to parasite susceptibility.
Despite the research efforts, our knowledge of the Striga seed bank-level, distribution, and dynamics is still very limited, attributable to various factors such as crop rotation, intercropping, wind, variable growing seasons, and anthropogenic spread. Notably, the assessment and characterization of Striga infestation distribution appear to be a crucial step prior to developing effective and rationally designed interventions for increased and sustainable pearl millet productivity. Moreover, attempts to elucidate S. hermonthica strains have been initiated (Ramaiah 1984; Kim et al. 1994; Freitag et al. 1996; Bozkurt et al. 2015). Two clusters of S. hermonthica populations were distinguished in Sudan, and the millet strain slightly differed and was more specific to its host (Ali et al. 2009). However, no study successfully identified S. hermonthica strains as it was done for S. gesnerioides (Cardwell and Lane 1995; Li and Timko 2009). Indeed, in the same area, a crop variety can prove to be resistant to S. hermonthica in one location and susceptible to another one. Besides, “intracrop specific” describes strains reacting in a different manner to cultivars of a single host crop, whereas “intercrop specific” describes strains reacting in a different manner to different host crops (Kim et al. 1994). The genetic variability of S. hermonthica has not been sufficiently evaluated relative to its wide distributions (Mohamed et al. 2007). The genotypic identification of S. hermonthica ecotypes and their mapping at the scale of each African country seems a prerequisite for a better understanding of the variability in the parasite aggressiveness, leading to the development of sustainable resistant pearl millet varieties.
Notably, it is postulated that the root system of host plants and the microorganisms from its rhizosphere interact, consequently influencing the quality and quantity of host root exudates responsible for the stimulation of Striga seeds (Bouwmeester et al. 2007). It is worth noting that a deeper understanding of the interactions Striga × soil microorganisms × pearl millet crop will provide insights into the variation in Striga infestation in the various farming systems. Importantly, the grain yield loss of the host crop due to Striga is positively correlated with the soil fertility level (Showemimo et al. 2002). Thus, modeling the levels of soil type and fertility, Striga ecotype and infestation, climate variability, and the resulting yield losses appears to be another area yet to explore in order to predict the impact of Striga parasitism on pearl millet production. This should be integrated with generating individual data about parasitic weeds (S. hermonthica, S. asiatica, Buchnera hispida) incidence on pearl millet production, yet to be documented. The availability of such data will increase awareness of the real Striga problem among producers and policymakers.
Climate envelope models can serve as a guideline for understanding the present distribution of parasitic weeds and predicting their potential future geographic distribution in light of climate and land use change. However, forecasts across African continental ranges prove difficult due to (1) the local climate variability and adaptations, (2) the host plant/management variations, and, (3) the local varieties within target species.
References
Abbasher AA, Hess DE, Sauerborn J, Kroschel J (1996) Effect of different Fusarium spp on seed germination of: Striga hermonthica (sorghum and millet strains); S. asiatica and S. gesnerioides. In: Monero MT, Cubero JI, Berner D, Musselman LJ, Parker C (eds) Advances in parasitic plant research. Dirección General de Investigación Agraria, Seville, pp 880–887
Ahanchede A, Gasquez J (1995) Mauvaises herbes des cultures pluviales au Nord-Est du Bénin. Agriculture et Développement 7:22–29
Ahonsi MO, Berner DK, Emechebe AM, Lagoke ST (2002) Selection of rhizobacterial strains for suppression of germination of Striga hermonthica (Del.) Benth. seeds. Biol Control 24(2):143–152
Ali RAMA, El-Hussein AA, Mohamed KI, Babiker AGT (2009) Specificity and genetic relatedness among Striga hermonthica strains in Sudan. Life Sci Int J 3(3):1159–1166
Aly R (2007) Conventional and biotechnological approaches for control of parasitic weeds. Vitr Cell Dev Biol Plant 43:304–317
Atera EA, Itoh K, Azuma T, Ishii T (2012) Farmers’ perspectives on the biotic constraint of Striga hermonthica and its control in western Kenya. Weed Biol Manag 12:53–62
Berner DK, Winslow MD, Awad AE, Cardwell KF, Mohan Raj DR, Kim SK (1997) Striga research methods – a manual, 2nd edn. International Institute of Tropical Agriculture (IITA), Ibadan
Bouillant ML, Miché L, Ouedraogo O, Alexandre G, Jacoud C, Sallé G, Bally R (1997) Inhibition of Striga seed germination associated with sorghum growth promotion by soil bacteria. Comptes Rendus de l'Académie des Sciences-Series III-Sciences de la Vie 320:159–162
Bouwmeester HJ, Roux C, Lopez-Raez JA, Becard G (2007) Rhizosphere communication of plants, parasitic plants and AM fungi. Trends Plant Sci 12:224–230
Bozkurt ML, Muth P, Parzies HK, Haussmann BIG (2015) Genetic diversity of east and west African Striga hermonthica populations and virulence effects on a contrasting set of sorghum cultivars. Weed Res 55:71–81
Cardwell KF, Lane JA (1995) Effect of soils, cropping system, and host phenotype on incidence and severity of Striga gesnerioides on cowpea in West Africa. Agric Ecosyst Environ 53:253–262
Chaudhary C, Dahiya S, Rani S, Pandey S (2018) Review and outlook of weed management in pearl millet. Int J Chem Stud 6(2):2346–2350
Cechin I, Press MC (1993) Nitrogen relations of the sorghum-Striga hermonthica host-parasite association: germination, attachment and early growth. New Phytol 124:681–687
Deat MM, Bockel N (1986) Identification des situations agricoles en Afrique sahélienne en vue d'améliorer leur adaptation à la sécheresse. CIRAD, Paris. 84 p
Deat M, Sement G, Fontenay P (1980) Premières observations sur la levée de quelques adventices tropicales. VIe Colloque International sur l'Ecologie, la Biologie et la systématique des mauvaises herbes. 7–8 Mai 1980. Montpellier, pp 321–328
Dembele B, Dembele D, Westwood JH (2005) Herbicide seed treatments for control of purple witchweed (Striga hermonthica) in sorghum and millet. Weed Technol 19:629–635
Dubey RP, Chethan CR, Choudhary VK, Mishra JS (2023) A review on weed management in millets. Indian J Weed Sci 55(2):141–148
Edgerton MD (2009) Increasing crop productivity to meet global needs for feed, food, and fuel. Plant Physiol 149(1):7–13
Ejeta G (2007) Breeding for Striga resistance in sorghum: exploitation of an intricate host-parasite biology. Crop Sci 47:S216–S227
Elzein A, Kroschel JK (2004) Fusarium oxysporum Foxy 2 shows potential to control both Striga hermonthica and S. asiatica. Weed Res 44:433–438
Elzein A, Kroschel J, Leth V (2006) Seed treatment technology: An attractive delivery system for controlling root parasitic weed Striga with mycoherbicide. Biocontrol Sci Technol 16(1):3–26
Emechebe AM, Ellis-Jones J, Schulz S, Chikoye D, Douthwaite B, Kurreh I, Tarawali IG, Hussaini MA, Kormawa P, Sanni A (2004) Farmers’ perception of the Striga problem and its control in northern Nigeria. Exp Agric 40:215–232
Eplee RE (1975) Ethylene: a witchweed seed germination stimulant. Weed Sci 23:433–436
Eplee RE, Norris RS (1987) Chemical control of Striga. In: Musselman LJ (ed) Parasitic weeds in agriculture: vol. I, Striga. CRC Press, Boca Raton, FI, pp 173–182
Fan S, Rue C (2020) The role of smallholder farms in a changing world. In: The role of smallholder farms in food and nutrition security. Springer International Publishing, Cham, pp 13–28
Fatondji D, Martius C, Vlek P (2001) Zaï - a traditional technique for land rehabilitation in Niger. ZEFnews 8:1–2
Freitag J, Hess DE, Welz HG (1996) Pearl millet and sorghum specific races of Striga hermonthica in Niger, pp 471–478. In: Monero M, Cubero JI, Berner D, Musselman IJ, Parker C (eds) Advances in parasitic plant research, sixth international parasitic weed symposium, April 16-18, Junta de Andalucia, 923 pp
Goldwasser Y, Rodenburg J (2013) Integrated agronomic management of parasitic weed seed banks. In: Parasitic Orobanchaceae: Parasitic mechanisms and control strategies. Springer Berlin Heidelberg, Berlin, Heidelberg, pp 393–413. Jun 9
Greathead DJ (1984) The natural enemies of Striga spp. and the prospects for their utilisation as biological control agents. In: Striga, biology and control, pp 133–160
Greathead DJ, Milner JE (1971) A survey of Striga spp.(Scrophulariaceae) and their insect natural enemies in East Africa with a discussion on the possibilities of biological control. Trop Agric 48(2):111–124
Gressel J, Hanafi A, Head G, Marasas W, Obilana B, Ochanda J, Souissi T, Tzotzos G (2004) Major heretofore intractable biotic constraints to African food security that may be amenable to novel biotechnological solutions. Crop Prot 23:661–689
Hamrick JL (1982) Plant population genetics and evolution. Am J Bot 69:1685–1693
Haussmann BIG, Hess DE, Welz HG, Geiger HH (2000) Improved methodologies for breeding Striga-resistant sorghums. Field Crop Res 66:195–211
Haussmann BIG, Hess DE, Omanya GO, Folkertsma RT, Reddy BVS, Kayentao M, Welz HG, Geiger HH (2004) Genomic regions influencing resistance to the parasitic weed Striga hermonthica in two recombinant inbred populations of sorghum. Theor Appl Genet 109:1005–1016
Hearne SJ (2009) Control - the Striga conundrum. Pest Manag Sci 65:603–614
Hess DE, Dodo H (2004) Potential for sesame to contribute to integrated control of Striga hermonthica in the west African Sahel. Crop Prot 23:515–522
Hoffmann G, Diarra C, Dembele B (1997) Les espèces parasites herbacées des cultures vivriéres en Afrique : biologie, in Dact, méthodes de lutte. Agriculture et développement 13:30–51
Iwoke FIO, Okonkwo SNC (1974) Facultative hemi-parasitism in Buchnera hispida Harm. Ex D. Don. Ann Bot 38:993–1002
Joel DM (2000) The long-term approach to parasitic weeds control: manipulation of specific developmental mechanisms of the parasite. Crop Prot 19:753–758
Kanampiu FK, Kabambe V, Massawe C, Jasi L, Friesen D, Ransom JK, Gressel J (2003) Multi-site, multi-season field tests demonstrate that herbicide seed-coating herbicide-resistance maize controls Striga spp. and increases yields in several African countries. Crop Prot 22:679–706
Khan ZR, Midega CAO, Amudavi DM, Hassanali A, Pickett JA (2008) On-farm evaluation of the ‘push–pull’ technology for the control of stemborers and Striga weed on maize in western Kenya. Field Crop Res 106:224–233
Kim SK, Adetimirin VO (1997) Responses of tolerant and susceptible maize varieties to timing and rate of nitrogen under Striga hermonthica infestation. Agron J 89:38–44
Kim SK, Akintunde AY, Walker P (1994) Responses of maize, sorghum and millet host plants to infestation by Striga hermonthica. Crop Prot 13(8):559–567
Koch W, Beshir ME, Unterladstatter R (1982) Crop losses due to weeds. Improving weed management. FAO plant production and protection paper, Rome 6-10 September, 1982, vol 44, pp 153–165
Kountche BA, Hash CT, Dodo H, Laoualy O, Sanogo MD, Timbeli A, Vigouroux Y, This D, Nijkamp R, Haussmann BI (2013) Development of a pearl millet Striga-resistant genepool: response to five cycles of recurrent selection under Striga-infested field conditions in West Africa. Field Crop Res 154:82–90
Kountche BA, Al-Babili S, Haussmann BIG (2016) Striga: a persistent problem on millets. In: Biotic stress resistance in millets. Elsevier, Amsterdam, pp 173–203
Kountche BA, Jamil M, Yonli D, Nikiema MP, Blanco-Ania D, Asami T, Zwanenburg B, Al-Babili S (2019) Suicidal germination as a control strategy for Striga hermonthica (Del.) Benth. in smallholder farms of sub-Saharan Africa. Plants People Planet 1:107–118
Koyama ML (2000) Molecular markers for studying pathogen variability: implications for breeding for resistance to Striga hermonthica. In: Haussmann BIG, Hess DE, Koyama ML, Grivet L, Rattunde HFW, Geiger HH (eds) Breeding for Striga resistance in cereals. Margraf Verlag, Weikersheim, Germany, pp 227–254
Kuchinda NC, Kureh I, Tarfa BD, Shinggu C, Omolehin R (2003) On-farm evaluation of improved maize varieties intercropped with some legumes in the control of Striga in the northern Guinea Savanna of Nigeria. Crop Prot 22:533–538
Li J, Timko PJ (2009) Gene-for-gene resistance in Striga-cowpea associations. Science 325:1094
Lybbert T, Sumner D (2010) Agricultural technologies for climate change mitigation and adaptation in developing countries: policy options for innovation and technology diffusion. ICTSD/Food & Agricultural Trade, issue brief no. 6, 32 pp
Marley PS, Ahmed SM, Shebayan JAY, Lagoke STO (1999) Isolation of Fusarium oxysporum with potential for biocontrol of the witchweed (Striga hermonthica) in the Nigerian savanna. Biocontrol Sci Tech 9:159–163
Marley PS, Aba DA, Shebayan JAY, Musa R, Sanni A (2004) Integrated management of Striga hermonthica in sorghum using a mycoherbicide and host plant resistance in the Nigerian Sudano-Sahelian savanna. Weed Res 44:157–162
Marley PS, Kroschel J, Elzein A (2005) Host specificity of Fusarium oxysporum Schlecht (isolate PSM 197), a potential mycoherbicide for controlling Striga spp. in West Africa. Weed Res 45:407–412
Marnotte P (1995) Utilisation des herbicides : contraintes et perspectives. Agriculture et Développement 7:12–21
Mason SC, Nouri MN, Pale S (2015) Pearl millet production practices in semi-arid West Africa: a review. Exp Agric 51(4):501–521
Mishra JS, Kumar R, Upadhyya PK, Hans H (2018) Weed management in millets. Indian Farm 68(11):77–79
Mohamed KI, Bolin JF, Musselman LJ, Peterson AT (2007) Genetic diversity of Striga and implications for control and modeling future distributions. In: Integrating new technologies for striga control: towards ending the witch-hunt. World Scientific Publishing, Singapore, pp 71–84
Nathaniels NQR (2005) Cowpea, farmer field schools and farmer-to-farmer extension: a Benin case study. AgREN Network Paper 148, p 15
Nicou R, Charreau C (1985) Soil tillage and water conservation in semi-arid West Africa. In: Ohm HW, Magy JG (eds) Appropriate technologies for farmers in semi-arid West Africa. Purdue University, West Lafayette, pp 9–32
Obilana AT, Ramaiah KV (1992) Striga (witchweeds) in sorghum and millet: knowledge and future research needs. In: de Milliano WAJ, Frederiksen RA, Bengston GD (eds) Sorghum and millets diseases: a second world review. International Crops Research Institute for the Semi-Arid Tropics (CP 741), Patancheru, pp 187–201
Oswald A (2005) Striga control – technologies and their dissemination. Crop Prot 24:333–342
Padgham J (2009) Agricultural development under a changing climate: opportunities and challenges for adaptation. Agriculture and Rural Development & Environment Departments, Joint Departmental discussion paper-issue 1, 168 pp
Parker C (1984) The influence of Striga species on sorghum under varying nitrogen fertilization. In: Proceedings of the Third International Symposium on Parasitic Weeds. International Center for Agricultural Research in the Dry Areas, pp 90–98
Parker C (2009) Observations on the current status of Orobanche and Striga problems worldwide. Pest Manag Sci 65:453–459
Parker C (2012) Parasitic weeds: a world challenge. Weed Sci 60:269–276
Parker C, Riches CR (1993) Parasitic weeds of the world: biology and control. Caslefield Press, Kettering. 332 pp
Parkinson V, Efron Y, Bello L, Dashiel K (1987) Trap crops as a cultural measure in Striga control in Africa. FAO Plant Prot Bull 35:51–54
Pennisi E (2010) Armed and dangerous. Science 327:804–805
Pennisi E (2015) How crop-killing witchweed senses its victims, vol 350. American Association for the Advancement of Science, New York, p 146
Pieterse AH (1991) The effect of nitrogen on the germination of seeds of Striga hermonthica and Orobanche crenata. In: Progress in Orobanche research. Proceedings of the International Workshop on Orobanche Research, Obermarchtal, Germany, pp 115–124
Ramaiah KV (1984) Physiological specialization of Striga hermonthica and crop speficity. In: Third international symposium on parasitic weeds, Allepo, Syria, pp 58–65
Rodenburg J, Bastiaans L, Weltzien E, Hess DE (2005) How can field selection for Striga resistance and tolerance in sorghum be improved? Field Crop Res 93:34–50
Rouamba A, Shimelis H, Drabo I, Laing M, Gangashetty P, Mathew I, Mrema E, Shayanowako AIT (2021) Constraints to pearl millet (Pennisetum glaucum) production and farmers’ approaches to Striga hermonthica management in Burkina Faso. Sustainability 13:8460
Samejima H, Babiker AG, Takikawa H, Sasaki M, Sugimoto Y (2016) Practicality of the suicidal germination approach for controlling Striga hermonthica. Pest Manag Sci 72:2035–2042
Showemimo FA, Kimbeng CA, Alabi SO (2002) Genotype response of sorghum cultivars to nitrogen fertilization in the control of Striga hermonthica. Crop Prot 21:867–870
Tank DC, Beardsley PM, Kelchner SA, Olmstead RG (2006) Review of the systematics of Scrophulariaceae and their current disposition. Aust Syst Bot 19:289–307
Teka HB (2014) Advance research on Striga control: a review. Afr J Plant Sci 8:492–506
Traore H, Maillet J (1992) Flore adventice des cultures céréalières annuelles du Burkina Faso. Weed Res 32:279–293
Traoré H, Yonli D (1999) Study of farmers’ perception of the Striga problem and inventory of indigenous Striga control methods in Burkina Faso. In: Kroschel J, Mercier-Quarshie H, Sauerborn J, Margra Verlag W (eds) Advances in parasitic weed control at on-farm level, vol I. Joint action to control Striga in Africa, Germany, pp 197–205
Traore D, Vincent C, Stewart RK (1996) Association and synchrony of Smicronys guineanus and Smicronys umbrinus (Coleoptera: Curculionidea) and the parasitic weed Striga hermonthica (Del.) Benth. (Scrophulariaceae). Biol Control 3:307–315
Traore H, Ouedraogo O, Salle G (2000) Contrôle du Striga hermonthica (Del.) Benth.par la combinaison de la variété, du 2,4-D ou de l’arrachage au Burkina Faso. Etudes et recherches sahéliennes N° 4–5:86–93
Traore H, Yonli D, Diallo D, Sereme P (2011) Suicidal germination of Striga hermonthica (Del.) Benth. by cotton, cowpea and groundnut Genotypes in Burkina Faso. Int J Agric Res 6(1):49–57
Tsanuo MK, Hassanali A, Hooper AM, Khan Z, Kaberia F, Pickett JA, Wadhams LJ (2003) Isoflavanones from the allelopathic aqueous root exudate of Desmodium uncinatum. Phytochemistry 64(1):265–273
Van de Fliert E (1993) Integrated pest management: farmer field schools generate sustainable practices. Wageningen Agricultural University Papers, Wageningen, The Netherlands, pp 3–93
Westwood JH, dePamphilis CW, Das M, Fernandez-Aparicio M, Honaas LA, Timko MP, Wafula EK, Wickett NJ, Yoder JI (2012) The parasitic plant genome project: new tools for understanding the biology of Orobanche and Striga. Weed Sci 60:295–306
Williams CN, Caswell GH (1959) An insect attacking Striga. Nature 184:1668–1668
Wilson JP, Hess DE, Hanna WW (2000) Resistance to Striga hermonthica in wild accessions of the primary gene pool of Pennisetum glaucum. Phytopathology 90:1169–1172
Wilson JP, Hess DE, Hanna WW, Kumar KA, Guptae SC (2004) Pennisetum glaucum subsp. monodii accessions with Striga resistance in West Africa. Crop Prot 23:865–870
Yoder JI, Scholes JD (2010) Host plant resistance to parasitic weeds; recent progress and bottlenecks. Curr Opin Plant Biol 13:478–484
Yonli D, Traore H, Hess DE, Sankara P, Sereme P (2006) Effect of growth medium, Striga seed burial distance and depth on efficacy of Fusarium isolates to control Striga hermonthica in Burkina Faso. Weed Res 46:73–81
Zahran E, Kohlschmid E, Sauerborn J, Abbasher AA, Muller-Stover D (2008) Does an alication as seed coating stabilize the efficacy of biological control agents of Striga hermonthica under field conditions? J Plant Dis Prot S 21:467–471
Zwanenburg B, Mwakaboko AS, Kannan C (2016) Suicidal germination for parasitic weed control. Pest Manag Sci 72:2016–2025
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2024 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Yonli, D., Traore, H., Kountche, B.A. (2024). Weed and Striga Management in Pearl Millet Production Systems in Sub-Saharan Africa. In: Tonapi, V.A., Thirunavukkarasu, N., Gupta, S., Gangashetty, P.I., Yadav, O. (eds) Pearl Millet in the 21st Century. Springer, Singapore. https://doi.org/10.1007/978-981-99-5890-0_15
Download citation
DOI: https://doi.org/10.1007/978-981-99-5890-0_15
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-99-5889-4
Online ISBN: 978-981-99-5890-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)