Abstract
Intranasal drug administration has enormous research potential, including both drugs designed specifically for intranasal use and off-label applications of commercially available generic medications. The olfactory canal architecture has a well-vascularized mucosal framework, which permits the drug molecule to be transported fast through a single epithelium layer and into the systemic bloodstream without having to go through hepatic and intestinal processing first. The majority of CNS therapy treatments now available rely on systemic medication delivery to the brain via the oral route and intravenous injection. The current CNS therapy treatments mostly rely on systemic medication delivery to the brain via the oral route and intravenous injection. The primary drawback of these approaches is the limited pharmacological access of therapeutic compounds in the blood supply to the brain, which is linked with decreased therapeutic efficacy and, eventually, increased peripheral adverse effects. The current study highlights the clinical status of numerous medications employed in intranasal drug delivery systems invented by diverse scientists for various brain illnesses. The paper also focuses on current advancements in intranasal medication delivery systems modeled for brain transport.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
1.1 Central Nervous System Overview
The central nervous system (CNS), one of the two basic divisions of the nervous system, is made up of the brain and spinal cord. The other division is the peripheral nervous system (PNS), which is found outside of the brain and spinal cord. The CNS’s primary role is to consolidate incoming information, coordinate, and impact the functioning of all bodily components. CNS and brain ailments impact more than a hundred million individuals globally, according to a World Health Organization (WHO) estimate, and require long-term therapy (World Health Organization 2006). There are various illnesses affecting the brain and CNS that impair a person’s motor functional activity. It has proven difficult to get drugs to the brain to treat neurological illnesses (Barchet and Amiji 2009; MacDonald et al. 2000). The most effective route to transport medications to any part of the body is orally, intranasally, or intravenously. There are several obstacles to these medication delivery pathways for brain targets (Bozdağ Pehlivan 2013).
2 Barriers to Brain Drug Delivery
Physiological barriers that prevent medications from accessing the brain commonly hamper the treatment of CNS diseases. Despite the availability of a number of brain targeting methods, none have proved successful in producing therapeutic concentrations in the brain (Lesniak and Brem 2004). Three primary barriers separate blood and the CNS: the BBB, the blood–cerebrospinal fluid barrier (BCSFB), and the blood–tumor barrier. Aside from these obstacles, efflux transporter proteins such as P-glycoprotein (P-gp) and others operate as gatekeepers, preventing drug molecules from reaching the brain (Ricci et al. 2006).
2.1 Blood–Brain Barrier
The blood–brain barrier (BBB) is a capillary-based endothelial barrier that runs throughout the brain parenchyma. Endothelial tight junctions contain intracellular junction proteins termed occludin and claudin. These structures provide a barrier to the flow of chemicals between the bloodstream and the brain (Sanchez-Covarrubias et al. 2014; Chen and Liu 2012).
The existence of tight junctions between cells, which inhibits paracellular transport owing to high electrical resistance, is one of the key properties of BBB (Abbott et al. 2006). The absence of fenestrations, the existence of P-gp, which is implicated in drug efflux, and the presence of fewer endocytic vesicles, which reduces transcellular flow (Chang et al. 2009), are all factors that limit transcellular flux (Sanchez-Covarrubias et al. 2014). As a result, most medications are limited from crossing the BBB, and only tiny lipophilic molecules can pass the endothelium/BBB (Krol 2012).
Important nutrients are transported across the BBB by particular transportation mechanisms. However, it prohibits xenobiotic entrance, implying that each peptide molecule has a unique pathway for entry (Pardridge 1983). Endogenous and exogenous solutes are metabolized by mitochondria and metabolizing enzymes such as adenylate cyclase, guanylate cyclase, etc. (Brownlees and Williams 1993). As a result, these tight connections prevent ion or molecule exchange between the systemic circulation and the central nervous system. As a result, active drug transport into brain tissue is reduced, resulting in the failure of CNS therapies for diseases including Alzheimer’s, schizophrenia, epilepsy, and depression (Löscher and Potschka 2005) (Fig. 19.1).
(a) Representation and (b) Scanning Electron Microscope Image of the Capillary (Pardridge 2006)
2.2 Blood–Cerebrospinal Fluid Barrier
The blood–cerebrospinal fluid barrier (BCB) controls the passage of blood-carried chemicals into the CSF. CSF and brain parenchymal interstitial fluid can exchange chemicals. Because of their intimate connections, the arachnoid membrane and choroid plexus provide an important function as barriers between the blood and CSF (Misra et al. 2003). Figure 19.2 shows a diagram of the BCB (Alam et al. 2010).
Blood–cerebrospinal Fluid Barrier Schematic Diagram (BCB) (Alam et al. 2010)
2.3 Blood–Tumor Barrier
The major issue in brain tumors is permeability. The first variable regarding capillary permeability is the tumor microvessel population, also known as the blood–tumor barrier, and the second variable is the spatial distribution of the target capillaries (Groothuis 2000).
3 Drug Transport Through Blood-Brain-Barrier (BBB)
Transport over the BBB is a major hurdle to drug absorption, and the BBB is reinforced by many parallel barriers, mainly the capillary bed in the vascular BBB, the BCB, and the choroid plexus, all of which offer strong endothelial resistance. Essential solutes enter the CNS via one of the two methods listed below (Pardridge 2006). Figure 19.3 depicts the route of drug penetration through the BBB.
3.1 Passive Diffusion
As illustrated in Fig. 19.4, highly lipophilic molecules of low molecular weight can extensively infiltrate the BBB via passive diffusion via multiple routes. Uncharged molecules partition easily between tissues and are absorbed in the brain due to their lipophilic nature (Jouyban and Soltani 2012). A molecule moving passively across the BBB has an average molecular mass of 357 Da, or less than 1 nm (Jain 2012).
(a) Efflux transport mechanisms prevent numerous chemicals from entering the brain. (b) Saturable transport is in charge of the transfer. (c) Transcellular lipophilic route for lipophilic agent diffusion. (d) A biological route that allows extremely tiny quantities of hydrophilic substances to diffuse. (e) Endocytosis of essential proteins such as insulin, transferrin, and cytokines is mediated by particular receptors. (f) Adsorptive endocytosis is caused by the cationization of certain molecules, such as albumin
There are two processes that control passive diffusion:
Free diffusion: Some molecules go between cells outside of the cell membrane (e.g., Sucrose). Because of the tight connections, transport is limited, particularly for hydrophobic substances, certain molecules travel transcellular across the cells (e.g., ethanol).
Facilitated diffusion: In this sort of transport, solutes are moved from one side of the membrane to the other by altering the conformation of certain membrane proteins (Alam et al. 2010). Molecular weight, lipophilicity, ionization, and hydrogen bonding are all variables that influence passive diffusion through the BBB.
3.2 Active Transport
Active transport is a sort of membrane transport that is energy driven and works in the opposite direction of the electrochemical gradient. This method is required to avoid any interruption in the passage of important substances such as glucose from the blood to the brain and vice versa for normal brain metabolism and activity (Nau et al. 2010; Begley 2004).
3.2.1 Receptor-Mediated Transcytosis
Large chemicals necessary for brain homeostasis enter the BBB via specialized receptors found in the endothelial membrane, such as insulin receptors, transferrin receptors, LDL receptors, and so on. Table 19.1 lists key receptor-mediated transporter systems at BBB (Patel et al. 2011).
3.2.2 Adsorptive-Mediated Transcytosis
Adsorptive-mediated transcytosis (AMT) (Gabathuler 2010) refers to the nonspecific transport of charged [cationic] peptide molecules such as protamine and albumin across the BBB. The transcytosis routes and enzymatic characteristics, together with the large number of mitochondria in endothelial cells, produce a shove for molecules to travel across the endothelial cytoplasm (Sauer et al. 2005; Xiao and Gan 2013).
3.3 Carrier-Mediated Transport
Select membrane-confined transporters carry endogenous chemicals, including vital nutrients, from the blood to the brain parenchyma. Changing the drug/substrate to imitate a nutrient/endogenous substrate capable of transporting, or employing one of the current carrier systems, are two options for CMT.
3.3.1 Drug Delivery Strategies Across the BBB
In the case of CNS disorder, conventional drug delivery enters the brain through systemic circulation. To get the appropriate therapeutic concentration at the intended site, systemic drug levels must be increased by the repeated dosage or extended administration. This might lead to an increase in toxicity. Invasive, non-invasive, and miscellaneous techniques are the three types of strategies for crossing the BBB.
3.3.2 Intranasal Drug Delivery Systems
In CNS disorders, conventional medication delivery enters the brain via systemic circulation. Systemic drug levels must be raised by repeated dosing or longer administration to achieve the optimal therapeutic concentration at the target location. This might lead to an increase in toxicity. There are three types of strategies for crossing the BBB: invasive, non-invasive, and various.
4 The Nasal Cavity’s Anatomy and Physiology
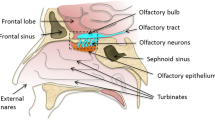
The human nasal cavity is around 12–14 cm long, with a total capacity of 15–20 mL. According to (Ali et al. 2010), the nasal cavity is divided into two parts by a vertical barrier called the nasal septum. In an anterior–posterior orientation, from the nostrils to the nasopharynx, the nasal cavity extends. Following delivery into the nasal canal, the drug might be deposited in one of three anatomically distinct sites. The drug must pass through the olfactory membrane as well as the arachnoid membrane, which covers the arachnoid space, which contains cerebrospinal fluid, before reaching the CNS through the nasal cavity (Vyas et al. 2005). Figure 19.5 depicts an overview of the nasal cavity.
4.1 Nasal Vestibule
The most anterior section of the nasal cavity is made up of a 0.6 cm2 patch with sebaceous glands that is composed of stratified squamous and keratinized epithelium. Although the nasal vestibule provides considerable resistance to hazardous environmental chemicals, medication absorption becomes extremely difficult in this location (Djupesland 2013).
4.2 Respiratory Region
In humans, the nasal respiratory area, also known as the conchae, is the biggest component, lining roughly 80–90% of the nasal cavity. It has a huge surface area and high vascularity, allowing for increased medication absorption via the respiratory epithelium. The cilia, which are hair-like projections on the surface of epithelial cells, are responsible for the epithelium’s expanded surface area. Furthermore, these cilia aid in the transmission of mucus to the nasopharynx (Ozsoy et al. 2009). Mucociliary clearance is the activity of clearing mucous into the gastrointestinal channel (MCC). This protective effect of mucous results in the quick removal of medications from the body. This protective effect of mucous results in the quick clearance of medicines from the nasal cavity by reducing the drug’s dwell time in the nasal cavity. Mucin, which is found in the mucous layer, is responsible for trapping big molecular weight drugs such as proteins and peptides (Charlton et al. 2007). Figure 19.6 displays an overview of respiratory mucosa with structural demonstration (Mandpe et al. 2013).
Overview of respiratory mucosa with structural demonstration (Mandpe et al. 2013)
4.3 Olfactory Region
The olfactory region is made of three types of cells: olfactory receptor cells, supportive epithelial cells, and basal cells. The only portion of the CNS that is connected directly to the physical realm is the neuroepithelium (Lochhead and Thorne 2012; Talegaonkar and Mishra 2004). Figure 19.7 shows an overview of the organization of the human olfactory region (Desai et al. 2015).
Human olfactory region (Desai et al. 2015)
4.4 Applications of Nasal Route for Delivery and Therapeutic Opportunities
Many benefits of the nasal mucosal structure have been identified, including the capacity to deliver medications with a rapid beginning of the action, increased bioavailability, and patient compliance.
4.4.1 Nasal Delivery of Systemic Drugs
Nasal delivery is regarded to be an interesting option for drugs that are having difficulty with current conventional techniques. Needle-free administration as an alternative to injections and lower chances of drug degradation, among other benefits, has resulted in the availability of systemically acting drugs with many currently being investigated (Gangurde et al. 2019).
4.4.2 Nasal Route for Vaccine Delivery
Nasal delivery is seen to be an intriguing possibility for treatments that are not working well with current methods. Needle-free delivery as a substitute for injections, as well as a reduced risk of drug degradation (Gangurde et al. 2019).
4.4.3 Nasal Route for Topical Delivery
Allergies impact between 5% and 10% of the global population. Topical steroids are the preferred treatment option for individuals suffering from chronic allergic and non-allergic mucosal inflammation, rhinitis, and sinusitis (Illum 2012). Treatment with topical steroids remains inadequate in certain instances due to insufficient distribution to the nose and sinuses. This opens up the possibility of developing innovative topical medications with unique nasal delivery methods to treat diverse allergy diseases and increase patient compliance (Illum 2004).
4.4.4 Nasal Route for Brain Delivery
Treatment of CNS illnesses such as Alzheimer’s disease, depression, epilepsy, and schizophrenia has many obstacles with the existing standard administration technique due to therapeutic agent delivery constraints. This channel delivers drug molecules to the brain by circumventing the BBB. The amount of drug entering systemic circulation, as well as the accompanying side effects, can be reduced in this way (Bahadur and Pathak 2012; Upadhyay et al. 2011).
5 Drug Absorption Through Nasal Mucosa
The passage of the mucous membrane is the initial stage in medication absorption from the nasal cavity. This layer is easily penetrated by small, unaltered particles. Large or charged particles, on the other hand, may have difficulties crossing them. Because of environmental changes, structural alterations in the mucous layer are conceivable (i.e., pH, temperature, etc.).
5.1 Nasal Drug Delivery Technique Benefits and Drawbacks
Advantages are as follows:
-
Non-invasive,
-
Bypass the BBB,
-
Therapeutic agents do not need any modification,
-
Does not require any trained person for medication,
-
Avoidance of environmental conditions like chemical and enzymatic degradation of drugs,
-
Rapid,
-
Feasible and convenient route of delivery to CNS,
-
Prevents first-pass metabolism,
-
Allowing increased bioavailability (Jadhav et al. 2014).
Nasal delivery has a few disadvantages as follows:
-
Including nasal irritation,
-
Tissue toxicity,
-
The risk of local side effects,
-
Nasal congestion,
-
Large molecular weight drugs cannot be given (Jadhav et al. 2014).
5.2 Factors Influencing Nasal Drug Absorption
To have a systemic influence or to penetrate the brain a medicine or formulation must pass through various membranes or barriers when administered intranasally. Furthermore, the rate and extent of nasal drug absorption are influenced by the drug’s physicochemical properties and many more as shown in Fig. 19.8.
5.2.1 Biological Factors
5.2.1.1 Structural Features
The nasal cavity is divided into five sections anatomically. The respiratory mucosa is the primary site of medication absorption via the nasal route. Columnar cells, vasculature, and other defense systems comprise it. The nasal cavity is well supplied with vasculature, which gets the greatest quantity of lachrymal secretion, which is responsible for the cavity’s warmth and humidification.
5.2.1.2 Biochemical Changes
The nasal route was chosen over the oral route to increase bioavailability and prevent first-pass metabolism. Drugs are partially broken down by metabolic enzymes which are released into mucus. Enzymatic degradants include mono and di amino peptides, serine, and cysteine (Boddupalli et al. 2010).
5.2.2 Physiological Factors
Blood supply and neural regulation, nasal cavity pH, mucociliary clearance (MCC), and ciliary beat frequency are all physiological factors that influence nasal drug absorption (CBF).
5.2.3 Pathological and Environmental Conditions
Because all systems are interrelated, any change in the micro-environment in the nose, such as MCC, CBF, pH, or any slight alteration in the structure of the mucous layer, triggers a defensive reaction, reducing medicine bioavailability (Costantino et al. 2007).
-
Enzymatic degradation.
-
Transporters and efflux systems.
-
Effect of pathological conditions.
5.2.4 Formulation Factors
Drug transport is affected by numerous factors that are explained in the following:
5.2.4.1 Size and Molecular Weight
Transcellular passive diffusion, which comprises molecules moving across the cell membrane between cells, allows polar molecules with a molecular weight of less than 1 KD (Kilo Dalton) to travel across the membrane. Molecules bigger than 1 KD, on the other hand, prefer passive diffusion outside of the cell, transcytosis, or carrier-mediated transport. This route, however, is only partially penetrated.
5.2.4.2 Lipophilicity
Though mucous is polar due to its high-water content and nasal membrane underneath, it is lipophilic, allowing lipophilic medicines to enter transcellular and achieve fast bioavailability.
5.2.4.3 pH and Partition Coefficient
Drug absorption is also affected by pH and partition coefficient. HLB modification allows for the successful delivery of drugs with a wide pH range. Many researchers determined in the early 1980s that medications with unionized states have more absorption/penetration than those with ionized states. Unionized forms of drugs absorb better in a wide pH range (Kushwaha et al. 2011).
5.2.4.4 Viscosity/Density
Higher viscous liquids improve permeability by coating the CBF and MCC; drug retention duration is enhanced, and absorption is improved. For example, the incorporation of hydroxyl propyl methyl cellulose (HPMC) helped improve drug absorption by the nose by extending the therapeutic action of the formulation through the development of a high-viscosity gel (Chaturvedi et al. 2011).
5.2.4.5 Drug Distribution
A dosage form is essential in the field of application. When opposed to aerosols, nasal drops have a greater spreadability (coverage area) and a longer retention period on the mucosa. Typically, solutions produce dosage uniformity issues (Greimel et al. 2007).
5.2.4.6 Solubility
In rare cases, intranasal administration of drugs with limited water solubility and/or higher doses might be difficult (Greimel et al. 2007).
5.2.4.7 Pharmaceutical Form
Higher viscosity formulations may have difficulties in administration. Metered dosage pumps and actuators have recently become accessible, allowing nasal spray systems to provide precise doses ranging from 25 to 200 μL.
6 Nasal Drug Absorption Methodologies
As previously stated, nasally given drugs have a number of limitations, including low drug solubility, limited membrane penetration quick enzymatic degradation, and rapid MCC. Table 19.2 explains the challenges and possible solutions for bioavailability problems through the intranasal route.
7 Brain Disorders
7.1 Epilepsy
Seizures are neurological disorders caused by abnormally high neuronal activity in the brain. This aberrant brain activity can emerge in a variety of ways, ranging from convulsive activity to experiential events that are not easily observable by an observer, depending on the distribution of discharges. During a seizure, certain brain cells send out aberrant signals that prevent other cells from functioning normally. This anomaly may result in a brief shift in sensation, behavior, movement, or awareness (El-Zaafarany et al. 2018). Drugs used in epilepsy and their clinical status are given in Table 19.3.
7.2 Parkinson’s Disease
Parkinson’s disease is characterized by a loss of dopaminergic neurons in the brain’s substantia nigra pars compacta. It is classified as a mobility disorder. Cerebral palsy, ataxia, and Tourette syndrome are examples of movement problems. They happen when a shift in the neurological system impairs a person’s capacity to move or remain motionless. It affects the neurological system, and the symptoms worsen over time. It occurs when nerve cells in the brain do not create enough dopamine, a brain neurotransmitter. The bulk of unfavorable neuronal loss in Parkinson’s disease is caused by complex 1 defects in the respiratory chain. The clinical status of drugs used in PD is given in Table 19.4.
7.3 Psychosis
Patients suffering from psychosis have a distorted worldview in which they are unable to discern between their personal, subjective experiences, and the reality of the outside world. They have hallucinations or delusions that they feel are real, and they may behave and converse inappropriately and incoherently. Delusions, hallucinations, and disorganized speech are some of the symptoms. The clinical status of drugs used in psychosis is given in Table 19.5.
7.4 Schizophrenia
Schizophrenia is a diverse condition characterized by disruptions in language, perception, thought, social behavior, emotion, and violation. Symptoms of schizophrenia include hallucinations, confused ideas, and emotions of fear and paranoia. Psychiatrists evaluate symptoms, examinations, and medical history before prescribing drugs and psychotherapy. There are no pathogenomic characteristics. Table 19.6 depicts the clinical status of drugs used in schizophrenia.
7.5 Alzheimer Disease
Alzheimer’s disease (AD) is a progressive decline of mental capacities that begins with moderate memory loss and progresses to loss of ability to keep employment, plan and execute normal tasks, reason, and exercise judgment. Skills in communication, emotions, and attitude are all harmed. Alzheimer’s disease is the fourth leading cause of death in individuals, behind heart disease, cancer, and stroke. Alzheimer’s Association (Alzheimer’s Association 2017); Alzheimer’s disease causes cognitive changes that follow a regular pattern, starting with memory loss and advancing to language and visuospatial problems. The symptom starts with memory loss, such as forgetfulness, and progresses to more distressing losses. The patient may forget the names of family members or what was stated at the start of a sentence. Table 19.7 explains the clinical status of drugs used in Alzheimer’s disease.
8 Novel Formulation Approaches for Intranasal Drug Delivery
The pharmacological characteristics, delivery mechanism, and nasal physiology should all be researched in order to generate a nasal formulation with acceptable performance and commercial features. It is better to concentrate on extending the stay and ensuring optimal medicine absorption. Table 19.8 summarizes recent research on intranasal drug delivery devices in the treatment of brain diseases.
8.1 Mucoadhesive Solutions
Mucoadhesive solutions, such as chitosan, cellulose polymers, polycarbophil, poloxamers, and other mucoadhesive polymers, have been found to increase intranasal drug penetration. Gel formulations with the appropriate rheological properties lengthen the time in contact with the mucosa at the absorption site. The polymer in the gel has mucoadhesive properties as well as the formulation’s physicochemical characteristics, restricting removal through protective mechanisms in the nose and eyes (Ugwoke et al. 1999; Vidgren et al. 1992). Several studies have stated that mucoadhesive polymers such as cellulose derivatives, polyacrylates, starch, and so on can be used in the formulation of mucoadhesive solutions. (Gavini et al. 2012) and colleagues created a mucoadhesive formulation that demonstrated regulated dopamine release in the brain following intranasal application.
8.2 Microspheres
Microspheres are tiny spherical particles that range in size from 1 to 1000 mm in diameter. Microspheres are also called microparticles in some situations. Microspheres may be made from a variety of polymeric materials, both natural/synthetic, as well as inorganic materials.
Microspheres, particularly mucoadhesive microspheres, are a prominent type of novel drug delivery technology. Microspheres may stay in touch with the nasal mucosa for extended periods, resulting in increased drug absorption rate and volume (Ugwoke et al. 2005). Microspheres for nasal applications are typically made from biocompatible materials such as starch, albumin, hyaluronic acid, chitosan, and gelatin, as well as HPMC, Carbopol 934P, dextran, and other polymer combinations (Patil and Sawant 2011; Takeuchi et al. 2003).
A team of researchers created a comparison report between Methotrexate (MTX) solution and MTX-chitosan loaded microspheres, which revealed that MTX-loaded chitosan microspheres exhibited rapid and controlled drug release after intranasal administration compared to MTX solution due to their ability to transport more drug to the brain. Furthermore, chitosan employed as a mucoadhesive polymer was found to be safe and aid in medication mucoadhesion in the olfactory area, resulting in enhanced drug bioavailability (Sun et al. 2012).
8.3 Liposomes
Liposomes are spherical vesicles that have a hydrophilic core within and an impenetrable lipophilic phospholipid bilayer on the outside, similar to cells. They may entrap drugs in both aqueous and lipid phases due to their unique structure. Lipophilic drugs are nearly completely enclosed in the lipid bilayers of liposomes, and because they are not water-soluble, problems like drug loss during storage are rare. Hydrophilic drugs can be located in the watery cores of liposomes or the surrounding water phase.
Liposome nasal clearance half-life was four times longer than usual human nasal clearance half-life, showing liposomal systems’ mucoadhesion capabilities and nasal application potential (Jaafari et al. 2010; Andresen et al. 2005). Furthermore, liposomes have the benefit of being able to be changed to efficiently target a specific region of interest, such as the BBB. As a result, targeted liposomes have mostly been explored for the detection and therapy of brain illnesses (Mourtas et al. 2011).
Liposome research has yielded encouraging findings in drug delivery via the nasal transport system. Seok Hong and colleagues explored how surface modification of liposomes with targeting moieties, PEGylation methods, and the integration of various carrier systems with liposomes can boost drug bioavailability.
8.4 Nanoparticles
The drug substance is either encapsulated inside the matrix or conjugated or adsorbs to the particle surface in nanoparticles. To enable delayed, regulated drug therapy, nanoparticles are frequently comprised of polymers, lipids, or a mix of the two.
8.5 Exosomes
8.5.1 Natural Bioactivities of Exosomes
Exosomes are membrane nanovesicles that develop spontaneously and have a diameter of 50–150 nm (Simons and Raposo 2009). They came from the endosomes of immune cells such as dendritic cells (Théry et al. 2006), macrophages (Bhatnagar et al. 2007), B-cells (Clayton et al. 2005), and T-cells (Nolte’t Hoen et al. 2009), as well as mesenchymal stem cells, cancer cells, and endothelial cells.
8.6 Nanoemulsions
Nanoemulsions (NEs) are surfactant-stabilized oil-in-water (O/W) or water-in-oil (W/O) colloidal carriers of two immiscible liquids with a mean droplet diameter of around 100 nm, but maximum size limitations of up to 300 nm have been recorded in the literature. Because the droplet size is much smaller than the wavelength of visible light, NEs seem translucent or milky-white (Rodrigues et al. 2005).
NEs can be made into a number of dosage forms, such as liquids (Sarker 2005), creams, gels, foams, sprays, and so on, and can be taken orally, parenterally, ocularly, or nasally. Much study has been done on nanoemulsions that target the brain via the nasal route.
8.7 Polymeric Micelles
Polymeric micelles are nano-shell structures generated by the co-polymerization of hydrophobic and hydrophilic block copolymers. These micelles have the ability to operate and change their shape for medication delivery (Wang et al. 2012). The medication, which is present inside the brain and targets the hydrophobic core, is stabilized from the external aqueous environment by the hydrophilic shell, which increases polymer water solubility. Karami et al. (2019) investigated magnetic brain targeting of naproxen-loaded polymeric micelles in recent work. The findings indicated that polymeric magnetic micelles with diameters less than 150 nm might be a suitable carrier for improving therapeutic drug accumulation in the brain for the treatment of CNS illnesses.
8.8 Nanogels
Nanogels are nano-sized polymeric systems having cross-linked swellable polymeric hydrophilic chains that can withstand significant amounts of water when immersed in an aqueous media without dissolving. They have several qualities, including the capacity to be ready physically or chemically manipulated, good swelling properties, high encapsulation efficiency, high permeability, stability, and so forth.
This polymeric system has received extensive investigation, highlighting its critical role in brain targeting. Because of their site-specificity, nanogels offer a wide range of applications in the treatment of cancers and neurological illnesses. Various studies show that nanogels may be used to deliver oligonucleotides, tiny therapies, proteins, as contrast agents, optical imaging, multimodal imaging, and diagnostic agents, among other things (Cherry 2006).
The effectiveness of cCHP-based nanoemulsion as a universal protein-based antigen-delivery vehicle for adjuvant-free intranasal vaccination has been demonstrated (Nochi et al. 2010). These nanogels are made by self-assembly in water and trap a variety of proteins via hydrophobic interactions, displaying chaperone-like activity due to protein hydration without aggregation. They may then be gradually released back to their original condition (Desale et al. 2013).
8.9 Dendrimers
Dendrimers are a new type of polymer with well-defined structures created by the sequential addition of layers to the branching groups around the core molecule. This results in structural regularity and minimal heterogeneity, making them an appealing candidate for next-generation nanomedicines (Harush-Frenkel et al. 2008). Their unusual design boosts their utility as radioligands, detecting agents, and targeting components, among other things. Recent improvements demonstrate their great utility in gene transfer, diagnostic agents, contrast agents, photodynamic treatment, and other fields. (Zaman et al. 2011) created a polyacrylate dendritic polymer-based completely synthesized peptide sub-unit vaccination candidate.
8.10 Niosomes
Niosomes are intriguing new nanocarriers in which the medication is encapsulated in multi-lamellar structures generated by the self-association of nonionic surfactants. Niosomes are biocompatible, biodegradable, non-immunogenic, non-toxic, and non-carcinogenic, among other properties (Mahale et al. 2012; Seleci et al. 2016). The capacity of a nonionic surfactant to generate bilayer vesicles is determined by the surfactant’s HLB value. The functioning of niosomes is similar to that of liposomes in that they improve medication bioavailability by decreasing clearance.
9 Conclusion
Intranasal medicine delivery systems have several advantages over typical oral dosage methods. Several intranasal medicine delivery systems have been created, allowing medications to be administered directly to the brain through the nasal mucosa. Recent trends and clinical research suggest that novel carrier systems with improved bioavailability, efficiency, and fewer side effects should be used. The properties of the drug candidate, the nose-to-brain transport pathway, and transit to and within the brain must all be better understood.
References
Abbott NJ, Rönnbäck L, Hansson E et al (2006) Astrocyte–endothelial interactions at the blood–brain barrier. Nat Rev Neurosci 7:41 –53
Al Asmari AK, Ullah Z, Tariq M et al (2016) Preparation, characterization, and in vivo evaluation of intranasally administered liposomal formulation of donepezil. Drug Des Dev Ther 10:205
Alam MI, Beg S, Samad A et al (2010) Strategy for effective brain drug delivery. Eur J Pharm Sci 40:385 –403
Alam T, Pandit J, Vohora D et al (2015) Optimization of nanostructured lipid carriers of lamotrigine for brain delivery: in vitro characterization and in vivo efficacy in epilepsy. Expert Opin Drug Deliv 12:181 –194
Ali J, Ali M, Baboota S et al (2010) Potential of nanoparticulate drug delivery systems by intranasal administration. Curr Pharm Des 16:1644 –1653
Alzheimer's Association (2017) Alzheimer's disease facts and figures. Alzheimers Dement 13:325 –373
Andresen TL, Jensen SS, Jørgensen K (2005) Advanced strategies in liposomal cancer therapy: problems and prospects of active and tumor specific drug release. Prog Lipid Res 44:68 –97
Arisoy S, Sayiner O, Comoglu T (2020) In vitro and in vivo evaluation of levodopa-loaded nanoparticles for nose to brain delivery. Pharm Dev Technol 25:735 –747
Bahadur S, Pathak K (2012) Physicochemical and physiological considerations for efficient nose-to-brain targeting. Expert Opin Drug Deliv 9:19 –31
Barchet TM, Amiji MM (2009) Challenges and opportunities in CNS delivery of therapeutics for neurodegenerative diseases. Expert Opin Drug Deliv 6:211 –225
Begley DJ (2004) Delivery of therapeutic agents to the central nervous system: the problems and the possibilities. Pharmacol Ther 104:29 –45
Bhatnagar S, Shinagawa K, Castellino FJ et al (2007) Exosomes released from macrophages infected with intracellular pathogens stimulate a pro-inflammatory response in vitro and in vivo. Blood 110:3234 –3244
Bhattamisra SK, Shak AT, Xi LW et al (2020) Nose to brain delivery of rotigotine loaded chitosan nanoparticles in human SH-SY5Y neuroblastoma cells and animal model of Parkinson's disease. Int J Pharm 579:119 –148
Boddupalli BM, Mohammed ZN, Nath RA et al (2010) Mucoadhesive drug delivery system: an overview. J Adv Pharm Technol Res 1:381
Bozdağ Pehlivan S (2013) Nanotechnology-based drug delivery systems for targeting, imaging and diagnosis of neurodegenerative diseases. Pharmaceutical Research. 30:2499–2511
Brownlees J, Williams CH (1993) Peptidases, peptides, and the mammalian blood–brain barrier. J Neurochem 60:793 –803
Chang J, Jallouli Y, Barras A et al (2009) Drug delivery to the brain using colloidal carriers. Prog Brain Res 180:2 –17
Charlton S, Jones NS, Davis SS et al (2007) Distribution and clearance of bio-adhesive formulations from the olfactory region in man: effect of polymer type and nasal delivery device. Eur J Pharm Sci 30:295 –302
Chaturvedi M, Kumar M, Pathak K et al (2011) A review on mucoadhesive polymer used in nasal drug delivery system. J Adv Pharm Technol Res 2:215
Chen Y, Liu L (2012) Modern methods for delivery of drugs across the blood–brain barrier. Adv Drug Deliv Rev 64:640 –665
Cherry SR (2006) Multimodality in vivo imaging systems: twice the power or double the trouble? Annu Rev Biomed Eng 8:35 –62
Clayton A, Turkes A, Navabi H et al (2005) Induction of heat shock proteins in B-cell exosomes. J Cell Sci 118:3631 –3638
Costantino HR, Illum L, Brandt G et al (2007) Intranasal delivery: physicochemical and therapeutic aspects. Int J Pharm 337:1 –24
Czapp M, Bankstahl JP, Zibell G et al (2008) Brain penetration and anticonvulsant efficacy of intranasal phenobarbital in rats. Epilepsia 49:1142 –1150
Desai P, Shete H, Adnaik R et al (2015) Therapeutic targets and delivery challenges for Alzheimer's disease. World J Pharmacol 4:236 –264
Desale SS, Cohen SM, Zhao Y et al (2013) Biodegradable hybrid polymer micelles for combination drug therapy in ovarian cancer. J Control Release 171:339 –348
Djupesland PG (2013) Nasal drug delivery devices: characteristics and performance in a clinical perspective—a review. Drug Deliv Transl Res 3:42 –62
El-Zaafarany GM, Soliman ME, Mansour S et al (2018) A tailored thermosensitive PLGA-PEG-PLGA/emulsomes composite for enhanced oxcarbazepine brain delivery via the nasal route. Pharmaceutics 10:217
Eskandari S, Varshosaz J, Minaiyan M et al (2011) Brain delivery of valproic acid via intranasal administration of nanostructured lipid carriers: in vivo pharmacodynamic studies using rat electroshock model. Int J Nanomedicine 6:363
Fang JY, Hung CF, Chi CH et al (2009) Transdermal permeation of selegiline from hydrogel-membrane drug delivery systems. Int J Pharm 380:33 –39
Fazil M, Md S, Haque S et al (2012) Development and evaluation of rivastigmine loaded chitosan nanoparticles for brain targeting. Eur J Pharm Sci 47:6 –15
Fine JM, Renner DB, Forsber AC et al (2015) Intranasal deferoxamine engages multiple pathways to decrease memory loss in the APP/PS1 model of amyloid accumulation. Neurosci Lett 584:362 –367
Gabathuler R (2010) Approaches to transport therapeutic drugs across the blood–brain barrier to treat brain diseases. Neurobiol Dis 37:48 –57
Gangurde PK, Ajitkumar BN, Kumar L (2019) Lamotrigine lipid nanoparticles for effective treatment of epilepsy: a focus on brain targeting via nasal route. J Pharm Innov 14:91 –111
Gavini E, Rassu G, Ciarnelli V et al (2012) Mucoadhesive drug delivery systems for nose-to-brain targeting of dopamine. J Neurosci Res 2:47 –55
Greimel A, Bernkop-Schnürch A, Dorly Del Curto M et al (2007) Transport characteristics of a beta sheet breaker peptide across excised bovine nasal mucosa. Drug Dev Ind Pharm 33:71 –77
Groothuis DR (2000) The blood-brain and blood-tumor barriers: a review of strategies for increasing drug delivery. J Neuro-Oncol 2:45 –59
Guo JW, Guan PP, Ding WY et al (2017) Erythrocyte membrane-encapsulated celecoxib improves the cognitive decline of Alzheimer's disease by concurrently inducing neurogenesis and reducing apoptosis in APP/PS1 transgenic mice. Biomaterials 145:106 –127
Hallschmid M, Benedict C, Schultes B et al (2008) Obese men respond to cognitive but not to catabolic brain insulin signaling. Int J Obes 32:275 –282
Hanafy AS, Farid RM, Helmy MW (2016) Pharmacological, toxicological and neuronal localization assessment of galantamine/chitosan complex nanoparticles in rats: future potential contribution in Alzheimer's disease management. Drug Deliv 23:3111 –3122
Harush-Frenkel O, Rozentur E, Benita S et al (2008) Surface charge of nanoparticles determines their endocytic and transcytotic pathway in polarized MDCK cells. Biomacromolecules 9:435 –443
Illum L (2004) Is nose-to-brain transport of drugs in man a reality? J Pharm Pharmcol 56:3 –17
Illum L (2012) Nasal drug delivery—recent developments and future prospects. J Control Release 161:254 –263
Jaafari MR, Tafaghodi M, Sa ST (2010) Evaluation of the clearance characteristics of liposomes in the human nose by gamma-scintigraphy. Iran J Pharm Res 20:3 –11
Jadhav AJ, Gondkar SB, Ravindra BS (2014) A review on nasal drug delivery system. WJPPS 3:231 –254
Jain KK (2012) Nanobiotechnology-based strategies for crossing the blood–brain barrier. Nanomedicine 7:1225 –1233
Jain R, Nabar S, Dandekar P (2010) Micellar nanocarriers: potential nose-to-brain delivery of zolmitriptan as novel migraine therapy. Pharm Res 27:655 –664
Jouyban A, Soltani S (2012) Blood brain barrier permeation. Toxic Drug Test 10:1 –24
Karami Z, Sadighian S, Rostamizadeh K et al (2019) Magnetic brain targeting of naproxen-loaded polymeric micelles: pharmacokinetics and biodistribution study. Mater Sci Eng C 100:771 –780
Katare YK, Daya RP, Sookram GC et al (2015) Brain targeting of a water insoluble antipsychotic drug haloperidol via the intranasal route using PAMAM dendrimer. Mol Pharm 12:3380 –3388
Katare YK, Piazza JE, Bhandari J et al (2017) Intranasal delivery of antipsychotic drugs. Schizophr Res 184:2 –13
Khosrow Tayebati S, Ejike Nwankwo I, Amenta F (2013) Intranasal drug delivery to the central nervous system: present status and future outlook. Curr Pharm Des 19:510 –526
Krol S (2012) Challenges in drug delivery to the brain: nature is against us. J Control Release 164:145 –155
Kumar M, Misra A, Babbar AK et al (2008) Intranasal nanoemulsion based brain targeting drug delivery system of risperidone. Int J Pharm 358:285 –291
Kushwaha SK, Keshari RK, Rai AK et al (2011) Advances in nasal trans-mucosal drug delivery. J Appl Pharm Sci 1:21
Lesniak MS, Brem H (2004) Targeted therapy for brain tumours. Nat Rev Drug Discov 3:499 –508
Lochhead JJ, Thorne RG (2012) Intranasal delivery of biologics to the central nervous system. Adv Drug Deliv Rev 64:614 –628
Löscher W, Potschka H (2005) Blood-brain barrier active efflux transporters: ATP-binding cassette gene family. NeuroRx 2:86 –98
MacDonald BK, Cockerell OC, Sander JW et al (2000) The incidence and lifetime prevalence of neurological disorders in a prospective community-based study in the UK. Brain 123:665 –676
Mahale NB, Thakkar PD, Mali RG et al (2012) Niosomes: novel sustained release nonionic stable vesicular systems—an overview. Adv Colloid Interf Sci 183:46 –54
Mandpe L, Kyadarkunte A, Pokharkar V (2013) Assessment of novel iloperidone- and idebenone-loaded nanostructured lipid carriers: brain targeting efficiency and neuroprotective potential. Ther Deliv 4:1365 –1383
Md S, Haque S, Fazil M et al (2014) Optimised nanoformulation of bromocriptine for direct nose-to-brain delivery: biodistribution, pharmacokinetic and dopamine estimation by ultra- HPLC/mass spectrometry method. Expert Opin Drug Deliv 11:827 –842
Meng Q, Wang A, Hua H et al (2018) Intranasal delivery of Huperzine a to the brain using lactoferrin-conjugated N-trimethylated chitosan surface-modified PLGA nanoparticles for treatment of Alzheimer’s disease. Int J Nanomedicine 13:705
Mischley LK, Conley KE, Shankland EG et al (2016) Central nervous system uptake of intranasal glutathione in Parkinson’s disease. Parkinson's Dis 2:1
Misra A, Ganesh S, Shahiwala A et al (2003) Drug delivery to the central nervous system: a review. J Pharm Pharm Sci 6:252 –273
Mittal D, Md S, Hasan Q et al (2016) Brain targeted nanoparticulate drug delivery system of rasagiline via intranasal route. Drug Deliv 23:130 –139
Mourtas S, Canovi M, Zona C et al (2011) Curcumin-decorated nanoliposomes with very high affinity for amyloid-β1-42 peptide. Biomaterials 32:1635 –1645
Musumeci T, Serapide MF, Pellitteri R et al (2018) Oxcarbazepine free or loaded PLGA nanoparticles as effective intranasal approach to control epileptic seizures in rodents. Eur J Pharm Biopharm 133:309 –320
Nasr M (2016) Development of an optimized hyaluronic acid-based lipidic nanoemulsion co- encapsulating two polyphenols for nose to brain delivery. Drug Deliv 23:1444 –1452
Nau R, Sörgel F, Eiffert H (2010) Penetration of drugs through the blood-cerebrospinal fluid/blood-brain barrier for treatment of central nervous system infections. Clin Microbiol Rev 23:858 –883
Nochi T, Yuki Y, Takahashi H et al (2010) Nanogel antigenic protein-delivery system for adjuvant-free intranasal vaccines. Nat Mater 9:572 –578
Noetzli M, Eap CB (2013) Pharmacodynamic, pharmacokinetic and pharmacogenetic aspects of drugs used in the treatment of Alzheimer's disease. Clin Pharmacokinet 52:225 –241
Nolte’t Hoen EN, Buschow SI, Anderton SM et al (2009) Activated T cells recruit exosomes secreted by dendritic cells via LFA-1. Blood 113:1977 –1981
Ozsoy Y, Gungor S, Cevher E (2009) Nasal delivery of high molecular weight drugs. Molecules 14:3754 –3779
Pailla SR, Talluri S, Rangaraj N et al (2019) Intranasal zotepine nanosuspension: intended for improved brain distribution in rats. Daru 27:541 –556
Pardridge WM (1983) Brain metabolism: a perspective from the blood-brain barrier. Physiol Rev 63:1481 –1535
Pardridge WM (2006) Molecular trojan horses for blood–brain barrier drug delivery. Curr Opin Pharmacol 6:494 –500
Patel P, Acharya S, Acharya N (2011) Potential of surface functionalized nanoparticles for improved therapy of refractory central nervous system disorders. Pharm Res 4:1093 –1099
Patel MR, Patel RB, Bhatt KK et al (2016a) Paliperidone microemulsion for nose-to-brain targeted drug delivery system: pharmacodynamic and pharmacokinetic evaluation. Drug Deliv 23:346 –354
Patel RB, Patel MR, Bhatt KK et al (2016b) Evaluation of brain targeting efficiency of intranasal microemulsion containing olanzapine: pharmacodynamic and pharmacokinetic consideration. Drug Deliv 23:307 –315
Patel RB, Patel MR, Bhatt KK et al (2016c) Micro-emulsion-based drug delivery system for transnasal delivery of carbamazepine: preliminary brain-targeting study. Drug Deliv 23:207 –213
Pathak R, Dash RP, Misra M et al (2014) Role of mucoadhesive polymers in enhancing delivery of nimodipine microemulsion to brain via intranasal route. Acta Pharm Sin B 4:151 –160
Patil SB, Sawant KK (2011) Chitosan microspheres as a delivery system for nasal insufflation. Colloids Surf B Biointerfaces 84:384 –389
Perez AP, Mundiña-Weilenmann C, Romero EL et al (2012) Increased brain radioactivity by intranasal 32P-labeled siRNA dendriplexes within in situ-forming mucoadhesive gels. Int J Nanomedicine 7:1373
Picone P, Sabatino MA, Ditta LA, Amato A et al (2018) Nose-to-brain delivery of insulin enhanced by a nanogel carrier. J Control Release 270:23 –36
Pokharkar V, Suryawanshi S, Dhapte-Pawar V (2020) Exploring micellar-based polymeric systems for effective nose-to-brain drug delivery as potential neurotherapeutics. Drug Deliv Transl Res 10:1019 –1031
Ravikrishna V, Janapareddi K (2019) Development and evaluation of clozapine intranasal mucoadhesive in situ gels for brain targeting. JDDT 9:198 –207
Ricci M, Blasi P, Giovagnoli S et al (2006) Delivering drugs to the central nervous system: a medicinal chemistry or a pharmaceutical technology issue? Curr Med Chem 13:1757 –1575
Rodrigues RF, Costa IC, Almeida FB et al (2005) Development and characterization of evening primrose (Oenothera biennis) oil nanoemulsions. Rev Bras Farmacogn 25:422 –425
Sanchez-Covarrubias L, Slosky LM, Thompson BJ et al (2014) Transporters at CNS barrier sites: obstacles or opportunities for drug delivery? Curr Pharm Des 20:1422 –1449
Sarker DK (2005) Engineering of nanoemulsions for drug delivery. Curr Drug Deliv 2:297 –310
Sauer I, Dunay IR, Weisgraber K et al (2005) An apolipoprotein E-derived peptide mediates uptake of sterically stabilized liposomes into brain capillary endothelial cells. Biochemistry 44:2021 –2029
Scarff JR, Casey DA (2011) Newer oral atypical anti-psychotic agents: a review. Pharm Ther 36:832
Seleci M, Seleci DA, Joncyzk R et al (2016) Smart multifunctional nanoparticles in nanomedicine. Bionanomaterials 17:33 –41
Shaji J, Poddar A, Iyer S (2009) Brain-targeted nasal clonazepam microspheres. Indian J Pharm Sci 71:715
Simons M, Raposo G (2009) Exosomes–vesicular carriers for intercellular communication. Curr Opin Cell Biol 21:575 –581
Stevens J, Ploeger BA, Van der Graaf PH et al (2011) Systemic and direct nose-to-brain transport pharmacokinetic model for remoxipride after intravenous and intranasal administration. Drug Metab Dispos 9:2275 –2282
Sun Y, Shi K, Wan F (2012) Methotrexate-loaded microspheres for nose to brain delivery: in vitro/in vivo evaluation. J Drug Deliv Sci Technol 22:167 –174
Takeuchi H, Matsui Y, Yamamoto H et al (2003) Mucoadhesive properties of carbopol or chitosan-coated liposomes and their effectiveness in the oral administration of calcitonin to rats. J Control Release 86:235 –242
Talegaonkar S, Mishra PR (2004) Intranasal delivery: an approach to bypass the blood brain barrier. Indian J Pharmacol 36:140
Théry C, Amigorena S, Raposo G et al (2006) Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr Protoc Cell Biol 30:3 –22
Ugwoke MI, Exaud S, Van Den Mooter G et al (1999) Bioavailability of apomorphine following intranasal administration of mucoadhesive drug delivery systems in rabbits. Eur J Pharm Sci 9:213 –219
Ugwoke MI, Agu RU, Verbeke N et al (2005) Nasal mucoadhesive drug delivery: background, applications, trends and future perspectives. Adv Drug Deliv Rev 57:1640 –1665
Upadhyay S, Parikh A, Joshi P et al (2011) Intranasal drug delivery system-a glimpse to become maestro. J Appl Pharm Sci 1:34 –44
Vidgren P, Vidgren M, Arppe J et al (1992) In vitro evaluation of spray-dried mucoadhesive micropheres for nasal administration. Drug Dev Ind Pharm 18:581 –597
Vyas TK, Shahiwala A, Marathe S et al (2005) Intranasal drug delivery for brain targeting. Curr Drug Deliv 2:165 –175
Wang Y, Wang C, Gong C et al (2012) Polysorbate 80 coated poly (ɛ-caprolactone)–poly (ethylene glycol)–poly (ɛ-caprolactone) micelles for paclitaxel delivery. Int J Pharm 434:1 –8
World Health Organization (2006) Neurological disorders: public health challenges. World Health Organization, Geneva
Xiao G, Gan LS (2013) Receptor-mediated endocytosis and brain delivery of therapeutic biologics. Int J Cell Biol 2013:703545 . https://doi.org/10.1155/2013/703545; Epub 2013 Jun 11. PMID: 23840214; PMCID: PMC3693099
Yang ZZ, Zhang YQ, Wang ZZ et al (2013) Enhanced brain distribution and pharmacodynamics of rivastigmine by liposomes following intranasal administration. Int J Pharm 452:344 –354
Zaman M, Skwarczynski M, Malcolm JM et al (2011) Self-adjuvanting polyacrylic nanoparticulate delivery system for group a streptococcus (GAS) vaccine. Nanomedicine 7:168 –173
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Ethics declarations
None of the authors have any conflict of interest.
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Arora, D., Bhatt, S., Kumar, M., Gautam, R.K., Taneja, Y., Chauhan, M. (2023). Significance of Intranasal Drug Delivery Systems: Recent Trends and Clinical Investigations in Brain Disorders. In: Singh, P.P. (eds) Recent Advances in Pharmaceutical Innovation and Research. Springer, Singapore. https://doi.org/10.1007/978-981-99-2302-1_19
Download citation
DOI: https://doi.org/10.1007/978-981-99-2302-1_19
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-99-2301-4
Online ISBN: 978-981-99-2302-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)