Abstract
In the current scenario of the emerging nation, the development of edible food packaging in a laboratory or commercial scale has immensely attracted the growing manufacturers. Based on this, the current chapter addresses various targeted biomaterials and synthesis strategies for fabricating edible food packaging. The chapter introduces the characteristics attributes of available biomaterials including polysaccharides, proteins, lipids, fats, and their modification strategies to overcome the existing shortcomings of biopolymers such as poor water solubility, inferior mechanical properties, hydrophilicity, and others in edible food packaging. Further, the use of nanomaterials in edible films and coatings has provided additional benefits in obtaining improved product quality and shelf life. In this regard, a brief overview of using several nanosystems-assisted edible food packaging such as polymeric nanoparticles, inorganic nanofillers, biocomposites, nanoencapsulation, and nanoemulsion has been made. However, the successful development of edible food packaging and its commercialization depends on both selected materials and processing techniques. The edible food packaging is generally availed in the form of edible films and coatings being developed using different approaches with existing benefits and shortcomings. The various processes for fabricating edible coatings including dipping, spraying (conventional and electrospraying), dripping, foaming, fluidized bed coating, brushing, panning, etc., have been detailed. Additionally, the adopted methods for fabricating edible films are solution casting, extrusion, compression molding, blown films, etc. The selection of processing techniques for the development of edible food packaging is dependent on batch size, targeted food products, food properties, geometry of food products, and other attributes. Thus, both the materials property and synthesis strategies play a major role in developing successful edible food packaging materials for highly perishable food products and others.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
2.1 Introduction
From very past years, edible food packaging being a kind of green packaging materials with eatable nature has attained great interest in food packaging industries with additional benefits of using natural resources. As discussed in the previous chapter, the “edible food packaging” is generally availed in the form of “edible films” or “edible coatings” to provide value-added food products with improved quality and shelf life. Among available packaging, the edible food packaging helps in keeping the food product safe and fresh from growing bacteria and spoilage. Additionally, the edible packaging incorporating active agents is one of the well-known packaging concepts in order to control the growth of unwanted microorganisms on the surfaces of the food. However, most of the food packaging products are wrapped in packaging, which are non-biodegradable in nature and create several environmental problems. Further, the use of biodegradable-based edible films for packaging food has been designed in a way to minimize health hazards and global transformation. The main objective of edible packaging films is to protect the food from microorganisms and preserve the foodstuffs. The edible films have been developed using many biodegradable materials targeting several applied areas such as oral-disintegrating films, edible oven bags, active packaging, wraps, fruit and vegetable leathers. The targeted packaging based on specific application is developed based on the selected materials and processing techniques. The successful development of edible coating depends on several factors such as employed method and properties of coating materials such as category, quantity, surface property, and chemical compositions. In this regard, the chapter discusses several biomaterials and synthesis strategies for developing edible food packaging for different applications.
The various renewable resources used in edible food packaging include polysaccharide, protein, lipid, and others, which are availed from several renewable resources [1]. The extensively available commercial edible packaged foods in terms of edible films or coatings developed from renewable resources are ice-cream cone, commercial waxing on fruits, collagen casing, chocolate coatings, cellulose ether-based water-soluble pouches, gelatin-based coatings, and others. The mentioned biomaterials are obtained from several available renewable sources. Interestingly, the used renewable resources for edible food packaging are also extracted from waste materials such as fruit peel, banana plant waste, sugarcane bagasse, corn stover, and sea-based waste. In this way, the utilization of natural resources in edible food packaging can be a strategic way to convert “waste into wealth” and further will help to reduce waste materials. However, the success of edible food packaging for obtaining in prolonging the shelf life of food products also depends on the structure, chemical composition, and film-forming properties of selected polymers, storage conditions, and the surface interaction (wettability and spreading coefficient) between fruit and coating materials. Additionally, the edible-coating process is also dependent on several factors including materials composition, surface morphology, geometry of product, etc. In this regard, the inclusion of nanosystem-assisted technique in developing edible coating has several advantages such as transportation of bioactive compounds to food products [2]. The application of nanotechnology in developing edible food packaging involves polymeric nanoparticle, nanocomposite, nanoencapsulation, nanoemulsion, inorganic nanocomposite, etc. The nanosystem can also transfer active components to food components in a controlled manner. The biopolymeric nanostructured materials are available in different morphologies such as nanocrystal form, nanofiber form, nanoparticles, and others, which deliver tailored-made properties of edible-coating materials. In this regard, an overview of the available nanostructured form that is utilized in developing edible food packaging materials has been discussed in the current chapter.
There are several strategies which are employed to develop edible films and coatings, where the selection of processing techniques is based on several factors such as targeted food products, quantity, and food property. Additionally, the processing strategies are applied to develop edible food packaging for food products as the technique is gaining interest as the packaging material can be eaten with the food products. Moreover, the consumer demands for ready-to-eat, high quality, safe and convenient food, which provide a great need in developing the large-scale production or commercialization of edible food packaging. However, the, challenging factors in edible food packaging are process equipment, food product to be packaged, and processing conditions, where several processings are suitable for different product types.
Based on the above discussion, the current chapter discusses the available biopolymeric materials and the modified forms to be utilized in developing edible food packaging. The edible biopolymeric materials such as polysaccharides (cellulose, chitosan, starch, glycogen, pectin, etc.), proteins, and lipids are used for developing edible food packaging. A detailed overview relating these biopolymeric materials, their modification strategies to overcome the existing shortcomings of materials such as hydrophilic property of some biopolymers, poor mechanical property, and low water solubility. In this regard, the chemical modifications and development of biocomposites and blends can modify the properties of biopolymers to obtain enhanced shelf life of food commodities. Further, a brief overview of the nanostructured materials used for the fabrication of edible food packaging has been done. The chapter also details several synthesis approaches for developing edible coating and films on food products. The processing steps that are adopted to develop edible-coated food products and edible films will be summarized in the subsequent section with their advantageous properties and associated shortcomings.
2.2 Biomaterials in Edible Food Packaging
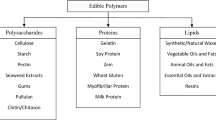
The principal components of edible packaging commonly include polysaccharides, proteins, and lipids-based materials. Further, the categories of biomaterials for developing edible food packaging are represented in Fig. 2.1. Besides, the other components of edible food packaging including the biopolymeric materials are (i) puree, juices obtained from fruits and vegetables; (ii) inorganic materials; (iii) natural fibers, nanomaterials as filler materials; (iv) alcohol and sugars as plasticizers; (v) antimicrobials, antioxidants, nutraceuticals, probiotics as functional additives; and (vi) browning inhibitors and cross-linking agents as additives. The used materials in edible packaging are biodegradable, reduce the solid waste management problems, and do not require any mode of recycling. The main characteristic features of edible food packaging (Fig. 2.2) include several attributes such as (i) packaging properties (barrier, mechanical, and thermal properties), (ii) sustainability developed from renewable and biodegradable materials, (iii) provide improved shelf life for offering low moisture content, low water activity, good stability, (iv) active functions such as antibacterial, antifungal, antioxidant, (v) sensory attributes such as texture, color, and flavor, and (vi) health benefits such as food fortification with nutraceuticals and others.
2.3 Polysaccharides in Edible Food Packaging
Polysaccharides are a class of carbohydrates consisting of chains of monosaccharide units linked together by glycosidic bonds. The other classes of carbohydrates are termed as monosaccharides and oligosaccharides consisting of single and 2–10 monosaccharide units, respectively. The polysaccharides are classified based on various factors such as repeating units, sources, and origin. The polysaccharides based on origin (as shown in Fig. 2.1) are categorized as (i) animal-based origins: chitosan (CS)/chitin, hyaluronic acid, and chondroitin sulfate obtained from marine wastes such as cartilage, fish eyeballs, and crustacean shells); (ii) microbial origin: xanthan gum, dextran; (iii) marine origin: agar, alginic acid, carrageenan; (iv) plant origin: starch (ST), cellulose, gum arabic, gum karaya, and guar gum. Additionally, the polysaccharides are also classified into main three types such as storage polysaccharides (ST, glycogen, and inulin), structural polysaccharides (cellulose, chitin, pectin), and mucosubstances (mucopolysaccharides). The classes of polysaccharides based on structure include linear and branched polysaccharides. The linear polysaccharides include cellulose, amylose, algin, pectin, and the branched polysaccharides include amylopectin, gum arabic, galactomannans, xanthan, and xylan. The several kinds of polysaccharides are considered as potential candidates to be used in edible food packaging (edible films and coatings) as biocomposites and blends. The presence of hydroxyl groups and other hydrophilic moieties in the chemical structure of polysaccharides serves a significant role in the film formation. In this regard, the chemical structure of several polysaccharides and their derivatives are represented in Table 2.1. The polysaccharides are also used in both edible and non-edible food packaging preparation as matrix and filler materials. Additionally, the polysaccharide-based films exhibit better adherence property to cut fruit surfaces and also have good gas barrier properties [3]. However, the low water resistance property, mechanical property, and cost effectiveness are the main drawbacks of polysaccharide-based materials to be used in food packaging. The structure of polysaccharides can be tuned to improve their physicochemical properties by salt addition, solvent change, pH change, and chemical modification of hydroxyl groups. In this regard, the composite preparation of hydroxypropyl methylcellulose (HPMC) reinforcing CS/sodium tripolyphosphate (STPP) nanoparticles can provide an improved barrier, thermal, and mechanical properties [4]. The STPP acts as a cross-linking agent in developing CS/STPP-based nanoparticles via ionic cross-linking process (at lower pH). Besides, there are several strategies are applied to modify the properties of polysaccharides to act as a better reinforcing agent to develop composite materials.
2.3.1 Cellulose and Its Derivatives
Cellulose is a class of structural polysaccharide, consisting of β (1 → 4) linked D-glucopyranose units which are extracted abundantly from plant -based sources and algae-based sources. Cellulose is the most abundantly available biopolymeric material in earth and is considered a potential candidate in developing edible films and coatings [5]. In 1838, cellulose was isolated for the first time from plant matter by Anselme Payen, a French chemist [6]. In 1870, the first successful development of celluloid, a cellulose-based thermoplastic polymer was obtained by Hyatt Manufacturing Company. These are insoluble in various aqueous solutions due to their rigid crystalline structure imparted due to the presence of hydroxyl groups (OH) between and within the strands. When the hydroxyl groups on the glycosyl units are substituted with the bulkier groups, it helps in separating the polymer chains of the crystalline structure by breaking the H-bonding. Cellulose, when untreated is insoluble in water but when chemically treated with alkali and further reacts with propylene oxide, methyl chloride, etc., become water soluble. As represented in Table 2.2, the forms of cellulose derivatives are carboxymethyl cellulose (CMC), methylcellulose (MC), ethylcellulose (EC), hydroxypropyl cellulose (HPC), HPMC, etc. These forms of cellulose ethers are the polymer substances that are formed by partially substituting OH groups with ether functionalities. These forms are water soluble and are used to develop edible polymer films having properties of transparency, odorless, tough, and flexible. These are usually poor water resistant, however provide resistant to fats and oils. Further, their hydrophilicities are arranged in the order of HPC < MC < HPMC < CMC. Interestingly, cellulose derivatives in combination with nanostructured cellulose materials are also used to develop food packaging materials with better properties [7]. Besides, MCC is also extensively utilized for developing edible films and coatings in combination with other biopolymeric materials for improved attributes. The cellulose microcrytsallites from cellulose can be developed via depolymerization technique which on drying provide MCC (Fig. 2.3a). The FESEM micrograph of MCC has been represented in Fig. 2.3b. The MCC as a reinforcement has several attributes to be used in edible food packaging applications such as mechanical property, water retaining property, surface area, and biological property.
-
Methylcellulose (MC). It is obtained from cellulose derived plant-based sources such as cotton, jute, bamboo, and pulps of plants. These are more biodegradable in nature which is produced by chemical treatments using alkali concentrated NaOH solution and further treating with methyl halide. The produced films from MC have application in the food packaging industry due to their low cost, transparency, high strength, easy processability, excellent film-forming properties, etc. [8, 9]. These polysaccharides are composed of linear chains of β (1 → 4) glycosidic units consisting of methyl substituent replacing the all or part of the three hydroxyl groups on cellulose, which imparts the films with sufficient oxygen and barrier property [9]. For many years, MC has been used to produce fine chemicals for the pharmaceuticals, packaging, civil construction, petrochemical industry, paints, adhesives, cosmetic industries, and others [10]. Also, it has been observed that MC has many application as emulsifiers, viscosity, and flow controllers. Due to the hydrophilic nature of MC, it has been used as an active material to protect the food from oxidation or food spoilage and further, which improves the shelf life. Further, nanoemulsions (food-grade) based on cellulose, its derivative, and essential oils can be used to prepare edible packaging and others to improve the quality and shelf-life of foods, which further helps in the delivery of functional compounds and antimicrobial agents into the food products [11]. Otoni et al. reported the fabrication of clove bud or oregano oil emulsified MC-based films, which provide antimicrobial property against spoilage fungi and further help in maintaining quality and shelf life of sliced bread [12]. Interestingly, MC-based films are more effective to be used as a coating for deep fat fried potato strips in order to minimize the oil uptake in comparison with HPMC films [13]. The addition of plasticizers such as sorbitol with cellulose derivatives may provide additional benefits in comparison with individual cellulose derivative films. The combination of modifiers such as propylene oxide and methyl chloride can be useful to generate polysaccharides with multiple modifications, viz. HPMC. In this regard, edible films based on HPMC composites using microcrystalline cellulose (MCC) nanoparticles are also developed to provide improved moisture barrier properties in comparison with neat HPMC films [14].
-
Ethylcellulose. In the food packaging applications, researchers are now focused mainly on biodegradable films. Ethylcellulose (EC) is one of the cellulose derivatives consisting of repeating structure of anhydrous glucose units with ethyl ether groups replacing some of the hydroxyl sites. It has attracted attention in the food and pharmaceutical sectors. Kaur and Yadav et al. prepared polymeric films for food packaging by using various ratio of EC and polyethylene glycol (PEG) as a plasticizer. The addition of PEG at a different ratio to EC film provides tailored property, where the fabrication of films with EC: PEG in the ratio of 2:1 has good flexibility and collapsibility providing the suitability for food packaging application. Further, the EC and PEG-based films can also withstand a maximum load of 2.65 kN for a long time showing good mechanical stability [15]. The EC-based edible coating is helpful in reducing the moisture loss from packaged meat in comparison with the agar-based coatings (considerable thickness: 0.8–0.9 mm) [16]. Moreover, acetylated glycerol monostearate coatings are slightly more water vapor permeable in comparison with EC, whereas the specific coatings based on acetylated glycerol monostearate provide less oxygen permeability in comparison with EC films [17]. Furthermore, the association of several other biopolymeric materials such as proteins and lipids can be added to cellulose derivatives to develop composite-based films with improved properties [18]. Romero-Bastida et al. prepared zein–EC films with the aid of plasticizer stearic acid (SA), which showed the lowest water sorption property in comparison with pure EC and zein films. Further, the biodegradability behavior of zein and EC film shows significant changes, where 100% zein-based films provide highest biodegradability [19]. Moreover, EC, having the properties of water insolubility, excellent membrane forming ability, drug release property, is used as films and microspheres in pharmaceutical purposes [20]. Li et al. prepared blends of EC/konjac glucomannan blend films provide increased thermal stability, moisture resistance, tensile strength, and elongation at break in comparison with the konjac glucomannan films [21]. In the year 2010, Guiga et al. designed multilayered nisin-loaded EC/HPMC/EC films using lecithin, a surface-active plasticizers, which impart antimicrobial activity to improve product packaging property [22]. Moreover, EC-based delivery systems have been studied which could serve as a potential for producing healthy and low calorie formulations at the industrial level.
-
Hydroxypropyl Cellulose (HPC). HPC is another cellulose derivative used for developing edible films or coating besides MC, CMC, and HPMC [23]. It is particularly used as the main component for multicomponent dispersion-coating applications. It is also used for dispersion-coating formulations for sustainable packaging applications with excellent convertibility with a grease barrier. It acts as a barrier for oil resistance and also the presence of additives like talc reduces the stickiness of the coated layer. The casted films made from MC, HPMC, HPC, and CMC generally have moderate strength, flexible, resistant to oil and fats, transparent, tasteless, and water soluble, and are moderate barriers to oxygen. The edible coatings made out of CMC, MC, HPC, and HPMC for various fruits serve as a barrier to oil and moisture transfer.
-
Hydroxypropyl Methylcellulose (HPMC). HPMC, also known as hypromellose, refers to the group of cellulosic ethers in which the substitution of the hydroxyl groups with methyl and hydroxyl propyl groups is done with hydroxyl groups that are present in the cellulose ring. The hydrophilic, biodegradable, and biocompatible nature of the HPMC makes it applicable in various fields such as in drug delivery, dyes, cosmetics, adhesives, coatings, agriculture, and textiles. It is a potential replacement for various synthetic plastics because of its excellent film-forming properties, flexibility, biodegradability, transparency, and its gas barrier properties. It is soluble in polar organic solvents both aqueous and non-aqueous solvents. It has a distinctive solubility property in both hot and cold organic solvents. As compared to other cellulosic derivatives, HPMC possesses increased thermoplasticity and organo-solubility with a gelation temperature of 75–90 ℃. Being biodegradable in nature, this type of polymer creates composites that are easily processable, eco-friendly in nature, and deliver desirable properties. Also, numerous studies have also been carried out to study the impact of various additives on the physicochemical properties of HPMC. The incorporation of antimicrobial agents such as nisin and potassium sorbate to tapioca ST and HPMC-based edible films provides an active packaging, where the combined use of ST and HPMC delivers increased elastic modulus and stress [24]. Additionally, the combined use of nisin and potassium sorbate is more advantageous in providing inhibitory spectrum. Further, studies based on edible films from HPMC with saturated and unsaturated fatty acids such as lauric acids (LA), myristic acid (MA), palmitic acid (PA), SA, and oleic acid (OA) provide tailored-made film properties in terms of microstructural and physical properties [25]. The addition of the film microstructure affects the several film properties including water barrier, mechanical, and optical property. The bigger lipid micellar structures are obtained in HPMC (aqueous) system when LA, MA, and PA are used in comparison with SA and OA. Besides, the HPMC films due to their hydrophilic nature have a shortcoming of providing lower moisture barrier properties. Thus, incorporation of lipids such as beeswax in the HPMC film increases the effectiveness toward water barrier properties, and it further helps to reduce the water vapor permeability (WVP), delivering an application of HPMC-lipid composite-coated papers in food packaging areas [26]. Polysaccharides and lipids-based coated papers are extensively used to develop packaging in food industry. The addition of carrageenan, plasticizers, and carnauba wax emulsions can improve the properties of HPMC-based edible films such as rheology and water vapor properties [27]. Also, the moisture barrier of any HPMC film can be improved by incorporation of SA in the film-forming solution. For the preparation of a composite film with both hydrophobic and hydrophilic compounds, chemical modification improves the hydrophobicity of the film. In this manner, a homogeneous packaging film can be prepared. Again, the cross-linking of HPMC can be another approach to produce biodegradable packaging materials with improved water vapor barrier properties [28]. The addition of SA to HPMC films helps in providing improved quality of edible films with improved film moisture barrier properties [29]. The studies also revealed that the plasticizer type and proportions can influence the properties of HPMC/beeswax-based edible films [30]. Interestingly, the effectiveness of these coatings depends on the type of the fruit and cultivar. In pharmaceutical industry, it is mainly used as a hard outer coating for tablets. It protects the medicine against light, moisture, and shape distortion. Further, HPMC, PEG, and calcium lactate pentahydrate-based coating are also used to develop white film for pharmaceutical products [31]. The use of pharmaceutical coatings on tablets helps to protect the tablets against photolytic degradation and other environmental agents. HPMC is further used in pharmaceuticals as binder, film coating, film-forming material, controlled release agent, biological adhesive, etc. [32]. This specific biopolymer, being a hydrophilic biopolymer, is used for the development of oral-controlled drug delivery system, where the mathematical modeling of HPMC-based delivery system can also be developed [33].
On the other hand, HPMC in combination with other biopolymeric materials is used to develop water-soluble edible pouches, to deliver dry food products. Sebti et al. (2002), developed a bioactive edible food packaging material by associating nisin and SA to HPMC, where the molecular interactions are pH-dependent and by adjusting the pH to 3, there induced a high film inhibitory activity [34]. Further, HPMC bears certain limitations and drawbacks in the food packaging applications for having limited moisture resistance. Recently, researchers have investigated on enhancing the barrier properties of HPMC by incorporating cellulose fibers [35], fatty acids [36], and essential oils [37]. In the year 2010, Pastor and others developed HPMC-based composite films incorporating extracts of propolis which provide improved WVP in comparison with neat HPMC films and also provide colored film with antifungal activity [38]. However, in case of food, pharmaceutical, and paint industries, lacquers and varnishes are used as a barrier against moisture. Shellac is comprised of polyesters and single ester, which can provide protection against moisture and has great applications in the area of food industry and pharmaceutical industry [39]. In this regard, the HPMC/shellac-based composite films with emulsifiers SA and LA provide tailored-made mechanical and moisture barrier properties, where LA is a more effective emulsifier than SA [40].
-
Carboxymethyl Cellulose (CMC). CMC is a typical anionic polysaccharide, which is one of the most important cellulose derivatives. It is water soluble in nature and is used in the food, pharmaceutical, and cosmetic industry as an effective additive. CMC consists of chains of linear β(1 → 4) linked glucopyranose units with carboxymethyl group bound to hydroxyl groups. In addition, CMC contains a hydrophobic polysaccharide backbone and many hydrophilic carboxyl groups and hence shows amphiphilic characteristics. It is non-toxic, biocompatible, biodegradable, hydrophilic, and possess good film-forming ability. CMC has been used in numerous edible film formulations. It is generally used as a hydrophilic polymer coating to food products and has many areas of application in food industry [41]. The formation of covalently bonded CMC-casein complex can be achieved by electrosynthesis and are very pH stable. These complexes also exhibit good emulsifying properties and thermal stability. Also, the Maillard reaction may occur when proteins are mixed with carbohydrates at higher temperatures. Additionally, the development of edible films is based on CMC and soy protein isolate (SPI) blends and glycerol (as a compatibilizer). The films showed improved mechanical properties with increased CMC content and further provide reduced water sensitivity and tailored morphological behavior of the SPI films [42]. Also, the use of citric acid (CA) as a cross-linker for the development of CMC/polyvinyl alcohol (PVA)/aloe vera (AV) films has been done. The films have improved moisture barrier and mechanical property, and the use of AV acts against UV radiation. The developed films are eco-friendly and water-resistant active packaging film for meat products [43].
2.3.2 Chitosan
The another polysaccharide material, CS, is the second most abundantly available material after cellulose. It is developed by deacetylation (using sodium hydroxide) of chitin, which is availed from sea-based waste [44]. It is a flexible biopolymer derived from various renewable resources and is considered a sustainable material widely used for edible food packaging. Additionally, it is a non-toxic, functional, and biodegradable material used for varied applications. It is a linear polysaccharide containing (1 → 4)-linked 2-amino-2-deoxy-β-D-glucan. It also serves as a natural antimicrobial agent from animal origin and is insoluble in water and any other organic solvent. It is soluble in slightly acidic water below pH 6.3 producing viscous solution, which is preferable for some edible-coating approaches. The solubility of CS depends on the molecular weight of the polymer. Although a precise mechanism has not been yet generated for CS’s antimicrobial mechanism. The several properties of CS may be imparted due to several reasons such as (a) electrostatic interaction between amine groups of the polymer and microbial cell membrane provides antimicrobial property; (b) it acts as a chelating agent; (c) it acts a penetrating agent through the microbial cell membrane and (d) modifies the cell surface and nutrient transport in the bacterial cells. In this regard, CS has antimicrobial properties offered by the polycationic nature of the molecule [45]. Vásconez et al. developed CS and ST-based edible coating and films to improve the quality of salmon muscle, where the addition of CS reduces the WVP and solubility of the films [46]. Additionally, CS-based blends with thermoplastic polymers are developed to attain humidity-resistant property [47]. CS having antimicrobial properties has attained great attraction in food packaging application, where the antimicrobial properties are affected by pH, intrinsic factors, CS-metal complex, etc. [48]. CS-based edible films or edible coatings are developed using CS blends or composites, which offer modified packaging properties to extend shelf life of food products [49].
2.3.3 Starch
ST, a kind of storage polysaccharide, is obtained from several renewable resources and consists of amylose (a linear polysaccharide) and amylopectin (a branched polysaccharide). In cereal kernels, ST is a constituent consisting of more than 60% of cereal kernels, which can be easily separated from other chemical components of the kernels [50]. Additionally, ST can be obtained from various other sources such as rice, oat, corn, wheat, maize, sago, potato, and cassava. [50, 51]. Based on the origin of ST, it may have varied shape, size, structure, and composition of amylose and amylopectin. This polysaccharide is naturally available, inexpensive, biodegradable, edible in nature, and a potential candidate for developing edible food packaging materials. ST is available as granules, which make them insoluble in water, where the constituent’s amylose is water soluble and amylopectin is water insoluble. However, properties of ST are modified through formulating blends, composites, and plasticizers to obtain improved properties.
The ST-based packaging materials are developed employing two types of techniques such as wet and dry process. The wet process for developing ST-based edible films is generally preferred over dry process. The dry process involves extrusion technique, where the thermoplastic materials are preferred. The ST is mixed with plasticizer at high temperature to deliver the thermoplastic behavior. The addition of plasticizers and other biopolymers can offer tailored flexibility of ST-based films. As already mentioned, ST is insoluble in water, however, get solubilized in hot water, thus ST is dissolved at high temperature to prepare edible films or coatings, where the ST granules lost its semicrystalline nature at high temperature. In wet process of developing edible films, the biopolymer is applied to food products via dipping, spraying, or brushing [52]. The ST being hydrophilic in nature has a poor WVP. However, the WVP of ST films are dependent on temperature, thickness of film, plasticizers content, etc., which further varied in terms of intermolecular interactions [53]. ST can provide excellent oxygen barrier properties by tailoring the crystalline or amylopectin part of the polymer. On the other hand, the brittle nature of ST provides poor mechanical property, which can further be modified by developing composites or blends of polymers. In this way, ST-based blends and composites with added plasticizers are extensively utilized to develop edible food packaging materials for storing food products.
2.3.4 Pectin
It is one of the main components of plant cells and a citrus by-product. It is basically a by-product of agricultural crops such as apple (1–1.5%), citrus peels (30%), oranges (0.5–3.5%), and carrots. However, pectin is widely extracted from apple pomace and dried citrus peels. It is commonly classified into two categories based on the degree of methylation (DM), low-methoxyl pectins (DM < 50%) ,and high-methoxyl pectins (DM > 50%) [54]. It consists of repeating units of α-1,4-linked galacturonic acid and a major plant cell wall component. The pectin-based edible films have poor thermomechanical, barrier, and low water resistance properties, which decrease its use in food packaging applications. However, pectin with other components is widely used in combination with other materials. In this regard, the pectin and green tea powder-based edible coating on irradiated pork patty have a positive impact on improving the product quality [55]. Further, films developed from pure pectin promoted microbial growth as pectin is generally used as a carbon source by the microorganisms. As, pectin serves poor antimicrobial properties, so addition of antimicrobial agents such as oolong tea, green tea, and others can improve edible food packaging quality. In this regard, pectin and oregano essential oils based edible coating provide antifungal activity in maintaining the quality of tomatoes [56]. Further, pectin is also used as a gelling agent in developing jams and jellies and also stabilizing agents in dairy products. Edible active packaging made from pectin with essential oils has been reported as potential antimicrobial coatings for strawberries for enhanced shelf life [57]. Recently, pectin incorporated with nanochitosan in the ratio of 50:50 provides improved edible film properties in terms of tensile strength, water solubility, WVP, and others [58]. Nano-CS being the most natural antifungal agent that has been used along with the pectin. Manrich et al. produced edible films (casting technique) made from tomato cutin and pectin in various proportions which provide low water uptake and solubility and thermal and mechanical stability similar to tomato peels [59]. Thus, it is blended with natural polymers and hydrophobic compounds to get the desired property.
2.3.5 Carrageenan
Carrageenan is an anionic and linear sulfated polysaccharide obtained from red edible seaweed. According to the Food and Drug Administration (FDA), carrageenan and alginates are generally recognized as safe (GRAS) materials for edible films and coatings. It is a combination of several polysaccharides. Carrageenan is composed of repeating β-d-galactose units and 3,6-anhydro-α-d-galactose (both sulfated and non-sulfated) linked by alternating α-(1,3) and β-(1,4) glycosidic linkages. The three major forms of carrageenan are kappa (κ), iota (ι), and lambda (λ) with varied degree of sulfation [60]. The chemical structures of different types of carrageenan have been represented in Table 2.3. The carrageenan-based edible films and coatings are widely applied to various food products such as meat products, oily foods, dry solid foods, and food products [61]. In this regard, thermo-reversible carrageenan gels can be used as food coatings to retard moisture loss from enrobed food by acting as a sacrificing agent. These are natural hydrophilic polymers, and the film formation follows a gelation mechanism to form a three-dimensional network and a solid film after evaporation of solvent. It is applicable in desserts, milkshakes, and condensed milks, where carrageenan helps to form gels with increased viscosity. The edible films based on semi-refined κ-carrageenan with plasticizers (glycerol and sorbitol) provide improved mechanical (tensile strength and elongation at break) and oil barrier properties in comparison with neat semi-refined κ-carrageenan [62]. Thus, modified carrageenan is used to obtain good film properties, where the carrageenan is found mostly in the seaweeds of Kappaphycus alvarezii and Eucheuma denticulatum [63]. Additionally, the alginate and carrageenan are best used as a film coating for meat and meat products. The coatings generally prevent shrinkage, microbial attack, and surface discoloration by delaying the moisture transport. The fabrication of edible films based on κ-carrageenan and locust bean gum provide improved functionality, physical properties, and further deliver improved barrier and mechanical properties [64]. In this way, several polysaccharides including CS, carrageenan, and others are utilized to develop edible coating to extend shelf life of food products [65].
2.3.6 Alginate
Alginates are derived from seaweeds and deliver good film-forming property. This biopolymer exhibits unique colloidal properties and is used as a deliverable in pharmaceuticals. In this regard, alginate beads containing 5-fluorouracil can be used for the treatment of breast cancer [66]. It has been used extensively as an impression-making material in dentistry and prosthetics. Moreover, alginates having the properties of film forming, gelling, and thickening has attained a great interest to be used as a food ingredient [67]. It is also used in the food industry, for thickening soups and jellies and forms strong films which exhibit poor water resistance because of their hydrophilic nature. The alginate and apple puree-based edible films attributes can be modified using plant essential oils such as oregano oil, lemongrass oil, and citral oils., where the essential oils provide antimicrobial property to the films [68].
2.4 Lipids in Edible Food Packaging
These are edible polymers that consist of fats, glycerides, and acetylated glycerides (monoglycerides, diglycerides, triglycerides), natural wax, and surfactants. The fats and oils in edible films and coatings include animals and vegetable oils and fats. The vegetable fats and oils are coconut oil, peanut oil, palm oil, cocoa, milk butter, etc. These are meant mainly for blocking moisture with their low polarity. The films with lipid-based materials are more hydrophobic and brittle in nature. So, these films are associated with the polymer matrix and other cellular derivatives to improve the mechanical strength. The biomaterials polysaccharides, proteins, and lipids with their combined form are used to develop edible-coating materials [69]. In this way, edible films based on ST-MC-lipid blends are used to improve the shelf life of dry bakery products, where the films properties can be modified according to the blending levels, concentration, plasticizers, etc. [69]. Additionally, the lipid-based materials can also develop a continuous layer over hydrophilic materials such as polysaccharides or proteins [70]. On the other hand, lipid can be used as a dispersed phase in the matrix materials to develop edible films and coatings [70]. The properties of lipid-based films are based on several factors such as type of lipid, volume fraction, drying condition, and processing type. Moreover, lipid is extensively used materials with polysaccharides and proteins for attaining improved moisture barrier properties [71].
2.5 Proteins in Edible Food Packaging
Proteins are linear randomly arranged copolymers made from different amino acid monomers. In the early twentieth century, these materials attracted many textiles and plastic industries. There are different classifications of protein based on sequence of amino acid, shape, and composition. Further, the different proteins are random coil (milk casein), fibrous (collagen), or globular (corn zein, soy protein, wheat gluten, whey protein), etc. [72]. The fibrous proteins are completely extended and are linked through H-bonding in different parallel structures to form fiber like structure. Similar to polysaccharides, protein films are also susceptible to more WVP. The limitation is due to the presence of hydrophilic plasticizers, viz. glycerin and sorbitol to impart flexibility to the films. The proteins and polysaccharides along with the lipids can form a homogeneous hydrocolloid matrix that can overcome the mechanical strength and WVP of the film. Researchers have found, fatty acids such as saturated fatty acids (LA, SA, and PA), and unsaturated fatty acids (oleic acid) provide tunable film properties of sodium caseinate in terms of water vapor barrier properties, mechanical properties, optical properties, etc. [73]. The intrinsic properties of proteins such as amino acid content, hydrophobicity and hydrophilicity, crystallinity, and molecular size make them exceptional starting materials for developing films and coatings. However, the extrinsic factors include processing temperature and its relative humidity, pH, ionic strength, salt type, shear, and pressure. The different protein sources used for developing edible films and coatings include whey, soy, wheat gluten, keratin, casein, etc. [74]. The protein-based films and coatings are biodegradable and compostable. These kinds of films serve as a source of nitrogen as they degrade on soil. These can be of plant and animal origin, and the films are generally prepared by combining protein, plasticizer, and solvent. The following are some of the proteins, which are widely used for developing edible films.
2.5.1 Animal Proteins
-
Milk Protein. This comprises whey protein and casein protein, and the casein contains 80% of the milk content, which comprises α, β, and κ components [75]. The α and β casein are calcium ion sensitive and are unable to participate in disulfide bond formation and cross-linking. These provide better barrier property but are rigid in nature. The obtained casein solution is generally treated with alkali to attain pH 7 and dried for further treatment. The whey protein is a by-product obtained from cheese-making process. It is prepared by precipitation of casein protein in milk at 4.6 pH and temperature of 20 °C.
-
Meat Protein. There are three types of meat proteins, viz. sarcoplasmic, stromal, and myofibrillar. The stromal proteins include collagen and elastin. The collagen is a by-product of meat processing. These are fibrous protein obtained from connective tissue, tendons, skin, bones, and vascular system. Gelatin is produced by treating collagen with acid or alkali solution. The pure gelatin formed is brittle in nature, and it forms a film once it is dissolved in hot water. Additionally, gelatin is extensively used in several areas including edible films, food, cosmetic coating, pharmaceuticals, etc. [76]. It is served in the food industry because of its good gelling property. It also offers excellent foaming property that helps in making a good edible film. It has been used as a coating for food and pharmaceutical industries for years, where the properties of gelatin can be modified using other biopolymeric materials such as CS and cellulose derivative.
-
Silk Protein. There are several silk-based proteins, among which the sericin is a glue-based protein produced by silkworm Bombyx mori, Antheraea assamensis, and many other sources. This type of protein is edible in nature, antibacterial, and it is oxygen and UV resistant. Studies have revealed that the incorporation of these proteins into some other macromolecular materials helps in enhancing the mechanical property of the blended system [77]. Wang et al. developed whey protein and sericin blended films, where sericin protein provides tunable properties of whey protein films in terms of WVP, solubility, and mechanical property [78]. The WVP of sericin and whey protein blended films has found to be reduced due to the sericin content. Also, studies have reported the preparation of silk fibroin-based thin films. By using a postprocessing water-based method to control the protein polymorphism which enable the modulation of diffusion of gases such as oxygen, carbon dioxide, and WVP, can be used as a parameter to confirm the freshness of the food and by using the dip-coating technique on fruits like strawberries and bananas, it is found, that the silk fibroin enhanced the shelf-life of the food at room temperature [79].
2.5.2 Plant Proteins
-
Wheat Protein. These constitute a major part of the bread-making industry. Wheat gluten, which contains a small proportion of charged amino acids (lysine, histidine, arginine) and a high proportion of non-polar amino acids, aggregates easily due to hydrophobic interactions. There are four main wheat protein fractions, based on their solubility: albumins (water soluble), globulins (dilute salt soluble), gliadins (soluble in 70–90% ethanol), and glutenin (insoluble under any conditions). The properties of wheat gluten-based edible films can be tailored using plasticizers such as water and glycerol, where the glycerol helps to improve the film extensibility [80]. Glutenin and gliadin are the major proteins making up 47 and 34% of the total wheat protein content, respectively [81]. Gliadin contains intramolecular disulfide bonds which are responsible for film formation.
-
Soy Protein. It is basically extracted from soya beans (38–44%). It comprises a mixture of globular proteins. It contains glycinin which is used as a gelling agent and provides film-forming property. These types of films are superior in film making as compared to lipids and polysaccharides based on their gas barrier property. Additionally, soy protein-based films can be modified using acids and oils [82]. In this regard, the use of lactic acid in developing soy protein-based films can improve the hydrophobicity and maintain the UV barrier properties.
-
Corn Zein. It is a polyamine and a main component extracted from corn. These are used for edible films and coatings. These are insoluble in water. The two major parts of zein are α and β zein. These are glossy, tough, and grease proof. They show low WVP as compared to most other agriculturally based protein films. It has been commercially used as a coating agent for pharmaceuticals [83].
2.6 Other Materials in Edible Food Packaging
The various materials such as fruit purees, pomace, and peel are also used to develop edible food packaging materials. Fruit purees are extracted from apple, oranges, banana, pear, etc. These are mainly composed of cellulose and pectin, which provide less flexible films. The flexible films using the specified materials can be fabricated by adding plasticizers, which makes it mechanically stable and improves the water barrier property. Also, fruit pomace is a cellulose extract from fruit juice industries used for making edible films. It also imparts flavor and color to the food. The development of edible films based on mango puree reinforced with cellulose nanofibers forms a fibrillary network within mango puree, which further delivers improved mechanical properties [84]. Another example, the carrot puree films are brittle and rigid in nature, so the addition of plasticizer makes the film more flexible and resistant to oxidation. However, several fruit purees such as peach, apricot, pear, apricot-based edible films can provide tailored permeability properties at different storage conditions [85].
2.7 Overview of Nanosystems Facilitated Materials in Edible Food Packaging
The nanosystem facilitated edible food packaging has attained great interest in developing edible food packaging to combat the available packaging materials with additional benefits. As shown in Fig. 2.4, the nanosystems in edible food packaging commonly include the use of biopolymeric nanostructured materials, inorganic nanomaterials, nanocomposites, nanoencapsulation, nanosystems, etc. The extensively used biopolymeric nanostructured materials in food packaging include cellulose, CS, ST, protein, and lipid-based nanostructured materials [86]. The nanostructured materials are available in different sizes and shapes such as nanocrystals, nanofibers, nanoparticles, and nanotubes. Additionally, several inorganic nanomaterials also used to develop edible food packaging, where some of the inorganic materials deliver antimicrobial, antibacterial activity to improve the shelf life of food products. The nanocomposites are defined as the dispersion of nanoscale materials (dispersing materials) in matrix materials (continuous phase), where developing nanocomposites provide improved properties in terms of barrier, mechanical, thermal, and optical properties. Additionally, nanoencapsulation is another technique to entrap active compounds using carrier materials such as gum arabic, ST, and maltodextrin.
2.8 Synthesis Strategies to Develop Edible-Coated Food Products
As mentioned in Fig. 2.5, the several methods used for developing edible-coated food products include dipping, spraying, fluidized-bed coating, dripping, foaming, panning, brushing, etc. In wet processes of developing edible coating, liquid forms of coat materials are used to coat regular and irregular products via dipping, spraying, electrospraying, and others followed by drying. Additionally, the selection of a particular coating method depends on several attributes such as surface property, selected coating materials (rheology and other property), and adhesion property. However, the stability between targeted food products and coating materials is essential to develop a good coat on food products. The layer by layer coating is another approach to improve the shelf life of food products, where the coating materials can be applied simultaneously or sequentially on food products. Further, the selection of processes also depends on large-scale coatings development and required time, where a discussion related to available techniques for developing edible coating has been made in the below sections.
2.8.1 Dip-Coating Method
The dip-coating method is extensively used laboratory scale approach for developing edible coating on food products, which provide several beneficial approaches in comparison with other available techniques such as simple, cost effective, and develop uniform coverage on both regular and irregular food products. The several processing steps for developing edible coating using dipping are represented in Fig. 2.6, which involves dipping of the selected food products in the developed edible-coating solution. However, to develop a dipping-based edible coating on fruit and vegetables, viscous solutions are preferred [87]. The dipped food products are further allowed to drain the excess materials from the food products and then dried in the open air or hot air dried to solidify the coating on food products followed by storage in specific conditions. This kind of coating process is commonly used to develop a uniform coating on food products. The coating thickness on food products is a critical parameter to consider in edible-coated food products, where the coating thickness on food products via dip-coating treatment depends on various factors such as dipping time, product temperature, and temperature of prepared coating materials. [88]. However, the controlling of processing conditions and continuous production of dipped coated food products are generally the shortcomings in developing dipped coated food products. Moreover, the dipping method involves the direct contact of the selected food products and coating solution, which may lead to microbial contamination in the coating solutions. In dip coating, the fruit products are washed using distilled water, disinfecting agents to avoid contamination before dipping into the coating solution. Further, the layer by layer coating can also be developed using dip-coating method, where the selected food products are dipped into different coating solutions followed by draining the excess solution after successive dipping [89].
The use of dip-coating method has been applied for developing coating on several categories of food products such as fruits and vegetables, meat and meat products, fish and fish products, and milk products. [88,89,90,91,92,93]. In this regard, the development of edible coating on several fruit products using dip-coating method has been represented in Table 2.4. The edible coating using dip coating can be given on whole (apple, pear fruit, strawberry) [94,95,96] and cut fruits such as apple slices [97], cut pineapple [98], fresh-cut pears [99], fresh-cut melon [100], fresh-cut kiwi fruits [100], and others, which are widely edible coated using dipped coating methods. Further, edible coating using dip-coating method is also developed by dipping in different solutions such as edible coating of cut pears via dipping in polysaccharide solution (first dipping) and calcium chloride solution with antibrowning agents (second dipping) [99]. The use of antibrowning agents can control the enzymatic browning and retard microbiological degradation of fruit products. The dipped coating of chicken breast meat can be obtained by first applying CS solution and then oregano essential oils under modified atmospheric packaging (MAP) conditions can retard lipid oxidation and significantly affected mesophilic count, lactic acid bacteria, yeast-molds, etc. [92]. The dip-coating of meat samples using CS, glycerol, tween 80 and Zataria multiflora essential oil (ZEO) is reported to perform, where development of edible coating on meat samples followed several steps such as (i) dipping in coating solution for 2 min, (ii) draining of excess coating solution from the samples, (iii) dipping again for 2 min, and (iv) drained for 5 h at 10 °C [91].
2.8.2 Spraying-Based Edible Coating
The spraying approach for developing edible coating delivers uniform coating on food products, with well-controlled processing as the product and the coating solution are in direct contact due to the spraying action. The use of CS-based preharvest spraying and postharvest coating on fruit products are beneficial to provide improved shelf life [102]. Among the available approaches utilized for developing edible coating, spray-based edible-coated food materials are available commercially. Additionally, a uniform thickness can also be developed on food products, where the spraying-based coating is suitable for low viscous solution. The spray-based edible coating is more acceptable when the coating is applied to a particular side of food products. The coating solution is sprayed on the food products by controlling the final drop size (drop size distribution up to 20 µm), which further depends on spray gun type, temperature of the nozzle, flow rates of air and liquids, drying conditions, etc. The spray-based edible coating is one of the most popular edible-coating methods applied for fruits and vegetables using atomizers and high-pressure spraying systems [103]. The processing steps in spray coating includes several steps such as (1) formation of droplets through atomization: This process begins by pumping coating materials through a nozzle; (2) drop impact spreading: This process involves spraying the drops of coating on the food surface, where based on the interaction between materials and food surface the drop may splash, rebound, or deposit on the food; (3) secondary leveling of droplets; (4) consolidation; and (5) drying of coating layer. The several wetting behaviors of drops onto food products or impacts of drops on food surface (such as banana, eggplant, purple cabbage) follow several stages such as kinematic phase (depends on the kinetic energy of the drops), spreading phase (the drop has a thin film shape), retraction, and relaxation. However, the coating solutions are optimized based on wettability (represents ability of coating materials to spread on a solid surface), adhesion forces (related to spreading the liquid on food surface), and cohesion forces (causes liquid to contract on food surfaces) [65]. Additionally, the control of drop impact on food product depends on several properties such as physicochemical properties of liquid (surface tension, temperature, viscosity, impact direction, kinetic energy of drop; surface roughness of food, surface energy of food, surface temperature of food, air temperature, and pressure) [104, 105]. The spraying-based edible coating has several beneficial attributes such as provide uniform coating, thickness control, multilayer of coating, continuous coating, and temperature control. However, operating cost is also a crucial parameter in selecting the nozzle type. The limitations of spray coating are further related to surface distribution and transfer efficiency. The development of spray-based edible coating depends on spray conditions such as product surface, spray surface, spray time, and distance between pneumatic nozzle and product. In this regard, to obtain a specific thickness, the several spray conditions need to be standardized. The spray-based edible coating on food products is a widely used technique to develop coating at large and continuous scale.
Spraying is used to develop an edible coating on different food products such as cheese, meat, and meat products, dry bakery products, fruits (strawberry, mandarin oranges, grapes, etc.). [69, 93, 103, 106]. The development of spray-based edible coating on a commercial cracker is obtained via developing coating on upper and lower surface separately. The emulsion-based coating is applied on the upper surface followed by drying at 60 °C for 2 h. The spraying of emulsion-based coating consisting of ST, MC, and soybean oil has been obtained at a pressure of 2 bar at 70 °C [69]. The development of antimicrobial edible nanocoating using spraying techniques on fruit products can enhance the product life, where the antimicrobial nanocoating is developed using supramolecular metal-organic coordination complex of ferric ions and tannic acid [106]. In this process, the spraying of tannic acid and ferric ions is applied sequentially and simultaneously on fruit products. In sequential spraying, the mentioned coating materials are applied to food products sequentially, whereas, in simultaneous spraying systems, the coating materials are applied to food products simultaneously, which is generally employed for bulk scale coating. Spray-based methods can develop a uniform coating on food products, further, bakery products require the formation of coating directly on food product, which can be developed using spray-based coating. The liquid droplet size depends on atomization pressure, where smaller sizes of liquid droplets can be obtained at higher pressure. The formulation of edible coating (via spraying technique) on grapefruits are obtained with the hydrocolloid materials such as HPMC, κ-carrageenan, glycerol, and cellulose nanofibers [103]. The spraying application of grapefruits is also optimized in terms of thickness of coating and percentage of coating and independent variables: liquid suspension flow, air pressure, and height of impact as 1–5 L/h, 50–200 kPa, and 0.3–0.5 m, respectively [103]. In spray-based technique, the optimized conditions for suspension flow rate, air pressure, and spray nozzle height are 1 L/h, 200 kPa, and 0.5 m, respectively, where coating thickness between 24.2 ± 0.9 and 38.5 ± 1.4 µm can be obtained. Further, the physical properties of liquids such as viscosity, density, surface tension, and others are different, thus the droplet formation and generation are different depending on the specific liquids.
2.8.3 Electrospraying-Based Edible Coating
Electrospraying technique is an efficient approach to develop an edible coating on complex food products and further help to reduce the processing cost in industries [107]. In the electrospraying technique, fine droplets of edible materials are obtained on account of electrostatic forces, which produce charged liquid surfaces. The electrospray process has an ability to develop fine droplets with a narrow size distribution of 0.1–1000 µm. The fine droplets provide thin coatings on food products. The electrospray technique is used to increase the shelf life of minimally processed food while maintaining the sensory properties. The electrostatic spraying method has many benefits over other common spraying techniques such as controlled droplet size, reduced waste of coating materials, increased droplet coverage, and develop homogeneous distribution. However, the electrostatic spraying approach is more preferred in comparison with non-electrostatic or conventional spray-based systems in delivering higher transfer efficiency and coating uniformity [107, 108]. The use of electrostatic spray-based approach for developing coating using materials alginate with carvacrol, and methyl cinnamate on strawberry fruits, helps in maintaining fruit firmness, weight loss, retain color, and others [108]. The sunflower oil and dark chocolates can be used to develop moisture barriers-based edible coating on hygroscopic tablets via electrospraying technique, where electrospraying of sunflower oil offers smaller droplets having higher charge to mass ratio [107]. Moreover, a study reports the use of electrospray-based technique to develop an edible coating on different food models such as apple slices and candy tablets, where chocolate and water-in-oil emulsion-based coating have been developed. The emulsion and chocolate-based coating help in reducing browning and water loss in apple slices [109]. Lipids are extensively used materials for developing electrospraying-based edible coating, however, lipids are non-conductive and not suitable for electrospraying [110]. In this regard, lecithin is an ionic surfactant and can be used to increase the conductivity of targeted lipid materials to be used for electrospraying. Further, emulsion-based edible coating is developed using electrospraying technique for food products [110, 111].
2.8.4 Other Methods
The other methods applied for developing edible coating on food products include foaming, brushing, dripping, fluidized bed coating, panning, and others. The foaming is an approach to develop emulsion-based coating, where foam-based edible-coating materials are developed using a foaming agent. The foam is also developed by blowing compressed air in the applicator tank. To achieve uniform distribution of foam, tumbling action is employed to developed foam and the foams are applied to moving food products. Further, brushes are used to attain a uniform coat of emulsion over the surface of food products. Interestingly, the drying of the developed edible-coating materials on food products requires less time. The application of edible coating on food products has been developed using brushing technique on several food products such as fruits and vegetables and meat and meat products. However, the brushing method of edible coating is applied to food products, where the moisture loss is a problem. In this regard, the AV gel matrix with gelling agents is applied to develop an edible coating on fresh fruits and vegetables via brushing approach [112]. Further, carrageenan, CA, and cinnamon oil are used to develop edible coating on chicken filets employing brushing approach [113]. Edible coating is further developed on chopped papaya fruits using clove oil using brushing technique [114]. Dripping-based edible coating is suitable for agro-based products, where coating can be applied on the surface directly or brushes can be used to apply the same. However, good coating is obtained, when tumbling action is applied over several brushes. The dripping-based edible-coating approach is the cost effective. In enrobing-based edible coating, the developed sticky coating solutions are applied to flow vertically to the food products, where the products are edible coated by the action of viscous and gravitational forces. In enrobing method, viscosity of the coating solution is a crucial parameter in maintaining good food quality and weight of the food products. The enrobing is better suited for developing edible coating on flat food products. Enrobing based edible coating on chocolate, meat industry, and milk products such as cheese are developed. The enrobing-based coating processes are generally used in chocolate industries using enrobe-based processes. The fluidized bed drier (FBD) is considered as a potential edible-coating approach to develop thin layer of edible materials on dry particles of small sizes and very low density. The several processing steps to coat food components have been represented in Fig. 2.7, which include spraying of coating materials on food components (step 1), wetting of coating materials on food components (step 2), drying of coated food components (step 3), and layering of coating materials on food components (step 4). Moreover, the several attributes of functional food materials such as flavors, preservatives, and fortifiers can be improved, which helps to improve texture and provide easy handling using FBD-based coating.
2.9 Synthesis Strategies to Develop Edible Film
The development of edible films is obtained via wet and dry processes. The wet processes of developing edible films include solution casting process, where the film-forming solutions are prepared by dispersing or solubilizing the biopolymers in a film-forming solution. On the other hand, the dry process for developing edible food packaging includes extrusion, where the biopolymer should possess thermoplastic properties. In this regard, some polymers can attain the properties of thermoplasticity by adding plasticizers and heating above the glass transition temperatures such as thermoplastic ST. However, the extruded films having the disadvantages of wrapping irregular food products, wet processes can be advantageous.
2.9.1 Solution Casting Methods
As shown in Fig. 2.8, the edible films using solution casting method are developed following several steps such as (1) dispersion or solubilization of selected biopolymers in solvents for a specific time; (2) casting of solution on pertiplates or Teflon plates, where before casting the solutions are kept for some duration to remove air bubbles; (3) solvent evaporation of solution casted films under hot air drying or open air; and (4) finally, peeling of the films followed by storage to obtain equilibrium conditions. The developed films are used to wrap food products or can be used as a sandwich material within food products. In fabricating solution casted edible films, the solvents for films materials are based on the properties of biopolymers (solubility), such as CS is soluble in acidic media (1% w/w acetic acid solution) [98], whereas the ST is soluble in hot water [115]. Further, the optimization of processing conditions for fabricating edible films can provide improved film properties. In this regard, the fabrication of edible films using cassava ST, glycerol, agar, and span 80 having concentrations of 1–3 g, 0.5–1.0 ml, 0.5–1.0 g, and 0.1–0.5 ml, respectively, are optimized using Box–Behnken experimental design to obtain second-order polynomial models for barrier and optical properties [116]. Further, the modeling of mechanical properties (tensile strength, Young’s modulus, elongation, puncture force, puncture deformation) for edible films based on cassava ST, glycerol, agar, and span 80 is developed by solution casting technique [117]. The emulsion-based edible films are also developed via solution casting process and used to protect food products, where animal waxes and plant waxes are extensively used lipid materials for developing emulsified films [118]. Further, the attributes of glycerol plasticized gelatin films (developed by casting technique) can be improved using cross-linking agents such as dialdehyde CMC [119]. In this regard, the properties of edible films are improved preparing composites and blends of biopolymers, using plasticizers, cross-linking agents, etc.
2.9.2 Extrusion Technique
The edible films are also focused to develop by extrusion followed by compression molding and blown films. The application of extrusion-based edible coating mainly depends on thermoplastic properties of the utilized materials and is considered as the well-utilized method for developing edible coatings for industrial application. The development of edible films using extrusion technique depends on several processing conditions such as extrusion temperature, concentration of biopolymers, and plasticizer content. The edible films based on fish gelatin are prepared using extrusion (twin screw extrusion at 110 and 120 °C) followed by compression molding (80 °C) [120]. In this regard, the extrusion and compression molding processes are more feasible to develop commercialized edible films in comparison with solution casting process. The edible films based on glycerol plasticized sodium caseinate are developed using twin screw and blown film extrusion methods, where the films can be produced in a large scale with no surface defects [121]. Moreover, active edible packaging based on plasticized sodium caseinate containing lysozyme can also be prepared using blown film extrusion method [122]. However, the processing temperature and glycerol have a great influential effect on lysozyme stability. The natural fiber of pectin and food hydrocolloids-based antimicrobial edible films are developed using extrusion technique followed by blown film or compression molding, where mild processing conditions are beneficial to protect bioactivity of antimicrobial agents [123]. The extrusion of pectin and gelatin/sodium alginate for developing blends-based edible films or casings can be tailored using corn oil or olive oil [124].
2.10 Conclusion
From very early days, the available biopolymeric materials are a potential candidate to develop edible food packaging for its noteworthy characteristics such as edible, biodegradability, availability, easy processing, and others. The biopolymers used for developing edible food packaging include cellulose and its derivatives, ST and its derivatives, CS, and its derivatives, protein and their modified forms, lipid, etc. Interestingly, the nanosystem-assisted edible food packaging has attained great interest in terms of using biopolymeric nanostructured materials, inorganic nanofillers, polymeric composites, and others to provide improved health benefits and shelf life of food products. The edible food packaging is developed as edible coatings and edible films. The fabrication of edible-coated food products generally follows three processing steps such as the development of a coating solution, application of the coating solution on the food products, and drying to solidify the coating materials on food products. Similarly, the development of edible films can be obtained via solution casting (laboratory scale approach), and extrusion (large-scale approach for commercialization) based on the required packaging category. However, the selection of synthesis approach for edible food packaging development also depends on the targeted materials which will be utilized to develop the packaging materials. Moreover, the processing conditions for developing edible food packaging need to be optimized to achieve better packaging properties with an improved storage life of food products.
Bibliography
Otoni CG, Avena-Bustillos RJ, Azeredo HM, Lorevice MV, Moura MR, Mattoso LH, McHugh TH (2017) Recent advances on edible films based on fruits and vegetables—a review. Compr Rev Food Sci Food Saf 16:1151–1169. https://doi.org/10.1111/1541-4337.12281
Zambrano-Zaragoza ML, González-Reza R, Mendoza-Muñoz N, Miranda-Linares V, Bernal-Couoh TF, Mendoza-Elvira S, Quintanar-Guerrero D (2018) Nanosystems in edible coatings: a novel strategy for food preservation. Int J Mol Sci 19:705. https://doi.org/10.3390/ijms19030705
Baldwin EA, Nisperos-Carriedo MO, Baker RA (1995) Edible coatings for lightly processed fruits and vegetables. HortScience 30:35–37
de Moura MR, Aouada FA, Avena-Bustillos RJ, McHugh TH, Krochta JM, Mattoso LH (2009) Improved barrier and mechanical properties of novel hydroxypropyl methylcellulose edible films with chitosan/tripolyphosphate nanoparticles. J Food Eng 92:448–453. https://doi.org/10.1016/j.jfoodeng.2008.12.015
Navarro-Tarazaga ML, Massa A, Pérez-Gago MB (2011) Effect of beeswax content on hydroxypropyl methylcellulose-based edible film properties and postharvest quality of coated plums (Cv. Angeleno). LWT-Food Sci Technol 44:2328–2334. https://doi.org/10.1016/j.lwt.2011.03.011
Adler E (1977) Lignin chemistry—past, present and future. Wood Sci Technol 11:169–218. https://doi.org/10.1007/BF00365615
Cazón P, Velazquez G, Ramírez JA, Vázquez M (2017) Polysaccharide-based films and coatings for food packaging: a review. Food Hydrocoll 68:136–148. https://doi.org/10.1016/j.foodhyd.2016.09.009
de Dicastillo CL, Bustos F, Guarda A, Galotto MJ (2016) Cross-linked methyl cellulose films with murta fruit extract for antioxidant and antimicrobial active food packaging. Food Hydrocoll 60:335–344. https://doi.org/10.1016/j.foodhyd.2016.03.020
De Dicastillo CL, Rodríguez F, Guarda A, Galotto MJ (2016) Antioxidant films based on cross-linked methyl cellulose and native Chilean berry for food packaging applications. Carbohydr Polym 136:1052–1060. https://doi.org/10.1016/j.carbpol.2015.10.013
Rodrigues Filho G, de Assunçao RM, Vieira JG, Meireles CDS, Cerqueira DA, da Silva Barud H, Ribeiro SJ, Messaddeq Y (2007) Characterization of methylcellulose produced from sugar cane bagasse cellulose: crystallinity and thermal properties. Polym Degrad Stabil 92:205–210. https://doi.org/10.1016/j.polymdegradstab.2006.11.008
Aswathanarayan JB, Vittal RR (2019) Nanoemulsions and their potential applications in food industry. Front Sustain Food Syst 3:95
Otoni CG, Pontes SF, Medeiros EA, Soares NDF (2014) Edible films from methylcellulose and nanoemulsions of clove bud (Syzygium aromaticum) and oregano (Origanum vulgare) essential oils as shelf life extenders for sliced bread. J Agric Food Chem 62:5214–5219. https://doi.org/10.1021/jf501055f
Garcıa MA, Ferrero C, Bertola N, Martino M, Zaritzky N (2002) Edible coatings from cellulose derivatives to reduce oil uptake in fried products. Innov Food Sci Emerg Technol 3:391–397. https://doi.org/10.1016/S1466-8564(02)00050-4
Bilbao-Sáinz C, Avena-Bustillos RJ, Wood DF, Williams TG, McHugh TH (2010) Composite edible films based on hydroxypropyl methylcellulose reinforced with microcrystalline cellulose nanoparticles. J Agric Food Chem 58:3753–3760. https://doi.org/10.1021/jf9033128
Kaur R, Yadav N (2019) Biodegradable polymeric film for food packaging. J Appl Packag Res 11:2
Gennadios A, Hanna MA, Kurth LB (1997) Application of edible coatings on meats, poultry and seafoods: a review. LWT-Food Sci Technol 30:337–350. https://doi.org/10.1006/fstl.1996.0202
Khan MI, Adrees MN, Tariq MR, Sohaib M (2013) Application of edible coating for improving meat quality: a review. Pak J Food Sci 23:71–79
Bourtoom T (2008) Edible films and coatings: characteristics and properties. Int Food Res J 15:237–248
Romero-Bastida CA, Flores-Huicochea E, Martin-Polo MO, Velazquez G, Torres JA (2004) Compositional and moisture content effects on the biodegradability of zein/ethylcellulose films. J Agric Food Chem 52:2230–2235. https://doi.org/10.1021/jf0350414
Shi P, Zuo Y, Zou Q, Shen J, Zhang L, Li Y, Morsi YS (2009) Improved properties of incorporated chitosan film with ethyl cellulose microspheres for controlled release. Int J Pharm 375:67–74. https://doi.org/10.1016/j.ijpharm.2009.04.016
Li X, Jiang F, Ni X, Yan W, Fang Y, Corke H, Xiao M (2015) Preparation and characterization of konjac glucomannan and ethyl cellulose blend films. Food Hydrocoll 44:229–236. https://doi.org/10.1016/j.foodhyd.2014.09.027
Guiga W, Swesi Y, Galland S, Peyrol E, Degraeve P, Sebti I (2010) Innovative multilayer antimicrobial films made with Nisaplin® or nisin and cellulosic ethers: physico-chemical characterization, bioactivity and nisin desorption kinetics. Innov Food Sci Emerg Technol 11:352–360. https://doi.org/10.1016/j.ifset.2010.01.008
Shit SC, Shah PM (2014) Edible polymers: challenges and opportunities. J Polym. https://doi.org/10.1155/2014/427259
Basch CY, Jagus RJ, Flores SK (2013) Physical and antimicrobial properties of tapioca starch-HPMC edible films incorporated with nisin and/or potassium sorbate. Food Bioproc Tech 6:2419–2428. https://doi.org/10.1007/s11947-012-0860-3
Jiménez A, Fabra MJ, Talens P, Chiralt A (2010) Effect of lipid self-association on the microstructure and physical properties of hydroxypropyl-methylcellulose edible films containing fatty acids. Carbohydr Polym 82:585–593. https://doi.org/10.1016/j.carbpol.2010.05.014
Sothornvit R (2009) Effect of hydroxypropyl methylcellulose and lipid on mechanical properties and water vapor permeability of coated paper. Food Res Int 42:307–311. https://doi.org/10.1016/j.foodres.2008.12.003
Osorio FA, Molina P, Matiacevich S, Enrione J, Skurtys O (2011) Characteristics of hydroxy propyl methyl cellulose (HPMC) based edible film developed for blueberry coatings. Procedia Food Sci 1:287–293. https://doi.org/10.1016/j.profoo.2011.09.045
Coma V, Sebti I, Pardon P, Pichavant FH, Deschamps A (2003) Film properties from crosslinking of cellulosic derivatives with a polyfunctional carboxylic acid. Carbohydr Polym 51:265–271. https://doi.org/10.1016/S0144-8617(02)00191-1
Hagenmaier RD, Shaw PE (1990) Moisture permeability of edible films made with fatty acid and hydroxypropyl methyl cellulose. J Agric Food Chem 38:1799–1803
Navarro-Tarazaga ML, Sothornvit R, Pérez-Gago MB (2008) Effect of plasticizer type and amount on hydroxypropyl methylcellulose–beeswax edible film properties and postharvest quality of coated plums (cv. Angeleno). J Agric Food Chem 56:9502–9509. https://doi.org/10.1021/jf801708k
Sakata Y, Shiraishi S, Otsuka M (2006) A novel white film for pharmaceutical coating formed by interaction of calcium lactate pentahydrate with hydroxypropyl methylcellulose. Int J Pharm 317:120–126. https://doi.org/10.1016/j.ijpharm.2006.02.058
Huichao W, Shouying D, Yang L, Ying L, Di WJJOC (2014) The application of biomedical polymer material hydroxy propyl methyl cellulose (HPMC) in pharmaceutical preparations. J Chem Pharm Res 6:155–160
Siepmann J, Peppas NAA (2012) Modeling of drug release from delivery systems based on hydroxypropyl methylcellulose (HPMC). Adv Drug Deliv Rev 64:163–174. https://doi.org/10.1016/j.addr.2012.09.028
Sebti I, Ham-Pichavant F, Coma V (2002) Edible bioactive fatty acid–cellulosic derivative composites used in food-packaging applications. J Agric Food Chem 50:4290–4294. https://doi.org/10.1021/jf0115488
De Moura MR, Avena-Bustillos RJ, McHugh TH, Wood DF, Otoni CG, Mattoso LH (2011) Miniaturization of cellulose fibers and effect of addition on the mechanical and barrier properties of hydroxypropyl methylcellulose films. J Food Eng 104:154–160. https://doi.org/10.1016/j.jfoodeng.2010.12.008
Vojdani F, Torres JA (1990) Potassium sorbate permeability of methylcellulose and hydroxypropyl methylcellulose coatings: effect of fatty acids. J Food Sci 55:841–846. https://doi.org/10.1111/j.1365-2621.1990.tb05244.x
Sánchez-González L, Vargas M, González-Martínez C, Chiralt A, Cháfer M (2009) Characterization of edible films based on hydroxypropylmethylcellulose and tea tree essential oil. Food Hydrocoll 23:2102–2109. https://doi.org/10.1016/j.foodhyd.2009.05.006
Pastor C, Sánchez-González L, Cháfer M, Chiralt A, González-Martínez C (2010) Physical and antifungal properties of hydroxypropylmethylcellulose based films containing propolis as affected by moisture content. Carbohydr Polym 82:1174–1183. https://doi.org/10.1016/j.carbpol.2010.06.051
Limmatvapirat S, Limmatvapirat C, Puttipipatkhachorn S, Nuntanid J, Luangtana-anan M (2007) Enhanced enteric properties and stability of shellac films through composite salts formation. Eur J Pharm Biopharm 67:690–698. https://doi.org/10.1016/j.ejpb.2007.04.008
Byun Y, Ward A, Whiteside S (2012) Formation and characterization of shellac-hydroxypropyl methylcellulose composite films. Food Hydrocoll 27:364–370. https://doi.org/10.1016/j.foodhyd.2011.10.010
Toğrul H, Arslan N (2004) Extending shelf-life of peach and pear by using CMC from sugar beet pulp cellulose as a hydrophilic polymer in emulsions. Food Hydrocoll 18:215–226. https://doi.org/10.1016/S0268-005X(03)00066-3
Su JF, Huang Z, Yuan XY, Wang XY, Li M (2010) Structure and properties of carboxymethyl cellulose/soy protein isolate blend edible films crosslinked by Maillard reactions. Carbohydr Polym 79:145–153. https://doi.org/10.1016/j.carbpol.2009.07.035
Kanatt SR, Makwana SH (2020) Development of active, water-resistant carboxymethyl cellulose-poly vinyl alcohol-Aloe vera packaging film. Carbohydr Polym 227:115303. https://doi.org/10.1016/j.carbpol.2019.115303
Kaur S, Dhillon GS (2014) The versatile biopolymer chitosan: potential sources, evaluation of extraction methods and applications. Crit Rev Microbiol 40:155–175. https://doi.org/10.3109/1040841X.2013.770385
Elsabee MZ, Abdou ES (2013) Chitosan based edible films and coatings: a review. Mater Sci Eng, C 33:1819–1841. https://doi.org/10.1016/j.msec.2013.01.010
Vásconez MB, Flores SK, Campos CA, Alvarado J, Gerschenson LN (2009) Antimicrobial activity and physical properties of chitosan–tapioca starch based edible films and coatings. Food Res Int 42:762–769. https://doi.org/10.1016/j.foodres.2009.02.026
van den Broek LA, Knoop RJ, Kappen FH, Boeriu CG (2015) Chitosan films and blends for packaging material. Carbohydr Polym 116:237–242. https://doi.org/10.1016/j.carbpol.2014.07.039
Aider M (2010) Chitosan application for active bio-based films production and potential in the food industry: review. LWT-Food Sci Technol 43:837–842. https://doi.org/10.1016/j.lwt.2010.01.021
Ghosh T, Katiyar V (2019) Chitosan-based edible coating: a customise practice for food protection. In: Katiyar V, Gupta R, Ghosh T (eds) Advances in sustainable polymers. Materials horizons: from nature to nanomaterials. Springer, Singapore. https://doi.org/10.1007/978-981-32-9804-0_8
Jiménez A, Fabra MJ, Talens P, Chiralt A (2012) Edible and biodegradable starch films: a review. Food Bioproc Tech 5:2058–2076. https://doi.org/10.1007/s11947-012-0835-4
Molavi H, Behfar S, Shariati MA, Kaviani M, Atarod S (2015) A review on biodegradable starch based film. J Microbiol Biotech Food Sci 4:456–461
Peressini D, Bravin B, Lapasin R, Rizzotti C, Sensidoni A (2003) Starch–methylcellulose based edible films: rheological properties of film-forming dispersions. J Food Eng 59:25–32. https://doi.org/10.1016/S0260-8774(02)00426-0
Bertuzzi MA, Vidaurre EC, Armada M, Gottifredi JC (2007) Water vapor permeability of edible starch based films. J Food Eng 80:972–978. https://doi.org/10.1016/j.jfoodeng.2006.07.016
Chaichi M, Hashemi M, Badii F, Mohammadi A (2017) Preparation and characterization of a novel bionanocomposite edible film based on pectin and crystalline nanocellulose. Carbohydr Polym 157:167–175. https://doi.org/10.1016/j.carbpol.2016.09.062
Kang HJ, Jo C, Kwon JH, Kim JH, Chung HJ, Byun MW (2007) Effect of a pectin-based edible coating containing green tea powder on the quality of irradiated pork patty. Food Control 18:430–435. https://doi.org/10.1016/j.foodcont.2005.11.010
Rodriguez-Garcia I, Cruz-Valenzuela MR, Silva-Espinoza BA, Gonzalez-Aguilar GA, Moctezuma E, Gutierrez-Pacheco MM, Ayala-Zavala JF (2016) Oregano (Lippia graveolens) essential oil added within pectin edible coatings prevents fungal decay and increases the antioxidant capacity of treated tomatoes. J Sci Food Agric 96:3772–3778. https://doi.org/10.1002/jsfa.7568
Guerreiro AC, Gago CM, Faleiro ML, Miguel MG, Antunes MD (2015) The use of polysaccharide-based edible coatings enriched with essential oils to improve shelf-life of strawberries. Postharvest Biol Tech 110:51–60. https://doi.org/10.1016/j.postharvbio.2015.06.019
Ngo TMP, Nguyen TH, Dang TMQ, Tran TX, Rachtanapun P (2020) Characteristics and antimicrobial properties of active edible films based on pectin and nanochitosan. Int J Mol Sci 21:2224. https://doi.org/10.3390/ijms21062224
Manrich A, Moreira FK, Otoni CG, Lorevice MV, Martins MA, Mattoso LH (2017) Hydrophobic edible films made up of tomato cutin and pectin. Carbohydr Polym 164:83–91. https://doi.org/10.1016/j.carbpol.2017.01.075
Stone AK, Nickerson MT (2012) Formation and functionality of whey protein isolate–(kappa-, iota-, and lambda-type) carrageenan electrostatic complexes. Food Hydrocoll 27:271–277. https://doi.org/10.1016/j.foodhyd.2011.08.006
Karbowiak T, Debeaufort F, Champion D, Voilley A (2006) Wetting properties at the surface of iota-carrageenan-based edible films. J Colloid Interface Sci 294:400–410. https://doi.org/10.1016/j.jcis.2005.07.030
Farhan A, Hani NM (2017) Characterization of edible packaging films based on semi-refined kappa-carrageenan plasticized with glycerol and sorbitol. Food Hydrocoll 64:48–58. https://doi.org/10.1016/j.foodhyd.2016.10.034
Largo DB, Fukami K, Nishijima T (1995) Occasional pathogenic bacteria promoting ice-ice disease in the carrageenan-producing red algae Kappaphycus alvarezii and Eucheuma denticulatum (Solieriaceae, Gigartinales, Rhodophyta). J Appl Phycol 7:545–554. https://doi.org/10.1007/BF00003941
Martins JT, Cerqueira MA, Bourbon AI, Pinheiro AC, Souza BW, Vicente AA (2012) Synergistic effects between κ-carrageenan and locust bean gum on physicochemical properties of edible films made thereof. Food Hydrocoll 29:280–289. https://doi.org/10.1016/j.foodhyd.2012.03.004
Ribeiro C, Vicente AA, Teixeira JA, Miranda C (2007) Optimization of edible coating composition to retard strawberry fruit senescence. Postharvest Biol Tech 44:63–70. https://doi.org/10.1016/j.postharvbio.2006.11.015
Arıca B, Çalış S, Kaş HS, Sargon MF, Hıncal AA (2002) 5-Fluorouracil encapsulated alginate beads for the treatment of breast cancer. Int J Pharm 242:267–269. https://doi.org/10.1016/S0378-5173(02)00172-2
Qin Y, Jiang J, Zhao L, Zhang J, Wang F (2018) Applications of alginate as a functional food ingredient. In: Biopolymers for food design. Academic Press, pp 409–429. https://doi.org/10.1016/B978-0-12-811449-0.00013-X
Rojas-Graü MA, Avena-Bustillos RJ, Olsen C, Friedman M, Henika PR, Martín-Belloso O, McHugh TH (2007) Effects of plant essential oils and oil compounds on mechanical, barrier and antimicrobial properties of alginate–apple puree edible films. J Food Eng 81:634–641. https://doi.org/10.1016/j.jfoodeng.2007.01.007
Bravin B, Peressini D, Sensidoni A (2006) Development and application of polysaccharide–lipid edible coating to extend shelf-life of dry bakery products. J Food Eng 76:280–290. https://doi.org/10.1016/j.jfoodeng.2005.05.021
Pérez-Gago MB, Rhim JW (2014) Edible coating and film materials: lipid bilayers and lipid emulsions. In: Innovations in food packaging. Academic Press, pp 325–350. https://doi.org/10.1016/B978-0-12-394601-0.00013-8
Morillon V, Debeaufort F, Blond G, Capelle M, Voilley A (2002) Factors affecting the moisture permeability of lipid-based edible films: a review. Crit Rev Food Sci Nutr 42:67–89. https://doi.org/10.1080/10408690290825466
Janjarasskul T, Krochta JM (2010) Edible packaging materials. Annu Rev Food Sci Technol 1:415–448
Fabra MJ, Jiménez A, Atarés L, Talens P, Chiralt A (2009) Effect of fatty acids and beeswax addition on properties of sodium caseinate dispersions and films. Biomacromol 10:1500–1507. https://doi.org/10.1021/bm900098p
Dangaran K, Tomasula PM, Qi P (2009) Structure and function of protein-based edible films and coatings. In: Edible films and coatings for food applications. Springer, New York, NY, pp 25–56. https://doi.org/10.1007/978-0-387-92824-1_2
Mohamed SA, El-Sakhawy M, El-Sakhawy MAM (2020) Polysaccharides, protein and lipid-based natural edible films in food packaging: a review. Carbohydr Polym 238:116178. https://doi.org/10.1016/j.carbpol.2020.116178
Mohamed SA, El-Sakhawy M, Nashy ESH, Othman AM (2019) Novel natural composite films as packaging materials with enhanced properties. Int J Biol Macromol 136:774–784. https://doi.org/10.1016/j.ijbiomac.2019.06.130
Teramoto H, Kakazu A, Yamauchi K, Asakura T (2007) Role of hydroxyl side chains in Bombyx mori silk sericin in stabilizing its solid structure. Macromolecules 40:1562–1569. https://doi.org/10.1021/ma062604e
Wang J, Shang J, Ren F, Leng X (2010) Study of the physical properties of whey protein: sericin protein-blended edible films. Eur Food Res Technol 231:109–116. https://doi.org/10.1007/s00217-010-1259-x
Marelli B, Brenckle MA, Kaplan DL, Omenetto FG (2016) Silk fibroin as edible coating for perishable food preservation. Sci Rep 6:25263. https://doi.org/10.1038/srep25263
Gontard N, Guilbert S, Cuq JL (1993) Water and glycerol as plasticizers affect mechanical and water vapor barrier properties of an edible wheat gluten film. J Food Sci 58:206–211. https://doi.org/10.1111/j.1365-2621.1993.tb03246.x
Embuscado ME, Huber KC (2009) Edible films and coatings for food applications, vol 222. Springer, London, New York
Guerrero P, Hanani ZN, Kerry JP, De La Caba K (2011) Characterization of soy protein-based films prepared with acids and oils by compression. J Food Eng 107:41–49. https://doi.org/10.1016/j.jfoodeng.2011.06.003
Shukla R, Cheryan M (2001) Zein: the industrial protein from corn. Ind Crops Prod 13:171–192. https://doi.org/10.1016/S0926-6690(00)00064-9
Azeredo HM, Mattoso LHC, Wood D, Williams TG, Avena‐Bustillos RJ, McHugh TH (2009) Nanocomposite edible films from mango puree reinforced with cellulose nanofibers. J Food Sci 74:N31–N35. https://doi.org/10.1111/j.1750-3841.2009.01186.x
McHugh TH, Huxsoll CC, Krochta JM (1996) Permeability properties of fruit puree edible films. J Food Sci 61:88–91. https://doi.org/10.1111/j.1365-2621.1996.tb14732.x
Mondal K, Ghosh T, Bhagabati P, Katiyar V (2019) Sustainable nanostructured materials in food packaging. In: Dynamics of advanced sustainable nanomaterials and their related nanocomposites at the bio-nano interface. Elsevier, Amsterdam, pp 171–213. https://doi.org/10.1016/B978-0-12-819142-2.00008-2
Valdés A, Burgos N, Jiménez A, Garrigós MC (2015) Natural pectin polysaccharides as edible coatings. Coatings 5:865–886. https://doi.org/10.3390/coatings5040865
Soares NM, Fernandes TA, Vicente AA (2016) Effect of variables on the thickness of an edible coating applied on frozen fish–establishment of the concept of safe dipping time. J Food Eng 171:111–118. https://doi.org/10.1016/j.jfoodeng.2015.10.016
Sipahi RE, Castell-Perez ME, Moreira RG, Gomes C, Castillo A (2013) Improved multilayered antimicrobial alginate-based edible coating extends the shelf life of fresh-cut watermelon (Citrullus lanatus). LWT-Food Sci Technol 51:9–15. https://doi.org/10.1016/j.lwt.2012.11.013
Cisneros-Zevallos L, Krochta JM (2003) Dependence of coating thickness on viscosity of coating solution applied to fruits and vegetables by dipping method. J Food Sci 68:503–510. https://doi.org/10.1111/j.1365-2621.2003.tb05702.x
Bazargani-Gilani B, Aliakbarlu J, Tajik H (2015) Effect of pomegranate juice dipping and chitosan coating enriched with Zataria multiflora Boiss essential oil on the shelf-life of chicken meat during refrigerated storage. Innov Food Sci Emerg Technol 29:280–287. https://doi.org/10.1016/j.ifset.2015.04.007
Petrou S, Tsiraki M, Giatrakou V, Savvaidis IN (2012) Chitosan dipping or oregano oil treatments, singly or combined on modified atmosphere packaged chicken breast meat. Int J Food Microbiol 156:264–271. https://doi.org/10.1016/j.ijfoodmicro.2012.04.002
Zhong Y, Cavender G, Zhao Y (2014) Investigation of different coating application methods on the performance of edible coatings on Mozzarella cheese. LWT-Food Sci Technol 56:1–8. https://doi.org/10.1016/j.lwt.2013.11.006
De León-Zapata MA, Sáenz-Galindo A, Rojas-Molina R, Rodríguez-Herrera R, Jasso-Cantu D, Aguilar CN (2015) Edible candelilla wax coating with fermented extract of tarbush improves the shelf life and quality of apples. Food Packag Shelf Life 3:70–75. https://doi.org/10.1016/j.fpsl.2015.01.001
Dave RK, Rao TR, Nandane AS (2017) Improvement of post-harvest quality of pear fruit with optimized composite edible coating formulations. J Food Sci Tech 54:3917–3927. https://doi.org/10.1007/s13197-017-2850-y
Del-Valle V, Hernández-Muñoz P, Guarda A, Galotto MJ (2005) Development of a cactus-mucilage edible coating (Opuntia ficus indica) and its application to extend strawberry (Fragaria ananassa) shelf-life. Food Chem 91:751–756. https://doi.org/10.1016/j.foodchem.2004.07.002
Rojas-Graü MA, Raybaudi-Massilia RM, Soliva-Fortuny RC, Avena-Bustillos RJ, McHugh TH, Martín-Belloso O (2007) Apple puree-alginate edible coating as carrier of antimicrobial agents to prolong shelf-life of fresh-cut apples. Postharvest Biol Technol 45:254–264. https://doi.org/10.1016/j.postharvbio.2007.01.017
Ghosh T, Teramoto Y, Katiyar V (2019) Influence of nontoxic magnetic cellulose nanofibers on chitosan based edible nanocoating: a candidate for improved mechanical, thermal, optical, and texture properties. J Agric Food Chem 67:4289–4299. https://doi.org/10.1021/acs.jafc.8b05905
Oms-Oliu G, Soliva-Fortuny R, Martín-Belloso O (2008) Edible coatings with antibrowning agents to maintain sensory quality and antioxidant properties of fresh-cut pears. Postharvest Biol Technol 50:87–94. https://doi.org/10.1016/j.postharvbio.2008.03.005
Roller S, Seedhar P (2002) Carvacrol and cinnamic acid inhibit microbial growth in fresh-cut melon and kiwifruit at 4 and 8 C. Lett Appl Microbiol 35:390–394. https://doi.org/10.1046/j.1472-765X.2002.01209.x
Menezes J, Athmaselvi KA (2016) Polysaccharide based edible coating on sapota fruit. Int Agrophys 30:4
Meng X, Li B, Liu J, Tian S (2008) Physiological responses and quality attributes of table grape fruit to chitosan preharvest spray and postharvest coating during storage. Food Chem 106:501–508. https://doi.org/10.1016/j.foodchem.2007.06.012
Silva-Vera W, Zamorano-Riquelme M, Rocco-Orellana C, Vega-Viveros R, Gimenez-Castillo B, Silva-Weiss A, Osorio-Lira F (2018) Study of spray system applications of edible coating suspensions based on hydrocolloids containing cellulose nanofibers on grape surface (Vitis vinifera L.). Food Bioproc Tech 11:1575–1585. https://doi.org/10.1007/s11947-018-2126-1
Andrade RD, Skurtys O, Osorio FA (2012) Atomizing spray systems for application of edible coatings. Compr Rev Food Sci Food Saf 11:323–337. https://doi.org/10.1111/j.1541-4337.2012.00186.x
Andrade R, Skurtys O, Osorio F (2013) Drop impact behavior on food using spray coating: fundamentals and applications. Food Res Int 54:397–405. https://doi.org/10.1016/j.foodres.2013.07.042
Park JH, Choi S, Moon HC, Seo H, Kim JY, Hong SP, Lee BS, Kang E, Lee J, Ryu DH, Choi IS (2017) Antimicrobial spray nanocoating of supramolecular Fe (III)-tannic acid metal-organic coordination complex: applications to shoe insoles and fruits. Sci Rep 7:6980. https://doi.org/10.1038/s41598-017-07257-x
Khan MKI, Schutyser M, Schroën K, Boom R (2014) Barrier properties and storage stability of edible coatings prepared with electrospraying. Innov Food Sci Emerg Technol 23:182–187. https://doi.org/10.1016/j.ifset.2014.03.001
Peretto G, Du WX, Avena-Bustillos RJ, Berrios JDJ, Sambo P, McHugh TH (2017) Electrostatic and conventional spraying of alginate-based edible coating with natural antimicrobials for preserving fresh strawberry quality. Food Bioproc Tech 10:165–174. https://doi.org/10.1007/s11947-016-1808-9
Khan MKI, Cakmak H, Tavman Ş, Schutyser M, Schroёn K (2014) Anti-browning and barrier properties of edible coatings prepared with electrospraying. Innov Food Sci Emerg Technol 25:9–13. https://doi.org/10.1016/j.ifset.2013.10.006
Khan MKI, Maan AA, Schutyser M, Schroën K, Boom R (2013) Electrospraying of water in oil emulsions for thin film coating. J Food Eng 119:776–780. https://doi.org/10.1016/j.jfoodeng.2013.05.027
Çakmak H (2017) Production of emulsion based edible coatings using electrospray method and their effects on the shelf-life of some fruits
Misir J, Brishti FH, Hoque MM (2014) Aloe vera gel as a novel edible coating for fresh fruits: a review. Am J Food Sci Technol 2:93–97. https://doi.org/10.12691/ajfst-2-3-3
Khare AK, Abraham RJ, Rao VA, Babu RN (2016) Utilization of carrageenan, citric acid and cinnamon oil as an edible coating of chicken fillets to prolong its shelf life under refrigeration conditions. Vet World 9:166–175. https://doi.org/10.14202/vetworld.2016.166-175
Kusrini E, Usman A, Wisakanti CD, Arbianti R, Nasution DA (2015) Improvement of quality of Carica papaya L. with clove oil as preservative in edible coating technology. Makara J Technol 19:148–152. https://doi.org/10.7454/mst.v19i3.3047
Ghanbarzadeh B, Almasi H, Entezami AA (2011) Improving the barrier and mechanical properties of corn starch-based edible films: effect of citric acid and carboxymethyl cellulose. Ind Crops Prod 33:229–235. https://doi.org/10.1016/j.indcrop.2010.10.016
Maran JP, Sivakumar V, Sridhar R, Thirugnanasambandham K (2013) Development of model for barrier and optical properties of tapioca starch based edible films. Carbohydr Polym 92:1335–1347. https://doi.org/10.1016/j.carbpol.2012.09.069
Maran JP, Sivakumar V, Sridhar R, Immanuel VP (2013) Development of model for mechanical properties of tapioca starch based edible films. Ind Crops Prod 42:159–168. https://doi.org/10.1016/j.indcrop.2012.05.011
Galus S, Kadzińska J (2015) Food applications of emulsion-based edible films and coatings. Trends Food Sci Technol 45:273–283. https://doi.org/10.1016/j.tifs.2015.07.011
Mu C, Guo J, Li X, Lin W, Li D (2012) Preparation and properties of dialdehyde carboxymethyl cellulose crosslinked gelatin edible films. Food Hydrocoll 27:22–29. https://doi.org/10.1016/j.foodhyd.2011.09.005
Krishna M, Nindo CI, Min SC (2012) Development of fish gelatin edible films using extrusion and compression molding. J Food Eng 108:337–344. https://doi.org/10.1016/j.jfoodeng.2011.08.002
Belyamani I, Prochazka F, Assezat G (2014) Production and characterization of sodium caseinate edible films made by blown-film extrusion. J Food Eng 121:39–47. https://doi.org/10.1016/j.jfoodeng.2013.08.019
Colak BY, Peynichou P, Galland S, Oulahal N, Assezat G, Prochazka F, Degraeve P (2015) Active biodegradable sodium caseinate films manufactured by blown-film extrusion: effect of thermo-mechanical processing parameters and formulation on lysozyme stability. Ind Crops Prod 72:142–151. https://doi.org/10.1016/j.indcrop.2014.12.047
Liu L, Jin T, Liu CK, Hicks K, Mohanty AK, Bhardwaj R, Misra M (2008) A preliminary study on antimicrobial edible films from pectin and other food hydrocolloids by extrusion method. J Nat Fibers 5:366–382. https://doi.org/10.1080/15440470802460643
Liu L, Kerry JF, Kerry JP (2006) Effect of food ingredients and selected lipids on the physical properties of extruded edible films/casings. Int J Food Sci Tech 41:295–302. https://doi.org/10.1111/j.1365-2621.2005.01063.x
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Ghosh, T., Das, D., Katiyar, V. (2021). Edible Food Packaging: Targeted Biomaterials and Synthesis Strategies. In: Nanotechnology in Edible Food Packaging. Materials Horizons: From Nature to Nanomaterials. Springer, Singapore. https://doi.org/10.1007/978-981-33-6169-0_2
Download citation
DOI: https://doi.org/10.1007/978-981-33-6169-0_2
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-33-6168-3
Online ISBN: 978-981-33-6169-0
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)