Abstract
Recently, carbohydrates and/or sugars have emerged as crucial components for improving plant tolerance to abiotic stress. Abiotic stressors such as drought, salinity, severe temperature, and so on can create an accumulation of soluble sugars as well as sugar alcohols or polyols. In particular, sugars function as storage compounds, energy reservoirs, structural components, and plant signaling molecules. In addition to their accumulation, sugar transport via transporters performs important functions in overall plant growth and development at different levels. Several studies have shown their important role in plant adaptation to various abiotic conditions. We tried to include and emphasize the significance of sugar(s) signaling and their various roles in plant abiotic stress tolerance. This chapter also examines some of the key regulatory aspects of sugar metabolic pathways and the challenges and impediments to enhancing abiotic stress tolerance by manipulating sugar metabolism. Several biotechnological research in the post-genomics age can assist in developing climate-resilient crop plants under various abiotic stressors. Such techniques for agricultural enhancement, sustainable agriculture, and producing stress-tolerant crops were considered. In a demanding context, we also highlight potential scientific challenges and future research directions in the involvement of plant sugar biology in enhancing abiotic stress.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
12.1 Introduction
Owing to the sedentary nature of plants, their exposure to environmental constraints is inevitable. These stressful environmental conditions, which include drought or low water accessibility; extreme temperature (heat or cold); inadequate light; soil pH, structure, or texture; and the availability of ions in the soil, are commonly called abiotic factors (Rosa et al. 2009a, b; Lunn et al. 2014; Salvi et al. 2022). These factors are expected to reduce global food yields by more than half and harm more than 80% of the world’s land surface (Cramer et al. 2011). Majorly the mechanism of any abiotic stress in plants involves three basic stages: sensing, signaling, and response (Gangola and Ramadoss 2018). When any of the abiotic variables are experienced by plants, their first response is to sense the change or adverse condition through numerous physical and biochemical processes. After sensing, with the aid of secondary messengers like calcium, reactive oxygen species (ROS), ADP, etc. trigger and amplify the plant cell’s signaling cascade, which activates the resistance or responsive machinery and leads to the third phase, i.e., response. The third phase encompasses the alterations in the physiological activities of plant cells. Persisting unfavorable or extreme conditions result in sets of changes like reduction in photosynthesis ability, inhibition of water transport, deficiency symptoms, overaccumulation of ions, ROS outburst, etc. that collectively affect the plant growth and development (Van den Ende and El-Esawe 2014). As a result, abiotic stress is one of the most severe threats to agricultural crop productivity, and it must be addressed on a priority basis to feed the world’s rising population (Bevan et al. 2017).
New strategies for designing varieties or cultivars with desirable traits which can endure and tolerate maximum production potential have become important. Even though most abiotic stressors are complicated and multigenic regulated, significant progress in breeding resistant crops has been accomplished. However, climate change-related issues have forced the use of new technologies to understand better stress perception, signal transduction, and plant stress tolerance systems (Zhang et al. 2018c; Vats et al. 2022). Carbohydrates and/or sugars have emerged as promising components for enhancing or boosting plant tolerance to abiotic stress in recent years (Sami et al. 2016; Kaur et al. 2021; Salvi et al. 2022). Carbohydrates are the fundamental cellular elements, characterized by the basic chemical formula [Cx(H2O)y], and contain carbon hydrates (Hernandez-Marin and Martínez 2012). Sugars are polyhydroxy aldehydes or ketones that have been classified mainly by molecular size, individual monomer properties, degree of polymerization (DP), and type of linkages. Based on the characteristics above, sugars are divided into four groups: monosaccharides (DP 1), disaccharides (DP 2), oligosaccharides (DP 3–9), and polysaccharides (DP >10) (Cummings and Stephen 2007). Sugars have a role in various metabolic, structural, and physiological aspects of a plant’s growth and development. They function as storage compounds as reserve energy, energy reserves to sink organs, and as a precursor for various metabolic activities (Gangola and Ramadoss 2018). They also function as osmoprotectants and a regulatory molecular switch for regulating many genes involved in the abiotic stress tolerance mechanism (Rosa et al. 2009b). So, they have been highly investigated for their crucial function in abiotic stress resistance and/or tolerance in the recent decade. Carbohydrate partitioning is the sugar absorption, transport, and distribution process from the source (leaves) to sink or storage organs that requires energy (Slama et al. 2015; Kaur et al. 2021). Plants may also govern glucose partitioning via several transporters, which coordinate signals in different stress responses, including biotic and abiotic stress (Diehn et al. 2019). Sucrose transporters (SUT), monosaccharide transporters (MST), and sugars will be exported transporter (SWEET) are examples of these (Chen et al. 2010; Salvi et al. 2022). At multiple levels, sucrose transporters are closely controlled, allowing plants to adjust to environmental stimuli such as light regime, temperature, pathogen attack, etc. These findings highlight the need to combine abiotic stress and sugar signaling into a functional paradigm and develop techniques to improve abiotic stress tolerance using biotechnological technologies (Saddhe et al. 2021).
This chapter highlights the importance of sugar(s) signaling and their diverse role as well as sugar partitioning via sugar transporters during plant abiotic stress tolerance. This chapter also discusses some important regulatory facets of sugar metabolic pathways and the challenges and obstacles in engineering the metabolic sugar process for improving abiotic stress tolerance. Several biotechnological studies can aid in developing climate-resilient crop plants under different abiotic stresses in the post-genomics era. We discussed such approaches for crop improvement, sustainable agriculture, and developing stress-tolerant crops. We also discuss possible scientific problems and future research paths in plant sugar transporter biology in a stressful environment.
12.2 Sugar and Its Associated Components in Plant: An Overview
Plants use light energy to fix water and carbon dioxide in their chloroplasts via photosynthesis, and sugars are formed. The plant produces various sugars that can be used for structural and non-structural purposes. Like cellulose and hemicelluloses, long-chain molecules are made up of structural carbohydrates that contribute to plant structure and biomass (Hartmann and Trumbore 2016). On the contrary, monosaccharides (trioses, tetroses, pentoses, and hexoses), disaccharides (sucrose, trehalose, and maltose), oligosaccharides (stachyose, raffinose), and polysaccharides (raffinose, stachyose) are non-structural or soluble sugars that regulate a variety of functions like energy reserve, precursors for many metabolic compounds, a signaling molecule, as well as an osmoprotectants (Salmon et al. 2020). Sucrose is the most important storage and transport molecule in most plants due to its non-reducing and little chemical activity. It consists of one glucose and fructose molecule that are connected by (1–2) glycosidic bond (Chibbar et al. 2016). Sucrose can be transported in either a symplastic or apoplastic manner to sink tissues and phloem cells. It can be maintained in the vacuole by tonoplast transporters or metabolized into glucose and fructose by invertase (Rosa et al. 2009b). Sucrose, along with proline and glycine-betaine, is the most prevalent osmolyte among monocot halophytes (Slama et al. 2015). In contrast, many soluble sugars like glucose, fructose, maltose, sucrose, and galactinol and sugar alcohols like mannitol, ononitol, pinitol, etc. are all prevalent osmolytes in dicot halophytes (Slama et al. 2015; Salvi et al. 2018). Next to sucrose, raffinose family oligosaccharides (RFOs) are the most prevalent soluble sugars that are found to be derivatives of galactosyl sucrose, and mainly include raffinose, stachyose, and verbascose (Martínez-Villaluenga et al. 2008; Salvi et al. 2016, 2020, 2021a). RFOs are essential photosynthetic transporter among the family members of Verbenaceae, Cucurbitaceae, Scrophulariaceae, Lamiaceae, and Oleaceae (Gangola and Ramadoss 2018).
Several abiotic stresses like drought, salinity, extreme temperature, low availability of nutrition, etc. can cause the accumulation of several soluble sugars like glucose, sucrose, trehalose, and sugar alcohols or polyols sorbitol and mannitol (Gangola and Ramadoss 2018). Sorbitol and/or mannitol are the major suitable solutes and antioxidants that protect Apium graveolens (celery) and many species of woody Rosaceae from different abiotic stresses. Glucose is a versatile signaling molecule and a metabolite that is involved in the control of various processes (Kiba et al. 2019). Hexokinase (HKX) detects glucose levels through a glucose HXK sensor, modulates cellular functions, and phosphorylates hexose carbohydrates for metabolic activity. The target of rapamycin (TOR) kinase signaling cascade controls the metabolism of stress-responsive carbohydrates such as glucose, sucrose, and starch. Also, it contains effector genes implicated in abiotic stress responses (Ahmad et al. 2020). Through HXK activity, glucose is converted to glucose 6-phosphate (G6P), which is then used to synthesize polyols such as mannitol, sorbitol, and inositol.
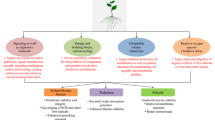
Similarly, sucrose is the most abundant sugar transportable between source and sink in plants, impacting physiological and cellular signaling pathways (Sakr et al. 2018). Several abiotic stimuli activate sucrose catabolic enzymes such as invertase and sucrose synthase (SUS), which generate sugars like fructose and glucose. Likewise, trehalose is an important disaccharide formed by two glucose molecules connected with the α-1-1 alpha bond and helps in maintaining the membrane lipids by acting as an osmolyte (Saddhe et al. 2021). Additionally, trehalose has been shown to preserve protein structure and scavenge ROS (Zulfiqar et al. 2019). Trehalose-6-phosphate (T6P) is an intermediate metabolite that plays a role in photosynthesis, sugar metabolism, and environmental response. G6P and T6P can inhibit snRK1 activity. T6P levels in cells are precisely proportional to sucrose concentrations, suggesting that T6P can act as an endogenous stimulus and control sucrose levels via a negative feedback regulation (Sakr et al. 2018). In the vacuole, fructosyltransferase (Fts) synthesizes fructans, which interact directly with the lipid group of the membrane to maintain lipid phase transitions and fluidity, contributing to cold and drought tolerance (Ahmad et al. 2020). Sugar and its associated components have a prominent and promising role in acquiring abiotic stress tolerance and can be used for further study (Fig. 12.1).
12.3 Sugar Signaling in Plant’s Metabolism
During abiotic stress tolerance, sugars serve as signaling molecules in plants and act as storage compounds, energy reservoirs, and structural molecules (Li and Sheen 2016). Sugar signaling also involves the same three basic phases of signaling mechanism sensing, signal transduction, and target gene(s) expression modulation. In plant cells, sugars are detected primarily by hexokinase (HKX)-dependent or HKX-independent mechanisms. HKX-dependent mechanisms can sense sugars with phosphorylation, whereas HKX-independent pathways can sense sugars without phosphorylation (Van den Ende and El-Esawe 2014). HKX is a multigenic family found in almost all plant species, including Arabidopsis thaliana (6), Zea mays (9), Solanum tuberosum (2), Nicotiana tabacum (9), Oryza sativa (10), Vitis vinifera (5), etc. (Paulina Aguilera-Alvarado and Sanchez-Nieto 2017; Gangola and Ramadoss 2018). Based on their subcellular location, HXKs are divided into four groups: type A HXKs (having one 30-amino-acid (aa)-long hydrophobic sequence with an N-terminal chloroplast signal), type B HXKs (having one 24-aa-long hydrophobic helix that attaches to the mitochondria), type C HXKs (lack signal peptide and membrane attachment), and type D HKX (mitochondrial HKX, but possess different peptide sequences from type B HKXs) (Paulina Aguilera-Alvarado and Sanchez-Nieto 2017). Among all four classes of HKXs, type B HXKs are the most investigated ones, commonly with nuclear-directing signals, and are critical for sugar signaling under normal and stressful environmental circumstances in plants. When glucose levels are high, the nuclear-localized HXK in collaboration with the 26S proteasome forms a glucose-signal complex that inhibits photosynthesis. However, low glucose level disrupts the HXK-mediated signal from abiotic stress. But the HXK’s intracellular sugar sensing location is still being investigated or unexplored; new findings will shed more light on the mechanism underlying (Valluru et al. 2016).
A sucrose-specific signaling route has been established to influence photosynthesis and the formation of fructan sugar and anthocyanin pigment. The balance between sucrose synthesis and degradation, which is controlled by circadian clocks and hormones in plants, determines sucrose buildup. Sucrose signaling has also been linked to additional signaling pathways activated by phytohormones like ABA and light that have been linked to calcium signaling in plants. Although no sucrose sensor has yet been found in plants, sucrose signaling is believed to be transduced to T6P signaling that controls anthocyanin production via MYB75, a transcription factor implicated in anthocyanin biosynthesis regulation (Van den Ende and El-Esawe 2014). Interestingly, HXK activity is maintained by glucose generated via invertase-catalyzed processes in the mitochondrion and cytoplasm, which supports the homeostasis of ROS (Valluru and Van den Ende 2011). SnRK1 is also a key regulator of carbon metabolism, serving as a backup supply of carbon, energy, and metabolites under abiotic stress tolerance (Emanuelle et al. 2016). SnRK1 has been demonstrated to be influenced by sugars or their derivatives, particularly glucose, G6P, and T6P. SnRK1-binding proteins have been demonstrated to regulate SnRK1 function in plant cells in a glucose-dependent manner, whereas G6P and T6P regulate SnRK1 activity via modifying SnRK1 confirmation via an unidentified intermediate molecule. Long-distance signaling in plants might be enabled via sugars and hormones (Salvi et al. 2021b). Hexokinase (HXK) and SnRK1 both interact with plant hormones, help protect plants from abiotic stressors, and are two major components of the sugar signaling cascade (Ljung et al. 2015). Transcription factor-like ABI4 and ANAC060 are two critical components of the sugar-ABA relationship. ABI4 binds to the promoters of sugar-responsive genes to control their expression. The sugar-ABA signaling route also uses ABI4 to induce the production of ANAC060, whose nuclear localization inhibits the sugar-ABA signaling pathway (Ljung et al. 2015). Auxin synthesis and signaling in plants depend on sucrose and glucose, respectively. Sucrose also links the sucrose-GA signaling cascade to brassinosteroids (BRs) and stabilizes the DELLA protein, a negative regulator of GA signaling important for plant development and stress response. In addition, starch metabolism is associated with amylase-mediated BR signaling, which functions as a maltose sensor in plant cells (Ljung et al. 2015; Gangola and Ramadoss 2018).
12.4 Molecular Roles of Sugars in Stress Tolerance
Sugars are chemically active macromolecules that play a key role in plants’ physical and chemical processes, such as respiration as respiratory agents, seed germination as energy reserve, photosynthesis as assimilatory compounds, and blooming and senescence as transporting molecules. Consequently, due to their multipurpose roles, any alteration in the sugar content in plants may help provide tolerance to several abiotic stress responses or adaption. Previous studies have also identified the sugars as playing various roles in abiotic stress, helpful in scavenging reactive oxygen species and as osmoprotectants.
12.4.1 Sugars as Scavenging Reactive Oxygen Species (ROS)
Hydrogen peroxide (H2O2), hydroxyl radicle (OH−), superoxide ion radical (O2), and singlet oxygen (O2) are the ROS examples in the living world. They are formed as a by-product of aerobic respiration, and their accumulation is in equilibrium with the plant cell’s antioxidant system under normal or stress conditions (Kwak et al. 2006). Abiotic stress, on the other hand, causes an increase in the generation of ROS in the cell, disrupting cellular redox equilibrium and leading to the breakdown of essential macromolecules such as proteins or peptides, lipids, or nucleic acid, and it represents the oxidative stress inside the plant cell (Torres et al. 2006). On the other hand, plant sugars have recently emerged as a novel class of antioxidant compounds. Monosaccharides are rarely found to act as direct antioxidants; instead, they are more likely to influence a plant cell’s antioxidative properties indirectly, either through polymerization or acting as second messengers, which increases the production or activity of various antioxidants. Sugars like sucrose, lactose, maltose, and trehalose are common disaccharides with a strong in vitro free-radical quenching effect (Bolouri-Moghaddam et al. 2010). Fructans were shown to have a higher capacity to scavenge ROS than the disaccharides studied (Peshev et al. 2013). However, while disaccharides such as sucrose appear to have the moderate antioxidant capacity, their small size and portability can play a major role in ROS control.
In addition, fructans are associated with increased accumulation of ascorbic acid salt and glutathione, suggesting that they are associated with the cytoplasmic antioxidant network (Bolouri-Moghaddam et al. 2010; Negi et al. 2017; Saxena et al. 2020). During abiotic stress, the vacuolar vesicle-derived extracellular pathway (TVE) can be used to transfer fructans from the vacuole to apoplasts, directly capturing hydroxyl radicals (Van Den Ende and Peshev 2013). In a nutshell, sucrose combines with hydroxyl radicals to generate sucrosyl radicals, which can go through four processes. Sucrosyl radicals can be converted into monosaccharide radicals and nonradicals with and without keto groups in two reactions; however, in the third reaction, sucrosyl radicals can be oxidized, giving hydrate products. Sucrosyl radicals may rejoin in the fourth step to generate distinct oligosaccharides with a greater degree of polymerization. The same principles may apply to other sugars found in plants, although no experimental evidence supports this (Gangola and Ramadoss 2018).
12.4.2 Sugars as Osmoprotectants
Major abiotic stresses like drought and salinity cause dehydration and osmotic stress to plant cells, which can cause hydrophilic connections to be disrupted, biomolecule structural breakdown (especially protein denaturation), organelle collapse, and cell membrane instability (Ozturk et al. 2021). Salt stress causes particular ions like Na+ and Cl− to become poisonous, reducing the intake of important minerals, including nitrogen, phosphorus, calcium, and potassium. The Na+/K+ ratio in the plant cell is also disrupted by Na+ toxicity, which is critical for regular cellular processes (Singh et al. 2015). Sugars like sucrose, RFOs, fructans, etc. are the osmoprotectants found in plants (Slama et al. 2015; Salvi et al. 2016). Sugar hydroxyl groups may substitute water molecules in plant cells to sustain hydrophilic contacts, which is critical for maintaining membrane integrity and structure and the native structure of macromolecules (Pukacka et al. 2009). The buildup of osmoprotective carbohydrates is thought to aid in ion partitioning and homeostasis in the plant cell, hence assisting in maintaining correct cell functioning and improving abiotic stress tolerance. Trehalose is the most promising osmotic protective sugar in terms of required concentration (Nahar et al. 2015) and can be replaced with sucrose and other sugars in plants. Sugar also helps plants develop drought-tolerant structures such as seeds and pollen. As mentioned earlier, the first way sugar provides drought tolerance is by replenishing water. “Vitrification” or glass formation in plant cells is another mechanism of desiccation tolerance. Cell solutions behave like solid plastic or highly viscous solutions. Vitrified cell solutions ensure cell stability by preventing diffusion (Angelovici et al. 2010). RFOs, coupled with LEA proteins and small HSPs, create a glassy cytosol that inhibits monosaccharide production, resulting in lower respiration and inhibition of the Maillard process (Pukacka et al. 2009; Salvi et al. 2016).
12.5 Regulation of Diverse Sugar Transporters Under Abiotic Stress
Sugar transporters play critical roles in plant growth and development at the cellular, tissue, and organ levels. Several studies have shown that they play an important role in plant adaptation to a variety of abiotic conditions (Chen et al. 2015; Saddhe et al. 2021; Salvi et al. 2022). As a result, learning their structure and function contributes to a better understanding of sugar transporters and their underlying mechanisms for developing stress-tolerant plants. The role of different sugar transporters in providing or enhancing the different abiotic stress tolerance has been summarized in Table 12.1.
12.5.1 SWEET Transporters
In plants, SWEET transporters belonging to the sugar efflux or bidirectional transporter family are known to play essential functions in pollen and seed development and nectar production (Chen et al. 2010). Significant progress has been made in understanding their distribution, phylogenetic relationships with other transporters, and structural and functional variations in several groups of plants, from algae to angiosperms, which were higher over the past decade (Doidy et al. 2012, 2019). An optical glucose sensing approach was used to identify this new family of sugar transporters in Caenorhabditis elegans, Homo sapiens, Arabidopsis thaliana, and Oryza sativa (Chen et al. 2010). Based on the number of existing MtN3 domains, all SWEET proteins should be classified into two large groups: one with two salivary MtN3 domains and the other with one salivary MtN3 domain.
The participation of the SWEET plant family in the control of sugar transport, abiotic stress tolerance, overall plant growth, seed and fruit development, and nectar secretion has achieved remarkable progress over the past decade (Jeena et al. 2019). Abiotic stressors disrupt metabolic and photosynthetic activities, disrupting sugar homeostasis. In a typical situation, plants maintain tight control over photosynthesis, sugar production, and the distribution of these substances to sink organs (Chen et al. 2012). AtSWEET15 is localized in the plasma membrane of Arabidopsis thaliana, whose transcriptional levels are significantly higher during drought, meaning that it plays a role in the release of sucrose apoplasts (Hennion et al. 2019).
AtSWEET15 is activated during leaf aging and osmotic stressors such as salt, cold, and drought via abscisic acid-dependent pathways (Julius et al. 2017). Plants that overexpress AtSWEET15 have faster leaf aging and are more susceptible to high salt stress, while AtSWEET15 variants are less susceptible to salt stress (Chen et al. 2015). Under cold, low-nitrogen conditions, studies demonstrated that AtSWEET16 and 17 largely regulate glucose or fructose levels in Arabidopsis leaf and root stem cells (Klemens et al. 2014). The single and double mutants of Arabidopsissweet11 sweet12 were more cold-tolerant than the wild type (Le Hir et al. 2015).
Wild-type Arabidopsis plants showed dramatically altered electrical conductivity compared to gene knockdown and AtSWEET4-overexpressing lines. In addition, increased hexose sugars (glucose and fructose) have been shown to protect plants from cold stress (Salvi et al. 2022). Salt stress has been reported to alter the expression of sucrose synthase (SUSY1) and several sugar transporters such as TMT and SWEET (Sellami et al. 2019). Hu et al. (2019) found that the M. truncatula genome contains 25 SWEET genes, and half showed a significant increase in transcripts during cold, salt, and drought stress. Drought, salt, and cold treatment dramatically changes the transcriptional levels of seven MtSWEET genes (Hu et al. 2019). Thirty SWEET genes have been found in Brassica oleracea, and their expression patterns suggest that five BoSWEET members are downregulated in response to cold stress (Zhang et al. 2019).
Gossypium hirsutum genome contains 55 SWEET genes, and transcript profiling reveals six GhSWEET genes with significant upregulation in heat, drought, cold, and saltwater conditions (Li et al. 2018). Transcript analysis revealed that GmSWEET6 and GmSWEET15 are highly upregulated under drought stress among 52 SWEET members of soybean (Patil et al. 2015; Du et al. 2020). Tomatoes (Solanum lycopersicum) overexpressing MdSWEET17 of apples (Malus domestica) showed increased fructose accumulation and drought tolerance (Lu et al. 2019). The SWEET gene family as a whole plays a variety of roles in stress responses and other physiological processes as well.
12.5.2 Sucrose Transporters (SUT)
The sucrose transporter is a member of one of the most important facilitator superfamilies, the glycoside pentose hexuronide (GPH) cationic symporter family (Reuscher et al. 2014). Members of the GPH family have a 12-transmembrane helix, having cytoplasmic facing N- and C-terminus. Plant growth, biomass degradation, pollen germination, fruit size control, and ethylene biosynthesis are all regulated by SUT. Nine sucrose transporter genes (SUT or SUC) have been found in Arabidopsis, but only five SUT members are in the rice genome (Kühn and Grof 2010). The sucrose transporter is involved in phloem loading in source tissue, sucrose absorption in sink cells, and migration of stored vacuoles (Slewinski et al. 2010). Several studies have also been conducted to functionally evaluate sucrose transporters for their use as candidate genes for abiotic stress tolerance (Julius et al. 2017). Low sucrose levels and salinity, osmolality, cold stress, and other abiotic stressors all cause alternation in the expression of AtSUC9 (Jia et al. 2015). In addition, the Atsuc9 mutant showed low levels of endogenous ABA under stress and suppressed ABA-inducible gene expression. Under salt stress, the Atsc4 mutant had higher levels of glucose, fructose, and sucrose in the shoots than in the roots, leading to an imbalance in sugar distribution (Gong et al. 2013). Salinity, osmotic stress, low temperature, and extrinsic abscisic acid promote AtSUC2 and AtSUC4 (Gong et al. 2015).
Rice OsSUT2 is upregulated in photosynthetic tissues under drought and salt stress, improving sucrose distribution in plants (Zhang et al. 2016). In response to drought, CBL-interacting protein kinases (CIPKs) phosphorylate the sucrose transporter MdSUT2.2 in Ser381 and Ser254 to improve salt tolerance (Chincinska et al. 2008). Overexpression of SUC27 in tobacco reduced abiotic stress by increasing the activity of reactive oxygen species and abscisic acid-related genes. Under water stress, SUT1 and SUT2 were downregulated with S. robustum, while SUT4 and SUT5 were upregulated with the leaf tissue of three Saccharum species. Drought stress has a significant impact on carbon uptake, partitioning, and tuber output in Solanum tuberosum. Under drought stress, the expression of key genes such as the sucrose transporter (StSUT2) was shown to be upregulated (Aliche et al. 2020).
12.5.3 Monosaccharide Sugar Transporter (MST)
MST is a member of the major facilitator superfamily and is involved in carbohydrate flux. These transporters contain 12 transmembrane domains. In Arabidopsis, the MST-like gene family comprises 53 genes divided into 7 subfamilies (Büttner 2010). MST regulates various physiological activities, including the distribution of sugars at the intracellular level, and is expressed in response to stress (Kong et al. 2019).
12.5.4 Sugar Transporter Protein (STP)
The STP of plants is a well-studied MST group. It is a sugar/H+ symporter in plants because it is a multipass transmembrane transporter (with 12 TM helices) (Büttner 2010). During phloem unloading, they are engaged in the absorption of hydrolyzed sucrose in the apoplast area. STP’s regulation functions under abiotic stress are well documented in the literature (Kong et al. 2019). The involvement of rice STP genes in floral development and abiotic and biotic challenges was revealed by expression analysis. OsSTP1, OsSTP3, OsSTP14, and OsSTP28 were upregulated in response to submergence, whereas OsSTP8, OsSTP11, OsSTP20, and OsSTP21 were increased in response to high temperatures. Any extremes in temperature on either side, like heat or cold and submergence stress, demonstrated upregulation of OsSTP14 (Kong et al. 2019). In a gene expression investigation, one study discovered that OsSTP2, OsSTP3, OsSTP4, OsSTP11, OsSTP19, OsSTP25, and OsSTP28 were upregulated in several abiotic responses like drought, salinity, and osmotic stress. OsSTP10, OsSTP1, and OsSTP14 were solely upregulated in response to osmotic stress (Deng et al. 2019). These investigations showed that STP has a variety of functions in drought and osmotic stress and also impacts overall plant growth and development.
12.5.5 Polyol Transporters
Polyols (also known as sugar alcohols) are sugar derivatives that can be classified as cyclic (myo-inositol, pinitol, and ononitol) or acyclic (inositol, myo-inositol, mannitol, and sorbitol) (Saxena et al. 2013; Bhattacharya and Kundu 2020). They provide a variety of physiological tasks, including carbon transfer between source and sink organs, osmoprotectant, and antioxidant defense against biotic and abiotic stressors (Noiraud et al. 2001; Bhattacharya and Kundu 2020). Polyols are thought to have osmoprotective properties by generating a hydration sphere around macromolecules, avoiding metabolic deactivation at low osmotic potential (Williamson et al. 2002; Schneider 2015). Under abiotic stress, the polyol transporters (PLT and INT) have distinct expression patterns.
In rice, OsPLT4 expression was shown to be greater in salt and drought stress than osmotic stress, whereas PLT13 expression was found to be higher in salt and osmotic stress than drought stress. In the case of OsPLT4 and 14, a similar differential expression was found. OsPLT14 was considerably upregulated during salt stress compared to osmotic and drought stress, but OsPLT3 was found upregulated under all salt, drought, and osmotic stresses (Deng et al. 2019). Similarly, under salt and osmotic stress, OsPLT13 was much more upregulated than under drought stress. Under salt stress, OsPLT14 upregulation was greater than under osmotic and dry stress. Under the three abiotic stressors, OsPLT3 was considerably upregulated. One study examined transcriptome data from two drought-tolerant Eruca vesicaria subs. sativa lines and found ERD6-like 12 transcripts were considerably upregulated when PEG treatment was applied (Hu et al. 2019). Although there are 19 ERD6-like members in Arabidopsis, only a handful have been functionally described. The varied functions of ERD6-like members in plant growth development under stress situations would be intriguing to investigate. Three TST genes are encoded by the A. thaliana genome and are found on the tonoplast membrane (Schulz et al. 2011).
In cold, drought, and salt stress, AtTMT1 and AtTMT2 were shown to be significantly upregulated. The research investigated the Beta vulgaris TST2.1 member, which is found in the vacuolar membrane and controls sucrose transport in taproot tissues via proton gradient energy (Klemens et al. 2014). Proteomics technique has been used to quantify abiotic stress-induced alterations in low abundant vacuolar transporters such as tonoplast monosaccharide transporter 2 (TMT2) and found that salt stress increased TMT2 abundance (Julius et al. 2017). Furthermore, TST2 transcript abundance was found to be highly sensitive to diverse abiotic stressors (salt, drought, and cold) (Hu et al. 2019). TST is a proton/sugar antiporter protein found in the vacuole that primarily transports glucose, fructose, and sucrose.
Furthermore, it is involved in fruit storage, organ growth, and sugar buildup in vacuoles. TST also plays an important function in maintaining cellular osmotic adjustment during abiotic stress by collecting excess carbohydrates in the vacuole. Few plant TST members have been functionally described under abiotic stress tolerance, yet additional research is needed to understand their functional diversity. In Arabidopsis, the plastid sugar transporter (pSuT) is involved in the export of glucose and sucrose (Klemens et al. 2014; Salvi et al. 2022). Chloroplast function, plant growth, and stress tolerance are all dependent on pSuT expression. This shows that, in addition to vacuolar sugar transfer, plastid sugar transport may play a role in stress tolerance development. Because there are so few studies on plastid glucose transporters, greater research on their physiological and functional insights under varied stress circumstances is essential.
12.6 Biotechnological Approaches for Developing Climate-Resilient Crop Plants in the Post-genomics Era
World agriculture faces issues as the human population grows, as well as the decrease in the agricultural land owing to industrialization, urbanization, climate change, and desertification. So far the breeding of agricultural crop plants has been beneficial in feeding an ever-increasing population; yet, 44 million metric tons of food would be required each year to feed the 9 billion people expected by 2050 (Godfray et al. 2010; Kaur et al. 2021). These yield differences are even more difficult to reconcile when it comes to the expected effects of global warming. As discussed here, sugar has an important and potential role in acquiring tolerance/resistance to different abiotic stresses. Sugar buildup in plants has long been thought to respond to abiotic stressors. It has also been well documented that to enhance stress response, abiotic stressors affect gene expression and the distribution of sugars (Gangola and Ramadoss 2018; Salvi et al. 2022).
Initially, traditional breeding methods were used to develop resistant cultivars by utilizing the genetic heterogeneity of crops at distinct gene pools. As a result, only a few abiotic stress-tolerant breeding lines in various crop species have been developed or created, most of which have failed to perform well in field testing (Manna et al. 2021). It makes traditional breeding procedures for developing stress-resistant cultivars of various agriculturally important crops more challenging (Saddhe et al. 2021). One approach was to use wild ancestors as the donor for resistance gene/s for agricultural crop manipulation to boost abiotic stress resistance. However, transferring tolerant genes for any specific abiotic resistance from wild varieties to domesticated crops is time-consuming and labor-intensive (Gangola and Ramadoss 2018; Manna et al. 2021).
Furthermore, reproductive barriers prevent beneficial genes from being passed down from wild relatives. As a result, genetic engineering has emerged as a viable option, and it is now being applied to increase abiotic stress tolerance worldwide. Recent research addressing these sugar genes’ molecular and functional control for building climate change resistance agricultural plants in various abiotic conditions are discussed in the coming sections.
12.6.1 Salt Stress
Plant physiology is altered by salt stress, which reduces cell division, photosynthesis, and nitrogen uptake, eventually affecting the plant’s overall development (Salvi et al. 2016; Kaur et al. 2021). Salinity affects 850 million hectares of land worldwide. Furthermore, salinity issues are growing at a 10% yearly rate worldwide, mostly in Asia (Ashraf and Foolad 2007). Moreover, modern agriculture and ineffective agronomic practices have resulted in increasing soil salinity of agricultural land. In most situations, saline soil has excessive Na+ and Cl− ions, which reduces water potential ion imbalance and overall plant development. Plant sugars operate as osmolytes, mitigating the negative effects of salt stress. Increases in glucose, fructose, and sucrose concentrations caused by salinity are critical for osmoprotectant, carbon storage, and ROS scavenging (Rosa et al. 2009a, b). Rice transgenics that express the trehalose gene are more resistant to several abiotic stresses, including salt, cold, and drought stress (Ashraf and Foolad 2007). Rice plants with the chimeric gene Ubi1:TPSP accumulated more trehalose, improving their resilience to salt and cold stresses (Jang et al. 2003). Mainly, trehalose-producing transgenic plants, on the other hand, exhibited pleiotropic effects that influenced other plant development pathways (Ashraf and Foolad 2007). In tobacco and wheat plants, the mt1D gene was shown to enhance salt stress resistance and mannitol accumulation (Abebe et al. 2003).
12.6.2 Drought Stress
Drought resistance breeding is undoubtedly the most challenging and time-consuming endeavor scientists face when striving to improve the genetic potential of various crop species. Drought accounts for more than 40% of crop failures, accounting for 89% of crop failures (Iordachescu and Imai 2008). Glucose improves plant resilience to drought and heat by promoting stomal closure (Osakabe et al. 2014). Furthermore, multiple investigations have discovered RFO buildup in seed desiccation events such as raffinose, verbascose, and stachyose (Bolouri-Moghaddam et al. 2010). Additionally, sugar accumulation under drought stress inhibits cell membrane oxidation (Arabzadeh 2012). Sugars also help to maintain leaf turbidity, membrane water levels, and osmotic potential (Sawhney and Singh 2002). Rice has bi-functional genes for trehalose biosynthesis that express TPP and TPS enzymes and help in the accumulation of more trehalose, which in turn is reported to increase drought, cold, and salinity tolerance in many plants (Jang et al. 2003).
12.6.3 Cold Stress
Another important ecological variable limiting plant distribution and its associated yield is temperature. Low temperatures impact the rates of reactions involved in biochemical processes differentially, resulting in metabolic pathway imbalances between partial processes. Furthermore, plants’ cold tolerance has been demonstrated to be influenced by changes in soluble sugar levels. Many soluble sugars, including sucrose, glucose, RFOs, etc., are known to give cold tolerance in plants (Jia et al. 2017). Soluble sugars also aid in acclimatization under cooling stress by interacting with lipid bilayers and aiding in their stability (Garg et al. 2002). For example, trehalose is generally present in very low concentrations, but it rises rapidly when subjected to cold stress (Fernandez et al. 2010). Moreover, sugars also influence the functions of housekeeping genes that are important throughout plant development. Advanced technologies might be employed to do more study on the role of specific or combination sugar in the cold response. These findings might help researchers better understand how sugar response pathways function during the cold stress response.
12.6.4 Heat Stress
Photosynthesis is the physiological function that suffers more when crop plants are subjected to heat stress, inhibiting overall plant development. The allocation of photoassimilates is also disrupted as a result of reduced photosynthesis. Indeed, when subjected to heat stress, the soluble glucose contents in the source leaves of many plants often decrease (Zhou et al. 2017). Sucrose transport and loading into the phloem were equally repressed in both maize and tomato plants under heat stress, suggesting that SWEETs and SUTs restrict phloem sucrose transport (Frey et al. 2015). However, in heat-stressed lemon and cucumber, glucose or fructose levels decreased, while sucrose levels increased, most likely due to increased sucrose biosynthesis (Aung et al. 2001). Heat shock proteins (HSPs) play a crucial role in how plants respond to heat stress. As per studies, sugars have a vital role in the modulation of HSP proteins, and these HSPs, in turn, regulate sugar metabolism. Heat-resistant tomato cultivars, for example, have higher invertase activity and sugar in tomato fruit (Li et al. 2012). Similarly, overexpression of the SlCIF1 gene coding for a small HSP protein resulted in a 1000-fold increase in SlHSP17.7 expression.
Furthermore, the silencing of SlCIF1 in tomatoes resulted in a drop in fructose and sucrose levels and the downregulation of numerous genes related to sugar metabolism (Zhang et al. 2018a). As a result, the plant’s response to heat stress is defined as a decrease in carbohydrate absorption followed by a drop in sugar levels in the leaves, resulting in altered sugar transporter performance (Julius et al. 2017). Heat stress regulates sugar transporters differently at different stages of development. As the temperature increased, the expression of the sucrose transporter 4 gene (OsSUT4) in embryo germination and pollen development increased. However, under prolonged heat treatment, the OsSUT4 transcript was downregulated in leaves, stems, and ears (Chung et al. 2014). They also discovered that the assimilate distribution between leaves and panicles was changed and that juvenile panicles were more susceptible to heat stress than fully matured panicles. Plasmodesmata deformation may cause delayed sucrose transport in plants under heat stress (Zhang et al. 2018b). Sugars, such as sucrose, play essential roles in thermo-tolerance control by modifying heat shock protein induction via the TOR-E2F signaling module, where E2F regulates the transcription of several HSP genes by regulating their promoters (Sharma et al. 2019, 2021).
12.7 Limitations and Challenges
Most of the studies and research were carried out on model plants like Arabidopsis and tobacco, which have demonstrated substantial resistance to various abiotic stressors. On the other hand, these model plants cannot anticipate the agriculturally significant crop plants. Although rice and wheat have been employed in different studies, they were all done under strictly controlled conditions. Most of the experiments were done when the plants were in the early stages of germination or vegetative growth. So, to better understand the significance of specific sugars and their associated gene or the signaling in crop plant abiotic stress tolerance, the practical strategy is to apply and reproduce the results directly to a crop of interest to access the gene’s true potential in the desired and natural environment (Salvi et al. 2018, 2022; Manna et al. 2021).
Furthermore, multilocation studies with the target crops are required to comprehend the activity and expression profile under natural conditions. Despite substantial efforts to produce abiotic resistant cultivars of varied agricultural plants using traditional plant breeding procedures, little progress toward the stated goal of creating viable variants has been made. It was believed that with the advent of molecular genetics and gene modification techniques, grown varieties resistant to diverse abiotic stresses and reasonably high throughput might be created, but the results are expected. Abiotic resistance features are likely to be complicated and controlled by several genes, with various biological, molecular, and physiological processes involved in abiotic resistance mechanisms.
Several studies have shed light on the significance of sugar signaling and its involvement in plant metabolism during the last few decades. The molecular basis of sugar transport, on the other hand, remains largely unknown. Despite research indicating that overexpression or downregulation of sugar transporters improves responses to a variety of abiotic stresses, efficient transformation of transporters depends on an understanding of their specific role and a virtual network with the linked biological mechanism (Salvi et al. 2016; Kaur et al. 2021). Sugar transporter modulation for increased abiotic stress responses is difficult because sugar transporters’ biological importance has been extended beyond just transporting sugar from source to sink. Some sugar transporters discovered so far also transport other substrates such as AtSWEET13 and AtSWEET14 that aid in transmitting gibberellin along with sugarMtN3/SWEET type (Kanno et al. 2016; Julius et al. 2017).
Transporter proteins play an important role in regulating many physiological processes by transporting various sugars and other metabolites. As a result, altered expression of the genes involved can adversely affect related cellular functions and developmental factors (Chen et al. 2015). Sugar signaling comprises a sophisticated network of phytohormone signaling, several transcription factors, and secondary messengers; therefore, altering sugar transporter genes may appear to have pleiotropic effects. Similarly, excessive sugar levels inside the leaves as a function of sugar exporter inhibition or downregulation could have detrimental implications on plant growth and mechanisms like photosynthesis. Reduced photosynthesis may eventually have a detrimental effect on the plant yield and also the associated environmental factors. Transforming C3 to C4 plants increases photosynthesis and output possibilities in field crops such as rice by improving CO2 fixation efficiency (Zhu et al. 2010; Baker et al. 2016). However, such a transformation would need a better knowledge of sugar transport.
12.8 Conclusions and Future Outlook
Sugars play diverse roles in plant development and mitigating unfavorable conditions. Due to their coordinated participation in stress resistance as osmoprotectants/antioxidants, role in several signaling pathways, and noteworthy relationship with photosynthesis or source-sink association, they are considered a potential target for balancing plant resilience to abiotic stresses. Sugars’ protective effect against abiotic stress has been studied to generate crop varieties with enhanced abiotic stress tolerance by altering their biosynthesis route (Kaur et al. 2021; Salvi et al. 2022). The challenge of discovering vital molecules or the genes involved, directly or indirectly, in abiotic stress tolerance has been improved by recent developments in molecular biology, particularly utilizing next-generation sequencing. However, there are few examples of generating a stable crop variety against some abiotic stress. As a result, agricultural and plant scientists must convert existing whole-genome data and omics approaches like transcriptomics, proteomics, and metabolomic data into abiotic stress-tolerant crop cultivars.
Environmental extremes caused by climate change have a recurring stress effect on plants, which has become a critical worry for maintaining high yield and plant production. Abiotic stress-tolerant cultivars have improved defense and yield due to both traditional and biotechnology techniques. Plants will need to adjust sugar transport and metabolism to counteract the detrimental effects of abiotic stressors and possess a defense arsenal. Under stress, research on the kinetics of starch to sucrose conversion has revealed multiple roles of sugars, including osmoprotectants; movement in various tissue, including sources and sink organs; and resources for long-term consideration (Kaur et al. 2021; Salvi et al. 2022). It’s also critical to understand how plants perceive and modify their cellular environment in response to specific stress such as drought, heat, or salt and how it can be interconnected when the plant senses more than one stress at a time. Stress-induced starch-sugar transformation, translocation, and relocation are also of interest, both topographically and transiently (Manna et al. 2021). Diverse pathways in these processes, both hereditarily and metabolically, might be ideal candidates for stress resistance development in agricultural plants. In any case, increased sugar accumulation might have various unintended consequences for plant development; stress-specific and tissue-specific acceptance should be addressed. In a nutshell, sugars and sugar transporters may play an important role in fine-tuning abiotic stress tolerance and agricultural productivity.
References
Abebe T, Guenzi AC, Martin B, Cushman JC (2003) Tolerance of mannitol-accumulating transgenic wheat to water stress and salinity. Plant Physiol 131:1748–1755. https://doi.org/10.1104/PP.102.003616
Ahmad F, Singh A, Kamal A (2020) Osmoprotective role of sugar in mitigating abiotic stress in plants. Prot Chem Agents Amelior Plant Abiotic Stress 53–70. https://doi.org/10.1002/9781119552154.CH3
Aliche EB, Theeuwen TPJM, Oortwijn M et al (2020) Carbon partitioning mechanisms in POTATO under drought stress. Plant Physiol Biochem 146:211–219. https://doi.org/10.1016/J.PLAPHY.2019.11.019
Angelovici R, Galili G, Fernie AR, Fait A (2010) Seed desiccation: a bridge between maturation and germination. Trends Plant Sci 15:211–218
Arabzadeh N (2012) Physiologic responses of haloxylon aphyllum to consecutive tensions of dryness and study of their role in improving resistance to dryness of vase twigs. Asian J Plant Sci 11:28–35. https://doi.org/10.3923/AJPS.2012.28.35
Ashraf M, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59:206–216. https://doi.org/10.1016/J.ENVEXPBOT.2005.12.006
Aung LH, Jenner JF, Houck LG (2001) Heat shock induced changes in exocarp soluble sugars of lemons from two climatic regions. J Hortic Sci Biotechnol 76:107–111
Baker EJ, Miles EA, Burdge GC et al (2016) Metabolism and functional effects of plant-derived omega-3 fatty acids in humans. Prog Lipid Res 64:30–56
Bevan MW, Uauy C, Wulff BBH et al (2017) Genomic innovation for crop improvement. Nature 543(7645):346–354. https://doi.org/10.1038/nature22011
Bhattacharya S, Kundu A (2020) Sugars and sugar polyols in overcoming environmental stresses. Prot Chem Agents Amelior Plant Abiotic Stress 71–101. https://doi.org/10.1002/9781119552154.CH4
Bolouri-Moghaddam MR, Le Roy K, Xiang L et al (2010) Sugar signalling and antioxidant network connections in plant cells. FEBS J 277:2022–2037
Büttner M (2010) The Arabidopsis sugar transporter (AtSTP) family: an update. Plant Biol 12:35–41. https://doi.org/10.1111/J.1438-8677.2010.00383.X
Cai Y, Tu W, Zu Y et al (2017) Overexpression of a grapevine sucrose transporter (VvSUC27) in tobacco improves plant growth rate in the presence of sucrose In vitro. Front Plant Sci 8:1069. https://doi.org/10.3389/FPLS.2017.01069/BIBTEX
Cai Y, Yin L, Tu W et al (2021) Ectopic expression of VvSUC27 induces stenospermocarpy and sugar accumulation in tomato fruits. Front Plant Sci 12:759047. https://doi.org/10.3389/FPLS.2021.759047/FULL
Cao H, Guo S, Xu Y et al (2011) Reduced expression of a gene encoding a Golgi localized monosaccharide transporter (OsGMST1) confers hypersensitivity to salt in rice (Oryza sativa). J Exp Bot 62:4595–4604. https://doi.org/10.1093/jxb/err178
Chen LQ, Hou BH, Lalonde S et al (2010) Sugar transporters for intercellular exchange and nutrition of pathogens. Nature 468:527–532. https://doi.org/10.1038/nature09606
Chen LQ, Qu XQ, Hou BH et al (2012) Sucrose efflux mediated by SWEET proteins as a key step for phloem transport. Science (80-) 335:207–211. https://doi.org/10.1126/science.1213351
Chen L-Q, Cheung LS, Feng L et al (2015) Transport of sugars. Annu Rev Biochem 84:865–894. https://doi.org/10.1146/annurev-biochem-060614-033904
Chibbar RN, Jaiswal S, Gangola M, Båga M (2016) Carbohydrate metabolism. Encycl Food Grains Second Ed 2–4:161–173. https://doi.org/10.1016/B978-0-12-394437-5.00089-9
Chincinska IA, Liesche J, Krügel U et al (2008) Sucrose transporter StSUT4 from potato affects flowering, tuberization, and shade avoidance response. Plant Physiol 146:515–528. https://doi.org/10.1104/pp.107.112334
Chung P, Hsiao HH, Chen HJ et al (2014) Influence of temperature on the expression of the rice sucrose transporter 4 gene, OsSUT4, in germinating embryos and maturing pollen. Acta Physiol Plant 36:217–229. https://doi.org/10.1007/S11738-013-1403-X
Cramer GR, Urano K, Delrot S et al (2011) Effects of abiotic stress on plants: a systems biology perspective. BMC Plant Biol 11:163. https://doi.org/10.1186/1471-2229-11-163
Cummings JH, Stephen AM (2007) Carbohydrate terminology and classification. Eur J Clin Nutr 61:S5–S18
Deng X, An B, Zhong H et al (2019) A novel insight into functional divergence of the MST gene family in rice based on comprehensive expression patterns. Genes 10:239. https://doi.org/10.3390/GENES10030239
Diehn TA, Bienert MD, Pommerrenig B, Liu Z, Spitzer C, Bernhardt N, Fuge J, Bieber A, Richet N, Chaumont F, Bienert GP (2019) Boron demanding tissues of Brassica napus express specific sets of functional Nodulin26-like Intrinsic Proteins and BOR 1 transporters. Plant J 100:68–82
Doidy J, Grace E, Kühn C et al (2012) Sugar transporters in plants and in their interactions with fungi. Trends Plant Sci 17:413–422
Doidy J, Vidal U, Lemoine R (2019) Sugar transporters in Fabaceae, featuring SUT MST and SWEET families of the model plant Medicago truncatula and the agricultural crop Pisum sativum. PLoS One 14:e0223173. https://doi.org/10.1371/JOURNAL.PONE.0223173
Du Y, Zhao Q, Chen L et al (2020) Effect of drought stress on sugar metabolism in leaves and roots of soybean seedlings. Plant Physiol Biochem 146:1–12. https://doi.org/10.1016/J.PLAPHY.2019.11.003
Durand M, Porcheron B, Hennion N et al (2016) Water deficit enhances C export to the roots in Arabidopsis thaliana plants with contribution of sucrose transporters in both shoot and roots. Plant Physiol 170:1460–1479. https://doi.org/10.1104/PP.15.01926
Emanuelle S, Doblin MS, Stapleton DI et al (2016) Molecular insights into the enigmatic metabolic regulator, SnRK1. Trends Plant Sci 21:341–353. https://doi.org/10.1016/J.TPLANTS.2015.11.001
Fernandez O, Béthencourt L, Quero A et al (2010) Trehalose and plant stress responses: friend or foe? Trends Plant Sci 15:409–417. https://doi.org/10.1016/J.TPLANTS.2010.04.004
Frey FP, Urbany C, Hüttel B et al (2015) Genome-wide expression profiling and phenotypic evaluation of European maize inbreds at seedling stage in response to heat stress. BMC Genomics 16:123. https://doi.org/10.1186/s12864-015-1282-1
Frost CJ, Nyamdari B, Tsai CJ, Harding SA (2012) The tonoplast-localized sucrose transporter in Populus (PtaSUT4) regulates whole-plant water relations, responses to water stress, and photosynthesis. PLoS One 7:44467. https://doi.org/10.1371/journal.pone.0044467
Gangola MP, Ramadoss BR (2018) Sugars play a critical role in abiotic stress tolerance in plants. In: Biochemical, physiological and molecular avenues for combating abiotic stress in plants. Elsevier, pp 17–38
Garg AK, Kim JK, Owens TG et al (2002) Trehalose accumulation in rice plants confers high tolerance levels to different abiotic stresses. Proc Natl Acad Sci U S A 99:15898–15903. https://doi.org/10.1073/pnas.252637799
Godfray HCJ, Beddington JR, Crute IR et al (2010) Food security: the challenge of feeding 9 billion people. Science 327(5967):812–818. https://doi.org/10.1126/science.1185383
Gong X, Liu ML, Wang C et al (2013) Sucrose transporter gene AtSUC4 regulates sucrose distribution and metabolism in response to salt stress in Arabidopsis thaliana. Adv Mater Res 726–731:217–221. https://doi.org/10.4028/WWW.SCIENTIFIC.NET/AMR.726-731.217
Gong X, Liu M, Zhang L et al (2015) Arabidopsis AtSUC2 and AtSUC4, encoding sucrose transporters, are required for abiotic stress tolerance in an ABA-dependent pathway. Physiol Plant 153:119–136. https://doi.org/10.1111/ppl.12225
Hartmann H, Trumbore S (2016) Understanding the roles of non-structural carbohydrates in forest trees—from what we can measure to what we want to know. New Phytol 211:386–403. https://doi.org/10.1111/NPH.13955
Hennion N, Durand M, Vriet C et al (2019) Sugars en route to the roots. Transport, metabolism and storage within plant roots and towards microorganisms of the rhizosphere. Physiol Plant 165:44–57. https://doi.org/10.1111/PPL.12751
Hernandez-Marin E, Martínez A (2012) Carbohydrates and their free radical scavenging capability: a theoretical study. J Phys Chem B 116:9668–9675. https://doi.org/10.1021/jp304814r
Hu B, Huang W, Dong L et al (2019) Molecular cloning and functional analysis of a sugar transporter gene (CsTST2) from cucumber (Cucumis sativus L.). Biotechnol Biotechnol Equip 33:118–127. https://doi.org/10.1080/13102818.2018.1555011
Iordachescu M, Imai R (2008) Trehalose biosynthesis in response to abiotic stresses. J Integr Plant Biol 50:1223–1229
Jang IC, Oh SJ, Seo JS et al (2003) Expression of a bifunctional fusion of the Escherichia coli genes for trehalose-6-phosphate synthase and trehalose-6-phosphate phosphatase in transgenic rice plants increases trehalose accumulation and abiotic stress tolerance without stunting growth. Plant Physiol 131:516–524. https://doi.org/10.1104/PP.007237
Jeena GS, Kumar S, Shukla RK (2019) Structure, evolution and diverse physiological roles of SWEET sugar transporters in plants. Plant Mol Biol 100:351–365
Jia W, Zhang L, Wu D et al (2015) Sucrose transporter AtSUC9 mediated by a low sucrose level is involved in Arabidopsis abiotic stress resistance by regulating sucrose distribution and ABA accumulation. Plant Cell Physiol 56:1574–1587. https://doi.org/10.1093/PCP/PCV082
Jia B, Zhu XF, Pu ZJ et al (2017) Integrative view of the diversity and evolution of SWEET and semiSWEET sugar transporters. Front Plant Sci 8:2178. https://doi.org/10.3389/FPLS.2017.02178
Julius BT, Leach KA, Tran TM et al (2017) Sugar transporters in plants: new insights and discoveries. Plant Cell Physiol 58:1442–1460. https://doi.org/10.1093/pcp/pcx090
Kanno Y, Oikawa T, Chiba Y et al (2016) AtSWEET13 and AtSWEET14 regulate gibberellin-mediated physiological processes. Nat Commun 7:1–11. https://doi.org/10.1038/ncomms13245
Kaur H, Manna M, Thakur T et al (2021) Imperative role of sugar signaling and transport during drought stress responses in plants. Physiol Plant 171:833–848. https://doi.org/10.1111/ppl.13364
Kiba T, Takebayashi Y, Kojima M, Sakakibara H (2019) Sugar-induced de novo cytokinin biosynthesis contributes to Arabidopsis growth under elevated CO2. Sci Rep 9:7765. https://doi.org/10.1038/S41598-019-44185-4
Klemens PAW, Patzke K, Trentmann O et al (2014) Overexpression of a proton-coupled vacuolar glucose exporter impairs freezing tolerance and seed germination. New Phytol 202:188–197. https://doi.org/10.1111/NPH.12642
Kong W, An B, Zhang Y et al (2019) Sugar Transporter Proteins (STPs) in gramineae crops: comparative analysis, phylogeny, evolution, and expression profiling. Cells 8:560. https://doi.org/10.3390/CELLS8060560
Kühn C, Grof CPL (2010) Sucrose transporters of higher plants. Curr Opin Plant Biol 13:287–297. https://doi.org/10.1016/J.PBI.2010.02.001
Kwak JM, Nguyen V, Schroeder JI (2006) The role of reactive oxygen species in hormonal responses. Plant Physiol 141:323–329. https://doi.org/10.1104/PP.106.079004
Le Hir R, Spinner L, Klemens PA et al (2015) Disruption of the sugar transporters AtSWEET11 and AtSWEET12 affects vascular development and freezing tolerance in Arabidopsis. Mol Plant 8(11):1687–1690. https://doi.org/10.1016/j.molp.2015.08.007
Li L, Sheen J (2016) Dynamic and diverse sugar signaling. Curr Opin Plant Biol 33:116–125. https://doi.org/10.1016/J.PBI.2016.06.018
Li Z, Palmer WM, Martin AP et al (2012) High invertase activity in tomato reproductive organs correlates with enhanced sucrose import into, and heat tolerance of, young fruit. J Exp Bot 63:1155–1166
Li W, Ren Z, Wang Z et al (2018) Evolution and stress responses of Gossypium hirsutum SWEET genes. Int J Mol Sci 19:769. https://doi.org/10.3390/IJMS19030769
Liu X, Zhang Y, Yang C et al (2016) AtSWEET4, a hexose facilitator, mediates sugar transport to axial sinks and affects plant development. Sci Rep 61(6):1–12. https://doi.org/10.1038/srep24563
Ljung K, Nemhauser JL, Perata P (2015) New mechanistic links between sugar and hormone signalling networks. Curr Opin Plant Biol 25:130–137. https://doi.org/10.1016/J.PBI.2015.05.022
Lu J, Sun M-h, Ma Q-j et al (2019) MdSWEET17, a sugar transporter in apple, enhances drought tolerance in tomato. J Integr Agric 18:2041–2051. https://doi.org/10.1016/S2095-3119(19)62695-X
Lunn JE, Delorge I, Figueroa CM et al (2014) Trehalose metabolism in plants. Plant J 79:544–567. https://doi.org/10.1111/TPJ.12509
Manna M, Thakur T, Chirom O et al (2021) Transcription factors as key molecular target to strengthen the drought stress tolerance in plants. Physiol Plant 172:847–868. https://doi.org/10.1111/PPL.13268
Martínez-Villaluenga C, Frias J, Vidal-Valverde C (2008) Alpha-galactosides: antinutritional factors or functional ingredients? Crit Rev Food Sci Nutr 48:301–316. https://doi.org/10.1080/10408390701326243
Miao H, Sun P, Liu Q et al (2017) Genome-wide analyses of SWEET family proteins reveal involvement in fruit development and abiotic/biotic stress responses in banana. Sci Rep 7:1–15. https://doi.org/10.1038/s41598-017-03872-w
Monfared HH, Chew JK, Azizi P et al (2020) Overexpression of a rice monosaccharide transporter gene (OsMST6) confers enhanced tolerance to drought and salinity stress in Arabidopsis thaliana. Plant Mol Biol Report 38:151–164. https://doi.org/10.1007/s11105-019-01186-x
Nahar K, Hasanuzzaman M, Fujita M (2015) Roles of osmolytes in plant adaptation to drought and salinity. Osmolytes Plants Acclim to Chang Environ Emerg Omics Technol 37–68. https://doi.org/10.1007/978-81-322-2616-1_4/COVER/
Negi B, Salvi P, Bhatt D et al (2017) Molecular cloning, in-silico characterization and functional validation of monodehydroascorbate reductase gene in Eleusine coracana. PLoS One 12:e0187793. Public Library of Science
Noiraud N, Maurousset L, Lemoine R (2001) Transport of polyols in higher plants. Plant Physiol Biochem 39:717–728
Osakabe Y, Yamaguchi-Shinozaki K, Shinozaki K, Tran LSP (2014) ABA control of plant macroelement membrane transport systems in response to water deficit and high salinity. New Phytol 202:35–49. https://doi.org/10.1111/NPH.12613
Ozturk M, Turkyilmaz Unal B, García-Caparrós P et al (2021) Osmoregulation and its actions during the drought stress in plants. Physiol Plant 172:1321–1335. https://doi.org/10.1111/ppl.13297
Patil G, Valliyodan B, Deshmukh R et al (2015) Soybean (Glycine max) SWEET gene family: insights through comparative genomics, transcriptome profiling and whole genome re-sequence analysis. BMC Genomics 16:1–16. https://doi.org/10.1186/S12864-015-1730-Y/FIGURES/5
Paulina Aguilera-Alvarado G, Sanchez-Nieto S (2017) Plant hexokinases are multifaceted proteins. Plant Cell Physiol 58:1151–1160. https://doi.org/10.1093/PCP/PCX062
Peshev D, Vergauwen R, Moglia A et al (2013) Towards understanding vacuolar antioxidant mechanisms: a role for fructans? J Exp Bot 64:1025–1038. https://doi.org/10.1093/jxb/ers377
Pukacka S, Ratajczak E, Kalemba E (2009) Non-reducing sugar levels in beech (Fagus sylvatica) seeds as related to withstanding desiccation and storage. J Plant Physiol 166:1381–1390
Reuscher S, Akiyama M, Yasuda T et al (2014) The sugar transporter inventory of tomato: genome-wide identification and expression analysis. Plant Cell Physiol 55:1123–1141. https://doi.org/10.1093/PCP/PCU052
Rosa M, Hilal M, González JA, Prado FE (2009a) Low-temperature effect on enzyme activities involved in sucrose–starch partitioning in salt-stressed and salt-acclimated cotyledons of quinoa (Chenopodium quinoa Willd.) seedlings. Plant Physiol Biochem 47:300–307. https://doi.org/10.1016/J.PLAPHY.2008.12.001
Rosa M, Prado C, Podazza G et al (2009b) Soluble sugars—metabolism, sensing and abiotic stress: a complex network in the life of plants. Plant Signal Behav 4:388–393. https://doi.org/10.4161/PSB.4.5.8294
Saddhe AA, Manuka R, Penna S (2021) Plant sugars: homeostasis and transport under abiotic stress in plants. Physiol Plant 171:739–755. https://doi.org/10.1111/PPL.13283
Sakr S, Wang M, Dédaldéchamp F et al (2018) (2018) The sugar-signaling hub: overview of regulators and interaction with the hormonal and metabolic network. Int J Mol Sci 19:2506. https://doi.org/10.3390/IJMS19092506
Salmon Y, Lintunen A, Dayet A et al (2020) Leaf carbon and water status control stomatal and nonstomatal limitations of photosynthesis in trees. New Phytol 226:690–703. https://doi.org/10.1111/NPH.16436
Salvi P, Saxena SC, Petla BP et al (2016) Differentially expressed galactinol synthase(s) in chickpea are implicated in seed vigor and longevity by limiting the age induced ROS accumulation. Sci Rep 6:35088. https://doi.org/10.1038/srep35088
Salvi P, Kamble NU, Majee M (2018) Stress-inducible galactinol synthase of chickpea (CaGolS) is implicated in heat and oxidative stress tolerance through reducing stress-induced excessive reactive oxygen species accumulation. Plant Cell Physiol 59:155. https://doi.org/10.1093/pcp/pcx170
Salvi P, Kamble NU, Majee M (2020) Ectopic over-expression of ABA-responsive Chickpea galactinol synthase (CaGolS) gene results in improved tolerance to dehydration stress by modulating ROS scavenging. Environ Exp Bot 171:103957. https://doi.org/10.1016/j.envexpbot.2019.103957
Salvi P, Kumar B, Kamble NU et al (2021a) A conserved NAG motif is critical to the catalytic activity of galactinol synthase, a key regulatory enzyme of RFO biosynthesis. Biochem J 478:3939–3955
Salvi P, Manna M, Kaur H et al (2021b) Phytohormone signaling and crosstalk in regulating drought stress response in plants. Plant Cell Rep 40:1305
Salvi P, Agarrwal R, Kajal et al (2022) Sugar transporters and their molecular tradeoffs during abiotic stress responses in plants. Physiol Plant 174(2):e13652. https://doi.org/10.1111/ppl.13652
Sami F, Yusuf M, Faizan M et al (2016) Role of sugars under abiotic stress. Plant Physiol Biochem 109:54–61
Sawhney V, Singh DP (2002) Effect of chemical desiccation at the post-anthesis stage on some physiological and biochemical changes in the flag leaf of contrasting wheat genotypes. Field Crops Res 77:1–6. https://doi.org/10.1016/S0378-4290(01)00192-7
Saxena SC, Salvi P, Kaur H et al (2013) Differentially expressed myo-inositol monophosphatase gene (CaIMP) in chickpea (Cicer arietinum L.) encodes a lithium-sensitive phosphatase enzyme with broad substrate specificity and improves seed germination and seedling growth under abiotic stresses. J Exp Bot 64:5623–5639. https://doi.org/10.1093/JXB/ERT336
Saxena SC, Salvi P, Kamble NU et al (2020) Ectopic overexpression of cytosolic ascorbate peroxidase gene (Apx1) improves salinity stress tolerance in Brassica juncea by strengthening antioxidative defense mechanism. Acta Physiol Plant 42:45. https://doi.org/10.1007/s11738-020-3032-5
Schneider S (2015) Inositol transport proteins. FEBS Lett 589:1049–1058. https://doi.org/10.1016/J.FEBSLET.2015.03.012
Schulz A, Beyhl D, Marten I et al (2011) Proton-driven sucrose symport and antiport are provided by the vacuolar transporters SUC4 and TMT1/2. Plant J 68:129–136. https://doi.org/10.1111/j.1365-313X.2011.04672.x
Sellami S, Le Hir R, Thorpe MR et al (2019) Salinity effects on sugar homeostasis and vascular anatomy in the stem of the Arabidopsis thaliana inflorescence. Int J Mol Sci 20:3167. https://doi.org/10.3390/IJMS20133167
Sharma M, Banday ZZ, Shukla BN, Laxmi A (2019) Glucose-regulated HLP1 acts as a key molecule in governing thermomemory. Plant Physiol 180:1081–1100
Sharma M, Muhammed Jamsheer K, Shukla BN et al (2021) Arabidopsis target of rapamycin coordinates with transcriptional and epigenetic machinery to regulate thermotolerance. Front Plant Sci 12:741965
Singh M, Kumar J, Singh S et al (2015) Roles of osmoprotectants in improving salinity and drought tolerance in plants: a review. Rev Environ Sci Bio/Technol 14:407–426. https://doi.org/10.1007/S11157-015-9372-8
Slama I, Abdelly C, Bouchereau A et al (2015) Diversity, distribution and roles of osmoprotective compounds accumulated in halophytes under abiotic stress. Ann Bot 115:433–447. https://doi.org/10.1093/AOB/MCU239
Slewinski TL, Garg A, Johal GS, Braun DM (2010) Maize SUT1 functions in phloem loading. Plant Signal Behav 5:687–690. https://doi.org/10.4161/PSB.5.6.11575
Torres MA, Jones JDG, Dangl JL (2006) Reactive oxygen species signaling in response to pathogens. Plant Physiol 141:373–378. https://doi.org/10.1104/PP.106.079467
Valluru R, Van den Ende W (2011) Myo-inositol and beyond—emerging networks under stress. Plant Sci 181:387–400. https://doi.org/10.1016/J.PLANTSCI.2011.07.009
Valluru R, Davies WJ, Reynolds MP, Dodd IC (2016) Foliar abscisic acid-to-ethylene accumulation and response regulate shoot growth sensitivity to mild drought in wheat. Front Plant Sci 7:461. https://doi.org/10.3389/fpls.2016.00461
Van den Ende W, El-Esawe SK (2014) Sucrose signaling pathways leading to fructan and anthocyanin accumulation: a dual function in abiotic and biotic stress responses? Environ Exp Bot 108:4–13
Van Den Ende W, Peshev D (2013) Sugars as antioxidants in plants. Crop Improv Under Advers Cond 285–307. https://doi.org/10.1007/978-1-4614-4633-0_13/COVER/
Vats S, Kumawat S, Brar J et al (2022) Opportunity and challenges for nanotechnology application for genome editing in plants. Plant Nano Biol 1:100001
Wang L, Yao L, Hao X et al (2018) Tea plant SWEET transporters: expression profiling, sugar transport, and the involvement of CsSWEET16 in modifying cold tolerance in Arabidopsis. Plant Mol Biol 96:577–592. https://doi.org/10.1007/S11103-018-0716-Y/FIGURES/7
Williamson JD, Jennings DB, Guo WW et al (2002) Sugar alcohols, salt stress, and fungal resistance: polyols—multifunctional plant protection? J Am Soc Hortic Sci 127:467–473. https://doi.org/10.21273/JASHS.127.4.467
Zhang Q, Hu W, Zhu F et al (2016) Structure, phylogeny, allelic haplotypes and expression of sucrose transporter gene families in Saccharum. BMC Genomics 17:1–18. https://doi.org/10.1186/S12864-016-2419-6/FIGURES/8
Zhang CX, Feng BH, Chen TT et al (2018a) Heat stress-reduced kernel weight in rice at anthesis is associated with impaired source-sink relationship and sugars allocation. Environ Exp Bot 155:718–733. https://doi.org/10.1016/j.envexpbot.2018.08.021
Zhang N, Shi J, Zhao H, Jiang J (2018b) Activation of small heat shock protein (SlHSP17. 7) gene by cell wall invertase inhibitor (SlCIF1) gene involved in sugar metabolism in tomato. Gene 679:90–99
Zhang Y, Li S, Xue S et al (2018c) Phylogenetic and CRISPR/Cas9 studies in deciphering the evolutionary trajectory and phenotypic impacts of rice ERECTA genes. Front Plant Sci 9:473. https://doi.org/10.3389/FPLS.2018.00473/BIBTEX
Zhang H, Dong J, Zhao X et al (2019) Research progress in membrane lipid metabolism and molecular mechanism in peanut cold tolerance. Front Plant Sci 10:838
Zhou R, Kjær KH, Rosenqvist E et al (2017) Physiological response to heat stress during seedling and anthesis stage in tomato genotypes differing in heat tolerance. J Agron Crop Sci 203:68–80. https://doi.org/10.1111/jac.12166
Zhou A, Ma H, Feng S et al (2018a) A novel sugar transporter from Dianthus spiculifolius, DsSWEET12, affects sugar metabolism and confers osmotic and oxidative stress tolerance in Arabidopsis. Int J Mol Sci 19:497. https://doi.org/10.3390/ijms19020497
Zhou A, Ma H, Feng S et al (2018b) DsSWEET17, a tonoplast-localized sugar transporter from Dianthus spiculifolius, affects sugar metabolism and confers multiple stress tolerance in Arabidopsis. Int J Mol Sci 19:1564. https://doi.org/10.3390/ijms19061564
Zhu K, Tang D, Yan C et al (2010) ERECT PANICLE2 encodes a novel protein that regulates panicle erectness in Indica rice. Genetics 184:343–350. https://doi.org/10.1534/GENETICS.109.112045
Zulfiqar F, Akram NA, Ashraf M (2019) Osmoprotection in plants under abiotic stresses: new insights into a classical phenomenon. Planta 251(1):1–17. https://doi.org/10.1007/S00425-019-03293-1
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Ethics declarations
Dr. Prafull Salvi thankfully acknowledges the research funding to his lab from the Department of Science and Technology (DST), Government of India, under the scheme of “DST-INSPIRE Faculty Award (DST/INSPIRE/04/2018/003425)” and SERB Core-Research-Grant (CRG/2021/000949), Government of India. He gratefully acknowledges the Executive Director, NABI, Mohali, for constant support. He also thanks the DBTe-Library Consortium (DeLCON) at National Agri-Food Biotechnology Institute for providing e-resource facilities.
Conflict of Interest
The authors declare no financial or commercial conflict of interest.
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Varshney, V., Singh, J., Salvi, P. (2023). Sugar Signaling and Their Interplay in Mitigating Abiotic Stresses in Plant: A Molecular Perspective. In: Sharma, D., Singh, S., Sharma, S.K., Singh, R. (eds) Smart Plant Breeding for Field Crops in Post-genomics Era . Springer, Singapore. https://doi.org/10.1007/978-981-19-8218-7_12
Download citation
DOI: https://doi.org/10.1007/978-981-19-8218-7_12
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-8217-0
Online ISBN: 978-981-19-8218-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)