Abstract
Clinically relevant depressive syndromes are estimated to occur in approximately 12% of women during pregnancy and the postpartum. Perinatal depression (PND) negatively impacts maternal self-care, nutrition, labor, and obstetric outcomes. Hormonal and physical modifications influence the circadian and homeostatic components of sleep regulation, leading to increased WASO, reduced TST, and a predominance of relatively more superficial NREM sleep, especially in the third trimester. Sleep-related breathing disorders, restless legs syndrome, insomnia, and circadian rhythm disorders also typically increase during pregnancy in predisposed women.
Of note, sleep disturbances are significantly more frequent in depressed pregnant women and might be an early predictor of mental health deterioration in the perinatal period. Therefore, sleep monitoring during pregnancy might contribute to the early detection of risk for PND. While polysomnography should be reserved for severe or diagnostically uncertain cases, sleep actigraphy can be used extensively due to its minor cost and major comfort, especially during pregnancy. Preliminary findings from this growing field of research encourage nonpharmacological approaches to sleep disturbances including clear communication of sleep hygiene to preserve positive sleep patterns, safe and effective chronotherapeutic measures such as bright light therapy to target circadian rhythm disruption, and cognitive-behavioral therapy to address insomnia. The combination of these interventions with conventional treatment approaches for PND is likely to improve mood and associated sleep disturbances in affected women.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction on Perinatal Depression
Perinatal depression (PND) is defined as a major depressive episode (MDE) with peripartum onset, that is, symptom onset during pregnancy or in the 4 weeks following delivery [1]. The extension of onset within pregnancy (antenatal depression, AD) is a major shift from the previous conceptualization of postpartum depression (PPD), classically restricted to the month following delivery. The American College of Obstetricians and Gynecologists (ACOG) has recently further extended the alert up to 1 year after delivery [2], suggesting the need to frame this disturbance in a span of 21 months (Fig. 18.1).
Current DSM-5 definition of Perinatal Depression and extension suggested by ACOG. The continuous curve indicates estimated prevalence course across the 21 months: 11.0% in the first trimester which drops to 8.5% in the second and third trimesters. After delivery, prevalence of major and minor depression begins to rise and peaks in the third month at 12.9%. In the fourth through seventh months postpartum, prevalence declines slightly, staying in the range of 9.9–10.6%, after which it declines to 6.5%. The dotted line indicates the 9.1% prevalence of any form of depression in non-pregnant women of childbearing age
PND has a pooled prevalence of 12% (95% CI 11.4–12.5), which is significantly higher in women from low- and middle-income countries compared to those from high-income countries (OR 1.8, 95% CI 1.4–2.2) [3]. PND should be clearly distinguished from the so-called “baby blues”: a brief, temporary state characterized by tearfulness and irritability that occurs in roughly 70% of new mothers. PND has been shown to negatively impact the early stages of mother–infant bonding and adversely affect child development [4]. PND is linked to poor maternal self-care, inadequate nutrition, premature labor, and adverse obstetric outcomes [5,6,7,8]. Early detection is of paramount importance given the negative consequences of untreated depression on the mother and her unborn child. Whether PND should be considered a subtype of major depression that occurs in the perinatal period rather than a different form of depression remains unclear [9]. Multiple epidemiological studies have found depression to be more common following childbirth than at other times in a woman’s life, suggesting an etiological link [10].
Several prenatal and perinatal risk factors for PND have been identified in childbearing women. The former group includes previous psychiatric disorders (especially history of PND or other mood/anxiety disorder), a family history of psychiatric disorders, a high level of neuroticism, and stressful life events in the previous year. In addition, lower socioeconomic status, low social support, daily hassles (work hassles, financial strain, time pressures), experiencing intimate partner violence, pregnancy complications, relatively younger (<24 years) or older (>35 years) age, and adverse obstetrical outcomes also increase risk for PND [11, 12].
2 Sleep and Pregnancy
Intense modifications of sleep can be observed among the several transformations that women endure during pregnancy, during which hormonal and physical modifications considerably impact both the circadian and homeostatic components of sleep regulation, leading to modifications of sleep architecture [13].
In human studies, progesterone and prolactin enhance non-rapid eye movement (NREM) sleep [14, 15], whereas rapid eye movement (REM) sleep is increased by estrogens and reduced by progesterone [16, 17]. Increased progesterone levels are also thought to be responsible for several modifications observed in sleep during the first trimester. Among these, increased urinary frequency, increased daytime sleepiness and shorter sleep onset latency (SOL) are noteworthy. Oxytocin peaks during the night, promoting uterine contractions leading to sleep fragmentation. Cortisol and growth hormone levels are also elevated, affecting sleep quality and inducing daytime sleepiness [13].
Nausea, shortness of breath, tender breasts, headache, constipation, and heartburn begin with pregnancy and often persist throughout, contributing further to sleep disruption. Sleep disruption and fragmentation typically characterize the antenatal period and the first few months after childbirth. The subjective quality of sleep is typically lower in pregnant women compared to control subjects, with perceived disruptions occurring as early as the first trimester despite an increase in sleep duration [18]. The amount of reported sleep begins to decrease in the second trimester. A recent meta-analysis showed that almost half of women experience poor sleep quality during pregnancy, with an average Pittsburgh Sleep Quality Index (PSQI) score of 6.4 (95% CI, 5.3–6.85) and a worsening trend from the second to the third trimester by an average of 1.68 points (95% CI, 0.42–2.94) [19]. However, pregnancy is a risk factor for developing major polysomnography-assessed sleep disorders only in women with predisposing factors, such as obesity or hypertension. The physiological changes occurring during pregnancy in these women may contribute to the onset of pathological conditions, especially sleep-disordered breathing.
Some evidence suggests that obstructive sleep apneas (OSAs), short sleep duration, and poor sleep quality may be associated with preterm birth (PTB). Very few available studies all point to a slight increase in the risk for stillbirth for women who sleep in supine position during the third trimester [20]. Severely disrupted sleep in the third trimester has been found to be associated with longer labors and a fivefold increase in cesarean deliveries [21].
Polysomnographic (PSG) studies have shown that sleep undergoes several physiological modifications during pregnancy. A recent systematic review on PSG in pregnancy reported a correlation between subjective worsening of sleep quality across gestation and objective changes in sleep macrostructure, which become particularly evident in the third trimester [13].
Women have been found to have shorter sleep duration, poorer sleep efficiency, more awakenings, more stage N2 sleep, less slow wave sleep, less rapid eye movement (REM) sleep, higher apnea-hypopnea index (AHI), and higher periodic limb movement (PLM) index in late compared to early pregnancy. Quantitative analyses revealed a progressive decrease of delta and theta powers and an increase of beta-2 power suggesting frequent disruption of sleep during this period [22].
Overall, the main changes identified by PSG are increased WASO, reduced TST, and a transition from N3 and REM sleep to more superficial NREM sleep stages (N1, N2). These modifications become particularly evident in the third trimester and have been confirmed both by studies comparing pregnant with age-matched nonpregnant women and by a recent large analysis of PSG data collected among the same mothers during early and late pregnancy [13].
Suboptimal sleep duration has been associated with impaired glucose tolerance and gestational diabetes mellitus (GDM). This association has been confirmed after controlling for age, trimester of pregnancy, lifestyle, and metabolic risk factors. However, pooled data from prospective as well as cross-sectional studies have shown a close relationship between extreme (i.e., ≤4 or ≥10 h per night) rather than short sleep duration during the first half of pregnancy and incident GDM [23].
3 Sleep Disorders During Pregnancy
In addition to the physiological modifications that disrupt sleep during pregnancy, specific sleep disorders may appear for the first time, or may worsen during pregnancy. A full overview of these conditions is beyond the scope of this chapter, so we will briefly outline the clinical features of the most common conditions.
3.1 Sleep-Related Breathing Disorders (SRBDs)
The spectrum of sleep-related breathing disorders includes a set of disorders characterized by irregularity of respiratory pattern and/or abnormality in the quantity of ventilation during sleep. As defined by DSM–5, SRBDs include obstructive sleep apnea/hypopnea, central sleep apnea, and sleep-related hypoventilation.
SRBDs are quite common: approximately 20% of the general population are estimated to have an apnea-hypopnea index (AHI), calculated as the number of apnea and hypopnea episodes per hour, more than 5. These disorders are two to three times more common in pregnant than nonpregnant females, particularly during the third trimester [24, 25]. Snoring, a common sleep disorder reported by 4% of nonpregnant women, increases to 25% during pregnancy [26].
The prevalence of obstructive sleep apnea hypopnea syndrome (OSAHS) is approximately 2% in nonpregnant women but suspected to rise to almost 25% during pregnancy. However, the actual prevalence of OSAHS during gestation is not known [27]. OSAHS is characterized by repetitive episodes of obstruction of the upper airway which causes partial or complete interruption of the airflow resulting in oxygen desaturations. Patients usually report snoring, witnessed apneas, nocturnal awakenings, and poor sleep quality resulting in daytime sleepiness. SRBDs may develop or worsen during pregnancy and have a negative impact on maternal and fetal health. Snoring and OSAHS during gestation have been associated with an increased risk of GDM, preeclampsia, and pregnancy-induced hypertension [27, 28].
Considering the potential negative impact of SRBDs on maternal and fetal health, and the relatively high prevalence of this disease during gestation, an assessment of sleep quality and a query on snoring and other symptoms of OSAH should always be included in prenatal care. Behavioral changes, such as lateral sleeping position and/or head elevation, treatment of nasal congestion and avoidance of sedatives, excessive weight gain and sleep deprivation, should be recommended for all cases of pregnancy SRBDs. First-line treatment for OSAHS is positive airway pressure therapy (PAP). Various devices can be proposed, differing in the way of delivering the positive airway pressure; among them, the most common provides continuous positive airway pressure (CPAP), other possibilities are the auto-titrating devices (Auto-PAP) or bilevel delivery devices (Bilevel-PAP). Considering the increasing dimensions of the pregnant uterus and the weight gain, pressure needs have to be adjusted over time during the gestation.
The presence and severity of OSAH should be reassessed in the postpartum, as the severity may improve following delivery and some women may no longer require the treatment (Table 18.1).
3.2 Restless Legs Syndrome
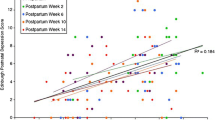
Restless legs syndrome (RLS, or Willis–Ekbom disease) is a sensory-motor disorder characterized by unpleasant leg sensation occurring at rest or in the horizontal position (sensitive component) and irresistible urge to move the legs to relieve the symptoms (motor component). The incidence of RLS in the general population is estimated to be 3.5–10%, and the syndrome is more common among women and older adults. Pregnancy is a risk factor for the emergency or exacerbation of preexisting RLS, and RLS symptoms are reported by as many as 27% of pregnant women [29]. RLS is strongly associated with sleep-wake disturbances (poor sleep quality, excessive daytime sleepiness, and poor daytime function) which are common in pregnancy and often overlap [30], as shown in Fig. 18.2.
The presence of transient RLS during gestation is a risk factor for subsequent development of chronic RLS. Furthermore, gestational RLS manifests in subsequent pregnancies in 60% of women [32].
For a long time, RLS was classified as idiopathic or secondary, depending on the presence of associated conditions. The idiopathic form is thought to be transmitted by autosomal dominant inheritance [33]. Conditions associated with RLS are iron deficiency anemia, folate deficiency, advanced renal disease, peripheral neuropathy, and pregnancy [34]. RLS can also emerge in association with Parkinson’s disease, rheumatoid arthritis, and fibromyalgia, but these associations are less common [35]. However, the concept of primary and secondary RLS has been recently challenged, and “secondary” factors are now considered as conditions that facilitate the manifestation of RLS symptoms [36].
The pathogenesis of RLS in pregnancy, as in the general population, is still unclear, but there is evidence that the underlying mechanism could involve iron deficiency, along with alterations of dopaminergic transmission [37].
The therapeutic approach depends essentially on the severity of symptoms. In case of mild or moderate new-onset RLS, the conservative approach is recommended: sleep hygiene, avoidance of dietary stimulants, stretching, and moderate exercise during daytime. Serum levels of ferritin and folate should always be assessed and treated if abnormal.
When RLS symptoms are severe, a pharmacological intervention is recommended. Nonetheless, most of the drugs approved for RLS are not considered safe during pregnancy due to the poverty of studies on gestational RLS treatment. Recently, the International RLS Study Group developed some guidelines for the treatment of RLS in pregnancy and lactation [38]. Specifically, nonpharmacological treatment such as yoga, moderate exercise, and massage should be considered as first-line treatment as well as iron implementation if serum ferritin level is <75 mcg/L. For refractory cases, medication prescription is recommended at the lowest possible dose and for the shortest possible period of time. When medications need to be considered for refractory RLS during pregnancy, the guidelines recommend low doses of benzodiazepines (second and third trimester of pregnancy), combinations of levodopa/carbidopa (25/100 mg or 50/200 mg extended release), or low doses of oxycodone for very severe, refractory RLS (first trimester) [38].
3.3 Insomnia
Pregnant women often report disrupted sleep, reduced total sleep time, and decreased quality of sleep, particularly during the third trimester. More than one in two pregnant women (52–61%) complain about insomnia on an almost nightly basis, particularly during the last 8 weeks of pregnancy [39].
Polysomnographic recordings in the third trimester confirm the presence of longer sleep latency, longer wake time after sleep onset, reduced sleep efficiency, and decreased deep sleep [40]. Patients complaining of insomnia during this period should be carefully assessed for OSA and RLS, which can present as difficulty in initiating or maintaining the sleep.
The presence of insomnia has a significantly negative impact in terms of quality of life and daytime functioning and has been linked with reduced ability to tolerate labor pain [41]. Moreover, some studies reported that women whose average sleep lasts 6 h tend to have longer labors, higher chance of cesarean sections, and increased risk of preterm birth compared to women sleeping for more than 6 h per night [21, 42].
Finally, insomnia is strongly associated with both preexisting and de novo peripartum depression [43, 44]. In case of pregnancy-related insomnia, the first recommendation is to rule out other sleep disorders, such as RLS or BRSD, or underlying depression. Therapeutic opportunities include nonpharmacological interventions based on sleep hygiene measures, dietary modifications, and sleep positioning adjustments. Some researchers reported efficacy of acupuncture, massages, yoga, and physical exercise [45]. Among nonpharmacological interventions, cognitive–behavioral therapy (CBT) is often effective and durable [46].
The use of sedative and hypnotic medications should be limited during pregnancy due to their potential for adverse effects on the developing fetus. Benzodiazepines (BDZ) and hypnotic benzodiazepine receptor agonist drugs (HBRA) may increase the risk of preterm birth, low birth weight, and/or small-for-gestational-age infants. However, lack of association between medication exposure and birth weight relative to gestational age and gender in a recent cohort of 82,038 pregnancies suggests the previously observed association with birth weight could be explained by earlier delivery rather than impaired intrauterine growth. These compounds are also generally not recommended in the third trimester to avoid neonatal withdrawal issues. Antihistamines are a possible alternative when medication is considered necessary, although the evidence for efficacy and safety is limited to very few studies.
Pharmacological treatment should be used for the shortest possible period, although, the benefits of these medications may outweigh the risks in particular cases [47].
3.4 Circadian Rhythm Sleep Disorders During Pregnancy
In physiological gestations, the normal circadian rhythm of melatonin secretion seems to be preserved, with the period of secretion proportional to the duration of darkness. The amplitude of the nocturnal increase of melatonin secretion tends to raise in the third trimester [48]. The literature concerning circadian rhythm disorders during pregnancy is scarce and essentially relies on studies of pregnant women working on rotating shifts and night-work. Circadian disruptions seem to have a negative impact during pregnancy; however, most of the evidence comes from animal studies [49]. Human studies on women exposed to shift work provided evidence of detrimental effects on pregnancy outcomes. An increased risk of fetal loss was confirmed among women working fixed night shifts, along with increased likelihood of delivering small-for-gestational-age babies, and a mean lower birthweight [48, 50, 51]. Of note, an association between circadian rhythm alterations and preeclampsia has also been reported [52].
The circadian system appears to play a role in term of labor as well. Specifically, spontaneous rupture of membranes mostly occurs during the night (12 am–4 am), and the onset of labor tends to peak around dawn and dusk, with lower rates during the day [53, 54]. In conclusion, although disruption of circadian rhythms during pregnancy appears to reflect both external (e.g. shift work) and endogenous (e.g. sleep disturbances) factors, the mechanisms leading to negative pregnancy outcomes are still unclear.
4 The Relationship Between Sleep Disturbances and Perinatal Depression
Karacan and colleagues were the first to hypothesize a link between sleep disturbances and postpartum depressed mood [55]. Empirical evidence accumulated over the following 40 years confirmed this strong relationship, with effect sizes ranging between 0.4 and 1.7 across studies [56]. Indeed, sleep disturbances have been reported to be more frequent in depressed than nondepressed pregnant women [57, 58]. During regular pregnancy, sleep can change due to physical factors (i.e., enlargement of fetus), hormonal abnormalities, or random sleep-wake patterns of the newborn.
Circadian rhythm dysregulation is a common feature among patients with mood disorders [59, 60]. However, studies on circadian rhythms during pregnancy and the postpartum remain scarce. Gonadal steroids (estrogen and progesterone) are endogenous modulators of circadian rhythm amplitude that undergo relevant modifications throughout the perinatal period. The relationship between mood and melatonin secretion, a typical marker of circadian rhythms, remains unclear. Plasma nocturnal melatonin concentrations, especially in the morning hours, were lower in depressed pregnant, but elevated in depressed postpartum women, compared with healthy controls of same gender. Melatonin timing measures were advanced in pregnant women with a personal or family history of depression. These findings implicate disturbances in the regulation of the melatonin-generating system in pregnancy and postpartum depression and suggest that abnormalities in melatonin timing parameters may be markers of vulnerability to depressive illness during pregnancy [61].
These preliminary data support circadian intervention (i.e., bright light therapy, BLT) as prevention measures in those women showing circadian vulnerability [62,63,64]. Sleep patterns during pregnancy have been studied with subjective (self-report questionnaires, rating scales, and sleep diaries) and objective sleep assessment tools such as polysomnography (PSG) and actigraphy [65,66,67]. Studies have generally shown a stronger correlation between self-perception of sleep quality and the development of depressive symptoms [68, 69]. The majority of longitudinal studies show that worsening in subjective sleep increases the likelihood of negative mood symptoms across the perinatal period [70]. Although most women report mild sleep disruption throughout the perinatal period, increased risk of depressive symptoms in the postpartum has been reported among those with a significant decline in sleep quality from early to late pregnancy and those who report poor subjective sleep quality throughout [71].
Subjective sleep quality and mood disturbances were recently assessed in 262 Chinese women from late pregnancy to 3 years after delivery with nine fixed time points of data collection [72]. This large, longitudinal study confirmed that women are vulnerable to poor sleep quality from late pregnancy onward, with disturbances persisting over time. The authors were able to distinguish three distinct trajectories that are shown in Fig. 18.3. Poor sleep quality, depression, and anxiety scores during late pregnancy appeared to anticipate the sleep trajectories. In particular, worse sleep quality predicted mood disturbances at 36 months postpartum.
Three distinct trajectories of subjective sleep quality in a cohort of 262 nonclinical pregnant women. The poorer sleep quality group demonstrated increased mood disturbances at 36 months postpartum [72]
Poor sleep quality during early stages of pregnancy may contribute to the development of higher levels of depressive symptoms close to delivery [73]. Similarly, sleep disruption in late pregnancy has been associated with more depressive symptoms in the first few weeks after childbirth [69, 74]. Even the time of delivery has been associated with emotional disturbances, that is, sleep loss due to labor and nocturnal delivery has been linked with more emotional distress in the early postpartum, when “baby blues” in most commonly experienced [74]. Some studies have shown a higher frequency of sleep-wake disturbances (poor sleep quality, daytime sleepiness, poor daytime function) in depressed compared to nondepressed pregnant women [56]. Furthermore, major depressive disorder has been associated with RLS, a typical cause of sleep disruption during pregnancy, especially in the third trimester [31, 75].
Krawczak et al. [76] compared sleep, daily activity rhythms, and mood during the third trimester and at 6–12 weeks postpartum between pregnant women with (“high-risk”) and without (“low-risk”) a history of mood disorder. By combining subjective (Edinburgh Postnatal Depression Scale, Pittsburgh Sleep Quality Index) and objective measures (actigraphy), they found that women with a history of depression reported worse mood, sleep, and daily rhythms during pregnancy; however, the only objective measure that significantly differed between the two groups was sleep efficiency (SE) in the postpartum period, suggesting a discrepancy between self-reported and objectively measured outcomes in their population. Variations of the interdaily stability, a measure of day-to-day stability of circadian rhythms, were found to correlate with day-to-day mood variation [77, 78]. High-risk women had lower variation amplitudes in pregnancy but not in the postpartum, when circadian rhythms are significantly influenced by the newborn. Sleep disturbances are reported in up to 90% of depressive episodes, but macro- and microstructural abnormalities often persist beyond the episode and have been proposed as markers which increase patients’ vulnerability to relapse and recurrence and predictors of negative therapeutic outcome [79,80,81,82,83,84]. Likewise, sleep monitoring in pregnant women might be crucial to predict the likelihood of PND given the bidirectional link between sleep and depressive symptoms [65, 85]. Furthermore, abnormalities of macro-structural parameters and micro-architectural sleep features have recently been described in infants born to depressed mothers. These findings suggest that intrinsic features of sleep might contribute to the transmission of depression vulnerability from mothers to children [86].
5 Sleep Loss and Puerperal Psychosis
Puerperal psychosis (PP) is the most severe form of postnatal psychiatric disorder observed in mothers. It is estimated to occur in 1–2/1000 women in the first 6 weeks following delivery. A previous history of puerperal psychosis or bipolar mood disorder increases the risk dramatically, to over 30% [87]. The clinical onset is often abrupt and should be carefully distinguished from neurological causes such as autoimmune encephalopathies precipitated by immunological modifications that occur during the early postpartum [88]. Indeed, puerperal psychosis often presents as a full-blown manic psychosis with racing thoughts, distractibility, insomnia, and delusions but also delirium-like signs such as disorientation or acute dissociative experiences that are more typically observed in neurological conditions. Given that sleep loss is a widely recognized trigger for both manic episodes and psychotic symptoms, the abrupt peripartum modifications of sleep are likely to play a role in the pathogenesis of puerperal psychosis [89]. Although very few studies directly addressed this relationship, some evidence suggests individual differences in the vulnerability to sleep-related mood regulation following sleep loss might explain the increased risk in women with bipolar disorder [90].
6 Routine Monitoring and Treatment
Monitoring sleep patterns during pregnancy appears to be a valuable preventive measure to detect the risk of PND. Polysomnography has provided the most objective evidence of a possible relationship between sleep disruption and depressive symptoms in the perinatal period. However, PSG should be reserved for severe or diagnostically uncertain cases. On the other hand, sleep actigraphy may be used extensively due to its minor cost and major comfort compared to PSG, especially during pregnancy. Preliminary findings from this growing field of research encourage the development of preventive interventions to ensure circadian rhythm and sleep stability throughout the perinatal period. These include clear communication of sleep hygiene to preserve positive sleep patterns, safe and effective chronotherapeutic measures such as BLT to target circadian rhythm disruption, and CBT to treat insomnia (iCBT) [91, 92]. Of note, a randomized controlled trial of almost 200 pregnant women without psychiatric morbidity confirmed that iCBT effectively reduces self-reported (but not objective) total wake time and might also have a positive impact on depressive symptoms [93].
In terms of pharmacological treatment, several available guidelines advise to continue antidepressants in women with a mood disorder who become pregnant, but clear evidence in support of this recommendation is lacking [94]. In the case of new episodes, most guidelines agree on psychosocial interventions for mild to moderate depression and pharmacological treatment with selective serotonin reuptake inhibitors (SSRIs) for severe depression. Among these, sertraline is generally recommended as first line due to its relatively low teratogenic risk, whereas paroxetine is discouraged due to a slight increase of risk for congenital heart malformations. Antidepressant treatment can be safely continued during breastfeeding, although guidelines encourage close observation of the newborn. Reliable, evidence-based information on the use of sleep medications during pregnancy and breastfeeding is still very limited [95]. Whenever appropriate, the pharmacological treatment of sleep disturbances must include an individualized assessment and cautious examination of risks and benefits of maternal and fetal pharmacologic exposure [96]. In all cases, intrapartum and immediate postpartum monitoring should be guaranteed for the newborn exposed to psychotropic medication in utero.
References
American Psychiatric Association. DSM 5 diagnostic and statistical manual of mental disorders. American Psychiatric Association (APA); 2013.
El-sayed Y, Heine RP, Wharton KR. Müllerian agenesis: diagnosis, management, and treatment. ACOG Committee Opinion No. 728. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2018;131(728):35–42.
Woody CA, Ferrari AJ, Siskind DJ, Whiteford HA, Harris MG. A systematic review and meta-regression of the prevalence and incidence of perinatal depression. J Affect Disord. 2017;219(May):86–92. https://doi.org/10.1016/j.jad.2017.05.003.
Schaffir J. Consequences of antepartum depression. Clin Obstet Gynecol. 2018;61(3):533–43.
Zuckerman B, Amaro H, Bauchner H, Cabral H. Depressive symptoms during pregnancy: relationship to poor health behaviors. Am J Obstet Gynecol. 1989;160(5):1107–11.
Chung TKH, Lau TK, Yip ASK, Chiu HFK, Lee DTS. Antepartum depressive symptomatology is associated with adverse obstetric and neonatal outcomes. Psychosom Med. 2001;63(5):830–4.
Dayan J, Creveuil C, Marks MN, Conroy S, Herlicoviez M, Dreyfus M, Tordjman S. Prenatal depression, prenatal anxiety, and spontaneous preterm birth: a prospective cohort study among women with early and regular care. Psychosom Med. 2006;68(6):938–46.
Kurki T, Hiilesmaa V, Raitasalo R, Mattila H, Ylikorkala O. Depression and anxiety in early pregnancy and risk for preeclampsia. Obstet Gynecol. 2000;95(4):487–90.
O’Brien S, Sethi A, Gudbrandsen M, et al. Is postnatal depression a distinct subtype of major depressive disorder? An exploratory study. Arch Womens Ment Health. 2000;24:329–33.
Di Florio A, Meltzer-Brody S. Is postpartum depression a distinct disorder? Curr Psychiatry Rep. 2015;17(10):1–6.
Eberhard-Gran M, Eskild A, Tambs K, Samuelsen SO, Opjordsmoen S. Depression in postpartum and non-postpartum women: prevalence and risk factors. Acta Psychiatr Scand. 2002;106(6):426–33.
Guintivano J, Manuck T, Meltzer-Brody S. Predictors of postpartum depression: a comprehensive review of the last decade of evidence. Clin Obstet Gynecol. 2018;61(3):591–603.
Garbazza C, Hackethal S, Riccardi S, Cajochen C, Cicolin A, D’Agostino A, Cirignotta F, Manconi M. Polysomnographic features of pregnancy: a systematic review. Sleep Med Rev. 2020;50:101249.
Driver HS, Dijk DJ, Werth E, Biedermann K, Borbély AA. Sleep and the sleep electroencephalogram across the menstrual cycle in young healthy women. J Clin Endocrinol Metab. 1996;81(2):728–35.
Frieboes RM, Murck H, Stalla GK, Antonijevic IA, Steiger A. Enhanced slow wave sleep in patients with prolactinoma. J Clin Endocrinol Metab. 1998;83(8):2706–10.
Thomson J, Oswald I. Effect of oestrogen on the sleep, mood, and anxiety of menopausal women. BMJ. 1977;2(6098):1317–9.
Schiff I, Regestein Q, Tulchinsky D, Ryan KJ. Effects of estrogens on sleep and psychological state of hypogonadal women. JAMA. 1979;242(22):2405–4.
Hedman C, Pohjasvaara T, Tolonen U, Suhonen-Malm AS, Myllylä V. Effects of pregnancy on mothers’ sleep. Sleep Med. 2002;3(1):37–42.
Sedov ID, Cameron EE, Madigan S, Tomfohr-Madsen LM. Sleep quality during pregnancy: a meta-analysis. Sleep Med Rev. 2018;38:168–76.
Warland J, Dorrian J, Morrison JL, O’Brien LM. Maternal sleep during pregnancy and poor fetal outcomes: a scoping review of the literature with meta-analysis. Sleep Med Rev. 2018;41:197–219.
Lee KA, Gay CL. Sleep in late pregnancy predicts length of labor and type of delivery. Am J Obstet Gynecol. 2004;191(6):2041–6.
Izci-Balserak B, Keenan BT, Corbitt C, Staley B, Perlis M, Pien GW. Changes in sleep characteristics and breathing parameters during sleep in early and late pregnancy. J Clin Sleep Med. 2018;14(7):1161–8.
Xu Y-H, Shi L, Bao Y-P, Chen S-J, Shi J, Zhang R-L, et al. Association between sleep duration during pregnancy and gestational diabetes mellitus: a meta-analysis. Sleep Med. 2018;52:67–74.
Izci B, Vennelle M, Liston WA, Dundas KC, Calder AA, Douglas NJ. Sleep-disordered breathing and upper airway size in pregnancy and post-partum. Eur Respir J. 2006;27(2):321–7.
Bourjeily G. Sleep disorders in pregnancy. Obstetric medicine. 2009;2(3):100–6.
O’Brien LM, Bullough AS, Owusu JT, Tremblay KA, Brincat CA, Chames MC, et al. Pregnancy-onset habitual snoring, gestational hypertension, and preeclampsia: prospective cohort study. Am J Obstet Gynecol. 2012;207(6):487.e1–9.
Olivarez SA, Ferres M, Antony K, Mattewal A, Maheshwari B, Sangi-Haghpeykar H, et al. Obstructive sleep apnea screening in pregnancy, perinatal outcomes, and impact of maternal obesity. Am J Perinatol. 2011;28(8):651–8.
Facco FL, Grobman WA, Kramer J, Ho KH, Zee PC. Self-reported short sleep duration and frequent snoring in pregnancy: impact on glucose metabolism. Am J Obstet Gynecol. 2010;203(2):142.e1–5.
Manconi M, Govoni V, De Vito A, Economou NT, Cesnik E, Casetta I, et al. Restless legs syndrome and pregnancy. Neurol Int. 2004;63(6):1065–9.
Dunietz GL, Lisabeth LD, Shedden K, Shamim-Uzzaman QA, Bullough AS, Chames MC, et al. Restless legs syndrome and sleep-wake disturbances in pregnancy. J Clin Sleep Med. 2017;13(7):863–70.
Lee HB, Hening WA, Allen RP, Kalaydjian AE, Earley CJ, Eaton WW, et al. Restless legs syndrome is associated with DSM-IV major depressive disorder and panic disorder in the community. J Neuropsychiatry Clin Neurosci. 2008;20(1):101–5.
Cesnik E, Casetta I, Turri M, Govoni V, Granieri E, Ferini Strambi L, et al. Transient RLS during pregnancy is a risk factor for the chronic idiopathic form. Neurology. 2010;75(23):2117–20.
Bonati MT, Ferini-Strambi L, Aridon P, Oldani A, Zucconi M, Casari G. Autosomal dominant restless legs syndrome maps on chromosome 14q. Brain. 2003;126(Pt 6):1485–92.
Allen RP, Earley CJ. Restless legs syndrome: a review of clinical and pathophysiologic features. J Clin Neurophysiol. 2001;18(2):128–47.
Bourjeily G, Raker CA, Chalhoub M, Miller MA. Pregnancy and fetal outcomes of symptoms of sleep-disordered breathing. Eur Respir J. 2010;36(4):849–55.
Trenkwalder C, Allen R, Högl B, Paulus W, Winkelmann J. Restless legs syndrome associated with major diseases: a systematic review and new concept. Neurology. 2016;86(14):1336–43.
Michaud M, Soucy JP, Chabli A, Lavigne G, Montplaisir J. SPECT imaging of striatal pre- and postsynaptic dopaminergic status in restless legs syndrome with periodic leg movements in sleep. J Neurol. 2002;249(2):164–70.
Allen RP, Picchietti DL, Garcia-Borreguero D, Ondo WG, Walters AS, Winkelman JW, et al. Restless legs syndrome/Willis–Ekbom disease diagnostic criteria: updated International Restless Legs Syndrome Study Group (IRLSSG) consensus criteria–history, rationale, description, and significance. Sleep Med. 2014;15(8):860–73.
Kizilirmak A, Timur S, Kartal B. Insomnia in pregnancy and factors related to insomnia. Sci World J. 2012;2012:197093.
Beebe KR, Lee KA. Sleep disturbance in late pregnancy and early labor. J Perinat Neonatal Nurs. 2007;21(2):103–8.
Pan PH, Lee S, Harris L. Chronobiology of subarachnoid fentanyl for labor analgesia. Anesthesiology. 2005;103(3):595–9.
Chang JJ, Pien GW, Duntley SP, Macones GA. Sleep deprivation during pregnancy and maternal and fetal outcomes: is there a relationship? Sleep Med Rev. 2010;14(2):107–14.
Dørheim SK, Bjorvatn B, Eberhard-Gran M. Insomnia and depressive symptoms in late pregnancy: a population-based study. Behav Sleep Med. 2012;10(3):152–66.
Okun ML, Kiewra K, Luther JF, Wisniewski SR, Wisner KL. Sleep disturbances in depressed and nondepressed pregnant women. Depress Anxiety. 2011;28(8):676–85.
Guerreiro Da Silva JB, Nakamura MU, Cordeiro JA, Kulay L. Acupuncture for insomnia in pregnancy—a prospective, quasi-randomised, controlled study. Acupunct Med. 2005;23(2):47–51.
Espie CA. Understanding insomnia through cognitive modelling. Sleep Med. 2007;8(Suppl 4):S3–8.
Okun ML, Ebert R, Saini B. A review of sleep-promoting medications used in pregnancy. Am J Obstet Gynecol. 2015;212(4):428–41.
Tamura H, Nakamura Y, Terron MP, Flores LJ, Manchester LC, Tan DX, et al. Melatonin and pregnancy in the human. Reprod Toxicol. 2008;25(3):291–303.
Summa KC, Vitaterna MH, Turek FW. Environmental perturbation of the Circadian clock disrupts pregnancy in the mouse. PLoS One. 2012;7(5):e37668.
Jin LZ, Hjollund NH, Andersen AMN, Olsen J. Shift work, job stress, and late fetal loss: the National Birth Cohort in Denmark. J Occup Environ Med. 2004;46(11):1144–9.
Niedhammer I, O’Mahony D, Daly S, Morrison JJ, Kelleher CC. Occupational predictors of pregnancy outcomes in Irish working women in the Lifeways cohort. BJOG An Int J Obstet Gynaecol. 2009;116(7):943–52.
Ditisheim AJ, Dibner C, Philippe J, Pechére-Bertschi A. Biological rhythms and preeclampsia. Front Endocrinol. 2013;4:47.
Ngwenya S, Lindow SW. 24 hour rhythm in the timing of pre-labour spontaneous rupture of membranes at term. Eur J Obstet Gynecol Reprod Biol. 2004;112(2):151–3.
Cagnacci A, Soldani R, Melis GB, Volpe A. Diurnal rhythms of labor and delivery in women: modulation by parity and seasons. Am J Obstet Gynecol. 1998;178(1 Pt 1):140–5.
Karacan I, Williams RL, Hursch CJ, McCaulley M, Heine MW. Some implications of the sleep patterns of pregnancy for postpartum emotional disturbances. Br J Psychiatry. 1969;115(525):929–35.
Bhati S, Richards K. A systematic review of the relationship between postpartum sleep disturbance and postpartum depression. J Obstet Gynecol Neonatal Nurs. 2015;44(3):350–7.
Swanson LM, Pickett SM, Flynn H, Armitage R. Relationships among depression, anxiety, and insomnia symptoms in perinatal women seeking mental health treatment. J Women’s Heal. 2011;20(4):553–8.
Jomeen J, Martin C. Assessment and relationship of sleep quality to depression in early pregnancy. J Reprod Infant Psychol. 2007;25(1):87–99.
Germain A, Kupfer DJ. Circadian rhythm disturbances in depression. Hum Psychopharmacol. 2008;23(7):571–85.
Palagini L, Domschke K, Benedetti F, Foster RG, Wulff K, Riemann D. Developmental pathways towards mood disorders in adult life: is there a role for sleep disturbances? J Affect Disord. 2019;243:121–32.
Parry BL, Meliska CJ, Sorenson DL, Lopez AM, Martinez LF, Nowakowski S, et al. Plasma melatonin circadian rhythm disturbances during pregnancy and postpartum in depressed women and women with personal or family histories of depression. Am J Psychiatry. 2008;165(12):1551–8.
Epperson CN, Terman M, Terman JS, Hanusa BH, Oren DA, Peindl KS, et al. Randomized clinical trial of bright light therapy for antepartum depression: preliminary findings. J Clin Psychiatry. 2004;65(3):421–5.
Oren DA, Wisner KL, Spinelli M, Epperson CN, Peindl KS, Terman JS, et al. An open trial of morning light therapy for treatment of antepartum depression. Am J Psychiatry. 2002;159(4):666–9.
Wirz-Justice A, Bader A, Frisch U, Stieglitz RD, Alder J, Bitzer J, et al. A randomized, double-blind, placebo-controlled study of light therapy for antepartum depression. J Clin Psychiatry. 2011;72(7):986–93.
Dorheim S, Bondevik GT, Eberhard-Gran M, Bjorvatn B. Sleep and depression in postpartum women. Sleep. 2009;32(7):847–55.
Goyal D, Gay CL, Lee KA. Patterns of sleep disruption and depressive symptoms in new mothers. J Perinat Neonatal Nurs. 2007;21(2):123–9.
Huang CM, Carter PA, Guo JL. A comparison of sleep and daytime sleepiness in depressed and non-depressed mothers during the early postpartum period. J Nurs Res. 2004;12(4):287–96.
Park EM, Meltzer-Brody S, Stickgold R. Poor sleep maintenance and subjective sleep quality are associated with postpartum maternal depression symptom severity. Arch Womens Ment Health. 2013;16(6):539–47.
Wolfson AR, Crowley SJ, Anwer U, Bassett JL. Changes in sleep patterns and depressive symptoms in first-time mothers: last trimester to 1-year postpartum. Behav Sleep Med. 2003;1(1):54–67.
Krawczak EM, Minuzzi L, Hidalgo MP, Frey BN. Do changes in subjective sleep and biological rhythms predict worsening in postpartum depressive symptoms? A prospective study across the perinatal period. Arch Womens Ment Health. 2016;19(4):591–8.
Tomfohr LM, Buliga E, Letourneau NL, Campbell TS, Giesbrecht GF. Trajectories of sleep quality and associations with mood during the perinatal period. Sleep. 2015;38(8):1237–45.
Wang G, Deng Y, Jiang Y, Lin Q, Dong S, Song Y, Zhu L, Zhu Q, Sun W, Zhang Y, Jiang F. Trajectories of sleep quality from late pregnancy to 36 months postpartum and association with maternal mood disturbances: a longitudinal and prospective cohort study. Sleep. 2018;41(12) https://doi.org/10.1093/sleep/zsy179.
Skouteris H, Germano C, Wertheim EH, Paxton SJ, Milgrom J. Sleep quality and depression during pregnancy: a prospective study. J Sleep Res. 2008;17(2):217–20.
Wilkie G, Shapiro CM. Sleep deprivation and the postnatal blues. J Psychosom Res. 1992;36(4):309–16.
Sarberg M, Bladh M, Svanborg E, Josefsson A. Postpartum depressive symptoms and its association to daytime sleepiness and restless legs during pregnancy. BMC Pregnancy Childb. 2016;16(1):1–8.
Krawczak EM, Minuzzi L, Simpson W, Hidalgo MP, Frey BN. Sleep, daily activity rhythms and postpartum mood: a longitudinal study across the perinatal period. Chronobiol Int. 2016;33:791–801.
Huang YL, Liu RY, Wang QS, Van Someren EJW, Xu H, Zhou JN. Age-associated difference in circadian sleep-wake and rest-activity rhythms. Physiol Behav. 2002;76(4–5):597–603.
Calogiuri G, Weydahl A, Carandente F. Methodological issues for studying the rest-activity cycle and sleep disturbances: a chronobiological approach using actigraphy data. Biol Res Nurs. 2013;15(1):5–12.
Tsuno N, Besset A, Ritchie K. Sleep and depression. J Clin Psychiatry. 2005;66(10):1254–69.
Perils ML, Giles DE, Buysse DJ, Tu X, Kupfer DJ. Self-reported sleep disturbance as a prodromal symptom in recurrent depression. J Affect Disord. 1997;42(2–3):209–12.
Lustberg L, Reynolds CF. Depression and insomnia: questions of cause and effect. Sleep Med Rev. 2000;4(3):253–62.
Clark C, Dupont R, Golshan S, Gillin JC, Rapaport MH, Kelsoe JR. Preliminary evidence of an association between increased REM density and poor antidepressant response to partial sleep deprivation. J Affect Disord. 2000;59(1):77–83.
Kupfer DJ, Frank E, Mceachran AB, Grochocinski VJ. Delta sleep ratio: a biological correlate of early recurrence in unipolar affective disorder. Arch Gen Psychiatry. 1990;47(12):1100–5.
Nissen C, Feige B, König A, Voderholzer U, Berger M, Riemann D. Delta sleep ratio as a predictor of sleep deprivation response in major depression. J Psychiatr Res. 2001;47(12):1100–5.
Lumley J, Austin MP. What interventions may reduce postpartum depression. Curr Opin Obstet Gynecol. 2001;13(6):605–11.
Bat-Pitault F, Sesso G, Deruelle C, Flori S, Porcher-Guinet V, Stagnara C, et al. Altered sleep architecture during the first months of life in infants born to depressed mothers. Sleep Med. 2017;30:195–203.
Wesseloo R, Kamperman AM, Munk-Olsen T, Pop VJM, Kushner SA, Bergink V. Risk of postpartum relapse in bipolar disorder and postpartum psychosis: a systematic review and meta-analysis. Am J Psychiatry. 2016;173(2):117–27.
Bergink V, Armangue T, Titulaer MJ, Markx S, Dalmau J, Kushner SA. Autoimmune encephalitis in postpartum psychosis. Am J Psychiatry. 2015;172(9):901–8.
Lewis KJS, Foster RG, Jones IR. Is sleep disruption a trigger for postpartum psychosis? Br J Psychiatry. 2016;208(5):409–11.
Lewis KJS, Di Florio A, Forty L, Gordon-Smith K, Perry A, Craddock N, et al. Mania triggered by sleep loss and risk of postpartum psychosis in women with bipolar disorder. J Affect Disord. 2018;225:624–9.
van Maanen A, Meijer AM, van der Heijden KB, Oort FJ. The effects of light therapy on sleep problems: a systematic review and meta-analysis. Sleep Med Rev. 2016;29:52–62.
Sedov ID, Goodman SH, Tomfohr-Madsen LM. Insomnia treatment preferences during pregnancy. J Obstet Gynecol Neonatal Nurs. 2017;46(3):e95–e104.
Manber R, Bei B, Simpson N, Asarnow L, Rangel E, Sit A, Lyell D. Cognitive behavioral therapy for prenatal insomnia: a randomized controlled trial. Obstet Gynecol. 2019;133(5):911–9.
Molenaar NM, Kamperman AM, Boyce P, Bergink V. Guidelines on treatment of perinatal depression with antidepressants: an international review. Aust N Z J Psychiatry. 2018;52(4):320–7.
Mcallister-Williams RH, Baldwin DS, Cantwell R, Easter A, Gilvarry E, Glover V, et al. British Association for Psychopharmacology consensus guidance on the use of psychotropic medication preconception, in pregnancy and postpartum 2017. J Psychopharmacol. 2017;31(5):519–52.
Huitfeldt A, Sundbakk LM, Skurtveit S, Handal M, Nordeng H. Associations of maternal use of benzodiazepines or benzodiazepine-like hypnotics during pregnancy with immediate pregnancy outcomes in Norway. JAMA Netw Open. 2020;3(6):e205860-10.1001/jamanetworkopen.2020.5860.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Fanti, V., Serrati, C., D’Agostino, A. (2022). Sleep and Perinatal Depression. In: Gupta, R., Neubauer, D.N., Pandi-Perumal, S.R. (eds) Sleep and Neuropsychiatric Disorders. Springer, Singapore. https://doi.org/10.1007/978-981-16-0123-1_18
Download citation
DOI: https://doi.org/10.1007/978-981-16-0123-1_18
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-0122-4
Online ISBN: 978-981-16-0123-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)