Abstract
Polyethylene glycol (PEG) is one of the most extensively used biocompatible polymer. PEG-modification improves the original properties of conjugates and thus being exploited in different fields. PEGs demonstrated their ability to bind DNA, dyes and proteins, in solution and in solid phase via amine and thiol groups. Covalent linkage of PEG to drug molecules improves water-solubility, bioavailability, pharmacokinetics, immunogenic properties, and biological activities. Entrapment of drugs into the PEG vesicle offers substantial benefits in the treatment of many diseases including type 2 diabetes over conventional injection-based therapies. Therapeutic enzymes are conjugated with PEG for targeted therapy of diseases in which the native enzyme was inefficient. PEG has been most extensively investigated polymers for gene delivery due to its capability to form stable complexes by electrostatic interactions with nucleic acids. Many PEG-enzymes conjugates have already obtained FDA approval for clinical implications. PEGylated copolymers have least cytotoxicity and cell-compatibility concern, high efficiency, safety and biocompatibility and thus considered as an attractive polymer for gene and drug delivery system. For instance, many tissue engineering applications, PEG and its derivatives are likely to precise control of cell behaviour in growing tissues. For this application numerous bioresponsive and intelligent biomaterials are developed and extensively used in bone and tissue regeneration. PEG-derived hydrogels increase gene expression of bone-specific markers, secretion of bone-related matrix, and mineralization and may have a potential impact on bone-engineering therapies. PEG-coated poly(amidoamine) exhibits low toxicity to human corneal epithelial cells and effectively used as antimicrobial agents.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Polyethylene glycol
- Drug delivery system
- Molecular imaging
- TISSUE engineering
- PEGylation
- Bioconjugation
- Gene therapy
- Nanocarriers
- Biomaterials
11.1 Introduction
Proteins are linear chain of amino acid residues that fold into three-dimensional structures to carry out a wide variety of function inside the cell. About 5–30% functional regions of proteins are disordered lacking well defined ensemble but majority of protein domain fold into ordered 3-dimensional conformations due to physical interaction within the chain (Dawson et al. 2017; Jones and Cozzetto 2015; Mitchell et al. 2018). The structural features of proteins, in turn, determine the broad range of functions from binding specificity, forming structure inside the cell, to catalysis of biochemical reactions, signal transduction or transport. Due to recent advance in high-throughput sequencing technology, gap is quickly growing between number of known protein sequence and number of those with experimentally characterized function. There are more than 60 million protein sequence deposited in the UniProt database (UniProt 2015), but less than 0.8% of these sequences have the function manually annotated in SwissProt (Boutet et al. 2016). Automated in silico protein function prediction thus become crucial for making use of the recent explosion of genomic sequencing data. In this chapter, we further explore the use of structural modification of protein and its application in medicine, gene delivery, pharmaceutical industries, cosmetics, food industries and bone and tissue engineering. Structural modification of protein mainly included here is PEGylation of protein surface.
Polyethylene glycol (PEG) is a semicrystalline polymer of ethylene glycol, have routinely used in biological research as crowding agent, in drug delivery. PEG is a best polymer of choice in drug delivery systems because of its tunable properties and well-established safety profile. Recently polymers of different sizes of PEGs are used on the basis of green chemistry principle for extraction of organic chemicals in food and pharmaceutical industry because of its inert, hydrophilic and hydrophobic properties (Guin and Gruebele 2019; Zinov’eva et al. 2020). In addition, it possesses the prime requisites for the selection of any ingredient in formulation development of drug delivery carrier. PEGylation technique is commonly used to improve the properties of biomolecules including, proteins, peptides, enzymes, antibody fragments, oligonucleotides, small synthetic drugs, etc. (D’souza and Shegokar 2016). Upon covalent attachment of PEG to any biomolecule, it increases the half-life, solubility, stability and reduced immunogenicity of that molecule. The hydrated PEG chain protects the conjugated biomolecules from proteases and thus reduces nonspecific degradation and improved solubility and stability. All these advantages of PEGylation are exploited in the pharmaceutical industry routinely.
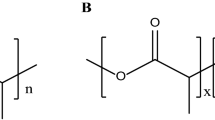
PEGs are amphiphilic polymers composed of repeating ethylene glycol subunits, and its number is represented by the whole integer n. Each ethylene glycol residue has a molecular mass of 44 Da, and n × 44 Da represents the number average molecular weight of the PEG chain. Chemically, PEGs are inert, nontoxic substance with hydroxyl groups at both termini that can be chemically activated for diverse function. In common practice, linear PEG chains, branched PEGs and polymer of more than one PEG monomers are joined either linearly or branched crosslink which are differ in their relative chemical reactivity and specificity (Suzuki et al. 1984).
PEG conjugation to biomolecules is currently a common practice to achieve persistent clinical responses and improved biological features due to thermal and mechanical stability, lower antigenicity and immunogenicity, reduced enzymatic degradation, improved solubility, optimal pharmacokinetics and pharmacodynamic properties, enhanced circulating half-life in body, decreased clearance and enhanced potency (Bailon and Berthold 1998; Zhang et al. 2014). The improved biological activity is attributed to the changes in electrostatic-binding properties, conformational changes, steric interference, hydrophobicity, local charge distribution and pI value of proteins. In addition, PEGylation significantly alters the binding affinities to the receptors attributed to the physicochemical changes, resulting in reduced activities in cell-based assays, in which incubation times are usually short (Inada et al. 1986). Degree of PEG-protein conjugate in the form of unmodified, mono-, di-, tri- pegylated have great advantages. Generally, with increase in the degree of pegylation, rate of absorption decreases which prolongs the availability of drug in circulation and receptor saturation.
Wide range of hydrogels are prepared from PEG which are being used in varieties of biomedical applications because of diverse nature of PEG and the versatility of mechanical and biological modification according to physiological requirements (Alexander et al. 2014; Mendez et al. 2018). The hydrogel formed by PEGs can release the drug over a long duration of time, meanwhile biocompatible and provide environment similar to soft tissue and allow diffusion of nutrients due to biodegradable properties with desired safety and efficacy. PEG prevents the molecules of the hydrogels from being dissolved in a swelling medium by holding the entire molecule together due to extensive cross linking. Some of the investigated PEG-based copolymers are currently used in biopharmaceutical industry and clinical research (Harris and Chess 2003). Many PEGylated drugs have been approved by FDA to address hepatitis, rheumatoid arthritis, neutropenia, various cancers and wound healing therapy (Katre 1993).
Because of enormous biological and clinical implications of PEG, this chapter aimed to provide a comprehensive discussion. We provide a brief discussion on protein structure prediction and different biological applications of PEG along with underlying mechanism of improved biochemical features.
11.2 Importance of Structure and Functional Dynamics in PEGylation
Genome sequencing efforts utilizing high-throughput technologies are yielding millions of protein encoding sequence that currently lack any functional characterization (Molloy et al. 2014). The function of a protein of interest can be inferred from other homologous protein with common ancestor which are functionally characterised as well. For this purpose, sequence and/or structure information can be used. Sequence comparisons methods were used for genome-wide sequence annotation that are based on sequence alignment to identify homologous proteins. BLAST, PROSITE and PFAM all are well-known sequence alignment tool for genome-wide functional annotation tool (Dhifli and Diallo 2016; Molloy et al. 2014). All these tools mentioned above are typically fast but restricted to identifying pair of proteins with significant sequence similarity (close homolog). Function of an uncharacterized query protein was determined when sequence alignment tool identify a homolog with known function and sequence similarity more than 30% with query. On the other hand, two proteins with identical function cannot be inferred based on sequence information alone. Sequence based functional annotation may miss detecting remote homolog which is either branching point or convergent evolution has resulted in high sequence diverge while preserving structure and function. First remote homolog identified was myoglobin and hemoglobin which have similar structure but different sequence. Evolutionary pressure is more on structure than sequence for preservation; methods that compare structure allow effectively casting wider lattice at detecting related proteins for functional annotation. Another word, three dimensional structure of a protein is highly conserved compared to the primary sequence. Thus, it is better to compare overall structure and shape of a protein and considered to be more eloquent way of assigning function of query protein. Structure-based function deduction promises to detect remote homolog and expand options for assigning function to novel protein sequences. So in order to implement PEGylation of proteins, one must go through these tools to predict the best results for the desired protein. These tools reduce the number of experiments and better strategy for PEGylation of proteins.
11.2.1 Protein Structure-Based Drug Design
Many therapeutic compounds currently available in the market were either discovered from the screening of natural or synthetic compound libraries or through serendipitously. These indiscriminate approaches entails testing large number of compound and developing countless high-throughput screening assay (Bonetta et al. 2016). Now days, a rational approach based on structure based route where the structure of target protein is determined and hypothetical ligand most of the case predicted by molecular modelling and movement of compounds were predicted by molecular dynamics simulation before the synthesis of screened molecules. These days protein structure is generally determined using three different methods (a) X-ray crystallography, (b) Nuclear Magnetic Resonance spectroscopy (NMR) and (c) cryoelectron microscopy (Cryo-EM). X-ray crystallography is oldest and commonly used technique, relies on the ability of protein to crystallise in a completely biologically unnatural condition in regular molecular array. NMR has advantage as it can perform in solution and no protein crystal required. Major problem associated with NMR is protein size, where it is unable to determine the structure of large proteins. Cryo-EM is just become available and soon be capable of providing structural information of proteins in solution and as good as X-ray crystallography. Present decade has witnessed rapid development of computer aided drug design with enough accuracy which allows frequent use in discovery of new therapeutics. Drug design based on protein structure prediction based on binding mode of small molecules and their relative affinity (Sledz and Caflisch 2018) is delivering better results.
11.2.2 Computer-Aided Drug Design
Computer aided drug design (CADD) methods have played fundamental role in drug discovery effort from many years. Nowadays, CADD become essential part of the discovery pipeline for pharmaceutical companies (Sledz and Caflisch 2018). Discovery of quite a lot of approved drugs including captopril, saquinavir, indinavir, ritonavir, and tirofiban, has benefited to a large extent from the application of CADD (Hillisch et al. 2015; Muegge et al. 2017). X-Ray structure generally provide proved quite static picture of the protein but when it is combined with computation techniques to obtain a clear idea of how protein function, the combination become awesome for drug desighning. CADD techniques are used principally for virtual screening, hit/lead optimization and design of novel compounds. In virtual screening a huge database of compounds is examined and subset of compound is picked out on the binding capacity for further in vitro testing. Further CADD is used for optimization of hit/lead compound driven by the rationalization of a structure–activity relationship. After the individuation of key elements for binding, the design of new compound can be attempted (Salmaso 2018).
11.2.3 Virtual Screening
Virtual screening is a computational method which is used to find potential ligand of interest by screening an in silico library of diverse chemical compounds. It acts as filter which allows to pass out unwanted ligands and retains which are of interest based on filter criteria like stereochemical similarity or stereochemical complementarity. Virtual screening identifies the potential ligands either based on similarity of ligand (ligand based approach) or binding affinity with receptor (receptor based approach). A comparison between ligand based and receptor methods of screening are shown in Table 11.1.
This chapter mainly focuses on receptor based or structure based virtual screening in which a library of small molecules (ligands) is docked to the binding site of protein in lock and key manner. In this approach three things are required; (1) Protein structure of protein either determined experimentally in crystal form and solution form or computationally modelled using homology modelling. One or more known protein structures with close sequence identity are used as template in homology modelling to model protein of interest. (2) A library of small molecules is required for the docking algorithm to calculate binding affinity of ligand with binding site on protein of interest. (3) Finally a docking protocol is required which defines the docking parameters used in docking experiment (Fig. 11.1).
11.2.4 Molecular Dynamic Simulation
Molecular Dynamic Simulation provides insight into protein dynamics ahead of that available crystallographically and unravels novel cryptic binding sites, expanding the druggability of the target. MD-simulations are applied in the investigation of numerous dynamic properties and process which is directly applied in structural biochemistry, enzymology, molecular biology, biophysics, biotechnology and pharmaceutical chemistry. It allows scientist to study the thermodynamic and kinetic properties of proteins and other biomolecules.
11.3 PEG in Drug Delivery
PEGs are amphiphilic in nature thus easily dissolved in organic and aqueous solvents. Because of its non-toxic nature and their property of being easily eliminated by renal and hepatic pathways, PEGs are ideal choice for drug delivery system (Rashmi Zabihi et al. 2020). These unusual properties of PEG along with their highly hydrated polyether backbone, capable as an acceptor hydrogen bonding, a large exclusion volume make it capable to entrap versatile drugs for the targeted delivery. Various efforts are being made to develop novel strategies to conjugate PEG with biomolecules to increase its clinical applications (Yang et al. 2020b). Currently, new strategies have been undertaken to develop PEG based drug delivery system which allow the delivery of different active classes of drugs (Han et al. 2019). Clinical implications of several PEG-conjugated drugs has been routinely reported. Entrapment of IL-2, growth hormone antagonist, haemoglobin, growth factor, anticancer drugs, cytokines, enzymes, hormones, lactoferrin, antibodies and antibody fragments etc., in the PEG-based polymers are frequently used in medical industry. In addition, many proteins are conjugated to the PEGylated polymers to extended circulating plasma life and easily clearance through renal filtration because of increased aqueous solubility, decreased immunogenicity, and permits binding of the proteins to their receptors (Kolate et al. 2014a). Furthermore, due to the increased overall volume and molecular mass of PEGylated polymer, it has a greater bioavailability, and thus results in less frequent dosing (Nucci et al. 1991). Generally, PEGylation is used for the modification of therapeutic molecules by conjugation with PEG. A natural clinical implication of PEGylated proteins is in the form of antibodies (both single chain and monoclonal) modifications to achieve greater solubility and longer circulating life in vivo. PEGylation with longer PEG chain was more efficient than multiple PEGylation with short PEG chain to increase serum half-life (Lee et al. 1999). A new and potentially useful application of PEG mAb to an ADEPT (antibody directed enzyme prodrug therapy) system which is generally used for targeted drug delivery for the impairment of genetic diseases (AlQahtani et al. 2019a). Recent advancement in the recombinant DNA technology has improved the production of proteins at large scale. But, their clinical applications are limited because of their antigenicity. Protein PEGylation significantly reduces the antigenicity of recombinant protein and thus associated antibody formation against them in limited, contributing to their prolonged activity with increased in vivo life to several hundred folds. During therapeutic uses, PEG protects protein from proteolytic digestion in the body (Veronese and Mero 2008).
Many small organic molecules and anticancer agents have been conjugated to PEG which can easily be delivered to the target without any side effect (Elechalawar et al. 2020). A successful PEGylation of drug molecules can improve the pharmacokinetic and pharmacodynamic outcomes. In addition, PEGylation extend blood residence, decrease enzymatic degradation and reduce immunogenicity of drugs as well as decrease uptake by reticuloendothelial system. Entrapment of anticancer drugs into the PEG helps to passively release at target tumor site with less frequent neutropenia, alopecia and cardiotoxicity (Plosker 2008). A PEG–daunorubicin conjugates have been recently synthesized and there in vitro antitumor activity has been evaluated (Greenwald 2001). PEGylation improves the physicochemical properties of drug molecules, including optimal degree of modification of molecular weight, size, hydrophilicity, conformation, steric hindrance and charge which lead to altered elimination kinetics and thus, several PEGylated therapeutics have entered into the clinical trials and their successful translation to the clinical use. However, an accurate assessment of pharmacokinetic and pharmacodynamic parameters of drug like molecules in animals and patients are highly needed. A sensitive in vivo quantification and fate of the bound drug in the host body is required after systemic administration stability, metabolism and bioavailability of PEGylated compounds (Kolate et al. 2014b). For target-specific therapy and imaging, nanocarrier based delivery systems have recently emerged as a promising delivery system for therapeutics with great potential (Rajan et al. 2014). It was shown that incorporation of small amounts of gangliosides, glucolipids, phosphatidylinositol impart a weak negative charge on surface of liposomes which can bypass the reticuloendothelial uptake. It was found from research charge influence that the uptake for hydrophobic and neutral to positively charged particles are more prone to reticuloendothelial uptake in comparison to the negatively charged particles.
11.4 Gene Therapy
Recently, gene therapy has drawn significant attention of scientific community for the treatment of diseases associated with the non-functional and mutated genes such as haemophilia, mucopolysaccharidosis, autoimmune diseases and cancer. The outcome of gene therapy is depending on the efficiency and safety of its delivery vectors. In comparison to the gene delivery using viral particle, PEGylation and hydrogel as gene delivery vectors are less oncogenic, immunogenic, easy to prepare and specially does not have any limitation of the size of gene to be transferred. In this context, PEG has gained remarkable attention because of its desired stability in the systemic circulation (Hashemi et al. 2019). Despite a great advantage of PEG for controlling the pharmacokinetics of the carriers, but is undesirable for the cellular association of gene carriers with tumors. To address this problem, various modified gene delivery systems have been developed. Hatakeyama et al. (2007) developed an interesting gene delivery system for cancer gene therapy, using a multifunctional envelope-type nano device modified with enzymatically cleavable PEG-lipid. They prepared a cleavable PEG-lipid which is composed of a PEG/matrix metalloproteinase (MMP)-substrate to be specifically cleaved by MMP in the extracellular space in tumor tissues (Hatakeyama et al. 2007). As we know that MMP expression is higher in the case of tumor cells, thus such system facilitate the site directed gene delivery. Kim et al. (2006) conjugated the siRNA of vascular endothelial growth factor to PEG via a disulfide linkage. A conjugate of siRNA-PEG interacting with cationic polyethylenimine form polyelectrolyte complex micelles and consequently showed a greater stability against enzymatic degradation. Under condition similar to reductive cytosolic environment, siRNA in its intact form was released from the siRNA–PEG conjugate after cleavage of the disulfide linkage which could be potentially applied to RNAi-based anti-angiogenic treatment of cancer in vivo. Hashemi et al., fabricated PEG coated, calcium doped magnetic nanograin (PEG/Ca(II)/Fe3O4) as a genome expression enhancer as a promising strategy for gene therapy (Hashemi et al. 2019). The potential of large scale production and biocompatibility make PEG-based carriers increasingly attractive for gene therapy. However, many intra- and extracellular obstacles needs to get addressed.
PEG-modified adenosine deaminase has been extensively used for the enzyme replacement therapy for immunodeficiency due to adenosine deaminase deficiency. Enzyme replacement therapy by modified PEG has restored the metabolic environment and thus immune function lost due to the adenosine deaminase deficiency (Hershfield 1995). In many cases, level of functions achieved through enzyme replacement therapy has been sufficient to protect against opportunistic and life-threatening infections and diseases (Liu et al. 2009). For gene therapy against HIV in peripheral blood mononuclear cells, a cationic carbosilane–PEG dendrimers were used (Sánchez-Nieves et al. 2014). An improved performance was observed as compared to a PEG-free carbosilane dendrimer. Toxicity significantly decreased in presence of PEG moety because dendrimers and siRNA interact weakly as compared to the homodendrimer analogous. Chemical features like well-defined size and structure, flexibility, monodispersity and multivalent molecular surface, lower toxicity and lower dendriplex strength, are key properties for use of these vectors for the gene delivery to target cells (Newkome et al. 2008).
11.5 Protein Folding Studies
PEG has been extensively used in research to mimic the cellular environment to investigate the role of crowding agent in reducing misfolding and aggregation, conformational change in protein to increase stability and activity. The extent of PEG-based stabilization of proteins depends on the size of PEG, PEGylation site, structure of the PEG–protein linker, etc. The strength of a noncovalent interaction, salt-bridge and hydrogen-bond strength within a protein depends strongly on its microenvironment and increasing in nonpolar versus aqueous environments. As we know that protein folds in a crowded cellular environment but most of the studied performed on protein folding were done on a single protein (Kinjo and Takada 2002; Tsao et al. 2010; van den Berg et al. 1999). The efficiency of macromolecular crowding agent is expected to be highest hydrodynamic dimensions of a tested protein. To mimic the natural cellular milieu, many crowding agent including PEG have been extensively used as crowding agents for various proteins (Ghosh et al. 2020; Nasreen et al. 2018; Shahid et al. 2019; Shahid et al. 2017). An expected outcome of the presence of PEG as a macromolecular crowding agent is their ability to improve the conformational stability of a globular protein due to the excluded volume effects including alteration of solvent properties (Christiansen et al. 2013; Stepanenko et al. 2016; Tokuriki et al. 2004). Behaviour of protein dynamics is affected by the hydrodynamic size and concentration of inert crowder where chemical nature of crowder molecules should not play any role in the modulation of conformational properties (Fonin et al. 2019). Conformational changes of d-glucose/d-galactose-binding protein (GGBP) were studied at different PEGs (Molecular weight, 12,000, 4000, and 600) in varying concentration and a noticeable structural changes was observed (Fonin et al. 2018). All PEGs promoted compaction of GGBP and lead to the increase in ordering of its structure. These data support the previous notion that the influence of macromolecular crowders on proteins is rather complex phenomenon that extends beyond the excluded volume effects (Fonin et al. 2017).
Stepanenko et al. (2016) has demonstrated the effects of different PEGs of varying molecular masses (PEG-600, PEG-8000, and PEG-12000) on the spectral properties and unfolding-refolding processes of the super-folder green fluorescent protein. The hydrodynamic radii of PEG-600, PEG-8000, and PEG-12000 are 5.6 Å ± 0.3 Å, 24.5 Å ± 1.9 Å, and 30.9 Å ± 2.5 Å, respectively (Kuznetsova et al. 2014). According to the excluded volume theory, the greatest effect on the unfolding-refolding processes should be provided by PEG-8000, whose hydrodynamic dimensions are closest to those of sfGFP. Ferreira et al., demonstrated that PEG and UCON of similar size produces different changes in the solvent properties of water in their solutions and induced morphologically different α-synuclein aggregates (Ferreira et al. 2015; Ferreira et al. 2016). The further extended study to get deeper insights into the behaviour of proteins in a crowded environment, a similar study was conducted to determine the secondary and tertiary structure and aromatic residue solvent accessibility ten different proteins. Both PEG and UCON polymers affects secondary and tertiary structures of folded and hybrid proteins in a limited fashion with a slight unfolding. Solvent accessibility of aromatic residues was significantly increased for the majority of the proteins in the presence of UCON but not PEG. PEG also accelerated the aggregation of protein into amyloid fibrils (Breydo et al. 2015). In addition, a large number of studies on PEG has been carried out to establish the role of PEG in protein folding and dynamics (Parray et al. 2019; Shahid et al. 2017; Xiao et al. 2019).
11.6 Cosmetic Industry
PEG is one of the best investigated polymer for use in bio-related application due to its inertness, biocompatibility, biodegradability and versatility. PEG is generally used in cosmetics, food processing, pharmaceuticals, agriculture, and industrial manufacturing (AlQahtani et al. 2019b). Because of two terminal alcohol groups, the PEGs can form a series of derivatives including, mono-, di- and polyesters, ethers, amines and acetals. PEGs can favour the addition reaction to form new compounds or complexes on their ether bridges. Some common derivatives of PEG in cosmetic industry are, fatty acid esters, PEG ethers, PEG amine ethers, PEG castor oils, PEG propylene glycols, and other derivates with required properties. PEG is found in many domestic and hygiene products, because of their solubility, viscosity and low toxicity. PEGs and their fatty acid esters produce a little irritation and have extremely low acute and chronic toxicities and thus considered as a highly acceptable polymer in the cosmetic industry (Fruijtier-Pölloth 2005). Due to the abundant presence of PEG, it likely to be present at or introduced to the inflammation site. PEG fatty esters, PEGs, and PEG sorbitan fatty esters are slightly irritating to the skin of rabbits and humans and thus commonly found in the antiseptic creams. The occurrence of PEG in close vicinity to highly active immune cells may be enough to elicit the stimulation of anti-PEG antibodies (Yang and Lai 2015). PEGylated pharmaceutical products are used as an indirect molecular probe for measuring mucosal permeability. Their unique osmotic characteristics in aqueous solutions, PEG provides solubility, stability and strength to the degrading substances (Fordtran and Hofmann 2017). In addition, carcinogenicity studies with PEGs have been conducted. Interestingly, in most of studies people found no indication of a tumorigenic effect of PEGs which will further allow the effective use of PEG in cosmetics. The PEGs and their fatty acid ethers and esters produce a negligible dermal irritation along with an extremely low acute and chronic toxicities as they do not readily penetrate intact skin and thus the sensitising potential of these compounds appears to be negligible (Jang et al. 2015). Furthermore, commonly used PEG derivatives in the cosmetic industry are free from reproductive and developmental toxicity, on genotoxicity and carcinogenic properties.
11.7 Food Industry
PEGs are widely used in medical and food industries because of their biologically inert, non-immunogenic, non-toxic and water-soluble nature. A mild to life-threatening immediate hypersensitivity for PEG are reported (Ahmed et al. 2018). PEG hypersensitivity should be considered during the diagnostic management and therapy. The Food and Drug Administration (FDA) has approved several biological application of PEGs including as a carrier, preservative of food, cosmetics and pharmaceuticals, including injectable and bowel solutions (Lim and Hong 2014). PEGs are extensively used as enhancer of solubility and oral bioavailability of compounds with poor aqueous solubility (Gullapalli and Mazzitelli 2015). The nanoparticles and essential oil loaded composite films of PEG are widely used in the food industry for packaging purposes. These films are capable of exhibiting antimicrobial effects against bacteria, and extend the shelf-life of meat. Such biocompatible films showed are manufactured commercially with increased thermal stability (Ahmed et al. 2018). The developed bionanocomposites are highly compatible to food packaging to control the spoilage and the pathogenic bacteria associated with the fresh chicken meat (Ahmed et al. 2018). Liu et at. developed EGylated chitosan modified silver nanoparticles which possess excellent antimicrobial activity against both Gram-negative and Gram-positive bacteria (Liu et al. 2017). Such antimicrobial coatings with excellent nonfouling property is available to resist bacterial attachment to the food materials for long term storage and transport. However, silver based nanoparticle coating with PEG has dramatically enhanced antibacterial property and ascribed to the synergism of PEG-based resistance of bacteria due the antibacterial property of silver. Colloidal silver nanoparticles prepared by chemical reduction using PEG as a reducing agent has been used as food simulants in apple juice.
11.8 Bone and Tissue Engineering
In recent years different types of biomaterials are developed to support the bone tissue regeneration process. A relatively new class of nanocomposite biomaterials are produced from PEG that incorporate a biopolymeric and biodegradable matrix structure render improved properties including large surface area, high mechanical strength and stability, enhanced cell adhesion, proliferation, and cell differentiation (Bharadwaz and Jayasuriya 2020). Cartilage production with the help of PEG and alginate was successfully done. Collagen, PEG encapsulated with poly-lactic or -glycolic groups and polyvinyl alcohol, are other examples of biological and synthetic polymers that have been combined with chondrocytes (Bryant and Anseth 2003). Recent development in bone tissue engineering have identified and propelled the need of PEG based biomaterials as a promising approach for effective bone regeneration because of its high tunable properties, excellent biocompatibility, controlled biodegradability and good mechanical strength (Wang et al. 2019). In bone regeneration, the biomaterial scaffold is required to be fabricated having adequate cell growth and bone tissue regeneration. Such properties may be gained by PEGs because of their high porosity, surface-to-volume ratio, and crystallinity. PEG based nanofiber mats exhibited excellent cell attachment and growth, along with adequate cytocompatibility (Scaffaro et al. 2017). PEG-based membrane incorporated with the nanocalcium phosphate was developed to reduce non-functional scar tissue defects with the help of guided bone regeneration. The addition of calcium phosphate particles increase in fiber diameter as the addition of the inorganic calcium phosphate may have reduced the conductivity of the electrospinning solution. Furthermore, osteogenic differentiation of the cells was aided by the addition of the calcium phosphate nanoparticles as they increased the concentration of calcium ions thereby facilitating better storage of the cells (Türkkan et al. 2017). For surgeries and regenerative engineering tissue adhesives are manufactured with the help of PEG to achieve a rapid crosslinking, strong wet adhesion and cohesion strengths, and minimal cytotoxicity remains a critical roadblock for clinical translation (Lu et al. 2020). Recently, Zhou et al. (2020) designed a (PEG)-based poly(urethane-urea) for bone tissue repair using cystine dimethyl ester as a cross-linker. The strength of material was further strengthened by physical blending of nano-hydroxyapatite. The obtained biocompatible material showed excellent mechanical strength, biocompatibility and osteogenic capability and thus has good prospects for bone tissue repair application.
11.9 Bioimaging and Radiotherapy
PEG is an FDA-approved compound, has been extensively used to modify various anticancer agents to increase long blood circulation, and enhancing their tumor accumulation. In recent years application of PEG-based nanoparticles have received greater attention in biomedicine, particularly in diagnostic imaging. Recently, Yang et al. designed a circular aptamer-PEG structure which is capable of prolonged blood circulation, and then responds to the acidic tumor microenvironment to deeply penetrate the solid tumor and selectively recognize cancer cells for in vivo targeted bioimaging (Yang et al. 2020a). A typical AS1411 aptamer containing G-rich oligonucleotide sequences with specific recognition of nucleolin was designed to detect a non-ribosomal protein normally existing in the nucleus and also highly expressed on the surface of cancer cells. Such materials having deep tumor tissue penetration and specific targeting of cancer cells, may be employed in efficient drug delivery strategy as well as bioimaging purposes. Photothermal therapy is an efficient approach employed for cancer treatment. A novel PEG based nanomaterials were synthesized which has significant T1-weighted performance to target tumor and localize for photothermal therapy. This approach holds significant potential for the clinical application especially in the case of tumor localization and targeted therapy (Meng et al. 2020).
Pretargeting strategies have recently emerged as an attractive imaging and therapy option for cancer patients. Different types of nanostars are prepared with has high potential to accumulate in the tumor tissue via an enhanced permeability and retention and thus implemented in the development of a passive delivery of cytotoxic compounds to cancer cells. To increase the contrast between uptake in the tumor and in surrounding healthy tissues, PEG based nanostars are designed for pretargeted positron emission tomography imaging, using radioligands that are labeled with the short-living positron emitter fluorine-18 (Goos et al. 2020a). The development of a pretargeting strategy based on the passive delivery of PEG based nanostars further expand the cancer imaging and treatment options, with a minimal radiation doses to healthy cells (Goos et al. 2020b). Recently, a new type of gold-PEG based nanoparticles are designed which possess excellent X-ray absorption coefficient, utilized as a contrast agent for computed tomography scan imaging. This nanoparticle shown a reliable aqueous dispensation, low cytotoxicity, and strong X-ray absorption ability subsequently enhances contrast, having long circulation time in the blood, and a negligible in vivo toxicity. Hence, such type of nanoparticles have great potential for clinical application in CT scan imaging (Gao et al. 2020).
Encapsulation of near infrared dyes, indocyanine green in the PEG based biocompatible nano carrier has been extensively used for NIR bioimaging, photothermal and photodynamic therapy. In is interesting to note that the encapsulated dyes remained stable for over long time and slowly accumulating in the liver and spleen having a wide range with deep penetration into the living tissues, may be exploited as a promising candidate for cancer treatment and diagnosis (Yeroslavsky et al. 2020). PEG incorporated silica nanoparticles are developed for improving efficacy of targeted radiotherapy in melanoma models. PEGylated carbon dots were engineered to bind alpha melanocyte stimulating hormone cyclic peptide analogs for targeting the melanocortin-1 receptor over-expressed on melanoma tumor cells. Such quantum dots are radiochemically stable, biologically active, and exhibited high affinity cellular binding properties and internalization (Zhang et al. 2020). An ultrasmall PEGylated quantum dots, covalently encapsulating the near infra-red emitting dye Cy5 were engineered to display MC1-R targeting cyclic DOTA-αMSH peptides on their surfaces. This superior property provide selective tumor uptake and favourable biodistribution properties and improved renal clearance. The unique and tunable surface features of this targeted radiotherapeutic quantum dots are coupled with favorable pharmacokinetic properties, substantially improved treatment efficacy and demonstrated a clear survival benefit in melanoma models.
11.10 Application of PEGs in Cutting Edge Technologies
PEGs are the most successful precipitants for the protein crystallization especially in the case of high molecular weight proteins (Gorrec 2016). Various parameters related to PEG such as types, concentration and molecular mass influence the crystallizing process. PEG has been used as a component in organ preservation solutions to reduce injury from cold perfusion in animal organs (Pasut et al. 2016; Valuckaite et al. 2013). PEGs 4000 and 3350 are commonly used in the gastroenterology from a long time. A series of PEGylated polymers are developed for tissue engineering. Implementing PEG-alt-thiol conjugates, biodegradable polymeric system showed in vivo biocompatible cell matrices for tissue engineering and wound healing (Wang et al. 2008). Further applications of PEG include in plastics and resins, in printing, as ingredients of soaps and detergents, in the rubber, in the textile and leather industry, in the paper industry, in the ceramics and glass industry, petroleum, mining and metal industries, for wood preservation and as chemical intermediates. Remarkably, PEG as a kind of stable, environmentally friendly and green surfactant. Many PEG-based aqueous solution are applied in extraction, separation and preconcentration of various constituents from complex mixture because it possesses the advantages of low cost, non-volatility, biodegradation, and non-toxicity (Zhang et al. 2017). Hydrogels composed of PEG and chitosan have been extensively used in the fields of wound dressing, tissue repair, water purification, drug delivery, and bone regeneration, and coatings in dental applications. Hydrogel developed for dental application shows a superior activity in early-stage adhesion inhibition and displays remarkably long-lasting colony-suppression activity. In addition, such nanomaterial antifouling and antimicrobial functions with excellent biocompatibility (Peng et al. 2020).
11.11 Miscellaneous Applications
PEGs are widely used in medical and food industries because of their biologically inert, non-immunogenic, non-toxic and water-soluble nature. A mild to life-threatening immediate hypersensitivity for PEG are reported. PEG hypersensitivity should be considered during the diagnostic management and therapy. The Food and Drug Administration (FDA) has approved several biological application of PEGs including as a carrier, preservative of food, cosmetics and pharmaceuticals, including injectable and bowel solutions (Lim and Hong 2014). PEG has been used as a component in organ preservation solutions to reduce injury from cold perfusion in animal organs (Pasut et al. 2016; Valuckaite et al. 2013). PEGs 4000 and 3350 are commonly used in the gastroenterology from a long time. PEGylated pharmaceutical products are used as an indirect molecular probe for measuring mucosal permeability. Their unique osmotic characteristics in aqueous solutions, PEG provides solubility, stability and strength to the degrading substances (Fordtran and Hofmann 2017). A series of PEGylated polymers are developed for tissue engineering. Implementing PEG-alt-thiol conjugates, biodegradable polymeric system showed in vivo biocompatible cell matrices for tissue engineering and wound healing (Wang et al. 2008). Cartilage production with the help of PEG and alginate was successfully done. Collagen, PEG encapsulated with poly-lactic or -glycolic groups and polyvinyl alcohol, are other examples of biological and synthetic polymers that have been combined with chondrocytes (Bryant and Anseth 2003). PEGs are extensively used as enhancer of solubility and oral bioavailability of compounds with poor aqueous solubility (Gullapalli and Mazzitelli 2015). PEGylation is one of the best approaches for targeting of anticancer therapeutics.
PEG is one of the best investigated polymer for use in bio-related application due to its inertness, biocompatibility, biodegradability and versatility. PEG is generally used in cosmetics, food processing, pharmaceuticals, agriculture, and industrial manufacturing (AlQahtani et al. 2019b). PEG is found in many domestic and hygiene products, thus our body got repeated exposure to PEG which may causes the development of anti-PEG antibodies. Due to the abundant presence of PEG, it likely to be present at or introduced to the inflammation site. The occurrence of PEG in close vicinity to highly active immune cells may be enough to elicit the stimulation of anti-PEG antibodies (Yang and Lai 2015). Recent development in bone tissue engineering have identified and propelled the need of PEG based biomaterials as a promising approach for effective bone regeneration because of its high tunable properties, excellent biocompatibility, controlled biodegradability and good mechanical strength (Wang et al. 2019). PEGs are the most successful precipitants for the protein crystallization especially in the case of high molecular weight proteins (Gorrec 2016). Various parameters related to PEG such as types, concentration and molecular mass influence the crystallizing process.
11.12 Conclusion
PEGs are most commonly used polymers which is inert in nature, non-immunogenic, non-antigenic which enhance the solubility of hydrophobic drugs and facilitate the potential for DNA transfection, siRNA delivery and tumor targeting. PEGylated drug delivery systems are very effective nano-carriers used to deliver anticancer drugs to the tumor site. The PEGylated polymers had lower cytotoxicity and hemolytic toxicity with increased solubility, less aggregation, lower opsonization by RES and higher tumor accumulation by the enhanced permeability and retention effect. PEGylation showed a great advantage in overcoming the unfavourable characteristics of biomaterials by keeping the unique properties. PEG has also been used as gene delivery vector for the targeted delivery of gene of interest. PEG-based copolymers play a crucial role as a biomedical material for biomedical applications, because of its biocompatibility, biodegradability, thermosensitivity and easy controlled characters. PEG–based hydrogel exhibits good gelling mechanical strength and minimizes the initial burst effect of the drug. The hydrogel developed from PEG is able to release the drug over a long duration of time, meanwhile is also biocompatible and provide environment similar to soft tissue and allow diffusion of nutrients due to biodegradable properties with desired safety and efficacy. Despite the extensive use of PEGs, future biomedical implications are the matter of concern among the scientists but still to procure the FDA approvals, a strong investigation in the clinical studies is necessary.
References
Ahmed, J., Arfat, Y. A., Bher, A., Mulla, M., Jacob, H., & Auras, R. (2018). Active chicken meat packaging based on polylactide films and bimetallic Ag-Cu nanoparticles and essential oil. Journal of Food Science, 83, 1299–1310.
Alexander, A., Ajazuddin Khan, J., Saraf, S., & Saraf, S. (2014). Polyethylene glycol (PEG)–poly(N-isopropylacrylamide) (PNIPAAm) based thermosensitive injectable hydrogels for biomedical applications. European Journal of Pharmaceutics and Biopharmaceutics, 88, 575–585.
AlQahtani, A. D., Al-Mansoori, L., Bashraheel, S. S., Rashidi, F. B., Al-Yafei, A., Elsinga, P., et al. (2019a). Production of “biobetter” glucarpidase variants to improve drug detoxification and antibody directed enzyme prodrug therapy for cancer treatment. European Journal of Pharmaceutical Sciences, 127, 79–91.
AlQahtani, A. D., O’Connor, D., Domling, A., & Goda, S. K. (2019b). Strategies for the production of long-acting therapeutics and efficient drug delivery for cancer treatment. Biomedicine & Pharmacotherapy, 113, 108750.
Bailon, P., & Berthold, W. (1998). Polyethylene glycol-conjugated pharmaceutical proteins. Pharmaceutical Science & Technology Today, 1, 352–356.
Bharadwaz, A., & Jayasuriya, A. C. (2020). Recent trends in the application of widely used natural and synthetic polymer nanocomposites in bone tissue regeneration. Materials Science & Engineering. C, Materials for Biological Applications, 110, 110698.
Bonetta, R., Ebejer, J. P., Seychell, B., Vella, M., Hunter, T., & Hunter, G. J. (2016). Role of protein structure in drug discovery. Journal of the Malta Chamber of Scientists, 4, 126–130.
Boutet, E., Lieberherr, D., Tognolli, M., Schneider, M., Bansal, P., Bridge, A. J., et al. (2016). UniProtKB/Swiss-Prot, the manually annotated section of the UniProt KnowledgeBase: How to use the entry view. Methods in Molecular Biology, 1374, 23–54.
Breydo, L., Sales, A. E., Frege, T., Howell, M. C., Zaslavsky, B. Y., & Uversky, V. N. (2015). Effects of polymer hydrophobicity on protein structure and aggregation kinetics in crowded milieu. Biochemistry, 54, 2957–2966.
Bryant, S. J., & Anseth, K. S. (2003). Controlling the spatial distribution of ECM components in degradable PEG hydrogels for tissue engineering cartilage. Journal of Biomedical Materials Research. Part A, 64, 70–79.
Christiansen, A., Wang, Q., Cheung, M. S., & Wittung-Stafshede, P. (2013). Effects of macromolecular crowding agents on protein folding in vitro and in silico. Biophysical Reviews, 5, 137–145.
D’souza, A. A., & Shegokar, R. (2016). Polyethylene glycol (PEG): A versatile polymer for pharmaceutical applications. Expert Opinion on Drug Delivery, 13, 1257–1275.
Dawson, N. L., Lewis, T. E., Das, S., Lees, J. G., Lee, D., Ashford, P., et al. (2017). CATH: An expanded resource to predict protein function through structure and sequence. Nucleic Acids Research, 45, D289–D295.
Dhifli, W., & Diallo, A. B. (2016). ProtNN: Fast and accurate protein 3D-structure classification in structural and topological space. BioDataMining, 9, 30.
Elechalawar, C. K., Hossen, M. N., Shankarappa, P., Peer, C. J., Figg, W. D., Robertson, J. D., et al. (2020). Targeting pancreatic cancer cells and stellate cells using designer Nanotherapeutics in vitro. International Journal of Nanomedicine, 15, 991–1003.
Ferreira, L. A., Cole, J. T., Reichardt, C., Holland, N. B., Uversky, V. N., & Zaslavsky, B. Y. (2015). Solvent properties of water in aqueous solutions of elastin-like polypeptide. International Journal of Molecular Sciences, 16, 13528–13547.
Ferreira, L. A., Madeira, P. P., Breydo, L., Reichardt, C., Uversky, V. N., & Zaslavsky, B. Y. (2016). Role of solvent properties of aqueous media in macromolecular crowding effects. Journal of Biomolecular Structure & Dynamics, 34, 92–103.
Fonin, A. V., Darling, A. L., Kuznetsova, I. M., Turoverov, K. K., & Uversky, V. N. (2018). Intrinsically disordered proteins in crowded milieu: When chaos prevails within the cellular gumbo. Cellular and Molecular Life Sciences, 75, 3907–3929.
Fonin, A. V., Silonov, S. A., Sitdikova, A. K., Kuznetsova, I. M., Uversky, V. N., & Turoverov, K. K. (2017). Structure and conformational properties of d-glucose/d-galactose-binding protein in crowded milieu. Molecules, 22, 244.
Fonin, A. V., Stepanenko, O. V., Sitdikova, A. K., Antifeeva, I. A., Kostyleva, E. I., Polyanichko, A. M., et al. (2019). Folding of poly-amino acids and intrinsically disordered proteins in overcrowded milieu induced by pH change. International Journal of Biological Macromolecules, 125, 244–255.
Fordtran, J. S., & Hofmann, A. F. (2017). Seventy years of polyethylene glycols in gastroenterology: The journey of PEG 4000 and 3350 from nonabsorbable marker to colonoscopy preparation to osmotic laxative. Gastroenterology, 152, 675–680.
Fruijtier-Pölloth, C. (2005). Safety assessment on polyethylene glycols (PEGs) and their derivatives as used in cosmetic products. Toxicology, 214, 1–38.
Gao, Y., Kang, J., Lei, Z., Li, Y., Mei, X., & Wang, G. (2020). Use of the highly biocompatible Au nanocages@PEG nanoparticles as a new contrast agent for in vivo computed tomography scan imaging. Nanoscale Research Letters, 15, 53.
Ghosh, S., Shahid, S., Raina, N., Ahmad, F., Hassan, M. I., & Islam, A. (2020). Molecular and macromolecular crowding-induced stabilization of proteins: Effect of dextran and its building block alone and their mixtures on stability and structure of lysozyme. International Journal of Biological Macromolecules, 150, 1238–1248.
Goos, J. A., Cho, A., Carter, L. M., Dilling, T. R., Davydova, M., Mandleywala, K., et al. (2020a). Delivery of polymeric nanostars for molecular imaging and endoradiotherapy through the enhanced permeability and retention (EPR) effect. Theranostics, 10, 567.
Goos, J. A. C. M., Davydova, M., Dilling, T. R., Cho, A., Cornejo, M. A., Gupta, A., et al. (2020b). Design and preclinical evaluation of nanostars for the passive pretargeting of tumor tissue. Nuclear Medicine and Biology, 84-85, 63–72.
Gorrec, F. (2016). Protein crystallization screens developed at the MRC Laboratory of molecular biology. Drug Discovery Today, 21, 819–825.
Greenwald, R. (2001). PEG drugs: an overview. Journal of Controlled Release, 74, 159–171.
Guin, D., & Gruebele, M. (2019). Weak chemical interactions that drive protein evolution: crowding, sticking, and quinary structure in folding and function. Chemical Reviews, 119, 10691–10717.
Gullapalli, R. P., & Mazzitelli, C. L. (2015). Polyethylene glycols in oral and parenteral formulations—A critical review. International Journal of Pharmaceutics, 496, 219–239.
Han, S., Sun, R., Su, H., Lv, J., Xu, H., Zhang, D., et al. (2019). Delivery of docetaxel using pH-sensitive liposomes based on D-alpha-tocopheryl poly(2-ethyl-2-oxazoline) succinate: Comparison with PEGylated liposomes. Asian Journal of Pharmaceutical Sciences, 14, 391–404.
Harris, J. M., & Chess, R. B. (2003). Effect of pegylation on pharmaceuticals. Nature Reviews Drug Discovery, 2, 214–221.
Hashemi, E., Mahdavi, H., Khezri, J., Razi, F., Shamsara, M., & Farmany, A. (2019). Enhanced gene delivery in bacterial and mammalian cells using PEGylated calcium doped magnetic Nanograin. International Journal of Nanomedicine, 14, 9879–9891.
Hatakeyama, H., Akita, H., Kogure, K., Oishi, M., Nagasaki, Y., Kihira, Y., et al. (2007). Development of a novel systemic gene delivery system for cancer therapy with a tumor-specific cleavable PEG-lipid. Gene Therapy, 14, 68–77.
Hershfield, M. S. (1995). PEG-ADA replacement therapy for adenosine deaminase deficiency: An update after 8.5 years. Clinical Immunology and Immunopathology, 76, S228–S232.
Hillisch, A., Heinrich, N., & Wild, H. (2015). Computational chemistry in the pharmaceutical industry: From childhood to adolescence. ChemMedChem, 10, 1958–1962.
Inada, Y., Takahashi, K., Yoshimoto, T., Ajima, A., Matsushima, A., & Saito, Y. (1986). Application of polyethylene glycol-modified enzymes in biotechnological processes: Organic solvent-soluble enzymes. Trends in Biotechnology, 4, 190–194.
Jang, H.-J., Shin, C. Y., & Kim, K.-B. (2015). Safety evaluation of polyethylene glycol (PEG) compounds for cosmetic use. Toxicological Research, 31, 105–136.
Jones, D. T., & Cozzetto, D. (2015). DISOPRED3: Precise disordered region predictions with annotated protein-binding activity. Bioinformatics, 31, 857–863.
Katre, N. V. (1993). The conjugation of proteins with polyethylene glycol and other polymers: Altering properties of proteins to enhance their therapeutic potential. Advanced Drug Delivery Reviews, 10, 91–114.
Kim, S. H., Jeong, J. H., Lee, S. H., Kim, S. W., & Park, T. G. (2006). PEG conjugated VEGF siRNA for anti-angiogenic gene therapy. Journal of Controlled Release, 116, 123–129.
Kinjo, A. R., & Takada, S. (2002). Effects of macromolecular crowding on protein folding and aggregation studied by density functional theory: Statics. Physical Review. E, Statistical, Nonlinear, and Soft Matter Physics, 66, 031911.
Kolate, A., Baradia, D., Patil, S., Vhora, I., Kore, G., & Misra, A. (2014a). PEG - a versatile conjugating ligand for drugs and drug delivery systems. Journal of Controlled Release, 192, 67–81.
Kolate, A., Baradia, D., Patil, S., Vhora, I., Kore, G., & Misra, A. (2014b). PEG — A versatile conjugating ligand for drugs and drug delivery systems. Journal of Controlled Release, 192, 67–81.
Kuznetsova, I. M., Turoverov, K. K., & Uversky, V. N. (2014). What macromolecular crowding can do to a protein. International Journal of Molecular Sciences, 15, 23090–23140.
Lee, L. S., Conover, C., Shi, C., Whitlow, M., & Filpula, D. (1999). Prolonged circulating lives of single-chain Fv proteins conjugated with polyethylene glycol: A comparison of conjugation chemistries and compounds. Bioconjugate Chemistry, 10, 973–981.
Lim, Y. J., & Hong, S. J. (2014). What is the best strategy for successful bowel preparation under special conditions? World journal of gastroenterology: WJG, 20, 2741.
Liu, G., Li, K., Luo, Q., Wang, H., & Zhang, Z. (2017). PEGylated chitosan protected silver nanoparticles as water-borne coating for leather with antibacterial property. Journal of Colloid and Interface Science, 490, 642–651.
Liu, P., Santisteban, I., Burroughs, L. M., Ochs, H. D., Torgerson, T. R., Hershfield, M. S., et al. (2009). Immunologic reconstitution during PEG-ADA therapy in an unusual mosaic ADA deficient patient. Clinical Immunology, 130, 162–174.
Lu, X., Shi, S., Li, H., Gerhard, E., Lu, Z., Tan, X., et al. (2020). Magnesium oxide-crosslinked low-swelling citrate-based mussel-inspired tissue adhesives. Biomaterials, 232, 119719.
Mendez, U., Zhou, H., & Shikanov, A. (2018). Synthetic PEG hydrogel for engineering the environment of ovarian follicles. Methods in Molecular Biology, 1758, 115–128.
Meng, X., Zhang, B., Yi, Y., Cheng, H., Wang, B., Liu, Y., et al. (2020). Accurate and real-time temperature monitoring during MR imaging guided PTT. Nano Letters, 20, 2522–2529.
Mitchell, A. L., Scheremetjew, M., Denise, H., Potter, S., Tarkowska, A., Qureshi, M., et al. (2018). EBI metagenomics in 2017: Enriching the analysis of microbial communities, from sequence reads to assemblies. Nucleic Acids Research, 46, D726–D735.
Molloy, K., Van, M. J., Barbara, D., & Shehu, A. (2014). Exploring representations of protein structure for automated remote homology detection and mapping of protein structure space. BMC Bioinformatics, 15(Suppl 8), S4.
Muegge, I., Bergner, A., & Kriegl, J. M. (2017). Computer-aided drug design at Boehringer Ingelheim. Journal of Computer-Aided Molecular Design, 31, 275–285.
Nasreen, K., Ahamad, S., Ahmad, F., Hassan, M. I., & Islam, A. (2018). Macromolecular crowding induces molten globule state in the native myoglobin at physiological pH. International Journal of Biological Macromolecules, 106, 130–139.
Newkome, G. R., Moorefield, C. N., & Vögtle, F. (2008). Dendritic molecules: Concepts, syntheses, perspectives. Weinheim: Wiley.
Nucci, M. L., Shorr, R., & Abuchowski, A. (1991). The therapeutic value of poly (ethylene glycol)-modified proteins. Advanced Drug Delivery Reviews, 6, 133–151.
Parray, Z. A., Ahamad, S., Ahmad, F., Hassan, M. I., & Islam, A. (2019). First evidence of formation of pre-molten globule state in myoglobin: A macromolecular crowding approach towards protein folding in vivo. International Journal of Biological Macromolecules, 126, 1288–1294.
Pasut, G., Panisello, A., Folch-Puy, E., Lopez, A., Castro-Benitez, C., Calvo, M., et al. (2016). Polyethylene glycols: An effective strategy for limiting liver ischemia reperfusion injury. World Journal of Gastroenterology, 22, 6501–6508.
Peng, L., Chang, L., Si, M., Lin, J., Wei, Y., Wang, S., et al. (2020). Hydrogel-coated dental device with adhesion-inhibiting and colony-suppressing properties. ACS Applied Materials & Interfaces, 12, 9718–9725.
Plosker, G. L. (2008). Pegylated liposomal doxorubicin: A review of its use in the treatment of relapsed or refractory multiple myeloma. Drugs, 68, 2535–2551.
Rajan, S. S., Turovskiy, Y., Singh, Y., Chikindas, M. L., & Sinko, P. J. (2014). Poly (ethylene glycol)(PEG)-lactic acid nanocarrier-based degradable hydrogels for restoring the vaginal microenvironment. Journal of Controlled Release, 194, 301–309.
Rashmi Zabihi, F., Singh, A. K., Achazi, K., Schade, B., Hedtrich, S., Haag, R., et al. (2020). Non-ionic PEG-oligoglycerol dendron conjugated nano-carriers for dermal drug delivery. International Journal of Pharmaceutics, 580, 119212.
Salmaso, V. (2018). Exploring protein flexibility during docking to investigate ligand-target recognition. Padova: University of Padova.
Sánchez-Nieves, J., Fransen, P., Pulido, D., Lorente, R., Muñoz-Fernández, M. Á., Albericio, F., et al. (2014). Amphiphilic cationic carbosilane–PEG dendrimers: Synthesis and applications in gene therapy. European Journal of Medicinal Chemistry, 76, 43–52.
Scaffaro, R., Lopresti, F., Maio, A., Botta, L., Rigogliuso, S., & Ghersi, G. (2017). Electrospun PCL/GO-g-PEG structures: Processing-morphology-properties relationships. Composites Part A: Applied Science and Manufacturing, 92, 97–107.
Shahid, S., Ahmad, F., Hassan, M. I., & Islam, A. (2019). Mixture of macromolecular crowding agents has a non-additive effect on the stability of proteins. Applied Biochemistry and Biotechnology, 188, 927–941.
Shahid, S., Hassan, M. I., Islam, A., & Ahmad, F. (2017). Size-dependent studies of macromolecular crowding on the thermodynamic stability, structure and functional activity of proteins: In vitro and in silico approaches. Biochimica et Biophysica Acta - General Subjects, 1861, 178–197.
Sledz, P., & Caflisch, A. (2018). Protein structure-based drug design: From docking to molecular dynamics. Current Opinion in Structural Biology, 48, 93–102.
Stepanenko, O. V., Stepanenko, O. V., Kuznetsova, I. M., Uversky, V. N., & Turoverov, K. K. (2016). Peculiarities of the super-folder GFP folding in a crowded milieu. International Journal of Molecular Sciences, 17, 1805.
Suzuki, T., Kanbara, N., Tomono, T., Hayashi, N., & Shinohara, I. (1984). Physicochemical and biological properties of poly(ethylene glycol)-coupled immunoglobuling G. Biochimica et Biophysica Acta (BBA) - Protein Structure and Molecular Enzymology, 788, 248–255.
Tokuriki, N., Kinjo, M., Negi, S., Hoshino, M., Goto, Y., Urabe, I., et al. (2004). Protein folding by the effects of macromolecular crowding. Protein Science, 13, 125–133.
Tsao, D., Minton, A. P., & Dokholyan, N. V. (2010). A didactic model of macromolecular crowding effects on protein folding. PLoS One, 5, e11936.
Türkkan, S., Pazarçeviren, A. E., Keskin, D., Machin, N. E., Duygulu, Ö., & Tezcaner, A. (2017). Nanosized CaP-silk fibroin-PCL-PEG-PCL/PCL based bilayer membranes for guided bone regeneration. Materials Science and Engineering: C, 80, 484–493.
UniProt, C. (2015). UniProt: A hub for protein information. Nucleic Acids Research, 43, D204–D212.
Valuckaite, V., Seal, J., Zaborina, O., Tretiakova, M., Testa, G., & Alverdy, J. C. (2013). High molecular weight polyethylene glycol (PEG 15-20) maintains mucosal microbial barrier function during intestinal graft preservation. Journal of Surgical Research, 183, 869–875.
van den Berg, B., Ellis, R. J., & Dobson, C. M. (1999). Effects of macromolecular crowding on protein folding and aggregation. The EMBO Journal, 18, 6927–6933.
Veronese, F. M., & Mero, A. (2008). The impact of PEGylation on biological therapies. BioDrugs, 22, 315–329.
Wang, J.-Z., You, M.-L., Ding, Z.-Q., & Ye, W.-B. (2019). A review of emerging bone tissue engineering via PEG conjugated biodegradable amphiphilic copolymers. Materials Science and Engineering: C, 97, 1021–1035.
Wang, N., Dong, A., Radosz, M., & Shen, Y. (2008). Thermoresponsive degradable poly(ethylene glycol) analogues. Journal of Biomedical Materials Research. Part A, 84, 148–157.
Xiao, Q., Draper, S. R. E., Smith, M. S., Brown, N., Pugmire, N. A. B., Ashton, D. S., et al. (2019). Influence of PEGylation on the strength of protein surface salt bridges. ACS Chemical Biology, 14, 1652–1659.
Yang, Q., & Lai, S. K. (2015). Anti-PEG immunity: Emergence, characteristics, and unaddressed questions. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology, 7, 655–677.
Yang, Y., Zhu, W., Cheng, L., Cai, R., Yi, X., He, J., et al. (2020a). Tumor microenvironment (TME)-activatable circular aptamer-PEG as an effective hierarchical-targeting molecular medicine for photodynamic therapy. Biomaterials, 246, 119971.
Yang, Z., Guo, Q., Cai, Y., Zhu, X., Zhu, C., Li, Y., et al. (2020b). Poly(ethylene glycol)-sheddable reduction-sensitive polyurethane micelles for triggered intracellular drug delivery for osteosarcoma treatment. Journal of Orthopaedic Translation, 21, 57–65.
Yeroslavsky, G., Umezawa, M., Okubo, K., Nigoghossian, K., Thi Kim Dung, D., Miyata, K., et al. (2020). Stabilization of indocyanine green dye in polymeric micelles for NIR-II fluorescence imaging and cancer treatment. Biomaterials Science, 8(8), 2245–2254.
Zhang, F., Liu, M. R., & Wan, H. T. (2014). Discussion about several potential drawbacks of PEGylated therapeutic proteins. Biological & Pharmaceutical Bulletin, 37, 335–339.
Zhang, L., Cheng, Z., Zhao, Q., & Wang, M. (2017). Green and efficient PEG-based ultrasound-assisted extraction of polysaccharides from superfine ground lotus plumule to investigate their antioxidant activities. Industrial Crops and Products, 109, 320–326.
Zhang, X., Chen, F., Turker, M. Z., Ma, K., Zanzonico, P., Gallazzi, F., et al. (2020). Targeted melanoma radiotherapy using ultrasmall 177Lu-labeled α-melanocyte stimulating hormone-functionalized core-shell silica nanoparticles. Biomaterials, 241, 119858.
Zhou, Z., Wang, Y., Qian, Y., Pan, X., Zhu, J., Zhang, Z., et al. (2020). Cystine dimethyl ester cross-linked PEG-poly(urethane-urea)/nano-hydroxyapatite composited biomimetic scaffold for bone defect repair. Journal of Biomaterials Science. Polymer Edition, 31, 407–422.
Zinov’eva, I. V., Zakhodyaeva, Y. A., & Voshkin, A. A. (2020). Data on the extraction of benzoic, salicylic and sulfosalicylic acids from dilute solutions using PEG-based aqueous two-phase systems. Data in Brief, 28, 105033.
Acknowledgements
This work was supported by the grant from the Indian Council of Medical Research (ICMR)BIC/12(16)/2014. The authors are very indebted to Sharda University, FIST Program (SR/FST/LSI-541/2012) and Jamia Millia Islamia (a Central University) for facilitating the research.
Conflict of Interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Raina, N., Singh, A.K., Islam, A. (2021). Biological Implications of Polyethylene Glycol and PEGylation: Therapeutic Approaches Based on Biophysical Studies and Protein Structure-Based Drug Design Tools. In: Singh, S.K. (eds) Innovations and Implementations of Computer Aided Drug Discovery Strategies in Rational Drug Design. Springer, Singapore. https://doi.org/10.1007/978-981-15-8936-2_11
Download citation
DOI: https://doi.org/10.1007/978-981-15-8936-2_11
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-8935-5
Online ISBN: 978-981-15-8936-2
eBook Packages: Computer ScienceComputer Science (R0)