Abstract
Biomolecules are the key factors for most of the biological processes. Immobilization is an impressive technique which is used to stabilize free biomolecules in solution. Immobilized biomolecules are more robust and more resistant to environmental changes. The main objective of the immobilization of biomolecules, namely., enzymes, is to enhance the economics of biocatalysis processes. Additionally, immobilization improves many properties of enzymes such as performance in organic solvents, pH tolerance, heat stability, and functional stability. More importantly, the heterogeneity of the immobilized biomolecule systems allows an easy recovery of both biomolecules and products, multiple reuse, continuous operation of enzymatic processes, rapid termination of reactions, and greater variety of bioreactor designs. This chapter presents an overview of the recent literature reports for biomolecule immobilization on nanomaterial surfaces by various techniques. It also demonstrates the need for immobilization and different applications at various platform.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
5.1 Introduction
Biomacromolecules, namely lipids, proteins, enzymes, and nucleic acids, are the most important systemic unit of cells. These show unique properties which majorly contribute as the center stage for different biochemical and physiological process of cells (Jia et al. 2018). With significant enhancement in R & D activities, biomacromolecules have been needed for various biomedical as well as industrial field. In the late 1920, the first enzyme immobilization (invertase) was reported by Nelson and Griffin on artificial aluminum hydroxide as a base via physical adsorption, which captured the interest of researchers to develop innovative strategies for immobilization (Zhang et al. 2015). Therefore, during 1920–1960, a lot of new and advantageous immobilization techniques were developed, for example, physical adsorption, covalent attachment, entrapment and encapsulation, and cross-linking attachment, etc., for the improvement of enzyme stability and productivity. Since 1960, these methods and processes were modified and applied at industrial and laboratory scale to improve the stability and activity of different biomolecules. But, few major challenges for using these molecules in artificial surroundings still remain, namely very limited use, problem in their upscaling, minimal recovery process, thermal and physiological instability, poor in vitro reaction ability, and poor binding efficiency toward substrate (Cao et al. 2003). Hence, there is an urgent need for alternative technologies to combat all these limitations. Product purification and repetitive usage of enzymes in long-term reaction followed by their recovery is also a big challenge for industrial applications. To overcome these obstacles, immobilization technique is the only preferred technology to economize the processed biomolecules on nanomaterial surface to improve stability, reusability, selectivity, and cost effectiveness (Ansari and Husain 2012; Brady and Jordaan 2009). Nanomaterials (NMs) act as a fruitful base or support for immobilization process, due to their various advantages such as large surface area to volume ratio, stabilizing biocatalysts efficiency, strong chemical as well as physical properties, etc. (Walia et al. 2019). Diffusion is a major problem in which there is high chances of substrate diffusion from reaction media. Additionally, many other obstacles are present during immobilization process, for example, false orientation and loss of catalytic activity of biomolecules (i.e., enzymes, proteins, ssDNA, etc.). Nowadays, nanoparticles are being used as a model system to combat the diffusion problems during immobilization process (Ansari and Husain 2012). Hence, the advantageous properties of NMs combine with innovative techniques would be an ideal approach. Here, in this chapter, we have described the advantages of different immobilization processes and their impact on related biological activities.
5.2 Methods of Immobilization and Their Advantages
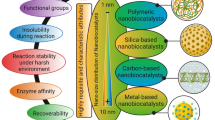
The proper selection of immobilization method is the only key to overcome the limitation of biomolecules’ unstability. In a specific reaction, it plays censorious role in evaluation of biomolecules activity and their characteristic features. The overall system requires a better activity, performance, reusability, minimal cost of the process, and minimal toxicity. There are a lot of methods available for immobilization process, for example, covalent attachment, physical adsorption and entrapment, affinity tag approach, polymer cross-linking, etc. (Pundir 2015). Features of different immobilization techniques are shown in Fig. 5.1.
5.2.1 Covalent Attachment
Covalent attachment is a process in which biomolecules are attached to a specific insoluble or soluble base with the help of chemically active functional moiety. This technique is highly useful due to its robust advantage over biomolecule leakage and instability (Pundir 2015). Major advantages of this techniques are strong binding capability of substrate to biomolecule, high heat stability, flexibility of design, and wide range of modifications that can be done via click chemistry. Click chemistry does not only rule the era of covalent immobilization but also gives the strong perspective for rapid and stable immobilization of biomolecules on NMs surface (Nemzer et al. 2010). Various literature reports suggest that stimulation of NMs surface via active chemical moiety is the most important factor which gives direction for enzymes and protein immobilization (Secundo 2013). Similar to non-covalent immobilization, it can be used for unmodified biomolecules too, which only rely on natural functional moiety. For example, if proteins are exposed to any ester active support, they can readily react and form unstable conjugation in aqueous environment and undergo ester hydrolysis due to nucleophilicity of thiol (from cysteine) in aqueous media. To overcome this disadvantage, an aldehyde group can be coupled with amine group of proteins in the presence of ß-mercaptoethanol or sodium borohydride for the formation of stable secondary amide linkage (Homaei et al. 2013). Based on thiol reactivity toward biomolecules and metallic nanoparticles nowadays, chemisorption is widely used for covalent immobilization. For example, single strand DNA (ssDNA) was covalently attached on gold nanoparticles (AuNPs) via chemisorption process (Li et al. 2007; Yu et al. 2013). Template-mediated covalent attachment is also widely used for oligomers immobilization, that is, two different templates containing phenol and benzene are covalently attached with oligomers via copper-catalyzed azide–alkyne cycloaddition (Núñez-Villanueva et al. 2019). Biomolecules like enzymes and proteins are well known for their therapeutic activities such as cancer treatment, treatment of genetic disorder, as food and nutritional supplements, etc. Hence, it is most important to maintain the stability and native activity of biomolecules. PEGylation of L-asparaginase via covalent attachment reduces its immunogenicity and short half-life along with high maximum saturation velocity (Vmax) and Michaelis constant (Km) (Meneguetti et al. 2019). Venditti et al. immobilized lipase on hydrophilic AuNPs via covalent attachment with high residual activity and loading capability of ~95% and 65–70%, respectively (Venditti et al. 2015).
5.2.2 Physical Adsorption and Entrapment
Unlike covalent attachment, physical adsorption is a process in which biomolecules are adsorbed on NMs surface via electrostatic interaction, van der Waals forces, hydrogen bonding, etc. between immobilizing biomolecules and NMs (Li et al. 2007). It can minimize the deactivation of biomolecules which is often caused by covalent chemical modification during immobilization process. Nitrocellulose membranes or hydrophobic polymer–coated substrates are majorly involved in this technique (Sharma et al. 2014). The advantages of using this technique is that neither does it involve any reducing agents nor does it require any modified biomolecules. However, the major disadvantage of this technique is involvement of weak and reversible interactions as compared to covalent immobilization. Hence, due to this poor attachment ability, biomolecules could be leached out from substrate, which results in contamination and loss of activity of immobilized assembly (Wang et al. 2015a, b). Ionic hydrogels are widely used in this technique. Based on hydrophobic and hydrogen bond interactions, these immobilizes biomolecules in the pattern of polymer. This noncovalent immobilization of biomolecules is affected by pH, temperature, concentration, and physicochemical properties of reaction mixture, etc. (Cacicedo et al. 2019).
5.2.3 Cross-linking Attachment
A carrier-free attachment in which biomolecules are immobilized via bifunctional cross-linking agents is known as cross-linking immobilization, that is, cross-linking enzymes (CLE) (Cipolatti et al. 2014). This is a simple process for immobilization without using any support materials as compared to covalent and noncovalent immobilization techniques. So, these are mostly used in the form of sol–gel–sol, hydrogel, gelatinous matrix, etc. As per the literature reports, there are various strategies which produce stable, highly efficient, and low-cost immobilization of enzymes and proteins via cross-linking approach (Cacicedo et al. 2019; Cipolatti et al. 2014). The commonly used cross-linker is glutaraldehyde in which two end aldehyde groups freely react with amino group of biomolecules (Hartmann and Kostrov 2013). This approach results in gelatinous immobilization of biomolecules which cannot be specifically controlled due to improper aggregation of immobilized enzymes. There are various reports suggesting that cross-linked enzyme aggregations (CLEAs) physically aggregate enzymes in the aqueous solvent via water miscible solvents, salts, and non-ionic polymer addition (Rodrigues et al. 2013). Chitosan-coated magnetic nanomaterials have been recently used for cross-linking immobilization of enzymes with enhanced biological activity (Díaz-Hernández et al. 2018; Khoshnevisan et al. 2019).
5.2.4 Affinity Tagging
Affinity tagging is an innovative approach mainly used in protein–protein or protein–molecule interactions in a specific orientation (Hood et al. 2014). Biotin- and streptavidin-mediated conjugation of proteins with metallic NMs is widely explored in sensing applications. Inorganic NPs are widely chosen for bioaffinity interaction with various biomolecules namely proteins, peptides, ssDNA, and lectins along with few of the enzymes (Compostella et al. 2017). Bioaffinity interactions of protein–NPs have gained a lot of attention recently. This type of interaction influenced by physicochemical properties of NPs such as hydrophobicity, surface charge, shape, and size, etc. (Dar et al. 2019). Nowadays, proteins or peptide–NPs interaction are widely used in nanomedicine applications due to versatile properties of biological peptide-mediated stealth effect and biocompatibility. Hence, these nano assemblies work as a tool for targeted delivery of nanoparticles without immune system detection. Most of the NMs, that is mesoporous silica NPs, liposomes, polymeric NMs, AuNPs, magnetic NPs, etc., are used in protein–NPs–based nanomedicine development (Spicer et al. 2018). The different physicochemical properties which affect the immobilization and catalytic activity of the biomolecules have been discussed in the following section.
5.3 Physicochemical Properties of Biomolecules
Physicochemical properties are the most important parameters to optimize the effect of the surface on biomolecule conjugation. Here, we briefly discuss about the importance of thermodynamic process as aid to understand the biomolecules–NMs interaction. Thermodynamics of reaction determines how ligand and receptors act during any interaction. The standard thermodynamic equation which characterize the steps of biomolecules–NMs conjugates is as follows:
where, ΔrG0rel is the standard Gibbs free energy change of released biomolecules from bioconjugates, ΔrG0bind is binding of free biomolecules to the substrate, and ΔrG0conj is the conjugation of substrate biomolecules to NMs (Rodriguez-Quijada et al. 2018).
5.3.1 Kinetic Parameters
Kinetic parameter describes the behavior of biomolecule binding with substrate. There are two well-known standard equations (Michaelis–Menten and Briggs–Haldane) which describe the dependency of biomolecule-catalyzed reaction rate on the substrate concentration via two parameters, namely, the catalytic constant (kcat) and Michaelis–Menten constant (Km). The maximum rate of reaction at saturated substrate concentration is determined via kcat estimation, whereas, Km is the substrate concentration at which the rate of reaction is half of the maximum velocity (Vmax) of reaction. These two parameters can be easily characterized by Lineweaver–Burk plot via linear transformation (Choi et al. 2017). Rodrigues et al. explained the factors which affect the kinetic parameters of bioconjugates, namely, rigidification, effect of medium, effect of substrate or product partition, diffusional limitations, and mixture of biomolecules and their environment. An improper immobilization methodology produces strong rigidity in biomolecule structure due to multipoint covalent attachments, which facilitate structural deformities in conjugated biomolecules (Rodrigues et al. 2013). Generally, biomolecules are always immobilized in the presence of the substrate so that active sites are not hampered. In the presence of inactivating agents such as hydrogen peroxide, free radicals, dissolved gases, organic solvents, etc., immobilized biomolecules significantly affect the soluble environment. This facilitates disturbance in the hydrophilic–lipophilic balance of working mixture of bioconjugates. So to minimize or stabilize the medium effect, biomolecules were further modified using suitable polymers (Brady and Jordaan 2009).
5.3.2 pH and Temperature Dependence
The milieu of biomolecule immobilization plays an important role in maintaining the integrity of biomolecules on solid substrate. pH changes in reaction mixture affect the active confirmation of biomolecules immobilized on substrate. The microenvironment of the immobilized biomolecules and reaction mixture usually has unequal partitioning of H+ and OH− concentrations due to electrostatic interactions with the substrate, which often leads to the change in the pH activity profile. The pH effect on the activity of free and immobilized laccase was examined in the pH range 2.0–7.0 at 30 °C. The optimum pH for free laccase activity was found at pH 4.0 which was similar to that reported previously. On the other hand, the optimum pH for immobilized laccase activity was found to shift to pH 5.0 and the thermal stability also shifted to 60 °C (Mazlan and Hanifah 2017). Immobilization of laccase on nanofiber matrix broaden the pH range, which suggests that immobilization process protects enzyme activity in a wider pH range (Fatarella et al. 2014). Garg et al. examined the pH dependent immobilization of urease on glutathione-modified AuNPs. Immobilized urease retained higher enzymatic activity at pH 7.4 as compared to free one (Garg et al. 2015). Biomolecules are highly sensitive toward temperature. Higher temperature causes irreversible loss of catalytic activity. Talebi et al. showed that thermal stability of α-amylase was improved after their immobilization on nanocomposite (Talebi et al. 2016).
5.3.3 Redox Potential
Biomolecules such as oxidoreductase and metalloproteins have redox potential in their reaction milieu. One-third of the cellular process is involved with these types of biomolecules for electron transfer, that is, photosynthesis, respiration, metabolism, and molecular signaling. The functional properties of the oxidoreductases/metalloproteins are determined by the nature of their redox centers. Biomolecule engineering is a powerful approach that is used to incorporate biological and abiological redox cofactors with predictable structures and desirable functions for important biological and chemical applications (Prabhulkar et al. 2012). The immobilization of enzymes on electrodes is of significant interest in the development of enzymatic fuel cells (EFC) and biosensors. Siepenkoetter et al. reported immobilized glucose dehydrogenase on nanoporous AuNPs which showed strong oxidative properties compared to free enzyme (Siepenkoetter et al. 2017). Carbon nanomaterials show better electron transfer process, so nowadays these are directly engineered and used as nanozyme, which resembles the redox enzyme activity (Le Goff and Holzinger 2018). Zhou et al. developed reagentless glucose sensing via glucose oxidase immobilization on carbon nanotubes (CNTs) (Zhou et al. 2017).
5.4 Biomolecules Immobilized on Nanomaterials
5.4.1 Enzyme and Proteins
Enzymes are well-known biological catalysts and are involved in vital physiological functions of different living cells of animals, plants, and microorganisms. Nowadays, enzymes are well explored in many fields of applications, such as industrial food processing, biosensing, therapeutics, etc. Wang et al. reported a new way of alkaline phosphatase (ALP) immobilization on magnetic nanoparticles (MNP). They synthesized amine-functionalized silica-coated magnetic nanoparticles (Fe3O4@SiO2@APES) and to this ALP was covalently attached with the help of sodium cyanoborohydride, which acts as reducing agent and form stable secondary amine linkage between enzyme and MNPs (Zhang et al. 2015). Likewise, lipase was covalently attached on MNP via carbodiimide-mediated direct binding between MNPs and immobilized enzyme and used for transesterification of fatty acid to biodiesel (Wang et al. 2013a, b). Li et al. reported immobilization process of glucose oxidase (GOx) on gold nanoparticle (AuNPs) via ethyl (dimethylaminopropyl) carbodiimide/N-hydroxysuccinimide (EDC/NHS) coupling reaction (Yu et al. 2013). Recently, cellulase was immobilized on MNP via cross-linking approach with high loading efficiency and increased reusability by ~50% (Ingle et al. 2017). Similarly, Lin et al. immobilized cellulase on chitosan-modified MNPs via carboxylmethyl cellulose as substrate for cross-linking and enhanced the reusability by ~84% (Lin et al. 2017). In the recent decades, immobilization process has increased the stability and activity of various therapeutic enzymes such as acetylcholinesterase, α-amylase, alcohol dehydrogenase, cytochrome P450, trypsin, peptides, proteins, etc., and these are mentioned in Table 5.1. Ganesana et al. reported the acetylcholinesterase immobilization on nickel NPs via site-specific 6-histidine affinity tagging (Ganesana et al. 2011). Similarly, Uygun et al. performed α-amylase immobilization on MNPs with enhanced activity and reusability by ~85% and ~10 times, respectively (Uygun et al. 2012). One of the important therapeutic enzyme L-asparaginase (L-ASNase) immobilization was reported by Monajati et al. They functionalized graphene oxide via aspartic acid and immobilized L-ASNase covalently with increased activity, recovery and reusability by 90%, 42%, and after 8 cycles, respectively (Monajati et al. 2018). Similarly, Baharman and Alemzadeh reported the immobilization of L-ASNase on calcium alginate beads via microencapsulation process and enhanced ~35% residual activity with ~46% loading efficiency (Bahraman and Alemzadeh 2017). Alcohol toxicity is a major concern nowadays, therefore, Ghannadi et al. immobilized alcohol dehydrogenase (ADH) on titania nanoparticles via physical adsorption with increased residual activity and reusability by ~84% and 10 cycles, respectively (Ghannadi et al. 2019). Urea detection is vital part of pathological analysis for early detection of kidney-related malfunction. Therefore, Eghbali et al. reported urease immobilization on zinc oxide nanoparticles via physical adsorption for urea sensing at a pH range of 6–8 (Eghbali et al. 2015). Proteolytic proteins pepsin and trypsin were immobilized on silica NP gels and MNPs, respectively, with increased thermal stability (Szałapata et al. 2016; Zhang et al. 2018).
5.4.2 Nucleic Acids
Nucleic acid is a key molecule of whole living entity, and these molecules have very specific activities. Nowadays single-stranded DNA (ssDNA) are utilized as a versatile tool for biosensing application. The simplicity and advantage of these molecules allow us to design these for targeted sequence and transduce to form DNA hybrid for detection (Wang et al. 2015a, b). Compatible and better technique of DNA or RNA immobilization promotes efficient reactivity and orientation of immobilized molecules (Xu et al. 2015). Benvidi et al. developed the process for single-strand DNA (ssDNA) immobilization. They drop cast-thiolated ssDNA probe on gold electrode and incubate it for overnight reaction at room temperature followed by immerging in 6-mercapto-1-hexanol (MCH) to minimize the unspecific attachment (Benvidi et al. 2015; Thiruppathiraja et al. 2011). Zheng et al. reported polyaniline/graphene composite for electrostatic absorption DNA probe (Zheng et al. 2015). Similarly, Wang et al. explained the covalent immobilization of ssDNA via thiol and amine linkage with 3′ or 5′ end of sequence (Wang et al. 2011). Yang et al. immobilized aminated DNA probe on graphene oxide-fabricated single-walled carbon nanotube (SWCNTs) via EDC–NHS coupling reaction. Thus, the covalent immobilization is widely applicable approach in DNA probe immobilization on stable and compatible substrates (Yang et al. 2015). Similarly, Wang et al. reported DNA probe immobilization on glassy carbon electrode via electro-polymerization of Eriochrome Black T (sulphonic dye) (Wang et al. 2014).
5.4.3 Peptide and Lectins
Peptides are small sequence of amino acids, which are specifically designed for the biomedical applications such as antimicrobial activity, fluorometric or visual sensing of biological entity, tissue engineering, targeted delivery of NPs, etc. Similarly, antibodies are the immune-mediated biomolecules which are secreted from B-cells and play vital role in defense process (Spicer et al. 2018). Poon et al. performed two different peptides (cyclic RGD and myxoma virus derived) conjugation on AuNPs via secondary amine linkage between thiol and amine in the presence of sodium borohydride (Poon et al. 2015). Similarly, Guerrero et al. reported AuNPs and amphipathic peptide (CLPFFD) conjugation via simple covalent-bonding ring thiol conjugation (Guerrero et al. 2010). Qie et al. immobilized full-length CD47 peptide on three different sized carboxylated polystyrene NPs via carbodiimide-mediated click chemistry (Qie et al. 2016). Similarly, Schöttler et al. used polystyrene NPs for plasma protein conjugation and compared these nanoparticles toward the PEGylated counterpart (Schöttler et al. 2016). He et al. conjugated antimicrobial peptide (AMP) HHC36 on poly-SBMA–modified silica NPs via surface-initiated atom transfer radical polymerization (SI-ATRP) (He et al. 2018). Similarly, Sur et al. reported indolicidin AMP conjugation on carbon nanotube (CNT) and AuNPs via EDC–NHS conjugation (Sur et al. 2015). Chaudhary et al. conjugate three different AMP (TP359, 226, and 557) on silver-coated CNT via EDC–NHS coupling reaction (Chaudhari et al. 2019). Lectins are versatile carbohydrate-binding proteins having various applications namely cellular surface recognition, cellular permeability, cellular integrity, food production, etc. Sakuma et al. immobilized mucosal lectin (acetyl-D-galactosamine) on fluorescent polystyrene nanosphere via affinity tagging (Sakuma et al. 2009; Kitamura et al. 2015). Wang et al. studied the conjugation of glycans on fluorescent label silica NPs via photocoupling reaction (Wang et al. 2013a, b).
5.5 Biological Applications of Immobilized Biomolecules
5.5.1 Biosensing
Biosensing is an important application in current scenario for targeted analyte detection via converting biological signal into measurable units. Biosensing mainly includes electrochemical, thermal, colorimetry, and optical sensing. It involves few idealistic features, that is, specificity, reliability, cost effectiveness, and operational stability (Asal et al. 2019). Biosensing mechanism depends upon different biomolecules which are mainly used in this process, namely, enzymes, proteins, antibodies, DNA probes, lectins, peptides, etc. Immobilization and stabilization of these molecules on bioreceptor is the most important part of biosensing. Nowadays, nanoparticles are widely used in biosensing application via biomolecule immobilization because of their versatile nature and structural stability (Lee and Du-Duong 2018). In late 1960, first glucose biosensing was reported by Clark and Lyons based on glucose oxidase immobilization (Yoo and Lee 2010). Kim et al. reported portable urea sensing in biological sample by the help of urease immobilization on silk fibroin (Kim et al. 2018a, b). Visual ethanol biosensor was reported by Kuswandi et al. by alcohol oxidase immobilization on polyaniline membrane (Kuswandi et al. 2014). Thandavan et al. immobilized xanthine oxidase on MNPs via covalent interaction for xanthine sensing (Thandavan et al. 2013). Nesakumar et al. immobilized acetylcholinesterase on zinc oxide nanoparticles for carbosulphan biosensing (Nesakumar et al. 2016). Hassan et al. reported carrageenan biosensing via immobilization of calf thymus double-stranded DNA (dsDNA) on carbon-based screening electrode (Hassan et al. 2019). Surface plasmon resonance biosensing of targeted oligonucleotide via thiolated ssDNA probe immobilized on AuNPs was reported by Drozd et al. (Drozd et al. 2018). Ebrahimi et al. performed DNA-based biosensing for Ag+ and Hg2+ ions in amalgam filling via covalent immobilization on modified multiwalled carbon nanotube (MWCNT) (Ebrahimi et al. 2018). He et al. reported electrochemical biosensor for silver deposition via specific prostate peptide and DNA probe conjugation on modified silica NPs (He et al. 2015).
5.5.2 Bioimaging
Bioimaging is a fluorescence-based imaging for tracking and deposition of synthesized NPs at targeting site. Fluorescent proteins and antibodies are widely used in this technique, namely, green fluorescent protein (GFP), red fluorescent protein (RFP), fluorescent tag secondary antibodies, etc. There are various developments in bioimaging techniques that have taken place in recent decades, but the precise tool and probe development for living cell imaging are still big challenging tasks. Skogs et al. clearly demonstrated the role of antibody in bioimaging process of endogenous proteins via GFP immobilization (Skogs et al. 2016). Miawaki et al. optimized long chain peptide–protein linker with enhanced FRET mediated bioimaging for targeted site (Miyawaki and Niino 2015). Fu et al. reported glycoprotein-mediated fluorescent nanoparticles synthesis which reduced the false and autofluorescence effect of live cells imaging (Fig. 5.2) (Fu et al. 2018).
(a)Fluorescent imaging of Hep-G2 cells with Gal-NSp/HAS (10 μM/100 μM) upon alternate UV/vis irradiation (green/red channel excitation: 460–490/520–550 nm; emission: 500–550/580–650 nm; scale bar = 100 μm). Hoechst 33342 were used to stain the cell nuclei. Error bars represent S. D. (n = 3). Intracellular fluorescence intensity switching between (b) Gal-NSp/HAS and Gal-NMr/HAS upon alternate UV/vis irradiation. (c) UV/vis cycling of the two imaging channels of Gal-NSp/HAS and Gal-NMr/HAS visualized by confocal laser-scanning microscopy (green/red channel excitation: 440/579 nm; emission: 535/603 nm; scale bar = 25 μm). (Adapted from Fu et al. 2018, reprint with permission)
5.5.3 Therapeutics
Therapeutics is a field of treatment, diagnostics, and prevention of disease site. Pharmacologically active biomolecules are widely used under therapeutics. Kouassi et al. immobilized cholesterol oxidase on MNPs for total cholesterol analysis present in serum for point-of-care solution (Kouassi et al. 2005). Similarly immobilization of lysozyme on chitosan nanofiber via cross-linkage for antimicrobial activity is another point-of-care application of immobilized biomolecule (Park et al. 2013). Lactose intolerance is one of the major problems in children where the indigestion of lactose causes severe stomach disturbance. Hence, Ansari et al. reported innovative approach for B-galactosidase immobilization on zinc oxide NPs via covalent binding for natural degradation of lactose (Ansari et al. 2011). Nanoparticles and protein interaction are very complex biophysical process due to which most of the NPs cannot properly act inside the body. Peptides are widely used biomolecules for specific targeted delivery of NPs and also these can bypass the protein corona formation due to their specificity toward the target. Cesbron et al. used HA2 biological peptide and immobilized on AuNPs via thiol conjugation, which improved the endosomal escape property of AuNPs in cellular environment (Cesbron et al. 2015). Brain is the most complex organ in human body, and it has lots of limitations toward the treatment related issues. Major obstacle of brain treatment is penetration of drug across blood–brain barrier (BBB). Endo-Takahashi et al. reported entrapment of angiopep-2 in liposomal nanocarrier which facilitates blood–brain barrier penetration of peptide conjugates (Endo-Takahashi et al. 2016). Similarly, Kale et al. reported dual mode application of liposome nanoparticle via immobilization of GFP plasmid and TATp peptide (Kale and Torchilin 2007).
5.5.4 Food Industry
Food production are the leading and foremost needed demand of market. Food products are packed and preserved via various biomolecules-mediated reaction, that is, potato fries are now treated with L-ASNase to minimize the content of acrylamide (Raveendran et al. 2018). Labrou et al. reported immobilization of L-ASNase on polyethylene glycol (PEG) for efficacy improvement of enzyme in food and clinical application (Labrou and Muharram 2016). Pasha et al. reported the utilization of pectinase in clarification of fruit juice (Pasha et al. 2013). Pectinase was immobilized on celite surface via adsorption to increase the stability and activity of the immobilized enzyme (Chauhan et al. 2015). Similarly, catalase was immobilized on magnetic-MWCNTs and has been used in food industry especially for hydrogen peroxide removal (Sirbu 2011; Rafiee et al. 2019). Peroxidase is widely used in food industry for nutritional and flavor quality development. Immobilization of peroxidase on porous silica increases the catalytic activity of enzyme, and it was used as catalytic reactor in industries (Regalado et al. 2004; Sahare et al. 2014). α-acetolactate dehydrogenase was immobilized on nanoflower/alginate microbeads and widely used in the beverage industry for shortening the time period of beer, liquor, and soft drink maturation process (Choi et al. 2015; Zhao et al. 2017). Lactose intolerance problems occur in many children and adults, and β-galactosidase deals with this issue. Hence, lactase (β-galactosidase) was immobilized on sodium alginate and l-carrageenan (1:5 ratio) beads and utilized as important enzyme in most of the milk food products (Souza et al. 2018). Similarly, phospholipase enzyme was immobilized on nanomagnetic carrier and widely used in breakdown of phospholipid to fatty acid, which is utilized in the production of vanaspati oil, ghee, etc. (Jiang et al. 2019a, b; Borrelli and Trono 2015).
5.5.5 Tissue Engineering
Tissue engineering is the advanced tool of biotechnology, which is the future of development of artificial organs, limbs, flesh, etc. In the recent decade, various nanoparticles are widely used for tissue engineering processes such as carbon nanomaterials, biodegradable polymeric nanomaterial, therapeutic enzyme immobilized nanocarrier, etc. Kim et al. reported that tunable hydrogel degradation facilitates the development of scaffolds for tissue regeneration. In this research, chitosan is cross-reacted with methacrylate to form hydrogels and the tunable degradation was occurred after lysozyme incorporation to hydrogels (Kim et al. 2018a, b). Sortase A is bacterial ligase protein which is recently explored for tissue engineering purpose. It was attached to biodegradable polymer via cross-linking in the presence of collagen which facilitates artificial tissue regeneration (Broguiere et al. 2018). Similarly, histone deacetylase (HDAC) is used in stem cell regeneration process. HDAC is an epigenetic factor which removes acetyl groups from histone so that chromatin assembly is tightly packed. This process facilitates stem cell for differentiation process (Lawlor and Yang 2019). Sivaraman et al. reported that 21 amino acid peptide, which was isolated from collagen, facilitate better adhesion property for HeLa cells and show significant lead in tissue regeneration (Sivaraman et al. 2019). Biomimetic peptides possess great potential for tissue regeneration. There are mainly three categories of peptides which can efficiently regenerate the tissue, namely, pro angiogenic peptide, anti-inflammatory peptide, and pro adherence peptide (Hosoyama et al. 2019).
5.6 Emerging Approach of Immobilized Biomolecules as Nanozyme
Nanomaterials with enzyme-mimicking properties are known as nanozymes. Nanozymes have effective catalytic sites for reactions which can serve as substitute of traditional enzymes for catalysis. Nanozyme possess inherent nanomaterial properties and can also act on a dynamic interfacing platform for complex biological environments (Zeng et al. 2018; Wu et al. 2019). Nowadays, nanomaterial engineering provides powerful biological interfaces to improve the performance of biosensors. Here, we have tried to compile few recent literature reports on biomolecules functionalized nano interfaces used for development of nanozyme. DNA, proteins, and peptides are utilized for functionalization of nanozyme. Different immobilization strategies for a range of DNA nanoscale biointerfaces, proteins recognitions, and peptide conjugations were optimized to modulate enzymatic and biological properties of the nanozyme system (Zhang et al. 2019). Fu et al. reported DNA-based platinum (Pt) nanomaterial for peroxidase mimic nanozyme application (Fu et al. 2014). Peptide-conjugated AuNPs were used as nanoprobe for quantification of integrin GPIIb/IIIa on the human erythroleukemia cell line. In addition, this nanoprobe also mimicked peroxidase activity which increased the sensitivity of integrin quantification on colorimetric basis (Gao et al. 2015). Similarly, anti-E.coli antibody was conjugated on platinum (Pt)–coated gold NPs for sensitive detection of bacteria via lateral flow immunoassay (Bradbury et al. 2019).
5.7 Conclusion and Future Aspects
Immobilization of biomolecules is a powerful tool for the development of novel biologically active nanomaterials. It is widely applicable for low-cost production and reusability of biomolecules. Within a few decades, scientific community and industries are witnessing remarkable achievements in immobilization techniques. The versatility of biomolecules plays an important role in the field of controlled utilization and modulation of these molecules with nanoparticles. Nanotechnology is providing solutions to all the limitations related to NPs–biomolecules conjugates. As we have seen in this chapter, many biomolecules can act in a similar manner under different immobilization conditions. Biological application of NPs and their conjugates are widely feasible due to enhanced stability and specificity. The rapid development in nanotechnology also makes the preparation of various nanoparticles more affordable. Biomolecules–NPs conjugates (bioconjugates) not only possess therapeutic and diagnostic activity but also inherit some intrinsic properties of nanoparticles, which benefit biosensing, diagnostics, cell killing, healing, and many other areas in the biomedical field. However, apart from impressive literature reports, several disadvantages of bioconjugates such as aggregation, precipitation, and bio-incompatibility are still challenging concerns. Despite some limitations, in immobilization process, biomolecules are still utilized for high-end applications. In the recent decade, tissue engineering application is the most attractive application in which carbon nanomaterials and polymeric nanomaterials are currently well explored. Recent literatures also suggest that biomolecules conjugated with nanomaterials can be used as surrogated artificial enzymes known as nanozymes. Thus, it can be commented that immobilization of biomolecules on the surface of advance substrates such as nanomaterials will pave the path for future applied science and technological outcomes.
Abbreviations
- AMP:
-
Antimicrobial peptide
- AuNPs:
-
Gold nanoparticles
- CLE:
-
Cross-linking enzymes
- CLEAs:
-
Cross-linked enzyme aggregations
- dsDNA:
-
Double-stranded deoxyribonucleic acid
- EDC:
-
Ethyl (dimethylaminopropyl) carbodiimide
- FRET:
-
Förster resonance energy transfer
- HDAC:
-
Histone deacetylase
- MCH:
-
6-mercato-1-hexanol
- MNPs:
-
Magnetic nanoparticles
- MWCNTs:
-
Multiwalled carbon nanotubes
- NHS:
-
N-hydroxysuccinimide
- NMs:
-
Nanomaterials
- NPs:
-
Nanoparticles
- PEG:
-
Polyethylene glycol
- R & D:
-
Research and development
- SI-ATRP:
-
Surface-initiated atom-transfer radical polymerization
- ssDNA:
-
Single-stranded deoxyribonucleic acid
- SWCNTs:
-
Single-walled carbon nanotubes
References
Ansari SA, Husain Q (2012) Potential applications of enzymes immobilized on/in nano materials: a review. Biotechnol Adv 30(3):512–523
Ansari SA, Husain Q, Qayyum S, Azam A (2011) Designing and surface modification of zinc oxide nanoparticles for biomedical applications. Food Chem Toxicol 49(9):2107–2115
Asal M, Özen Ö, Şahinler M, Baysal HT, Polatoğlu İ (2019) An overview of biomolecules, immobilization methods and support materials of biosensors. Sens Rev 39(3):377–386
Bahraman F, Alemzadeh I (2017) Optimization of l-asparaginase immobilization onto calcium alginate beads. Chem Eng Commun 204(2):216–220
Bahrami A, Vincent T, Garnier A, Larachi F, Boukouvalas J, Iliuta MC (2017) Noncovalent immobilization of optimized bacterial cytochrome P450 BM3 on functionalized magnetic nanoparticles. Ind Eng Chem Res 56(39):10981–10989
Benvidi A, Firouzabadi AD, Tezerjani MD, Moshtaghiun SM, Mazloum-Ardakani M, Ansarin A (2015) A highly sensitive and selective electrochemical DNA biosensor to diagnose breast cancer. J Electroanal Chem 750:57–64
Borrelli GM, Trono D (2015) Recombinant lipases and phospholipases and their use as biocatalysts for industrial applications. Int J Mol Sci 16(9):20774–20840
Bradbury DW, Azimi M, Diaz AJ, Pan AA, Falktoft CH, Wu BM, Kamei DT (2019) Automation of biomarker preconcentration, capture, and Nanozyme signal enhancement on paper-based devices. Anal Chem 91(18):12046–12054
Brady D, Jordaan J (2009) Advances in enzyme immobilisation. Biotechnol Lett 31(11):1639–1650
Broguiere N, Formica F, Barreto G, Zenobi-Wong M (2018) Sortase A as a cross-linking enzyme in tissue engineering. Acta Biomater 77:182–190
Cacicedo ML, Manzo RM, Municoy S, Bonazza HL, Islan GA, Desimone M, Castro GR (2019) Immobilized enzymes and their applications. In: Advances in enzyme technology. Elsevier, Amsterdam, pp 169–200
Cao L, Langen LV, Sheldon RA (2003) Immobilised enzymes: carrier bound or carrier-free? Curr Opin Biotechnol 14(4):387–394
Cesbron Y, Shaheen U, Free P, Lévy R (2015) TAT and HA2 facilitate cellular uptake of gold nanoparticles but do not lead to cytosolic localisation. PLoS One 10(4):e0121683
Chapurina YE, Drozdov AS, Popov I, Vinogradov VV, Dudanov IP (2016) Streptokinase@ alumina nanoparticles as a promising thrombolytic colloid with prolonged action. J Mater Chem B 4(35):5921–5928
Chaudhari AA, Joshi S, Vig K, Sahu R, Dixit S, Baganizi R, Pillai S (2019) A three-dimensional human skin model to evaluate the inhibition of Staphylococcus aureus by antimicrobial peptide-functionalized silver carbon nanotubes. J Biomater Appl 33(7):924–934
Chauhan S, Vohra A, Lakhanpal A, Gupta R (2015) Immobilization of commercial pectinase (Polygalacturonase) on celite and its application in juice clarification. J Food Process Preserv 39(6):2135–2141
Choi EJ, Ahn HW, Kim WJ (2015) Effect of α-acetolactate decarboxylase on diacetyl content of beer. Food Sci Biotechnol 24(4):1373–1380
Choi B, Rempala GA, Kim JK (2017) Beyond the Michaelis-Menten equation: accurate and efficient estimation of enzyme kinetic parameters. Sci Rep 7(1):17018
Cipolatti EP, Silva MJA, Klein M, Feddern V, Feltes MMC, Oliveira JV, de Oliveira D (2014) Current status and trends in enzymatic nanoimmobilization. J Mol Catal B Enzym 99:56–67
Compostella F, Pitirollo O, Silvestri A, Polito L (2017) Glyco-gold nanoparticles: synthesis and applications. Beilstein J Org Chem 13(1):1008–1021
Dar AI, Walia S, Acharya A (2019) Molecular recognition based rapid diagnosis of immunoglobulins via proteomic profiling of protein-nanoparticle complexes. Int J Biol Macromol 138:156–167
Devi KS, Mahalakshmi VT, Ghosh AR, Kumar AS (2017) Unexpected co-immobilization of lactoferrin and methylene blue from milk solution on a Nafion/MWCNT modified electrode and application to hydrogen peroxide and lactoferrin biosensing. Electrochim Acta 244:26–37
Díaz-Hernández A, Gracida J, García-Almendárez BE, Regalado C, Núñez R, Amaro-Reyes A (2018) Characterization of magnetic nanoparticles coated with chitosan: a potential approach for enzyme immobilization. J Nanomater 9468574:11
Drozd M, Pietrzak MD, Malinowska E (2018) SPRi-based biosensing platforms for detection of specific DNA sequences using thiolate and dithiocarbamate assemblies. Front Chem 6:173
Ebrahimi M, Raoof JB, Ojani R (2018) Sensitive electrochemical DNA-based biosensors for the determination of Ag+ and Hg 2+ ions and their application in analysis of amalgam filling. J Iran Chem Soc 15(8):1871–1880
Eghbali M, Farahbakhsh A, Rohani A, Pour AN (2015) Urea biosensor based on immobilization of urease on ZnO nanoparticles. Orient J Chem 31(2):1237–1242
Endo-Takahashi Y, Ooaku K, Ishida K, Suzuki R, Maruyama K, Negishi Y (2016) Preparation of angiopep-2 peptide-modified bubble liposomes for delivery to the brain. Biol Pharm Bull 39(6):977–983
Fatarella E, Spinelli D, Ruzzante M, Pogni R (2014) Nylon 6 film and nanofiber carriers: preparation and laccase immobilization performance. J Mol Catal B Enzym 102:41–47
Fu J, Reinhold J, Woodbury NW (2011) Peptide-modified surfaces for enzyme immobilization. PLoS One 6(4):e18692
Fu Y, Zhao X, Zhang J, Li W (2014) DNA-based platinum nanozymes for peroxidase mimetics. J Phys Chem C 118(31):18116–18125
Fu Y, Han HH, Zhang J, He XP, Feringa BL, Tian H (2018) Photocontrolled fluorescence “double-check” bioimaging enabled by a glycoprobe–protein hybrid. J Am Chem Soc 140(28):8671–8674
Ganesana M, Istarnboulie G, Marty J-L, Noguer T, Andreescu S (2011) Site-specific immobilization of a (His) 6-tagged acetylcholinesterase on nickel nanoparticles for highly sensitive toxicity biosensors. Biosens Bioelectron 30:43–48
Gao L, Liu M, Ma G, Wang Y, Zhao L, Yuan Q, Zhao Y (2015) Peptide-conjugated gold nanoprobe: intrinsic nanozyme-linked immunsorbant assay of integrin expression level on cell membrane. ACS Nano 9(11):10979–10990
Garg S, De A, Mozumdar S (2015) pH-dependent immobilization of urease on glutathione-capped gold nanoparticles. J Biomed Mater Res Part A 103(5):1771–1783
Gebreyohannes AY, Mazzei R, Marei Abdelrahim MY, Vitola G, Porzio E, Manco G, Giorno L (2018) Phosphotriesterase-magnetic nanoparticle bioconjugates with improved enzyme activity in a biocatalytic membrane reactor. Bioconjug Chem 29(6):2001–2008
Ghannadi S, Abdizadeh H, Miroliaei M, Saboury AA (2019) Immobilization of alcohol dehydrogenase on titania nanoparticles to enhance enzyme stability and remove substrate inhibition in the reaction of formaldehyde to methanol. Ind Eng Chem Res 58(23):9844–9854
Guerrero S, Araya E, Fiedler JL, Arias JI, Adura C, Albericio F, Kogan MJ (2010) Improving the brain delivery of gold nanoparticles by conjugation with an amphipathic peptide. Nanomedicine 5(6):897–913
Hartmann M, Kostrov X (2013) Immobilization of enzymes on porous silica–benefits and challenges. Chem Soc Rev 42(15):6277–6289
Hassan RA, Heng LY, Tan LL (2019) Novel DNA biosensor for direct determination of carrageenan. Sci Rep 9(1):6379
He Y, Xie S, Yang X, Yuan R, Chai Y (2015) Electrochemical peptide biosensor based on in situ silver deposition for detection of prostate specific antigen. ACS Appl Mater Interfaces 7(24):13360–13366
He J, Chen J, Hu G, Wang L, Zheng J, Zhan J, Wang Y (2018) Immobilization of an antimicrobial peptide on silicon surface with stable activity by click chemistry. J Mater Chem B 6(1):68–74
Heid S, Unterweger H, Tietze R, Friedrich R, Weigel B, Cicha I, Lyer S (2017) Synthesis and characterization of tissue plasminogen activator—functionalized superparamagnetic iron oxide nanoparticles for targeted fibrin clot dissolution. Int J of Mol Sci 18(9):1837
Homaei AA, Sariri R, Vianello F, Stevanato R (2013) Enzyme immobilization: an update. J Chem Bio 6(4):185–205
Hood ED, Chorny M, Greineder CF, Alferiev IS, Levy RJ, Muzykantov VR (2014) Endothelial targeting of nanocarriers loaded with antioxidant enzymes for protection against vascular oxidative stress and inflammation. Biomaterials 35(11):3708–3715
Hosoyama K, Lazurko C, Muñoz M, McTiernan CD, Alarcon EI (2019) Peptide-based functional biomaterials for soft-tissue repair. Front Bioeng Biotechnol 7:205
Hosseini SM, Kim SM, Sayed M, Younesi H, Bahramifar N, Park JH, Pyo SH (2019) Lipase-immobilized chitosan-crosslinked magnetic nanoparticle as a biocatalyst for ring opening esterification of itaconic anhydride. Biochem Eng J 143:141–150
Ingle AP, Rathod J, Pandit R, da Silva SS, Rai M (2017) Comparative evaluation of free and immobilized cellulase for enzymatic hydrolysis of lignocellulosic biomass for sustainable bioethanol production. Cellulose 24(12):5529–5540
Jia M, Li S, Zang L, Lu X, Zhang H (2018) Analysis of biomolecules based on the surface enhanced Raman spectroscopy. Nano 8(9):730
Jiang Y, Du J, Tang H, Zhang X, Li W, Wang L, Yu D (2019a) Synthesis and application of nanomagnetic immobilized phospholipase C. J Chem 2019:5951793
Jiang Y, Liu H, Wang L, Zhou L, Huang Z, Ma L, He Y, Shi L, Gao J (2019b) Virus-like organosilica nanoparticles for lipase immobilization: characterization and biocatalytic applications. Biochem Eng J 144:125–134
Kale AA, Torchilin VP (2007) Enhanced transfection of tumor cells in vivo using “smart” pH-sensitive TAT-modified pegylated liposomes. J Drug Target 15(7–8):538–545
Khoshnevisan K, Poorakbar E, Baharifar H, Barkhi M (2019) Recent advances of cellulase immobilization onto magnetic nanoparticles: an update review. Magnetochemistry 5(2):36
Kim S, Cui ZK, Koo B, Zheng J, Aghaloo T, Lee M (2018a) Chitosan–lysozyme conjugates for enzyme-triggered hydrogel degradation in tissue engineering applications. ACS Appl Mater Interfaces 10(48):41138–41145
Kim K, Lee J, Moon B, Seo Y, Park C, Park M, Sung G (2018b) Fabrication of a urea biosensor for real-time dynamic fluid measurement. Sensors 18(8):2607
Kitamura T, Sakuma S, Shimosato M, Higashino H, Masaoka Y, Kataoka M, Koike S (2015) Specificity of lectin-immobilized fluorescent nanospheres for colorectal tumors in a mouse model which better resembles the clinical disease. Contrast Media Mol Imaging 10(2):135–143
Kouassi GK, Irudayaraj J, McCarty G (2005) Examination of cholesterol oxidase attachment to magnetic nanoparticles. J Nanobiotechnol 3(1):1
Kuswandi B, Irmawati T, Hidayat M, Ahmad M (2014) A simple visual ethanol biosensor based on alcohol oxidase immobilized onto polyaniline film for halal verification of fermented beverage samples. Sensors 14(2):2135–2149
Labrou NE, Muharram MM (2016) Biochemical characterization and immobilization of Erwinia carotovora L-asparaginase in a microplate for high-throughput biosensing of L-asparagine. Enzyme Microb Technol 92:86–93
Lawlor L, Yang XB (2019) Harnessing the HDAC–histone deacetylase enzymes, inhibitors and how these can be utilised in tissue engineering. Int J Oral Sci 11(2):20
Le Goff A, Holzinger M (2018) Molecular engineering of the bio/nano-interface for enzymatic electrocatalysis in fuel cells. Sustain Energy Fuels 2(12):2555–2566
Lee CK, Au-Duong AN (2018) Enzyme immobilization on nanoparticles: recent applications. Emerg Areas Bioeng 1:67–80
Li D, He Q, Cui Y, Duan L, Li J (2007) Immobilization of glucose oxidase onto gold nanoparticles with enhanced thermostability. Biochem Biophys Res Commun 355(2):488–493
Lin Y, Liu X, Xing Z, Geng Y, Wilson J, Wu D, Kong H (2017) Preparation and characterization of magnetic Fe3O4–chitosan nanoparticles for cellulase immobilization. Cellulose 24(12):5541–5550
Mazlan SZ, Hanifah SA (2017) Effects of temperature and pH on immobilized laccase activity in conjugated methacrylate-acrylate microspheres. Int J Poly Sci 2017:5657271
Meneguetti GP, Santos JHPM, Obreque KMT, Barbosa CMV, Monteiro G, Farsky SHP, Pessoa-Junior A (2019) Novel site-specific PEGylated L-asparaginase. PLoS One 14(2):0211951
Miyawaki A, Niino Y (2015) Molecular spies for bioimaging—fluorescent protein-based probes. Mol Cell 58(4):632–643
Mogharabi M, Nassiri-Koopaei N, Bozorgi-Koushalshahi M, Nafissi-Varcheh N, Bagherzadeh G, Faramarzi MA (2012) Immobilization of laccase in alginate-gelatin mixed gel and decolorization of synthetic dyes. Bioinorg Chem Appl 2012:823830
Monajati M, Borandeh S, Hesami A, Mansouri D, Tamaddon AM (2018) Immobilization of L-asparaginase on aspartic acid functionalized graphene oxide nanosheet: enzyme kinetics and stability studies. Chem Eng J 354:1153–1163
Nemzer LR, Schwartz A, Epstein AJ (2010) Enzyme entrapment in reprecipitated polyaniline nano- and microparticles. Macromolecules 43(9):4324–4330
Nesakumar N, Sethuraman S, Krishnan UM, Rayappan JBB (2016) Electrochemical acetylcholinesterase biosensor based on ZnO nanocuboids modified platinum electrode for the detection of carbosulfan in rice. Biosens Bioelectron 77:1070–1077
Núñez-Villanueva D, Ciaccia M, Iadevaia G, Sanna E, Hunter CA (2019) Sequence information transfer using covalent template-directed synthesis. Chem Sci 10(20):5258–5266
Park JM, Kim M, Park HS, Jang A, Min J, Kim YH (2013) Immobilization of lysozyme-CLEA onto electrospun chitosan nanofiber for effective antibacterial applications. Int J Bio Macromol 54:37–43
Pasha KM, Anuradha P, Subbarao D (2013) Applications of pectinases in industrial sector. Int J Pure Appl Sci Technol 16(1):89
Poon W, Zhang X, Bekah D, Teodoro JG, Nadeau JL (2015) Targeting B16 tumors in vivo with peptide-conjugated gold nanoparticles. Nanotechnology 26(28):285101
Prabhulkar S, Tian H, Wang X, Zhu JJ, Li CZ (2012) Engineered proteins: redox properties and their applications. Antioxid Redox Signal 17(12):1796–1822
Pundir CS (ed) (2015) Enzyme noparticles, vol 69. William Andrew Publishing, Boston
Qie Y, Yuan H, Von Roemeling CA, Chen Y, Liu X, Shih KD, Kim BY (2016) Surface modification of nanoparticles enables selective evasion of phagocytic clearance by distinct macrophage phenotypes. Sci Rep 6:26269
Rafiee-Pour HA, Nejadhosseinian M, Firouzi M, Masoum S (2019) Catalase immobilization onto magnetic multi-walled carbon nanotubes: optimization of crucial parameters using response surface methodology. New J Chem 43(2):593–600
Raveendran S, Parameswaran B, Beevi Ummalyma S, Abraham A, Kuruvilla Mathew A, Madhavan A, Pandey A (2018) Applications of microbial enzymes in food industry. Food Technol Biotechnol 56(1):16–30
Regalado C, García-Almendárez BE, Duarte-Vázquez MA (2004) Biotechnological applications of peroxidases. Phytochem Rev 3(1–2):243–256
Rodrigues RC, Ortiz C, Berenguer-Murcia Á, Torres R, Fernández-Lafuente R (2013) Modifying enzyme activity and selectivity by immobilization. Chem Soc Rev 42(15):6290–6307
Rodriguez-Quijada C, Sánchez-Purrà M, de Puig H, Hamad-Schifferli K (2018) Physical properties of biomolecules at the nanomaterial interface. J Phys Chem B 122(11):2827–2840
Sahare P, Ayala M, Vazquez-Duhalt R, Agrawal V (2014) Immobilization of peroxidase enzyme onto the porous silicon structure for enhancing its activity and stability. Nanoscale Res Lett 9(1):409
Sakuma S, Yano T, Masaoka Y, Kataoka M, Hiwatari KI, Tachikawa H, Tang L (2009) In vitro/in vivo biorecognition of lectin-immobilized fluorescent nanospheres for human colorectal cancer cells. J Control Release 134(1):2–10
Schöttler S, Becker G, Winzen S, Steinbach T, Mohr K, Landfester K, Wurm FR (2016) Protein adsorption is required for stealth effect of poly (ethylene glycol)-and poly (phosphoester)-coated nanocarriers. Nat Nanotechnol 11(4):372
Secundo F (2013) Conformational changes of enzymes upon immobilisation. Chem Soc Rev 42(15):6250–6261
Sharma B, Mandani S, Sarma TK (2014) Enzymes as bionanoreactors: glucose oxidase for the synthesis of catalytic Au nanoparticles and Au nanoparticle–polyaniline nanocomposites. J Mater Chem B 2(26):4072–4079
Siepenkoetter T, Salaj-Kosla U, Xiao X, Conghaile PÓ, Pita M, Ludwig R, Magner E (2017) Immobilization of redox enzymes on nanoporous gold electrodes: applications in biofuel cells. Chem Plus Chem 82(4):553–560
Sirbu T (2011) The searching of active catalase producers among the microscopic fungi. An Univ Oradea Fasc Biol 18(2):164–167
Sivaraman K, Muthukumar K, Shanthi C (2019) A potential bioactive peptide candidate for biomaterial and tissue engineering applications. Life Sci 226:140–148
Skogs M, Stadler C, Schutten R, Hjelmare M, Gnann C, Björk L, Lundberg E (2016) Antibody validation in bioimaging applications based on endogenous expression of tagged proteins. J Proteome Res 16(1):147–155
Souza CJ, Garcia-Rojas EE, Favaro-Trindade CS (2018) Lactase (β-galactosidase) immobilization by complex formation: impact of biopolymers on enzyme activity. Food Hydrocoll 83:88–96
Spicer CD, Jumeaux C, Gupta B, Stevens MM (2018) Peptide and protein nanoparticle conjugates: versatile platforms for biomedical applications. Chem Soc Rev 47(10):3574–3620
Sur A, Pradhan B, Banerjee A, Aich P (2015) Immune activation efficacy of indolicidin is enhanced upon conjugation with carbon nanotubes and gold nanoparticles. PLoS One 10(4):e0123905
Szałapata K, Osińska-Jaroszuk M, Bryjak J, Jaszek M, Jarosz-Wilkołazka A (2016) Novel application of porous and cellular materials for covalent immobilization of pepsin. Braz J Chem Eng 33(2):251–260
Talebi M, Vaezifar S, Jafary F, Fazilati M, Motamedi S (2016) Stability improvement of immobilized α-amylase using nano pore zeolite. Iran J Biotechnol 14(1):33
Thandavan K, Gandhi S, Sethuraman S, Rayappa JBB, Krishnan UM (2013) Development of electrochemical biosensor with nano-interface for xanthine sensing–a novel approach for fish freshness estimation. Food Chem 139(1-4):963–969
Thiruppathiraja C, Kamatchiammal S, Adaikkappan P, Santhosh DJ, Alagar M (2011) Specific detection of Mycobacterium sp. genomic DNA using dual labeled gold nanoparticle based electrochemical biosensor. Anal Biochem 417(1):73–79
Uygun DA, Öztürk N, Akgöl S, Denizli A (2012) Novel magnetic nanoparticles for the hydrolysis of starch with Bacillus licheniformis α-amylase. J Appl Poly Sci 123(5):2574–2581
Venditti I, Palocci C, Chronopoulou L, Fratoddi I, Fontana L, Diociaiuti M, Russo MV (2015) Candida rugosa lipase immobilization on hydrophilic charged gold nanoparticles as promising biocatalysts: activity and stability investigations. Colloids Surf B Biointerfaces 131:93–101
Walia S, Shukla AK, Sharma C, Acharya A (2019) Engineered bright blue-and red-emitting carbon dots facilitate synchronous imaging and inhibition of bacterial and cancer cell progression via 1O2-mediated DNA damage under photoirradiation. ACS Biomater Sci Eng 5(4):1987–2000
Wang Q, Zhang B, Lin X, Weng W (2011) Hybridization biosensor based on the covalent immobilization of probe DNA on chitosan–mutiwalled carbon nanotubes nanocomposite by using glutaraldehyde as an arm linker. Sens Actuators B Chem 156(2):599–605
Wang X, Matei E, Deng L, Koharudin L, Gronenborn AM, Ramström O, Yan M (2013a) Sensing lectin–glycan interactions using lectin super-microarrays and glycans labeled with dye-doped silica nanoparticles. Biosens Bioelectron 47:258–264
Wang S, Su P, Huang J, Wu J, Yang Y (2013b) Magnetic nanoparticles coated with immobilized alkaline phosphatase for enzymolysis and enzyme inhibition assays. J Mater Chem B 1(12):1749–1754
Wang L, Liao X, Ding Y, Gao F, Wang Q (2014) DNA biosensor based on a glassy carbon electrode modified with electropolymerized Eriochrome Black T. Microchim Acta 181(1-2):155–162
Wang XY, Jiang XP, Li Y, Zeng S, Zhang YW (2015a) Preparation Fe3O4@ chitosan magnetic particles for covalent immobilization of lipase from Thermomyces lanuginosus. Int J Bio Macromol 75:44–50
Wang K, Lei Y, Zhong GX, Zheng YJ, Sun ZL, Peng HP, Lin XH (2015b) Dual-probe electrochemical DNA biosensor based on the “Y” junction structure and restriction endonuclease assisted cyclic enzymatic amplification for detection of double-strand DNA of PML/RARα related fusion gene. Biosens Bioelectron 71:463–469
Wu J, Wang X, Wang Q, Lou Z, Li S, Zhu Y, Wei H (2019) Nanomaterials with enzyme-like characteristics (nanozymes): next-generation artificial enzymes (II). Chem Soc Rev 48(4):1004–1076
Xu B, Zheng D, Qiu W, Gao F, Jiang S, Wang Q (2015) An ultrasensitive DNA biosensor based on covalent immobilization of probe DNA on fern leaf-like α- Fe2O3 and chitosan Hybrid film using terephthalaldehyde as arm-linker. Biosens Bioelectron 72:175–181
Yang L, Li X, Yan S, Wang M, Liu P, Dong Y, Zhang C (2015) Single-walled carbon nanotubes–carboxyl-functionalized graphene oxide-based electrochemical DNA biosensor for thermolabile hemolysin gene detection. Anal Methods 7(12):5303–5310
Yoo EH, Lee SY (2010) Glucose biosensors: an overview of use in clinical practice. Sensors 10(5):4558–4576
Yu CY, Huang LY, Kuan I, Lee SL (2013) Optimized production of biodiesel from waste cooking oil by lipase immobilized on magnetic nanoparticles. Int J Mol Sci 14(12):24074–24086
Zeng C, Lu N, Wen Y, Liu G, Zhang R, Zhang J, Zhang M (2018) Engineering nanozymes using DNA for catalytic regulation. ACS Appl Mater Interfaces 11(2):1790–1799
Zhang Y, Ge J, Liu Z (2015) Enhanced activity of immobilized or chemically modified enzymes. ACS Catal 5(8):4503–4513
Zhang L, Wang B, Wang S, Zhang W (2018) Recyclable trypsin immobilized magnetic nanoparticles based on hydrophilic polyethylenimine modification and the proteolytic characteristics. Anal Methods 10(4):459–466
Zhang L, Qi Z, Zou Y, Zhang J, Xia W, Zhang R, Li J (2019) Engineering DNA–nanozyme interfaces for rapid detection of dental Bacteria. ACS Appl Mater Interfaces 11(34):30640–30647
Zhao F, Wang Q, Dong J, Xian M, Yu J, Yin H, Wang J (2017) Enzyme-inorganic nanoflowers/alginate microbeads: an enzyme immobilization system and its potential application. Process Biochem 57:87–94
Zheng Q, Wu H, Shen Z, Gao W, Yu Y, Ma Y, Ding K (2015) An electrochemical DNA sensor based on polyaniline/graphene: high sensitivity to DNA sequences in a wide range. Analyst 140(19):6660–6670
Zhou J, Li H, Yang H, Cheng H, Lai G (2017) Immobilization of glucose oxidase on a carbon nanotubes/dendrimer-ferrocene modified electrode for reagent less glucose biosensing. J Nanosci Nanotech 17(1):212–216
Acknowledgment
The authors would like to thank Director, Council of Scientific and Industrial Research–Institute of Himalayan Bioresource Technology (CSIR-IHBT) for his constant support and encouragement. AA acknowledges financial assistance in the form of project grant MLP-0201 from CSIR and GAP-0214 (EMR/2016/003027) from Department of Science & Technology (DST), Government of India. AKS and MV acknowledge CSIR-GATE, and DST, Government of India for their respective fellowships. The CSIR-IHBT communication number of this manuscript is 4504.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Shukla, A.K., Verma, M., Acharya, A. (2020). Biomolecules Immobilized Nanomaterials and Their Biological Applications. In: Acharya, A. (eds) Nanomaterial - Based Biomedical Applications in Molecular Imaging, Diagnostics and Therapy. Springer, Singapore. https://doi.org/10.1007/978-981-15-4280-0_5
Download citation
DOI: https://doi.org/10.1007/978-981-15-4280-0_5
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-4279-4
Online ISBN: 978-981-15-4280-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)