Abstract
Tropical home gardens are characterized by biodiversity making them beneficial agroforestry ecosystems. Home gardens associated with each dwelling provide direct benefits like food and fodder in addition to indirect benefits and ecosystem services like microclimate regulation and enhancement of the soil carbon content. The most important ecologically relevant fauna reported in home gardens is the soil microarthropods which are considered to be determinants of soil quality. Their features like edaphic adaptations which are helpful in participating in soil biogeochemical cycles through litter decomposition have made them important tools in assessing soil quality. Soil microarthropods being sensitive to soil temperature and soil moisture can alter in number and species composition in relation to seasonal perturbations and soil ecosystem alterations, thus functioning as an efficient tool in biomonitoring studies. The effect of temperature on soil microarthropods and common indices used in biomonitoring of tropical home gardens are discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Home gardens are considered to be valuable traditional agro-biodiversity systems (Kumar et al. 1994; Sankar and Chandrashekara 2002; Soumya et al. 2007) in the tropical regions, as they along with livelihood security provide ecosystem services like efficient nutrient cycling, low use of external inputs, and soil conservation (Torquebiau 1992; Jensen 1993a, b; Jose and Shanmugaratnam 1993) in addition to regulation of microclimate of the corresponding areas where they are present. About 15–20% of the world’s food supply is provided by traditional multiple cropping systems including home gardens (Altieri 1999). They also contribute to enhancing food security of a nation (Mellisse et al. 2018). The home gardens are small-holder agroforestry systems with high tree densities and are ecologically significant ecosystems with high carbon stocks (Nero et al. 2018; Subba et al. 2018). It is reported that these traditional agroforestry systems in the tropics hold a number of annual and perennial plant species per field and most species have their economic importance as construction materials, firewood, medicine, food, and livestock feed. These trees also protect soil nutrients by reducing soil erosion. Another important contribution of tropical home gardens to soil biodiversity is by maintaining a population of soil microarthropods which are prominent players in soil organic matter recycling, thereby contributing to soil quality enhancement (Vreeken-Buijs et al. 1998). The active involvement of soil microarthropods in soil food webs helps in maintaining a healthy soil ecosystem. A recent approach to evaluation of the soil quality of home gardens is by utilizing the indicator value of soil microarthropods (van Straleen and Verhoef 1997) which has been used to estimate the soil quality (Paolo et al. 2010; Madej and Kozub 2014) in different land areas and has proved to be efficient. According to Giller (1996), soil communities help to sustain high biodiversity, but due to the lack of knowledge in the community structure and presence of gaps in description of the soil community species, they are called “the poor man’s tropical rainforests” (Usher et al. 1979).
2 General Features of the Home Gardens in Kerala
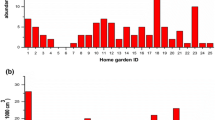
The home garden generally represents an ecosystem with mixed plant functional groups like grasses, forbs, shrubs, trees, and climbers. The plant groups found in tropical home garden agro-ecosystems have economic significance in terms of their use as medicinal, flowering, and fruiting plants. The good canopy cover offered by trees and the extensive taproot systems help in reducing the overall temperature of the home gardens, reduce soil erosion, and maintain the groundwater table. The home gardens generally show horizontal and vertical stratification. In most home gardens in Kerala, vertical stratification is evident with grasses, herbs, shrubs, and trees. A study related to soil microarthropods was undertaken in rural home gardens in Kerala, India (Lakshmi and Joseph 2017). The home gardens in rural areas like Chengamanad generally have four strata with a height ranging from 70 cm to 28 m (Fig. 17.1), while home gardens in other urban areas generally represent less floral diversity and soil faunal composition.
The diversity of soil microarthropods in the home gardens is an important issue to be addressed considering its importance in soil health of home gardens which are the most important agro-ecosystems linked to livelihood security in coastal areas. This study addresses the soil microarthropods in home gardens in the light of a study undertaken in a rural area in Kerala, India, along with the findings from published research papers on soil microarthropods.
3 Diversity of Soil Microarthropods in Tropical Home Gardens
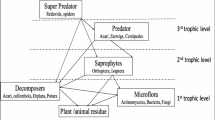
Soil microarthropods are edaphic fauna inhabiting soil and organic debris layer above the soil. Soil microarthropods, considered as “resource biota” contributing to litter decomposition, are important biodiversity component in the tropical home gardens (Fig. 17.2). The arthropod populations under different soil conditions were studied long back by Ford (1937) and later by Dhillon and Gibson (1962). From various studies, it has been understood that soil microarthropods are most abundant near the soil surface characterized by favorable moisture, aeration, and organic matter content (Murphy 1953). The soil microarthropod composition also varies in accordance with soil depth. According to Price and Benham (1977), most arthropod groups declined rapidly in abundance with increasing soil depth. It is also found that the species composition and abundance of soil microarthropods are influenced by geographical location, physicochemical properties of soil, type of vegetation cover, as well as nature and depth of litter.
The attempts to correlate soil fauna with soil fertility date back to Soudek. Later, Fujikawa (1970) stressed on the role of soil microarthropods in litter decomposition and release of nutrients which has a positive effect on soil formation and soil fertility. The significance of Collembola and mites in the breakdown of organic matter and soil formation had also been pointed out by many (Fujikawa 1970; Wolters 2000; Eaton et al. 2004). Due to their widespread presence and abundance in tropical home garden agro-ecosystems, soil microarthropods can be used for the study of similar habitats with different management practices or different habitats within the same geographical area as the changes in the home gardens will be reflected as the presence, absence, or change in number of soil microarthropods. Soil microarthropods include Acarina, Collembola, Protura, Pauropoda, Diplura, and Symphyla groups (Lakshmi and Joseph 2017), among which Acarina and Collembola are found to be most abundant in home garden soils. The occurrence of soil microarthropods in soils is related to various factors such as temperature, moisture, organic matter, etc. For example, in a study of home gardens in a rural area (Lakshmi and Joseph 2017), microarthropod abundance was found to be higher during monsoon compared to summer (Fig. 17.3). Such regional studies point out that the various factors influencing microarthropod population in home gardens need a thorough study. Some of these are discussed here.
Distribution of soil microarthropods during summer season (a) and monsoon season (b) in home garden of a village in Kerala, India (Lakshmi and Joseph 2017)
3.1 Effect of Soil Temperature on Soil Microarthropods
Temperature is one of the determining factors for the survival of soil microarthropods. Studies have proved that soil microarthropods can survive only in soils with optimum temperature which favors their physiological activities. Excess temperature will lead to heat stress in the organisms which responds to the increasing temperature in the form of movement to other soil ecosystems with more favorable conditions. The inability to tolerate increased temperature will ultimately lead to the death of soil microarthropods. Most Collembola and Oribatida mites have been reported to have their lethal temperature limits between 35 °C and 40 °C (Madge 1965; Hodkinson et al. 1996). Vannier (1994) presented a thermo-biological scale for insects, in which he suggested that the optimum temperature for the survival of insects is just above 20 °C. But certain studies have found that a temperature less than even 40 °C can be lethal to soil microarthropods (Malmstrom 2008). This normally occurs during events like forest fires. As sunlight plays an important role in maintaining the soil temperature of home gardens and direct sunlight is a limiting factor for soil microarthropod survival, it has been found that the soils which do not receive direct sunlight show a higher abundance of soil microarthropods compared to those which receive direct sunlight. This has been particularly noted in forest soils with a high canopy cover acting as a barrier for direct sunlight from entering the soil. The same principle is applicable for home gardens with thick canopy cover. Sunlight has a negative relationship with soil moisture, as direct sunlight will cause the evaporation of soil water leading to soil moisture deficit. The reduction in soil moisture will prompt the soil microarthropods to shift their niches to more favorable areas with good soil moisture content. Reduction in soil water content can also cause increased soil temperature leading to heat stress in soil microarthropods. Another observation to be noted is that in the regions where heat-absorbing black humus layer is exposed to direct sunlight, soil microarthropods can be subjected to mortality resulting from the elevated temperature (Malmstrom 2008). It is true that the thermal buffering of soil protects itself from changes in atmospheric temperature (Whitford 1992), and as we go deeper into the soil, the temperature variation decreases with depth to some extent. The saturation of soil with soil water can also reduce the threat of desiccation (Ghilarov 1977) caused due to increased temperature on soil microarthropods. But those soil microarthropods which survive above the soil are more prone to desiccation and death due to heat stress, while the soil microarthropod species that live on the litter surface have the ability to tolerate higher temperatures than species living further down in the soil.
3.1.1 Temperature and Seasonal Distribution of Soil Microarthropods
Soil temperature shows considerable variations in different seasons, especially in tropical countries which experience summer and rainy seasons. Hence the population of soil microarthropods also varies in tropical regions (Parwez and Abbas 2012). Studies have reported a high population of soil microarthropods during monsoon/wet months than during dry months (Verma and Yadav 2014; Begum et al. 2014; Lakshmi and Joseph 2017) in tropical regions. It is also reported that water is the most important factor influencing the population size of soil microarthropods (Badejo 1990). A study on Acari population by Badejo (1990) gives the finding that soil moisture content has a positive relationship with the population of Acari. Generally, a high abundance in soil microarthropods is noticed during wet seasons due to favorable conditions like increased soil moisture content due to precipitation and reduced evaporation. The increased soil moisture content during wet seasons can support a good population of soil microarthropods provided there is sufficient organic matter or litter content in the soil. During dry months, the soil conditions become reversed due to reduced soil moisture content as a result of increased evaporation and reduced precipitation. This can have a negative effect on the survival and physiology of soil microarthropods resulting in reduction of their numbers. For the survival of soil microarthropods, it has been found that a range of species-specific intermediate temperatures is found to be more suitable (Wallwork 1970) than a common range of temperature.
3.1.2 Effect of Fire on Survival of Soil Microarthropods
Forest fires are found to reduce the number of soil microarthropods. Since the temperature of the tropical regions is found to be increasing during summer, it can cause fire in home gardens as well. Fire can also occur due to burning of materials like waste during cleaning of households which the home gardens belong to. Fire releases enormous amount of energy, which increases the soil temperature. For understanding the effect of forest fires on soil microarthropods, a laboratory study was done, and it has been found that the determining factors for increased soil temperature during a fire are the rate of the burn, amount and quality of fuel consumed, soil moisture, and soil conductivity (Fisher and Binkley 2012). Rapidly advancing fires that consume little fuel have minor effects on soil temperature, while slower fires that consume more fuel may result in temperatures exceeding 70 °C at the soil surface, which decline to normal levels only at 15–30 cm depths (DeBano et al. 1998). This can be deteriorative to the majority of soil microarthropods living in the upper soil layers (Hagvar 1983), in addition to bottom-dwelling species of soil microarthropods. After a fire, it will take time for the soil microarthropod community to re-establish through secondary succession.
3.2 Effect of Canopy Cover on the Survival of Soil Microarthropods
Canopy cover in the home gardens indirectly affects soil microarthropod survival by increasing soil moisture content and reducing soil evaporation, thereby providing favorable microhabitat conditions (Lakshmi and Joseph 2015). The canopy cover acts as a barrier preventing direct sunlight from reaching the soil which reduces excessive heating of the soil and loss of soil moisture due to evaporation and increased soil temperature. The water drops on the leaves formed due to precipitation also fall down into the soil, adding to soil moisture content. Usually, a home garden with thick canopy cover offers a cool habitat for soil microarthropods throughout the year compared to with little canopy cover. The canopy cover also holds a good litter layer on the soil, which is invaded by soil microarthropods, helping in their degradation, thereby enhancing soil fertility. Reduced canopy cover often results in higher daytime temperature and in turn leads to reduced soil moisture and higher moisture loss from litter (Zhang and Zak 1995) ultimately resulting in unfavorable soil microarthropod habitats, reduction in microarthropod number, and thereby reduced rate of decomposition.
3.3 Climate Change and Soil Microarthropods
Warming and changes in precipitation can directly alter soil temperature and moisture, thereby affecting soil microarthropod reproduction and development rates (Uvarov 2003) in the home gardens. The impact of warming on soil microarthropods has been studied using different experiments (Coulson et al. 1996; Huhta and Hanninen 2001). According to Sjursen and Michelsen (2005), warming may affect soil microarthropod communities by changing the abundance and composition of soil organisms which they depend for food. Elevated temperature can also affect soil microarthropod communities by causing a change in plant physiology in the home gardens where they belong to. This happens during extreme summer where the soil becomes devoid of moisture causing water stress to the plants in home gardens. Due to reduced soil moisture content, the biomass from the trees falling on soil undergoes slow decomposition, reducing the litter content in the soil. Since the soil microarthropods are litter dwellers, the reduction in the litter content may affect their survival, ultimately resulting in reduction of their numbers. Kardol et al. (2011) has studied the effect of climate change on soil microarthropods using laboratory experiments. According to Harte and Rawa (1996), warming increased microarthropod abundance and biomass under wet conditions compared to dry conditions. This may be because the soil moisture present during wet conditions offers a favorable environment for the survival of soil microarthropods, while the reduced moisture content during dry seasons causes a threat to their survival.
4 Soil Microarthropods and Soil Health
The concept of “soil health” refers to a soil ecosystem which can support the existence and growth of plants and soil organisms like soil microbes and other edaphic fauna like soil invertebrates and vertebrates. While the layman’s concept of soil health refers to a soil rich in nutrients which are helpful for the survival of plants, in scientific terms, soil health is a broader concept which includes all the organisms which are directly or indirectly affected by soil. This network includes organisms which have direct contact with the soil as well as those who indirectly depend on the benefits from the soil. According to FAO, “Soil health is the capacity of soil to function as a living system, with ecosystem and land use boundaries, to sustain plant and animal productivity, maintain or enhance water and air quality, and promote plant and animal health. Healthy soils maintain a diverse community of soil organisms that help to control plant disease, insect and weed pests, form beneficial symbiotic associations with plant roots; recycle essential plant nutrients; improve soil structure with positive repercussions for soil water and nutrient holding capacity, and ultimately improve crop production.” This definition can be extended in an environmental perspective as follows: A healthy soil is one that does not pollute the environment and contributes to buffer climate change by maintaining/increasing the carbon content.
Soil microarthropods being inhabitants of soil can provide excellent information regarding soil health of tropical home gardens. Among the soil microarthropods reported, the commonly used indicators for soil health are Collembola and Acari. According to recent findings, soil quality of home gardens can be quantified using the bioindication value of soil microarthropods through different indices. The different steps involved in determining soil quality indices are soil sampling, soil microarthropod extraction, microarthropod specimen preservation, determination of biological forms, and calculation of indices (Parisi 2001; Madej et al. 2011).
4.1 Ecomorphological Index (EMI)
The ecomorphological index (EMI ) is an index of soil quality given to the soil microarthropods, based on the morphotype of each organism. EMI values ranging from 1 to 20 are assigned to the soil microarthropods considering the morphological features like body length and edaphic adaptations like the absence of wings and visual apparatus. The EMI score uses the presence of eu-edaphic soil microarthropod as the criterion for estimating soil quality (Yan et al. 2012), rather than their abundance. A higher EMI value refers to a higher level of adaptation of soil microarthropods. Since eu-edaphic soil microarthropods are highly adapted to soil habitat, they usually have an EMI score closer to 20 compared to the epi-edaphic and hemi-edaphic organisms having a score closer to 1 (Parisi et al. 2005). Whenever two ecomorphological forms are present in the same group, the final score is determined by the higher EMI . In other words, the most highly adapted microarthropods belonging to a group determine the overall EMI score for that group. The EMI scores of various soil microarthropod groups are given in Table 17.1 (Parisi 2001).
4.2 QBS (Qualità Biologica del Suolo) Score
QBS index (“Qualità Biologica del Suolo”) or Biological Quality of Soil proposed by Parisi (2001) is based on the concept that the higher the soil quality, the higher the number of soil microarthropod groups adapted to soil habitat. The QBS index is calculated as the sum of EMI values in each soil (Parisi 2001). Two types of QBS index are reported, namely, QBS-ar and QBS-c. While QBS-ar relies on the soil quality index determination using soil microarthropod fauna present in a given area obtained by adding up the EMI scores, QBS-c is based on the Collembola-Acari ratio. For estimation of QBS-c, the Collembolans are separated into six groups, namely Podurid, Onychiurid, Isotomid, Entomobryid, Neelid, and Sminthurid. The biological form with the higher EMI value is recorded, and the EMI scores are summed to get QBS-c index. According to Parisi (2001), based on the QBS score, seven soil quality classes can be identified in a home garden (Table 17.2).
The EMI and QBS scores can be effectively used as scores for assessing the soil health of the tropical home gardens due to two reasons: (1) soil health is dependent on soil temperature and associated factors like soil moisture content and soil organic carbon and (2) the higher the soil temperature, the lower will be the QBS and EMI scores. The relation of soil quality indices with soil health was studied in different home gardens. Since tropical countries have a climate that is variable, showing gradation of temperatures and soil moisture and organic carbon content, the soil quality indices will act as a measurement of soil quality because the soil microarthropod fauna also change under the influence of soil temperature and thereby the associated factors like soil organic carbon and soil moisture content.
A simple representation regarding the effect of soil temperature on soil quality of home gardens is shown in Fig. 17.4.
Apart from soil temperature, soil moisture content and soil organic matter content also determine the QBS and EMI scores. Since all of these are dependent, it can be concluded that a change in any one of these will affect the soil microarthropod population and thereby the QBS score. The formation of organic matter in soil is favored by increased soil moisture content and increased amount of litterfall. The increased litterfall in the tropical home gardens can support a high number of eu-edaphic soil microarthropod fauna which will in turn increase the QBS value of the home garden soils. Since an increased QBS-ar value is a determinant of higher soil quality, the home gardens with higher QBS-ar scores are the ones with better soil quality. As management practices like cleaning the home gardens remove the litter cover from the soil, the soil quality of well-managed home gardens is found to be less when compared to the unmanaged home gardens (Gope and Ray 2006; Lakshmi and Joseph 2017). Hence unmanaged tropical home gardens with good canopy cover are a very good storehouse of soil microarthropods which can enhance the soil quality compared to intensively managed home gardens with domesticated plants.
5 Conclusion
Soil microarthropods, the determinants of soil quality of tropical home gardens, are affected by change in soil temperature, soil moisture, soil organic carbon, and anthropogenic activities like land management practices. The relationship of soil microarthropods to soil temperature can be utilized for the assessment of soil quality of tropical home gardens with the help of soil quality indices, and this can serve as a good support for the implementation of proper management practices of tropical home gardens in the future. This approach will also help in developing a common database regarding the soil quality of home gardens in the tropical regions and its changes in the future.
References
Altieri MA (1999) The ecological role of biodiversity in agro ecosystems. Agric Ecosyst Environ 74(1):19–31
Badejo MA (1990) Seasonal abundance of soil mites (Acarina) in two contrasting environments. Biotropica 22(4):382–390
Begum F, Bajracharya RM, Sitaula BK, Sharma S, Ali S, Ali H (2014) Seasonal dynamics and land use effect on soil microarthropod communities in the mid-hills of Nepal. Int J Agron Agric Res 5(2):114–123
Coulson SJ, Hodkinson ID, Webb NR, Block W, Bale JS, Strathdee AT, Worland MR, Wooley C (1996) Effects of experimental temperature elevation on high-arctic soil microarthropod populations. Polar Biol 16:147–153
DeBano LF, Neary DG, Ffolliott PF (1998) Fire’s effects on ecosystems. Wiley, New York
Dhillon BS, Gibson BHE (1962) A study of the Acarina and Collembola of agricultural soils. Pedobiologia 1:189–209
Eaton RJ, Barbercheck M, Buford M, Smith W (2004) Effects of organic matter removal, soil compaction, and vegetation control on Collembolan populations. Pedobiologia 48(2):121–128
Fisher RF, Binkley D (2012) Ecology and management of forest soils, 3rd edn. Wiley, New York
Ford J (1937) Fluctuations in natural populations of Collembola and Acarina. J Anim Ecol 7(2):98–111
Fujikawa T (1970) Distribution of soil animals in three forests of northern Hokkaido: II. Horizontal and vertical distribution of Oribatid mites (Acarina: Cryptostigmata). Appl Entomol Zool 5(4):208–212
Ghilarov MS (1977) Why so many species and so many individuals can coexist in the soil. In: Soil organisms as components of ecosystems. Ecol Bull Stockholm 25:593–597
Giller PS (1996) The diversity of soil communities, the ‘poor man’s tropical rainforest. Biodivers Conserv 5(2):135–168
Gope R, Ray D (2006) Dynamics of soil Acari (Arthropoda: Arachnida) under managed and unmanaged land use of Barak Valley, Assam (North Eastern India). Bull Natl Inst Ecol 17:17–23
Hagvar S (1983) Collembola in Norwegian coniferous forest soils II. Vertical distribution. Pedobiologia 25:383–401
Harte J, Rawa A, Price V (1996) Effects of manipulated soil microclimate on mesofaunal biomass and diversity. Soil Biol Biochem 28:313–322
Hodkinson ID, Coulson SJ, Webb NR, Block W (1996) Can high Arctic soil microarthropods survive elevated summer temperatures? Funct Ecol 10:314–321
Huhta SM, Hanninen SM (2001) Effects of temperature and moisture fluctuations on an experimental soil microarthropod community. Pedobiology 45:279–286
Jensen M (1993a) Soil conditions, vegetation structure and biomass of a Javanese home garden. Agrofor Syst 24:171–186
Jensen M (1993b) Productivity and nutrient cycling in a Javanese home garden. Agrofor Syst 24:187–201
Jose D, Shanmugaratnam N (1993) Traditional home gardens of Kerala: a sustainable human ecosystem. Agrofor Syst 24(2):203–213
Kardol P, Reynolds WN, Norby RJ, Classen AT (2011) Climate change effects on soil microarthropod abundance and community structure. Appl Soil Ecol 47(1):37–44
Kumar BM, George SJ, Chinnamani S (1994) Diversity, structure and standing stock of wood in the home gardens of Kerala in Peninsular India. Agrofor Syst 25:243–262
Lakshmi G, Joseph A (2015) The effect of vegetation cover on the soil microarthropod abundance of home gardens. In Jaya DS (eds) Proceedings of national seminar on soil pollution and paradigms for sustainable soil management (December 4–5, 2015), University of Kerala, Thiruvananthapuram, Kerala, India, pp 83–89
Lakshmi G, Joseph A (2017) Soil microarthropods as indicators of soil quality of tropical home gardens in a village in Kerala, India. Agrofor Syst 91(3):439–450
Madej G, Kozub M (2014) Possibilities of using soil microarthropods, with emphasis on mites (Arachnida, Acari, Mesostigmata), in assessment of successional stages in a reclaimed coal mine dump (Pszów, S Poland). Biol Lett 51(1):19–36
Madej G, Barczyk G, Gdawiec M (2011) Evaluation of soil biological quality index (QBS-ar): its sensitivity and usefulness in the post-mining chronosequence–preliminary research. Pol J Environ Stud 20(5):1367–1372
Madge DS (1965) The effects of lethal temperatures on oribatid mites. Acarologia 7:121–130
Malmstrom A (2008) Temperature tolerance in soil microarthropods: simulation of forest-fire heating in the laboratory. Pedobiologia 51(5):419–426
Mellisse BT, Descheemaeker K, Giller KE, Abebe T, van de Ven GW (2018) Are traditional home gardens in southern Ethiopia heading for extinction? Implications for productivity, plant species richness and food security. Agric Ecosyst Environ 252:1–13
Murphy PW (1953) The biology of forest soils with special reference to the mesofauna or meiofauna. J Soil Sci 4(2):155–193
Nero B, Callo-Concha D, Denich M (2018) Structure, diversity, and carbon stocks of the tree Community of Kumasi, Ghana. Forests 9(9):519
Paolo AG, Boccelli R, Ampollini D, Del Re AA, Capri E (2010) Assessment of soil-quality index based on microarthropods in corn cultivation in northern Italy. Ecol Indic 10(2):129–135
Parisi V (2001) La qualità biologica del suolo. Un metodo basato sui microartropodi. Acta Naturalia de L’Ateno Parmense 37:97–106
Parisi V, Menta C, Gardi C, Jacomini C, Mozzanica E (2005) Microarthropod communities as a tool to assess soil quality and biodiversity: a new approach in Italy. Agric Ecosyst Environ 105:323–333
Parwez H, Abbas MJ (2012) Seasonal diversity, habitat quality and species specific differences of micro arthropods abundance in two different managed agro-ecosystems at Aligarh. Int J Geol Earth Environ Sci 2(2):206–217
Price DW, Benham GS Jr (1977) Vertical distribution of soil-inhabiting microarthropods in an agricultural habitat in California. Environ Entomol 6(4):575–580
Sankar S, Chandrashekara UM (2002) Development and testing of sustainable agroforestry models in different agroclimatic zone of Kerala with emphasis on socio-cultural, economic, technical and institutional factors affecting the sector. Research Report 234, Kerala Forest Research Institute, Peechi
Sjursen H, Michelsen A, Jonasson S (2005) Effects of long-term soil warming and fertilisation on microarthropod abundances in three sub-arctic ecosystems. Appl Soil Ecol 30:148–161
Soumya M, Nair PKR, Long AJ (2007) An assessment of ecological diversity in home gardens: a case study from Kerala State, India. J Sustain Agric 29(4):135–153
Subba M, Pala NA, Shukla G, Chakravarty S (2018) Study of the variability of home gardens influencing carbon stock under sub-humid tropical zone of West Bengal, India. Indian Forester 144(1):60–66
Torquebiau E (1992) Are tropical agroforestry home gardens sustainable? International Centre for Research in Agroforestry (ICRAF). Agric Ecosyst Environ 41:189–207
Usher MB, Davis P, Harris J, Longstaff B (1979) A profusion of species? Approaches towards understanding the dynamics of the populations of microarthropods in decomposer communities. In: Anderson RM, Turner BD, Taylor LR (eds) Population dynamics. Blackwell Scientific, Oxford, pp 359–384
Uvarov AV (2003) Effects of diurnal temperature fluctuations on population responses of forest floor mites. Pedobiologia 47(4):331–339
van Straleen N, Verhoef H (1997) The development of a bioindicator system for soil acidity based on arthropod pH preferences. J Appl Ecol 34:217–232
Vannier G (1994) The thermobiological limits of some freezing intolerant insects: the supercooling and thermostupor points. Acta Oecol 15(1):31–42
Verma D, Yadav R (2014) Preliminary study of population diversity of collembola in Agra regions. Bull Environ Pharmacol Life Sci 3(4):7–9
Vreeken-Buijs MJ, Hassink J, Brussard L (1998) Relationships on soil microarthropods biomass with organic matter and pore size distribution in soil under different land use. Soil Biol Biochem 30:97–106
Wallwork JA (1970) Ecology of soil animals. McGraw Hill, New York
Whitford W (1992) Effects of climate change on soil biotic communities and soil processes. In: Peters R, Laveforg T (eds) Global warming and biological diversity. Yale University Press, USA, pp 124–136
Wolters V (2000) Invertebrate control of soil organic matter stability. Biol Fertil Soils 31(1):1–19
Yan S, Singh AN, Fud S, Liao C, Wang S, Li Y, Cui Y, Huf L (2012) A soil fauna index for assessing soil quality. Soil Biol Biochem 47:158–165
Zhang Q, Zak JC (1995) Effects of gap size on litter decomposition and microbial activity in a subtropical forest. Ecology 76:2196–2204
Acknowledgments
The authors would like to thank the School of Environmental Studies, Cochin University of Science and Technology, Kerala, for providing research facilities. The research was carried out with the help of the Junior Research Fellowship from University Grants Commission (UGC), India, awarded to the first author for conducting research in Environmental Science.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Lakshmi, G., Joseph, A. (2020). Soil Microarthropods as Indicators of Soil Health of Tropical Home Gardens in Kerala,India. In: Dagar, J.C., Gupta, S.R., Teketay, D. (eds) Agroforestry for Degraded Landscapes. Springer, Singapore. https://doi.org/10.1007/978-981-15-4136-0_17
Download citation
DOI: https://doi.org/10.1007/978-981-15-4136-0_17
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-4135-3
Online ISBN: 978-981-15-4136-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)