Abstract
Sulfur (S) is an important macronutrient that plays a significant role in plant growth and development. In the past few decades, efforts focused on reducing sulfur dioxide emission for environment protection had limited the use of S-based fertilizers in agriculture, thereby causing S deficiency in food crops. It also triggered the susceptibility of crop plants to environmental stresses as S assimilation and synthesis of different S compounds modulate several metabolic processes to induce tolerance against various abiotic stresses. The use of inorganic fertilizers containing S has increased tremendously in recent years due to its significance in enhancing crop yield and quality. Therefore, in this chapter, we discuss recent studies on effects of S fertilizers on growth and yield of major cereals (wheat, maize, rice), legumes (mung bean, chickpea, black gram), and oilseeds (sunflower, brassica, soybean). An overview of current state of knowledge on S-mediated physiological and biochemical alterations in food crops may facilitate in developing appropriate fertilizer management strategies to improve yield and quality under abiotic stress conditions.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
14.1 Introduction
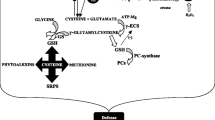
Sulfur (S) is recognized as the fourth major nutrient after nitrogen (N), phosphorus (P), and potassium (K). It is an essential nutrient required to improve crop yield and quality due to its key role in protein synthesis (Yi et al. 2010). It is the main constituent of proteins, thioredoxin (Trx), methionine (Met), cysteine (Cys), vitamins (Vit), sulfo-lipids (SL), and Fe-S cluster system that play an important role in the regulation of physiological metabolism of plants (Khan et al. 2013). Increased S demand during metabolic adaptation processes suggests the key role of S-containing compounds (Anjum et al. 2015). Metabolism of S influences the accumulation of osmolytes and osmo-protectants (Gill et al. 2013), whereas S-containing compounds such as glutathione (GSH) interplay with signaling pathways to ensure sufficient production of metabolites for ABA synthesis (Herrmann et al. 2014). Sulfate interacts with ABA and acts as a chemical signal to initiate stomatal closure in leaves under water deficit conditions (Hasanuzzaman et al. 2018). Anti-transparent effect of ABA is increased by the presence of sulfate in stomata of plant leaves (Ernst et al. 2010). Assimilation of S results in the formation of several S-containing defense compounds including GSH and phytochelatins (PCs) involved in plant survival under various abiotic stresses (Honsel et al. 2011) (Fig. 14.1). Moreover, interplay of S with phytohormones helps to regulate crucial metabolic processes in plants (Noctor et al. 2012). S metabolism is directly linked to polyamines and ethylene through salvage pathway involved in plant response to drought stress (Sauter et al. 2013). Deficiency of S markedly affects the yield potential of plants even under well-watered conditions (Rasheed et al. 2004). Low S levels in soil influence the uptake of nutrients and nitrate reductase metabolism in plants (Prosser et al. 2001).
Plants uptake S in metabolically inactive form known as sulfate (SO4−2) from soil surface. It is reduced into sulfide (S−2) and assimilated into Cys by the activity of ATP sulfurylase (Herrmann et al. 2014). A variety of S compounds such as GSH, Met, and PCs are synthesized from Cys residues which play an important role in alleviating the drastic effects of environmental stresses like drought (Anjum et al. 2015). Sulfur metabolism induces alterations at metabolic and transcriptional levels to activate enzymes and increase root development for maximum uptake of nutrients and water (Ahmad et al. 2016a).
Judicious use of fertilizers and their management is essential to obtain high yield potential in crop plants. Optimum S supplementation significantly influences growth, yield, and quality of plants (Shao et al. 2008). Ahmad et al. (2016b) observed that S supplementation affects N uptake and use efficiency in maize. They applied different levels of S (0, 20, 30, and 40 kg ha−1) in combination with various N application rates (0, 120, and 150 kg ha−1). It was noted that S application significantly increased N uptake in maize at an increasing rate. They recommended S fertilization to improve growth, N use efficiency, and yield of maize. In a greenhouse study, Khan et al. (2015) evaluated the role of S and N fertilization on growth and yield of hybrid sunflower. Although N deficiency delayed all development stages of sunflower, a merked effect of S deficiency was only recorded at floret initiation and anthesis stages. Moreover, S deficiency also reduced Cys and Met by 30% but increased arginine by 34% in achenes. They suggested that timely availability of S is needed to prevent floret abortion in sunflower. Deficiency of S in sunflower at seed filling stage results in kernels with low levels of essential S-containing amino acids. Wani et al. (2001) noted a marked increase in achene yield and quality of sunflower by increasing S doses. A field study on chickpea involving two S sources (gypsum and ammonium sulfate) showed that S supply increased yield by 17% compared to no S application (Islam 2012). It was observed that ammonium sulfate was more efficient than gypsum to improve nutrients uptake and yield in chickpea.
The abovementioned literature highlights the importance of S in plant growth and development. In this chapter, we present a comprehensive overview of S-mediated physiological and biochemical processes involved in improving abiotic stress tolerance in crops (Table 14.1). In addition, comparative effects of different methods of exogenous S supply as well as application of various S sources and their doses are also discussed.
14.2 Role of S in Improving Abiotic Stress Tolerance in Cereals
14.2.1 Wheat
Deficiency of S results in light green to yellow color in young wheat leaves along with stunted growth and spindly stalks. High rainfall and poor drainage during the growth season result in leaching of available S below the root zone. Similarly, fine-textured soils are more likely to be S deficient compared to coarse-textured soil (Shah et al. 2018). Availability of S also facilitates N assimilation in wheat seeds (Tea et al. 2007).
Hydrogen sulfide (H2S) has emerged as a new stress-signalling molecule with multiple effects on plant metabolism to increase tolerance against various environmental stresses. It is considered an important part of S-induced plant defense mechanisms (Aroca et al. 2018). Application of H2S donor, i.e., NaHS, was observed to influence ABA metabolism pathway as well as ABA concentration in roots and leaves of wheat seedlings exposed to drought stress (Ma et al. 2016). Moreover, it also upregulated ABA reactivation and catabolism genes in roots. In another study, NaHS application modulated GSH and ascorbate assimilation in wheat leaves under drought stress (Shan et al. 2011). Pretreatment of NaHS markedly decreased malondialdehyde (MDA) content and electrolyte leakage and upregulated dehydroascorbate reductase (DHAR), gamma-glutamylcysteine synthetase (γGCS), glutathione reductase (GR), ascorbate peroxidase (APX), and gamma-glutamylcysteine synthetase (γGCS) compared to no NaHS supply. NaHS-induced upregulation of drought-responsive genes facilitates phytohormones’ signal transduction, amino acids’ metabolism, and translocation of iron from root to shoot to improve drought tolerance in wheat (Liu et al. 2017).
Salinity stress severely hampers wheat growth and productivity. Exposure to salt stress of 6–8 dS m−1 causes a marked reduction in wheat yield (Royo and Abió 2003). S-containing compounds play a significant role in enhancing salinity tolerance through upregulation of specific genes and modulating several physiological and biochemical processes (Khan et al. 2014). A negative correlation was observed between sodium (Na) content and S application in wheat exposed to saline-sodic soil (Ali et al. 2012). Increasing S doses (0, 25, 50, and 75 kg ha−1) significantly improved nutrient uptake and yield of wheat resulting in high K and calcium (Ca) content in grains to alleviate salinity/sodicity. In a similar study, Arshadullah et al. (2013) observed a negative correlation between Ca and Na ions in wheat plants treated with different doses of CaSO4 (0, 50, 100, 150, 200 kg ha−1) under saline-sodic soil (ECe = 5.32 dS m−1). In contrast, K and Ca ions were positively correlated resulting in maximum wheat yield by application of 150 kg ha−1 CaSO4. Recently, Kausar et al. (2016) reported a marked increase in nutrient uptake and biomass accumulation by treating wheat plants with various K2SO4 doses (0.50, 100, 150, 200 mM) under saline conditions.
Heavy metal (HM) contamination of agricultural soils is a major concern for plant scientists due to potential harms on animals and human health. Wheat is a major food crop in most parts of the world, and high concentration of HMs in wheat grains poses serious health risks (Ivezić et al. 2013). Toxic concentrations of cadmium (Cd), nickel (Ni), chromium (Cr), lead (Pb), and zinc (Zn) in wheat flour may cause health disorders like kidney and liver failure. Treatment of seedlings with different S concentrations (0, 1, 5, and 10 mM) was observed to reduce MDA content, improved growth attributes, and increased total carbohydrate accumulation in wheat exposed to Cd toxicity (Gaafar et al. 2012). Khan et al. (2015) evaluated combined effects of S and Se to minimize Cd toxicity in wheat. A significant increase in Cd tolerance was correlated with GSH and proline synthesis due to reduced activity of proline oxidase (PROX) and high glutamyl kinase (GK) activity in leaf tissues. Pretreatment with NaHS (0, 0.4, 0.8, and 1.2 mM) was observed to improve PSII photochemistry, photochemical quenching, and antioxidative activities in wheat seedlings exposed to copper (Cu) stress (Dai et al. 2016). Environmental pollutants like nickel (Ni) interfere with uptake and distribution of mineral nutrients including S to reduce growth and quality of wheat. Matraszek et al. (2016) conducted a series of water culture experiments to evaluate the effects of Ni toxicity on macronutrient composition in wheat. They found that intensive S-SO42− nutrition partially improved biomass, enhanced ionic equilibrium, and increased nutrient accumulation in shoots of wheat plants exposed to Ni toxicity.
14.2.2 Maize
Deficiency of S has been more prevalent in maize in recent past due to reduced deposition of atmospheric S, use of crop residues, and no tillage. Low S supply results in yellowing of leaves including interveinal chlorosis. Application of poultry manure may somewhat supplement the soil with organic S however; it needs to undergo mineralization by soil microorganisms to make it available for plants. Microbial inoculation in combination with inorganic S fertilizers has also been found effective in maize. Combined effects of zinc sulfate (ZnSO4) and vesicular arbuscular mycorrhizal (AM) fungus on biomass accumulation and production of maize were investigated by Sajedi et al. (2010). The plants were applied with three different levels of ZnSO4 (0, 25, and 45 kg ha−1). The results showed that maize growth is not significantly affected by AM fungus under well-watered conditions. However, increasing level of ZnSO4 in combination with AM fungus significantly improved maize biomass and yield under water deficit conditions. They concluded that combined application of ZnSO4 (45 kg ha−1) and AM fungus might be utilized as an effective approach to improve growth and yield of maize. A recent report by Shahzad et al. (2017) showed that foliar application of K2SO4 reduces pre-anthesis abortion of maize kernel resulting in higher yield under drought stress conditions. In another study, Vazin (2012) found that foliar application of various ZnSO4 doses (0, 0.5%, 1%, and 1.5%) markedly improved yield attributes of maize under water deficit conditions.
High accretion of Na+ in the leaves is one of the main reasons for susceptibility of maize to salinity stress (Farooq et al. 2015). Accumulation of Na+ markedly reduces K content in symplast of maize leaves that disturbs stomatal undulations under saline conditions (Jafar et al. 2012). Exposure to salt stress induces high Cys production due to increased activity of O-acetylserine (thiol) lyase (OASTL) leading to better salt tolerance in plants (Fediuc et al. 2005). In a pot study, Riffat and Ahmad (2016a) found a significant effect of S application on biomass accumulation and nutrient content of maize seedlings exposed to salt stress. In another study, they observed a marked increase in germination attributes of different maize cultivars treated with K2SO4 under saline conditions (Riffat and Ahmad 2016b). High S supply may cause a reduction in maize yield under salinity stress as reported by Manesh et al. (2013). Maize plants were subjected to saline environment (water salinity 9.79 dSm−1 and soil salinity 9.3 dSm−1) and treated with various S doses, i.e., 0, 250, 500, 750, and 1000 kg ha−1. Exogenous S supply up to 750 kg ha−1 caused a linear increase in maize yield; however, higher S dose of 1000 kg ha−1 markedly reduced the yield. Shan et al. (2014) suggested that application of S sources like H2S helps to maintain the redox states of ascorbate and GSH to prevent electrolyte leakage that ultimately enhances salt tolerance in maize. Increased GSH content in roots of salt-stressed maize seedlings may be ascribed to high salt tolerance (AbdElgawad et al. 2016), which may also serve as a signal for ABA biosynthesis in shoot (Bittner et al. 2001). Maize seedlings exposed to S deficiency exhibited a marked increase in catalase (CAT) and superoxide dismutase (SOD) activities in leaf sheaths and blades due to increased ROS production under salinity stress (Chorianopoulou et al. 2012).
High concentration of toxic metals in agricultural soils has severely affected yield and nutritive value of maize. Availability of S plays a pivotal role in enhancing tolerance to metal toxicity through regulation of electron transport chain in Fe-S clusters and detoxification of HMs and xenobiotics (Hell and Hillebrand 2001). Efficacy of applied S doses may vary with the soil depth as both sulfate and elemental S exhibited a stronger effect at 0–40 cm soil layer compared to 40–80 cm horizon (Skwierawska et al. 2012). Applied S doses did not significantly affect Cu and Mn content, whereas they increased Cd and reduced Ni content in soils at a depth of 0–40 cm. Xu et al. (2008) observed toxic effects of high Zn content on pigments of maize seedlings. Supplementation of S in nutrient solution mitigated Zn toxicity and reduced the damage to young seedlings. In a similar study, Cui and Zhao (2011) reported positive effect of S application on antioxidative activities of CAT and SOD to alleviate Zn toxicity in maize seedlings. Contrarily, treatment with 50 mmol kg−1 S markedly reduced CAT and peroxidase (POX) activities in leaves of maize seedlings exposed to Cd toxicity suggesting that plant response to metal toxicity varies with intensity of stress, metal concentration, and plant species (Cui and Wang 2006).
14.2.3 Rice
Rice (Oryza sativa L.) is a staple food of nearly half of the world’s population; hence, it is imperative to overcome challenges limiting rice productivity worldwide. S deficiency rarely occurs in irrigated rice and usually affects vegetative stage. It is essential to evaluate the S requirement of rice crop since low S availability results in reduced protein synthesis and causes delayed plant development and maturity (Tsujimoto et al. 2017). In contrast, high S supply may result in reduced nutrient uptake and root respiration due to sulfide toxicity, particularly in low Fe soils. Rice is considered sensitive to drought stress due to presence of shallow root system, little circular wax, and rapid stomatal closure under water deficit conditions (Ji et al. 2012). Liu et al. (2017) evaluated the effect of NaHS on antioxidative mechanism of rice seedlings subjected to PEG-induced oxidative stress. Pretreatment with NaHS markedly enhanced germination rate and prevented the degradation of soluble protein content. In addition, it significantly reduced accumulation of H2O2 in seeds and helped to maintain cell membrane stability resulting in slow disintegration and senescence of seedlings.
Among cereals, rice is considered the most sensitive to salinity stress. Exposure to salt concentration of even less than 40 mM may cause a significant loss in rice growth and productivity (Munns and Tester 2008). Apoplastic uptake and distribution of Na+ cause toxicity which may be prevented by rapid downregulation of OsHKT2;1 in rice roots. An important GSH transporter in rice (OsGT1) is weakly expressed under normal conditions suggesting that it may play a key role in S transport under environmental stresses like salinity (Zhang et al. 2004; Srivalli and Khanna-Chopra 2008). S starvation reduced GSH level by 70% in rice seedlings that ultimately decreased PSII efficiency and the ability of PSI to photoreduce NADP+ by 31 and 61%, respectively. However, no significant effect of S deficiency was observed on metabolites of Calvin or tricarboxylic acid (TCA) cycle (Lunde et al. 2008). Ahmed et al. (2016) found that S fertilization markedly improved rice yield under saline conditions. A positive effect of S application was also noted on soil chemical properties such as such as SAR, pH, and ECe. Maintenance of Na+/K+ balance was observed to increase salt tolerance in rice seedlings treated with NaHS (Mostofa et al. 2015). Supplementation of this H2S donor also helped to maintain oxidative metabolism and mineral homeostasis by inhibiting Na+ uptake in the seedlings.
Consumption of rice contaminated with toxic metals may cause serious health hazards in humans. Low S availability affects oxidative thiol modifications, thereby increasing HM translocation from root to shoot (Leichert et al. 2008). In young rice seedlings, S application was reported to increase Fe and Mn accumulation in rhizosphere to reduce arsenic (As) toxicity (Wu et al. 2014). S availability positively influences thiol metabolism and glycolytic enzymes to promote amino acid accumulation in leaves of rice seedlings exposed to AS toxicity (Dixit et al. 2015). Fahad et al. (2015) found that application of ZnSO4 in combination with rubber ash alleviated Cd toxicity in different rice cultivars. Foliar application of ZnSO4 at panical initiation and milking stages significantly increased the number of spikelets and panicles as well as spikelet fertility to improve grain yield by 73% under salt stress conditions. In a similar study, Fan et al. (2010) observed significant reduction in Cd accumulation in brown rice by excessive S supply. Pretreatment of rice seedlings with NaHS was reported to reduce mercury (Hg) toxicity in rice seedlings exposed to HgCl2 (100 μM) for 3 days (Chen et al. 2017). The seedlings treated with NaHS exhibited increased expression of prominent thiol-containing compounds (OsMT-1 and NPT) to prevent Hg transport from root to shoot. Moreover, NaHS supplementation helped to scavenge or inhibit H2O2 and O2− (peroxy radicals) as well as CAT or SOD inhibitors (AT and DDC) even in the presence of Hg.
14.3 Role of S in Improving Abiotic Stress Tolerance in Legumes
14.3.1 Mung Bean
Pulses are part of healthy and balanced human diet and play an important role in preventing many acute diseases. They belong to family Leguminosae so they also increase soil C and N, reduce soil erosion, and help to control soil pathogens (Bagayoko et al. 2000; Sainju et al. 2005). Moreover, legumes positively influence growth of the following crops when grown in rotations with cereals. Hence, promoting legumes cultivation in developing countries can be an effective approach to reduce poverty and hunger in poor or developing countries (Abate et al. 2012).
Mung bean is an important pulse crop or food legume that is grown primarily for dry seeds and very occasionally used as a forage (Tomooka 2002). Although mung bean is considered tolerant to limited water supply, low water availability at reproductive and grain filling stages significantly reduces its yield and quality (Ahmad et al. 2015). Exposure to drought stress after 6 weeks of sowing can lead to decline in number of leaves, root nodules, dry matter, and plant height of mung bean (Ranawake et al. 2011). Reduction in plant production under drought stress is related to reduction in absorption, translocation, and redistribution of nutrients (Rouphael et al. 2012). Drought stress increases the concentration of sulfate compared to other ions like phosphate or nitrate, providing evidence that sulfate demand increases under limited water conditions (Ernst et al. 2010). Sulfate interacts with ABA and acts as a chemical signal to initiate stomatal closure in leaves under water deficit conditions. Usman et al. (2014) evaluated the role of ZnSO4 (10, 15, and 20 kg ha−1) on growth and yield of mung bean and reported a significant increase in growth and yield attributes. It was found that soil application of 20 kg ha−1 ZnSO4 resulted in maximum growth and yield attributes. However, soil applied ZnSO4 (15 kg ha−1) resulted in maximum plant height. In addition, protein content was found higher in plants supplemented with 10 kg ha−1 ZnSO4.
Recently, Jamal et al. (2018) reported that application of FeSO4 at 5 kg ha−1 in combination with ZnSO4 at 10 kg ha−1 significantly improved the biological yield, grain yield, straw yield, and nodule numbers in mung bean. Similarly, application of K2SO4 significantly improved yield and yield attributes in mung bean grown under arid climate (Abbas et al. 2011). Ali et al. (2014) suggested that foliar spray of iron sulfate (FeSO4) at branching and flowering stages can significantly increase the growth and yield attributes of mung bean. In addition, protein and iron contents of mung bean plants supplemented with S were also found higher compared to control plant, i.e., no S supply. Muniswamy et al. (2018) evaluated the role of foliar applied Zn (0 and 0.1%), N (15 and 25 kg ha−1), and S (0, 20, and 40 kg ha−1) on yield and quality of mung bean. They reported that application of 25 kg ha−1 N combined with 40 kg ha−1 S and 0.1% Zn (at both pre-flowering and pod initiation stages) markedly increased yield and quality of mung bean.
Salinity stress in mung bean leads to significant reduction in yield (Saha et al. 2010) due to poor germination and seedling growth (Promila and Kumar 2000; Misra and Dwivedi 2004). In a pot study, Pandey et al. (2010) found that exogenous ZnSO4 application may reduce the suppressing effects of salt stress on growth of mung bean seedlings. Similarly, Pandey et al. (2010) compared the effects of ZnSO4 seed treatment and foliar spray on mung bean under induced salt stress conditions. They observed that seed priming and foliar spray of ZnSO4 are effective strategies to alleviate the harmful effects of salt stress in mung bean.
Toxic metals negatively affect microbial population in the soil and may affect population size, diversity, and overall activity of soil microbiota (Kelly et al. 2003). Among various HMs, silver (Ag) and Pb were found to exert the most toxic effects on growth of soil microorganisms that delayed seed germination in mung bean (Ashraf and Ali 2015). They found that application of ZnSO4 positively influenced microbial population and helped to increase germination percentage under Ag or Pb toxicity. Positive effects of S supply on microbial population in a mung bean field were also reported by Bahadur and Tiwari (2014). They observed that application of 15 kg ha−1 S increased Rhizobium and Azotobacter population that ultimately increased grain yield in mung bean. Recent reports of Islam et al. (2017) indicate that S supply in combination with boron (B) was more effective than individual application of these nutrients to improve grain yield in mung bean.
14.3.2 Chickpea
Seed proteins in chickpea, like other legumes, are deficient in S-containing compounds such as Cys, Met, and amino acids (Chiaiese et al. 2004). Exogenous S supply significantly improves accumulation of S-enriched seed proteins in legumes including chickpea (Sexton et al. 1998). In a controlled study, Chiaiese et al. (2004) found that addition of a transgene, sunflower seed albumin (SSA), encoding Cys and Met reduced accumulation of S-containing amino acids in chickpea seeds. Excess S supply downregulated SSA expression and improved seed protein composition in chickpea. S deficiency also leads to flower abortion and infertility of pollen and ovule resulting in low yield in chickpea. Combined application of K2SO4 and calcium ammonium nitrate (CAN) was observed to ameliorate negative effects of drought stress on yield attributes of chickpea (Mahmood et al. 2017). Application of 1.5% K2SO4 + 2 and 3% CAN resulted in maximum increase in yield and yield attributes when applied at 40 and 60 days after sowing.
Chickpea is considered sensitive to salinity as exposure to salt stress results in poor germination and seedling growth (Zawude and Shanko 2017). Seyedi (2011) conducted a laboratory experiment to evaluate the role of S supply in improving salinity tolerance in chickpea. They observed that chickpea seeds primed with ZnSO4 exhibited enhanced germination and seedling establishment under saline conditions. In a similar study, Nautiyal and Shukla (2013) found that S supplementation improved seedling dry mass, seed Zn content, as well as Zn uptake and translocation in salt-stressed chickpea plants raised from ZnSO4-primed seeds. Positive effects of S application were found related with increased carbonic anhydrase and SOD activity in ZnSO4-treated seeds. Islam (2012) conducted a field trial to evaluate the comparative effects of two different S sources, viz., gypsum and (NH4)2SO4, on yield of chickpea. They found that exogenous S supply improved yield by 17%; however, (NH4)2SO4 was found more effective than gypsum regarding increment in nutrient uptake and yield.
14.3.3 Black Gram
Pulse crops, viz., black gram, green gram, cowpea, and horse gram, exhibited increased yield when supplemented with foliar spray of nutrient solution containing 2% DAP, 1% KCl, 1% boron, 1% MgSO4, and 1% ZnSO4 at 30 and 45 DAS of pulse crop (Maheswari and Karthik 2017). Patel et al. (2018) evaluated the effects of different S sources, viz., elemental S, gypsum, and ammonium sulfate, and S levels, viz., 0, 20, 40, and 60 kg S ha−1, on growth and yield of black gram. Among different sources, gypsum was found superior in terms of growth and yield. Among various levels, 40 kg ha−1 gypsum was found more superior compared to others. Srivastava and Shukla (2016) reported an adverse effect of arsenic (As), cadmium (Cd), cobalt (Co), chromium (Cr), and lead (Pb) on growth of black gram. They found that heavy metal toxicity followed the order As > Cr > Cd > Co > Pb in terms of damage caused to black gram plants. Plants grown in As toxic soils failed to survive and wilted within 1 week. A significant loss of pigments and proteins was noted in plants exposed to Cr and Co toxicity compared to control plants. Antioxidant enzymes’ activity was found higher in plants grown in Cr and Co toxic soils compared to Cd and Pb plants. It was concluded that black gram is highly sensitive to As followed by Cr and Pb was found least toxic for growth.
14.4 Role of S in Improving Abiotic Stress Tolerance in Oilseeds
14.4.1 Sunflower
Sunflower seeds are used to make high-quality vegetable oil (Okoko et al. 2008). Its yield depends upon the availability of water at different growth stages, and flowering stage is considered as most critical stage regarding water availability. It was reported that application of S at the rate of 25 kg ha−1 significantly improved the plant height, head diameter, no. of seeds per achene, biological yield, and achene yield of sunflower plants (Poonia 2000). Wani et al. (2001) suggested that increasing S doses might significantly improve seed yield and quality of sunflower seeds. Recently, Ullah et al. (2019) found a significant increase in sunflower yield by applying S in combination with K. The maximum grain yield and oil quality were recorded in plants supplemented with 60 kg ha−1 S + 90 kg ha−1 K. Similarly, S fertilization combined with exogenous Zn supply alleviated the adverse effects of drought stress at different growth stages of sunflower (Zafar et al. 2014). Application of S using gypsum at the rate of 60 kg ha−1 not only increased the plant height, leaf area index, and dry matter of sunflower but also improved the pH of the soil (Usha Rani et al. 2009). Cheema et al. (2014) investigated the effects of various ZnSO4 concentrations (15, 30, 45, 60, and 75 kg ha−1) on sunflower. They reported that plants supplemented with 45 kg ha−1 ZnSO4 resulted in maximum stem diameter, number of achenes per head, achene yield, head diameter, and biological yield. Moreover, harvest index and 1000-achene weight were found maximum in plants supplemented with 60 kg ha−1 ZnSO4. A study involving exposure of sunflower to drought stress at vegetative and reproductive stages showed that foliar application of 1% ZnSO4 significantly ameliorated water stress to improve achene yield in sunflower (Shahri et al. 2012). Likewise, foliar application of FeSO4 was found helpful in improving growth and yield of sunflower under normal and drought stress conditions (Ebrahimian and Bybordi 2011).
Nano-particles of FeSO4 were found to significantly reduce salt-induced oxidative stress in sunflower (Torabian et al. 2017). S supplementation markedly reduced Na+ content and increased net CO2 assimilation rate, leaf area, shoot dry weight, chlorophyll content, sub-stomatal CO2 concentration, and Fe content to improve salinity tolerance in sunflower.
14.4.2 Brassica
In general, Brassica species require S at vegetative stage to synthesize essential proteins for improved oil quality (Blake-Kalff et al. 2001). It is estimated that production of 1 ton rape seed requires about 16 kg S compared to wheat seed that only requires 2–3 kg S (Lee et al. 2016). Moreover, these species contain high amount (20%) of organic S in the form of S-containing metabolites like glucosinolate (Aghajanzadeh et al. 2014). Deficiency of S significantly reduces the incorporation of total amount of S into proteins by 62% (Lee et al. 2013). A study involving two contrasting Brassica napus species, viz., Mosa and Saturnin, showed that genotype possessing better sulfur use efficiency exhibited higher tolerance to drought stress (Lee et al. 2014). Dhruw et al. (2017) found that application of S using ZnSO4 (40 kg ha−1) along with basal dose of N-P-K @ 120:60:40 can significantly improve growth, yield, and oil contents of B. juncea plants. A field study by Mishra et al. (2010) showed that S fertilization in combination with P resulted in maximum mustard oil production. Khan et al. (2016) compared the effects of ZnSO4 and manganese sulfate (MnSO4) in improving drought tolerance in B. juncea. They reported a significant increase in growth attributes, biochemical aspects (chlorophyll and carotenoids), and relative water content of B. juncea treated with S under drought stress.
Salt stress tolerance in Brassica species is related to modulation in several physiological and biochemical processes (Ashraf and McNeilly 2004). Siddiqui et al. (2012) reported that exogenous S supply in combination with N can alleviate the negative effects of salt stress in B. juncea. It was observed that decrease in nitrate reductase activity, N content, growth, and photosynthetic activity under salinity was restored with soil application of N and S at the rate of 100 mg kg−1 soil. Moreover, combined application of N and S resulted in better growth and proline accumulation compared to individual application of these nutrients.
Exposure to HMs not only poses serious threat to productivity of food crops but also threatens the safety of human nutrition. S nutrition helps to reduce toxic effects of metal elements in Brassica species. For example, Bashir et al. (2015) suggested that S pool is needed to synthesize GSH, PCs, and non-protein thiols to alleviate Cd toxicity in B juncea. They found that S-deficient plants exhibited higher oxidative activity under Cd stress. Contrarily, S application markedly enhanced CAT, APX, and GR activities to alleviate Cd-induced oxidative stress in B. juncea. Similarly, Zhong et al. (2012) found that S-treated B. napus plants exhibited lower As content in roots and grain compared to S-deficient plants. They were of the view phytoremediation capacity of rapeseed could be increased by S fertilization.
14.4.3 Soybean
The production of soybean is threatened by climatic change with more frequent occurrence of drought stress around the globe (Dai 2013; Foyer et al. 2016). Studies conducted under field and controlled conditions have shown that water stress can cause 25–50% yield reductions in soybeans (Frederick et al. 2001; Sadeghipour and Abbasi 2012). It has been well documented that S nutrition plays a significant role against drought stress in crop plants. Its metabolite GSH has been reported to scavenge reactive oxygen species (Astolfi et al. 2012). Exogenous H2S application increased the biomass and survival of soybean plants grown under terminal drought stress (Zhang et al. 2010). Water deficit conditions drastically decreased the chlorophyll contents and antioxidant activity of soybean seedlings. Foliar application of H2S significantly improved the antioxidant activity and chlorophyll contents of seedlings. Moreover, delay in the accumulation of hydrogen peroxide, superoxide anion, and malondialdehyde was also observed in plants sprayed with H2S compared to control. Cigelske (2017) investigated the combined effects of N and S on soybean. It was reported that application of N significantly improved the vigor and yield but decreased the nodulation in soybean plants. However, S application decreased protein content but significantly increased nodulation in rhizosphere. Seed priming with sole and mixed solutions of ZnSO4 and FeSO4 alleviated the detrimental effects of drought stress in soybean at reproductive stage (Dehnavi and Sheshbahre 2017). They found a significant increment in photosynthetic rate, transpiration rate, stomatal conductance, and photosynthetic characteristics of plants raised from nutrient-primed seeds compared to hydropriming. Similarly, Heidarzade et al. (2016) reported that FeSO4 application in combination with molybdenum markedly enhanced the yield attributes of soybean under drought stress. Comparative effects of KCl and K2SO4 application on physiological activities of soybean seedlings exposed to salt stress were investigated by Adhikari et al. (2019). They found that application of K2SO4 at 2.5% was more effective than KCl to improve growth, flavonoids, total phenols, antioxidant activity, carotenoids, and chlorophyll contents of salt-stressed soybean seedlings.
14.5 Conclusion
Considering the importance of S-containing metabolites in improving abiotic stress tolerance in plants, the application of S-containing fertilizers has become imperative to improve yield and quality of food crops in recent past. Low S availability severely hampers uptake, distribution, and assimilation of plant nutrients such as N, P, K, Zn, B, and Ca. Moreover, plants grown in S-deficient soils exhibit high susceptibility to environmental stresses like drought, salinity, and metal ions. Despite extensive research on S metabolism and assimilation in crop plants in recent years, there are still many questions answered. Future studies focused on increasing our understanding about specific gene expression in response to S application would help us to develop crop species tolerant to a wide array of environmental extremities. Studies triggered to boost our knowledge about interactive pathways of S assimilation and phytohormones regulation are of particular importance in this regard.
Abbreviations
- (NH4)2SO4:
-
Ammonium sulfate
- γGCS:
-
Gamma-glutamylcysteine synthetase
- ABA:
-
Abscisic acid
- ABA:
-
Abscisic acid
- AM:
-
Arbuscular mycorrhizal
- APX:
-
Ascorbate peroxidase
- As:
-
Arsenic
- CAN:
-
Calcium ammonium nitrate
- CaSO4:
-
Calcium sulfate
- CAT:
-
Catalase
- Cd:
-
Cadmium
- Cr:
-
Chromium
- Cys:
-
Cysteine
- DHAR:
-
Dehydroascorbate reductase
- FeSO4:
-
Iron sulfate
- GPX:
-
Glutathione peroxidase
- GR:
-
Glutathione reductase
- GSH:
-
Reduced glutathione
- GST:
-
Glutathione S-transferases
- H2S:
-
Hydrogen sulfide
- HMs:
-
Heavy metals
- K2SO4:
-
Potassium sulfate
- Met:
-
Methionine
- NaHS:
-
Sodium hydrogen sulfide
- Ni:
-
Nickel
- PCs:
-
Phytochelatins
- ROS:
-
Reactive oxygen species
- S:
-
Sulfur
- SOD:
-
Superoxide dismutase
- Trx:
-
Thioredoxins
- Vit:
-
Vitamins
- Zn:
-
Zinc
- ZnSO4:
-
Zinc sulfate
References
Abate T, Alene AD, Bergvinson D, Shiferaw B, Silim S, Orr A, Asfaw S (2012) Tropical grain legumes in Africa and south Asia: knowledge and opportunities. Int J Multidiscip Adv Res 10:1–12
Abbas G, Aslam M, Malik AU, Abbas Z, Ali M, Hussain F (2011) Potassium sulfate effects on growth and yield of mungbean (Vigna radiata L.) under arid climate. Int J Agric Appl Sci 2:72–75
AbdElgawad FK, Khalil WK, El-Kady AA, Waly AI, Abdel-Wahhab MA (2016) Carboxymethyl chitosan modulates the genotoxic risk and oxidative stress of perfluorooctanoic acid in Nile tilapia (Oreochromis niloticus). J Saudi Soc Agric Sci 15:57–66
Adhikari B, Dhungana SK, Kim ID, Shin DH (2019) Effect of foliar application of potassium fertilizers on soybean plants under salinity stress. J Saudi Soc Agric Sci. In Press
Aghajanzadeh T, Hawkesford MJ, De Kok LJ (2014) The significance of glucosinolates for sulfur storage in Brassicaceae seedlings. Front Plant Sci 5:1–10. https://doi.org/10.3389/fpls.2014.00704
Ahmad A, Selim MM, Alderfasi AA, Afzal M (2015) Effect of drought stress on mung bean (Vigna radiata L.) under arid climatic conditions of Saudi Arabia. In: Miralles i Garcia JL, Brebbia CA (eds) Ecosystem and sustainable development. WIT Press, Southampton, pp 185–193
Ahmad N, Malagoli M, Wirtz M, Hell R (2016a) Drought stress in maize causes differential acclimation responses of glutathione and sulfur metabolism in leaves and roots. BMC Plant Biol 16:247–262
Ahmad R, Dawar K, Iqbal J, Wahab S (2016b) Effect of sulfur on nitrogen use efficiency and yield of maize crop. Adv Environ Biol 10:85–91
Ahmed K, Qadir G, Jami AR, Saqib AI, Nawaz MQ, Kamal MA, Haq E (2016) Strategies for soil amelioration using sulphur in salt affected soils. Cer Agron Mold 49:5–16
Ali A, Arshadullah M, Hyder SI, Mahmood IA (2012) Effect of different levels of sulfur on the productivity of wheat in a saline sodic soil. Soil Environ 31:91–95
Ali B, Ali A, Tahir M, Ali S (2014) Growth, Seed yield and quality of mungbean as influenced by foliar application of iron sulfate. Pak J Life Soc Sci 12:20–25
Anjum NA, Gill R, Kaushik M, Hasanuzzaman M, Pereira E, Ahmad I, Tuteja N, Gill SS (2015) ATP-sulfurylase, sulfur-compounds, and plant stress tolerance. Front Plant Sci 6:1–9
Aroca A, Gotor C, Romero LC (2018) Hydrogen sulfide signaling in plants: Emerging roles of protein persulfidation. Front Plant Sci 9:1–8
Arshadullah M, Hyder SI, Arshad A, Mahmood IA (2013) Cumulative effect of sulfur and calcium on wheat growth and yield under saline-sodic soils. Pak J Agric Res 26:46–53
Ashraf R, Ali TA (2015) Effect of heavy metals on soil microbial community and mung beans seed germination. Pak J Bot 39:629–636
Ashraf M, McNeilly T (2004) Salinity tolerance in brassica oilseeds. Crit Rev Plant Sci 23:157–174
Astolfi S, Zuchi S, Neumann G, Cesco S, Sanita di Toppi L, Pinton R (2012) Response of barley plants to Fe deficiency and Cd contamination as affected by S starvation. J Exp Bot 63:1241–1250
Bagayoko M, Buerkert A, Lung G, Bationo A, Römheld V (2000) Cereal/legume rotation effects on cereal growth in Sudano-Sahelian West Africa: soil mineral nitrogen, mycorrhizae and nematodes. Plant and Soil 218:103–116
Bahadur L, Tiwari DD (2014) Nutrient management in mung bean (Vigna radiata L.) through sulphur and biofertilizers. Legum Res 37:180–187
Bashir H, Ibrahim MM, Bagheri R, Ahmad J, Arif IA, Baig MA, Qureshi MI (2015) Influence of sulfur and cadmium on antioxidants, phytochelatins and growth in Indian mustard. AoB Plants 7:1–13
Bittner F, Oreb M, Mendel RR (2001) ABA3 is a molybdenum cofactor sulfurase required for activation of aldehyde oxidase and xanthine dehydrogenase in Arabidopsis thaliana. J Biol Chem 276:40381–40384
Blake-Kalff M, Zhao J, Hawkesford M, McGrath S (2001) Using plant analysis to predict yield losses by sulphur deficiency. Ann Appl Biol 138:123–127. https://doi.org/10.1111/j.1744-7348.2001.tb00093.x
Cheema MA, Wahid MA, Ghaffar A, Sattar A, Abbas S (2014) Yield response of autumn planted sunflower hybrids to zinc sulfate application. J Agric Res 52:523–533
Chen Z, Chen M, Jiang M (2017) Hydrogen sulfide alleviates mercury toxicity by sequestering it in roots or regulating reactive oxygen species productions in rice seedlings. Plant Physiol Biochem 111:179–192
Chiaiese P, Ohkama-Ohtsu N, Molvig L, Godfree R, Dove H, Hocart C, Fujiwara T, Higgins TJV, Tabe LM (2004) Sulphur and nitrogen nutrition influence the response of chickpea seeds to an added, transgenic sink for organic sulphur. J Exp Bot 55:1889–1901
Chorianopoulou SN, Giamouroglou M, Bouranis DL (2012) Differential early fluctuations in superoxide dismutase and catalase activities are included in the responses of young maize organs to s-deprivation. Am J Plant Sci 3:338–345
Cigelske B (2017) Soybean response to nitrogen and sulfur fertilization. Doctoral dissertation, North Dakota State University
Cui Y, Wang Q (2006) Physiological responses of maize to elemental sulphur and cadmium stress. Plant Soil Environ 52:523–529
Cui Y, Zhao N (2011) Oxidative stress and change in plant metabolism of maize (Zea mays L.) growing in contaminated soil with elemental sulfur and toxic effect of zinc. Plant Soil Environ 57:34–39
Dai A (2013) Increasing drought under global warming in observations and models. Nat Clim Change 3:52–58. https://doi.org/10.1038/nclimate1811
Dai H, Xu Y, Zhao L, Shan C (2016) Alleviation of copper toxicity on chloroplast antioxidant capacity and photosystem II photochemistry of wheat by hydrogen sulfide. Braz J Bot 39:787–793
Dehnavi MM, Sheshbahre MJ (2017) Soybean leaf physiological responses to drought stress improved via enhanced seed zinc and iron concentrations. J Plant Proc Funct 5:13–21
Dhruw SS, Swaroop N, Swamy A, Upadhayay Y (2017) Effects of different levels of NPK and sulphur on growth and yield attributes of Mustard (Brassica juncea L.) Cv. Varuna. Int J Cur Microbiol Appl Sci 6:1089–1098
Dixit G, Singh AP, Kumar A, Singh PK, Kumar S, Dwivedi S, Trivedi PK, Pandey V, Norton GJ, Dhankher OP, Tripathi RD (2015) Sulfur mediated reduction of arsenic toxicity involves efficient thiol metabolism and the antioxidant defense system in rice. J Hazard Mater 298:241–251
Ebrahimian E, Bybordi A (2011) Effect of iron foliar fertilization on growth, seed and oil yield of sunflower grown under different irrigation regimes. Middle East J Sci Res 9:621–627
Ernst L, Goodger JQ, Alvarez S, Marsh EL, Berla B, Lockhart E, Jung J, Li P, Bohnert HJ, Schachtman DP (2010) Sulphate as a xylem-borne chemical signal precedes the expression of ABA biosynthetic genes in maize roots. J Exp Bot 61:3395–3405
Fahad S, Hussain S, Khan F, Wu C, Saud S, Hassan S, Ahmad N, Gang D, Ullah A, Huang J (2015) Effects of tire rubber ash and zinc sulfate on crop productivity and cadmium accumulation in five rice cultivars under field conditions. Environ Sci Pollut Res 22:12424–12434
Fan JL, Hu ZY, Ziadi N, Xia X (2010) Excessive sulfur supply reduces cadmium accumulation in brown rice (Oryza sativa L.). Environ Pollut 158:409–415
Farooq M, Hussain M, Wakeel A, Siddique KH (2015) Salt stress in maize: effects, resistance mechanisms, and management. A review. Agron Sustain Dev 35:461–481
Fediuc E, Lips SH, Erdei L (2005) O-acetylserine (thiol) lyase activity in Phragmites and Typha plants under cadmium and NaCl stress conditions and the involvement of ABA in the stress response. J Plant Physiol 162:865–872
Foyer CH, Lam HM, Nguyen HT, Siddique KHM, Varshney R et al (2016) Neglecting legumes has compromised global food and nutritional security. Nat Plant 2:16112. https://doi.org/10.1038/nplants.2016.112
Frederick JR, Camp CR, Bauer PJ (2001) Drought-stress effects on branch and mainstem seed yield and yield components of determinate soybean. Crop Sci 41:759–763
Gaafar ARZ, Ghdan AA, Siddiqui MH, Al-Whaibi MH, Basalah MO, Ali HM, Sakran AM (2012) Influence of sulfur on cadmium (Cd) stress tolerance in Triticum aestivum L. Afr J Biotechnol 11:10108–10114
Gill SS, Anjum NA, Hasanuzzaman M, Gill R, Trivedi DK, Ahmad I, Pereira E, Tuteja N (2013) Glutathione and glutathione reductase: a boon in disguise for plant abiotic stress defense operations. Plant Physiol Biochem 70:204–212
Hasanuzzaman M, Oku H, Nahar K, Bhuyan MB, Al Mahmud J, Baluska F, Fujita M (2018) Nitric oxide-induced salt stress tolerance in plants: ROS metabolism, signaling, and molecular interactions. Plant Biotech Rep 12:77–92
Heidarzade A, Esmaeili M, Bahmanyar M, Abbasi R (2016) Response of soybean (Glycine max) to molybdenum and iron spray under well-watered and water deficit conditions. J Exp Biol Agric Sci 4:37–46
Hell R, Hillebrand H (2001) Plant concepts for mineral acquisition and assimilation. Curr Opin Biotechnol 12:161–168
Herrmann J, Ravilious GE, McKinney SE, Westfall CS, Lee SG, Baraniecka P, Giovannetti M, Kopriva S, Krishnan HB, Jez JM (2014) Structure and mechanism of soybean ATP sulfurylase and the committed step in plant sulfur assimilation. J Biol Chem 289:10919–10929
Honsel A, Kojima M, Haas R, Frank W, Sakakibara H, Herschbach C, Rennenberg H (2011) Sulphur limitation and early sulphur deficiency responses in poplar: significance of gene expression, metabolites, and plant hormones. J Exp Bot 63:1873–1893
Hu ZY, Zhu YG, Li M, Zhang LG, Cao ZH, Smith FA (2007) Sulfur (S)-induced enhancement of iron plaque formation in the rhizosphere reduces arsenic accumulation in rice (Oryza sativa L.) seedlings. Environ Pollut 147:387–393
Islam M (2012) The effect of different rates and forms of sulfur on seed yield and micronutrient uptake by chickpea. Plant Soil Environ 58:399–404
Islam MS, Sabagh EL, Hasana A, Akhter K, Hasan M, Barutçulard C (2017) Growth and yield response of mung bean (Vigna radiata L.) as influenced by sulphur and boron application. Sci. J Crop Sci 6:153–160
Ivezić V, Lončarić Z, Engler M, Kerovec D, Singh BR (2013) Comparison of different extraction methods representing available and total concentrations of Cd, Cu, Fe, Mn and Zn in soil. Poljoprivreda 19:53–58
Jafar MZ, Farooq M, Cheema MA, Afzal I, Basra SMA, Wahid MA, Aziz T, Shahid M (2012) Improving the performance of wheat by seed priming under saline conditions. J Agron Crop Sci 198:38–45
Jamal A, Ismail MK, Tariq M, Fawad M (2018) Response of Mung bean crop to different levels of applied iron and zinc. J Hortic Plant Res 3:13–22
Ji K, Wang Y, Sun W, Lou Q, Mei H, Shen S, Chen H (2012) Drought-responsive mechanisms in rice genotypes with contrasting drought tolerance during reproductive stage. J Plant Physiol 169:336–344
Kausar A, Ashraf MY, Gull M, Ghafoor R, Ilyas M, Zafar S, Niaz M, Akhtar N, Kanwal H, Iqbal N, Aftab K (2016) Alleviation of salt stress by K2SO4 in two wheat (Triticum aestivum L.) cultivars. Appl Ecol Environ Res 14:137–147
Kelly JJ, Häggblom MM, Tate RL (2003) Effects of heavy metal contamination and remediation on soil microbial communities in the vicinity of a zinc smelter as indicated by analysis of microbial community phospholipid fatty acid profiles. Biol Fertil Soils 38:65–71
Khalid S, Afridi MZ, Munsif F, Imranuddin I, Ullah I, Nadia U (2018) Effect of sulphur foliar application on yield and yield components of Brassica napus L. Int J Agric Environ Res 2:232–236
Khan MI, Asgher M, Iqbal N, Khan NA (2013) Potentiality of sulphur-containing compounds in salt stress tolerance. In: Ecophysiology and responses of plants under salt stress. Springer, New York, pp 443–472
Khan NA, Khan MIR, Asgher M, Fatma M, Masood A, Syeed S (2014) Salinity tolerance in plants: revisiting the role of sulfur metabolites. J Plant. Biochem Physiol 2:1–8
Khan MIR, Nazir F, Asgher M, Per TS, Khan NA (2015) Selenium and sulfur influence ethylene formation and alleviate cadmium-induced oxidative stress by improving proline and glutathione production in wheat. J Plant Physiol 173:9–18
Khan R, Gul S, Hamayun M, Shah M, Sayyed A, Ismail H, Gul H (2016) Effect of foliar application of zinc and manganese on growth and some biochemical constituents of Brassica juncea grown under water stress. Am Eur J Agric Environ Sci 16:984–997
Lee BR, Muneer S, Kim KY, Avice JC, Ourry A, Kim TH (2013) S-deficiency responsive accumulation of amino acids is mainly due to hydrolysis of the previously synthesized proteins – not to de novo synthesis in Brassica napus. Physiol Plant 147:369–380. https://doi.org/10.1111/j.1399-3054.2012.01669.x
Lee BR, Muneer S, Jung WJ, Avice JC, Ourry A, Kim TH (2014) Partitioning of newly absorbed and previously stored nitrogen and sulphur under sulphate deficient nutrition. J Plant Nutr 37:1702–1716. https://doi.org/10.1080/01904167.2014.889148
Lee BR, Zaman R, Avice JC, Ourry A, Kim TH (2016) Sulfur use efficiency is a significant determinant of drought stress tolerance in relation to photosynthetic activity in Brassica napus cultivars. Front Plant Sci 7:1–11
Leichert LI, Gehrke F, Gudiseva HV, Blackwell T, Ilbert M, Walker AK, Strahler JR, Andrews PC, Jakob U (2008) Quantifying changes in the thiol redox proteome upon oxidative stress in vivo. Proc Natl Acad Sci 105:8197–8202
Liu J, Zhang H, Yin Y, Chen H (2017) Effects of exogenous hydrogen sulfide on antioxidant metabolism of rice seed germinated under drought stress. J South Agric 48:31–37
Lunde C, Zygadlo A, Simonsen HT, Nielsen PL, Blennow A, Haldrup A (2008) Sulfur starvation in rice: the effect on photosynthesis, carbohydrate metabolism, and oxidative stress protective pathways. Physiol Plant 134:508–521
Ma D, Ding H, Wang C, Qin H, Han Q, Hou J, Lu H, Xie Y, Guo T (2016) Alleviation of drought stress by hydrogen sulfide is partially related to the abscisic acid signaling pathway in wheat. PLoS One 11:1–16
Maheswari UM, Karthik A (2017) Effect of foliar nutrition on growth, yield attributes and seed yield of pulse crops. Adv Crop Sci Technol 5:1–3
Mahmood I, Razzaq A, Qayyum A, Ali Khan A (2017) Mitigating the terminal drought stress in chickpea (Cicer Arietinum L.) through exogenous application of nutrients. J Agric Res 55:291–302
Manesh AK, Armin M, Moeini MJ (2013) The effect of sulfur application on yield and yield components of corn in two different planting methods in saline conditions. Int J Agron Plant Prod 4:1474–1478
Matraszek R, Hawrylak-Nowak B, Chwil S, Chwil M (2016) Macronutrient composition of nickel-treated wheat under different sulfur concentrations in the nutrient solution. Environ Sci Pollut Res 23:5902–5914
Mishra SV, Deepak M, Gupta G (2010) Effect of phosphorus and sulphur and their interaction on mustard crop. Asian Sci 5:79–84
Misra N, Dwivedi UN (2004) Genotypic difference in salinity tolerance of green gram cultivars. Plant Sci 166:1135–1142. https://doi.org/10.1016/j.plantsci.2003.11.028
Mostofa MG, Saegusa D, Fujita M, Tran LSP (2015) Hydrogen sulfide regulates salt tolerance in rice by maintaining Na+/K+ balance, mineral homeostasis and oxidative metabolism under excessive salt stress. Front Plant Sci 6:1055
Muniswamy RS, Singh V, Mithare P (2018) Response of nitrogen, Sulphur and foliar application of zinc on yield and quality of greengram (Vigna radiata L.). J Pharma Phytochem 7:517–522
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681
Nautiyal N, Shukla K (2013) Evaluation of seed priming zinc treatments in chickpea for seedling establishment under zinc deficient conditions. J Plant Nutr 36:251–258
Noctor G, Mhamdi A, Chaouch S, Han YI, Neukermans J, Marquez-Garcia B, Queval G, Foyer CH (2012) Glutathione in plants: an integrated overview. Plant Cell Environ 35:454–484
Okoko NEK, Mahasi MJ, Kidula N, Ojowi M, Makini F (2008) Participatory sunflower production, technology dissemination and value addition in Southwest Kenya. Afr J Agric Res 3:396–399
Pandey SK, Bahuguna RN, Madan P, Trivedi AK, Hemantaranjan A, Srivastava JP (2010) Effects of pre-treatment and foliar application of zinc on growth and yield components of mungbean (Vigna radiata L.) under induced salinity. Indian J Plant Physiol 15:164–167
Pathak GC, Gupta B, Pandey N (2012) Improving reproductive efficiency of chickpea by foliar application of zinc. Brazilian J Plant Physiol 24:173–180
Patel AK, Nath T, Prajapati A, Singh VK, Pandey SK (2018) Effect of doses and sources of sulphur on growth and yield of black gram (Vigna mungo L. Hepper) under rainfed condition of Vindhyan Soil. J Pharmacog Phytochem 1:91–94
Poonia KL (2000) Effect of planting geometry, nitrogen and sulfur on growth and yield of sunflower (Helianthus annuus L.). J Eco Physiol 3:59–71
Promila K, Kumar S (2000) Vigna radiata seed germination under salinity. Biol Plant 43:423–426. https://doi.org/10.1093/pcp/pcs/040
Prosser IM, Purves JV, Saker LR, Clarkson DT (2001) Rapid disruption of nitrogen metabolism and nitrate transport in spinach plants deprived of sulphate. J Exp Bot 52:113–121
Ranawake AL, Dahanayaka N, Amarasingha UGS, Rodrigo WDRJ, Rodrigo UTD (2011) Effect of water stress on growth and yield of mung bean (Vigna radiata L). Trop Agric Res Ext 14:76–79
Rasheed M, Ali H, Mahmood T (2004) Impact of nitrogen and sulfur application on growth and yield of maize (Zea mays L.) crop. J Res Sci 15:153–157
Rehman UH, Iqbal Q, Farooq M, Wahid A, Afzal I, Basra SM (2013) Sulphur application improves the growth, seed yield and oil quality of canola. Acta Physiol Plant 35:2999–3006
Riffat A, Ahmad MS (2016a) Amelioration of adverse effects of salt stress on maize (Zea mays L.) cultivars by exogenous application of sulfur at seedling stage. Pak J Bot 48:1323–1334
Riffat A, Ahmad MS (2016b) Amelioration of adverse effects of salt stress on maize (Zea mays L.) cultivars by exogenous application of sulfur at seedling stage. Pak J Bot 48:1323–1334
Rouphael Y, Cardarelli M, Schwarz D, Franken P, Colla G (2012) Effects of drought on nutrient uptake and assimilation in vegetable crops. In: Aroca R (ed) Plant responses to drought stress. Springer, Berlin, pp 171–195
Royo A, Abió D (2003) Salt tolerance in durum wheat cultivars. Spanish J Agric Res 1:27–35
Sadeghipour O, Abbasi S (2012) Soybean response to drought and seed inoculation. World Appl Sci J 17:55–60
Saha P, Chatterjee P, Biswas AK (2010) NaCl pretreatment alleviates salt stress by enhancement of antioxidant defense and osmolyte accumulation in mungbean (Vigna radiata L. Wilczek). Indian J Exp Biol 48:593–600
Sainju UM, Whitehead WF, Singh BP (2005) Biculture legume–cereal cover crops for enhanced biomass yield and carbon and nitrogen. Agron J 97:1403–1412
Sajedi NA, Ardakani MR, Rejali F, Mohabbati F, Miransari M (2010) Yield and yield components of hybrid corn (Zea mays L.) as affected by mycorrhizal symbiosis and zinc sulfate under drought stress. Physiol Mol Biol Plants 16:343–351
Sauter M, Moffatt B, Saechao MC, Hell R, Wirtz M (2013) Methionine salvage and S-adenosylmethionine: essential links between sulfur, ethylene and polyamine biosynthesis. Biochem J 1:145–154
Sexton PJ, Naeve SL, Paek NC, Shibles R (1998) Sulphur availability, cotyledon nitrogen:sulphur ratio, and relative abundance of seed storage proteins of soybean. Crop Sci 38:983–986
Seyedi M (2011) The effect of seed priming with zinc sulphate on germination characteristics and seedling growth of chickpea (Cicer arietinum L.) under salinity stress. Euphrates J Agric Sci 3:205–210
Shah S, Hussain M, Jalal A, Khan MS, Shah T, Ilyas M, Uzair M (2018) Nitrogen and sulfur rates and timing effects on phenology, biomass yield and economics of wheat. Sarhad J Agric 34:671–679
Shahri ZB, Zamani GR, Sayyari-Zahan MH (2012) Effect of drought stress and zinc sulfate on the yield and some physiological characteristics of sunflower (Helianthus. annuus L.). Adv Environ Biol 6:518–525
Shahzad AN, Fatima A, Sarwar N, Bashir S, Rizwan M, Qayyum MF, Qureshi MK, Javaid MH, Ahmad S (2017) Foliar application of potassium sulfate partially alleviates pre-anthesis drought-induced kernel abortion in maize. Int J Agric Biol 19:495–501
Shan CJ, Zhang SL, Li DF, Zhao YZ, Tian XL, Zhao XL, Wu YX, Wei XY, Liu RQ (2011) Effects of exogenous hydrogen sulfide on the ascorbate and glutathione metabolism in wheat seedlings leaves under water stress. Acta Physiol Plant 33:2533
Shan C, Liu H, Zhao L, Wang X (2014) Effects of exogenous hydrogen sulfide on the redox states of ascorbate and glutathione in maize leaves under salt stress. Biol Plant 58:169–173
Shao HB, Chu LY, Shao MA, Jaleel CA, Hong-mei M (2008) Higher plant antioxidants and redox signaling under environmental stresses. C R Biol 331:433–441
Siddiqui MH, Mohammad F, Khan MM, Al-Whaibi MH (2012) Cumulative effect of nitrogen and sulphur on Brassica juncea L. genotypes under NaCl stress. Protoplasma 249:139–153
Skwierawska M, Zawartka L, Skwierawski A, Nogalska A (2012) The effect of different sulfur doses and forms on changes of soil heavy metals. Plant Soil Environ 58:135–140
Srivalli S, Khanna-Chopra R (2008) Role of glutathione in abiotic stress tolerance. In: Sulfur assimilation and abiotic stress in plants. Springer, Berlin, Heidelberg, pp 207–225
Srivastava S, Shukla AK (2016) Differential response of black gram towards heavy metal stress. Environ Pollut Prot 1:89–96
Tea I, Genter T, Naulet N, Lummerzheim M, Kleiber D (2007) Interaction between nitrogen and sulfur by foliar application and its effects on flour bread-making quality. J Sci Food Agric 87:2853–2859
Tomooka N (2002) Two new species, new species combinations and sectional designations in Vigna subgenus Ceratotropis (Piper) Verdcourt (Leguminosae, Phaseoleae). Kew Bull 57:613–624
Torabian S, Zahedi M, Khoshgoftar AH (2017) Effects of foliar spray of nano-particles of FeSO4 on the growth and ion content of sunflower under saline condition. J Plant Nutr 40:615–623
Tsujimoto Y, Inusah B, Katsura K, Fuseini A, Dogbe W, Zakaria AI, Fujihara Y, Oda M, Sakagami JI (2017) The effect of sulfur fertilization on rice yields and nitrogen use efficiency in a floodplain ecosystem of northern Ghana. Field Crop Res 211:155–164
Ullah S, Anwar S, Khan GR, Anjum MM, Ali N, Jalal A, Ali K, Zaman KU, Miraj M, Sohail A (2019) Effect of potassium and sulfur on grain yield, oil concentration and fatty acid profile of sunflower. Pure Appl Biol 8:139–150
Usha Rani K, Sharma KL, Nagasri K, Srinivas K, Vishnu Murthy T, Maruthi Shankar GR, Korwar GR, Sridevi Sankar K, Madhavi M, Kusuma Grace J (2009) Response of sunflower to sources and levels of sulfur under rainfed semi-arid tropical conditions. Commun Soil Sci Plant Anal 40:2926–2944
Usman M, Tahir M, Majeed MA (2014) Effect of zinc sulphate as soil application and seed treatment on green gram (Vigna radiata L.). Pak J Life Social Sci 12:87–91
Vazin F (2012) Effect of zinc sulfate on quantitative and qualitative characteristics of corn (Zea mays) in drought stress. Cer Agron Mold 45:15–24
Wani MA, Agha FA, Malik MA, Rather ZA (2001) Response of sunflower to sulphur application under Kashmir conditions. Appl Biol Res 3:19–22
Wu CYH, Lu J, Hu ZY (2014) Influence of sulfur supply on the iron accumulation in rice plants. Commun Soil Sci Plant Anal 45:1149–1161
Xu J, Qu D, Zhou LN (2008) Effects of sulfur nutrition on the chlorophyll content of maize leaf under zinc and drought stress. Agric Res Arid Areas 2. In Chinese
Yi H, Galant A, Ravilious GE, Preuss ML, Jez JM (2010) Sensing sulfur conditions: simple to complex protein regulatory mechanisms in plant thiol metabolism. Mol Plant 3:269–279
Zafar S, Nasri M, Moghadam HRT, Zahedi H (2014) Effect of zinc and sulfur foliar applications on physiological characteristics of sunflower (Helianthus annuus L.) under water deficit stress. Int J Biosci 5:87–96
Zawude S, Shanko D (2017) Effects of salinity stress on chickpea (Cicer arietinum L.) landraces during early growth stage. Int J Sci Rep 3:214–219
Zhang MY, Bourbouloux A, Cagnac O, Srikanth CV, Rentsch D, Bachhawat AK, Delrot S (2004) A novel family of transporters mediating the transport of glutathione derivatives in plants. Plant Physiol 134:482–491
Zhang H, Jiao H, Jiang CX, Wang SH, Wei ZJ, Luo JP, Jones RL (2010) Hydrogen sulfide protects soybean seedlings against drought-induced oxidative stress. Acta Physiol Plant 32:849–857
Zhong L, Hu C, Tan Q, Liu J, Sun X (2012) Effects of sulfur application on sulfur and arsenic absorption by rapeseed in arsenic-contaminated soil. Plant Soil Environ 57:429–434
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Nawaz, F. et al. (2020). Sulfur-Mediated Physiological and Biochemical Alterations to Improve Abiotic Stress Tolerance in Food Crops. In: Hasanuzzaman, M. (eds) Plant Ecophysiology and Adaptation under Climate Change: Mechanisms and Perspectives II. Springer, Singapore. https://doi.org/10.1007/978-981-15-2172-0_14
Download citation
DOI: https://doi.org/10.1007/978-981-15-2172-0_14
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-2171-3
Online ISBN: 978-981-15-2172-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)