Abstract
Though Mars is a cold and dry planet now, Mars would have harbored a large amount of liquid water on the surface early in its history. Mars could have been similar to the early Earth from which life arose 4 billion years ago, and life may have also emerged on Mars during this period. Although the Viking mission in 1976, which explored life on Mars, did not find evidence for life, many findings associated with the possibility of life have been discovered since the Viking mission: past and present aqueous environments, organic compounds, methane, reduced compounds suitable for microorganism energy sources, and so on. These findings suggest that life might exist on Mars. Habitable environments may be deep subsurface, but it may also be on or near the surface where physical and chemical conditions on which even terrestrial microorganisms to survive are found. Life detection instruments have been developed since the Viking mission. Traces or existence of Martian life might be found by future exploration.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The possibility of life on Mars has enamored many people for many years. In the 1970s, the Viking landers conducted search-for-life experiments and failed to detect evidence of life on the planet. After the Viking mission, both the National Aeronautics and Space Administration (NASA) and the European Space Agency (ESA) focused on investigating ancient habitability in their Mars exploration programs. Evidence of past liquid water activities has been reported: large outflow channels found by the Mars Global Surveyor (Malin and Carr 1999; Malin and Edgett 2000), H2O ice under tens of centimeters of soil found by the Mars Odyssey Neutron Spectrometer (Feldman et al. 2002), hydrated sulfate and phyllosilicates found by the Mars Express spacecraft (Gendrin et al. 2005), sedimentary rocks found by the Mars Exploration Rover Opportunity (Squyres and Knoll 2005), and an H2O ice table found by the Phoenix lander (Smith et al. 2009). These findings suggest that ancient Mars sustained large quantities of liquid water and contained suitable conditions for life (see Chap. 21; Lasue et al. 2013). Assuming that life emerges in suitable conditions, it can be hypothesized that, since life arose within a few hundred million years after the formation of Earth’s surface (McKay and Davis 1991), life could have also arisen on early Mars.
Even on Mars today, locations indicating the possible presence of liquid water have been found. Furthermore, organic compounds and energy sources used by terrestrial microorganisms have been discovered on the surface. This chapter reviews the possibility of life on Mars and the potential for exploration.
2 The Viking Mission and Recent Reexaminations
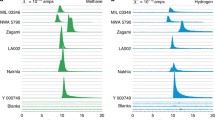
NASA conducted life exploration experiments with the Viking mission in 1976; however, the existence of life could not be verified from the surface soil samples (Klein 1977, 1978, 1979, 1992; Margulis et al. 1979). The Viking landers carried out three biological experiments on the Mars surface: the pyrolytic release (PR) experiment for detecting carbon assimilation, the labeled release (LR) experiment for detecting the decomposition of organic compounds, and the gas exchange (GEX) experiment for detecting changes in gas composition caused by metabolic reactions (Fig. 23.1).
In the PR experiment, carbon dioxide and carbon monoxide labeled with radioactive carbon-14 (14C) were added to the soil sample with water and irradiated with light. In the presence of organisms, 14C is incorporated, and organic compounds are produced. These compounds were pyrolyzed, and the released 14C was measured with a radiation detector. Although small amounts of CO2/CO were incorporated, this incorporation was considered non-biological, because similar levels of incorporation were seen after heating the samples at 90 °C for 2 h.
In the GEX experiment, a nutrient medium containing organic substances, such as amino acids and vitamins, and a mixed gas composed of CO2 and Kr (in He) were added to the sample chamber. The changes in the gas composition were analyzed by gas chromatography after several days. If organisms were present, the gas composition would be changed by the release of carbon dioxide. Although the CO2 evolution was observed, it was thought to have come from the oxidation of organics in the nutrient medium by indigenous oxidants like Fe2O3 (Oyama and Berdahl 1977).
The LR experiment was conducted by adding seven liquid nutrients (formate, glycolate, glycine, D-alanine, L-alanine, D-lactate, and L-lactate) labeled with 14C, to the samples. The release of radioactive carbon (such as 14CO2) was expected, if organisms metabolized the nutrients. The results showed positive responses that were consistent with biological activities: radioactive gas was evolved, and the gas evolution was reduced or not observed when samples were heated at 46 °C or 160 °C, respectively (Levin and Straat 1977). However, the results were still interpreted to be non-biological responses. This interpretation was based on the following results: (1) organic compounds were not detected at levels above the detection limit of the thermal volatilization–gas chromatography–mass spectrometry (TV-GCMS) instrument, and, although chlorinated organics (chloromethane and dichloromethane) were detected, they were interpreted as terrestrial contamination (Biemann et al. 1977); (2) the results could be explained by the presence of oxidants in the regolith (Klein 1978; Margulis et al. 1979). Therefore, the most acceptable conclusion of the Viking experiments was that no organisms were present within the detection limits of these experiments (Klein 1977, 1998, 1999).
However, after the Viking mission, instrumental limitations were reported. The Viking TV-GCMS was not specifically designed to identify the presence of living cells, and the pyrolysis products of cells would not have been detected if living cells were present in quantities less than 107 cells per gram (Glavin et al. 2001). Nonvolatile salts of organic acids and low levels of organic compounds would not have been easily detected by the TV-GCMS (Benner et al. 2000; Navarro-Gonzalez et al. 2006). Thus, the existence of organic compounds on Mars could not be accurately determined by the Viking instrument.
3 Habitability
In the years since the Viking mission, both NASA and ESA have looked for evidence of ancient habitability, such as traces of past water activities. An ancient possible habitable environment was discovered by the Curiosity Rover at Yellowknife Bay in Gale Crater (Grotzinger et al. 2014). The site was determined to be an ancient lake, with neutral pH and low salinity. Reduced iron and sulfur, as possible microbial energy sources, as well as biogenic elements (C, H, O, S, N, P), have also been detected. On modern-day Mars, life (most likely microorganisms) might exist at least locally since organic compounds, possible liquid water, and energy sources have been found. Figure 23.2 provides a schematic drawing of the possible habitability of present-day Mars. To learn more about both past and present habitability on Mars, see a review by Cockell (2014).
3.1 Organic Compounds
If life existed on Mars, organic compounds would be present. The detection and interpretation of organic compounds on Mars are complicated by the existence of perchlorates. While the Viking TV-GCMS did not identify any organic compounds, the Phoenix lander conducted a chemical analysis and detected the presence of 0.4–0.6 wt% perchlorate anion (ClO4 −) from the surface soil (Hecht et al. 2009). Perchlorate is rare on the surface of Earth; it occurs naturally in hyperarid environments, such as the Atacama Desert in Chile (Catling et al. 2010) and the Antarctic Dry Valleys (Kounaves et al. 2010). Though perchlorate salts are stable at low temperature, they become strong oxidants when heated, decomposing organic compounds.
Recently, the Curiosity Rover detected chlorinated compounds (chlorobenzene and C2 to C4 dichloroalkanes) (Ming et al. 2014; Freissinet et al. 2015) and thiophenic, aromatic, and aliphatic compounds (Eigenbrode et al. 2018) in the mudstones in Gale Crater. The chlorinated compounds were interpreted to be the reaction products of pyrolysis between oxychlorine compounds, such as perchlorate, and indigenous organic compounds (Freissinet et al. 2015). However, identification of the original organic compounds is difficult due to the complex reactions during pyrolysis. Thus, it is uncertain whether these compounds are derived from Martian sources (igneous, hydrothermal, atmospheric, or biological) or exogenous sources (meteorites, comets, or interplanetary dust particles (IDPs)) (Freissinet et al. 2015). Due to the discovery of perchlorates, it has been noted that the chlorinated compound found by the Viking TV-GCMS might also be the reaction product of indigenous organic compounds and perchlorates during pyrolysis of the soil samples at 500 °C (Steininger et al. 2012; Lasne et al. 2016). Although the discovery of organic compounds does not indicate evidence of life, it also does not rule out the possibility of life. The identification and characterization of organic compounds will be important for future Mars missions.
3.2 Liquid Water
Liquid water is a fundamental requirement for life. As described in Chap. 21, ancient Mars had a large amount of liquid water on the surface, but liquid water is unstable on the present Martian surface because of low temperatures and pressures. Water on the surface exists mainly in the form of ice, which organisms cannot use. Ground ice has been identified near the surface of the planet by orbiting neutron detectors on the gamma-ray spectrometer carried by Mars Odyssey (Feldman et al. 2002), and the Phoenix lander has shown a shallow H2O ice table at depths of 5–18 cm in the northern arctic region (Smith et al. 2009).
The possibility of liquid water has also been reported on the Martian surface. For example, recurring slope lineae (RSL), narrow dark streaks on steep slopes that appear during warm seasons in equatorial regions, could be a result of liquid water flow (McEwen et al. 2014) as hydrated salts of magnesium perchlorate, magnesium chlorate, and sodium perchlorate were observed at some of the flows sites (Ojha et al. 2015), although, as another interpretation, it could be dry granular flows of sand and dust (Dundas et al. 2017). The salt solutions can lower the freezing point and the evaporation rate of water. For example, highly concentrated perchlorate solutions remain at liquid state below about −70 °C (Möhlmann and Thomsen 2011). In addition to RSL, the Curiosity Rover demonstrated that transit liquid brine is formed at night in the shallow subsurface at Gale Crater, based on the meteorological analysis. Recently the Mars Advanced Radar for Subsurface and Ionosphere Sounding (MARSIS) instrument on the Mars Express spacecraft has detected the evidence of stable liquid water, possibly perchlorate brines, about 1.5 km below the surface at the southern polar ice cap (Orosei et al. 2018). Liquid brines could be abundant on Mars, since perchlorates are widespread on the surface (Martín-Torres et al. 2015). Although it is uncertain whether the Martian brines have sufficient water activity to support life (Martín-Torres et al. 2015; Edwards and Piqueux 2016), microbial growth on Earth is known to occur in highly concentrated salt solutions (Grant 2004), and some halophilic microorganisms can use perchlorate as an electron acceptor for respiration in anaerobic conditions (Oren et al. 2014). It can be inferred, therefore, that microorganisms may survive in the briny environments on Mars.
3.3 Energy Sources
Life requires energy for its growth, reproduction, and survival. Terrestrial organisms that obtain chemical energy from the oxidation of reduced compounds (energy sources) are named chemotrophs, while organisms that use light as an energy source are named phototrophs. Chemotrophs are further classified into chemoheterotrophs, which use organic compounds as energy sources, and chemolithoautotrophs, which use inorganic compounds as energy sources. Electron transport from reduced compounds (electron donors) to oxidative compounds (electron acceptors) generates energy for the organisms, which can be calculated as Gibbs free energy. There are many combinations of electron donors and electron acceptors in terrestrial microorganisms, some of which may be used by Martian microorganisms.
Table 23.1 provides examples of potential energy sources for chemotrophs on Mars, which are known to exist or are strongly inferred to exist on the planet. The metabolism of these sources, including H2, CH4, S0, S2−, Fe2+, CO, and organic compounds, has been confirmed in terrestrial microorganisms (Cockell 2014; Rummel et al. 2014; Westall et al. 2015; Cockell et al. 2016). Potential electron acceptors in anaerobic conditions are CO2, Fe3+, SO4 2−, S0, NO3 −, MnO2, ClO4 −, H2O, and organic compounds. Oxygen that is produced by photolysis/radiolysis of water might be used as an electron acceptor, although the amount of O2 is much lower in the Martian atmosphere than on Earth (Westall et al. 2015). Low concentration of molecular hydrogen (H2) was detected in the upper atmosphere by the space-based telescopes from Earth, which was likely produced by photolysis of water vapor (Krasnopolsky and Feldman 2001). Although H2 has not been directly measured on Mars surface yet, it is inferred from the presence of olivine and serpentine (Oze and Sharma 2005; Schulte et al. 2006). Fe-bearing minerals and elemental sulfur have been identified on Mars surface (Morris et al. 2007), as well as reduced sulfur such as sulfides (Ming et al. 2014). Nitrate has not been directly detected yet, but detection of NO by the Curiosity Rover suggests the possible presence of nitrate (Stern et al. 2015). Indigenous organics may also be energy sources for chemoheterotrophs, although their structures and accessibility are unknown.
Among these energy sources, methane is a molecule with special interest, because it can be an energy source for methane-oxidizing microorganisms (methanotrophs). Methane generation is associated with microbial activities on Earth, where around 80% of natural emissions of methane originate from living microorganisms (Etiope et al. 2011).
Methane in the Martian atmosphere has been reported using a variety of methods (see Chap. 22): Earth-based telescopic observations (Krasnopolsky et al. 1997; Krasnopolsky et al. 2004; Mumma et al. 2009), the Planetary Fourier Spectrometer on board the ESA Mars Express (Formisano et al. 2004; Geminale et al. 2011) ranging from several to tens of parts per billion by volume (ppbv), and the tunable laser spectrometer in the Sample Analysis at Mars on the Curiosity Rover at ~7.2 ppbv (Webster et al. 2015). Spatial and seasonal variations of methane (Mumma et al. 2009; Webster et al. 2018), combined with its relatively short lifetime in the Martian atmosphere of about 300 years (Krasnopolsky et al. 2004) and potentially less than 200 days (Lefevre and Forget 2009), indicate that methane has been released into the atmosphere locally and periodically. Though the origins of this methane are uncertain, several generation processes have been proposed, including biotic (microbial) and abiotic processes.
Biotic methane is produced by microorganisms called methanogens, which are anaerobic ones belong to the domain Archaea. Most methanogens use H2 as an energy source and CO2 for a carbon source, and some methanogens use CO, acetate, methanol, etc. as energy sources. An important H2 origin could be subsurface serpentinization (Atreya et al. 2007). Serpentinization is a reaction of olivine- and pyroxene-rich rocks with liquid water, liberating H2 in the process (Schulte et al. 2006). Carbon dioxide may be derived from the atmosphere, magma degassing, or the thermal decomposition of carbonates on Mars (Oehler and Etiope 2017).
Hydrogen is used as an energy source not only for methanogens but also for a wide variety of chemolithoautotrophic microorganisms on Earth (Table 23.1). Since higher temperatures and pressures would sustain liquid water stably at depths below a few kilometers, a microbial community may exist in the Martian subsurface (Chapelle et al. 2002; Clifford et al. 2010; Michalski et al. 2013).
Possible abiotic methane production mechanisms include geological productions like hydrogeochemical Fischer-Tropsch-type (FTT) reactions after serpentinization (Oze and Sharma 2005); thermogenesis of organics delivered to Mars by meteorites, IDPs, or possible biotic organics (Etiope et al. 2011); geothermal reactions at high temperatures (Oehler and Etiope 2017); ultraviolet degradation of meteoritic organics (Keppler et al. 2012); production by the impact of comets (Krasnopolsky 2006); and volcanic degassing (Atreya et al. 2007). Among them, FTT reactions, which produce methane from the reaction of H2 with CO2, could be important, since they are major abiotic producers of methane on Earth that occur over a wide range of temperatures (<100 to ~500 °C) (Etiope and Sherwood Lollar 2013).
Biotic or abiotic methane could be released into the atmosphere directly and/or via clathrates (methane-hydrates) or gas-absorbing regolith. Thus, the presence of methane today does not require the presence of living methanogens; it may have been produced by past methanogens and preserved (Max and Clifford 2000; Atreya et al. 2007).
3.4 Physical and Chemical Conditions
Terrestrial microorganisms inhabit a wide range of environmental conditions (see Chap. 20). Although present Martian environments are hostile to life, some microorganisms may survive near the surface (Yamagishi et al. 2010). Microorganisms isolated from a Siberian permafrost sample, for example, were capable of growth under simulated Mars conditions: low temperature (0 °C), low pressure (7 hPa), and an anoxic CO2-dominated atmosphere (Nicholson et al. 2013).
Radiation would be a serious limiting factor for microbial survivability, since organic compounds are likely to be destroyed by ionizing radiation and UV radiation (Benner et al. 2000; Kminek and Bada 2006). The total dose of ionizing radiation on the Martian surface was measured as 76 mGy/year by the Curiosity Rover (Hassler et al. 2013). However, a radiation-tolerant microbe, Deinococcus radiodurans, can survive 5 kGy without loss of viability (Cox and Battista 2005; Dartnell et al. 2007); thus, ionizing radiation would not seriously damage these microorganisms. Though UV radiation is harmful, it would be shielded by thin layers (less than a millimeter) of dust or regolith (Mancinelli and Klovstad 2000). A depth of several centimeters from the surface, therefore, could provide sufficient covering for microorganisms to survive.
4 Life Detection Instruments and Possible Explorations
There are many biosignatures for the targets of life explorations, including organic compounds, metabolic activities, cell-like morphology, and stable isotope patterns. Organic compounds are important targets, and the GCMS is an effective instrument for the detection of organic compounds. However, as mentioned earlier in this chapter, analyses by the TV-GCMS on Mars were affected by perchlorates, which react with indigenous organics during pyrolysis. In forthcoming missions, including the Mars 2020 mission and the ExoMars 2020 mission, instruments designed to detect organic compounds without pyrolysis have been selected.
The Mars 2020 rover will detect organic compounds with the Scanning Habitable Environments with Raman and Luminescence for Organics and Chemicals (SHERLOC), which will detect and characterize minerals and organic compounds, such as aromatic hydrocarbons, with a resonance Raman spectrometer and a fluorescence spectrometer that utilizes a deep-UV laser (<250 nm) (Abbey et al. 2017). It has a context imager with a spatial resolution of 30 μm to visualize surface textures, morphology, and visible features correlated with the spectral signatures (Beegle et al. 2015). The rover will also select and cache the highest value samples for a future sample-return mission, which will take the samples to laboratories on Earth for advanced analysis.
The ExoMars 2020 rover will be equipped with a drill to collect materials from outcrops at depths down to 2 m. The organic compounds will be detected by the Mars Organic Molecule Analyzer (MOMA), which includes two different types of analysis methods, laser desorption mass spectrometry (LD-MS) and TV-GCMS, with or without derivatization agents (Vago et al. 2017). The LD-MS method is not affected by perchlorates, and the derivatization process will be useful for detecting refractory molecules like carboxylic and amino acids. It will be also equipped with a Raman laser spectrometer that will identify minerals and organic compounds (Vago et al. 2017).
The candidates for landing sites in both the Mars 2020 and ExoMars 2020 missions are places with evidence of past water activities (Ono et al. 2016; Kereszturi et al. 2016). Although RSL where possible liquid water/brine exists could indicate attractive sites at which to search for extant life, RSL are not considered indicators for high-priority sites for either project. Both missions have mainly focused on investigating ancient habitability; additionally, explorations of RSL would include Committee on Space Research (COSPAR) Planetary Protection constraints to protect Mars from contamination from terrestrial organisms (Rummel and Conley 2017). The current COSPAR Planetary Protection Policy defines Mars special regions as locations in which Earth life could propagate, where the temperature is at or above −28 °C and water activity is at least 0.5 (Kminek and Rummel 2015; Rummel and Conley 2017). Although no confirmed special regions have been shown on Mars, RSL indicate possible candidates (Rummel et al. 2014; Rummel and Conley 2017). Exhaustive discussion will be required for explorations in RSL areas.
Other attractive sites where to search for present life include methane seepage sites. Even though methanogens and other microorganisms may exist in the deep subsurface, it is difficult explore those areas. Methanotrophs, however, may be found near the surface (Yamagishi et al. 2010). Some methanotrophs on Earth utilize MnO2, Fe(OH)3, and SO4 2− as electron acceptors (Beal et al. 2009), all of which have been found on the Martian surface. Potential methane seepage sites have been indicated on Mars, such as mud volcano-like mounds, ancient springs, and rims of large impact craters (Oehler and Etiope 2017). When future work by the Trace Gas Orbiter or surface rovers determines the locations of methane seepage sites, those sites could become candidates for future exploration.
A microscopic instrument would be a powerful tool for searching for extant microorganisms, but it has not been used in space missions yet (Nadeau et al. 2008; Yamagishi et al. 2010). Microscopes directly image life forms and identify their shapes, sizes, and other morphological structures. The Life Detection Microscope (Fig. 23.3) proposed by Yamagishi et al. (2018) detects organic compounds at a spatial resolution of 1 μm, differentiating among organic compounds surrounded by membranes or with enzyme activity by staining the samples with fluorescent pigments. This technique is especially useful for the detection of living microorganisms.
Conceptual design of LDM (Life Detection Microscope) (Adapted from Yamagishi et al. 2018 (Ⓒ 2018 by the Japan Society for Aeronautical and Space Sciences and ISTS))
The search for living microorganisms is important not only for scientific interest but for planetary protection. Before future human missions begin, surveys investigating the presence of living microorganisms should be conducted to mitigate the risk of human contact with Martian microorganisms, which may be harmful to human health. Microscopic instruments would be effective tools for this purpose.
5 Conclusions
Although the Viking mission failed to detect Martian life, recent findings, such as the presence of organic compounds, energy sources, and possible liquid water, have suggested the possibility of life near the planet’s surface. Life detection instruments have been developed since the Viking mission that use Raman spectrometry and LD-MS without pyrolysis to avoid the problem caused by the reaction between indigenous organic compounds and perchlorates. Microscopic instruments that are particularly superior for detecting living microorganisms have also been proposed. These in situ instruments and a future sample-return mission might reveal the existence of life on Mars.
References
Abbey WJ, Bhartia R, Beegle LW, DeFlores L, Paez V, Sijapati K, Sijapati S, Williford K, Tuite M, Hug W (2017) Deep UV Raman spectroscopy for planetary exploration: the search for in situ organics. Icarus 290:201–214
Atreya SK, Mahaffy PR, Wong A-S (2007) Methane and related trace species on Mars: origin, loss, implications for life, and habitability. Planet Space Sci 55(3):358–369
Beal EJ, House CH, Orphan VJ (2009) Manganese- and iron-dependent marine methane oxidation. Science 325(5937):184–187
Beegle L, Bhartia R, White M, DeFlores L, Abbey W, Wu Y-H, Cameron B, Moore J, Fries M, Burton A (2015) SHERLOC: scanning habitable environments with Raman & luminescence for organics & chemicals. In Aerospace Conference, 2015 IEEE, pp 1–11
Benner SA, Devine KG, Matveeva LN, Powell DH (2000) The missing organic molecules on Mars. Proc Natl Acad Sci U S A 97(6):2425–2430
Biemann K, Oro J, Toulmin PIII, Orgel LE, Nier AO, Anderson DM, Simmonds PG, Flory D, Diaz AV, Rushneck DR, Biller JE, Lafleur AL (1977) The search for organic substances and inorganic volatile compounds in the surface of Mars. J Geophys Res 82(28):4641–4658. https://doi.org/10.1029/JS082i028p04641
Catling D, Claire M, Zahnle K, Quinn R, Clark B, Hecht M, Kounaves S (2010) Atmospheric origins of perchlorate on Mars and in the Atacama. J Geophys Res: Planets 115(E1):E00E11
Chapelle FH, O’neill K, Bradley PM, Methé BA, Ciufo SA, Knobel LL, Lovley DR (2002) A hydrogen-based subsurface microbial community dominated by methanogens. Nature 415(6869):312–315
Clifford SM, Lasue J, Heggy E, Boisson J, McGovern P, Max MD (2010) Depth of the Martian cryosphere: revised estimates and implications for the existence and detection of subpermafrost groundwater. J Geophys Res: Planets 115(E7):E07001
Cockell CS (2014) Trajectories of Martian habitability. Astrobiology 14(2):182–203
Cockell C, Bush T, Bryce C, Direito S, Fox-Powell M, Harrison J, Lammer H, Landenmark H, Martin-Torres J, Nicholson N (2016) Habitability: a review. Astrobiology 16(1):89–117
Cox MM, Battista JR (2005) Deinococcus radiodurans – the consummate survivor. Nat Rev Microbiol 3(11):882–892
Dartnell LR, Desorgher L, Ward J, Coates A (2007) Modelling the surface and subsurface Martian radiation environment: implications for astrobiology. Geophys Res Lett 34(2):L02207
Dundas CM, McEwen AS, Chojnacki M, Milazzo MP, Byrne S, McElwaine JN, Urso A (2017) Granular flows at recurring slope lineae on Mars indicate a limited role for liquid water. Nat Geosci 10(12):903–907
Edwards CS, Piqueux S (2016) The water content of recurring slope lineae on Mars. Geophys Res Lett 43(17):8912–8919
Eigenbrode JL, Summons RE, Steele A, Freissinet C, Millan M, Navarro-González R, Sutter B, McAdam AC, Franz HB, Glavin DP, Archer PD, Mahaffy PR, Conrad PG, Hurowitz JA, Grotzinger JP, Gupta S, Ming DW, Sumner DY, Szopa C, Malespin C, Buch A, Coll P (2018) Organic matter preserved in 3-billion-year-old mudstones at Gale crater. Mar Sci 360(6393):1096–1101
Etiope G, Sherwood Lollar B (2013) Abiotic methane on Earth. Rev Geophys 51(2):276–299
Etiope G, Oehler D, Allen C (2011) Methane emissions from Earth’s degassing: implications for Mars. Planet Space Sci 59(2):182–195
Feldman WC, Boynton WV, Tokar RL, Prettyman TH, Gasnault O, Squyres SW, Elphic RC, Lawrence DJ, Lawson SL, Maurice S, McKinney GW, Moore KR, Reedy RC (2002) Global distribution of neutrons from Mars: results from Mars odyssey. Science 297(5578):75–78
Formisano V, Atreya S, Encrenaz T, Ignatiev N, Giuranna M (2004) Detection of methane in the atmosphere of Mars. Science 306(5702):1758–1761. https://doi.org/10.1126/science.1101732
Freissinet C, Glavin DP, Mahaffy PR, Miller KE, Eigenbrode JL, Summons RE, Brunner AE, Buch A, Szopa C, Archer PD, Franz HB, Atreya SK, Brinckerhoff WB, Cabane M, Coll P, Conrad PG, Marais DJD, Dworkin JP, Fairén AG, François P, Grotzinger JP, Kashyap S, ILt K, Leshin LA, Malespin CA, Martin MG, Martin-Torres FJ, McAdam AC, Ming DW, Navarro-González R, Pavlov AA, Prats BD, Squyres SW, Steele A, Stern JC, Sumner DY, Sutter B, Zorzano MP (2015) Organic molecules in the Sheepbed Mudstone, Gale Crater, Mars. J Geophys Res: Planets 120(3):495–514. https://doi.org/10.1002/2014JE004737
Geminale A, Formisano V, Sindoni G (2011) Mapping methane in Martian atmosphere with PFS-MEX data. Planet Space Sci 59(2):137–148
Gendrin A, Mangold N, Bibring JP, Langevin Y, Gondet B, Poulet F, Bonello G, Quantin C, Mustard J, Arvidson R, LeMouelic S (2005) Sulfates in Martian layered terrains: the OMEGA/Mars express view. Science 307(5715):1587–1591
Glavin DP, Schubert M, Botta O, Kminek G, Bada JL (2001) Detecting pyrolysis products from bacteria on Mars. Earth Planet Sci Lett 185(1–2):1–5
Grant W (2004) Life at low water activity. Philos Trans R Soc Lond B: Biol Sci 359(1448):1249–1267
Grotzinger JP, Sumner DY, Kah L, Stack K, Gupta S, Edgar L, Rubin D, Lewis K, Schieber J, Mangold N (2014) A habitable fluvio-lacustrine environment at Yellowknife Bay, Gale Crater. Mar Sci 343(6169):1242777
Hassler DM, Zeitlin C, Wimmer-Schweingruber RF, Ehresmann B, Rafkin S, Eigenbrode JL, Brinza DE, Weigle G, Böttcher S, Böhm E (2013) Mars’ surface radiation environment measured with the Mars Science Laboratory’s Curiosity rover. Science 343:1244797
Hecht MH, Kounaves SP, Quinn RC, West SJ, Young SM, Ming DW, Catling DC, Clark BC, Boynton WV, Hoffman J, Deflores LP, Gospodinova K, Kapit J, Smith PH (2009) Detection of perchlorate and the soluble chemistry of Martian soil at the Phoenix lander site. Science 325(5936):64–67. https://doi.org/10.1126/science.1172466
Keppler F, Vigano I, McLeod A, Ott U, Früchtl M, Röckmann T (2012) Ultraviolet-radiation-induced methane emissions from meteorites and the Martian atmosphere. Nature 486(7401):93
Kereszturi A, Bradák B, Chatzitheodoridis E, Ujvari G (2016) Indicators and methods to understand past environments from ExoMars rover drills. Orig Life Evol Biospheres 46(4):435–454
Klein HP (1977) The Viking biological investigation: general aspects. J Geophys Res 82(28):4677–4680
Klein HP (1978) The Viking biological experiments on Mars. Icarus 34(3):666–674
Klein HP (1979) The Viking mission and the search for life on Mars. Rev Geophys 17(7):1655–1662
Klein HP (1992) The Viking biology experiments: epilogue and prologue. Orig Life Evol Biospheres 21(4):255–261
Klein HP (1998) The search for life on Mars: what we learned from Viking. J Geophys Res 103(E12):28463–28466. https://doi.org/10.1029/98je01722
Klein HP (1999) Did Viking discover life on Mars? Orig Life Evol Biospheres 29(6):625–631
Kminek G, Bada JL (2006) The effect of ionizing radiation on the preservation of amino acids on Mars. Earth Planet Sci Lett 245(1–2):1–5
Kminek G, Rummel J (2015) COSPAR’s planetary protection policy. Space Res Today 193:7–18
Kounaves SP, Stroble ST, Anderson RM, Moore Q, Catling DC, Douglas S, McKay CP, Ming DW, Smith PH, Tamppari LK (2010) Discovery of natural perchlorate in the Antarctic Dry Valleys and its global implications. Environ Sci Technol 44(7):2360–2364
Krasnopolsky VA (2006) Some problems related to the origin of methane on Mars. Icarus 180(2):359–367
Krasnopolsky VA, Feldman PD (2001) Detection of molecular hydrogen in the atmosphere of Mars. Science 294(5548):1914–1917
Krasnopolsky V, Bjoraker G, Mumma M, Jennings D (1997) High-resolution spectroscopy of Mars at 3.7 and 8 μm: a sensitive search for H2O2, H2CO, HCl, and CH4, and detection of HDO. J Geophys Res: Planets 102(E3):6525–6534
Krasnopolsky VA, Maillard JP, Owen TC (2004) Detection of methane in the Martian atmosphere: evidence for life? Icarus 172(2):537–547
Lasne J, Noblet A, Szopa C, Navarro-González R, Cabane M, Poch O, Stalport F, François P, Atreya SK, Coll P (2016) Oxidants at the surface of Mars: a review in light of recent exploration results. Astrobiology 16(12):977–996
Lasue J, Mangold N, Hauber E, Clifford S, Feldman W, Gasnault O, Grima C, Maurice S, Mousis O (2013) Quantitative assessments of the Martian hydrosphere. Space Sci Rev 174(1–4):155–212
Lefevre F, Forget F (2009) Observed variations of methane on Mars unexplained by known atmospheric chemistry and physics. Nature 460(7256):720
Levin GV, Straat PA (1977) Recent results from the Viking labeled release experiment on Mars. J Geophys Res 82(28):4663–4667. https://doi.org/10.1029/JS082i028p04663
Malin MC, Carr MH (1999) Groundwater formation of Martian valleys. Nature 397(6720):589–591. https://doi.org/10.1038/17551
Malin MC, Edgett KS (2000) Sedimentary rocks of early Mars. Science 290(5498):1927–1937
Mancinelli RL, Klovstad M (2000) Martian soil and UV radiation: microbial viability assessment on spacecraft surfaces. Planet Space Sci 48(11):1093–1097
Margulis L, Mazur P, Barghoorn ES, Halvorson HO, Jukes TH, Kaplan IR (1979) The Viking Mission: implications for life on Mars. J Mol Evol 14(1):223–232
Martín-Torres FJ, Zorzano M-P, Valentín-Serrano P, Harri A-M, Genzer M, Kemppinen O, Rivera-Valentin EG, Jun I, Wray J, Madsen MB (2015) Transient liquid water and water activity at Gale crater on Mars. Nat Geosci 8(5):357–361
Max MD, Clifford SM (2000) The state, potential distribution, and biological implications of methane in the Martian crust. J Geophys Res: Planets 105(E2):4165–4171
McEwen AS, Dundas CM, Mattson SS, Toigo AD, Ojha L, Wray JJ, Chojnacki M, Byrne S, Murchie SL, Thomas N (2014) Recurring slope lineae in equatorial regions of Mars. Nat Geosci 7(1):53–58
McKay CP, Davis WL (1991) Duration of liquid water habitats on early Mars. Icarus 90(2):214–221
Michalski JR, Cuadros J, Niles PB, Parnell J, Rogers AD, Wright SP (2013) Groundwater activity on Mars and implications for a deep biosphere. Nat Geosci 6(2):133
Ming D, Archer P, Glavin D, Eigenbrode J, Franz H, Sutter B, Brunner A, Stern J, Freissinet C, McAdam A (2014) Volatile and organic compositions of sedimentary rocks in Yellowknife Bay, Gale Crater. Mar Sci 343(6169):1245267
Möhlmann D, Thomsen K (2011) Properties of cryobrines on Mars. Icarus 212(1):123–130
Morris R, Ming D, Yen A, Arvidson R, Gruener J, Humm D, Klingelhöfer G, Murchie S, Schröder C, Seelos IV F, Squyres S., Wisema S., Wolff M., the MER and CRISM Science Teams (2007) Possible evidence for iron sulfates, iron sulfides, and elemental sulfur at Gusev Crater, Mars, from MER, CRISM, and analog data. In Seventh International Conference on Mars
Mumma MJ, Villanueva GL, Novak RE, Hewagama T, Bonev BP, Disanti MA, Mandell AM, Smith MD (2009) Strong release of methane on Mars in northern summer 2003. Science 323(5917):1041–1045. https://doi.org/10.1126/science.1165243
Nadeau JL, Perreault NN, Niederberger TD, Whyte LG, Sun HJ, Leon R (2008) Fluorescence microscopy as a tool for in situ life detection. Astrobiology 8(4):859–874. https://doi.org/10.1089/ast.2007.0043
Navarro-Gonzalez R, Navarro KF, de la Rosa J, Iniguez E, Molina P, Miranda LD, Morales P, Cienfuegos E, Coll P, Raulin F, Amils R, McKay CP (2006) The limitations on organic detection in Mars-like soils by thermal volatilization-gas chromatography-MS and their implications for the Viking results. Proc Natl Acad Sci U S A 103(44):16089–16094. https://doi.org/10.1073/pnas.0604210103
Nicholson WL, Krivushin K, Gilichinsky D, Schuerger AC (2013) Growth of Carnobacterium spp. from permafrost under low pressure, temperature, and anoxic atmosphere has implications for Earth microbes on Mars. Proc Natl Acad Sci 110(2):666–671. https://doi.org/10.1073/pnas.1209793110
Oehler DZ, Etiope G (2017) Methane seepage on Mars: where to look and why. Astrobiology 17(12):1233–1264. https://doi.org/10.1089/ast.2017.1657
Ojha L, Wilhelm MB, Murchie SL, McEwen AS, Wray JJ, Hanley J, Masse M, Chojnacki M (2015) Spectral evidence for hydrated salts in recurring slope lineae on Mars. Nat Geosci 8:829–832. https://doi.org/10.1038/ngeo2546
Ono M, Rothrock B, Almeida E, Ansar A, Otero R, Huertas A, Heverly M (2016) Data-driven surface traversability analysis for Mars 2020 landing site selection. In: Aerospace Conference, IEEE, pp 1–12
Oren A, Bardavid RE, Mana L (2014) Perchlorate and halophilic prokaryotes: implications for possible halophilic life on Mars. Extremophiles 18(1):75–80
Orosei R, Lauro SE, Pettinelli E, Cicchetti A, Coradini M, Cosciotti B, Di Paolo F, Flamini E, Mattei E, Pajola M, Soldovieri F, Cartacci M, Cassenti F, Frigeri A, Giuppi S, Martufi R, Masdea A, Mitri G, Nenna C, Noschese R, Restano M, Seu R (2018) Radar evidence of subglacial liquid water on Mars. Science 361:eaar7268
Oyama VI, Berdahl BJ (1977) The Viking gas exchange experiment results from Chryse and Utopia surface samples. J Geophys Res 82(28):4669–4676. https://doi.org/10.1029/JS082i028p04669
Oze C, Sharma M (2005) Have olivine, will gas: serpentinization and the abiogenic production of methane on Mars. Geophys Res Lett 32(10):L10203
Rummel J, Conley C (2017) Four fallacies and an oversight: searching for Martian life. Astrobiology 17(10):1–4
Rummel JD, Beaty DW, Jones MA, Bakermans C, Barlow NG, Boston PJ, Chevrier VF, Clark BC, de Vera J-PP, Gough RV (2014) A new analysis of Mars “special regions”: findings of the second MEPAG Special Regions Science Analysis Group (SR-SAG2). Astrobiology 14(11):887–968
Schulte M, Blake D, Hoehler T, McCollom T (2006) Serpentinization and its implications for life on the early Earth and Mars. Astrobiology 6(2):364–376
Smith PH, Tamppari LK, Arvidson RE, Bass D, Blaney D, Boynton WV, Carswell A, Catling DC, Clark BC, Duck T, DeJong E, Fisher D, Goetz W, Gunnlaugsson HP, Hecht MH, Hipkin V, Hoffman J, Hviid SF, Keller HU, Kounaves SP, Lange CF, Lemmon MT, Madsen MB, Markiewicz WJ, Marshall J, McKay CP, Mellon MT, Ming DW, Morris RV, Pike WT, Renno N, Staufer U, Stoker C, Taylor P, Whiteway JA, Zent AP (2009) H2O at the phoenix landing site. Science 325(5936):58–61. https://doi.org/10.1126/science.1172339
Squyres SW, Knoll AH (2005) Sedimentary rocks at Meridiani Planum: origin, diagenesis, and implications for life on Mars. Earth Planet Sci Lett 240(1):1–10
Steininger H, Goesmann F, Goetz W (2012) Influence of magnesium perchlorate on the pyrolysis of organic compounds in Mars analogue soils. Planet Space Sci 71(1):9–17
Stern JC, Sutter B, Freissinet C, Navarro-González R, McKay CP, Archer PD, Buch A, Brunner AE, Coll P, Eigenbrode JL (2015) Evidence for indigenous nitrogen in sedimentary and aeolian deposits from the Curiosity rover investigations at Gale crater, Mars. Proc Natl Acad Sci 112(14):4245–4250
Vago JL, Westall F, Coates AJ, Jaumann R, Korablev O, Ciarletti V, Mitrofanov I, Josset J-L, De Sanctis MC, Bibring J-P (2017) Habitability on early Mars and the search for biosignatures with the ExoMars Rover. Astrobiology 17(6–7):471–510
Webster CR, Mahaffy PR, Atreya SK, Flesch GJ, Mischna MA, Meslin P-Y, Farley KA, Conrad PG, Christensen LE, Pavlov AA (2015) Mars methane detection and variability at Gale crater. Science 347(6220):415–417
Webster CR, Mahaffy PR, Atreya SK, Moores JE, Flesch GJ, Malespin C, McKay CP, Martinez G, Smith CL, Martin-Torres J, Gomez-Elvira J, Zorzano M-P, Wong MH, Trainer MG, Steele A, Archer D, Sutter B, Coll PJ, Freissinet C, Meslin P-Y, Gough RV, House CH, Pavlov A, Eigenbrode JL, Glavin DP, Pearson JC, Keymeulen D, Christensen LE, Schwenzer SP, Navarro-Gonzalez R, Pla-García J, Rafkin SCR, Vicente-Retortillo Á, Kahanpää H, Viudez-Moreiras D, Smith MD, Harri A-M, Genzer M, Hassler DM, Lemmon M, Crisp J, Sander SP, Zurek RW, Vasavada AR (2018) Background levels of methane in Mars’ atmosphere show strong seasonal variations. Science 360(6393):1093–1096
Westall F, Foucher F, Bost N, Bertrand M, Loizeau D, Vago JL, Kminek G, Gaboyer F, Campbell KA, Bréhéret J-G (2015) Biosignatures on Mars: what, where, and how? Implications for the search for Martian life. Astrobiology 15(11):998–1029
Yamagishi A, Yokobori S, Yoshimura Y, Yamashita M, Hashimoto H, Kubota T, Yano H, Haruyama J, Tabata M, Kobayashi K, Honda H, Utsumi Y, Saiki T, Itoh T, Miyakawa A, Hamase K, Naganuma T, Mita H, Tonokura K, Sasaki S, Miyamoto H (2010) Japan Astrobiology Mars Project (JAMP): search for microbes on the Mars surface with special interest in methane-oxidizing bacteria. Biol Sci Space 24(2):67–82
Yamagishi A, Satoh T, Miyakawa A, Yoshimura Y, Sasaki S, Kobayashi K, Kebukawa Y, Yabuta H, Mita H, Imai E, Naganuma T, Fujita K, Usui T (2018) LDM (life detection microscope): in situ imaging of living cells on surface of Mars. Transaction of the Japan Society for Aeronautical and Space Sciences. Aerosp Technol Jpn 16(ISTS31):299–305
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Yoshimura, Y. (2019). The Search for Life on Mars. In: Yamagishi, A., Kakegawa, T., Usui, T. (eds) Astrobiology. Springer, Singapore. https://doi.org/10.1007/978-981-13-3639-3_23
Download citation
DOI: https://doi.org/10.1007/978-981-13-3639-3_23
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-3638-6
Online ISBN: 978-981-13-3639-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)