Abstract
Salinity is a major problem in India. It not only hampers the growth of plant and productivity but also decreases the soil productivity. Some plants develop several mechanisms to cope up Salinity stress like ion regulation by Na/H antiporter; synthesis of amino acids like valine, aspartic acid, and proline; etc. Various antioxidants play crucial role in combating salinity stress. Biochar (product obtained after pyrolysis of any organic compound) enhances the fertility of soil as it improves the soil cation exchange capacity and water holding capacity. That in turn improves the nutrient capacity of the soil. A biochar property is also dependent on the type of material and pyrolysis temperature. After biochar amendment to the saline soil, its physicochemical parameters improve like organic carbon, CEC, available phosphorous, etc. Thus, biochar not only enhances the crop productivity but also improves soil enzymatic activity.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
6.1 Introduction
The genesis of biochar is linked to the ancient Amerindian populations of the Amazon region, regionally known as Terra Preta de Indio, where dark earth was created through the use of slash-and-char techniques (Lehmann 2009; Lehmann and Joseph 2009a). Research on Terra Preta soils (hortic anthrosols) discloses various effects of biochars on the functionality of soils in the Amazonia. Generally, biochar ameliorates with the soil for maintaining soil fertility and sustainability. Biochar is also recognized as a very crucial tool for the management of the environment (Lehmann and Joseph 2009b). Biochar is a newly fabricated scientific term, which means a carbon (C)-rich product is obtained when organic substance like agricultural residues. Wood or manure is heated in a closed chamber in absence of air or little air (Lehmann and Joseph 2009a). Further, Shackley et al. (2012) defined biochar more precisely as “the porous carbonaceous solid material generated by the thermochemical conversion of organic materials in an anaerobic atmosphere that owns physicochemical properties suitable for safe and long-term storage of carbon in the environment.” The International Biochar Initiative (IBI) standardized its definition as “a solid material obtained from the thermochemical conversion of biomass in an oxygen-limited environment” (IBI 2012). All of these definitions are directly or indirectly related to the biochar production condition and its application to soil. Lehmann and Joseph (2009b) differentiate biochar operationally from charcoal. Chiefly, the difference lies in the end use of biochar and charcoal. Charcoal is used for producing fuel and energy, and basically it is a charred organic matter, whereas biochar is usually applied for carbon sequestration and environmental management. The term hydrochar is closely related to biochar; however, it is distinguished by different conditions like the hydrothermal carbonization of biomass (Libra et al. 2011). Broadly, biochar is generated by dry carbonization or pyrolysis and gasification of biomass, whereas hydrochar is produced as slurry in water by hydrothermal carbonization of biomass under pressure. These two chars varied widely in physical as well as chemical properties (Bargmann et al. 2013). Four major areas where biochar is being used in environmental management are (1) soil reclamation, (2) waste management, (3) climate change mitigation, and (4) energy generation (Lehmann and Joseph 2009a).
6.2 Biochar Properties
6.2.1 Biomass Pyrolysis
Biomass resources may be limited for the biochar production in a sustainable way. For example, biomass obtained from agricultural crops and trees (forests) may cause diminution of forest areas and increase soil erosion, hence decreasing soil fertility (Cowie et al. 2012). To overcome this situation, Brick (2010) categorized feedstocks into two groups: (1) primarily produced biomass as a resource of bioenergy and biochar and (2) by-products as waste biomass. Nowadays, waste biomass has been used extensively for production of biochar because of its cost-effectiveness and food security advantages compared to other types of biomass (Brick 2010). Biochar is usually made by thermochemical decomposition of biomass at temperatures between 200 and 900 °C in anaerobic condition, which is commonly known as pyrolysis (Demirbas and Arin 2002). Pyrolysis is further categorized into fast, intermediate, and slow depending on the residence time and temperature (Table 6.1; Mohan et al. 2006). Fast pyrolysis process have a short residence time (less than 2 s) and usually generate about 75% of bio-oil from biomass (Mohan et al. 2006). Biochar production around 25–35% comes from slow and intermediate pyrolysis procedure which has a residence time between a few minutes and several hours (Brown 2009).
Gasification is different from general pyrolysis process. In gasification, organic biomass is transformed into gases which are usually rich in carbon monoxide and hydrogen; this is obtained when biomass is burned at high temperature (more than 800 °C) in a controlled aerobic environment. The resulting gas mixture is known as synthetic gas or syngas (Mohan et al. 2006; Sohi et al. 2009).
6.2.2 Factors Affecting Biochar Properties
There are various factors that affect biochar properties, and they are feedstock type, pyrolysis temperature, and heating rate. Table 6.2 showed various types of feedstock and biochar properties.
Commonly, animal litter and solid waste produced a large amount of biochar compared to the biochar obtained from agricultural fields and wood biomasses (Enders et al. 2012). The high yield is associated with the higher inorganic constituents of the raw materials, as indicated by their high ash content. Biochar derived from animal litter contains inherent metals which protect the biochar material from the loss of volatile matter present in biochar by charging the bond dissociation energies of organic and inorganic C bonds (Cantrell et al. 2012). This finding supported by Raveendran et al. (1995) also suggested that high-yield biochar was obtained from rice husk, groundnut shells, coir pith, and wheat straw due to the higher levels of K and Zn. Usually, materials with high lignin percent produce good amount of biochar (Sohi et al. 2010a). Increased heating rate from 5 to 15 °C min−1 showed a slight decrease in biochar yield (Karaosmanoğlu et al. 2000). Pyrolysis temperature plays an influential role in determining biochar characteristics. In a study on cottonseed hulls, Uchimiya et al. (2011a) revealed that the biochar yield was affected by the pyrolysis temperatures. A swift reduction in biochar generation was found at more than 400 °C because of the loss of volatile matter and noncondensable gases (CO2, CO, H2, and CH4). The biochar yield was stable at less than 400 °C in cottonseed hulls due to low lignin content. A quick decline in biochar yields was observed at more than 300 °C due to initial degradation reactions, in the case of biochar obtained from grass and wood materials (Keiluweit et al. 2010). However, lignin content of grass is less than the lignin content of wood which causes thermal breakdown at low temperature (200–400 °C).
An enhanced temperature during pyrolysis increases the carbon content, whereas hydrogen and oxygen contents were decreased (Table 6.2). This results in lower molar H/C and O/C ratios, thereby indicating dehydration and deoxygenation of the biomass (Keiluweit et al. 2010). Biochar derived from sewage sludge and poultry manure does not undergo depolymerization due to the absence of lignocellulosic compounds. They are generally rich in N content. Regarding other elements, insufficient database is available for S and P contents of biochar. Usually, no significant change was observed in pyrolysis temperature of various feedstocks (Table 6.2) related to N content of biochar obtained from various sources. S containing functional groups (present in biochar) increases ammonia retention on the surface of char due to ammonium sulfate salts (Petit et al. 2010). Hence, the functional groups in biochar affect the nutrient cycling in soil. Pyrolysis temperature highly influences the morphology and surface structure in biochar (Liu et al. 2010; Uchimiya et al. 2011a). In general, increase in pyrolysis temperature enhances the surface area of char. Although at 700 °C a reduction in surface area has also been reported (Uchimiya et al. 2011b). At high pyrolysis temperature, the aliphatic alkyl and ester groups are disrupted or dissolved which exposes the aromatic lignin core which may be responsible for an increase in surface area (Chen and Chen 2009). Downie et al. (2009) suggested that the pore size distribution is a key factor responsible for the increase in surface area in biochar. Biochars obtained from animal litter and solid waste displayed lower surface areas, whereas biochars obtained from crop residue and wood biomass showed higher surface area even at high temperatures (Table 6.2). This may be due to the low C content and high molar H/C and O/C ratios in the latter biomass samples, leading to the formation of extensive cross-linkages (Bourke et al. 2007; Ahmad et al. 2014a).
6.3 Physiological and Biochemical Response of Plant Under Salt Stress
Salinity is the most important environmental factor that limits plant growth and productivity (Allakhverdiev et al. 2000). The harmful effects of high salt content on plants can be observed such as reduced growth or the death of plants. Many plants create different ways to eliminate salt from their cells or to agglomerate the salt in the cells. During the initiation and development of the salt stress within a plant, all crucial processes such as photosynthesis, protein synthesis, and energy and lipid metabolism are strongly affected. The prompt response is to reduce the speed of blade surface expansion, followed by a termination of expansion as the stress increases. The growth of the plant increases again when the stress is relieved. The photosynthetic rate is generally lower for plants under salt stress.
6.3.1 Salt Tolerance of Plants
Plants which are salt tolerant have a capability to grow and complete their life cycle on a medium containing high amount of salts. Plants that sustain high concentrations of salt in their rhizospheric zone and grow well are known as halophytes. Depending on their salt tolerance, halophytes are obligate or facultative. Obligate halophytes are characterized by low morphology and taxonomic diversity with relatively higher growth rates. In facultative halophytes, they are found in less healthy habitats along the border between saline and nonsaline lands and characterized by a greater physiological diversity that allows them to cope with saline conditions and nonsaline situations.
6.3.2 Mechanism Adopted by the Plant Against Salt Tolerance
Plants follow a series of molecular and biochemical strategies to deal with salt stress. Biochemical processes leading to various products which in turn stimulate various processes that help the plants to cope up with the salinity stress (Iyengar and Reddy 1996). Biochemical mechanism includes (1) selective agglomeration or elimination of ions, (2) selective ion uptake by the roots and transfer it to the leaves, (3) compartmentalization and fixation of ions at the cellular levels, (4) production of various compatible solutes, (5) alterations in photosynthetic pathway, (6) alteration in membrane structure, (7) stimulation of various enzymatic and nonenzymatic antioxidants, and (8) induction of plant hormones. Salt tolerance mechanisms operated inside the plants are generally low complex or high complex. Low-complex mechanism involves alterations in various biochemical pathways. High-complex mechanisms involve changes in crucial process like chromosomal structural changes, viz., DNA methylation and DNA elimination (Walbot and Cullis 1985), and in photosynthesis and respiration processes, i.e., water use efficiency and plasma membrane–cell wall interactions (Botella et al. 1994). It is usually found that high-complex mechanism works in coordination with low-complex mechanism (Bohnert et al. 1995).
6.3.2.1 Ion Modulation and Compartmentalization
Plant generally restricts salt uptake under saline condition or compartmentalizes the salts within various tissues (Adams et al. 1992). Plants, whether glycophyte or halophyte, cannot sustain high concentration of salts within the cytoplasm, and thus they are confined within the vacuole or fix the ions in various tissues to promote normal metabolic functions of the cell (Reddy et al. 1992; Iyengar and Reddy 1996; Zhu 2003). Cheeseman (1988) reported that glycophytes restrict Na uptake or store them in eldest tissues which will eventually be sacrificed (Cheeseman 1988). Salt-inducible enzymes expel sodium from the cytoplasm or confined within the vacuoles and this is usually done by a Na+/H+ antiporter (Apse et al. 1999). Two types of electrogenic H+ pumps exist. The first one is vacuolar-type H+-ATPase (V-ATPase) and the second one is vacuolar pyrophosphatase (V-PPase). Dietz et al. (2001) reported that H+ pumps work in a synchronized way and exist at the membranes of secretory pathways of plants. V-ATPase is the dominant H+ pump found at the endomembranes of most of the plant cells. Under normal conditions, the V-ATPase is imperative for plant growth because it provides energy to secondary transport, maintains solute homeostasis, and also enhances vesicle fusion. During abiotic stress conditions like salinity, drought, anoxia, excess heavy metals, etc. in the soil, survival of the plant cells largely resides on retaining the activity of the V-ATPase. Modulation of gene expression and its activity based on the V-ATPase adapting capability lies on long- and short-term bases. A study by Otoch et al. (2001) on salt stress in hypocotyls of Vigna unguiculata seedlings revealed that tonoplast regulates salt by H+-pumping, V-ATPase, and H+-pyrophosphatase. During salt stress, V-ATPase activity was enhanced, whereas V-PPase activity was restricted (Otoch et al. 2001). The crucial mechanism in halophytes (Suaeda salsa) against salinity stress is the modulation of V-ATPase activity, which in turn activates the tonoplast to facilitate ion uptake inside the vacuole; however, V-PPase plays a secondary role (Wang et al. 2001). When the plant is under salt stress, it maintains high concentrations of K+ and lower concentrations of Na+ in the cytosol. This is maintained by K+ and Na+ transporters, and the driving force for transport is generated by H+ pumps (Zhu et al. 1993). Some salt stress sensors have been identified which help sustain the plant during stress. Zhu et al. (1993) demonstrated that calcium signal activates the myristoylated calcium-binding protein SOS3 and the serine/threonine protein kinase SOS2. This protein kinase complex then phosphorylates and triggers various ion transporters, such as the plasma membrane Na+/H+ antiporter SOS1. Arabidopsis thaliana (AtNHX1 gene) encodes a vacuolar Na+/H+ antiporter which is crucial for salt tolerance. A similar study by Shi and Zhu (2002) reported modulation of AtNHX1 expression along with ABA production during salt stress. Experimental evidence revealed that salts like NaCl, KCl, or ABA modulate the level of AtNHX1 transcript. AtNHX1 promoter (GUS) analysis in transgenic Arabidopsis displayed that AtNHX1 is expressed in all plant tissues except the root tip. High GUS expression was observed in guard cells which further disclosed that AtNHX1 played crucial role in regulation of K+ homeostasis into the specialized cells. NaCl, KCl, or ABA regulates the expression of AtNHX1 at the transcriptional level. AtNHX1 store Na+ in the enlarged vacuoles of the root hair cells.
Studies (Liu and Zhu 1997; Lauchli and Schubert 1989) showed that Ca2+ plays a major role in salt adaptation by the plants. It also stimulates K+/Na+ transporters. High salinity also induces high cytosolic Ca2+ which is shifted from the intracellular compartments and apoplast (Knight et al. 1997). The resultant transient Ca2+ increase potentiates stress signal transduction and leads to salt adaptation (Mendoza et al. 1994; Knight et al. 1997). Other strategies of salt modulation are salt secretion and selective salt agglomeration or elimination. Salt secretion takes place with the help of unique cellular structure known as salt glands. Salts are expelled from these glands from the leaf surface and maintain ion concentration inside the cell (Hogarth 1999). Salt ejection occurs through the roots in many halophytes (Levitt 1980). Selective agglomeration of ions enables the plants to make osmotic adjustments which results in enhanced water retention and Na exclusion.
6.3.2.2 Induced Biosynthesis of Compatible Solutes
The cytoplasm cumulates low molecular mass compounds known as compatible solutes to harmonize ionic balance in vacuoles, as it does not disrupt the normal metabolic reactions rather it replaces water from the biochemical reactions (Yancey et al. 1982; Ford 1984; Ashihara et al. 1997; Hasegawa et al. 2000; Zhifang and Loescher 2003). Osmolytes maintain the osmotic balance of the tissues by water influx (or reduced efflux) to protect plant structure from salt stress. Compatible solutes contain mainly proline (Khatkar and Kuhad 2000; Singh et al. 2000), glycine betaine (GB) (Rhodes and Hanson 1993; Khan et al. 2000; Wang and Nil 2000), sugars (Kerepesi and Galiba 2000; Bohnert and Jensen 1996; Pilon-Smits et al. 1995), and polyols (Ford 1984; Popp et al. 1985; Orthen et al. 1994; Bohnert et al. 1995). Polyols have various functions, viz., as low-molecular-weight chaperones and also act as scavengers of oxygen radicals (stress-induced) (Smirnoff and Cumbes 1989; Bohnert et al. 1995). There are two types of polyols: acyclic (e.g., mannitol) and cyclic (e.g., pinitol). Mannitol serves as a compatible solute to deal with salt stress, as it is produced through the action of a mannose-6-phosphate reductase (M6PR) in celery. A bacterial gene is used to engineer mannitol biosynthesis in plants to make up the plants to cope salt stress. For example, A. thaliana (nonmannitol-producer) has been introduced with M6PR gene under control of the CaMV 35S promotor. After transformation, Arabidopsis M6PR transformants started accumulating mannitol throughout the plants, and it ranges between 0.5 and 6 mmol g_1 fresh weight. A unique compound (i.e. mannitol), neither found in celery or Arabidopsis. In the absence of NaCl, all transformants are phenotypically the same as the wild type; however, in the presence of NaCl, mature transgenic plants show a high level of salt tolerance (Zhifang and Loescher 2003). Salt stress enhances various reducing sugars like glucose, fructose, sucrose, and fructans in various plants (Kerepesi and Galiba 2000; Khatkar and Kuhad 2000; Singh et al. 2000). However, Gadallah (1999) reported decreased soluble and hydrolyzable sugars in Vicia faba due to salinity. Alamgir and Ali (1999) revealed that enhanced sugar content was observed in some genotypes of rice; however, reduction in sugar content was also observed in some genotypes under salinity stress. During salinity stress, Parida et al. (2002) reported that a decrease in starch content vis-a-vis an enhanced content of both reducing and nonreducing sugar has been found in the leaves of Bruguiera parviflora. Other studies (Khavarinejad and Mostofi 1998) illustrated that the content of soluble sugars and total saccharides in tomato was increased, but the starch content was not affected by NaCl treatment. Similarly, Gao et al. (1998) have reported upraised sucrose content along with sucrose phosphate in tomato (Lycopersicon esculentum L.), but reduction in acid invertase activity was observed due to salinity stress. A number of nitrogen-containing compounds (NCC) are agglomerated in the plants when subjected to salinity stress. The most common NCC is polyamines, amides, quaternary ammonium compounds, imino acids, and proteins. The specific NCC accumulation varies from plant to plant. Wang and Nil (2000) found that during salt stress, glycine and betaine content increase in various plants. Parida et al. (2002) also confirmed that the proline content is also known to increase under saline condition in the leaves of B. parviflora.
Amino acid like cysteine, arginine, and methionine contents were reduced in wheat plants due to NaCl treatments. However, proline, isoleucine, aspartic acid, and valine contents were enhanced in the salinity stress condition (Elshintinawy and Elshourbagy 2001).
6.3.2.3 Induction of Antioxidative Enzymes
Salt stress introduces water paucity due to osmotic effects on various metabolic activities (Greenway and Munns 1980; Cheeseman 1988). This condition leads to the formation of reactive oxygen species (ROS) such as superoxide (O2 ˙¯), hydrogen peroxide (H2O2), hydroxyl radical (OH) (Halliwell and Gutteridge 1985), and singlet oxygen (1O2) (Elstner 1987). These reactive oxygen species (ROS) can cause severe injury to the normal metabolism via lipid peroxidation (Fridovich 1986; Wise and Naylor 1987) and damage to protein and nucleic acids (Fridovich 1986; Imlay and Linn 1988). O2 concentration is very high during photosynthesis; thus, the chloroplast is more likely to generate ROS (Asada and Takahashi 1987). Superoxide (O2 ˙¯) dismutates, either enzymatically or nonenzymatically, to produce H2O2 and O2. Further, H2O2 may interact with certain metal ions or metal chelates to produce highly reactive ˙OH (Imlay and Linn 1988). Plants are equipped with a series of antioxidants to protect themselves from reactive oxygen species. The metalloenzyme superoxide dismutase (SOD) converts O2 ˙¯ to H2O2. Catalase and a variety of peroxidases (Chang et al. 1984) activate the catalysis of H2O2. However, catalase is absent in the chloroplast; H2O2 can be detoxified by an ascorbate-specific peroxidase (through the ascorbate–glutathione cycle) that is present in high concentration in this organelle (Chen and Asada 1989; Halliwell and Gutteridge 1986; Asada 1992). Plants that have high levels of antioxidants (either constitutive or induced) have been reported to cope up better against the oxidative damage. The enhanced activities of various antioxidative enzymes such as catalase (CAT), ascorbate peroxidase (APX), guaiacol peroxidase (POD), glutathione reductase (GR), and superoxide dismutase are in correlation with the high levels of salinity stress (Benavides et al. 2000; Lee et al. 2001; Mittova et al. 2003). Comba et al. (1998) revealed that soybean root nodules under salt stress showed decline of ascorbate peroxidase and glutathione reductase activities, whereas superoxide dismutase and reduced glutathione content were increased, and malondialdehyde (MDA) and total protein content remain unchanged. Willkens et al. (1997) reported that tobacco plant displayed enhanced sensitivity against salinity stress. Studies (Orr and Sohal 1992; Allen et al. 1997; Noctor and Foyer 1998) revealed that transgenic plants showed overexpression of genes which leads to enhanced activities of Mn-SOD, Fe-SOD, chloroplastic Cu/Zn-SOD, bacterial catalase, and glutathione S-transferase (GST)/glutathione peroxidase (GPX) under salinity stress (Roxas et al. 2000). Takemura et al. (2002) observed that salt stress causes stimulation of superoxide in the cytosol which in turn builds up the tolerance capacity of plants (B. gymnorrhiza).
6.3.2.4 Stimulation of Plant Hormones
A huge amount of salt stimulate plant hormones like ABA and cytokinins (Vaidyanathan et al. 1999). Abscisic acid is liable for the changes in salt stress-induced genes (De-Bruxelles et al. 1996). Gupta et al. (1998) suggested that in rice, ABA-induced genes play critical role in salt tolerance. An enhanced level of ABA, aminocyclopropane-1-carboxylic acid, and ethylene was observed in Citrus sinensis (GomezCadenas et al. 1998) during salt stress. ABA lightens the stress generated due to NaCl (Popova et al. 2002). ABA favors stomatal closure by swiftly changing ion fluxes in guard cells under stress conditions. ABA is also involved in modifications of gene expression and the diversity of potential cis-acting regulatory elements. Chen et al. (2001) demonstrated the experimental evidence which showed that the enhanced Ca2+ uptake is correlated with high levels of ABA under salt stress which further maintain membrane integrity. GomezCadenas et al. (2002) reported that ABA caused diminution of ethylene release and leaf absorption in citrus under stress. This is probably due to the decrease in the accumulation of Cl− ion in the leaves. Salt tolerance of facultative halophytic (Lophopyrum elongatum) and less salt-tolerant wheat (T. aestivum L.) increased in due course of time, when they are gradually acclimatized with the salt condition rather than suddenly shocked (Noaman et al. 2002). This acclimation to salt stress is regulated by ABA.
6.3.2.5 Change in Photosynthetic Pathway
A high salt concentration inhibits the photosynthesis by reducing the water potential. To cope up from this, plants enhance their water use efficiency under salt stress. Facultative halophytic plants such as M. crystallinum change their C3 mode to CAM photosynthesis, so as to reduce their water loss by facilitating the opening of stomata at night (Cushman et al. 1989). Plants like Atriplex lentiformis change their mode of photosynthesis from C3 to C4 pathway in response against salinity stress (Zhu and Meinzer 1999).
6.3.2.6 Molecular Mechanism of Salt Tolerance
Metabolic adaptation at cellular level makes the plant to sustain against salt stress, and a large number of genes have also been identified that copes the plant against stress (Ingram and Bartels 1996; Bray 1997; Shinozaki et al. 1998). Salt tolerance is a multigenic trait, and various genes having different functional groups are responsible for encoding salt stress proteins: (1) genes for photosynthetic enzymes, (2) genes for synthesis of compatible solutes, (3) genes for vacuolar-sequestering enzymes, and (4) genes for radical scavenging enzymes. Majority of the genes in the functional groups have been classified as salt inducible under stress conditions. Wu et al. (1996) identified mutants by a salt hypersensitivity assay in Arabidopsis, which caused K uptake due to salt stress. Kawasaki et al. (2001) investigated the transcript regulation in rice-tolerant variety Pokkali with microarrays. Adjustment of microorganisms to particular environmental stress is deeply related to the expression of various genes present in the microorganism. Kanesaki et al. (2002) found that hyperosmotic stress showed different effects on the cytoplasmic volume and gene expression in Synechocystis sp. PCC 6803. DNA microarray analysis revealed that salt stress stimulates genes for some ribosomal proteins. However, hyperosmotic stress also strongly triggers the genes for 3-ketoacyl-acyl carrier protein reductase and rare lipoprotein A. Each kind of stress stimulates a number of genes for proteins of unknown function.
6.4 How Biochar Mitigates Salinity Stress in Plants
The world population is increasing at an alarming rate and is expected to reach 9.6 billion by 2050 (FAO 2009). Increased population puts pressure for food requirement, and generation of more food in turn creates pressure on natural resources. Agricultural crops are usually subjected to abiotic stresses like salinity, drought, and heavy metal stress (Osakabe et al. 2014; Parihar et al. 2015; Rizwan et al. 2016a). Among the abiotic stresses, salinity and drought are the most serious threats to agricultural production. According to Wicke et al. (2011), more than 1100 million hectares (1128 Mha) of land surface is affected by salinity. Due to salinity problem, approximately an annual economic loss of 27.2 billion USD in terms of crop loss in irrigated agriculture land (Qadir et al. 2014). Loss in revenue increased day by day, and it will reach up to 69% if no precautionary measure is taken to mitigate the deteriorated land. However, the C emanation from degraded lands enhances the cost of reclamation. Plants under salinity have to deal with two types of stresses: one is osmotic stress and the other is ionic stress. The osmotic stress raises the salt level in the soil solution which surrounds the roots that results in water uptake hindrance and interferes in lateral bud development (Munns and Tester 2008). On the other hand, in ionic stress, when Na+ concentration is more than the threshold, it leads to leaf mortality, chlorosis, necrosis, and inhibition of photosynthesis (Glenn et al. 1999; Panuccio et al. 2014). Biochar is a charcoal-like material gathered after heating any organic material in anaerobic condition (process known as pyrolysis). It has a great demand in the agricultural sector as it enhances the soil physicochemical properties (e.g., soil water holding capacity, aggregate stability, aeration, bulk density, nutrient holding capacity, EC, pH, surface area, and CEC) when amended with degraded soil (Lehmann and Joseph 2009a; Sohi et al. 2010b; Andrenelli et al. 2016; Bamminger et al. 2016). The overall mean increase in crop productivity due to increased plant growth and biomass reported in the literature with biochar amendment was about 10–12% (Jeffery et al. 2011; Haider et al. 2015; Kim et al. 2016). Some studies have shown negative effects on crop productivity when biochar was added in the soil, which may be due to specific types of biochar (Liu et al. 2013). Biochar consists of a large degree of recalcitrant carbon (C) (Cheng et al. 2008), which may remain in the soil for 100–1000 years, and thus, biochar could be very effective in fixing the carbon in the soil and hence reducing global greenhouse gas (GHG) emission (Lehmann 2007; Sohi et al. 2010a; Chowdhury et al. 2014). There are plenty of reports which illustrate the remediation of toxic/contaminated soils (Beesley et al. 2011; Uchimiya et al. 2012; Zhang et al. 2013; Ahmad et al. 2014b; Samsuri et al. 2014). The crucial factor or mechanism behind the reclamation of contaminated soil with biochar was due to its high adsorption capacity (Samsuri et al. 2014; Zhang et al. 2013). Thomas et al. (2013) also reported high salt sorption potential of biochar, and therefore, it can alter the negative consequences of salinity by reducing Na+ uptake or by eliminating Na+ from the plant cells. The adsorption capacity of biochar is mainly governed by the characteristics of biochar (Lehmann and Joseph 2009a) and the type of feedstock and also the pyrolysis conditions under which the biochar is produced (Chen et al. 2011). The determining feature of biochar which makes it jack of all trades is the high adsorption capacity which includes its high surface area and cation exchange properties (Fig. 6.1).
6.4.1 Functional Properties of Biochar
Adsorption capacity of biochar is mainly because of the presence of functional groups generated during the pyrolysis process which in turn depends on raw material (feedstock) and the pyrolysis temperature (Chun et al. 2004; Ahmad et al. 2012). During pyrolysis at temperature around 300 °C, organic material loses various degradable compounds such as cellulose and lignin. As the temperature is intensified, the aromatic compounds become more condensed which leads to enhanced surface area of biochar (Inyang et al. 2016). The functional properties of biochar which are derived from agricultural residues can be assessed through FTIR, Boehm titrations, and scanning electron microscopy (Zhang and Luo 2014). Other studies by Zhang et al. (2014) found that the functional properties like carboxyl, lactones, and phenolic groups were observed more in wood-derived biochar than the biochar obtained from bamboo, rice husk, and rice husk ash. A similar study by Qayyum et al. (2012) illustrated high aromaticity in biochar derived from wood as compared to biochar derived from sewage sludge. Biochar derived from pine needles contains various functional groups (Ahmad et al. 2013). The presence of these and other functional groups in biochar makes them a suitable and economical choice for the adsorption of various salts present in the soils, thus mitigating the salinity of the soil (Ahmad et al. 2014a; Rajapaksha et al. 2016).
6.4.2 Salt Stress Effect on Soil Properties
Salt stress negatively affects the soil properties, plant growth, and overall productivity of plants (Ohashi et al. 2014; Rath and Rousk 2015). Commonly, salt-affected soils are further divided into saline, sodic, or saline–sodic which are mainly based on their electrical conductivity (EC), sodium adsorption ratio (SAR), and exchangeable sodium percentage (ESP) of the saturated paste extracts (Richards 1954). Among these three, the saline–sodic soils are highly deteriorated and deprived of nutrient which in turn corresponds to least productivity (Rengasamy and Olsson 1991). Salinity is inversely proportional to the soil properties such as organic matter and C:N ratio (Morrissey et al. 2014). Due to salinity and sodicity, the microbial biomass and microbial activity alter in the soil and gradually decline (Yan et al. 2015). A similar study was done by Rath and Rousk (2015); they found that soil respiration and soil microbial enzyme activities were restricted due to short- or long-term salinity and it also affects the C and nutrient cycling in the soil. However, the net respiration also depends on the residue properties. The adverse consequences of salinity are more prompt in the degraded or infertile soil having materials that are less degradable (Hasbullah and Marschner 2014).
6.4.3 Biochar Effect on Soil Properties Under Salt Stress
Biochar is well known to enhance the plant tolerance against salt stress. Amelioration of soil with the help of biochar was studied extensively; it improves the physicochemical properties of soil. Studies (Chaganti et al. (2015), Diacono and Montemurro (2015), Sun et al. (2016)) revealed that biochar not only improves the soil characteristics but also related to sodium removal as sodium leaching and EC. Biochar also rectifies the physical, chemical, and biological properties of soil under abiotic stresses (Rizwan et al. 2016b). Lu et al. (2015) reported during maize cultivation, when biochar derived from poultry manure compost (BPC) amended in saline soil showed increased microbial biomass carbon content and along with the enhanced activities of urease, invertase, and phosphatase in rhizospheric soils. Similar studies were done by Bhaduri et al. (2016); they illustrated that saline soil mitigation by biochar depends upon the amount of biochar, incubation time, biochar material, and type of soil enzymes. However, an organic amendment also improves the physicochemical properties of saline soil (Wang et al. 2014a, b). Not much literature is available on the effect of biochar on the saline soil properties. Thomas et al. (2013) showed that application of biochar (30 g m−2) on salt-affected soil did not alter the pH, although increase in EC was observed over the control. Similarly, enhanced SOC and CEC were observed after furfural biochar was applied in saline soil (Wu et al. 2014). Biochar enhanced the soil organic matter along with the CEC and inhibited the exchangeable Na (Luo et al. 2017). Aforesaid studies showed that addition of biochar in saline soils mitigates the salinity in soil and also enhances the soil microbiota.
6.4.4 Regulation of Stomatal Conductance and Reduction in Oxidative Stress
Various studies (Akhtar et al. 2015a; Lashari et al. 2015) revealed that biochar reduces salt stress in plants by lower production of phytohormones. Akhtar et al. (2015a) reported that under salinity stress, biochar restricts the ABA content in leaf and xylem sap of potato. When biochar is amended in saline soil with endophytic bacteria, it inhibits the ABA concentration in xylem of wheat and maize as compared to the unamended controls (Akhtar et al. 2015b, c). Stomatal conductance is known to be enhanced in plants grown in biochar-mediated soil under salt stress. Many recent studies (Thomas et al. 2013; Akhtar et al. 2015b, c) have revealed that biochar application to sodic/saline soils ameliorates the stomatal density and conductance in various herbaceous plants like wheat and tomato. Biochar not only improves soil properties but also enhances the soil moisture and sodium-binding capacity to biochar-amended soil which in turn reduces the root reactivity to osmotic stress (Akhtar et al. 2015c). Thus, the generation of ABA declines in the root which in turn enhances the stomatal conductance and leaf growth.
Salinity induces oxidative stress in plants by the excessive production of reactive oxygen species (Parihar et al. 2015; Farhangi-Abriz and Torabian 2017). Tartoura et al. (2014) showed that organic amendments lessen the salinity stress in plants by modulating the synthesis of antioxidant enzymes. A few studies reported that biochar mitigates the oxidative stress in plants grown in saline soils. Kim et al. (2016) found that maize under salinity stress generates high amount of ROS, but after amendment with biochar, the ascorbate peroxidase (APX) and glutathione reductase (GR) activities were declined as compared to control. Similarly, study by Lashari et al. (2015) revealed that compost of poultry manure plus diluted pyroligneous solution when applied on maize field under salt stress decreases MDA content in leaf sap. A very recent study (Farhangi-Abriz and Torabian 2017) reported that biochar when amended in saline soil decreases the antioxidant enzyme activities of plants and releases the oxidative stress generated in bean seedlings. These studies formulated that biochar could improve plant growth and biomass under salt stress by reducing oxidative stress.
6.4.5 Effects on Plant Growth, Biomass, and Photosynthesis
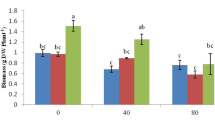
Employment of biochar in salt-affected soils is proclaimed to augment the soil properties which in turn promote plant growth and photosynthesis. For example, biochar derived from wood, when amended in saline soil, enhanced the shoot biomass, tuber yield, and Pn in potato (Akhtar et al. 2015a). Another study by Kim et al. (2016) restores the tidal land (which contained high concentration of exchangeable sodium) with the help of biochar derived from rice hull, which further promotes the maize growth. Biochar addition enhances the tomato growth and biomass under saline condition (3.6 dS m−1) (Usman et al. 2016). A study by Thomas et al. (2013) reported that biochar (pyrolyzed at 378 °C and applied at the rate of 50 t ha−1) enhances the growth of two herbaceous plant (Abutilon theophrasti and Prunella vulgaris) under salt stress; however, photosynthetic carbon gains and chlorophyll fluorescence (Fv/Fm) value have not been affected by the presence of biochar.
Overall, the biochar response under salt stress varied from plant species to species. During salinity stress, biochar derived from compost when amended in soil could also promote the plant growth and biomass. For example, Lashari et al. (2013) reported that employment of biochar poultry manure compost (BPC) along with pyroligneous solution (PS) spray for 6 weeks in the saline soil enhanced the wheat grain production as compared to the control. In another study in maize field, BPC and PS treatments in salt-affected soil promote plant height, leaf area index, and photosynthetic pigments along with the increased grain yield (Lashari et al. 2015). However, when composted biochar was applied on two halophyte species under saline conditions, similar result was reported (Luo et al. 2017). During saline conditions, biochar application with the suitable microbial inoculants like plant growth-promoting rhizobacteria (PGPR) further improved the growth of plant as compared to the control and biochar-only treatments (Nadeem et al. 2013; Fazal and Bano 2016; Akhtar et al. 2015b, c). Another study by Hammer et al. (2015) found that arbuscular mycorrhizal (AM) fungi when incorporated with biochar under salinity stress showed increased growth of lettuce as compared to the treatments alone. The beneficial effect of biochar on ion homeostasis under salinity stress could be further enhanced by co-application of biochar with endophytic bacteria (Akhtar et al. 2015a, b). However, the combined application of wheat straw-derived biochar and P increased the phosphate precipitation/sorption in the saline–sodic soil and decreased P concentrations in plants (Xu et al. 2016). These studies illustrated that biochar might be very effective in mitigating salinity stress from the soil and also inhibits Na+ uptake by plants grown in saline soils which in turn enhanced the mineral nutrients in plants. For example, the biochar and AM application promotes the P and Mn content in lettuce plants (Hammer et al. 2015). However, further research is needed to establish the mechanisms of biochar-mediated mineral uptake by plants under saline conditions both at the soil and plant levels.
6.5 Effect on Soil Properties After Addition of Biochar
6.5.1 Modification of the Soil Habitat by Biochar
The material properties of biochar are very diverse from that of the uncharred organic matter in the soil (Schmidt and Noack 2000) and are evidently changed over a period of time due to weathering, interactions with soil mineral and organic matter, and oxidation by microorganisms in soil (Lehmann et al. 2005; Cheng et al. 2008; Cheng and Lehmann 2009; Nguyen et al. 2010). The mechanism of physical and chemical characteristics of biochar and its interaction with soil biota and microorganism are poorly understood.
6.5.1.1 Basic Properties: Organic and Inorganic Composition
Biochar composition can be divided into recalcitrant C, labile or leachable C, and ash. The major chemical difference between biochar and other organic matter is the much larger proportion of aromatic C and, specifically, the occurrence of fused aromatic C structures, in contrast to other aromatic structures of soil organic matter such as lignin (Schmidt and Noack 2000). The fused aromatic structure of biochars, when obtained at lower temperature, includes amorphous C, whereas when obtained at higher temperature, turbostratic C was formed (Keiluweit et al. 2010; Nguyen et al. 2010). The stability of biochar depends largely on these C structures (Nguyen et al. 2010). However, exact mechanism of the stability to the aromatic C structures in soil is not yet clear. The microbiota present in soil does not readily absorb the C (from the biochar) although a fraction of C may be leached and therefore mineralizable (Lehmann et al. 2009), and in some studies, it has been shown that biochar stimulates the microbial activity and hence abundance (Steiner et al. 2008). At present, such fractions may be quantified by incubation studies and are frequently referred to as “volatile matter” or the labile fraction. Volatile matter refers to an ASTM standard methodology that was developed to evaluate the quality of coals as fuels and determine the stability of biochar (Deenik et al. 2010; Zimmerman 2010). However, such quantified volatile matter (5–37% of C in the study by Zimmerman 2010) is typically much larger than the corresponding mineralization (2–18% of C over 1 year). This may indicate that the mineralizable fraction is imperfectly captured by volatile matter. The next main component consists of minerals present as ash inclusions in biochar. These minerals include several significant macro- and micronutrients for biological absorption and thus the concept of valuable resources in the soil food chain. The presence of these elements during pyrolysis stimulates organo-metal reactions which are thermodynamically favorable at high temperatures. For example, N may replace one or two C atoms in aromatic compounds (Leinweber et al. 2007) with largely unknown effects on biochar behavior in soil. Iron (Fe)-rich biochars made from peat and investigated by 57Fe Mossbauer spectroscopy show the formation of Fe3C bonds and small ferromagnetic iron clusters at pyrolysis temperatures over 600 °C (Freitas et al. 2002). Grass and a variety of common raw materials (rice shells, sludge from purification plants, etc.) also contain significant amounts of amorphous silica (>2 wt%). The Si-C bonds usually participate in cross-links between aromatic domains and crystallites (Freitas et al. 2000). At temperatures of 400–600 °C, pyrolysis alters the chemical structure of biosilicates, with a continuous increase of SiO4 relative to SiO2–3 with increasing heat treatment temperature (Freitas et al. 2000). Silicates can absorb a significant proportion (>14% for corn cobs and 88% for rice shells) of biochar pore volume (Bourke et al. 2007; Freitas et al. 2000).
6.5.2 Responses of the Soil Biota to Biochar
The use of biochar as a targeted strategy for controlling soil biota is an issue of increasing interest, and unintended changes to the soil’s biota as a result of biochar application are of the same concern. This line of research is important as the health and diversity of soil microbial populations are crucial for soil functional and ecosystem services, which in turn affect the soil structure and stability, nutrient cycle, aeration, water utilization efficiency, disease resistance, and C storage capacity (Brussaard 1997). Incorporation of various organic matters to the soil is one of the most important means of managing biodiversity in soils (Brussaard et al. 2007). The distributions of organic amendments, quantity, and quality affect the trophic structure of the soil food web (Moore et al. 2004). Therefore, all three of these aspects should be considered in the use of biochar as a soil management tool. In the following section, we discuss how the biochar affects the:
-
I.
Growth of microorganism
-
II.
Nutrient transformation
In the following sections, we consider how biochar affects soil biota on several trophic levels, including root dynamics, and discuss the reasons behind observed changes with respect to different biochar properties.
6.5.3 Abundance of Microorganisms
Microbial abundance has been assured in biochar-amended soil by various methods like total genomic DNA extraction (Jin 2010), culturing and plate counting (Jackson 1958), substrate-induced respiration (Steiner et al. 2004; Kolb et al. 2009), fumigation extraction (Jin 2010; Liang et al. 2010), phospholipid fatty acid (PLFA) extraction (Birk et al. 2009), staining, and direct observation of individual biochar particles (Jackson 1958; Pietikäinen et al. 2000; Warnock et al. 2007; Jin 2010; Fig. 6.2). The microbial reproduction rate has been uplifted in some biochar-amended soils (Steiner et al. 2004) and in wastewater (Koch et al. 1991). In case of bio-digesters which are used to evolve methane (CH4) (as an energy source) after the addition of biochar led to an increase in anaerobic and cellulose-hydrolyzing bacteria (Kumar et al. 1987). The changes in microbial abundance vary within the groups of microorganism. In case of mycorrhizal fungi (arbuscular and ectomycorrhizal), enhanced growth was observed (Warnock et al. 2007). Mycorrhizal response in the host plant is most commonly measured by measuring the root colonization. Both formation rate and tip number of EM infection of larch seedling roots were increased by 19–157% with biochar additions (Makoto et al. 2010). Likewise, AM colonization of wheat roots was found to increase to 20–40% 2 years after Eucalyptus wood biochar additions of 0.6–6 t ha−1, in comparison to a colonization rate of 5–20% in unamended controls (Solaiman et al. 2010). Biochar derived through hydrothermal carbonization stimulates the spore germination of AM fungi, which further enhances the populations of these symbionts in the soil (Rillig et al. 2010). Although, some studies (Gaur and Adholeya 2000; Birk et al. 2009; Warnock et al. 2010) revealed that after biochar addition AM abundance decreases. The reasons behind the decline in population are still not clear; however, it could be because of (1) a reduced requirement for mycorrhizal symbiosis due to increased in nutrient and water availability to plants which cause decreases in mycorrhizal abundance, for example, with greater P availability in soil (Gryndler et al. 2006); (2) changes in soil conditions, e.g., due to modifications of pH or water relations (discussed below); (3) direct negative effects from high contents of mineral elements or organic compounds detrimental to the fungi, such as high salt or heavy metal contents (Killham and Firestone 1984; Killham 1985); and (iv) sorption of organic C and organically bound nutrients may influence their availability (Pietikäinen et al. 2000; Chan and Xu 2009).
The nutrient and C availability change the increase or decrease in microbial biomass, depending on (1) the existing nutrient and C availability in soil, (2) the magnitude of change, and (3) the microorganism group (Warnock et al. 2010). Bacteria may sorb to biochar surfaces and make them less susceptible to leaching in soil (Pietikäinen et al. 2000). Thus, it increases bacterial abundance although it has no effect on fungal abundance. The ability of biochars to retain bacteria will vary greatly depending on the biochar properties including the ash content, pore size, and volatile content that are highly variable (Bond 2010). Formation of surfactants by microorganisms (Ron and Rosenberg 2001) may additionally facilitate adhesion to biochars.
6.5.4 Effect of Biochar on Nutrient Transformation
Biochar can have influential effects on microbially mediated transformation of nutrients in soil. In forest soils, nitrification has been enhanced after the amelioration of soil with the biochar (MacKenzie and DeLuca 2006; Ball et al. 2010) which in turn increases sorption of phenolic that would otherwise inhibit nitrification (Zackrisson et al. 1996; DeLuca et al. 2006) and an increase in ammonia-oxidizing bacteria (Ball et al. 2010). However, biochar additions to agricultural soil show no change in net N mineralization (DeLuca et al. 2006) and less N availability to the plants (Lehmann et al. 2003). The higher the mineralizable fraction of biochar (often quantified and described as volatile matter), the greater the N immobilization with resultant decreases in N uptake and growth of crops (Deenik et al. 2010). A high microbial biomass content was observed after biochar additions. An enhanced activity of alkaline phosphatase, aminopeptidase, and N-acetylglucosaminidase was found to increase with biochar applications (Bailey et al. 2010; Jin 2010). Alkaline phosphatase increased by 615% and aminopeptidase by 15% with increasing rates of corn biochar application to an alfisol (Jin 2010). Biochar triggers the growth of fine roots and root hairs which in turn raised the production of organic N and P mineralization enzymes (Bailey et al. 2010). Biochar induces changes in the bacterial community similar to rhizosphere effects.
6.5.5 Biochar and Plant Roots
Various studies (Breazeale 1906; Nutman 1952) showed that the biochar materials have been reported to stimulate the root growth (Breazeale 1906; Nutman 1952). The different properties of biochar in comparison to the soil cause the improved root growth; however, roots may grow into the biochar pores (Lehmann et al. 2003; Joseph et al. 2010). After forest fire, a layer of char enhances the root biomass (47%) and also root tip number (64%) (Makoto et al. 2010). The root length of rice was also increased with biochar additions (Noguera et al. 2010). Germination and rooting of fir embryos (Abies numidica) significantly increased from 10% to 20% without additions to 32–80% of embryos when activated carbon was added to various growth media (Vookova and Kormutak 2001). Therefore, not only abundance but also growth behavior of roots may change in response to the presence of biochar. When the soils are amended with biochar, it improves the physicochemical properties of the soil like water availability, pH, and aeration which likely enhance root growth and also enhance shoot to root ratio (Wilson 1988). A study by Breazeale (1906) and Dachnowski (1908) have illustrated the pronounced increase in root growth after additions of carbon black (soot) to soil with sorption of allelopathic compounds that were phytotoxic. Inderjit and Callaway (2003) found that activated carbon neutralizes the phytotoxic compounds present in soil. Although these results have been challenged by Lau et al. (2008), they revealed that nutrients leaching from the activated carbons and that the addition of carbonaceous adsorbents may have multiple effects on soil. No studies have been published which showed the toxic effect of biochar on plant growth, i.e., plant growth decreased while shoot to root ratio increased.
6.6 Conclusion and Future Perspectives
Crop growth and yields have seriously been affected due to salt stress. Studies reported that the biochar application enhances the plant growth and biomass during salt stress. Biochar when applied in soil enhances photosynthesis and nutritional uptake and modified gas exchange properties in plants. Biochar inhibits Na+ uptake while increased K+ uptake in salt-stressed plants. The biochar amelioration increases the capability of the plant to tolerate high salt condition by the reduction of Na+ uptake, accumulation of minerals, and regulation of stomatal conductance and plant hormones. Overall, this chapter develops a better understanding of the biochar-mediated tolerance mechanisms in plants under salt stress. However, more research is needed to establish a different mechanism for biochar-mediated mineral uptake at plant and soil. The effect of biochar on soil also depends on the type of biochar (raw material), pyrolysis temperature, and the type of soil.
References
Adams P, Thomas JC, Vernon DM, Bohnert HJ, Jensen RG (1992) Distinct cellular and organismic responses to salt stress. Plant Cell Physiol 33:1215–1223
Ahmad M, Lee SS, Dou X, Mohan D, Sung JK, Yang JE, Ok YS (2012) Effects of pyrolysis temperature on soybean stover- and peanut shell derived biochar properties and TCE adsorption in water. Bioresour Technol 118:536–544
Ahmad M, Lee SS, Rajapaksha AU, Vithanage M, Zhang M, Cho JS, Lee SE, Ok YS (2013) Trichloroethylene adsorption by pine needle biochars produced at various pyrolysis temperatures. Bioresour Technol 143:615–622
Ahmad M, Rajapaksha AU, Lim JE, Zhang M, Bolan N, Mohan D, Vithanage M, Lee SS, Ok YS (2014a) Biochar as a sorbent for contaminant management in soil and water: a review. Chemosphere 99:19–33
Ahmad M, Lee SS, Lim JE, Lee SE, Cho JS, Moon DH, Hashimoto Y, Ok YS (2014b) Speciation and phytoavailability of lead and antimony in a small arms range soil amended with mussel shell, cow bone and biochar: EXAFS spectroscopy and chemical extractions. Chemosphere 95:433–441
Akhtar SS, Andersen MN, Liu F (2015a) Biochar mitigates salinity stress in potato. J Agron Crop Sci 201:368–378
Akhtar SS, Andersen MN, Liu F (2015b) Residual effects of biochar on improving growth, physiology and yield of wheat under salt stress. Agric Water Manag 158:61–68
Akhtar SS, Andersen MN, Naveed M, Zahir ZA, Liu F (2015c) Interactive effect of biochar and plant growth-promoting bacterial endophytes on ameliorating salinity stress in maize. Funct Plant Biol 42:770–781
Alamgir ANM, Ali MY (1999) Effect of salinity on leaf pigments, sugar and protein concentrations and chloroplast ATPase activity of rice (Oryza sativa L.) Bangladesh J Bot 28:145–149
Allakhverdiev SI, Sakamoto A, Nishiyama Y, Inaba M, Murata N (2000) Ionic and osmotic effects of NaCl-induced inactivation of photosystems I and II in Synechococcus sp. Plant Physiol 123:1047–1056
Allen RD, Webb RP, Schake SA (1997) Use of transgenic plants to study antioxidant defenses. Free Radic Biol Med 23:473–479
Andrenelli MC, Maienza A, Genesio L, Miglietta F, Pellegrini S, Vaccari FP, Vignozzi N (2016) Field application of pelletized biochar: short term effect on the hydrological properties of a silty clay loam soil. Agric Water Manag 163:190–196
Apse MP, Aharon GS, Snedden WA, Blumwald E (1999) Salt tolerance conferred by overexpression of a vacuolar Na+/H+ antiport in Arabidopsis. Science 285:1256–1258
Asada K (1992) Ascorbate peroxidase—a hydrogen peroxide scavenging enzyme in plants. Physiol Plant 85:235–241
Asada K, Takahashi M (1987) Production and scavenging of active oxygen radicals in photosynthesis. In: Kyle DJ, Osmond CB, Arntzen CJ (eds) Photoinhibition, vol 9. Elsivier, Amsterdam, pp 227–288
Ashihara H, Adachi K, Otawa M, Yasumoto E, Fukushima Y, Kato M, Sano H, Sasamoto H, Baba S (1997) Compatible solutes and inorganic ions in the mangrove plant Avicennia marina and their effects on the activities of enzymes. Z Naturforsh 52:433–440
Bailey VL, Fansler SJ, Smith JL, Bolton JH (2010) Reconciling apparent variability in effects of biochar amendment on soil enzyme activities by assay optimization. Soil Biol Biochem 43:296–301
Ball PN, MacKenzie MD, DeLuca TH, Holben WE (2010) Wildfire and charcoal enhance nitrification and ammonium-oxidizing bacteria abundance in dry montane forest soils. J Environ Qual 39:1243–1253
Bamminger C, Poll C, Sixt C, Högy P, Wüst D, Kandeler E, Marhan S (2016) Short-term response of soil microorganisms to biochar addition in a temperate agroecosystem under soil warming. Agric Ecosyst Environ 233:308–317
Bargmann I, Rilling MC, Buss W, Kruse A, Kuecke M (2013) Hydrochar and biochar effects on germination of spring barley. J Agron Crop Sci 199:360–373
Beesley L, Moreno-Jimenez E, Gomez-Eyles JL, Harris E, Robinson B, Sizmur T (2011) A review of biochars’ potential role in the remediation, revegetation and restoration of contaminated soils. Environ Pollut 159:3269–3282
Benavides MP, Marconi PL, Gallego SM, Comba ME, Tomaro ML (2000) Relationship between antioxidant defence systems and salt tolerance in Solanum tuberosum. Aust J Plant Physiol 27:273–278
Bhaduri D, Saha A, Desai D, Meena HN (2016) Restoration of carbon and microbial activity in salt-induced soil by application of peanut shell biochar during short-term incubation study. Chemosphere 148:86–98
Birk JJ, Steiner C, Teixeira WC, Zech W, Glaser B (2009) Microbial response to charcoal amendments and fertilization of a highly weathered tropical soil. In: Woods WI, Teixeira WG, Lehmann J, Steiner C, WinklerPrins AMGA, Rebellato L (eds) Amazonian dark earths: Wim Sombroek’s vision. Springer, Berlin, pp 309–324
Bohnert HJ, Jensen RG (1996) Strategies for engineering waterstress tolerance in plants. Trends Biotechnol 14:89–97
Bohnert HJ, Nelson DE, Jensen RG (1995) Adaptations to environmental stresses. Plant Cell 7:1099–1111
Bond DR (2010) Electrodes as electron acceptors, and the bacteria who love them. In: Barton LL, Mandl M, Loy A (eds) Geomicrobiology: molecular and environmental perspective. Springer, Berlin, pp 385–399
Botella MA, Quesada MA, Kononowicz AK, Bressan RA, Pliego F, Hasegawa PM, Valpuesta V (1994) Characterization and in-situ localization of a salt-induced tomato peroxidase messenger-RNA. Plant Mol Biol 25:105–114
Bourke J, Harris MM, Fushimi C, Dowaki K, Nunoura T, Antal MJ Jr (2007) Do all carbonized charcoals have the same chemical structure? 2. A model of the chemical structure of carbonized charcoal. Ind Eng Chem Res 46:5954–5967
Bray EA (1997) Plant responses to water deficit. Trends Plant Sci 2:48–54
Breazeale JF (1906) Effect of certain solids upon the growth of seedlings in water cultures. Bot Gaz 41:54–63
Brick S (2010) Biochar: assessing the promise and risks to guide US policy. Natural Resource Defense Council, New York
Brown R (2009) Biochar production technology. In: Lehmann J, Joseph S (eds) Biochar for environmental management science and technology. Earthscans, London, pp 127–146
Brussaard L (1997) Biodiversity and ecosystem functioning in soil. Ambio 26:563–570
Brussaard L, de Ruiter PC, Brown GG (2007) Soil biodiversity for agricultural sustainability. Agric Ecosyst Environ 121:233–244
Cantrell KB, Hunt PG, Uchimiya M, Novak JM, Ro KS (2012) Impact of pyrolysis temperature and manure source on physicochemical characteristics of biochar. Bioresour Technol 107:419–428
Chaganti VN, Crohn DM, Šimůnek J (2015) Leaching and reclamation of a biochar and compost amended saline–sodic soil with moderate SAR reclaimed water. Agric Water Manag 158:255–265
Chan KY, Xu Z (2009) Biochar: nutrient properties and their enhancement. In: Lehmann J, Joseph S (eds) Biochar for environmental management: science and technology. Earthscan, London, pp 67–84
Chang H, Siegel BZ, Siegel SM (1984) Salinity-induced changes in isoperoxidases in taro Colocasia esculenta. Phytochemistry 23(2):233–235
Cheeseman JM (1988) Mechanism of salinity tolerance in plants. Plant Physiol 87:547–550
Chen G, Asada K (1989) Ascorbate peroxidase in tea leaves: occurrence of two isozymes and the differences in their enzymatic and molecular properties. Plant Cell Physiol 30:987–998
Chen B, Chen Z (2009) Sorption of naphthalene and 1-naphthol by biochars of orange peels with different pyrolytic temperatures. Chemosphere 76:127–133
Chen S, Li J, Wang S, Huttermann A, Altman A (2001) Salt, nutrient uptake and transport, and ABA of Populus euphratica; a hybrid in response to increasing soil NaCl. Trees Struct Funct 15:186–194
Chen B, Zhou D, Zhu L (2008) Transitional adsorption and partition on nonpolar and polar aromatic contaminants by biochars of pine needles with different pyrolytic temperatures. Environ Sci Technol 42:5137–5143
Chen X, Chen G, Chen L, Chen Y, Lehmann J, McBride MB, Hay AG (2011) Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour Technol 102(19):8877–8884
Cheng CH, Lehmann J (2009) Ageing of black carbon along a temperature gradient. Chemosphere 75:1021–1027
Cheng CH, Lehmann J, Thies JE, Burton SD (2008) Stability of black carbon in soils across a climatic gradient. J Geophys Res 113:20–27
Chowdhury MA, de Neergaard A, Jensen LS (2014) Potential of aeration flow rate and bio-char addition to reduce greenhouse gas and ammonia emissions during manure composting. Chemosphere 97:16–25
Chun Y, Sheng G, Chiou CT, Xing B (2004) Compositions and sorptive properties of crop residue-derived chars. Environ Sci Technol 38:4649–4655
Comba ME, Benavides MP, Tomaro ML (1998) Effect of salt stress on antioxidant defence system in soybean root nodules. Aust J Plant Physiol 25:665–671
Cowie AL, Downie AE, George BH, Singh B, Zweiten LV, O’Connell D (2012) Is sustainability certification for biochar the answer to environmental risks? Pesqui Agrop Brasil 47:637–648
Cushman JC, Meyer G, Michalowski CB, Schmitt JM, Bohnert HJ (1989) Salt stress leads to differential expression of two isogenes of PEP case during CAM induction in the common ice plant. Plant Cell 1:715–725
Dachnowski A (1908) The toxic property of bog water and bog soil. Bot Gaz 46:130–143
De-Bruxelles GL, Peacock WJ, Dennies ES, Dolferus R (1996) Abscisic acid induces the alcohol dehydrogenase gene in Arabidopsis. Plant Physiol 111:381–391
Deenik JL, McClellan T, Uehara G, Antal NJ, Campbell S (2010) Charcoal volatile matter content influences plant growth and soil nitrogen transformations. Soil Sci Soc Am J 74:1259–1270
DeLuca TH, MacKenzie MD, Gundale MJ, Holben WE (2006) Wildfire-produced charcoal directly influences nitrogen cycling in ponderosa pine forests. Soil Sci Soc Am J 70:448–453
Demirbas A, Arin G (2002) An overview of biomass pyrolysis. Energy Sources 24(5):471–482
Diacono M, Montemurro F (2015) Effectiveness of organic wastes as fertilizers and amendments in salt-affected soils. Agriculture 5:221–230
Dietz KJ, Tavakoli N, Kluge C, Mimura T, Sharma SS, Harris GC, Chardonnens AN, Golldack D (2001) Significance of the V-type ATPase for the adaptation to stressful growth conditions and its regulation on the molecular and biochemical level. J Exp Bot 52:1969–1980
Downie A, Crosky A, Munroe P (2009) Physical properties of biochar. In: Lehmann J, Joseph S (eds) Biochar for environmental management science and technology. Earthscans, London, pp 13–32
Elshintinawy F, Elshourbagy MN (2001) Alleviation of changes in protein metabolism in NaCl-stressed wheat seedlings by thiamine. Biol Plant 44:541–545
Elstner EF (1987) Metabolism of activated oxygen species. In: Davies DD (ed) The biochemistry of plants. Vol. II, biochemistry of metabolism. Academic, San Diego, pp 252–315
Enders A, Hanley K, Whitman T, Joseph S, Lehmann J (2012) Characterization of biochars to evaluate recalcitrance and agronomic performance. Bioresour Technol 114:644–653
FAO (2009) www.fao.org/How to feed the world in 2050
Farhangi-Abriz S, Torabian S (2017) Antioxidant enzyme and osmotic adjustment changes in bean seedlings as affected by biochar under salt stress. Ecotoxicol Environ Saf 137:64–70
Fazal A, Bano A (2016) Role of plant growth-promoting rhizobacteria (pgpr), biochar, and chemical fertilizer under salinity stress. Commun Soil Sci Plant Anal 47:1985–1993
Ford CW (1984) Accumulation of low molecular solutes in water stress tropical legumes. Phytochemistry 23:1007–1015
Freitas JCC, Emmerich FG, Bonagamba TJ (2000) High-resolution solid-state NMR study of the occurrence and thermal transformations of silicon containing species in biomass materials. Chem Mater 12:711–718
Freitas JCC, Passamani EC, Orlando MTD, Emmerich FG, Garcia F, Sampaio LC, Bonagamba TJ (2002) Effects of ferromagnetic inclusions on 13C MAS NMR spectra of heat-treated peat samples. Energy Fuel 16:1068–1075
Fridovich I (1986) Biological effects of the superoxide radical. Arch Biochem Biophys 247:1–11
Gadallah MAA (1999) Effects of proline and glycinebetaine on Vicia faba response to salt stress. Biol Plant 42:249–257
Gao ZF, Sagi M, Lips SH (1998) Carbohydrate metabolism in leaves and assimilate partitioning in fruits of tomato (Lycopersicon esculentum L.) as affected by salinity. Plant Sci 135:149–159
Gaur A, Adholeya A (2000) Effects of the particle size of soil-less substrates upon AM fungus inoculum production. Mycorrhiza 10:43–48
Glenn EP, Brown JJ, Blumwald E (1999) Salt tolerance and crop potential of halophytes. Crit Rev Plant Sci 18:227–255
GomezCadenas A, Tadeo FR, PrimoMillo E, Talon M (1998) Involvement of abscisic acid and ethylene in the responses of citrus seedlings to salt shock. Plant Physiol 103:475–484
GomezCadenas A, Arbona V, Jacas J, PrimoMillo E, Talon M (2002) Abscisic acid reduces leaf abscission and increases salt tolerance in citrus plants. J Plant Growth Regul 21:234–240
Greenway H, Munns R (1980) Mechanisms of salt tolerance in nonhalophytes. Annu Rev Plant Physiol 31:149–190
Gryndler M, Larsen J, Hrselova H, Rezaccova V, Gryndlerova H, Kuba J (2006) Organic and mineral fertilization, respectively, increase and decrease the development of external mycelium of arbuscular mycorrhizal fungi in a longterm field experiment. Mycorrhiza 16:159–166
Gupta S, Chattopadhyay MK, Chatterjee P, Ghosh B, SenGupta DN (1998) Expression of abscisic acid-responsive elementbinding protein in salt tolerant indica rice (Oryza sativa L. cv. Pokkali). Plant Mol Biol 137:629–637
Haider G, Koyro HW, Azam F, Steffens D, Müller C, Kammann C (2015) Biochar but not humic acid product amendment affected maize yields via improving plant-soil moisture relations. Plant Soil 395:141–157
Halliwell B, Gutteridge JMC (1985) Free radicals in biology and medicine. Clarendon Press, Oxford
Halliwell B, Gutteridge JMC (1986) Free radicals in biology and medicine. Oxford University Press, London
Hammer EC, Forstreuter M, Rillig MC, Kohler J (2015) Biochar increases arbuscular mycorrhizal plant growth enhancement and ameliorates salinity stress. Appl Soil Ecol 96:114–121
Hasbullah H, Marschner P (2014) Residue properties influence the impact of salinity on soil respiration. Biol Fertil Soils 51:99–111
Hasegawa PM, Bressan RA, Zhu JK, Bohnert HJ (2000) Plant cellular and molecular responses to high salinity. Annu Rev Plant Physiol Plant Mol Biol 51:463–499
Hogarth PJ (1999) The biology of mangroves. Oxford University Press, New York
IBI (2012) Standardized product definition and product testing guidelines for biochar that is used in soil. International Biochar Initiative April 2012
Imlay JA, Linn S (1988) DNA damage and oxygen radical toxicity. Science 240:1302–1309
Inderjit, Callaway RM (2003) Experimental designs for the study of allelopathy. Plant Soil 256:1–11
Ingram J, Bartels D (1996) The molecular basis of dehydration tolerance in plants. Annu Rev Plant Physiol Plant Mol Biol 47:377–403
Inyang MI, Gao B, Yao Y, Xue Y, Zimmerman A, Mosa A, Pullammanappallil P, Ok YS, Cao X (2016) A review of biochar as a low-cost adsorbent for aqueous heavy metal removal. Crit Rev Environ Sci Technol 46:406–433
Iyengar ERR, Reddy MP (1996) Photosynthesis in highly salt tolerant plants. In: Pesserkali M (ed) Handbook of photosynthesis. Marshal Dekar, Baten Rose, USA, pp 897–909
Jackson RM (1958) The ecology of fungi in the soil with special reference to fungistasis. PhD thesis, University of London, Rothamsted Experimental Station, Harpenden, UK
Jeffery S, Verheijen GAM, Vander-Velde V, Bastos AC (2011) A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agric Ecosyst Environ 144:175–187
Jin H (2010) Characterization of microbial life colonizing biochar and biochar amended soils. PhD. dissertation, Cornell University, Ithaca, NY
Joseph SD, Camps-Arbestain M, Lin Y, Munroe P, Chia CH, Hook J, van Zwieten L, Kimber S, Cowie A, Singh BP, Lehmann J, Foidl N, Smernik RJ, Amonette JE (2010) An investigation into the reactions of biochar in soil. Aust J Soil Res 48:501–515
Kanesaki Y, Suzuki I, Allakhverdiev SI, Mikami K, Murata N (2002) Salt stress and hyperosmotic stress regulate the expression of different sets of genes in Synechocystis sp. PCC 6803. Biochem Biophys Res Commun 290:339–348
Karaosmanoǧlu F, Işıḡıgür-Ergüdenler A, Sever A (2000) Biochar from the straw-stalk of rapeseed plant. Energy Fuel 14(2):336–339
Kawasaki S, Borchert C, Deyholos M, Wang H, Brazille S, Kawai K, Galbraith D, Bohnert HJ (2001) Gene expression profiles during the initial phase of salt stress in rice. Plant Cell 13:889–905
Keiluweit M, Nico PS, Johnson MG, Kleber M (2010) Dynamic molecular structure of plant biomass-derived black carbon (biochar). Environ Sci Technol 44:1247–1253
Kerepesi I, Galiba G (2000) Osmotic and salt stress-induced alteration in soluble carbohydrate content in wheat seedlings. Crop Sci 40:482–487
Khan MA, Ungar IA, Showalter AM (2000) Effects of sodium chloride treatments on growth and ion accumulation of the halophyte Haloxylon recurvum. Commun Soil Sci Plant Anal 31:2763–2774
Khatkar D, Kuhad MS (2000) Short-term salinity induced changes in two wheat cultivars at different growth stages. Biol Plant 43:629–632
Khavarinejad RA, Mostofi Y (1998) Effects of NaCl on photosynthetic pigments, saccharides, and chloroplast ultrastructure in leaves of tomato cultivars. Photosynthetica 35:151–154
Killham K (1985) A physiological determination of the impact of environmental stress on the activity of microbial biomass. Environ Pollut Ser A 38:283–294
Killham K, Firestone MK (1984) Salt stress control of intracellular solutes in Streptomyces indigenous to saline soils. Appl Environ Microbiol 47:301–306
Kim HS, Kim KR, Yang JE, Ok YS, Owens G, Nehls T, Wessolek G, Kim KH (2016) Effect of biochar on reclaimed tidal land soil properties and maize (Zea mays L.) response. Chemosphere 142:153–159
Kloss S, Zehetner F, Dellantonio A, Hamid R, Ottner F, Liedtke V, Schwanninger M, Gerzabek MH, Soja G (2012) Characterization of slow pyrolysis biochars: effects of feedstocks and pyrolysis temperature on biochar properties. J Environ Qual 41:990–1000
Knight H, Trewavas AJ, Knight MR (1997) Calcium signaling in Arabidopsis thaliana responding to drought and salinity. Plant J 12:1067–1078
Koch B, Ostermann M, Höke H, Hempel DC (1991) Sand and activated carbon as biofilm carriers for microbial degradation of phenols and nitrogen-containing aromatic compounds. Water Res 25:1–8
Kolb SE, Fermanich KJ, Dornbush ME (2009) Effect of charcoal quantity on microbial biomass and activity in temperate soils. Soil Sci Soc Am J 73:1173–1181
Kumar S, Jain MC, Chhonkar PK (1987) A note on the stimulation of biogas production from cattle dung by addition of charcoal. Biol Wastes 20:1209–1215
Lashari MS, Liu Y, Li L, Pan W, Fu J, Pan G, Zheng J, Zheng J, Zhang X, Yu X (2013) Effects of amendment of biochar-manure compost in conjunction with pyroligneous solution on soil quality and wheat yield of a salt-stressed cropland from Central China Great Plain. Field Crop Res 144:113–118
Lashari MS, Ye Y, Ji H, Li L, Kibue GW, Lu H, Zheng J, Pan G (2015) Biochar–manure compost in conjunction with pyroligneous solution alleviated salt stress and improved leaf bioactivity of maize in a saline soil from central China: a 2-year field experiment. J Sci Food Agric 95:1321–1327
Lau JA, Puliafico KP, Kopshever JA, Steltzer H, Jarvis EP, Schwarzländer M, Strauss SY, Hufbauer RA (2008) Inference of allelopathy is complicated by effects of activated carbon on plant growth. New Phytol 178:412–423
Lauchli A, Schubert S (1989) The role of calcium in the regulation of membrane and cellular growth processes under salt stress. NATO ASI Ser G19:131–137
Lee DH, Kim YS, Lee CB (2001) The inductive responses of the antioxidant enzymes by salt stress in the rice (Oryza sativa L.) J Plant Physiol 158:737–745
Lehmann J (2007) A handful of carbon. Nature 447:143–144
Lehmann J (2009) Terra preta Nova – where to from here? In: Woods WI, Teixeira WG, Lehmann J, Steiner C, Prins AW (eds) Amazonian dark earths: Wim Sombroek’s vision. Springer, Berlin, pp 473–486
Lehmann J, Joseph S (2009a) Biochar for environmental management: science and technology. Earthscan, London
Lehmann J, Joseph S (2009b) Biochar for environmental management: an introduction. In: Lehmann J, Joseph S (eds) Biochar for environmental management science and technology. Earthscans, London, pp 1–12
Lehmann J, da Silva Jr JP, Steiner C, Nehls T, Zech W, Glaser B (2003) Nutrient availability and leaching in an archaeological Anthrosol and a Ferralsol of the Central Amazon basin: fertilizer, manure and charcoal amendments. Plant Soil 249:343–357
Lehmann J, Liang B, Solomon D, Lerotic M, Luizão F, Kinyangi F, Schäfer T, Wirick S, Jacobsen C (2005) Near-edge X-ray absorption fine structure (NEXAFS) spectroscopy for mapping nano-scale distribution of organic carbon forms in soil: application to black carbon particles. Glob Biogeochem Cycles 19:10–13
Lehmann J, Czimczik C, Laird D, Sohi S (2009) Stability of biochar in soil. In: Lehmann J, Joseph S (eds) Biochar for environmental management: science and technology. Earthscan, London, pp 183–205
Leinweber P, Kruse J, Walley FL, Gillespie A, Eckardt K-U, Blyth R, Regier T (2007) Nitrogen K-edge XANES an overview of reference compounds used to identify ‘unknown’ organic nitrogen in environmental samples. J Synchrotron Radiat 14:500–511
Levitt J (1980) Responses of plant to environmental stress chilling, freezing, and high temperature stresses, 2nd edn. Academic, New York
Liang B, Lehmann J, Sohi SP, Thies JE, O’Neill B, Trujillo L, Gaunt J, Solomon D, Grossman J, Neves EG, Luizão FJ (2010) Black carbon affects the cycling of non-black carbon in soil. Org Geochem 41(2):206–213
Libra JA, Ro KS, Kammann C, Funke A, Berge ND, Neubauer Y, Titirici MM, Fühner C, Bens O, Kern J, Emmerich KH (2011) Hydrothermal carbonization of biomass residuals: a comparative review of the chemistry, processes and applications of wet and dry pyrolysis. Biofuels 2:89–124
Liu J, Zhu JK (1997) An Arabidopsis mutant that requires increased calcium for potassium nutrition and salt tolerance. Proc Natl Acad Sci U S A 94:14960–14964
Liu Z, Zhang FS, Wu J (2010) Characterization and application of chars produced from pinewood pyrolysis and hydrothermal treatment. Fuel 89:510–514
Liu X, Zhang A, Ji C, Joseph S, Bian R, Li L, Pan G, Paz-Ferreiro J (2013) Biochar’s effect on crop productivity and the dependence on experimental conditions—a meta-analysis of literature data. Plant Soil 373:583–594
Lu W, Kang C, Wang Y, Xie Z (2015) Influence of biochar on the moisture of dark brown soil and yield of maize in Northern China. Int J Agric Biol 17:1007–1012
Luo X, Liu G, Xia Y, Chen L, Jiang Z, Zheng H, Wang Z (2017) Use of biochar-compost to improve properties and productivity of the degraded coastal soil in the Yellow River Delta, China. J Soils Sediments 17:780–789
MacKenzie MD, DeLuca TH (2006) Charcoal and shrubs modify soil processes in ponderosa pine forests of western Montana. Plant Soil 287:257–267
Makoto K, Tamai Y, Kim YS, Koike T (2010) Buried charcoal layer and ectomycorrhizae cooperatively promote the growth of Larix gmelinii seedlings. Plant Soil 327:143–152
Mendoza I, Rubio F, Rodriguez-Navarro A, Prado JM (1994) The protein phosphatase calcineurin is essential for NaCl tolerance of Saccharomyces cerevisiae. J Biol Chem 269:8792–8796
Mittova V, Tal M, Volokita M, Guy M (2003) Up-regulation of the leaf mitochondrial and peroxisomal antioxidative systems in response to salt-induced oxidative stress in the wild salt-tolerant tomato species Lycopersicon pennellii. Plant Cell Environ 26:845–856
Mohan D, Pittman CU, Steele PH (2006) Pyrolysis of wood/biomass for bio-oil: a critical review. Energy Fuel 20:848–889
Moore JC, Berlow EL, Coleman DC, de Ruiter PC, Dong Q, Hastings A, Johnson NC, McCann KS, Melville K, Morin PJ, Nadelhoffer K, Rosemond AD, Post DM, Sabo JL, Scow KM, Vanni MJ, Wall DH (2004) Detritus, trophic dynamics and biodiversity. Ecol Lett 7:584–600
Morrissey EM, Gillespie JL, Morina JC, Franklin RB (2014) Salinity affects microbial activity and soil organic matter content in tidal wetlands. Glob Chang Biol 20:1351–1362
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681
Nadeem S, Zahir Z, Naveed M, Nawaz S (2013) Mitigation of salinityinduced negative impact on the growth and yield of wheat by plant growth-promoting rhizobacteria in naturally saline conditions. Ann Microbiol 63:225–232
Nguyen B, Lehmann J, Hockaday WC, Joseph S, Masiello CA (2010) Temperature sensitivity of black carbon decomposition and oxidation. Environ Sci Technol 44:3324–3331
Noaman MM, Dvorak J, Dong JM (2002) Genes inducing salt tolerance in wheat, Lophopyrum elongatum and amphiploid and their responses to ABA under salt stress. Prospects Saline Agric (Ser: Tasks Veg Sci) 37:139–144
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49:249–279
Noguera D, Rondon M, Laossi K-R, Hoyos V, Lavelle P, de Carvalho MHC, Barot S (2010) Contrasted effect of biochar and earthworms on rice growth and resource allocation in different soils. Soil Biol Biochem 42:1017–1027
Nutman PS (1952) Host factors influencing infection and nodule development in leguminous plants. Proc R Soc Lond Ser B Biol Sci 139:176–185
Ohashi M, Kume T, Yoshifuji N, Kho LK, Nakagawa M, Nakashizuka T (2014) The effects of an induced short-term drought period on the spatial variations in soil respiration measured around emergent trees in a typical Bornean tropical forest, Malaysia. Plant Soil 387:337–349
Orr WC, Sohal RS (1992) The effects of catalase gene overexpression on life span and resistance to oxidative stress in transgenic Drosophila melanogaster. Arch Biochem Biophys 297:35–41
Orthen B, Popp M, Smirnoff N (1994) Hydroxyl radical scavenging properties of cyclitols. Proc R Soc Edinburg Sect B 102:269–272
Osakabe Y, Osakabe K, Shinozaki K, Tran LP (2014) Response of plants to water stress. Front Plant Sci 5:1–19
Otoch MDL, Sobreira ACM, deAragao MEF, Orellano EG, Lima MDS, deMelo DF (2001) Salt modulation of vacuolar H+-ATPase and H+-pyrophosphatase activities in Vigna unguiculata. J Plant Physiol 158:545–551
Panuccio MR, Jacobsen SE, Akhtar SS, Muscolo A (2014) Effect of saline water irrigation on seed germination and early seedling growth of the halophyte quinoa. AoB Plants 6:plu047
Parida A, Das AB, Das P (2002) NaCl stress causes changes in photosynthetic pigments, proteins and other metabolic components in the leaves of a true mangrove, Bruguiera parviflora, in hydroponic cultures. J Plant Biol 45:28–36
Parihar P, Singh S, Singh R, Singh VP, Prasad SM (2015) Effect of salinity stress on plants and its tolerance strategies: a review. Environ Sci Pollut Res 22:4056–4075
Petit C, Kante K, Bandosz TJ (2010) The role of sulfur-containing groups in ammonia retention on activated carbons. Carbon 48:654–667
Pietikäinen J, Kiikkilä O, Fritze H (2000) Charcoal as a habitat for microbes and its effects on the microbial community of the underlying humus. Oikos 89:231–242
Pilon-Smits EAH, Ebskamp MJM, Paul MJ, Jeuken MJW, Weisbeek PJ, Smeekens SCM (1995) Improved performance of transgenic fructan-accumulating tobacco under drought stress. Plant Physiol 107:125–130
Popova OV, Ismailov SF, Popova TN, Dietz KJ, Golldack D (2002) Salt-induced expression of NADP-dependent isocitrate dehydrogenase and ferredoxin-dependent glutamate synthase in Mesembryanthemum crystallinum. Planta 215:906–913
Popp M, Larther F, Weigel P (1985) Osmotic adaptation in Australian mangroves. Vegetation 61:247–254
Qadir M, Quillérou E, Nangia V, Murtaza G, Singh M, Thomas RJ, Drechsel P, Noble AD (2014) Economics of salt-induced land degradation and restoration. Nat Res Forum 38:282–295
Qayyum MF, Steffens D, Reisenauer HP, Schubert S (2012) Kinetics of carbon mineralization of biochars compared with wheat straw in three soils. J Environ Qual 41:1210–1220
Rajapaksha AU, Chen SS, Tsang DC, Zhang M, Vithanage M, Mandal S, Gao B, Bolan NS, Ok YS (2016) Engineered/designer biochar for contaminant removal/immobilization from soil and water: potential and implication of biochar modification. Chemosphere 148:276–291
Rath KM, Rousk J (2015) Salt effects on the soil microbial decomposer community and their role in organic carbon cycling: a review. Soil Biol Biochem 8:108–123
Raveendran K, Ganesh A, Khilar KC (1995) Influence of mineral matter on biomass pyrolysis characteristics. Fuel 74:1812–1822
Reddy MP, Sanish S, Iyengar ERR (1992) Photosynthetic studies and compartmentation of ions in different tissues of Salicornia brachiata Roxb. under saline conditions. Photosynthetica 26:173–179
Rengasamy P, Olsson K (1991) Sodicity and soil structure. Aust J Soil Res 29:935–952
Rhodes D, Hanson AD (1993) Quaternary ammonium and tertiary sulphonium compounds in higher plants. Annu Rev Plant Physiol Plant Mol Biol 44:357–384
Richards LA (1954) Diagnosis and improvement of saline and alkali soils. In: Agricultural handbook No. 60. U. S. Salinity Laboratory, Riverside, California
Rillig MC, Wagner M, Salem M, Antunes PM, George C, Ramke HG, Titirici MM, Antonietti M (2010) Material derived from hydrothermal carbonization: effects on plant growth and arbuscular mycorrhiza. Appl Soil Ecol 45:238–242
Rizwan M, Ali S, Qayyum MF, Ibrahim M, Rehman MZ, Abbas T, Ok YS (2016a) Mechanisms of biochar-mediated alleviation of toxicity of trace elements in plants: a critical review. Environ Sci Pollut Res 23:2230–2248
Rizwan M, Meunier JD, Davidian JC, Pokrovsky OS, Bovet N, Keller C (2016b) Silicon alleviates Cd stress of wheat seedlings (Triticum turgidum L. cv. Claudio) grown in hydroponics. Environ Sci Pollut Res 23:1414–1427
Ro KS, Cantrell KB, Hunt PG (2010) High-temperature pyrolysis of blended animal manures for producing renewable energy and value-added biochar. Ind Eng Chem Res 49:10125–10131
Ron EZ, Rosenberg E (2001) Natural roles of biosurfactants. Environ Microbiol 3:229–236
Roxas VP, Lodhi SA, Garrett DK, Mahan JR, Allen RD (2000) Stress tolerance in transgenic tobacco seedlings that overexpress glutathione S-transferase/glutathione peroxidase. Plant Cell Physiol 41:1229–1234
Samsuri AW, Sadegh-Zadeh F, Seh-Bardan BJ (2014) Characterization of biochars produced from oil palm and rice husks and their adsorption capacities for heavy metals. Int J Environ Sci Technol 11:967–976
Schmidt MWI, Noack AG (2000) Black carbon in soils and sediments: analysis, distribution, implications, and current challenges. Glob Biogeochem Cycles 14:777–793
Shackley S, Carter S, Knowles T, Middelink E, Haefele S, Sohi S, Cross A, Haszeldine S (2012) Sustainable gasification-biochar systems? A case-study of rice-husk gasification in Cambodia, Part 1: Context, chemical properties, environmental and health and safety issues. Energ Policy 42:49–58
Shi HZ, Zhu JK (2002) Regulation of expression of the vacuolar Na+/H+ antiporter gene AtNHX1 by salt stress and abscisic acid. Plant Mol Biol 50:543–550
Shinozaki K, Yamaguchi-Shinozaki K, Mizoguchi T, Uraro T, Katagiri T, Nakashima K, Abe H, Ichimura K, Liu QA, Nanjyo T, Uno Y, Luchi S, Srki M, Lto T, Hirayama T, Mikami K (1998) Molecular responses to water stress in Arabidopsis thaliana. J Plant Res 111:345–351
Singh SK, Sharma HC, Goswami AM, Datta SP, Singh SP (2000) In vitro growth and leaf composition of grapevine cultivars as affected by sodium chloride. Biol Plant 43:283–286
Smirnoff N, Cumbes QJ (1989) Hydroxyl radical scavenging activity of compatible solutes. Phyotochemistry 28:1057–1060
Sohi S, Loez-Capel S, Krull E, Bol R (2009) Biochar’s roles in soil and climate change: a review of research needs. CSIRO Land Water Sci Rep 5(9):64
Sohi SP, Krull E, Lopez-Capel E, Bol R (2010a) A review of biochar and its use and function in soil. Adv Agron 105:47–82
Sohi SP, Krull E, Lopez-Capel E, Bol R (2010b) A review of biochar and its use and function in soil. In: Sparks DL (ed) Advances in agronomy. Academic, Burlington, pp 47–82
Solaiman ZM, Blackwell P, Abbott LK, Storer P (2010) Direct and residual effect of biochar application on mycorrhizal colonization, growth and nutrition of wheat. Aust J Soil Res 48:546–554
Steiner C, Teixeira WG, Lehmann J, Zech W (2004) Microbial response to charcoal amendments of highly weathered soils and Amazonian dark earths in Central Amazonia – preliminary results. In: Glaser B, Woods WI (eds) Amazonian dark earths: explorations in time and space. Springer, Berlin, pp 195–212
Steiner C, Das KC, Garcia M, Förster B, Zech W (2008) Charcoal and smoke extract stimulate the soil microbial community in a highly weathered xanthic ferralsol. Pedobiologia Int J Soil Biol 51:359–366
Sun J, He F, Shao H, Zhang Z, Xu G (2016) Effects of biochar application on Suaedasalsa growth and saline soil properties. Environ Earth Sci 75:1–6
Takemura T, Hanagata N, Dubinsky Z, Karube I (2002) Molecular characterization and response to salt stress of mRNAs encoding cytosolic Cu/Zn superoxide dismutase and catalase from Bruguiera gymnorrhiza. Trees Struct Funct 16:94–99
Tartoura KA, Youssef SA, Tartoura ESA (2014) Compost alleviates the negative effects of salinity via up-regulation of antioxidants in Solanum lycopersicum L. plants. Plant Growth Regul 74:299–310
Thomas SC, Frye S, Gale N, Garmon M, Launchbury R, Machado N, Melamed S, Murray J, Petroff A, Winsborough (2013) Biochar mitigates negative effects of salt additions on two herbaceous plant species. J Environ Manag 129:62–68
Tong SJ, Li JY, Yuan JH, Xu RK (2011) Adsorption of Cu(II) by biochars generated from three crop straws. Chem Eng J 172:828–834
Uchimiya M, Wartelle LH, Lima IM, Klasson KT (2010) Sorption of deisopropylatrazine on broiler litter biochars. J Agric Food Chem 58:12350–12356
Uchimiya M, Chang S, Klasson KT (2011a) Screening biochars for heavy metal retention in soil: role of oxygen functional groups. J Hazard Mater 190:432–441
Uchimiya M, Klasson KT, Wartelle LH, Lima IM (2011b) Influence of soil properties on heavy metal questration by biochar amendment: 1. Copper sorption isotherms and the release of cations. Chemosphere 82:1431–1437
Uchimiya M, Bannon DI, Wartelle LH (2012) Retention of heavy metals by carboxyl functional groups of biochars in small arms range soil. J Agric Food Chem 60:1798–1809
Usman ARA, Al-Wabel MI, Abdulaziz AH, Mahmoud WA, El-Naggar AH, Ahmad M, Al-Faraj AAO (2016) Conocarpus biochar induces changes in soil nutrient availability and tomato growth under saline irrigation. Pedosphere 26:27–38
Vaidyanathan R, Kuruvilla S, Thomas G (1999) Characterization and expression pattern of an abscisic acid and osmotic stress responsive gene from rice. Plant Sci 140:21–30
Vookova B, Kormutak A (2001) Effect of sucrose concentration, charcoal, and indole-3-butyric acid on germination of Abies numidica somatic embryos. Biol Plant 44:181–184
Walbot V, Cullis CA (1985) Rapid genomic change in higher plants. Annu Rev Plant Physiol Plant Mol Biol 36:367–396
Wang Y, Nil N (2000) Changes in chlorophyll, ribulose biphosphate carboxylase–oxygenase, glycine betaine content, photosynthesis and transpiration in Amaranthus tricolor leaves during salt stress. J Hortic Sci Biotechnol 75:623–627
Wang BS, Luttge U, Ratajczak R (2001) Effects of salt treatment and osmotic stress on V-ATPase and V-PPase in leaves of the halophyte Suaeda salsa. J Exp Bot 52:2355–2365
Wang L, Chen W, Zhou W (2014a) Assessment of future drought in Southwest China based on CMIP5 multimodel projections. Adv Atmos Sci 31:1035–1050
Wang L, Sun X, Li S, Zhang T, Zhang W, Zhai P (2014b) Application of organic amendments to a coastal saline soil in north China: effects on soil physical and chemical properties and tree growth. PLoS One 9:1–9
Warnock DD, Lehmann J, Kuyper TW, Rillig MC (2007) Mycorrhizal responses to biochar in soil e concepts and mechanisms. Plant Soil 300:9–20
Warnock DD, Mummey DL, McBride B, Major J, Lehmann J, Rillig MC (2010) Influences of non-herbaceous biochar on arbuscular mycorrhizal fungal abundances in roots and soils: results from growth-chamber and field experiments. Appl Soil Ecol 46:450–456
Wicke B, Smeets E, Dornburg V, Vashev B, Gaiser T, Turkenburg W, Faaij A (2011) The global technical and economic potential of bioenergy from salt-affected soils. Energy Environ Sci 4:2669–2681
Willkens H, Chamnogopol S, Davey M, Schraudner M, Langebartels C (1997) Catalase is a sink for H2O2 and is indispensable for stress defense in C3 plants. EMBO J 16:4806–4816
Wilson JB (1988) A review of the evidence on the control of shoot: root ratio in relation to models. Ann Bot 61:433–449
Wise RR, Naylor AW (1987) Chilling-enhanced photooxidation: evidence for the role of singlet oxygen and endogenous antioxidants. Plant Physiol 83:278–282
Wu S, Ding L, Zhu J (1996) SOS1, a genetic locus essential for salt tolerance and potassium acquisition. Plant Cell 8:617–627
Wu Y, Xu G, Shao HB (2014) Furfural and its biochar improve the general properties of a saline soil. Solid Earth 5:665–671
Xu G, Zhang Y, Sun J, Shao H (2016) Negative interactive effects between biochar and phosphorus fertilization on phosphorus availability and plant yield in saline sodic soil. Sci Total Environ 568:910–915
Yan N, Marschner P, Cao W, Zuo C, Qin W (2015) Influence of salinity and water content on soil microorganisms. Int Soil Water Conserv Res 3:316–323
Yancey P, Clark ME, Had SC, Bowlus RD, Somero GN (1982) Living with water stress: evolution of osmolyte system. Science 217:1214–1222
Zackrisson O, Nilsson M-C, Wardle DA (1996) Key ecological function of charcoal from wildfire in the Boreal forest. Oikos 77(1):10
Zhang Y, Luo W (2014) Adsorptive removal of heavy metal from acidic wastewater with biochar produced from anaerobically digested residues: kinetics and surface complexation modeling. Biol Res 9:2484–2499
Zhang J, Lü F, Luo C, Shao L, He P (2014) Humification characterization of biochar and its potential as a composting amendment. J Environ Sci 26:390–397
Zhang X, Wang H, He L, Kouping L, Sarmah A, Li J, Bolan NS, Pei J, Huang H (2013) Using biochar for remediation of soils contaminated with heavy metals and organic pollutants. Environ Sci Pollut Res 20(12):8472–8483
Zhifang G, Loescher WH (2003) Expression of a celery mannose 6- phosphate reductase in Arabidopsis thaliana enhances salt tolerance and induces biosynthesis of both mannitol and a glucosyl-mannitol dimmer. Plant Cell Environ 26:275–283
Zhu JK (2003) Regulation of ion homeostasis under salt stress. Curr Opin Plant Biol 6:441–445
Zhu J, Meinzer FC (1999) Efficiency of C-4 photosynthesis in Atriplex lentiformis under salinity stress. Aust J Plant Physiol 26:79–86
Zhu J-K, Shi J, Singh U, Wyatt SE, Bressan RA, Hasegawa PM, Carpita NC (1993) Enrichment of vitronectinand fibronectin-like proteins in NaCl-adapted plant cells and evidence for their involvement in plasma membrane-cell wall adhesion. Plant J 3(5):637–646
Zimmerman A (2010) Abiotic and microbial oxidation of laboratory-produced black carbon (biochar). Environ Sci Technol 44:1295–1301
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Patel, A., Khare, P., Patra, D.D. (2017). Biochar Mitigates Salinity Stress in Plants. In: Shukla, V., Kumar, S., Kumar, N. (eds) Plant Adaptation Strategies in Changing Environment. Springer, Singapore. https://doi.org/10.1007/978-981-10-6744-0_6
Download citation
DOI: https://doi.org/10.1007/978-981-10-6744-0_6
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-6743-3
Online ISBN: 978-981-10-6744-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)