Abstract
Developmental dysplasia of the hip (DDH) is a common cause of secondary osteoarthritis. Various types of periacetabular osteotomy that reorient the dysplastic acetabulum have been developed to prevent the early onset of secondary osteoarthritis. Bernese periacetabular osteotomy and rotational acetabular osteotomy are now commonly used as surgical treatments for symptomatic DDH. Periacetabular osteotomies are technically demanding procedures that require detailed anatomical knowledge of the pelvic anatomy and three-dimensional (3D) cognitive skills because surgeons must avoid intra-articular perforation of chisels, thin acetabular fragments, posterior column fracture, and vascular and nerve injury. Preoperative 3D simulation of the osteotomy would be useful to avoid these perioperative complications. Computer-assisted systems such as navigation and custom cutting guides would be powerful tools with which to accurately execute the 3D osteotomy plan. A few reports have described the clinical application of a navigation system or custom cutting guide for periacetabular osteotomy. However, no navigation system that can track movement of the acetabular fragment has been developed. Some researchers have been developing such navigation systems with a focus on tracking the acetabular fragment.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Bernese periacetabular osteotomy
- Rotational acetabular osteotomy
- Curved periacetabular osteotomy
- Navigation
- Patient-specific surgical guide
1 Need for Computer Technology in Periacetabular Osteotomy
Developmental dysplasia of the hip (DDH) is a common cause of secondary osteoarthritis. Various types of periacetabular osteotomy that reorient the dysplastic acetabulum have been developed to prevent the early onset of secondary osteoarthritis [1,2,3], including Eppright’s dial osteotomy [4], Wagner’s spherical acetabular osteotomy [5], Ganz’s Bernese periacetabular osteotomy [1], rotational acetabular osteotomy (RAO) [2], and curved periacetabular osteotomy (CPO) [6]. Bernese periacetabular osteotomy and RAO are now commonly used as surgical treatments for symptomatic DDH in Europe, North America, and Japan [7]. Clinical application of computer technology has also been reported mainly for Bernese periacetabular osteotomy, RAO, and CPO.
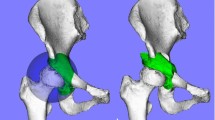
Bernese periacetabular osteotomy was first described in 1988 by Ganz et al. [1] and has been performed in Europe and North America. Its polygonal osteotomy design involves three cuts in the ilium, pubis, and ischial bones through a modified Smith-Petersen approach and maintains the integrity of the posterior column. RAO is a reorientational periacetabular osteotomy first described by Ninomiya and Tagawa in 1984 [2]. The surgical approaches and procedures of RAO have been modified, and it is currently widely used throughout Japan for patients with the early stages of osteoarthritis secondary to DDH. The design of the osteotomy is spherical, allowing the acetabular fragment to easily rotate and increasing the bone contact area between fragments for stability and bone healing (Fig. 12.1). RAO also maintains the integrity of the posterior column. CPO was modified from periacetabular osteotomy by Naito et al. [6]; the osteotomy design is spherical and performed through a modified Smith-Petersen approach.
(a) Design of rotational acetabular osteotomy and (b) position and alignment of a reoriented acetabulum were simulated three-dimensionally. Reprinted from [32]
The common aims of these techniques are to improve femoral head vertical coverage with articular cartilage and restore the femoral head from its subluxated position. Most of these osteotomies are technically demanding procedures. Insufficient lateral and anterior acetabular coverage as well as excessive anterior coverage after periacetabular osteotomy reportedly leads to early osteoarthritis [8]. Thus, many researchers have attempted to develop a three-dimensional (3D) system with which to evaluate the acetabular coverage of the femoral head [9,10,11,12,13], a 3D planning system [10, 14, 15], and a surgical guidance system for periacetabular osteotomy that includes navigation [16,17,18,19,20,21,22] and a custom cutting guide [23, 24]. Periacetabular osteotomy requires detailed anatomical knowledge of the pelvic anatomy and 3D cognitive skills because surgeons must avoid intra-articular perforation of chisels, thin acetabular fragments, posterior column fracture, and vascular and nerve injury [25,26,27].
Preoperative 3D simulation of the osteotomy would help to avoid these perioperative complications. Computer-assisted systems such as navigation and custom cutting guides would be powerful tools with which to accurately execute the 3D osteotomy plan. Many authors have reported the usefulness of navigation in total hip arthroplasty to reduce malpositioning of components and minimize leg-length discrepancy regardless of the incision length, approach, or surgeon’s experience [28]. However, a few reports have described the clinical application of a navigation system or custom cutting guide for periacetabular osteotomy (Table 12.1) [16,17,18,19, 22,23,24, 29,30,31,32].
2 Periacetabular Osteotomy Using Navigation
Langlotz et al. [16] developed their original image-guided freehand navigation of surgical instruments and applied it to Bernese periacetabular osteotomy for 12 patients with 12 dysplastic hips. Their system did not incorporate the preoperative planning of osteotomies, reorientation, or refixation, but could visualize the osteotome in real time. The real-time visualization initiated a modification of the established surgical technique in the form of an alteration of the direction of the pubis osteotomy. The system indicated that the risk of endangering the hip joint during this osteotomy could be reduced by a less markedly inclined cut. The authors also reported the outcomes of 14 cases of computer-navigated osteotomy [17]. They found that although the operative time was 20–30 min longer and the operative blood loss volume was higher than those for nonimage-guided interventions, no intraoperative or postoperative complications were observed, and an accurate and safe pelvic osteotomy could be performed.
Mayman et al. [19] developed an original CT-based navigation system and applied it to periacetabular osteotomy through a transtrochanteric approach for seven patients with eight dysplastic hips. The iliac and ischial osteotomies were marked in three dimensions using an optically tracked drill following the preoperative plan. The superior pubic rami osteotomy was performed under fluoroscopic guidance. They reported that computer enhancement of periacetabular osteotomy allows the surgeon to take the preoperative plan to the operating room and carry out that plan with accuracy, reliability, and safety. This exposure allows the experienced hip surgeon to perform the procedure safely under direct vision, even if the computer enhancement fails.
Nakahodo and Sugano et al. [18] developed an original CT-based navigation system that could guide both the surgical instruments and movement of the fragment as well as reconstruct the intraoperative change in the acetabular fragment model. Three screws were placed around the acetabular rim just before the osteotomy, and the movement of the acetabular fragment was determined by measuring the movement of the position of each screw head. They applied the navigation system not only to two cases of RAO but also to two cases of Chari medialization osteotomy. Sugano et al. [22] reported the 8-year follow-up outcomes of RAO using this CT-based navigation system. The position of the acetabular fragment was estimated by touching the edge points of the rim and osteotomy line with a navigation probe. No perioperative complications such as infection, nonunion, avascular necrosis, or neurovascular injuries occurred, but variations in the postoperative center-edge (CE) angle and acetabular roof angle were not smaller than the preoperative angles despite the fact that the preoperative plan targeted a CE angle of 35°. This indicates that the landmark matching technique to evaluate the position of the reorientated acetabulum is not as accurate as real-time tracking of the navigated osteotome. An additional tool with which to track the acetabular fragment, such as a fiducial marker or a tracker, might improve the accuracy of reorientation. Radiographic progression of osteoarthritis was found in one hip, but no hips were converted to total hip arthroplasty.
Hsieh et al. [30] performed a randomized comparative study of 36 patients undergoing periacetabular osteotomy through a transtrochanteric approach using either the commercially available CT-based navigation system (VectorVision Hip Navigation System; Brainlab Inc., Westchester, IL) or the conventional technique. They reported a reduced number of intraoperative radiographic images and a reduced operation time using the navigation system than using the conventional technique. No significant differences in the operative blood loss volume, transfusion requirement, radiographic correction of deformity, or clinical functional improvement were found after a 2-year follow-up. The authors concluded that the navigation system offers little additional benefit when the surgery is performed by an experienced surgeon. However, their navigation system did not show the preoperative plan superimposed on the bone model data, and only the tip of the osteotome was shown as a line on the monitor. Therefore, the small number of cases and the use of navigation with a primitive user interface without preoperative planning may have failed to show a benefit.
Akiyama et al. [29] applied a commercially available CT-based navigation system (Stealth Station TRIA Plus; Medtronic Surgical Navigation Technologies, Louisville, CO, USA) to CPO through a modified Smith-Petersen approach and achieved an accurate osteotomy without intraoperative complications. The navigation system neither shows the intraosseous location and direction of the osteotome nor follows an osteotomy line during an osteotomy. It cannot track the rotation of the osteotomized acetabular fragment.
Inaba et al. [31] applied a commercially available CT-based navigation system (OrthoMap 3D Navigation System; Stryker Orthopaedics, Mahwah, NJ, USA) to RAO in 23 hips of 23 patients and compared the clinical and radiographic results with those of RAO performed in 23 hips of 20 patients without navigation before introduction of the navigation system. No significant difference in the operative time or blood loss was observed between the two groups. Additionally, no significant differences in the radiological assessments were observed between the navigation and non-navigation groups. The authors performed precise 3D preoperative planning, and the navigation system could track the tip position of the osteotome and vitalize the osteotomy line and target position of the acetabular fragment; however, it could not track the movement of the acetabular fragment.
Takao et al. [32] also applied the abovementioned CT-based navigation system (OrthoMap 3D Navigation System; Stryker Orthopaedics) to RAO in 25 hips of 24 patients. They compared the outcomes of RAO performed by experienced surgeons (16 hips) and less-experienced surgeons (9 hips) using this navigation system. The navigation system could track the tip position of the osteotome and vitalize the osteotomy line and target position of the acetabular fragment (Fig. 12.2), but it could not track the movement of the acetabular fragment. There were no significant differences in the clinical or radiographic results after a minimum 2-year follow-up between the high-experience and low-experience groups. The use of navigation combined with a preoperative CT-based plan enabled less-experienced surgeons to perform RAO through a mini-incision as safely and reliably as that performed by experienced surgeons. The authors also evaluated the accuracy of the osteotomy position and acetabular movement using image registration between the preoperative and postoperative CT data.
Display of the navigation system shows the position and direction of the curved chisels during (a, b) iliac osteotomy, (c, d) ischial osteotomy, and (e, f) pubic osteotomy on (a) coronal, (c) axial, and (e) sagittal images centered on the tip of the chisel. (b, d, f) Three-dimensional views. (a, c, e) Green lines and (b, d, f) blue lines represent the tangential direction of the tip of the curved chisel. Reprinted from [32]
Preoperative planning and accurate bone cutting are major benefits of CT-based navigation. However, no commercially available navigation system can track movement of the acetabular fragment. Sugano et al. [22] attempted to estimate the position of the acetabular fragment by touching the edge points of the rim and osteotomy line with a navigation probe. However, there were two outliers (6%) that showed >10° of difference from the targeted CE angle of 35°. Takao et al. [32] confirmed anterior and lateral femoral head coverage with the moved acetabular fragment by visualization of the planned position of the acetabular fragments on the navigation monitor. The position of the reoriented acetabulum was assessed by touching the anterior and lateral edges of the reoriented acetabulum with the navigation pointer with reference to the 3D plan on the navigation monitor (Fig. 12.3). However, there were five overcorrections (20%) that showed a CE angle of >40°. A navigation system that can track the acetabular fragment is necessary to improve the accuracy of both rotation of the acetabular fragment and medialization of the femoral head [20, 21]. Some researchers have developed a navigation system that focuses on tracking of the acetabular fragment. Pflugi et al. [20] developed a cost-effective surgical navigation system to measure the orientation of the acetabular fragment during Bernese periacetabular osteotomy using commercially available inertial measurement units. They used plastic bones in the operating room to compare a previously developed optical navigation system with their inertial-based navigation system. The mean absolute difference was <4°. Murphy et al. [21] developed a biomechanical guidance system that incorporated intraoperative acetabular fragment tracking and acetabular characterization through radiographic angles and joint biomechanics and used the system for 11 patients who underwent 12 Bernese periacetabular osteotomies. The system collected information on acetabula positioning by digitizing the four divots created on the acetabular fragment by a bone burr. The measured acetabular positioning showed strong agreement with the postoperative CT measurements (−0.3° to 9.2°; mean, 3.7°).
Acquired lateral femoral head coverage by the acetabulum was checked on a (a) coronal image and a (b) three-dimensional image by touching the lateral edge of the reoriented acetabulum with the navigation pointer. The acquired anterior femoral head coverage was checked on a (c) sagittal image and a (d) three-dimensional image by touching the anterior edge of the reoriented acetabulum with the navigation pointer. (a, c) Green lines and (b, d) blue lines represent the navigation pointer. Reprinted from [32]
3 Periacetabular Osteotomy Using a Custom Cutting Guide
Navigation has several advantages such as real-time tracking of surgical tools, intraoperative validation of registration using landmarks, and intraoperative confirmation of the reoriented acetabular position. However, it has also some drawbacks such as expensive equipment and a learning curve for preparation and registration. Some researchers have attempted to develop a custom guide for hip osteotomy using 3D printing technology to overcome these drawbacks.
Radermacher et al. [23] developed individual templates to create a cutting guide of triple osteotomy, which involves three individual osteotomies in the ilium, pubis, and ischium. They applied individual templates for iliac osteotomy and compared 13 interventions using individual templates with 11 interventions performed in the conventional manner. The mean effective time of intraoperative X-ray monitoring was reduced by >75%, and the mean time of intervention was reduced by 25% compared with the conventional procedures. They concluded that the exact positioning of the osteotomies resulted in optimal mobility of the acetabulum.
Otsuki et al. [24] designed a custom cutting guide made of titanium for CPO and applied it to the C-shaped osteotomy of seven patients. They evaluated the accuracy of the osteotomy position by measuring the actual cutting radius and the planned cutting radius at three different points of the C-shaped osteotomy. The difference was <5 mm in all measurements.
4 Target 3D Acetabular Coverage in Periacetabular Osteotomy
Technological advancements in navigation systems and custom surgical guide systems might improve clinical outcomes by reducing the risk of postoperative instability due to undercorrection or impingement due to over-rotation. However, the optimal range of acetabular coverage of the femoral head remains unclear. The postoperative lateral acetabular coverage of the femoral head is a very important factor because it is reportedly a determinant of long-term outcomes of periacetabular osteotomy [8, 33]. Hartig-Andreasen et al. [34] reported that the postoperative lateral CE angle of periacetabular osteotomy should be 30–40° at the 4- to 12-year follow-up evaluations. They found the risk of conversion to total hip arthroplasty to be doubled if acetabular reorientation was not confined to this range. Albers et al. [8] found a lateral CE angle of <22° to be a risk factor for failure. Tannast et al. [35] reported that a normal lateral CE angle is within the range of 23–32°, which is quite a narrow target for periacetabular osteotomy.
The appropriate anterior acetabular coverage after periacetabular osteotomy is also a controversial topic. Overcorrection of anterior acetabular coverage may reportedly cause reduced range of motion [36] and postoperative femoral acetabular impingement [8]. Nakahara et al. [37, 38] analyzed acetabular and femoral morphologies on 3D CT images and found that the anterior and lateral acetabular coverage of both normal and dysplastic hips showed wide variations. In normal hips, the mean lateral 3D CE angle was 35.6° (range, 21.4–59.2°), and the mean anterior 3D CE angle was 58.6° (34.6–73.9°). Hamada et al. [36] simulated the range of motion of 52 DDH-affected hips after RAO with several patterns of femoral head coverage and compared them with those of 73 normal hips using computer models reconstructed from CT images. After RAO with a lateral 3D CE angle of 30° and an anterior 3D CE angle of 55° producing coverage similar to that of normal hips, the maximal flexion and maximal internal rotation at 110° flexion with 20° adduction were significantly smaller than those of the normal group. Yasunaga et al. [39] reported that the radiographic crossover sign did not influence the radiographic progression of osteoarthritis after RAO during a mean 13-year follow-up. Imai et al. [40] reported that patients with a postoperative vertical-center-anterior angle of >46° had impairment of activities of daily living associated with limitations in hip flexion after RAO. The vertical-center-anterior angle was measured on the false-profile radiographic view obtained with the patients in the standing position; thus, it was necessary to estimate changes in pelvic sagittal inclination from the supine to standing position in CT-based planning. Postoperative range of motion was determined by acetabular and femoral morphologies, so individual biomechanical planning with range-of-motion simulation and estimation of peak contact pressure during activities of daily living would be necessary.
5 Computer-Assisted Orthopedic Surgery for Proximal Femoral Osteotomy
Intertrochanteric osteotomy had been performed mainly for the adult sequelae of DDH (varus, valgus, or derotational according to the final geometry of the proximal femur). Because of the success of periacetabular osteotomy, isolated intertrochanteric osteotomy is indicated only occasionally [41]. Transtrochanteric rotational osteotomy (TRO) [42] and curved varus osteotomy (CVO) [43] are performed to preserve the joint in patients with femoral head osteonecrosis. Because of the success of total hip arthroplasty, the technical demanding nature of the procedure, and the unstable clinical results, TRO and CVO are also indicated occasionally. Navigation systems have been used in the proximal femoral region in surgeries including stem orientation in total hip arthroplasty [44], femoral component placement in hip resurfacing [45,46,47,48], removal of bone cement in revision total hip arthroplasty [49], and open reduction and fixation of proximal femur fractures [50, 51]. To the best of our knowledge, no reports have described the clinical application of a navigation system in proximal femoral osteotomy. In TRO and CVO for femoral head osteonecrosis, the ratio of the transposed intact articular surface of the femoral head to the weight-bearing surface of the acetabulum on postoperative anteroposterior radiographs (postoperative intact ratio) is the most critical factor in preventing progression of femoral head collapse [42, 43, 52,53,54,55,56]. An appropriate indication and surgical procedure are needed to obtain good postoperative outcomes of TRO and CVO. Preoperative 3D simulation of TRO using 3D CT images and/or 3D magnetic resonance images is reportedly useful to determine the proper indications for these procedures [57].
References
Ganz R, Klaue K, Vinh TS, Mast JW. A new periacetabular osteotomy for the treatment of hip dysplasias. Technique and preliminary results. Clin Orthop Relat Res. 1988;232:26–36.
Ninomiya S, Tagawa H. Rotational acetabular osteotomy for the dysplastic hip. J Bone Joint Surg Am. 1984;66(3):430–6.
Murphy SB, Ganz R, Muller ME. The prognosis in untreated dysplasia of the hip. A study of radiographic factors that predict the outcome. J Bone Joint Surg Am. 1995;77(7):985–9.
Eppright RH. Dial osteotomy of the acetabulum in the treatment of dysplasia of the hip. J Bone Joint Surg Am. 1975;57(8):1172.
Wagner H. Experiences with spherical acetabular osteotomy for the correction of the dysplastic acetabulum. In: Weil UH, editor. Acetabular dysplasia: skeletal dysplasias in childhood. Berlin: Springer; 1978. p. 131–45. https://doi.org/10.1007/978-3-642-66737-4_7.
Naito M, Shiramizu K, Akiyoshi Y, Ezoe M, Nakamura Y. Curved periacetabular osteotomy for treatment of dysplastic hip. Clin Orthop Relat Res. 2005;433:129–35.
Yasunaga Y, Yamasaki T, Ochi M. Patient selection criteria for periacetabular osteotomy or rotational acetabular osteotomy. Clin Orthop Relat Res. 2012;470(12):3342–54. https://doi.org/10.1007/s11999-012-2516-z.
Albers CE, Steppacher SD, Ganz R, Tannast M, Siebenrock KA. Impingement adversely affects 10-year survivorship after periacetabular osteotomy for DDH. Clin Orthop Relat Res. 2013;471(5):1602–14. https://doi.org/10.1007/s11999-013-2799-8.
Klaue K, Wallin A, Ganz R. CT evaluation of coverage and congruency of the hip prior to osteotomy. Clin Orthop Relat Res. 1988;232:15–25.
Millis MB, Murphy SB. Use of computed tomographic reconstruction in planning osteotomies of the hip. Clin Orthop Relat Res. 1992;274:154–9.
Dutoit M, Zambelli PY. Simplified 3D-evaluation of periacetabular osteotomy. Acta Orthop Belg. 1999;65(3):288–94.
Mechlenburg I, Nyengaard JR, Romer L, Soballe K. Changes in load-bearing area after Ganz periacetabular osteotomy evaluated by multislice CT scanning and stereology. Acta Orthop Scand. 2004;75(2):147–53. https://doi.org/10.1080/00016470412331294395.
Armiger RS, Armand M, Lepisto J, Minhas D, Tallroth K, Mears SC, Waites MD, Taylor RH. Evaluation of a computerized measurement technique for joint alignment before and during periacetabular osteotomy. Comput Aided Surg. 2007;12(4):215–24. https://doi.org/10.3109/10929080701541855.
Hipp JA, Sugano N, Millis MB, Murphy SB. Planning acetabular redirection osteotomies based on joint contact pressures. Clin Orthop Relat Res. 1999;364:134–43.
Liu L, Ecker TM, Schumann S, Siebenrock KA, Zheng G. Evaluation of constant thickness cartilage models vs. patient specific cartilage models for an optimized computer-assisted planning of periacetabular osteotomy. PLoS One. 2016;11(1):e0146452. https://doi.org/10.1371/journal.pone.0146452.
Langlotz F, Stucki M, Bachler R, Scheer C, Ganz R, Berlemann U, Nolte LP. The first twelve cases of computer assisted periacetabular osteotomy. Comput Aided Surg. 1997;2(6):317–26. https://doi.org/10.1002/(sici)1097-0150(1997)2:6<317::aid-igs1>3.0.co;2-2.
Langlotz F, Bachler R, Berlemann U, Nolte LP, Ganz R. Computer assistance for pelvic osteotomies. Clin Orthop Relat Res. 1998;354:92–102.
Nakahodo K, Sasama T, Sato Y, Sugano N, Ohzono K, Nishii T, Nishihara S, Yonenobu K, Ochi T, Tamura S. Intraoperative update of 3D bone model during computer navigation of pelvic osteotomies using real-time 3D position data. In: Lemke H, Vannier M, Inamura K, Farman A, Doi K, editors. Computer assisted radiology and surgery. 14th International Symposium and Exhibition (CARS 2000), San Francisco, CA, 2000. Amsterdam: Elsevier; 2000. p. 252–6.
Mayman DJ, Rudan J, Yach J, Ellis R. The Kingston periacetabular osteotomy utilizing computer enhancement: a new technique. Comput Aided Surg. 2002;7(3):179–86. https://doi.org/10.1002/igs.10041.
Pflugi S, Liu L, Ecker TM, Schumann S, Larissa Cullmann J, Siebenrock K, Zheng G. A cost-effective surgical navigation solution for periacetabular osteotomy (PAO) surgery. Int J Comput Assist Radiol Surg. 2016;11:271. https://doi.org/10.1007/s11548-015-1267-1.
Murphy RJ, Armiger RS, Lepisto J, Mears SC, Taylor RH, Armand M. Development of a biomechanical guidance system for periacetabular osteotomy. Int J Comput Assist Radiol Surg. 2015;10(4):497–508. https://doi.org/10.1007/s11548-014-1116-7.
Sugano N, Takao M, Sakai T, Nishii T, Miki H. Safety and accuracy of CT-based navigation for rotational acetabular osteotomy. Formosan J Musculoskelet Disord. 2016;7(1):44–50. https://doi.org/10.6492/fjmd.20151007.
Radermacher K, Portheine F, Anton M, Zimolong A, Kaspers G, Rau G, Staudte HW. Computer assisted orthopaedic surgery with image based individual templates. Clin Orthop Relat Res. 1998;354:28–38.
Otsuki B, Takemoto M, Kawanabe K, Awa Y, Akiyama H, Fujibayashi S, Nakamura T, Matsuda S. Developing a novel custom cutting guide for curved peri-acetabular osteotomy. Int Orthop. 2013;37(6):1033–8. https://doi.org/10.1007/s00264-013-1873-x.
Azuma H, Taneda H. Rotational acetabular osteotomy in congenital dysplasia of the hip. Int Orthop. 1989;13(1):21–8.
Kambe T, Naito M, Asayama I, Koga K, Fujisawa M, Yamaguchi T, Yatsunami M. Vascular anatomy for rotational acetabular osteotomy: cadaveric study. J Orthop Sci. 2003;8(3):323–8. https://doi.org/10.1007/s10776-002-0630-7.
Matsui M, Masuhara K, Nakata K, Nishii T, Sugano N, Ochi T. Early deterioration after modified rotational acetabular osteotomy for the dysplastic hip. J Bone Joint Surg. 1997;79(2):220–4.
Xu K, Li YM, Zhang HF, Wang CG, Xu YQ, Li ZJ. Computer navigation in total hip arthroplasty: a meta-analysis of randomized controlled trials. Int J Surg (London, England). 2014;12(5):528–33. https://doi.org/10.1016/j.ijsu.2014.02.014.
Akiyama H, Goto K, So K, Nakamura T. Computed tomography-based navigation for curved periacetabular osteotomy. J Orthop Sci. 2010;15(6):829–33. https://doi.org/10.1007/s00776-010-1520-y.
Hsieh PH, Chang YH, Shih CH. Image-guided periacetabular osteotomy: computer-assisted navigation compared with the conventional technique: a randomized study of 36 patients followed for 2 years. Acta Orthop. 2006;77(4):591–7. https://doi.org/10.1080/17453670610012656.
Inaba Y, Kobayashi N, Ike H, Kubota S, Saito T. Computer-assisted rotational acetabular osteotomy for patients with acetabular dysplasia. Clin Orthop Surg. 2016;8(1):99–105. https://doi.org/10.4055/cios.2016.8.1.99.
Takao M, Nishii T, Sakai T, Sugano N. Comparison of rotational acetabular osteotomy performed with navigation by surgeons with different levels of experience of osteotomies. Int J Comput Assist Radiol Surg. 2017;12:841. https://doi.org/10.1007/s11548-016-1494-0.
Hasegawa Y, Iwase T, Kitamura S, Kawasaki M, Yamaguchi J. Eccentric rotational acetabular osteotomy for acetabular dysplasia and osteoarthritis: follow-up at a mean duration of twenty years. J Bone Joint Surg Am. 2014;96(23):1975–82. https://doi.org/10.2106/jbjs.m.01563.
Hartig-Andreasen C, Troelsen A, Thillemann TM, Soballe K. What factors predict failure 4 to 12 years after periacetabular osteotomy? Clin Orthop Relat Res. 2012;470(11):2978–87. https://doi.org/10.1007/s11999-012-2386-4.
Tannast M, Hanke MS, Zheng G, Steppacher SD, Siebenrock KA. What are the radiographic reference values for acetabular under- and overcoverage? Clin Orthop Relat Res. 2015;473(4):1234–46. https://doi.org/10.1007/s11999-014-4038-3.
Hamada H, Takao M, Nakahara I, Sakai T, Nishii T, Sugano N. Hip range-of-motion (ROM) is less than normal after rotational acetabular osteotomy for developmental dysplasia of the hip: a simulated ROM analysis. J Orthop Res. 2016;34:217. https://doi.org/10.1002/jor.23024.
Nakahara I, Takao M, Sakai T, Miki H, Nishii T, Sugano N. Three-dimensional morphology and bony range of movement in hip joints in patients with hip dysplasia. Bone Joint J. 2014;96-b(5):580–9. https://doi.org/10.1302/0301-620x.96b5.32503.
Nakahara I, Takao M, Sakai T, Nishii T, Yoshikawa H, Sugano N. Gender differences in 3D morphology and bony impingement of human hips. J Orthop Res. 2011;29(3):333–9. https://doi.org/10.1002/jor.21265.
Yasunaga Y, Yamasaki T, Matsuo T, Ishikawa M, Adachi N, Ochi M. Crossover sign after rotational acetabular osteotomy for dysplasia of the hip. J Orthop Sci. 2010;15(4):463–9. https://doi.org/10.1007/s00776-010-1489-6.
Imai H, Kamada T, Takeba J, Shiraishi Y, Mashima N, Miura H. Anterior coverage after eccentric rotational acetabular osteotomy for the treatment of developmental dysplasia of the hip. J Orthop Sci. 2014;19(5):762–9. https://doi.org/10.1007/s00776-014-0592-5.
Turgeon TR, Phillips W, Kantor SR, Santore RF. The role of acetabular and femoral osteotomies in reconstructive surgery of the hip: 2005 and beyond. Clin Orthop Relat Res. 2005;441:188–99.
Sugioka Y. Transtrochanteric anterior rotational osteotomy of the femoral head in the treatment of osteonecrosis affecting the hip: a new osteotomy operation. Clin Orthop Relat Res. 1978;130:191–201.
Sakano S, Hasegawa Y, Torii Y, Kawasaki M, Ishiguro N. Curved intertrochanteric varus osteotomy for osteonecrosis of the femoral head. J Bone Joint Surg Br. 2004;86(3):359–65.
Kitada M, Nakamura N, Iwana D, Kakimoto A, Nishii T, Sugano N. Evaluation of the accuracy of computed tomography-based navigation for femoral stem orientation and leg length discrepancy. J Arthroplasty. 2011;26(5):674–9. https://doi.org/10.1016/j.arth.2010.08.001.
Kitada M, Sakai T, Murase T, Hanada T, Nakamura N, Sugano N. Validation of the femoral component placement during hip resurfacing: a comparison between the conventional jig, patient-specific template, and CT-based navigation. Int J Med Robot Comput Assist Surg. 2013;9(2):223–9. https://doi.org/10.1002/rcs.1490.
Bailey C, Gul R, Falworth M, Zadow S, Oakeshott R. Component alignment in hip resurfacing using computer navigation. Clin Orthop Relat Res. 2009;467(4):917–22. https://doi.org/10.1007/s11999-008-0584-x.
Olsen M, Schemitsch EH. Avoiding short-term femoral neck fracture with imageless computer navigation for hip resurfacing. Clin Orthop Relat Res. 2011;469(6):1621–6. https://doi.org/10.1007/s11999-010-1607-y.
Schnurr C, Michael JW, Eysel P, Konig DP. Imageless navigation of hip resurfacing arthroplasty increases the implant accuracy. Int Orthop. 2009;33(2):365–72. https://doi.org/10.1007/s00264-007-0494-7.
Akiyama H, Kawanabe K, Goto K, Ohnishi E, Nakamura T. Computer-assisted fluoroscopic navigation system for removal of distal femoral bone cement in revision total hip arthroplasty. J Arthroplasty. 2007;22(3):445–8. https://doi.org/10.1016/j.arth.2006.11.010.
Hamelinck HK, Haagmans M, Snoeren MM, Biert J, van Vugt AB, Frolke JP. Safety of computer-assisted surgery for cannulated hip screws. Clin Orthop Relat Res. 2007;455:241–5. https://doi.org/10.1097/01.blo.0000238815.40777.d2.
Liebergall M, Ben-David D, Weil Y, Peyser A, Mosheiff R. Computerized navigation for the internal fixation of femoral neck fractures. J Bone Joint Surg Am. 2006;88(8):1748–54. https://doi.org/10.2106/jbjs.e.00137.
Zhao G, Yamamoto T, Ikemura S, Motomura G, Mawatari T, Nakashima Y, Iwamoto Y. Radiological outcome analysis of transtrochanteric curved varus osteotomy for osteonecrosis of the femoral head at a mean follow-up of 12.4 years. J Bone Joint Surg Br. 2010;92(6):781–6. https://doi.org/10.1302/0301-620x.92b6.23621.
Atsumi T, Kajiwara T, Hiranuma Y, Tamaoki S, Asakura Y. Posterior rotational osteotomy for nontraumatic osteonecrosis with extensive collapsed lesions in young patients. J Bone Joint Surg Am. 2006;88(Suppl 3):42–7. https://doi.org/10.2106/jbjs.f.00767.
Zhao G, Yamamoto T, Ikemura S, Motomura G, Iwasaki K, Yamaguchi R, Nakashima Y, Mawatari T, Iwamoto Y. Clinico-radiological factors affecting the joint space narrowing after transtrochanteric anterior rotational osteotomy for osteonecrosis of the femoral head. J Orthop Sci. 2012;17(4):390–6. https://doi.org/10.1007/s00776-012-0238-4.
Hamanishi M, Yasunaga Y, Yamasaki T, Mori R, Shoji T, Ochi M. The clinical and radiographic results of intertrochanteric curved varus osteotomy for idiopathic osteonecrosis of the femoral head. Arch Orthop Trauma Surg. 2014;134(3):305–10. https://doi.org/10.1007/s00402-013-1919-y.
Sugano N, Takaoka K, Ohzono K, Matsui M, Saito M, Saito S. Rotational osteotomy for non-traumatic avascular necrosis of the femoral head. J Bone Joint Surg Br. 1992;74(5):734–9.
Koyama T, Sugano N, Nishii T, Miki H, Takao M, Sato Y, Yoshikawa H, Tamura S. MRI-based surgical simulation of transtrochanteric rotational osteotomy for femoral head osteonecrosis. J Orthop Res. 2009;27(4):447–51. https://doi.org/10.1002/jor.20568.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Takao, M., Sakai, T., Hamada, H., Sugano, N. (2018). Computer-Assisted Orthopedic Surgery for Hip Osteotomy. In: Sugano, N. (eds) Computer Assisted Orthopaedic Surgery for Hip and Knee. Springer, Singapore. https://doi.org/10.1007/978-981-10-5245-3_12
Download citation
DOI: https://doi.org/10.1007/978-981-10-5245-3_12
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-5244-6
Online ISBN: 978-981-10-5245-3
eBook Packages: MedicineMedicine (R0)