Abstract
In terms of global production, wheat among cereals ranks third after rice and maize, contributing about 35% of the total food grain production. Wheat due to high nutritional value is considered one of the important dietary constituents and, hence, has become one of the better food choices around the world. For growth and development, wheat requires large amounts of major plant nutrients especially phosphorus (P). Application of sufficient amounts of P has many beneficial impacts on wheat including its role in growth, grain formation, and development, and in straw yield. Phosphorus deficiency, however, may adversely affect the growth and, therefore, hampers the physiological processes leading eventually to overall stunting of the plant. In order to circumvent the phosphorus problems and hence to achieve optimum yields, wheat growers usually apply excessive amounts of chemical phosphatic fertilizer which is both expensive and destructive to soil fertility. To overcome these problems, a physiologically versatile array of microorganisms especially belonging to phosphate-solubilizing group has been introduced into the agricultural system for improving wheat production. The P-solubilizing microorganisms (PSM) solubilize unavailable soil P and make it available for uptake by plants. The use of microbial phosphatic fertilizer (microphos) in wheat production system is considered an eco-friendly strategy without adversely affecting the soil health. Despite numerous informations available on the impact of P-solubilizing microorganisms on various plants, literature suggesting the use of PSM in wheat production is limited. Realizing the importance of PSM in enhancing the overall performance of wheat, attempt has been made to better understand as to how the PSM affects wheat production in variable agricultural practices. Also, efforts will be made to find PSM which could be applied to facilitate the growth and development of wheat grown in different agroecological niches. Constant and sustainable application of PSM is expected to decrease the use of fertilizers in wheat production strategies.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
3.1 Introduction
Wheat (Triticum aestivum) is one of the important cereal crops which is extensively cultivated and used as food worldwide. Wheat is cultivated in variable climatic conditions that range between 47°S and 57°N latitudes on different soils including sandy and clayey soil and has the highest adaptation among all the crop species (Naresh et al. 2014). Globally, about 680 million tons of wheat was produced during 2008–2012, while in 2011, the production rate reached to almost 700 million tons (FAO Stat http://faostat.fao.org/site/291/default.aspx). Moreover, the statistics reveal that more than 600 million tons of wheat is harvested annually. Among various wheat-producing countries, India is the second largest wheat-producing country with 11.9% production from approximately 12% of total area (USDA 2010). In India, wheat is cultivated in about 30 million hectares land resulting in 93 million tons yield. The national productivity is estimated as 2.98 tons ha−1. However, despite being an important food crop of the country, the average production of wheat is slowly dwindling due to several reasons (Ray et al. 2012). Chief among them has been the declining cultivable lands, fluctuating environmental conditions (global warming) and excessive usage of chemical fertilizers in order to obtain maximum yields.
The consistently increasing cost of chemical fertilizers and their deleterious impact on soil fertility and human health (via food chain) are some of the vital problems of farmers growing wheat across different regions (Eman et al. 2008; Singh et al. 2008). In order to reduce the use of fertilizers in wheat production, scientists require searching for some other inexpensive alternatives. In this regard, biofertilizers, defined as a biological product containing living microorganisms which, when applied to seed, plant surfaces, or soil, colonize the rhizosphere or the interior of the plant and facilitate growth by enhancing the supply or availability of primary nutrients to the host plant (Vessey 2003), involving PSM that have offered a better alternative to the expensive and environmentally unfriendly fertilizers in wheat production (Sarker et al. 2014; Sharma et al. 2013). The chief benefit of applying PSM in wheat cultivation is their ability to better colonize and establish in the rhizosphere and to make P available to plants constantly (Sharma 2002). Apart from P, PSM also facilitates the growth of plants by providing gibberellins, cytokinins, and IAA (Priya et al. 2013; Sharma et al. 2012), improves uptake of water and nutrients (Abbasniayzare et al. 2012; Khan et al. 2007), secretes antibiotics and other toxic products (Shanmugam et al. 2011; Lipping et al. 2008), and supplies vitamins belonging to β-group (Revillas et al. 2000). Also, PSM improves the wheat growth by inhibiting plant pathogens (Salma et al. 2014). For instance, P-solubilizing bacteria like Pseudomonas and Bacillus species when used alone or in combination profoundly increased grain yield, tiller formation, and seed P of wheat (Afzal et al. 2005). Similarly, the pre-sowing application of PSM inoculated wheat resulted in a considerable increase in yield relative to uninoculated wheat (Dwivedi et al. 2004). These and other similar studies suggest that the sole or combined application of PSM could be used to enhance the overall performance of wheat.
3.2 Nutritional Importance of Wheat
Wheat is one of the main staple food crops for majority of the world population and is the major staple food in many Asian countries. Wheat is an important cereal crop of human dietary systems all over the world. Nutritionally, it is highly rich in carbohydrate and serves as a good source of energy. Interestingly, proteins and fibers are also the major constituents of wheat composition. In addition, wheat also contains significant amounts of vitamins, minerals, lipids, and a few phytochemicals. The nutrient composition of wheat is represented in Table 3.1.
3.3 Role of Phosphorus in Promoting Wheat Growth
Plants in general require variety of nutrient elements for survival and growth. These elements are categorized as macro- and micronutrients depending upon the requirement of various crop plants. Phosphorus among plant nutrients is required in larger quantities by plants, the deficiency of which severely affects the whole metabolism of many plants including wheat. Phosphorus plays an important part in photosynthesis, energy transfer, utilization of sugar and starch, nucleus formation and cell division, signal transduction, macromolecular biosynthesis and respiration (Khan et al. 2010), and N2 fixation (Wibisono et al. 2015). It also initiates root growth and development and maintains the overall health of the plants.
In wheat, P plays a prime role in strengthening of the straw and results in better fruit production (Anonymous 1988). Presence of P also accounts for a better tillering in wheat, promotes early maturation of plant, and assists seed formation. For example, in a study, the combined effect of irrigation and phosphorus demonstrated a positive impact on the developmental stages of wheat. Furthermore, an increase in the number and weight of the grains was recorded. Topical application of P further increased the size of wheat grains (Hossain et al. 1996; Turk and Tawaha 2002). Phosphorus fertilization is therefore a major input in crop production across different regions, because some soils lack sufficient P to optimize crop quality and yields (Griffith 2009). Effective nutrient management, hence, requires that P be available in adequate amounts when needed by the plant (Sarfraz et al. 2009). In order to supply available and soluble form of P to wheat plants, a group of soil microorganisms collectively called as phosphate-solubilizing microorganisms (PSM) have been applied. To substantiate this further, Islam and Hossain (2012) proposed that the use of synthetic phosphatic fertilizer could be reduced if the insoluble soil P is solubilized naturally by PSM and is made available to plants. In some other experiments, P-solubilizing bacteria (PSB) when applied as bioinoculants have been shown to solubilize the fixed soil P, thereby improving the crop yield (Gull et al. 2004) including those of wheat (Afzal and Asghari 2008). In addition to the sole application of PSM, there has been considerable reports where PSM in synergism with other organisms, for example, AM fungi, has been found to enhance uptake of solubilized soil P (Barea et al. 2002) and concurrently increased nutrient uptake and yield of wheat and maize (Raja et al. 2002).
3.4 Rationale for Using Phosphate-Solubilizing Microorganisms in Wheat Production
Phosphorus, one of the major plant nutrients, affects many stages of plant growth and enhances grain yield and yield components. On the contrary, in many agricultural production systems, P has been identified as the most deficient essential nutrient after N. And hence, nutrient supply to agronomic production systems has increased to achieve optimum yields in order to sustain the growing populations demand around the world. When soil is deficient in available P, phosphatic fertilizers are applied to soils. Although inorganic fertilizers are readily available, they are slowly converted to unavailable forms due to precipitation/complexation. Among cereals, wheat requires more nutrients than other crops. Worldwide, the commercial production of fertilizers needs substantial amounts of energy, and, hence, it becomes costly. Moreover, phosphatic fertilizers are consistently used in wheat production to achieve higher yields. The excessive and continued application of phosphatic fertilizers, however, destruct the soil fertility (Younis et al. 2013). High dose of P fertilizers causes abrupt shoot growth, while it limits root growth. Also, following accumulation within soils, P can pollute the ground water resources. Protein digestion inhibitors deposited in plant cell vacuoles were not taken up by sucking herbivores but hampered the chewing herbivores (Mattson 1980). Moreover, the uptake of P by plants is considerably low due to its rapid fixation with Fe and Al oxides in acidic soils (Goldstein 1986; Norrish and Rosser 1983) and calcium in neutral or calcareous soils (Lindsay et al. 1989). Due to these, approximately 75–90% of the P fertilizers applied to soil are lost, and, hence, plants generally suffer from P deficiency. The combined use of the phosphatic fertilizers to maximize the wheat production without experiencing any toxicological hazards is therefore urgently required. In order to overcome the cost of production, abolish the toxic effect of fertilizers, and fulfill the P requirements of wheat, it has become imperative to search for some newer and inexpensive option that could solve such difficulties. To address such problems, the focus is shifted toward the use of PSM both singly (Kumar et al. 2014a) and as mixture with fertilizers (Babana et al. 2013) or as coculture (Upadhyay et al. 2012) to improve soil fertility leading to increase in wheat yields (Zaidi and Khan 2005). When applied properly, PSM in agricultural practices has been found to decrease the use of costly phosphatic fertilizers (Ali et al. 2014; Dalve et al. 2009). For example, Ramlakshmi and Bhrathiraja (2015) in a study conducted for marigold production have suggested that the mixture of Paenibacillus polymyxa (phospho-bacterium) and Glomus fasciculatum (AM fungi) could decrease the application of P fertilizer by 25%. The integrated nutrient management (Chaitra and Patil 2007) which involves the use of PSM carrying variable characteristics has, therefore, motivated wheat growers worldwide (Naqvi and Ahmad 2012; Goes et al. 2012). Phosphate-solubilizing microorganisms also act as biological control agents (Zaidi et al. 2014) and by limiting the phytopathogens increase the performance of plants (Basharat et al. 2011).
3.5 PSM Improves Wheat Production
3.5.1 PSM: Definition, Origin, and Selection of Phosphate-Solubilizing Microorganisms
Phosphate-solubilizing microorganisms are a group of useful microorganisms which hydrolyze organic and inorganic P (Chen et al. 2006b). Numerous PSM have been recovered from non-rhizosphere (Onyia and Anyanwu 2013) and rhizosphere soils (Qiao et al. 2013), rhizoplane (Sarkar et al. 2012), phyllosphere (Mwajita et al. 2013), rock phosphate (RP) deposit area soil (Mardad et al. 2013), marine environment (Mujahid et al. 2014), and polluted soils (Susilowati and Syekhfani 2014). Some of the important P-solubilizing bacteria belongs to genera Achromobacter (Ma et al. 2009 ), Acinetobacter (Gulati et al. 2010), Sphingomonas and Burkholderia (Panhwar et al. 2014; Song et al. 2008), Bacillus (Tallapragada and Usha 2012), Serratia (Selvakumar et al. 2008), Enterobacter (Frank and Julius 2012), Micrococcus (Reena et al. 2013), Pseudomonas (Mehnaz et al. 2010), rhizobia (Kumar et al. 2014; Kenasa et al. 2014), and actinomycetes (Saif et al. 2014). The important P-solubilizing fungi belong to genera Penicillium (Reena et al. 2013), Aspergillus (Coutinho et al. 2012), and Trichoderma (Yasser et al. 2014). However, among various phosphate solubilizers, P-solubilizing fungi (PSF) in general have been found superior P solubilizer compared to PSB (Venkateswarlu et al. 1984). Like any other plant, wheat too represents a habitat for diverse PSM, which colonize the (i) rhizosphere (Majeed et al. 2015; Kundu et al. 2009), (ii) the phyllosphere (Verma et al. 2016a), (iii) PSM living inside tissues (endophytes) (Verma et al. 2016a), and (iv) stressed environment.
Rhizosphere PSM
The region of soil that is directly influenced by root exudates and associated soil microbiota is generally termed as rhizosphere. The term rhizosphere (Greek word “rhizo” meaning root and “sphere” is one field of action, influence, or existence) was introduced by Hiltner in 1904. The rhizosphere is generally rich in rhizodeposition (sloughed-off plant cells), proteins, and sugars released by roots. These exudates support the growth of various microbial communities including PSM. Like many other microbial communities, PSB have been recovered from many crop rhizospheres including those of wheat. Some of them have been identified as Pseudomonas aeruginosa (Kumar et al. 2015), P. fluorescens, P. putida (Zabihi et al. 2011), P. stutzeri (Venieraki et al. 2011), Bacillus (Ogut and Er 2016; Ogut et al. 2011), Lysinibacillus sphaericus, Paenibacillus polymyxa, Staphylococcus succinus, Sporosarcina sp. (Verma et al. 2016b), Azotobacter chrococcum (Kumar and Narula 1999), Thiobacillus sp. (Babana et al. 2016), Vibrio splendidus (Babana et al. 2013), Proteus sp. (Billah and Bano 2015), Azospirillum brasilense (Venieraki et al. 2011), Acinetobacter (Ogut et al. 2010), Stenotrophomonas sp. AJK3 (Majeed et al. 2015), Enterobacter sp., Arthrobacter chlorophenolicus (Kumar et al. 2014a), and Serratia marcescens (Lavania and Nautiyal 2013). Of the filamentous fungi involved in solubilization of insoluble P, Aspergillus niger (Shrivastava and D’Souza 2014), Penicillium bilaii (Ram et al. 2015), Penicillium oxalicum (Xiao et al. 2013), and Mucor ramosissimus (Xiao et al. 2009) are the most important PSF, while strains of Candida krissi (Xiao et al. 2009) have also been identified as P solubilizer which solubilized insoluble P by secreting organic acids.
Phyllosphere PSM
The term phyllosphere refers to the total aboveground portions of plants inhabited by microorganisms (Last 1955; Ruinen 1956). Phosphate-solubilizing microbes in the wheat phyllospheres have been reported and identified using PCR technique. For instance, Verma et al. (2014) isolated wheat-associated epiphytic bacteria from five locations in central zone (one of the wheat agroecological zones) in India. The phosphate-solubilizing bacteria isolated from phyllosphere (N = 89) belonged to genera Arthrobacter, Bacillus, Corynebacterium, Methylobacterium, Paenibacillus, Pseudomonas, and Psychrobacter. Of these, Arthrobacter humicola showed the highest P-solubilizing activity. Other genera identified to species level using 16S rRNA gene sequencing and subsequent molecular phylogeny analysis included Paenibacillus amylolyticus, Bacillus aryabhattai, Methylobacterium extorquens, Methylobacterium mesophilicum, Methylobacterium radiotolerans, Psychrobacter fozii, and Pseudomonas fuscovaginae.
Endophyte PSM
An endosymbiont (bacterium or fungus) often called an endophyte resides inside plant tissues (Hardoim et al. 2008) for longer periods of its life cycle but causes no diseases to plants (Vijayabharathi et al. 2016; Puri et al. 2015 ; Hardoim et al. 2015). Also, the endophytic bacteria improve plant growth and nutrition more efficiently compared to rhizospheric bacteria because they show more intimate relationship with plant tissues. The endophytes have an ecological advantage over epiphyte microbes because they are shielded from unfavorable environmental conditions such as high temperature, salinity, drought, pH, osmotic potentials, and ultraviolet radiation (Seghers et al. 2004). The endophytes generally adhere to root hair zone of apical roots and enter through a crack or damage. Following entry inside, they colonize the differentiation zone and intercellular spaces in epidermis (Raven et al. 2009). After crossing the exodermal barrier, they colonize different regions such as point of entry, deep inside cortex, and the cortical intercellular spaces. The plant tissue type, plant growth stage, and soil fertilizer treatment all contribute to composition of endophyte bacterial community in wheat (Robinson et al. 2015). Like rhizosphere/phyllosphere microbial communities, endophytes also facilitate the growth of plants by various mechanisms (Gaiero et al. 2013) including P-solubilization (Wakelin et al. 2004). There are other studies also which suggest that soil inoculation with P-solubilizing Bacillus spp. can solubilize unavailable soil P and applied P, leading to a better plant development and greater yields (Canbolat et al. 2006). The root endophytes Bacillus, Enterobacter, Micrococcus, and Pseudomonas genera identified by Mbai et al. (2013) have also shown to have potential to solubilize P. In other study, Jha and Kumar (2009) isolated a diazotrophic bacterium identified as Achromobacter xylosoxidans WM234C–3 from surface-sterilized roots and culms of wheat variety Malviya 234 which had a significant P-solubilizing activity. Zinniel et al. (2002) also isolated diazotrophic endophytic bacteria from wheat, whereas the filamentous Actinobacteria and some fungi were observed in wheat plants by Coombs and Franco (2003). Recently, Oteino et al. (2015) reported that majority of the endophytic Pseudomonas strains produced gluconic acid (GA) (14-169 mM) and demonstrated moderate to high P-solubilizing activity (400–1300 mgl−1). Thus, the study of endophytes is important primarily for two reasons – (i) it helps to better understand its ecology and (ii) the bioactive molecules secreted by endophytes facilitate the growth of plants in sustainable agricultural practices. Therefore, concerted efforts should be directed to find some new and potentially exciting endophytes for ultimate use in different agricultural production systems across different ecological niches.
Stress-Tolerant PSM
Phosphate-solubilizing microorganisms in general have regularly been isolated from conventional environment. However, the isolation of PSM from derelict/stressed environment would be advantageous because such stress-tolerant PSM could be beneficial for crops growing in stressed/polluted soils. In this context, various P-solubilizing bacteria have been recovered from wheat growing in disturbed environments, for example, low temperature (Mishra et al. 2011; Verma et al. 2015a), drought (Verma et al. 2014), acidic soil (Verma et al. 2013), and salinity (Egamberdieva et al. 2008; Tiwari et al. 2011) using culture-dependent techniques. Verma et al. (2016a) in a recent investigation assessed the diversity and functional attributes of thermotolerant bacteria hosting leaves, shoots, roots, and rhizospheric soils of wheat growing in the peninsular zone of India. Majority of the isolated genera demonstrated P-solubilizing activity and belonged to genera Achromobacter, Alcaligenes, Arthrobacter, Bacillus, Methylobacterium, Pseudomonas, Rhodobacter, Salmonella, and Staphylococcus. Verma et al. (2016b) in a follow-up experiment recovered the epiphytic bacterial strains identified as B. amyloliquefaciens, A. faecalis, and P. poae from the wheat phyllosphere. In a similar study, A. faecalis and P. poae were isolated from wheat growing at arid land and high-temperature regions and were identified for the first time as epiphytic PGPB (Joo et al. 2005). Similar studies were carried out by Verma et al. (2013) to reveal acidotolerant P-solubilizing bacteria exhibiting other PGP activities from phyllosphere of two varieties of wheat growing in acidic soil in the southern hills zone of India. Among these PSB, Variovorax soli (21.52 ± 1.3 μg P mg−1 day−1) was isolated from wheat var. HD2833 and Methylobacterium sp. (36.35 ± 1 μg P mg−1 day−1) and M. radiotolerans (21.35 ± 1 μg P mg−1 day−1) from wheat var. HW3094. Apart from bacteria and fungi, the P-solubilizing actinomycetes can also survive in extreme environments (e.g., drought, fire), and through their ability to secrete antibiotics and phytohormone-like compounds, they can enhance plant growth. Micromonospora aurantiaca and Streptomyces griseus, for example, have shown the greatest stimulatory effect on wheat due to their P-solubilizing efficiency and plant growth-promoting activities (Hamdali et al. 2008, Jog et al. 2012). This ability of actinomycetes of surviving under extremes of environmental conditions has therefore attracted greater attention toward their use as biological agents in stressful conditions. Stress-tolerant microbial inoculants are required for inoculation under extreme environments like high temperature so that such organisms could survive under such adverse environment while maintaining their plant growth-promoting activities. And hence, the selection of thermotolerant P-solubilizing microorganisms carrying numerous PGP traits could be used to produce inoculants for crops grown in the arid, subarid, high-plateau, and high-temperature zones. Furthermore, considering the variety of PSM’s widely spread in different habitats, there is ample scope to find many more prospective microorganisms from variable environments for eventual transfer to end users/farmers.
3.6 Selection of Phosphate Solubilizers
P-solubilizing microorganisms have been recovered from conventional (Surapat et al. 2013) to derelict soils (Susilowati and Syekhfani 2014) and from rhizosphere (Ranjan et al. 2013) to endophytic (Resende et al. 2014) environment. They have subsequently been used in various agronomic practices with greater positive impact on different crops (Sonmez and Tufenkci 2015) including wheat (Sial et al. 2015) under different production systems. And hence, the use of P-solubilizing organisms in crop production is increasing which is likely to substitute or/decrease the use of phosphatic fertilizers considerably (Adesemoye et al. 2009). Considering the importance of PSM in sustainable crop production, many workers have isolated such beneficial microbes (Ahemad and Khan 2012) employing serial plate dilution technique or enrichment culture method. Generally, the PSM are isolated using media containing insoluble tricalcium phosphate (TCP), and the best suitable and most widely used medium for this purpose has been the Pikovskaya medium (Pikovskaya 1948) (g/l: glucose 10, Ca3 (PO4) 2 5, (NH4)2 SO4 0.5, NaCl 0.2, MgSO4.7H2O 0.1, KCl 0.1, yeast extract 0.5, MnSO4 and FeSO4 trace, and pH 7). Rhizospheric or non-rhizospheric soil samples are diluted serially and spread plated (100 μl) or streaked or spot (10 μl) inoculated on Pikovskaya agar plates or any plates having insoluble P and properly incubated for 5–7 days (bacteria) and 3–5 days (fungi and actinomycetes) at 28 ± 2 °C. Organisms showing PS activities are identified by the appearance of zone of solubilization (clear halo) near microbial growth (Plate 3.1) on TCP/insoluble P supplemented plates. The consistency of this technique is, however, not accepted by many workers since numerous bacterial isolates in other studies have even though failed to yield any visible zone of P-solubilization on agar plates but instead solubilized insoluble P in culture medium. However, considering halo formation as a positive indicator of P-solubilizers, the organisms (colonies) showing halo on plates are picked up and used to determine their capability to solubilize insoluble P under liquid medium. After evaluating their P-solubilizing ability on agar plates and in liquid medium, the PSM are assessed for their in vitro potential to secrete plant growth-promoting bioactive molecules. The most putative strains are then identified to species level employing molecular techniques, for example, 16S rDNA sequencing and phylogenetic method. The organisms identified by biochemical (identified up to genus level only) or molecular methods (identified up to species level) and showing single or many plant growth-promoting activities apart from their intrinsic P-solubilizing activity are selected and checked in pots and fields using seed treatment, seedling dipping, or soil application methods for their final transfer to growers for consequent application in agronomic practices as an inexpensive phosphatic option.
3.7 Phosphate Solubilization: How It Occurs?
The heterogenous microbial populations in different habitat include PSM also. Out of the total P-solubilizing microorganisms, 1–50% is contributed by PSB, whereas only 0.1–0.5% populations account for PSF (Chen et al. 2006a). Both phosphate-solubilizing bacteria and fungi convert the insoluble form of phosphorus (inorganic and organic) into soluble and available form of P which is taken up as a source of P by plants. Broadly, the solubilization/mineralization of inorganic/organic P occurs by one of the three mechanisms: (a) production of organic acids, (b) excretion of H+ions, and (c) synthesis of enzymes (Kapri and Tewari 2010; Arcand and Schneider 2006). Of these, solubilization of inorganic P through the organic acids secreted by PSM (Khan et al. 2014) is one of the most extensively accepted theories of P-solubilization. The organic acids produced by PSM largely include gluconic acid, oxalic acid, ketogluconic acid, citric acid etc. (Table 3.2). The release of organic acids is directly associated with the lowering of pH of the medium (Mardad et al. 2013; Whitelaw 2000; Maliha et al. 2004). The effectiveness of solubilization/mineralization, however, depends on the types and amounts of organic acids/enzymes secreted into the liquid medium. Also, the inherent properties of the organic acids are vital than the whole amount of acids released by P-solubilizers (Scervino et al. 2010). Moreover, the insoluble P is also converted into soluble P without the secretion of OA by microbes (Illmer and Schinner 1992). For example, ammonia assimilation resulting in proton extrusion has been found as an alternative mechanism for P-solubilization (Parks et al. 1990; Ogut et al. 2011). Phosphate solubilization by PSM also occurs by enhancing the process of chelation of cations that are bound to the soil P or by generating soluble compounds with cations linked with insoluble soil P so as to discharge the P into the soil system. Another mechanism of P-solubilization includes the production of enzymes like (a) phosphatases (meant for dephosphorylation of phospho-ester bonds), (b) phytases (responsible for the release of P from phytic acid), and (c) phosphatases (enzymes that cleave the C-P linkage in organophosphonates). The phosphatases and phytases together mediate the P mineralization process. The phosphatases play an important role in releasing the inorganic phosphates through scavenging of phospho-ester bonds, whereas most of the phytases are involved in the cleavage of C-P bonds. Another interesting role attributed to these enzymes is the degradation of phytate, thereby leading to mineralization of organic P present within the soil (Behera et al. 2014).
3.8 How Phosphate Solubilizers Facilitate Wheat Growth
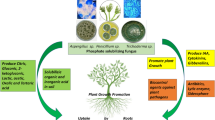
Indeed, the plant growth enhancement by P-solubilizing microorganisms in P-deficient soil occurs greatly through solubilization of insoluble P. The soluble P is then taken up as a major nutrient element by plants. Apart from making soluble P available to plants, the PSM also secretes some important active biomolecules (Table 3.3) that directly or indirectly enhance the growth and productivity of crop plants (Fig. 3.1). Chief among them is the release of siderophores (Ghosh et al. 2015), indole acetic acid (Chitraselvi et al. 2015), and gibberellic acid (Jha and Subramanian 2014). Synthesis of siderophores, an iron-chelating substance by PSB, for instance, Pantoea agglomerans and Burkholderia anthina, may indirectly affect the growth of plants (Datta and Chakrabartty 2014; Walpola and Yoon 2013b, c). Siderophores released by PSB form a complex with iron (Fe3+) in the rhizosphere and limit its availability to the phytopathogens and concomitantly prevent phytopathogens from causing damage to plants. Thus, PSB due their ability to secrete siderophores could be developed as biocontrol agent as well. Secretion of IAA by phosphate solubilizers, for example, Azospirillum, Bacillus, and Enterobacter, (Tahir et al. 2013) is yet another microbiological trait that has shown greater positive impact on overall performance of wheat plants. IAA secreted as a secondary metabolite due to rich supply of substrates by PSB control cell elongation and division, phototropism, and apical dominance in plants (Remnas et al. 2008; Ali et al. 2009). Also, IAA helps in the expansion of roots and increases the number of root hairs and lateral roots which participate in uptake of nutrients from soil (Datta and Basu 2000). Indole acetic acid also inhibits or impedes the abscission of leaves inducing flowering and fruiting (Zhao 2010). Interestingly, phosphate-solubilizing organism, for example, Bacillus, also secretes cyanogenic compounds (Agrawal and Agrawal 2013), and Burkholderia tropica (Tenorio-Salgado et al. 2013) and phosphate-solubilizing actinomycetes Streptomyces spp. (Jog et al. 2014) exhibited antifungal activity which suppress the fungal phytopathogens and indirectly promote the growth of plants (Singh et al. 2014). Among various P-solubilizing bacteria, some bacterial strains, like Stenotrophomonas rhizophila, Enterobacter cloacae etc., have been reported to fix atmospheric nitrogen (Majeed et al. 2015; Singh and Jha 2015). This property of N2 fixation by PSB could be of special interest for growers for soils deficient in both N and P since application of single PSB expressing dual activity of P-solubilization and N2 fixation together are likely to overcome the N and P deficiency in P- and N-deficient soils. Among plant growth regulators, ACC deaminase is an important plant growth regulator that induces metabolic changes and, hence, increases the growth of plants indirectly by hindering/reducing ethylene secretion (Glick et al. 2007; Bal et al. 2013; Magnucka and Pietr 2015). In a recent study, Singh and Jha (2015) isolated a phosphate-solubilizing bacterium Enterobacter cloacae from rhizospheric roots of Aerva javanica carrying various plant growth-promoting activities such as the isolate was able to produce ACC deaminase, display nitrogen fixation ability, synthesize IAA, and secrete ammonia. Considering the multifarious traits of this bacterium, it was suggested that such bacteria could be used as biofertilizers for increasing the production of crops including those growing in salt-stressed environment.
3.9 Examples of Sole and Composite Effects of PSM on Wheat
With ever increasing human populations, there is greater pressure on agriculture to satisfy human food demands across different regions. Indeed, modern agriculture especially green revolution has reduced some of the human problems by producing more and more foods. To achieve optimum yields, practitioners have, however, extensively used agrochemicals including fertilizers and pesticides in agronomic practices. The expensive and injudicious applications of agrochemicals have undoubtedly increased food production, but their use over the years has backfired as well. The excessive application of agrichemicals has resulted in destruction of microbial diversity and consequently the loss of soil fertility. To obviate such threatening problems of soil pollution and to restore soil fertility, the use of inexpensive natural resources, for example, PSM, has been practiced in different production systems in recent times. Wheat is a high P-demanding cereal crop, and at global scale, wheat production suffers from certain problems; one of the key limitations in enhancing wheat production is the inappropriate use of plant nutrients, especially P and K, and the mean P uptake of wheat is about 3.8 kg P/t of grains (Timsina and Connor 2001). The recovery of P by wheat from fertilizers is quite low, and it is estimated that about 15–20% of the applied P is recovered, while the 80–85% P is fixed as insoluble soil P (Rodríguez and Fraga 1999). It is reported that only 0.1% of the total P remains in soluble form which is available for uptake by plants. Constantly increasing costs of synthetic phosphatic fertilizers together with its high complexation ability in soil have warranted the search for alternative and viable means of P nutrition of wheat. In this context, the sole (Agrawal and Pathak 2010) or composite (Saxena et al. 2014) application of PSM have been considered as a suitable and practicable choice for providing soluble P to wheat while reducing dependence on chemical fertilizers (Table 3.4).
3.10 Inoculation Impact of Phosphate-Solubilizing Bacteria on Wheat
Numerous bacterial species identified as PS bacteria have been used as biofertilizer (microbial inoculants) in agricultural practices largely because of their immense ability to improve the availability of applied and soil P (Vessey 2003). In general, the valuable effects of PSB on crop production have been extensively reported (Khan et al. 2007; Zaidi et al. 2009), but the application of PSB as microbial inoculants (biofertilizer) in wheat cultivation is limited. Considering the importance of PSB and lack of sufficient information on the role of PSB in wheat productivity, an attempt is made here to highlight the impact of single or dual culture of PSB in the improvement of wheat grown under different agroecological niches.
Application of PS bacterium Bacillus megaterium var. phosphaticum [M-13] in the presence of P fertilizers greatly increased the grain and straw yield of wheat when grown in pots. Also, an increase of 27.3%–53.3% in number of spikes per square meter was observed in the presence of inoculated PSB strain over control treatment (Bulut 2013). In a follow-up study, Hossain and Sattar (2014) investigated the effect of mineral P fertilizer and phosphate-solubilizing bacteria (Pseudomonas sp. and Klebsiella sp.) used singly or as mixture on the growth, yield, nutrient uptake, and P use efficiency of wheat grown in field soils treated with varying levels of inorganic phosphorus (triple super phosphate) fertilizer. The highest grain and straw yield (2.13 and 2.84 t ha−1) were observed when Pseudomonas sp. and Klebsiella sp. were applied with 15 kg P ha−1 at Pabna and Rajshahi, respectively. Inoculation of Pseudomonas sp. for Pabna and Klebsiella sp. for Rajshahi in the presence of triple super phosphate resulted in better yield and nutrient uptake of wheat and quality of soil compared to other treatments. When used alone, PS bacteria increased the efficiency of P during crop production, and a positive significant correlation was found between yield contributing characters and grain yield of wheat. This study clearly indicated that PSB could solubilize unavailable P to available form and made it available to crops resulting in greater nutrient uptake and yield of wheat.
Similarly, Afzal et al. (2005) found a significant enhancement in grain and biological yield of wheat grown in presence of PSB (Pseudomonas and Bacillus species) used either alone or in combinations. Moreover, a statistically significant improvement in seed P content and tillers per m2 over control was recorded. It was concluded from this study that P-solubilizing microorganisms when used singly or jointly with other organisms showed a significant impact on grain and biological yield, tillers per m2, and seed P content. An increase in straw and grain yields of wheat following interaction between levels of phosphatic fertilizers and PSB inoculations have been reported (Dwivedi et al. 2004). In a similar study, a synergistic relationship between P-solubilizing microorganisms, for example, Pseudomonas striata and Penicillium, and asymbiotic N2 fixer A. chroococcum facilitated a better uptake of poorly soluble P and, consequently, enhanced dry biomass, grain yield, and P uptake of wheat plants (Zaidi and Khan 2005). Later on, Sarker et al. (2014) observed a considerable increment in growth and nutrient uptake of Pseudomonas inoculated wheat plants. Following inoculation with Pseudomonas sp., the dry biomass of shoots increased significantly over uninoculated control. Additionally, the concentration of macronutrients like, N, P, and K in root and shoot tissues were found maximum in inoculated wheat plants. Kumar et al. (2001), in a pot experiment carried out in greenhouse, assayed the survival of P-solubilizing strains of A. chroococcum, including soil isolates and their mutants, in the rhizosphere, and their influence on biological characteristics (growth and root biomass) of three genetically diverse wheat cultivars. Wheat plants inoculated with/without microbial cultures were grown in soils treated with different dose rates of N and P fertilizers. Seeds of wheat bacterized with P-solubilizing and plant hormone producing A. chroococcum displayed superior response relative to uninoculated controls. Furthermore, grain and straw yields were increased significantly by 12.6% and 11.4%, respectively, following inoculation of mutant strains of A. chroococcum over control. The survival of mutant strain of A. chroococcum in the rhizosphere was enhanced by 12–14% as compared to parent soil isolate. Of the mutant strain, strain M37 was found superior for all three varieties and significantly increased grain yield and root biomass by 14% and 11.4%, respectively, over control. In an experiment, the application of P-solubilizing bacteria (Thiobacillus thiooxidans) in combination with fertilizers (Tilemsi rock phosphate) has resulted in a tremendous increase in wheat yields. Formulation of RP fertilizers along with T. thiooxidans AHB411 and T. thiooxidans AHB417 increased the yield up to 33.3% and 11.9%, respectively. Other biological parameters like number of tillers per plant and length of panicle and seed characteristics such as grains per panicle and 1000 grain weight were significantly improved. Mixed inoculation of T. thiooxidans and Bio TRP1 increased the grain yield of wheat by 46%, whereas straw yield was enhanced by 74% relative to control (Babana et al. 2016).
3.11 Response of Wheat to PSF Inoculations
Apart from phosphate-solubilizing bacteria, fungi have also been found as a better P-solubilizing organism (Khan et al. 2010; Yasser et al. 2014), and upon inoculation, they have shown considerable improvement in wheat production (Wahid and Mehana 2000). For instance, Ram et al. (2015) in a recent field experiment determined the effect of seed treatment with PSF, Penicillium bilaii at varying levels of P on growth, P concentration in leaves, and production of wheat. In the absence of P, the single application of PSF profoundly enhanced grain yield by 12.6% over uninoculated control. On the contrary, PSF in the presence of 50% P fertilizer augmented wheat yield which was equal to the application of 100% P but without PSF inoculation. The interaction between PSF inoculation and P levels affected the spike density significantly. When the P levels were 0 and 50%, the spike density increased significantly to about 7% as compared to control, without PSF application. The PS fungus Penicillium bilaii was capable of enhancing the number of grains per spike and grain yield of wheat remarkably when compared with uninoculated treatments. A 3.7% increase in the 1000 grain weight was recorded following PSF application in wheat relative to control. Also, the application of P. bilaii and phosphatic fertilizer together increased the concentration of P both in grains and straw of wheat plants. When measured at 30 DAS, the P content in the leaves of P. bilaii inoculated wheat plants was found to increase. The study in general reflected a growth enhancement in wheat as a result of PSF inoculation as well as application of phosphatic fertilizer (Ram et al. 2015). In a similar experiment performed by Singh and Reddy (2011), the growth of wheat plants was enhanced due to inoculation with Penicillium oxalicum. Penicillium oxalicum in combination with rock phosphate (RP) increased the shoot length by 1.5 times compared to uninoculated plants. Moreover, the dry biomass of shoots and roots of inoculated plants grown in soil treated with rock phosphate was comparatively higher than control. The mixture of P. oxalicum and RP, however, also increased the yield by 42%. The total P content of wheat plants also increased in the presence of P. oxalicum. Generally, the P accumulation within various plant organs like shoot, root, and grains of wheat plants inoculated with mixture of P. oxalicum and RP was almost three times higher than the P accumulated in untreated plants. Apart from increase in P, the phosphatase and phytase activity were also enhanced following P. oxalicum. The overall improvement in the performance of wheat plants was, therefore, attributed to the inoculation of phosphate-solubilizing fungus P. oxalicum which increased the soil available P and concurrently facilitated the growth of wheat plants.
3.12 Influence of Composite Inoculations on Wheat Production
Wheat crop requires a larger quantity of some essential plant nutrients, such as N and P. The deficiency of such nutrient elements restricts the growth of plants severely. And hence, to supply such plant nutrients, inoculation of inexpensive and favorably interacting microorganisms have been found effective and viable. Also, where P is limited, it has been reported that plants inoculated with AM-fungi, either singly or as co-culture with PSM enhanced the uptake of P by wheat plants (Raja et al. 2002). In view of this, Saxena et al. (2014) studied the interactive effect of an AMF, for instance, Glomus etunicatum, and a PSB, Burkholderia cepacia BAM-6, on wheat plants sown in pots having low available P in order to find bioinoculants for semiarid regions. The composite application of G. etunicatum and B. cepacia increased the growth and yield in comparison to the single application of G. etunicatum and B. cepacia. Crop yield was increased by more than 50%, while N concentration was enhanced by 90%, due to the co-inoculation. The root colonization infected by AMF and population of PSB in rhizosphere also increased with time in soil. This study suggested that B. cepacia BAM-6 interacted synergistically with AMF and enhanced the growth and nutrient uptake of wheat plants. Therefore, the mixture of two unrelated organisms could be used as biofertilizer for wheat crop grown in arid to semiarid regions. In other study, Tomar et al. (1998) used various combinations of Azotobacter, AM fungi, PSB, and NPK fertilizers in wheat production practices. The highest (3.80 tons ha−1) grain yield was recorded with dual inoculation of AM fungi and P-solubilizing bacteria in the presence of NPK which was followed by 3.41 tons ha−1 with NPK only and 2.63 tons ha−1 for control. In a similar experiment, the synergistic effects of plant growth-promoting rhizobacteria and an AM fungus G. fasciculatum on plant growth, yield, and nutrient uptake of wheat plants grown under field conditions were assayed by Khan and Zaidi (2007). The tripartite combination of asymbiotic nitrogen fixer A. chroococcum with PS bacterium Bacillus and G. fasciculatum significantly augmented the dry biomass by 2.6-fold compared to control. At 135 days after sowing (DAS), the grain yield of wheat plants bacterized with A. chroococcum, Bacillus sp., and G. fasciculatum was twofold greater in comparison to non-inoculated plants. Grain protein (GP) was maximum (255.2 mg g−1) in plants treated with four cultures namely, A. chroococcum, Bacillus sp., G. fasciculatum, and Penicillium variabile (PSF), while the minimum GP (113.7 mg g–1) was obtained with sole application of G. fasciculatum. The N and P contents were maximum (33.6 and 67.8 mg plant−1, respectively) in wheat plants co-inoculated with A. chroococcum, Bacillus sp., and G. fasciculatum. However, the N and P contents of soil measured at 135 DAS varied among treatments. Use of P. variabile along with single or dual cultures had negative impact on the measured parameters. Percentage root infection, spore density of the AM fungus, populations of A. chroococcum, and P-solubilizing microorganisms were enhanced at 80 DAS. This result demonstrated that the various combinations of PGPR constantly amplified the growth and yield, N and P contents, and grain quality of wheat. In a field study conducted consecutively for 2 years, Kaur and Reddy (2015) used two phosphate-solubilizing bacteria, Pantoea cypripedii (PSB-3) and Pseudomonas plecoglossicida (PSB-5), which were applied singly or as mixture with RP against maize and wheat crops, and their impact was compared with chemical P fertilizer (diammonium phosphate, DAP). Application of PSB along with RP improved the shoot height, shoot and root dry matter, grain yield, and total P concentration in both maize and wheat crops in comparison to the other treatments. Available soil P, enzyme activities, and PSB populations in both maize and wheat rhizosphere were significantly increased due to inoculation of PSB X RP fertilization relative to DAP application. The mixed application of PSB and RP was found more economical, and, hence, it was suggested that the composite application of PSB and RP would be a suitable alternative to phosphatic fertilizer in sustainable production of wheat. The composite culture of phosphate-solubilizing bacterial strains Pseudomonas fluorescens (BAM-4) and B. cepacia (BAM-12) and Glomus etunicatum enhanced the shoot and root dry biomass and grain yields of wheat plants relative to the uninoculated plants (Saxena et al. 2013). The solubilization of insoluble P is carried out effectively by PSB, whereas the process of P uptake by plant roots is attributed to AM fungi which assist the transportation of solubilized P through plant roots. A composite inoculation of PSB and AM fungus showed better growth and yield of wheat plants in comparison to the plants inoculated with single microbial culture (Minaxi et al. 2013). Also, the AM fungi enhance the P uptake of plants by enhancing the contact surface and volume of soil (Clark and Zeto 2000).
3.13 Inoculation Effects of Immobilized Culture on Wheat Production
Immobilization of bacterial cells has commonly been used in agriculture, pharmaceutical, food, and other industries to obtain a defensive structure or a capsule that could allow immobilization, protection, release, and function of active ingredients. And hence, bacterial cells face little challenge from adverse environmental conditions since encapsulation helps bacterial cells to stabilize and enhance their viability and stability during production, storage, and handling of cultures. Also, encapsulation provides extra protection to bacterial cells during rehydration and lyophilization. In addition, the use of microbial cultures into soil has shown that some microbial inoculants can augment plant uptake of nutrients and consequently increase the use efficiency of applied chemical fertilizers (Adesemoye and Kloepper 2009). In this context, rhizobacteria can play an important role in creating a suitable system for crop production. However, the application of free-living PSB into soil is difficult because it is not easy to maintain the survivability of cells around plant roots largely because they are highly susceptible to environmental variables, for example, temperature, humidity, and stressor molecules. The variation in PSB effect on plants is chiefly due to the differences in the quality of microbial inoculants. Due to these, the efforts should be directed to find an adequate formulation that could be developed as a commercial inoculants product. Considering these, Schoebitz et al. (2013) evaluated the P-solubilizing ability of rhizobacteria using RP as insoluble P and the assimilation of soluble P by wheat plants in quartz sand potted experiments. For this, two P-solubilizing bacteria such as P. fluorescens and Serratia sp. were encapsulated in sodium alginate and potato starch beads. They were further tested for enzyme activity (alkaline and acid phosphatase) and P-solubilization in Pikovskaya liquid medium. A considerable decrease in pH was obtained following P-solubilization. A total of 89 and 93 μg P ml−1 was solubilized by immobilized P-solubilizing bacteria, which was significantly greater than those observed for autoclaved alginate-starch beads. An appreciable increase of 64% in P uptake by wheat plants was observed after 60 days of growth when wheat plants were treated with immobilized P. fluorescens + 3.25 ppm of P. This finding suggests that use of the immobilized rhizobacteria could be a viable option for increasing the P level in wheat grown in different agroecological niches.
Conclusion
The phosphate-solubilizing microorganisms are a boon to the agricultural system. It is indeed an inexpensive and an environmentally friendly strategy to reduce the use of chemical fertilizers in farming practices. The enhancement in biological and chemical properties of wheat plants has been reported due to inoculation with variety of phosphate solubilizers including bacteria, fungi, actinomycetes, and mycorrhizae etc. The yield and other growth parameters of wheat have been enhanced, in general, following inoculation with PSM when used singly or as mixture with other free-living PGPR/AM fungi. Another positive aspect of using these microorganisms is that the health of soil is not compromised at any stage of plant growth. Thus, the phosphate-solubilizing microorganisms in general are considered a useful soil microflora which could be developed at commercial scale as bioinoculants for enhancing the production of wheat while reducing dependence on chemical fertilizer.
References
Abaid-Ullah M, Hassan MN, Jamil M, Brader G, Shah MKN, Sessitsch A, Hafeez FY (2015) Plant growth promoting rhizobacteria: an alternate way to improve yield and quality of wheat (Triticum aestivum). Int J Agric Biol 17:51–60
Abbasniayzare SK, Sedaghathoor S, Dahkaei MNP (2012) Effect of biofertilizer application on growth parameters of Spathiphyllum illusion. Am Eurasian J Agric Environ Sci 12:669–673
Adesemoye AO, Torbert HA, Kloepper JW (2009) Plant growth-promoting rhizobacteria allow reduced application rates of chemical fertilizers. Microb Ecol 58:921–929
Adesemoye AO, Kloepper JW (2009) Plant–microbes interactions in enhanced fertilizer-use efficiency. Appl Microbiol Biotechnol 85:1–12
Afzal A, Asghari B (2008) Rhizobium and phosphate solubilizing bacteria improve the yield and phosphorus uptake in wheat (Triticum aestivum). Int J Agric Biol 10:85–88
Afzal A, Ashraf M, Asad SA, Farooq M (2005) Effect of phosphate solubilizing microorganisms on phosphorus uptake, yield and yield traits of wheat (Triticum aestivum L.) in rainfed area. Int J Agric Biol 7:207–209
Agrawal S, Pathak RK (2010) Response of phosphate solubilizing microorganism on quality of wheat (Triticum aestivum L.) plant grown conventionally in temperate climate. J Biofertil Biopestici 2:2
Agrawal DPK, Agrawal S (2013) Characterization of Bacillus sp. strains isolated from rhizosphere of tomato plants (Lycopersicon esculentum) for their use as potential plant growth promoting rhizobacteria. Int J Curr Microbiol App Sci 2:406–417
Ahemad M, Khan MS (2012) Effect of fungicides on plant growth promoting activities of phosphate solubilizing Pseudomonas putida isolated from mustard (Brassica campestris) rhizosphere. Chemosphere 86(9):945–950
Ali A, Tahir M, Rashid H, Ajmal B, Sajjad RN, Adeel A (2014) Investigation of biofertilizers influence on vegetative growth, flower quality, bulb yield and nutrient uptake in gladiolus (Gladiolus grandiflorus L.) Intern J Plant Anim Environ Sci 4:94–99
Ali B, Sabri AN, Ljung K, Hasnain S (2009) Quantification of indole-3-acetic acid from plant associated Bacillus spp. and their phytostimulatory effect on Vigna radiata (L.) World J Microbiol Biotechnol 25:519–526
Amara U, Wang YX, Cui XL, Khalid R, Ali S, Shabbir G, Hayat R (2015) Screening and identification of soil bacteria for growth promotion of wheat (Triticum aestivum L.) J Bio Env Sci 7(3):87–99
Amraei B, Ardakani MR, Rafiei M, Paknejad F, Rejali F (2015) Effect of Mycorrhiza and Azotobacter on concentration of macroelements and root colonization percentage in different cultivars of wheat (Triticum aestivum L). Biol Forum An Int J 7:895–900
Anonymous (1988) Better crops with plant food. PPI, Atlanta, pp 26
Arcand MM, Schneider KD (2006) Plant- and microbial-based mechanisms to improve the agronomic effectiveness of phosphate rock: a review. An Acad Bras Cienc 78:791–807
Babana AH, Antoun H, Dicko AH, Maïga K, Traoré D (2012) Effect of Pseudomonas sp. on wheat roots colonization by mycorrhizal fungi and phosphate-solubilizing microorganisms, wheat growth and P-uptake. Int J Microbiol 1(1):1–7
Babana AH, Dicko AH, Maïga K, Traoré D (2013) Characterization of rock phosphate-solubilizing microorganisms isolated from wheat (Triticum aestivum L.) rhizosphere in Mali. J Microbiol Res 1(1):1–6
Babana AH, Kassogué A, Dicko AH, Maîga K, Samaké F, Traoré D, Fané R, Faradji FA (2016) Development of a biological phosphate fertilizer to improve wheat (Triticum aestivum L.) production in Mali. Procedia Eng 138:319–324
Bal HB, Das S, Dangar TK, Adhya TK (2013) ACC deaminase and IAA producing growth promoting bacteria from the rhizosphere soil of tropical rice plants. J Basic Microbiol 53:972–984
Barea JM, Toro M, Orozco MO, Campos E, Azcón R (2002) The application of isotopic 32P and 15N-dilution techniques to evaluate the interactive effect of phosphate-solubilizing rhizobacteria, mycorrhizal fungi and Rhizobium to improve the agronomic efficiency of rock phosphate for legume crops. Nutr Cycl Agroecosyst 63:35–42
Basharat N, Sobita S, Vijay SR (2011) Effect of Pseudomonas fluorescens on Fusarium oxysporum f. sp. gladioli causing cormrot disease of gladiolus. J Plant Dis Sci 6:51–53
Behera BC, Singdevsachan SK, Mishra RR, Dutta SK, Thatoi HN (2014) Diversity, mechanism and biotechnology of phosphate solubilizing microorganism in mangrove- a review. Biocatal Agric Biotechnol 3(2):97–110
Billah M, Bano A (2015) Role of plant growth promoting rhizobacteria in modulating the efficiency of poultry litter composting with rock phosphate and its effect on growth and yield of wheat. Waste Mgmt Res 33:63–72
Bulut S (2013) Evaluation of yield and quality parameters of phosphorous-solubilizing and N-fixing bacteria inoculated in wheat (Triticum aestivum L.) Turk J Agric For 37:545–554
Canbolat MY, Bilen S, Cakmakci RS, Ahin F, Aydın A (2006) Effect of plant growth-promoting bacteria and soil compaction on barley seedling growth, nutrient uptake, soil properties and rhizosphere microflora. Biol Fertil Soils 42:350–357
Chaitra R, Patil VS (2007) Integrated nutrient management studies in China aster (Callistephus chinensis Nees) cv. Kamini Karnataka. J Agric Sci 20:689–690
Chen YP, Rekha PD, Arunshen AB, Lai WA, Young CC (2006a) Phosphate solubilizing bacteria from subtropical soil and their tricalcium phosphate solubilizing abilities. Appl Soil Ecol 34:33–41
Chen BD, Zhu YG, Smith FA (2006b) Effect of arbuscular mycorrhizal inoculation on uranium and arsenic accumulation by Chinese brake fern (Pteris vittata L.) from uranium mining impacted soil. Chemosphere 62:1464–1473
Chitraselvi RPE, Kalidass S, Kant R (2015) Efficiency of rhizosphere bacteria in production of indole acetic acid, siderophore and phosphate solubilization. Int J Chem Tech Res 7(6):2557–2564
Clark RB, Zeto SK (2000) Mineral acquisition by arbuscular mycorrhizal plants. J Pl Nutr 23:867–902
Coombs JT, Franco CMM (2003) Isolation and identification of actinobacteria from surface-sterilized wheat roots. Appl Environ Microbiol 69:5603–5608
Coutinho FP, Felix WP, Yano-Melo AM (2012) Solubilization of phosphates in vitro by Aspergillus spp. and Penicillium spp. Ecol Eng 42:85–89
Dalve PD, Mane SV, Nimbalkar RR (2009) Effect of biofertilizers on growth, flowering and yield of gladiolus. Asian J Hortic 4:227–229
Datta C, Basu P (2000) Lndole acetic acid production by a Rhizobium species from root nodules of a leguminous shrub Cajanus cajan. Microbiol Res 155:123–127
Datta B, Chakrabartty PK (2014) Siderophore biosynthesis genes of Rhizobium sp. isolated from Cicer arietinum L. 3 Biotech 4:391–401
Dwivedi BS, Singh VK, Dwivedi V (2004) Application of phosphate rock, with or without Aspergillus awamori inoculation, to meet phosphorus demands of rice–wheat systems in the indo–Gangetic Plains of India. Austr J Exptl Agric 44:1041–1050
Egamberdieva D, Kamilova F, Validov S, Gafurova L, Kucharova Z, Lugtenberg B (2008) High incidence of plant growth-stimulating bacteria associated with the rhizosphere of wheat grown on salinated soil in Uzbekistan. Environ Microbiol 10:1–9
Eman AA, Monem AE, Saleh MMS, Mostafa EAM (2008) Minimizing the quantity of mineral nitrogen fertilization grapevine by using humic acid organic and biofertilizers. Res J Agric Sci 4:46–50
Frank O, Julius O (2012) Some characteristics of a plant growth promoting Enterobacter sp. isolated from the roots of maize. Adv Microbiol 2:368–374
Gaiero JR, Mc Call CA, Thompson KA, Day NJ, Best AS, Dunfield KE (2013) Inside the root microbiome: bacterial root endophytes and plant growth promotion. Amer J Bot 100:1738–1750
Ghosh P, Rathinasabapathi B, Ma LQ (2015) Phosphorus solubilization and plant growth enhancement by arsenic-resistant bacteria. Chemosphere 134:1–6
Glick BR, Todorovic B, Czarny J, Cheng Z, Duan J, McConkey B (2007) Promotion of plant growth by bacterial ACC deaminase. Crit Rev Plant Sci 26:227–242
Goes KCGPD, Cattelan AJ, De Carvalho CGP (2012) Biochemical and molecular characterization of high population density bacteria isolated from sunflower. J Microbiol Biotechnol 22:437–447
Goldstein AH (1986) Bacterial mineral phosphate solubilization: historical perspective and future prospects. Am J Alternat Agric 1:57–65
Griffith DB (2009) Efficient fertilizer use – phosphorus. Sci Agric 433:23–67
Gulati A, Sharma N, Vyas P, Sood S, Rahi P, Pathania V, Prasad R (2010) Organic acid production and plant growth promotion as a function of phosphate solubilization by Acinetobacter rhizosphaerae strain BIHB 723 isolated from the cold deserts of the trans-Himalayas. Arch Microbiol 192:975–983
Gull M, Hafeez FY, Saleem M, Malik KA (2004) Phosphorus uptake and growth promotion of chickpea by co-inoculation of mineral phosphate solubilising bacteria and a mixed rhizobial culture. Aust J Exp Agric 44:623–628
Hamdali H, Hafidi M, Virolle MJ, Ouhdouch Y (2008) Growth promotion and protection against damping-off of wheat by two rock phosphate solubilizing actinomycetes in a P-deficient soil under greenhouse conditions. Appl Soil Ecol 40(3):510–517
Haque MA, Sattar MA, Islam MR, Hashem MA, Khan MK (2013) Performance of phosphate solubilizing bacteria with various phosphorus levels on wheat in pot culture. J Environ Sci Nat Res 6:221–226
Hardoim PR, van Overbeek LS, Berg G, Pirttilä AM, Compant S, Campisano A, Döring M, Sessitsch A (2015) The hidden world within plants: ecological and evolutionary considerations for defining functioning of microbial endophytes. Microbiol Mol Biol Rev 79:293–320
Hardoim PR, Van Overbeek LS, Van Elsas JD (2008) Properties of bacterial endophytes and their proposed role in plant growth. Trends Microbiol 16:463–471
Herrera SD, Grossi C, Zawoznik M, Groppa MD (2016) Wheat seeds harbour bacterial endophytes with potential as plant growth promoters and biocontrol agents of Fusarium graminearum. Microbiol Res 186:37–34
Hossain MA, Begum S, Rahman AKMM, Arabinda S, Salahuddin ABM (1996) Growth analysis of mustard and rapeseed in relation to grain filling period and yield potential. J Agric Res 34:59–369
Hossain MB, Sattar MA (2014) Effect of inorganic phosphorus fertilizer and inoculants on yield and phosphorus use efficiency of wheat. J Environ Sci Natural Res 7:75–79
Illmer P, Schinner F (1992) Solubilization of inorganic phosphates by microorganisms isolated from forest soil. Soil Biol Biochem 24:389–395
Islam MT, Hossain MM (2012) Plant probiotics in phosphorus nutrition in crops, with special reference to Rice. In: Maheshwari DK (ed) Bacteria in agrobiology: plant probiotics. Springer, Berlin/Heidelberg, pp 325–363
Jha P, Kumar A (2009) Characterization of novel plant growth promoting endophytic bacterium Achromobacter xylosoxidans from wheat plant. Microb Ecol 58:179–188
Jha Y, Subramanian RB (2014) Characterization of root-associated bacteria from paddy and its growth promotion efficacy. 3 Biotech 4:325–330
Jog R, Nareshkumar G, Rajkumar S (2012) Plant growth promoting potential and soil enzyme production of the most abundant Streptomyces spp. from wheat rhizosphere. J Appl Microbiol 113:1154–1164
Jog R, Pandya M, Nareshkumar G, Rajkumar S (2014) Mechanism of phosphate solubilization and antifungal activity of Streptomyces spp. isolated from wheat roots and rhizosphere and their application in improving plant growth. Microbiol 160:778–788
Joo HS, Hirai M, Shoda M (2005) Characteristics of ammonium removal by heterotrophic nitrification-aerobic denitrification by Alcaligenes faecalis no. 4. J Biosci Bioeng 100:184–191
Kang SM, Radhakrishnan R, You YH, Joo GJ, Lee IJ, Lee KE, Kim JH (2014) Phosphate solubilizing Bacillus megaterium mj 1212 regulates endogenous plant carbohydrates and amino acid contents to promote mustard plant growth. Indian J Microbiol 54:427–433
Kapri A, Tewari L (2010) Phosphate solubilization potential and phosphatase activity of rhizospheric Trichoderma spp. Braz J Microbiol 41:787–795
Kaur G, Reddy MS (2015) Effects of phosphate-solubilizing bacteria, rock phosphate and chemical fertilizers on maize-wheat cropping cycle and economics. Pedosphere 25:428–437
Kenasa G, Jida M, Assefa F (2014) Characterization of phosphate solubilizing faba bean (Vicia faba L.) nodulating rhizobia isolated from acidic soils of Wollega, Ethiopia. Sci Technol Arts Res J 3:11–17
Khan MS, Zaidi A, Ahemad M, Oves M, Wani PA (2010) Plant growth promotion by phosphate solubilizing fungi – current perspective. Arch Agron Soil Sci 56:73–98
Khan MS, Zaidi A (2007) Synergistic effects of the inoculation with plant growth-promoting rhizobacteria and an arbuscular mycorrhizal fungus on the performance of wheat. Turk J Agric For 31:355–362
Khan MS, Zaidi A, Wani PA (2007) Role of phosphate solubilizing microorganisms in sustainable agriculture: a review. AgronSustain Dev 27:29–43
Khan MS, Zaidi A, Ahmad E (2014) Mechanism of phosphate solubilization and physiological functions of phosphate solubilizing microorganisms. In: Phosphate solubilising microorganisms: principles and application of microphos technology. Springer, Switzerland; p 31–62
Kumar A, Maurya BR, Raghuwanshi R (2014a) Isolation and characterization of PGPR and their effect on growth, yield and nutrient content in wheat (Triticum aestivum L.) Biocatal Agric Biotechnol 3:121–128
Kumar A, Shukla UK, Singh A, Poonam AK, Prasad S, Singh SK, Kumar D (2014) Evaluation of Pseudomonas isolates from wheat for some important plant growth promoting traits. Afr J Microbiol Res 8:2604–2608
Kumar A, Maurya BR, Raghuwanshi R (2015) Characterization of bacterial strains and their impact on plant growth promotion and yield of wheat and microbial populations of soil. Afr J Agri Res 10:1367–1375
Kumar V, Narula N (1999) Solubilization of inorganic phosphates and growth emergence of wheat as affected by Azotobacter chroococcum mutants. Biol Fertil Soils 28(3):301–305
Kumar V, Behl RK, Narula N (2001) Establishment of phosphate-solubilizing strains of Azotobacter chroococcum in the rhizosphere and their effect on wheat cultivars under green house conditions. Microbiol Res 156:87–93
Kundu BS, Nehra K, Yadav R, Tomar M (2009) Biodiversity of phosphate solubilizing bacteria in rhizosphere of chickpea, mustard and wheat grown in different regions of Haryana. Indian J Microbiol 49:120–127
Last FT (1955) Seasonal incidence of Sporobolomyces on cereal leaves. Trans Br Mycol Soc 38:221–239
Lavania M, Nautiyal CS (2013) Solubilization of tricalcium phosphate by temperature and salt tolerant Serratia marcescens NBRI1213 isolated from alkaline soils. Afr J Microbiol Res 7:4403–4413
Li RX, Cai F, Pang G, Shen QR, Li R, Chen W (2015) Solubilisation of phosphate and micronutrients by Trichoderma harzianum and its relationship with the promotion of tomato plant growth. PLoS One 10(6):e0130081. doi:10.1371/journal.pone.0130081
Lindsay WL, Vlek PLG, Chien SH (1989) Phosphate minerals. In: Dixon JB, Weed SB, (eds) Soil environment, 2nd ed. Soil Science Society of America, Madison, pp 1089–1130
Lipping Y, Jiatao X, Daohong J, Yanping F, Guoqing L, Fangcan L (2008) Antifungal substances produced by Penicillium oxalicum strain PY-1 potential antibiotics against plant pathogenic fungi. World J Microbiol Biotechnol 24:909–915
Ma Y, Rajkumar M, Freitas H (2009) Inoculation of plant growth promoting bacterium Achromobacter xylosoxidans strain Ax10 for the improvement of copper phytoextraction by Brassica juncea. J Environ Manag 90:831–837
Magnucka EG, Pietr SJ (2015) Various effects of fluorescent bacteria of the genus Pseudomonas containing ACC deaminase on wheat seedling growth. Microbiol Res 181:112–119
Majeed A, Abbasi MK, Hameed S, Imran A, Rahim N (2015) Isolation and characterization of plant growth-promoting rhizobacteria from wheat rhizosphere and their effect on plant growth promotion. Front Microbiol 6:1–10
Maliha R, Samina K, Najma A, Sadia A, Farooq L (2004) Organic acids production and phosphate solubilization by phosphate solubilizing microorganisms under in vitro conditions. Pak J Biol Sci 7:187–196
Mardad I, Serrano A, Soukri A (2013) Solubilization of inorganic phosphate and production of organic acids by bacteria isolated from a Moroccan mineral phosphate deposit. Afr J Microbiol Res 7:626–635
Marra LM, Longatti SMO, Soares CRFS, Lima JM, Olivares FL, Moreira FMS (2015) Initial pH of medium affects organic acid production but do not affect phosphate solubilization. Braz J Microbiol 46:367–375
Mattson WJ Jr (1980) Herbivory in relation to plant nitrogen content. Annu Rev Ecol Syst 11:119–161
Mbai FN, Magiri EN, Matiru VN, Nganga J, Nyambati VCS (2013) Isolation and characterisation of bacterial root endophytes with potential to enhance plant growth from kenyan basmati rice. Amer Intern J Contem Res 3:25–40
Mehnaz S, Baig DN, Lazarovits G (2010) Genetic and phenotypic diversity of plant growth promoting rhizobacteria isolated from sugarcane plants growing in Pakistan. J Microbiol Biotechnol 20:1614–1623
Mihalache G, Zamfirache MM, Mihasan M, Ivanov I, Stefan M, Raus L (2015) Phosphate solubilizing bacteria associated with runner bean rhizosphere. Arch Biol Sci 67:793–800
Minaxi SJ, Chandra S, Nain L (2013) Synergistic effect of phosphate solubilizing rhizobacteria and arbuscular mycorrhiza on growth and yield of wheat plants. J Soil Sci Pl Nutr 13:511–525
Mishra PK, Bisht SC, Ruwari P, Selvakumar G, Joshi GK, Bisht JK, Bhatt JC, Gupta HS (2011) Alleviation of cold stress in inoculated wheat (Triticum aestivum L.) seedlings with psychrotolerant pseudomonads from NW Himalayas. Arch Microbiol 193:497–513
Moreira FS, Costa PB, Rd S, Beneduzi A, Lisboa BB, Vargas LK, Passaglia LM (2016) Functional abilities of cultivable plant growth promoting bacteria associated with wheat (Triticum aestivumL.) crops. Genet Mol Biol 39:111–121
Mujahid TY, Siddiqui K, Ahmed R, Kazmi SU, Ahmed N (2014) Isolation and partial characterization of phosphate solubilizing bacteria isolated from soil and marine samples. Pak J Pharm Sci 27:1483–1490
Mwajita MR, Murage H, Tani A, Kahangi EM (2013) Evaluation of rhizosphere, rhizoplane and phyllosphere bacteria and fungi isolated from rice in Kenya for plant growth promoters. SpringerPlus 2:606
Naqvi SDY, Ahmad S (2012) Effect of Pseudomonas fluorescens on Fusarium oxysporum f.sp. gladioli causing corm rot disease of gladiolus. J Stored Prod Postharvest Res 3:49–51
Naresh R, Tomar S, Purushottam S, Kumar D, Pratap B, Kumar V, Nanher A (2014) Testing and evaluation of planting methods on wheat grain yield and yield contributing parameters in irrigated agro-ecosystem of western Uttar Pradesh, India. Afr J Agric Res 9:176–182
Narula N, Kumar V, Behl RK (2005) Effect of phosphate solubilizing strains of Azotobacter chroococcum on yield traits and their survival in the rhizosphere of wheat genotypes under field conditions. Acta Agronomica Hungarica 49:141–149
Nath R, Sharma GD, Barooah M (2012) Efficiency of tricalcium phosphate solubilization by two different Endophytic Penicillium sp. isolated from tea (Camellia sinensis L.) Eur J Exp Biol 2:1354–1358
Norrish K, Rosser H (1983) Mineral phosphate soils: an Australianviewpoint. Sponsored by the Division of Soils, Commonwealth Scientific and Industrial Research Organization. Academic Press/CSIRO, Melbourne /London . pp 335–361
Ogut M, Er F (2016) Mineral composition of field grown winter wheat inoculated with phosphorus solubilizing bacteria at different plant growth stages. J Plant Nutr 39:479–490
Ogut M, Er F, Neumann G (2011) Increased proton extrusion of wheat roots by inoculation with phosphorus solubilising microorganims. Plant Soil 339:285–297
Ogut M, Er F, Kandemir N (2010) Phosphate solubilization potentials of soil Acinetobacter strains. Biol Fertil Soils 46:707–715
Onyia CE, Anyanwu CU (2013) Comparative study on solubilization of tri-calcium phosphate (TCP) by phosphate solubilizing fungi (PSF) isolated from Nsukka pepper plant rhizosphere and root free soil. J Yeast Fungal Res 4:52–57
Oteino N, Lally RD, Kiwanuka S, Lloyd A, Ryan D, Germaine KJ, Dowling DN (2015) Plant growth promotion induced by phosphate solubilizing endophytic Pseudomonas isolates. Front Microbiol 6:745. doi:10.3389/fmicb.2015.00745
Padmavathi T (2015) Optimization of phosphate solubilization by Aspergillus niger using plackett-burman and response surface methodology. J Soil Sci Pl Nutr 15(3):781–793
Panhwar QA, Naher UA, Jusop S, Othman R, Latif MA, Ismail MR (2014) Biochemical and molecular characterization of potential phosphate solubilizing bacteria in acid sulphate soils and their beneficial effects on rice growth. PLoS One 9 : PMC4186749; e97241
Parks EJ, Olson GJ, Brinckman FE, Baldi F (1990) Characterization by high performance liquid chromatography (HPLC) of the solubilization of phosphorus in iron ore by a fungus. J Ind Microbiol Biotechnol 5:183–189
Pikovskaya RI (1948) Mobilization of phosphorus in soil in connection with vital activity of some microbial species. Microbiologiya 17:362–370
Priya S, Panneerselvam T, Sivakumar T (2013) Evaluation of indole-3-acetic acid in phosphate solubilizing microbes isolated from rhizosphere soil. Int J Curr Microbiol App Sci 2:29–36
Promwee A, Issarakraisila M, Intana W, Chamswarng C, Yenjit P (2014) Phosphate solubilization and growth promotion of rubber tree (Hevea brasiliensis Muell. Arg.) by Trichoderma strains. J Agric Sci 6:8. doi:10.5539/jas.v6n9p8
Puri A, Padda KP, Chanway CP (2015) Can a diazotrophic endophyte originally isolated from lodgepole pine colonize an agricultural crop (corn) and promote its growth? Soil Biol Biochem 89:210–216
Qiao ZW, Hong JP, Xie YH, Li LX (2013) Screening, identification and phosphate-solubilizing characteristics of Rahnella sp. phosphate-solubilizing bacteria in calcareous soil. Ying Yong Sheng Tai Xue Bao 24:2294–2300
Raja AK, Shah KH, Aslam M, Memon MY (2002) Response of phosphobacterial and mycorrhizal inoculation in wheat. Asian J Plant Sc 1:322–323
Rajput L, Imran A, Mubeen F, Hafeez FY (2013) Salt tolerant PGPR strain Planococcus rifietoensis promotes the growth and yield of wheat (Triticum aestivum L.) cultivated in saline soil. Pak J Bot 45:1955–1962
Ram H, Malik SS, Dhaliwal SS, Kumar B, Singh Y (2015) Growth and productivity of wheat affected by phosphorus-solubilizing fungi and phosphorus levels. Plant Soil Environ 61:122–126
Ramesh A, Sharma SK, Sharma MP, Yadav N, Joshi OP (2014) Plant growth-promoting traits in Enterobacter cloacae subsp. dissolvens MDSR9 isolated from soybean rhizosphere and its impact on growth and nutrition of soybean and wheat upon inoculation. Agri Res 3:53–66
Ramlakshmi R, Bharathiraja S (2015) AM fungi and phosphate solubilizing bacteria (Paenibacillus polymyxa) as a potential bioinoculant for marigold. Intern J Curr Res 7:12264–12266
Rana A, Joshi M, Prasanna R, Shivay YS, Nain L (2012) Biofortification of wheat through inoculation of plant growth promoting rhizobacteria and cyanobacteria. Eur J Soil Biol 50:118–126
Ranjan A, Mahalakshmi M, Sridevi M (2013) Isolation and characterization of phosphate solubilizing bacterial species from different crop fields of Salem, Tamil Nadu, India. Intern J Nutr, Pharmacol, Neurol dis 3:29–33
Raven JA, Beardall J, Flynn KJ, Maberly SC (2009) Phagotrophy in the origins of photosynthesis in eukaryotes and as complementary mode of nutrition in phototrophs: relation to Darwin’s insectivorous plants. J Exp Bot 60:3975–3987
Ray DK, Ramankutty N, Mueller ND, West PC, Foley JA (2012) Recent patterns of crop yield growth and stagnation. Nat Commun 3:1293
Reena TD, Deepthi H, Pravitha MS, Lecturer D (2013) Isolation of phosphate solubilizing bacteria and fungi from rhizospheres soil from banana plants and its effect on the growth of Amaranthus cruentus L. IOSR J Pharmacy Biol Sci 5:06–11
Remans R, Beebe S, Blair M, Manrique G, Tovar E, Rao I, Croonenborghs A, Torres-Gutierrez R, El-Howeity M, Michiels J, Vanderleyden J (2008) Physiological and genetic analysis of root responsiveness to auxin-producing plant growth-promoting bacteria in common bean (Phaseolus vulgaris L.) Plant Soil 302:149–161
Resende MP, Jakoby ICMC, dos Santos LCR, Soares MA, Pereira FD, Souchie EL, Silva FG (2014) Phosphate solubilization and phytohormone production by endophytic and rhizosphere Trichoderma isolates of guanandi (Calophyllum brasiliense Cambess). Afr J Microbiol Res 8(27):2616–2623
Revillas JJ, Rodelas B, Pozo C, Martı’nez-Toledo V, Gonza’lez-Lo´ pez J (2000) Production of B-group vitamins by two Azotobacter strains with phenolic compounds as sole carbon source under diazotrophic and adiazotrophic conditions. J Appl Microbiol 89:486–493
Robinson RJ, Fraaije BA, Clark IM, Jackson RW, Hirsch PR, Mauchline TH (2015) Endophytic bacterial community composition in wheat (Triticum aestivum) is determined by plant tissue type, developmental stage and soil nutrient availability. Plant Soil 405:381–396
Rodríguez H, Fraga R (1999) Phosphate solubilizing bacteria and their role in plant growth promotion. Biotechnol Adv 17:319–339
Ruinen J (1956) Occurrence of Beijerinckia species in the phyllosphere. Nature 178:220–221
Saber Z, Pirdashti H, Esmaeili M, Abbasian A, Heidarzadeh A (2012) Response of wheat growth parameters to co-inoculationof plant growth promoting Rhizobacteria (PGPR) and different levels of inorganic nitrogen and phosphorus. World Appl Sci J 16:213–219
Safari D, Jamali F, Nooryazdan HR, Bayat F (2016) Screening fluorescent pseudomonads isolated from wheat rhizosphere for plant growth- promoting and salt tolerance properties. Biol Forum – An Int Journal 8:35–42
Saif S, Khan MS, Zaidi A, Ahmad E (2014)Role of phosphate solubilizing actinomycetes in plant growth promotion: current perspective. In: Khan MS, Zaidi A, Musarrat J (eds) Phosphate solubilising microorganisms: principles and application of microphos technology. Springer, Switzerland; pp 137–156
Salma Z, Sindhu SS, Ahlawat VP (2014) Suppression of Fusarium wilt disease in gladiolus by using rhizobacterial strains. J Crop Weed 10:466–471
Sane SA, Mehta SK (2015) Isolation and evaluation of rock phosphate solubilizing fungi as potential biofertilizer. J Fertil Pestic 6(2):156–160
Sarfraz M, Abid M, Mehdi SM (2009) External and internal phosphorus requirements of wheat in Rasulpur soil series of Pakistan. Soil Environ 28:38–44
Sarkar A, Islam T, Biswas GC, Alam S, Hossain M, Talukder NM (2012) Screening for phosphate solubilizing bacteria inhabiting the rhizoplane of rice grown in acidic soil in Bangladesh. ActaMicrobiol Immunol Hung 59:199–213
Sarker A, Talukder NM, Islam MT (2014) Phosphate solubilizing bacteria promote growth and enhance nutrient uptake by wheat. Plant Sci Today 1:86–93
Satyanandam T, Babu K, Rosaiah G, Vijayalakshmi M (2014) Screening of Rhizobium strains isolated from the root nodules of Vigna mungo cultivated in rice fallows for their phosphate solubilizing ability and enzymatic activities. Brit Microbiol Res J4:996–1006
Saxena J, Minaxi, Jha A (2014) Impact of a phosphate solubilizing bacterium and an arbuscular mycorrhizal fungus (Glomus etunicatum) on growth, yield and P concentration in wheat plants. Clean (Weinh) 42:1248–1252
Saxena J, Chandra S, Nain L (2013) Synergistic effect of phosphate solubilizing rhizobacteria and arbuscular mycorrhiza on growth and yield of wheat plants. J Soil Sci Pl Nutr 13:511–525
Scervino JM, Mesa MP, Mónica ID, Recchi M, Moreno NS, Godeas A (2010) Soil fungal isolates produce different organic acid patterns involved in phosphate salts solubilization. Biol Fertil Soils 46:755–763
Schoebitz M, Ceballos C, Ciampi L (2013) Effect of immobilized phosphate solubilizing bacteria on wheat growth and phosphate uptake. J Soil Sci Plant Nutr 13(1):1–10
Selvakumar G, Mohan M, Kundu S, Gupta AD, Joshi P, Nazim S, Gupta HS (2008) Cold tolerance and plant growth promotion potential of Serratia marcescens strain SRM (MTCC 8708) isolated from flowers of summer squash (Cucurbita Pepo). Lett Appl Microbiol 46:171–175
Seghers D, Wittebolle L, Top EM, Verstraete W, Siciliano SD (2004) Impact of agricultural practices on the Zea mays L. endophytic community. Appl Environ Microbiol 70:1475–1482
Shanmugam V, Kanoujia N, Singh M, Singh S, Prasad R (2011) Biocontrol of vascular wilt and corm rot of gladiolus caused by Fusarium oxysporum f. Sp. gladioli using plant growth promoting rhizobacterial mixture. Crop Prot 30:807–813
Sharma AK (2002) Bio-fertilizers for sustainable agriculture. AgrobiosIndia, Jodhpur, p 407
Sharma SK, Johri BN, Ramesh A, Joshi OP, Prasad SS (2011) Selection of plant growth-promoting Pseudomonas spp. that enhanced productivity of soybean-wheat cropping system in Central India. J Microbiol Biotechnol 21:1127–1142
Sharma A, Rawat US, Yadav BK (2012) Influence of phosphorus levels and phosphorus solubilizing fungi on yield and nutrient uptake by wheat under sub-humid region of Rajasthan, India. ISRN Agronomy. Article ID 234656, 9 pages. doi:10.5402/2012/234656
Sharma SB, Sayyed RZ, Trivedi MH, Gobi TA (2013) Phosphate solubilizing microbes: sustainable approach for managing phosphorus deficiency in agricultural soils. SpringerPlus 2:587
Shrivastave M, D’Souza SF (2014) Bio-solubilization of rock phosphate and plant growth promotion by Aspergillus niger TMPS1 in ultisol and vertisol. International symposium on managing soils for food security and climate change adaptation and mitigation. Vienna, Austria
Sial NA, Memon MY, Abro SA, Shah JA, Depar ND, Abbas M (2015) Effect of phosphate solubilizing bacteria (Bacillus megaterium) and phosphate fertilizer on yield and yield components of wheat. Pak J Biotechnol 12:35–40
Singh H, Reddy MS (2011) Effect of inoculation with phosphate solubilizing fungus on growth and nutrient uptake of wheat and maize plants fertilized with rock phosphate in alkaline soils. Eur J Soil Biol 47:30–34
Singh P, Kumar V, Agrawal S (2014) Evaluation of phytase producing bacteria for their plant growth promoting activities. Int J Microbiol 2014; Article eID 426483, 7 pages, doi:10.1155/2014/426483
Singh RP, Jha PN (2015) Plant growth promoting potential of ACC deaminase rhizospheric bacteria isolated from Aerva javanica: a plant adapted to saline environments. Int J Curr Microbiol App Sci 4(7):142–152
Singh N, Pandey P, Dubey RC, Maheshwari DK (2008) Biological control of root rot fungus Macrophomina phaseolina and growth enhancement of Pinus roxburghii (Sarg.) by rhizosphere competent Bacillus subtilis BN1. World J Microbiol Biotechnol 24:1669–1679
Song OR, Lee SJ, Lee YS, Lee SC, Kim KK, Choi YL (2008) Solubilization of insoluble inorganic phosphate by Burkholderia cepacia DA 23 isolated from cultivated soil. Braz J Microbiol 39:151–156
Sönmez F, Tüfenkçi Ş (2015) Investigation the effects of different doses organic fertilizers and phosphate solubilizing bacterias on yield and nutrient contents in chickpea (Cicer arietinum L.) Int J Second Metab (IJSM) 2:43–53
Surapat W, Pakahuta C, Rattanachaikunsopon P, Aimi T, Boonlue S (2013) Characteristics of phosphate solubilization by phosphate solubilizing bacteria isolated from agricultural chilli soil and their efficiency on the growth of chilli (Capsicum frutescens L. Cv. Hua Rua). Chiang Mai J Sci 40:11–25
Susilowati LE, Syekhfani (2014) Characterization of phosphate solubilizing bacteria isolated from Pb contaminated soils and their potential for dissolving tricalcium phosphate. J Degrad Min Lands Manag 1:57–62
Tahir M, Mirza MS, Zaheer A, Dimitrov MR, Smidt H, Hameed S (2013) Isolation and identification of phosphate solubilizer Azospirillum, Bacillus and Enterobacter strains by 16SrRNA sequence analysis and their effect on growth of wheat (Triticum aestivum L.) Aus J Crop Sci 7:1284
Tallapragada P, Usha S (2012) Phosphate-solubilizing microbes and their occurrence in the rhizospheres of Piper betel in Karnataka, India. Turk J Biol 36:25–35
Timsina J, Connor DJ (2001) Productivity and management of rice-wheat cropping systems: issues and challenges. Field Crop Res 59:93–132
Tiwari S, Singh P, Tiwari R, Meena KK, Yandigeri M, Singh DP, Arora DK (2011) Salt-tolerant rhizobacteria-mediated induced tolerance in wheat (Triticum aestivum) and chemical diversity in rhizosphere enhance plant growth. Biol Fertil Soils 47:907–916
Tenorio-Salgado S, Tinoco R, Vazquez-Duhalt R, Caballero-Mellado J, Perez-Rueda E (2013) Identification of volatile compounds produced by the bacterium Burkholderia tropica that inhibit the growth of fungal pathogens. Bioengineered 4:236–243
Tomar US, Tomar IS, Badaya AK (1998) Response of chemical and biofertilizer on some matric traits in wheat. Crop Res 16:408–410
Turk MA, Tawaha AM (2002) Impact of seedling rate, seeding date, rate and method of phosphorus application in faba bean (Vicia faba L. Minor) in the absence of moisture stress. Biotechnol Agron Soc Environ 6:171–178
Upadhyay SK, Singh JS, Saxena AK, Singh DP (2012) Impact of PGPR inoculation on growth and antioxidant status of wheat under saline conditions. Plant Biol 14:605–611
USDA (2010) Grain Report No. IN 1011. Washington, Global Agricultural Information Network
Venkateswarlu B, Rao AV, Raina P (1984) Evaluation of phosphorous solubilization by microorganisms isolated from aridisols. J Ind Soc Soil Sc 32:273–277
Venieraki A, Dimou M, Pergalis P, Kefalogianni I, Chatzipavlidis I, Katinakis P (2011) The genetic diversity of culturable nitrogen-fixing bacteria in the rhizosphere of wheat. Microb Ecol 61:277–285
Verma P, Yadav AN, Kazy SK, Saxena AK, Suman A (2013) Elucidating the diversity and plant growth promoting attributes of wheat (Triticum aestivum) associated acidotolerant bacteria from southern hills zone of India. Natl J Life Sci 10:219–226
Verma P, Yadav AN, Kazy SK, Saxena AK, Suman A (2014) Evaluating the diversity and phylogeny of plant growth promoting bacteria associated with wheat (Triticum aestivum) growing in central zone of India. Int J Curr Microbiol App Sci 3:432–447
Verma P, Yadav AN, Khannam KS, Mishra S, Kumar S, Saxena AK, Suman A (2016a) Appraisal of diversity and functional attributes of thermotolerant wheat associated bacteria from the peninsular zone of India. Saudi J Biol Sci. doi:10.1016/j.sjbs.2016.01.042
Verma P, Yadav AN, Khannam KS, Kumar S, Saxena AK, Suman A (2016b) Molecular diversity and multifarious plant growth promoting attributes of bacilli associated with wheat (Triticum aestivum L.) rhizosphere from six diverse agro-ecological zones of India. J Basic Microbiol 56:44–58
Verma P, Yadav AN, Khannam KS, Panjiar N, Kumar S, Saxena AK, Suman A (2015a) Assessment of genetic diversity and plant growth promoting attributes of psychrotolerant bacteria allied with wheat (Triticum aestivum) from the northern hills zone of India. Ann Microbiol 65:1885–1899
Vessey KJ (2003) Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 255:123–112
Vijayabharathi R, Sathya A, Gopalakrishnan S (2016) A renaissance in plant growth-promoting and biocontrol agents by endophytes. In: Microbial inoculants in sustainable agricultural productivity. Springer, India; pp 37–60.
Wahid OA, Mehana TA (2000) Impact of phosphate-solubilizing fungi on the yield and phosphorus-uptake by wheat and faba bean plants. Microbiol Res 5(3):221–227
Wakelin S, Warren R, Harvey P, Ryder M (2004) Phosphate solubilization by Penicillium spp. closely associated with wheat roots. Biol Fertil Soils 40:36–43
Walpola BC, Yoon MH (2013a) In vitro solubilization of inorganic phosphates by phosphate solubilizing microorganisms. Afr J Microbiol Res 7:3534–3541
Walpola BC, Yoon MH (2013b) Phosphate solubilizing bacteria: assessment of their effect on growth promotion and phosphorus uptake of mung bean (Vigna radiata [L.]R. Wilzcek). Chilean J Agric Res 73:275–281
Walpola C, Yoon MH (2013c) Isolation and characterization of phosphate solubilizing bacteria and their co-inoculation efficiency on tomato plant growth and phosphorous uptake. Afr J Microbiol Res 7:266–275
Whitelaw MA (2000) Growth promotion of plants inoculated with phosphate solubilizing fungi. Adv Agron 69:99–151
Wibisono MG, Veneklaas E, Mendham DS, Hardiyanto EB (2015) Nitrogen fixation of Acacia mangium Willd. From two seed sources grown at different levels of phosphorus in an ultisol, South Sumatra, Indonesia. South Forests A J Forest Sci 77:59–64
Xiao C, Chi R, He H, Qiu G, Wang D, Zhang W (2009) Isolation of phosphate-solubilizing fungi from phosphate mines and their effect on wheat seedling growth. Appl Biochem Biotechnol 159(2):330–342
Xiao CQ, Chi RA, Hu LH (2013) Solubilization of aluminum phosphate by specific Penicillium spp. J Cent South Univ 20:2109–2114
Yasser MM, Mousa ASM, Massoud ON, Nasr SH (2014) Solubilization of inorganic phosphate by phosphate solubilizing fungi isolated from Egyptian soils. J Biol Earth Sci 4:B83–B90
Younis A, Riaz A, Ikram S, Nawaz T, Hameed M, Fatima S, Batool R, Ahmad F (2013) Salinity-induced structural and functional changes in three cultivars of Alternanthera bettzickiana (Regel) G. Nicholson. Turk J Agri For 37:674–687
Zabihi HR, Savaghebi GR, Khavazi K, Ganjali A, Miransari M (2011) Pseudomonas bacteria and phosphorous fertilization, affecting wheat (Triticum aestivum L.) yield and P uptake under greenhouse and field conditions. Acta Physiol Planta 33:145–152
Zaidi A, Ahmad E, Khan MS (2014) Role of phosphate solubilising microbes in the management of plant diseases. In: Khan MS,Zaidi A, Musarrat J (eds) Phosphate solubilising microorganisms: principles and application of Microphos technology. Springer, Switzerland; pp 225–256
Zaidi A, Khan MS, Ahemad M, Oves M (2009) Plant growth promotion by phosphate solubilizing bacteria. Acta Microbiol Immunol Hung 56:263–284
Zaidi A, Khan MS (2005) Interactive effect of rhizospheric microorganisms on growth yield and nutrient uptake of wheat. J Plant Nutr 28:2079–2092
Zhao Y (2010) Auxin biosynthesis and its role in plant development. Annu Rev Plant Biol 61:49–64
Zia-ul-Hassan ATS, Shah AN, Jamro GM, Rajpar I (2015) Biopriming of wheat seeds with rhizobacteria containing ACC deaminase and phosphate solubilizing activities increases wheat growth and yield under phosphorus deficiency. Pak J Agri Agril Engg Vet Sci 31:24–32
Zinniel DK, Lambrecht P, Harris NB, Feng Z, Kuezmarski D, Higley P, Ishimaru CA, Arunakumari A, Barletta RG, Vidaver AK (2002) Isolation and characterization of endophytic colonizing bacteria from agronomic crops and prairie plants. Appl Environ Microbiol 68:2198–2208
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Khan, M.S., Rizvi, A., Saif, S., Zaidi, A. (2017). Phosphate-Solubilizing Microorganisms in Sustainable Production of Wheat: Current Perspective. In: Kumar, V., Kumar, M., Sharma, S., Prasad, R. (eds) Probiotics in Agroecosystem. Springer, Singapore. https://doi.org/10.1007/978-981-10-4059-7_3
Download citation
DOI: https://doi.org/10.1007/978-981-10-4059-7_3
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-4058-0
Online ISBN: 978-981-10-4059-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)