Abstract
Image-guided percutaneous thermal ablation therapy, such as radiofrequency or microwave, has been widely used for liver tumors. However, when the tumors are located in the hepatic dome, it is difficult to perform ablation because of poor visualization due to the presence of pulmonary air, which can obstruct the transmission of ultrasound or make it impossible to identify a safe puncture path. In order to perform therapy on liver tumors located in the hepatic dome, the artificial pleural effusion technique has been applied. This chapter introduces indications for artificial pleural effusion, procedure for artificial pleural effusion, the category of solution injected, and our study concerning the efficacy of artificial pleural effusion.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keyword
1 Introduction
Image-guided percutaneous thermal ablation therapy, such as radiofrequency or microwave, has been widely used for liver tumors, especially for small hepatocellular carcinoma [1–10]. For imaging guidance, ultrasound has many advantages such as easy availability and real-time monitoring capability, which make make it the most common imaging guidance approach [11–15]. However, when the tumors are located in the hepatic dome, it is difficult to perform ablation because of poor visualization owing to the presence of pulmonary air, which can obstruct the transmission of ultrasound or make it impossible to identify a safe puncture path [16–20].
In order to perform therapy on liver tumors located in the hepatic dome, the artificial pleural effusion technique has been applied. Shinya Shimada et al. in 2001 first reported the application of an artificial hydrothorax in microwave coagulation therapy of liver tumors, and concluded that the technique is a simple, inexpensive, and accurate therapy for the treatment of liver tumors in the hepatic dome [21]. After that, a series of articles about the application of artificial pleural effusion in microwave or radiofrequency ablation of liver tumor were published, which proved that artificial pleural effusion with percutaneous MWA or radiofrequency ablation could be used to perform therapy on liver tumors in the hepatic dome [18–20, 22–25].
2 Indication of Artificial Pleural Effusion
So far, the purpose of artificial pleural effusion in all papers was to extend the indications of percutaneous thermal ablation for liver tumors in the hepatic dome. Therefore, the indications for artificial pleural effusion is as follows: firstly, liver tumors in the hepatic dome can not be revealed by ultrasound, or liver tumors can only be showed partially which would cause an incomplete ablation necrosis zone, and secondly, no proper puncture path can be identified because of restriction in the lungs. Other indications are the same as the general indications of liver cancer ablation. Contraindications include all kinds of acute or severe chronic respiratory insufficiency and cardiac insufficiency, chest infection, and empyema.
3 Procedure for Artificial Pleural Effusion
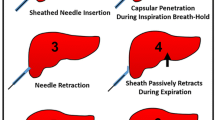
The basic instruments for artificial pleural effusion are an ultrasonic instrument with puncture needle holder, a 16G trocar, and infusion apparatus. The position of patients is semi-erect and left lateral. The region of the pleural cavity can be identified on ultrasound between the right anterior axillary line and the right posterior axillary line in patients. After local anesthesia, the 16G trocar is inserted through the costophrenic angle. Interventional radiologists can monitor the process of puncture by ultrasound and tell patient to hold their breath. When the sudden reduction of resistance is felt, 10 ml of 0.9 % saline solution is injected from the trocar to identify whether the tip of the needle has entered into the pleural cavity. If the tip of the needle is in the pleural cavity, the outer soft sheath is pushed into the pleural cavity to avoid injuring the lung, and the stylet of the needle is extracted. Then, the outer sheath is connected with the infusion set, and the fluctuation of the infusion in relation to the rhythm of breath can be observed. A sufficient amount of 0.9 % saline solution is injected until the tumor can be well-revealed or a safe path for puncture is fully identified. The volume of solution injected is about 500–1,500 ml. At the same time, the vital signs and oxygen saturation of the blood must be checked. After the therapy, the pleural effusion will be drawn out.
Different methods or instruments for artificial pleural effusion have been reported. Shinya Shimada et al., who first reported artificial pleural effusion, used the 14-F trocar to carry out thoracocentesis under general anesthesia using one-lung ventilation. Considering safety and avoiding injuring the lung, artificial pleural effusion as an innovative technology was performed under general anesthesia using one-lung ventilation at the beginning. Nowadays, artificial pleural effusion can be performed under local anesthesia. Akimichi Kume et al. reported two kinds of method for inducing artificial pleural effusion, which were called the two-step method and the one-step method [22]. The two-step method was performed as follows. First, a 23-gauge needle was inserted into the pleural space. After 200 ml of saline was injected into the pleural space, the 23-gauge needle was withdrawn and a central venous access kit was catheterized to instill more saline. Although a little complicated and time-consuming, this was a relatively safe method. Takahide Uehara et al. also reported a similar method to induce artificial pleural effusion [19]. The one-step method was performed as follows. Akimichi Kume et al. used an18-gauge Tuohy needle which was connected to a drip infusion set with 500 ml saline to puncture the thorax. The needle tip was inserted into the thorax under ultrasound monitoring, which can also be confirmed by the drip of saline. In addition, Yasunori Minami et al. [20] and Masahiko Koda et al. [23] reported the use of a Veress needle (Olympus Optical, Tokyo, Japan). The Veress needle consists of a blunt-tipped inner stylet and a sharp outer needle. Once the needle entered the pleural cavity, the blunt-tipped inner stylet extended to push the lung away to prevent injury. In a short, any of the methods mentioned above can acquire a satisfactory result.
4 Category of Solution Injected
Considering the kinds of fluid instilled into thorax, Paul F. Laeseke et al. reported their research with regard to the relative effectiveness of 0.9 % saline and 5 % dextrose in water for protecting the diaphragm and lung during radiofrequency [26, 27]. They draw a conclusion that “Instillation of 5 % dextrose in water into the peritoneal cavity before hepatic radiofrequency ablation decreases the risk and severity of diaphragm and lung injuries compared with 0.9 % saline in an animal model.” Because the heating effect of radiofrequency ablation is caused by the friction of ions, 5 % dextrose in water which is isotonic and nonionic could be an ideal buffer to infuse into the pleural cavity. However, when the method of ablation is microwave, 0.9 % saline solution as an artificial fluid is recommended, firstly because the heating theory of microwave ablation is the vibration of dipolar molecules rather than the friction of ions, and secondly because the use of 5 % dextrose in water is not suitable for diabetes patients, which restricts the use of 5 % dextrose in water.
5 Safety of Artificial Pleural Effusion
With regard to the safety of artificial pleural effusion, potential complications related to the procedure are bleeding, tumor seeding, and pleuritis. No major complications or deaths related to the artificial pleural effusion procedure have been identified in the published literature. Minor complications such as moderate pain, cough, mild dyspnea, low-grade fever, temporary elevation of liver transaminase, few subcutaneous hydrops, etc. disappeared spontaneously without any special treatment. Tae Wook Kang et al. [25] reported thermal injury to the diaphragm, and used computed tomography to observe the thickness of the diaphragm. Some papers reported that only a few patients had moderate pain, which aggravated when deeply breathing or changing position, and the pain disappeared spontaneously without any special treatment within 2 weeks. To avoid tumor seeding in the pleural cavity, puncture biopsy through artificial pleural effusion is forbidden. If it is necessary to insert a microwave antenna into tumor through artificial pleural effusion because there was no other safe path, repeated punctures are forbidden, and the applicator track must be heated with sufficient microwave energy by stopping the cooling-shaft water dump. No tumor seeding in pleural cavity has occurred in the existing literature.
6 Efficacy of Artificial Pleural Effusion
In reviewing the efficacy of artificial pleural effusion, our department summarized the data of 112 sessions of MWA with artificial pleural effusion which were performed on 102 consecutive patients with 119 liver tumors (17 of the patients had two tumors). Induction of artificial pleural effusion was achieved successfully in 110 of 112 sessions (98.2 %). Two sessions of artificial pleural effusion failed. One session had to stop inducing saline solution at the beginning of the procedure because of dyspnea. The other one was a recurring case, and artificial pleural effusion had been induced previously. The saline solution could not be induced into the thorax, and some liquid effused into the chest wall.
Among 110 successful artificial pleural effusion sessions, 82/83 sessions (98.8 %) made tumors clearly visible by gray-scale ultrasound (n = 74) and contrast-enhanced ultrasound (n = 8); 26/27 sessions (96.3 %) acquired a safe puncture path for percutaneous MWA. Two sessions failed to achieve the preoperative objective. One patient’s tumor was located at segment VII. The size of the tumor was about 0.9 × 0.7 × 0.7 cm. Even after about 800 ml 0.9 % saline solution had been injected, the tumor could not be revealed clearly. The other patient’s tumor was located at segment II. After about 1,500 ml 0.9 % saline solution had been injected, there was still no safe puncture path to avoid injuring the lung (Figs. 13.1 and 13.2).
Images in a 58-year-old man with 1.8 cm hepatocellular carcinoma treated by percutaneous microwave ablation (MWA) with artificial pleural effusion. (a) Contrast-enhanced magnetic resonance imaging (MRI) before MWA shows the neoplasm appearing in hyper-enhancement (small arrow) on arterial phrase and hypo-enhancement (large arrow) on venous phrase. (b) Left sonogram shows the puncture of the pleural cavity with a 16-gauge BD angiocath needle (small arrow). The middle sonogram shows the hyper-enhanced nodule on contrast-enhanced ultrasound, which is unclear on grey scale (large arrow). (c) Sonogram shows the procedure of MWA. The ablating area appears as a hyper-echoic region (large arrows) with artificial pleural effusion surrounded on grey-scale (small arrows). (d) Contrast-enhanced MRI 1 month after MWA shows the ablation zone appearing unenhanced in the arterial phrase (small arrow) and the venous phrase (large arrow)
Images in a 58-year-old man with 2.6 cm hepatocellular carcinoma treated by percutaneous MWA with artificial pleural effusion. (a) Contrast-enhanced MRI before MWA shows the neoplasm appearing as a high signal on T2WI (small arrow) and in hyper-enhancement (large arrow) on arterial phrase. (b) Left sonogram shows the puncture of the pleural cavity with a 16-gauge BD angiocath needle (small arrow). The middle sonogram shows the hyper-enhanced nodule on contrast-enhanced ultrasound (large arrow) with the artificial pleural effusion surrounded (triangle). (c) Sonogram shows the procedure of MWA. The ablating area appears as a disturbed blood flow on color-Doppler (small arrow) for microwave emitting, and as a hyper-echoic region on grey-scale (large arrow). (d) Contrast-enhanced MRI 1 month after MWA shows the ablation zone appearing as a low signal on T2WI (small arrow) and unenhanced in the arterial phrase (large arrow)
7 Efficacy of Percutaneous MWA with Artificial Pleural Effusion
To examine the efficacy of percutaneous MWA with artificial pleural effusion, we designed a case-control study, and compared the primary technique effectiveness rate, local tumor progression rate, and tumor-free survival rate between an artificial pleural effusion group and a control group which was matched in terms of tumor differentiation, tumor size, and tumor location. In previous studies [1, 3], tumor differentiation and tumor size were chosen as the independent prognostic factors affecting recurrence of hepatocellular carcinoma after microwave ablation treatment. Therefore, tumor differentiation and tumor size were selected as a matching standard to make sure that the two groups were balanced as far as possible. In addition, we matched tumor location between the two groups to compare complications after treatment. The results of our study showed that the primary technique effectiveness rate, the 1-, 2-, and 3-year local tumor progression rates, and the 1-, 2-, and 3-year tumor-free survival rates in the two groups had no significant difference. Therefore, percutaneous MWA with artificial pleural effusion for liver tumors located in the hepatic dome has a similar therapeutic effect to that of percutaneous MWA for liver tumors with good ultrasonic visibility.
8 Conclusion
Artificial pleural effusion could be a feasible, safe, and effective assistive technique for the expansion of indications of microwave or radiofrequency ablation for liver tumors located in the hepatic dome.
Abbreviations
- MWA:
-
Microwave ablation
References
Liang P, Dong B, Yu X, Yu D, Wang Y, Feng L, Xiao Q. Prognostic factors for survival in patients with hepatocellular carcinoma after percutaneous microwave ablation. Radiology. 2005;235(1):299–307.
N’Kontchou G, Mahamoudi A, Aout M, Ganne-Carrie N, Grando V, Coderc E, Vicaut E, Trinchet JC, Sellier N, Beaugrand M, Seror O. Radiofrequency ablation of hepatocellular carcinoma: long-term results and prognostic factors in 235 Western patients with cirrhosis. Hepatology. 2009;50(5):1475–83.
Dong B, Liang P, Yu X, Su L, Yu D, Cheng Z, Zhang J. Percutaneous sonographically guided microwave coagulation therapy for hepatocellular carcinoma: results in 234 patients. AJR Am J Roentgenol. 2003;180(6):1547–55.
Lencioni R, Cioni D, Crocetti L, Franchini C, Pina CD, Lera J, Bartolozzi C. Early-stage hepatocellular carcinoma in patients with cirrhosis: long-term results of percutaneous image-guided radiofrequency ablation. Radiology. 2005;234(3):961–7.
Yoon HM, Kim JH, Shin YM, Won HJ, Kim PN. Percutaneous radiofrequency ablation using internally cooled wet electrodes for treatment of colorectal liver metastases. Clin Radiol. 2012;67(2):122–7.
Nishikawa H, Osaki Y, Iguchi E, Takeda H, Ohara Y, Sakamoto A, Hatamaru K, Saito S, Nasu A, Kita R, Kimura T. Percutaneous radiofrequency ablation therapy for recurrent hepatocellular carcinoma. Anticancer Res. 2012;32(11):5059–65.
Zacharoulis D, Asopa V, Navarra G, Nicholls JP, Jensen SL, Habib NA. Hepatectomy using intraoperative ultrasound-guided radiofrequency ablation. Int Surg. 2003;88(2):80–2.
Yokoyama T, Egami K, Miyamoto M, Watanabe H, Hasegawa H, Iida S, Suzuki S, Nakamura Y, Okawa K, Hagiwara N, Takashima Y, Yoshioka M, Tajiri T, Onda M. Percutaneous and laparoscopic approaches of radiofrequency ablation treatment for liver cancer. J Hepatobiliary Pancreat Surg. 2003;10(6):425–7.
Crucitti A, Danza FM, Antinori A, Vincenzo A, Pirulli PG, Bock E, Magistrelli P. Radiofrequency thermal ablation (RFA) of liver tumors: percutaneous and open surgical approaches. J Exp Clin Cancer Res. 2003;22(4 Suppl):191–5.
Machi J, Uchida S, Sumida K, Limm WM, Hundahl SA, Oishi AJ, Furumoto NL, Oishi RH. Ultrasound-guided radiofrequency thermal ablation of liver tumors: percutaneous, laparoscopic, and open surgical approaches. J Gastrointest Surg. 2001;5(5):477–89.
Xu HX, Wang Y, Lu MD, Liu LN. Percutaneous ultrasound-guided thermal ablation for intrahepatic cholangiocarcinoma. Br J Radiol. 2012;85(1016):1078–84.
Carrafiello G, Fontana F, Cotta E, Petulla M, Brunese L, Mangini M, Fugazzola C. Ultrasound-guided thermal radiofrequency ablation (RFA) as an adjunct to systemic chemotherapy for breast cancer liver metastases. Radiol Med. 2011;116(7):1059–66.
Hofer S, Oberholzer C, Beck S, Looser C, Ludwig C. Ultrasound-guided radiofrequency ablation (RFA) for inoperable gastrointestinal liver metastases. Ultraschall Med. 2008;29(4):388–92.
Chiou YY, Hwang JI, Chou YH, Wang HK, Chiang JH, Chang CY. Percutaneous ultrasound-guided radiofrequency ablation of intrahepatic cholangiocarcinoma. Kaohsiung J Med Sci. 2005;21(7):304–9.
Xu HX, Xie XY, Lu MD, Chen JW, Yin XY, Xu ZF, Liu GJ. Ultrasound-guided percutaneous thermal ablation of hepatocellular carcinoma using microwave and radiofrequency ablation. Clin Radiol. 2004;59(1):53–61.
Kim PN, Choi D, Rhim H, Rha SE, Hong HP, Lee J, Choi JI, Kim JW, Seo JW, Lee EJ, Lim HK. Planning ultrasound for percutaneous radiofrequency ablation to treat small (</=3 cm) hepatocellular carcinomas detected on computed tomography or magnetic resonance imaging: a multicenter prospective study to assess factors affecting ultrasound visibility. J Vasc Interv Radiol. 2012;23(5):627–34.
Nesher N, Ben Haim M, Pevni D, Kessler A, Paz Y. Ultrasound-guided, video-assisted transdiaphragmatic radiofrequency ablation for primary liver malignancy or metastatic nodules. Innov (Phila). 2011;6(5):337–40.
Liu LN, Xu HX, Lu MD, Xie XY. Percutaneous ultrasound-guided thermal ablation for liver tumor with artificial pleural effusion or ascites. Chin J Cancer. 2010;29(9):830–5.
Uehara T, Hirooka M, Ishida K, Hiraoka A, Kumagi T, Kisaka Y, Hiasa Y, Onji M. Percutaneous ultrasound-guided radiofrequency ablation of hepatocellular carcinoma with artificially induced pleural effusion and ascites. J Gastroenterol. 2007;42(4):306–11.
Minami Y, Kudo M, Kawasaki T, Chung H, Ogawa C, Inoue T, Sakaguchi Y, Sakamoto H, Shiozaki H. Percutaneous ultrasound-guided radiofrequency ablation with artificial pleural effusion for hepatocellular carcinoma in the hepatic dome. J Gastroenterol. 2003;38(11):1066–70.
Shimada S, Hirota M, Beppu T, Shiomori K, Marutsuka T, Matsuo A, Tanaka E, Ogawa M. A new procedure of percutaneous microwave coagulation therapy under artificial hydrothorax for patients with liver tumors in the hepatic dome. Surg Today. 2001;31(1):40–4.
Kume A, Nimura Y, Kamiya J, Nagino M, Kito Y. Percutaneous ethanol injection via an artificially induced right hydrothorax for hepatocellular carcinoma in the hepatic dome. Cardiovasc Intervent Radiol. 2003;26(6):543–9.
Koda M, Ueki M, Maeda Y, Mimura K, Okamoto K, Matsunaga Y, Kawakami M, Hosho K, Murawaki Y. Percutaneous sonographically guided radiofrequency ablation with artificial pleural effusion for hepatocellular carcinoma located under the diaphragm. AJR Am J Roentgenol. 2004;183(3):583–8.
Minami Y, Kudo M, Kawasaki T, Chung H, Ogawa C, Shiozaki H. Percutaneous radiofrequency ablation guided by contrast-enhanced harmonic sonography with artificial pleural effusion for hepatocellular carcinoma in the hepatic dome. AJR Am J Roentgenol. 2004;182(5):1224–6.
Kang TW, Rhim H, Lee MW, Kim YS, Choi D, Lee WJ, Lim HK. Radiofrequency ablation for hepatocellular carcinoma abutting the diaphragm: comparison of effects of thermal protection and therapeutic efficacy. AJR Am J Roentgenol. 2011;196(4):907–13.
Hinshaw JL, Laeseke PF, Winter 3rd TC, Kliewer MA, Fine JP, Lee Jr FT. Radiofrequency ablation of peripheral liver tumors: intraperitoneal 5% dextrose in water decreases postprocedural pain. AJR Am J Roentgenol. 2006;186(5 Suppl):S306–10.
Laeseke PF, Sampson LA, Brace CL, Winter 3rd TC, Fine JP, Lee Jr FT. Unintended thermal injuries from radiofrequency ablation: protection with 5% dextrose in water. AJR Am J Roentgenol. 2006;186(5 Suppl):S249–54.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Zhang, D., Liang, P. (2015). Application of Artificial Pleural Effusion in Microwave Ablation of Liver Tumor. In: Liang, P., Yu, Xl., Yu, J. (eds) Microwave Ablation Treatment of Solid Tumors. Springer, Dordrecht. https://doi.org/10.1007/978-94-017-9315-5_13
Download citation
DOI: https://doi.org/10.1007/978-94-017-9315-5_13
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-017-9314-8
Online ISBN: 978-94-017-9315-5
eBook Packages: MedicineMedicine (R0)