Abstract
Circulating tumor cells (CTCs) have been considered an indicator of tumor invasion. CTCs have recently been detected in breast cancer patients and have become a target for evaluating breast cancer progression, prognosis, and diagnosis. CTCs are a heterogenous population with phenotypes ranging from epithelial to mesenchymal. CTCs express various markers including epithelial cell adhesion molecule, cytokeratins, and MUC-1, depending on the stage of epithelial–mesenchymal transition. Breast CTCs are usually detected and confirmed via two steps, including enrichment and identification. These methods have become powerful tools for diagnosis and for predicting response to systemic therapies. This chapter aims to review breast CTC biology and the role of CTC detection in breast cancer prognosis and diagnosis. Recent advances in CTC research mean that CTCs are becoming a strong tool for the prognosis and diagnosis of breast cancer.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Breast cancer is one of the most common cancers among women worldwide, with 1.15 million new cases each year [1], including an estimated 207,090 new cases and 39,840 deaths from metastasis in the USA in 2010 [2]. Metastasis is a major cause of death in all cancer patients, and managing metastasis is therefore an important step in treating breast cancer. Breast cancer is currently classified by TNM classification and differentiation grade, complemented by estrogen and progesterone receptor (ER/EP) status and HER2/neu expression [3–7]. Recent advances have been made in relation to the molecular characterization of tumors, and in predicting the effects of therapy, including using breast cancer gene profiling by reverse transcription-polymerase chain reaction [8–11].
Breast Circulating Tumor Cells
Circulating tumor cells (CTCs) are biomarkers for evaluating metastasis in breast cancer. Since 2000, breast CTCs have been considered a unique target for understanding disease progression, prognosis, and treatment in breast cancer pathogenesis. CTCs are tumor cells that have been shed into the vasculature from the primary tumor and are thus present in the blood. They are found in the blood of patients with many different types of carcinomas, especially in those with metastatic disease [12, 13]. Some studies found that the presence of CTCs in breast cancer patients was associated with significantly shorter progression-free survival (PFS) and overall survival (OS) [14–17], as well as with a high risk of recurrence [18].
Disseminated tumor cells (DTCs) differ from CTCs and were a focus of earlier studies. DTCs are defined as tumor cells in the bone marrow and were present in 30 % of breast cancer patients [19]. The presence of DTCs was considered a significant predictor of outcome. However, bone marrow isolation is an invasive technique, and DTCs were only present at low levels in the bone marrow, with as few as 3 % of bone marrow aspirates containing tumor cells [20]. CTCs are therefore of more interest as targets for the diagnosis and prognosis of cancer, particularly breast cancer.
Morphology
Breast CTCs may be larger than other cells in the blood, such as leukocytes, erythrocytes, and thrombocytes. A commonly quoted range for CTCs is 12–25 μm, which is larger than 90–95 % of the largest blood cell population [21]. Size-based sorting thus represents an attractive, label-free, isolation method. Another study found similar nucleus–cytoplasm ratios in CTCs and tumor cells from a solid metastasis, suggesting that the cell populations in the two sites were similarly differentiated. However, the average size of CTCs is smaller than that of tumor cells in a solid metastasis [22], suggesting either a filtering effect of the capillary beds proximal to the site of blood collection or morphologically undetectable apoptotic effects on the circulating cells.
Immunophenotype
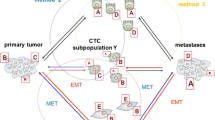
Epithelial to mesenchymal transition (EMT) is a process relating to a set of molecular changes, whereby carcinoma cells increase motility, invasion, and intravasation, as shown in Fig. 21.1. EMT induces proteases that trigger the degradation of the extracellular matrix, allowing carcinoma cells to be released into the blood [23, 24]. CTCs have therefore been considered as EMT-phenotype carcinoma cells in the blood. The EMT phenotype is also associated with a stem cell phenotype [25], and CTCs were demonstrated to include a small population with a cancer stem cell phenotype [26–28].
CTCs are considered as EMT forms of primary tumor cells. Carcinoma cells initially activate the trans-differentiation EMT program, during which they acquire the traits needed to execute the multiple steps of metastasis [29]. During the metastatic process, carcinoma cells in primary breast tumors gradually change from an epithelial phenotype (in the primary tumor), via EMT to a mesenchymal phenotype (in the blood), to an epithelial phenotype again (at the metastatic sites). CTCs in the blood thus exhibit the EMT phenotype.
Breast CTCs in the blood have been recognized with different phenotypes ranging from epithelial to mesenchymal, depending on the stage of EMT. CTCs usually express an epithelial phenotype during the early stage of EMT, both epithelial and mesenchymal phenotypes in the intermediate stage, and a mesenchymal phenotype in late EMT. Pecot et al. recently showed that CTC capture based on cytokeratin (CK) expression would be likely to miss populations of CTCs that had undergone EMT because CTCs in the late stage of EMT lack expression of CK markers [30].
CTCs with Epithelial Phenotypes in Early Epithelial–Mesenchymal Transition
One epithelial marker detected in CTCs is the surface epithelial cell adhesion molecule (EpCAM). EpCAM is a pan-epithelial differentiation antigen expressed on most carcinomas. CTCs express high levels of this marker. Indeed, EpCAM levels are 100- to 1,000-fold greater in primary and metastatic breast cancers than in normal epithelial cells, and this molecule is implicated in tumor invasion and migration [31]. EpCAM has been suggested as a potential therapeutic target in patients with advanced malignancies [19]. EpCAM-positive CTCs could reflect both the volume of metastatic disease in breast cancer patients and the tumor biology in terms of aggressiveness, drug resistance, and mutation.
CTCs have also been defined as mononuclear cells lacking CD45 (which is expressed in blood cells) but expressing CKs (which are strongly expressed in epithelial cells) [32]. Some specific CKs such as CK8, CK18, CK19, and CK20 have been used as markers of CTCs, included breast CTCs [33–36]. Some commercially available methods for isolating CTCs have been developed based on antibodies to these marker proteins, in combination with CD45-negative selection to eliminate white blood cells.
Zhao et al. demonstrated that EpCAM-, CK19-, and hMAM-positive cells were detected in 50 (51.0 %), 43 (43.9 %), and 68 (69.4 %) of 98 patients, respectively, and triple-marker-positive CTCs were detected in 86 of the 98 (87.8 %) patients, which was a significantly higher rate than in the control group [37]. In a recent study, Tunca et al. showed that CK20 was a novel biomarker of breast CTCs, which could be used to identify CTCs as well as to predict breast cancer progression. The CK20-positivity rate was 28.57 % (24/84) [38]. CD227 (Mucin-1 or Ca 15.3) is highly expressed by virtually all mucosal epithelial tissues and is aberrantly expressed in most human breast cancers. It is also used as marker of breast cancer cells and breast CTCs [39–41].
CTCs with Epithelial–Mesenchymal Phenotype in Intermediate Epithelial–Mesenchymal Transition
During the intermediate stage of the EMT process, CTCs exhibit both epithelial and mesenchymal traits. In patients with metastatic CTCs, more than 80 % of CTCs co-express epithelial proteins, such as EpCAM, CKs, and E-cadherin, together with mesenchymal proteins including vimentin, N-cadherin and O-cadherin, and the stem cell marker CD133 [42]. Another study showed that 77 % of CTCs in early breast cancer patients expressed vimentin, Twist, and CK, while 100 % of CTCs expressed these proteins in patients with metastatic breast cancer. CTCs in patients with metastatic disease thus express antigens characteristic of EMT more than those with early cancer, suggesting that EMT is involved in the metastatic potential of CTCs [43].
CTCs with Mesenchymal Phenotype in Late Epithelial–Mesenchymal Transition
A recent study by Gorges et al. used the AdnaTest to detect CTCs in metastatic breast cancer patients. However, the kit failed to detect CTCs because of the downregulation of EpCAM, whereas mesenchymal markers such as Twist and epidermal growth factor receptor were upregulated, indicating that the CTCs in these patients were in the late stage of EMT. They also recognized that the late stage of EMT was associated with metastatic cancers [44].
CTCs Express Breast Cancer Stem Cell Phenotypes
Breast cancer stem cells (BCSCs) are known to have specific phenotypes, such as EpCAM+CD44+CD24−/dim or CD44+CD24−/dim. Based on the BCSC phenotype, CTCs contain 20–30 % BCSCs with the EpCAM+CD44+CD24−/dimCD45− phenotype. Other CTC phenotypes are also found in breast cancer patients, including EpCAM+CD44−CD24−/dimCD45−, EpCAM+CD44+CD24+CD45−, and EpCAM+CD44+CD24+CD45dim [26]. Theodoropoulos et al. detected CTCs in 66.7 % patients, with 35.2 % of CTCs positive for the BCSC phenotype CD44+CD24−/dim. CTCs also exhibited another BCSC phenotype aldehyde dehydrogenase 1 (ALDH1)hiCD24−/dim in 17.7 % patients [27]. Like BCSCs that are the most tumorigenic cells in solid tumors, CTCs expressing BCSC markers are also known as breast circulating tumor stem cells (CTSCs). Breast CTSCs are determined as CD45−EpCAM+CD44+CD24− cells in the blood. The percentage of CTSCs in CD45-negative cells detected by flow cytometry increased with increasing TNM stage (0, 0.00 ± 0.00 %; I, 0.03 ± 0.05 %; II, 0.06 ± 0.14 %; III, 0.10 ± 0.09 %; IV, 0.29 ± 0.35 %; P = 0.034) [28].
Notably, the expression of ALDH1 on CTCs was found to correlate with the stage of disease and with the expression of vimentin and fibronectin [45]. CTCs also express some other BCSC markers such as NOTCH1, a gene associated with self-renewing cancer stem cells [46], and almost 70 % of CTCs were found to express ALDH1 [47, 48]. Similarly to BCSCs, CTCs were also shown to be triple negative for ER, PR, and HER2 [47, 48].
Breast CTCs and HER2, ER, PR Tumor Phenotype
HER2/neu is considered as a target for breast treatment. Some studies have evaluated the correlation between HER2/neu expression in CTCs and in primary tumor cells, as well as HER2/neu expression in primary tumor cells with some specific genes in individual CTCs [48–55]. Evidence to date suggests that HER2/neu expression in CTCs does not depend strictly on HER2/neu expression in the primary tumor cells; indeed, HER2/neu-positive cells were detected in patients with HER2/neu-negative tumors [51, 54]. These results have also been confirmed by other groups [56, 57]. Punnoose et al., however, reported that there was a concordance between HER2 status in CTCs and in the primary tumor tissue in the majority of patients (89 %), though the HER2 status in CTCs differed from that in the primary tumor in a subset of patients (11 %) [58]. Thus, some patients negative for HER2/neu in the primary tumor cells may still benefit from HER-2-directed therapy [59]. Moreover, HER-2-positive CTCs were more common in women with HER-2-positive primary tumors [54].
Similarly to HER2/neu expression, ER/PR expression in CTCs does not depend on ER/PR expression in the primary tumor. Aktas et al. showed that the expression profiles of CTCs and the primary tumor differed with regard to ER/PR/HER2 positivity [47], with concordance rates of 29, 25, and 53 %, respectively [48].
Detection Methods for Breast Circulating Tumor Cells
Detection of breast CTCs is an important and essential step in their use for breast cancer diagnosis and prognosis. This step determines not only the existence of CTCs but also the number of CTCs. Like many special kinds of cells, CTCs in a heterogenous population can be detected based on their unique characteristics. However, CTCs are rare cells (one CTC/106–107 mononuclear cells), and they must therefore be enriched before detection. Hence, CTC detection methods are usually composed of two steps. The initial step enriches the CTCs using morphological and immunological techniques that are nonspecific for CTCs, while the second step involves their identification using protein and nucleic acid-based techniques that are specific to CTCs.
Based on the particular proteins or/and nucleic acids (usually RNA), CTCs can be not only detected but also quantified. CTC detection results can help in the diagnosis of breast cancer, as well as tracking cancer status or drug response during treatment. The CTC isolation and detection process therefore needs to be highly specific.
Enrichment Methods
Size-Based Enrichment
As noted above, CTCs are larger than most other blood cells, and some enrichment methods have therefore utilized this size difference. Size-based enrichment strategies for CTCs include centrifugation, microfiltration, and hydrodynamic sorting. Centrifugation is the most popular method used for cell and stem cell enrichment, while microfiltration and hydrodynamic sorting are relatively new technologies.
Centrifugation
Centrifugation uses centrifugal force to separate cells based on their density. Blood cells will separate into distinct zones in the centrifugation tube according to their different densities. CTCs, leukocytes, and thrombocytes exist in the same zone called the buffy coat layer. Density gradient centrifugation using a separating medium can then be used to further separate cells within the distinct zone. Two separating media are commonly used: Ficoll (GE Healthcare) and OncoQuick (Greiner Bio-One). OncoQuick is an improved form of Ficoll that limits cross-contamination between different zones by using a porous membrane. OncoQuick has thus been shown to produce a higher recovery [60, 61].
Microfiltration
Alternatively, CTCs can be enriched using a microfiltration device [62–66]. As noted above, CTCs are mostly epithelial cells that are larger than blood cells [62, 67, 68]. Some studies have shown that a microfiltration device with a pore size of around 8 μm in diameter is optimal for CTC retention [69]. This technique initially used polycarbonate filters fabricated using track etching [70], which generates pores at random locations. However, the low pore density and multiple pore fusion led to low CTC capture efficiency (around 50–60 %) and frequent sample clogging on the filter [67, 71]. To increase the capture efficiency, improved microfabricated filters with high-density uniform circular pores were developed as 2D [21, 68] and 3D microfiltration devices [72].
Hydrodynamic Sorting
Hydrodynamic sorting uses fluid flow in combination with microdevice geometries or parallel fluids at different flow rates to sort or separate tumor cells. The main advantage of this technique is that the cells do not pass through any physical constriction and shear forces are therefore reduced. In addition, these devices typically operate at relatively high flow rates, resulting in high throughout.
Dielectrophoresis
Dielectrophoresis (DEP) is a new technique based on differences in cell polarizability and size. When an electrical field is applied to cells, they become polarized. Cells will interact with an electric field. DEP forces can be used in two ways to separate cells: DEP migration, where different types of particles migrate to different regions based on their relative polarizabilities, and DEP retention, where DEP forces are used in competition with fluid-flow forces. In the case of CTC isolation from blood, electrophoretic mobility distinguishes tumor cells in terms of their attraction toward the electrode, and normal blood cells migrate in the electrical field into an eluant.
Immunological-Based Methods
Immunological-based methods select cells based on their surface markers. Two selection methods are commonly used: positive selection and negative selection.
Positive Selection
Most CTCs originate from epithelial cells and thus express epithelial cell-specific markers such as EpCAM and CKs. These markers can be efficiently used to enrich CTCs. Some tumor-specific markers relating to specific cancers, such as alpha-fetoprotein, HER2/neu, MUC1/MUC2, mammaglobulin, and carcinoembryonic antigen, can also be used to enrich CTCs. Some approaches have combined popular epithelial cell markers and tumor-specific markers to improve the enrichment results. Two immunomagnetic methods can be used: (1) ferrous beads with anti-EpCAM that retain EpCAM-positive cells in magnetic columns (the so-called magnetic-activated cell sorting (MACS)) and (2) anti-EpCAM proteins bound in arrays to which EpCAM-positive cells can then attach.
However, as suggested, CTCs in the blood stream express a gradient of EMT markers, and some CTCs express low or no EpCAM. Immunomagnetic methods that positively select EpCAM-positive cells are therefore unable to detect them [73]. Indeed, a previous study considered that the evaluation of CTCs as prognostic markers should include both EpCAM-positive and EpCAM-negative cells [74].
Negative Selection
Most blood cells are positive for CD45, and anti-CD45 negative selection of leukocytes is thus used to enrich cell populations with CTCs that are negative for CD45. This selection technique uses antibodies labeled with magnetic microbeads and magnets. The most popular system used for negative selection is MACS (Miltenyi Biotec).
Many commercial systems currently exist for the enrichment and isolation of CTCs, such as MACS, CellSearch, RARE, AdnaTest, CTC chip, ELISPOT, MAINTRAC, Ikoniscope, and Ariol. The characteristics of these methods are summarized in Table 21.1. To increase the enrichment efficacy, some systems have combined selection by anti-CK and anti-EpCAM antibodies with CD45 depletion or with other markers.
Identification Methods
Identification is essential to confirm the cells as CTCs. CTC confirmation is usually based on specific markers expressed at the transcriptional or translational level. Nucleic acid-based methods are used at the transcriptional level, while antibody-based assays are used at the translational level. Recently, four methods have been used to identify and confirm the identity of breast CTCs, including (1) flow cytometry, (2) image-based approaches such as classic immunocytochemistry (CellSearch, Ariol system, laser scanning system), (3) protein-based assays such as ELISPOT that detect secreted proteins from CTCs, and (4) reverse transcription–polymerase chain reaction (RT-PCR) (qRT-PCR, multiplex RT-PCR).
Nucleic Acid-Based Methods
In most cases, RT-PCR or real-time RT-PCR is used to identify specific gene expression. However, there is currently no specific gene profile that can be used to confirm breast cancer CTCs. RT-PCR is commonly used to amplify and identify genes relating to EMT as well as breast cancer. Such genes may include CK19, mammaglobin-A (MGB1), HER2, and MUC1 [75–80].
Some commercial systems combine several markers to improve the results of RT-PCR. For example, AdnaTest BreastCancerSelect uses multiplex RT-PCR for HER2, MUC1, and EpCAM genes to confirm breast cancer CTCs [81]. Xi et al. showed that MGB2 was a marker of breast cancer CTCs [82]. The limited availability of CTCs means that single-cell PCR can be an ideal platform.
In addition to RT-PCR, fluorescence in situ hybridization (FISH) is also a valid method for CTC confirmation. Expression of HER2 on breast cancer CTCs evaluated by FISH showed 93 % concordance with the expression in primary breast cancer cells [83]. Using FISH, Hayashi et al. detected polysomy 17 in a small population CTCs in patients with metastatic breast cancer [84].
Cytometric-based Methods
At the translational level, CTCs can be confirmed using cytometric-based methods. In all cytometric-based methods, CTCs are stained with monoclonal antibodies specific for CTCs. There are three popular cytometric-based methods currently used for CTC confirmation: flow cytometry, image-based approaches (immunocytochemistry, fiber-optic array scanning technology (FAST), laser scanning cytometer (LSC)), and EPISPOT (epithelial immunospot).
Most cytometric-based methods use CKs and EpCAM as common markers to identify CTCs. Moreover, CD45 can also be used to detect leukocytes to increase the specificity. However, not all CTCs express CKs and EpCAM, and during EMT, CTCs may change from an epithelial phenotype expressing CKs or EpCAM to a mesenchymal phenotype with lower expression of these markers.
Flow Cytometry
Flow cytometry is a conventional method for identifying and counting CTCs. This technique has certain advantages compared with PCR, including high specificity, high statistical significance based on counting large numbers of cells in the blood, and analysis of multiple parameters including DNA content, cell size, and cell viability, as well as intracellular markers. However, it also has some limitations, particularly low sensitivity (one tumor cell/104–105 blood cells) compared with RT-PCR approaches (one tumor cell/106 blood cells) [10, 85].
Image-Based Approaches
CTCs are usually enriched and then stained with specific markers by immunocytochemical techniques to confirm their identity. This technique has been used by pathologists for decades to identify certain types of tumor cells. However, it has some limitations, including being time-consuming, and automated systems have therefore been developed to capture the stained CTCs, such as the LSC (Compucyte Corporation, Cambridge, MA), automated cellular imaging system (ACIS, Dako, Denmark), and Ariol (Applied Imaging Corp. San Jose, CA). The LSC makes it possible to scan and relocate epithelial-positive cells immunolabeled for multiple markers such as EpCAM combined with the lymphocyte marker CD45 automatically. ACIS and Ariol allow the rapid and automatic identification of CTCs based on their morphological evaluation.
Fiber-optic array scanning technology (FAST) is another, more sensitive technique for confirming CTCs. This system is equipped with an exceptionally large (50,341 mm) field of view without sacrifice of collection efficiency, which makes it possible to locate immunofluorescently labeled CTCs on glass substrates at a scan rate 500 times faster than conventional automated digital microscopy, allowing FAST to detect CTCs without the need for an enrichment procedure [86, 87]. Moreover, the process is very rapid, with up to 300,000 cells scanned per second. However, there are currently no reports of validation studies in clinical settings.
EPISPOT
EPISPOT (epithelial immunospot) is another antibody-based approach and is an immunological assay based on enzyme-linked immunosorbent assay (ELISA) technology. EPISPOT identifies CTCs by detecting specific proteins (CKs, MUC, prostate-specific antigen) secreted by them. However, one of the features of EPISPOT is that it can only detect viable CTCs, because dying CTCs do not secrete adequate amounts of proteins [88]. The sensitivity of EPISPOT is superior to that of ELISA by two orders of magnitude in terms of detecting released CK19 from cancer cells [89]. However, validation studies in clinical settings are still awaited.
Commercial Circulating Tumor Cell Detection Systems for Breast Cancer
Significant effort has recently been devoted to the development of automated techniques for detecting CTCs, and several commercial systems now exist for detecting breast cancer CTCs, including CellSearch, CTC chip, The CTChip, MagSweeper, MAINTRAC, Ariol, and AdnaTest, which combine enrichment and confirmation steps, as shown in Table 21.2. However, only CellSearch has currently been approved by the FDA for detecting breast cancer CTCs [90–92].
CellSearch is used worldwide to detect breast CTCs and has been approved in more than 17 countries. This system comprises two steps: an initial step to enrich epithelial cells by selecting EpCAM-positive cells and a second step to identify epithelial carcinoma cells by double staining with CD45 and CK antibodies, using DAPI to visualize the cell nucleus. Analysis of cells using CellSearch is thus performed as follows: first, peripheral blood is mixed with iron particles coated with EpCAM to confer magnetic properties on all the epithelial cells, and then, anti-CK antibodies are used to identify these epithelial cells, while anti-CD45 antibodies are used to rule out lymphocytes; the nuclear dye DAPI is applied to fluorescently label cell nuclei for microscopic visualization of the enriched cell population. After incubation, washing, magnetic separation, and fixation, the immunomagnetically separated cell population can be viewed and counted by automated digital fluorescent microscopy [90]. CellSearch requires about 7.5 ml of blood for each analysis. Figure 21.2 shows some approaches in breast CTC enrichment and identification.
Circulating Tumor Cells for Diagnosis and Prognosis
Many studies have demonstrated that the presence of CTCs is significantly associated with shorter PFS, disease-free survival (DFS), and OS. CTCs are considered as a stable prognosticator in patients with early-stage and metastatic breast cancer. By counting CTCs before and after treatment in patients with metastatic breast cancer, Cristofanilli et al. showed that detection of CTCs before initiation of first-line therapy in patients with metastatic breast cancer was highly predictive of PFS and OS [93].
CTCs Associate with Progression-Free, Disease-Free, and Overall Survival
Zhao et al. performed a meta-analysis of published literature to assess whether the detection of CTCs in patients diagnosed with primary breast cancer could be used as a prognostic factor. A total of 24 eligible studies with 4,013 cases and 1,333 controls were included. Meta-analyses were performed using a random-effects model, with the hazard ratio (HR) and 95 % confidence intervals (95 % CIs) as effect measures. The results showed that the positive detection of CTCs was significantly associated with poor OS (HR = 3.00 [95 % CI 2.29–3.94], n = 17, P < 0.0001) and recurrence-free survival (RFS) (HR = 2.67 [95 % CI 2.09–3.42], n = 22, P < 0.0001) [94]. Another meta-analysis identified 49 eligible studies enrolling 6,825 patients and showed that the presence of CTCs was significantly associated with shorter survival in the total population. The prognostic value of CTCs was significant in both early (DFS, HR 2.86; 95 % CI 2.19–3.75; OS, HR 2.78; 95 % CI 2.22–s3.48) and metastatic breast cancer (PFS, HR 1.78; 95 % CI 1.52–2.09; OS, HR 2.33; 95 % CI 2.09–2.60) [95].
The association between CTCs and survival also depends on the number of CTCs. Patients with no CTCs at baseline had a significantly better prognosis, while an increase in number of CTCs was associated with increased risk for both PFS and OS, though the rate of increase lessened above approximately five CTCs/7.5 ml. CTCs increasing up to a maximum of five represented a prognostic factor in metastasis [96]. In another study, Hayes et al. analyzed the correlation between the number of CTCs and OS and found that the median OS for patients with <5 CTCs/7.5 ml from blood drawn at five time points was >18.5 months, while OS for patients with ≥5 CTCs/7.5 ml was significantly shorter, by 10.9, 6.3, 6.3, 6.6, and 6.7 months, respectively [15]. Liu et al. also observed shorter PFS for patients with ≥5 CTCs at 3–5 weeks and at 7–9 weeks after the start of treatment [97]. Median OS and PFS in the subgroup with ≥5 circulating tumor cells/7.5 ml of blood at baseline were significantly shorter (5 months and 3 months, respectively) compared with the subgroup with <5 circulating tumor cells (8 months and 7 months, respectively) (P = 0.003 and P < 0.001, respectively). The number of metastatic sites was significantly associated with OS and PFS and correlated with the number of CTCs [98].
In a recent study, Pierga et al. showed that ≥1 CTC/7.5 ml was a strong prognostic factor for PFS (P < 0.0001), while the threshold of ≥5 CTCs/7.5 ml was statistically significant for PFS and OS (P = 0.03) in multivariate analysis [18].
CTCs Associate with Axillary Lymph Node Metastasis
The main reason why CTCs are related with shorter OS and PFS is the high incidence of metastasis in patients with CTCs. The risk of progression for patients with ≥5 CTCs/7.5 ml was several times higher than that for patients with 0–4 CTCs/7.5 ml at the same time point. Multivariate analysis showed that axillary lymph node metastasis, serum CA15-3-positivity, and the presence of EpCAM-, CK19-, and hMAM-positive CTCs had a significant impact on PFS, while axillary lymph node metastasis and the presence of EpCAM-, CK19-, and hMAM-positive CTCs had a significant impact on OS.
CTCs and Histological Grade
CTC positivity is significantly associated with high histological grade, tumor size, and nodal status of breast cancer. Detection of CTCs in the peripheral blood indicates a poor prognosis in patients with primary breast cancer. Larger clinical studies are required to further evaluate the role of these markers in clinical practice [94].
CTCs and Clinical Treatment Response
CTC follow-up was considered as an indicator of treatment efficacy. Some studies found a correlation between a reduction of CTCs upon therapy initiation and the final tumor response [99, 100].
During chemotherapy, >6 CTCs/7.5 ml was correlated with a poorer prognosis in patients with metastatic disease. CTC levels, rather than the presence of CTCs, were associated with PFS and showed borderline significance in terms of OS. The differential prognoses and OS rates in patients with and without elevated CTCs before and at the end of chemotherapy are of particular interest in patients with no clinical evidence of metastasis [101].
Pachmann et al. evaluated the number of CTCs prior to each chemotherapy cycle and at the completion of treatment in 91 breast cancer patients [102]. CTCs were detected using the MAINTRAC method. Three groups of CTC changes were recorded: a ≥10-fold decrease, marginal change, or a ≥10-fold increase. Relapses were seen in 1/28 patients with CTC decreases, 5/30 patients with minimal change, and 14/33 with a CTC increases, demonstrating that an increase in CTC level of ≥10-fold predicted early breast cancer relapse. Similarly, Pachmann et al. showed that escalating numbers of CTCs during tamoxifen treatment were strong predictors of relapse [102].
In another study, advanced breast cancer patients were treated with docetaxel chemotherapy, and the treatment response was evaluated by [18F] 3′-deoxy-3′-fluorothymidine positron emission tomography (FLT-PET) and levels of CTCs. In the individuals in whom CTCs could be detected, a decreased CTC count correlated with reduced FLT-PET signal within 2 weeks [103].
Conclusion and Future Perspective
CTCs have been shown to play prognostic and diagnostic roles and to be associated with PFS, DFS, and OS in breast cancer patients. Early breast cancer patients with CTCs are at high risk of metastasis. Recent results have also demonstrated a correlation between the presence of CTCs and poor histological grade of primary tumors. Evaluation of CTCs during treatment can provide information on treatment efficacy, as well as recurrence risk. Moreover, analysis of CTC molecular characteristics can provide information on protein targets for treatment and chemoresistant profiles. However, further progress is needed before CTCs can be applied as a powerful tool for the diagnosis and prognosis of breast cancer, including determining specific markers for breast CTCs, developing highly sensitive and specific methods to detect CTCs, and exploring the molecular characterization of CTCs, especially in terms of CTC marker profiles related to cancer progress, recurrence, and metastasis. However, rapid increases in breast CTC research will allow CTCs to become powerful tools for use in breast cancer diagnosis and prognosis in the near future.
References
Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics. CA Cancer J Clin. 2005;55:74–108.
Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277–300.
Boyages J, Chua B, Taylor R, Bilous M, Salisbury E, Wilcken N, et al. Use of the St Gallen classification for patients with node-negative breast cancer may lead to overuse of adjuvant chemotherapy. Br J Surg. 2002;89:789–96.
Boyages J, Taylor R, Chua B, Ung O, Bilous M, Salisbury E, et al. A risk index for early node-negative breast cancer. Br J Surg. 2006;93:564–71.
Colomer R, Vinas G, Beltran M, Izquierdo A, Lluch A, Llombart-Cussac A, et al. Validation of the 2001 St Gallen risk categories for node-negative breast cancer using a database from the Spanish Breast Cancer Research Group (GEICAM). J Clin Oncol. 2004;22:961–2.
Lundin J, Lehtimaki T, Lundin M, Holli K, Elomaa L, Turpeenniemi-Hujanen T, et al. Generalisability of survival estimates for patients with breast cancer—a comparison across two population-based series. Eur J Cancer. 2006;42:3228–35.
Olivotto IA, Bajdik CD, Ravdin PM, Speers CH, Coldman AJ, Norris BD, et al. Population-based validation of the prognostic model ADJUVANT! for early breast cancer. J Clin Oncol. 2005;23:2716–25.
Perou CM, Sorlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, et al. Molecular portraits of human breast tumours. Nature. 2000;406:747–52.
Foekens JA, Atkins D, Zhang Y, Sweep F, Harbeck N, Paradiso A, et al. Multicenter validation of a gene expression-based prognostic signature in lymph node-negative primary breast cancer. J Clin Oncol. 2006;24:1665–71.
Wang YX, Klijn JGM, Zhang Y, Sieuwerts A, Look MP, Yang F, et al. Gene-expression pro-files to predict distant metastasis of lymph-node-negative primary breast cancer. Lancet. 2005;365:671–9.
West M, Blanchette C, Dressman H, Huang E, Ishida S, Spang R, et al. Predicting the clinical status of human breast cancer by using gene expression profiles. Proc Natl Acad Sci U S A. 2001;98:11462–7.
Gupta GP, Massagué J. Cancer metastasis: building a framework. Cell. 2006;127(4):679–95.
Allard WJ, Matera J, Miller MC, Repollet M, Connelly MC, Rao C, et al. Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin Cancer Res. 2004;10(20):6897–904.
De Giorgi U, Valero V, Rohren E, Dawood S, Ueno NT, Miller MC, et al. Circulating tumor cells and [18F]fluorodeoxyglucose positron emission tomography/computed tomography for outcome prediction in metastatic breast cancer. J Clin Oncol. 2009;27(20):3303–11.
Hayes DF, Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Miller MC, et al. Circulating tumor cells at each follow-up time point during therapy of metastatic breast cancer patients predict progression-free and overall survival. Clin Cancer Res. 2006;12(14 Pt 1):4218–24.
Budd GT, Cristofanilli M, Ellis MJ, Stopeck A, Borden E, Miller MC, et al. Circulating tumor cells versus imaging-predicting overall survival in metastatic breast cancer. Clin Cancer Res. 2006;12:6403–9.
Dawood S, Broglio K, Valero V, Reuben J, Handy B, Islam R, et al. Circulating tumor cells in metastatic breast cancer from prognostic stratification to modification of the staging system? Cancer. 2008;113:2422–30.
Pierga JY, Hajage D, Bachelot T, Delaloge S, Brain E, Campone M, et al. High independent prognostic and predictive value of circulating tumor cells compared with serum tumor markers in a large prospective trial in first-line chemotherapy for metastatic breast cancer patients. Ann Oncol. 2012;23(3):618–24.
Braun S, Hepp F, Kentenich CR, Janni W, Pantel K, Riethmüller G, et al. Monoclonal antibody therapy with edrecolomab in breast cancer patients: monitoring of elimination of disseminated cytokeratin-positive tumor cells in bone marrow. Clin Cancer Res. 1999;5:3999–4004.
Giuliano M, Giordano A, Jackson S, Hess KR, De Giorgi U, Mego M, et al. Circulating tumor cells as prognostic and predictive markers in metastatic breast cancer patients receiving first-line systemic treatment. Breast Cancer Res. 2011;13(3):R67.
Zheng S, Lin H, Liu JQ, Balic M, Datar R, Cote RJ, et al. Membrane microfilter device for selective capture, electrolysis and genomic analysis of human circulating tumor cells. J Chromatogr A. 2007;1162(2):154–61.
Schram ED, Kolatkar A, Yoshioka C, Scuderi R, Lazar D, Malchiodi M, et al. Comparative morphometric analysis of breast-circulating tumor cells and their corresponding solid tumor cytology: a case study. J Clin Oncol. 2010;28(15s).
Ota I, Li XY, Hu Y, Weiss SJ. Induction of a MT1-MMP and MT2-MMP-dependent basement membrane transmigration program in cancer cells by Snail1. Proc Natl Acad Sci U S A. 2009;106(48):20318–23.
Bonnomet A, Brysse A, Tachsidis A, Waltham M, Thompson EW, Polette M, et al. Epithelial-to-mesenchymal transitions and circulating tumor cells. J Mammary Gland Biol Neoplasia. 2010;15(2):261–73.
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY, et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell. 2008;133(4):704–15.
Bao H, Burke PA, Chen X, Shi X, Czapiga M, Li Y, et al. Analysis and characterization of subpopulations of circulating tumor cells in patients with breast cancer. J Clin Oncol. 2011;29:2011.
Theodoropoulos PA, Polioudaki H, Agelaki S, Kallergi G, Saridaki Z, Mavroudis D, et al. Circulating tumor cells with a putative stem cell phenotype in peripheral blood of patients with breast cancer. Cancer Lett. 2010;288(1):99–106.
Wang N, Shi L, Li H, Hu Y, Du W, Liu W, et al. Detection of circulating tumor cells and tumor stem cells in patients with breast cancer by using flow cytometry: a valuable tool for diagnosis and prognosis evaluation. Tumour Biol. 2012;33(2):561–9.
Thiery JP. Epithelial-mesenchymal transitions in development and pathologies. Curr Opin Cell Biol. 2003;15(6):740–6.
Pecot CV, Bischoff FZ, Mayer JA, Wong KL, Pham T, Bottsford-Miller J, et al. A novel platform for detection of CK+ and CK− CTCs. Cancer Discov. 2011;1(7):580–6.
Osta WA, Chen Y, Mikhitarian K, Mitas M, Salem M, Hannun YA, et al. EpCAM is overexpressed in breast cancer and is a potential target for breast cancer gene therapy. Cancer Res. 2004;64:5818–24.
Kagan M, Howard D, Bendele T. A sample preparation and analysis system for identification of circulating tumor cells. J Clin Lig Assay. 2002;25:104–10.
Nagrath S, Sequist LV, Maheswaran S, Bell DW, Irimia D, Ulkus L. Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature. 2007;450:1235–9.
Moll R, Divo M, Langbein L. The human keratins: biology and pathology. Histochem Cell Biol. 2008;129:705–33.
Deng G, Herrler M, Burgess D, Manna E, Krag D, Burke JF. Enrichment with anti-cytokeratin alone or combined with anti-EpCAM antibodies significantly increases the sensitivity for circulating tumor cell detection in metastatic breast cancer patients. Breast Cancer Res. 2008;10:R69.
Stott SL, Hsu CH, Tsukrov DI, Yu M, Miyamoto DT, Waltman BA, et al. Isolation of circulating tumor cells using a microvortex-generating herringbone-chip. Proc Natl Acad Sci U S A. 2010;107:18392–7.
Zhao S, Yang H, Zhang M, Zhang D, Liu Y, Liu Y, et al. Circulating tumor cells (CTCs) detected by triple-marker EpCAM, CK19, and hMAM RT-PCR and their relation to clinical outcome in metastatic breast cancer patients. Cell Biochem Biophys. 2013;65(2):263–73.
Tunca B, Egeli U, Cecener G, Tezcan G, Gökgöz S, Tasdelen I, et al. CK19, CK20, EGFR and HER2 status of circulating tumor cells in patients with breast cancer. Tumori. 2012;98(2):243–51.
Dick JE. Breast cancer stem cells revealed. Proc Natl Acad Sci U S A. 2003;100:3547–9.
Hauch S, Zimmermann S, Lankiewicz S, Zieglschmid V, Bocher O, Albert WH. The clinical significance of circulating tumour cells in breast cancer and colorectal cancer patients. Anticancer Res. 2007;27:1337–41.
Hayes DF, Smerage J. Is there a role for circulating tumor cells in the management of breast cancer? Clin Cancer Res. 2008;14:3646–50.
Armstrong AJ, Marengo MS, Oltean S, Kemeny G, Bitting RL, Turnbull JD, et al. Circulating tumor cells from patients with advanced prostate and breast cancer display both epithelial and mesenchymal markers. Mol Cancer Res. 2011;9(8):997–1007.
Kallergi G, Papadaki MA, Politaki E, Mavroudis D, Georgoulias V, Agelaki S. Epithelial to mesenchymal transition markers expressed in circulating tumour cells of early and metastatic breast cancer patients. Breast Cancer Res. 2011;13(3):R59.
Gorges TM, Tinhofer I, Drosch M, Röse L, Zollner TM, Krahn T, et al. Circulating tumour cells escape from EpCAM-based detection due to epithelial-to-mesenchymal transition. BMC Cancer. 2012;12:178.
Raimondi C, Gradilone A, Naso G, Vincenzi B, Petracca A, Nicolazzo C, et al. Epithelial-mesenchymal transition and stemness features in circulating tumor cells from breast cancer patients. Breast Cancer Res Treat. 2011;130(2):449–55.
Reuben JM, Lee BN, Li C, Gao H, Broglio KR, Valero V, et al. Circulating tumor cells and biomarkers: implications for personalized targeted treatments for metastatic breast cancer. Breast J. 2010;16:327–30.
Aktas B, Tewes M, Fehm T, Hauch S, Kimmig R, Kasimir-Bauer S. Stem cell and epithelial-mesenchymal transition markers are frequently overexpressed in circulating tumor cells of metastatic breast cancer patients. Breast Cancer Res. 2009;11(4):R46.
Fehm T, Hoffmann O, Aktas B, Becker S, Solomayer EF, Wallwiener D, et al. Detection and characterization of circulating tumor cells in blood of primary breast cancer patients by RT-PCR and comparison to status of bone marrow disseminated cells. Breast Cancer Res. 2009;11:R59.
Meng S, Tripathy D, Shete S, Ashfaq R, Haley B, Perkins S, et al. HER-2 gene amplification can be acquired as breast cancer progresses. Proc Natl Acad Sci U S A. 2004;101:9393–8.
Bozionellou V, Mavroudis D, Perraki M, Stathopoulou A, Lianidou E, Georgoulias V, et al. Trastuzumab administration can effectively target chemotherapy-resistant cytokeratin- 19 messenger RNA-positive tumor cells in the peripheral blood and bone marrow of patients with breast cancer. Clin Cancer Res. 2004;10:8185–94.
Fehm T, Müller V, Aktas B, Janni W, Schneeweiss A, Stickeler E, et al. HER2 status of circulating tumor cells in patients with metastatic breast cancer: a prospective, multicenter trial. Breast Cancer Res Treat. 2010;124:403–12.
Tewes M, Aktas B, Welt A, Mueller S, Hauch S, Kimmig R, et al. Molecular profiling and predictive value of circulating tumor cells in patients with metastatic breast cancer: an option for monitoring response to breast cancer related therapies. Breast Cancer Res Treat. 2009;115:581–90.
Riethdorf S, Müller V, Zhang L, Rau T, Loibl S, Komor M, et al. Detection and HER2 expression of circulating tumor cells: prospective monitoring in breast cancer patients treated in the neoadjuvant GeparQuattro trial. Clin Cancer Res. 2010;16(9):2634–45.
Ignatiadis M, Rothé F, Chaboteaux C, Durbecq V, Rouas G, Criscitiello C, et al. HER2-positive circulating tumor cells in breast cancer. PLoS One. 2011;6(1):e15624.
Flores LM, Kindelberger DW, Ligon AH, Capelletti M, Fiorentino M, Loda M, et al. Improving the yield of circulating tumour cells facilitates molecular characterisation and recognition of discordant HER2 amplification in breast cancer. Br J Cancer. 2010;102:1495–502.
Pestrin M, Bessi S, Galardi F, Truglia M, Biggeri A, Biagioni C, et al. Correlation of HER2 status between primary tumors and corresponding circulating tumor cells in advanced breast cancer patients. Breast Cancer Res Treat. 2009;118(3):523–30.
Sieuwerts AM, Mostert B, Bolt-de Vries J, Peeters D, de Jongh FE, Stouthard JM, et al. mRNA and microRNA expression profiles in circulating tumor cells and primary tumors of metastatic breast cancer patients. Clin Cancer Res. 2011;17(11):3600–18.
Punnoose EA, Atwal SK, Spoerke JM, Savage H, Pandita A, Yeh RF, et al. Molecular biomarker analyses using circulating tumor cells. PLoS One. 2010;5(9):e12517.
Hartkopf AD, Banys M, Fehm T. HER2-positive DTCs/CTCs in breast cancer. Recent Results Cancer Res. 2012;195:203–15.
Rosenberg R, Gertler R, Friederichs J, Fuehrer K, Dahm M, Phelps R, et al. Comparison of two density gradient centrifugation systems for the enrichment of disseminated tumor cells in blood. Cytometry. 2002;49(4):150–8.
Gertler R, Rosenberg R, Fuehrer K, Dahm M, Nekarda H, Siewert JR. Detection of circulating tumor cells in blood using an optimized density gradient centrifugation. Recent Results Cancer Res. 2003;162:149–55.
Vona G, Sabile A, Louha M, Sitruk V, Romana S, Schütze K, et al. Isolation by size of epithelial tumor cells: a new method for the immunomorphological and molecular characterization of circulating tumor cells. Am J Pathol. 2000;156(1):57–63.
Vona G, Béroud C, Benachi A, Quenette A, Bonnefont JP, Romana S, et al. Enrichment, immunomorphological, and genetic characterization of fetal cells circulating in maternal blood. Am J Pathol. 2002;160(1):51–8.
Vona G, Estepa L, Béroud C, Damotte D, Capron F, Nalpas B, et al. Impact of cytomorphological detection of circulating tumor cells in patients with liver cancer. Hepatology. 2004;39(3):792–7.
Kahn HJ, Presta A, Yang LY, Blondal J, Trudeau M, Lickley L, et al. Enumeration of circulating tumor cells in the blood of breast cancer patients after filtration enrichment: correlation with disease stage. Breast Cancer Res Treat. 2004;86(3):237–47.
Pinzani P, Salvadori B, Simi L, Bianchi S, Distante V, Cataliotti L, et al. Isolation by size of epithelial tumor cells in peripheral blood of patients with breast cancer: correlation with real-time reverse transcriptase-polymerase chain reaction results and feasibility of molecular analysis by laser microdissection. Hum Pathol. 2006;37(6):711–8.
Lara O, Tong X, Zborowski M, Chalmers JJ. Enrichment of rare cancer cells through depletion of normal cells using density and flow-through, immunomagnetic cell separation. Exp Hematol. 2004;32(10):891–904.
Lin HK, Zheng S, Williams AJ, Balic M, Groshen S, Scher HI, et al. Portable filter-based microdevice for detection and characterization of circulating tumor cells. Clin Cancer Res. 2010;16(20):5011–8.
Zabaglo L, Ormerod MG, Parton M, Ring A, Smith IE, Dowsett M. Cell filtration-laser scanning cytometry for the characterisation of circulating breast cancer cells. Cytometry A. 2003;55(2):102–8.
Fleischer RL, Alter HW, Furman SC, Price PB, Walker RM. Particle track etching. Science. 1972;178(4058):255–63.
Rostagno P, Moll JL, Bisconte JC, Caldani C. Detection of rare circulating breast cancer cells by filtration cytometry and identification by DNA content: sensitivity in an experimental model. Anticancer Res. 1997;17(4A):2481–5.
Zheng S, Lin HK, Lu B, Williams A, Datar R, Cote RJ, et al. 3D microfilter device for viable circulating tumor cell (CTC) enrichment from blood. Biomed Microdevices. 2011;13(1):203–13.
Sieuwerts AM, Kraan J, Bolt J, van der Spoel P, Elstrodt F, Schutte M, et al. Anti-epithelial cell adhesion molecule antibodies and the detection of circulating normal-like breast tumor cells. J Natl Cancer Inst. 2009;101(1):61–6.
Königsberg R, Obermayr E, Bises G, Pfeiler G, Gneist M, Wrba F, et al. Detection of EpCAM positive and negative circulating tumor cells in metastatic breast cancer patients. Acta Oncol. 2011;50(5):700–10.
Slade MJ, Smith BM, Sinnett HD, Cross NC, Coombes RC. Quantitative polymerase chain reaction for the detection of micrometastases in patients with breast cancer. J Clin Oncol. 1999;17:870–9.
De Cremoux P, Extra JM, Denis MG, Pierga JY, Bourstyn E, Nos C, et al. Detection of MUC1-expressing mammary carcinoma cells in the peripheral blood of breast cancer patients by real-time polymerase chain reaction. Clin Cancer Res. 2000;6:3117–22.
Reinholz MM, Nibbe A, Jonart LM, Kitzmann K, Suman VJ, Ingle JN, et al. Evaluation of a panel of tumor markers for molecular detection of circulating cancer cells in women with suspected breast cancer. Clin Cancer Res. 2005;11:3722–32.
Zehentner BK, Secrist H, Hayes DC, Zhang X, Ostenson RC, Loop S, et al. Detection of circulating tumor cells in peripheral blood of breast cancer patients during or after therapy using a multigene real-time RT-PCR assay. Mol Diagn Ther. 2006;10:41–7.
Van der Auwera I, Peeters D, Benoy IH, Elst HJ, Van Laere SJ, Prove A, et al. Circulating tumour cell detection: a direct comparison between the Cell Search System, the AdnaTest and CK-19/mammaglobin RT-PCR in patients with metastatic breast cancer. Br J Cancer. 2010;102:276–84.
Markou A, Strati A, Malamos N, Georgoulias V, Lianidou ES. Molecular characterization of circulating tumor cells in breast cancer by a liquid bead array hybridization assay. Clin Chem. 2011;57:421–30.
Zieglschmid V, Hollmann C, Bocher O. Detection of disseminated tumor cells in peripheral blood. Crit Rev Clin Lab Sci. 2005;42:155–96.
Xi L, Nicastri DG, El-Hefnawy T, Hughes SJ, Luketich JD, Godfrey TE. Optimal markers for real-time quantitative reverse transcription PCR detection of circulating tumor cells from melanoma, breast, colon, esophageal, head and neck, and lung cancers. Clin Chem. 2007;53(7):1206–15.
Mayer JA, Pham T, Wong KL, Scoggin J, Sales EV, Clarin T, et al. FISH-based determination of HER2 status in circulating tumor cells isolated with the microfluidic CEE™ platform. Cancer Genet. 2011;204(11):589–95.
Hayashi N, Nakamura S, Yagata H, Shimoda Y, Ota H, Hortobagyi GN, et al. Chromosome 17 polysomy in circulating tumor cells in patients with metastatic breast cancer: a case series. Int J Clin Oncol. 2011;16(5):596–600.
Hu Y, Fan L, Zheng J, Cui R, Liu W, He Y, et al. Detection of circulating tumor cells in breast cancer patients utilizing multiparameter flow cytometry and assessment of the prognosis of patients in different CTCs levels. Cytometry A. 2010;77(3):213–9.
Krivacic RT, Ladanyi A, Curry DN, Hsieh HB, Kuhn P, Bergsrud DE, et al. A rare-cell detector for cancer. Proc Natl Acad Sci U S A. 2004;101(29):10501–4.
Hsieh HB, Marrinucci D, Bethel K, Curry DN, Humphrey M, Krivacic RT, et al. High speed detection of circulating tumor cells. Biosens Bioelectron. 2006;21(10):1893–9.
Alix-Panabières C, Vendrell JP, Pellé O, Rebillard X, Riethdorf S, Müller V, et al. Detection and characterization of putative metastatic precursor cells in cancer patients. Clin Chem. 2007;53(3):537–9.
Alix-Panabières C, Vendrell JP, Slijper M, Pellé O, Barbotte E, Mercier G, et al. Full-length cytokeratin-19 is released by human tumor cells: a potential role in metastatic progression of breast cancer. Breast Cancer Res. 2009;11(3):R39.
Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Matera J, Miller MC, et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med. 2004;351(8):781–91.
Cohen SJ, Punt CJ, Iannotti N, Saidman BH, Sabbath KD, Gabrail NY, et al. Relationship of circulating tumor cells to tumor response, progression-free survival, and overall survival in patients with metastatic colorectal cancer. J Clin Oncol. 2008;26(19):3213–21.
Danila DC, Heller G, Gignac GA, Gonzalez-Espinoza R, Anand A, Tanaka E, et al. Circulating tumor cell number and prognosis in progressive castration-resistant prostate cancer. Clin Cancer Res. 2007;13(23):7053–8.
Cristofanilli M, Hayes DF, Budd GT, Ellis MJ, Stopeck A, Reuben JM, et al. Circulating tumor cells: a novel prognostic factor for newly diagnosed metastatic breast cancer. J Clin Oncol. 2005;23(7):1420–30.
Zhao S, Liu Y, Zhang Q, Li H, Zhang M, Ma W, et al. The prognostic role of circulating tumor cells (CTCs) detected by RT-PCR in breast cancer: a meta-analysis of published literature. Breast Cancer Res Treat. 2011;130(3):809–16.
Zhang L, Riethdorf S, Wu G, Wang T, Yang K, Peng G, et al. Meta-analysis of the prognostic value of circulating tumor cells in breast cancer. Clin Cancer Res. 2012;18(20):5701–10.
Botteri E, Sandri MT, Bagnardi V, Munzone E, Zorzino L, Rotmensz N, et al. Modeling the relationship between circulating tumour cells number and prognosis of metastatic breast cancer. Breast Cancer Res Treat. 2010;122(1):211–7.
Liu MC, Shields PG, Warren RD, Cohen P, Wilkinson M, Ottaviano YL, et al. Circulating tumor cells: a useful predictor of treatment efficacy in metastatic breast cancer. J Clin Oncol. 2009;27(31):5153–9.
Consoli F, Grisanti S, Amoroso V, Almici C, Verardi R, Marini M, et al. Circulating tumor cells as predictors of prognosis in metastatic breast cancer: clinical application outside a clinical trial. Tumori. 2011;97(6):737–42.
Camara O, Rengsberger M, Egbe A, Koch A, Gajda M, Hammer U, Jörke C, et al. The relevance of circulating epithelial tumor cells (CETC) for therapy monitoring during neoadjuvant (primary systemic) chemotherapy in breast cancer. Ann Oncol. 2007;18(9):1484–92.
Pachmann K, Camara O, Kavallaris A, Schneider U, Schünemann S, Höffken K. Quantification of the response of circulating epithelial cells to neoadjuvant treatment for breast cancer: a new tool for therapy monitoring. Breast Cancer Res. 2005;7(6):R975–9.
Serrano MJ, Sánchez-Rovira P, Delgado-Rodriguez M, Gaforio JJ. Detection of circulating tumor cells in the context of treatment: prognostic value in breast cancer. Cancer Biol Ther. 2009;8(8):671–5.
Pachmann K, Camara O, Kohlhase A, Rabenstein C, Kroll T, Runnebaum IB, et al. Assessing the efficacy of targeted therapy using circulating epithelial tumor cells (CETC): the example of SERM therapy monitoring as a unique tool to individualize therapy. J Cancer Res Clin Oncol. 2011;137(5):821–8.
Contractor K, Aboagye EO, Jacob J, Challapalli A, Coombes RC, Stebbing J. Monitoring early response to taxane therapy in advanced breast cancer with circulating tumor cells and [(18)F] 3′-deoxy-3′-fluorothymidine PET: a pilot study. Biomark Med. 2012;6(2):231–3.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer India
About this chapter
Cite this chapter
Van Pham, P. (2014). Breast Circulating Tumor Cells: Potential Biomarkers for Breast Cancer Diagnosis and Prognosis Evaluation. In: Barh, D. (eds) Omics Approaches in Breast Cancer. Springer, New Delhi. https://doi.org/10.1007/978-81-322-0843-3_21
Download citation
DOI: https://doi.org/10.1007/978-81-322-0843-3_21
Published:
Publisher Name: Springer, New Delhi
Print ISBN: 978-81-322-0842-6
Online ISBN: 978-81-322-0843-3
eBook Packages: MedicineMedicine (R0)