Abstract
Neutrophils are essential components of the innate immune system. They participate in a variety of tissue reactions, including antimicrobial responses and damage repair. Neutrophils are exquisitely sensitive to migratory stimuli, which enables them to rapidly home into injured tissues, including the skin, where they exert their effector functions. The latter include the release of preformed mediators from granules, production of reactive oxygen species, and release of DNA traps into the extracellular space. Based on these activities, neutrophils play a crucial role in cutaneous immune responses, and patients with neutrophil defects are prone to bacterial and fungal skin infections. Nevertheless, neutrophils may also directly cause tissue damage, and are the driving force behind the pathology of a number of inflammatory skin conditions. This chapter describes the molecular mechanisms underlying neutrophil function in the skin, and reviews our current understanding of the role of neutrophils in cutaneous biology and disease.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Skin
- Innate immunity
- Neutrophil
- Infection
- Staphylococcus aureus
- Neutrophilic dermatoses and neutrophil deficiencies
1 Introduction
The skin, with its sentinel immune cells, forms the first line of defence against invading pathogens and noxious insults. The presence of specialized immune cell subsets resident in the epidermis, for example, Langerhans cells, coupled with a vast array of antimicrobial peptides secreted by epithelial cells, deters the growth of pathogenic microbes [43, 53, 58]. Although the epidermis and its constituents act as an efficient barrier against microbes and environmental factors, occasionally these mechanisms fail due to either pathogen-mediated neutralisation of host defence or due to physical barrier breach. The detection of these microbes (commensal or pathogenic) and/or tissue damage results in the activation of a pro-inflammatory cascade leading to the rapid recruitment of leukocytes from the bloodstream. Neutrophils are the prime responders to such stimuli due to their high numbers in the circulation and their exquisite sensitivity for chemoattractants. These cells are replete with an array of cytotoxic granules containing preformed mediators that can be rapidly unleashed within tissues. As such, neutrophils are essential for the clearance of bacterial and parasitic pathogens as well as tissue debris. Nevertheless, in cases where the influx of neutrophils persists over prolonged periods of time or where negative regulatory mechanisms fail neutrophils may also contribute to tissue damage. In this chapter we outline the role of neutrophils in cutaneous immune responses and discuss conditions associated with impaired neutrophil functions and their consequences on skin physiology and pathology.
2 Neutrophil Residence in Bone Marrow and Release into Circulation
Neutrophils are short-lived bone marrow-derived cells whose development and release are intimately linked to signals originating at inflammatory sites. Neutrophils are produced in the haematopoietic cords in the bone marrow and their production is tightly regulated by cytokines and a large number of transcription factors [12]. Normal adult human and murine bone marrow produces 1011 and 107 neutrophils per day, respectively [13, 31], with only 1–2% of these cells being present in the circulation. Their production is tightly regulated by the IL-23/IL-17A/G-CSF cytokine axis [70, 108, 130]. IL-23 produced by macrophages in the periphery stimulates γδ T cells and a subset of NKT cells to produce IL-17A, which in turn stimulates production of G-CSF from bone marrow stromal cells leading to granulocytopoiesis [70, 119]. Neutrophil release from the bone marrow is mediated by differential signalling via the CXCR4 and CXCR2 chemokine receptors. CXCL12 expression in osteoblasts results in CXCR4-mediated signalling that promotes retention of neutrophils in the bone marrow, whereas CXCL1/2 expression in endothelial cells facilitates CXCR2-mediated neutrophil egress [35, 76, 106, 109, 120, 122, 133]. During inflammation, G-CSF produced by immune cells in the periphery skews the chemotactic response towards CXCR2 by increasing the expression of CXCL2 on endothelial cells in the bone marrow while reducing both CXCL12 producing osteoblasts [26, 35, 133] resulting in rapid mobilisation of neutrophils into systemic circulation. While in the blood stream, neutrophils have a short half-life (<12 h in adult mice). In addition to their central antimicrobial role (discussed below), during their short circulatory life neutrophils can also perform housekeeping functions such as intraluminal removal of damaged endothelial cells in coordination with Nr4a1-dependent patrolling monocytes [18]. Apoptotic and senescent neutrophils are cleared from the circulation by resident macrophages in the liver, spleen, and bone marrow; a study using radiolabelled neutrophils revealed that all three organs contribute equally in clearing neutrophils [41, 102]. Although terminal trafficking of neutrophils into the intestinal lumen has also been reported [66], its physiological relevance in neutrophil clearance is unknown.
3 Neutrophil Entry into the Skin and Interstitial Dermal Migration
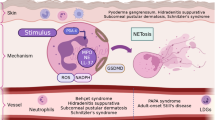
Following an insult in peripheral organs, such as the skin, neutrophils are rapidly recruited into the damaged interstitium. The inflammatory cascade underlying this recruitment has traditionally been viewed as being initiated by skin-resident cells. These cells include dendritic cells, macrophages, and mast cells, as well as epithelial cells and stromal cells, all of which are capable of sensing the presence of tissue damage or pathogens via germline encoded pattern recognition receptors (PRRs) that bind to damage-associated molecular patterns (DAMPs) or pathogen-associated molecular patterns (PAMPs) . In addition, low numbers of neutrophils themselves have been found to scan the noninflamed dermis actively, where they likely serve as first-line responders to tissue damage [71, 90]. Signalling via PRRs in skin-resident cells leads to the release of pro-inflammatory mediators, including the cytokines TNFα, IL-1β, IL-17 family members, histamine, and/or lipid mediators (Fig. 9.1a). This is followed by the activation of blood vessel endothelium and recruitment of neutrophils into the interstitium via the ‘leukocyte adhesion cascade’.
Neutrophils in the skin. Schematic representation of molecular and cellular processes initiated during neutrophil antibacterial response in the dermis. The antibacterial response is initiated by sensing of DAMPs and PAMPs by resident cells in the interstitium (a) resulting in rapid recruitment of neutrophils from circulation via leukocyte adhesion cascade (b). These recruited neutrophils initiate multiple antibacterial responses (c) and activate endothelial cells for recruitment of monocytes and other immune cells (d). In addition to these functions, neutrophils can also transport microbial pathogens to draining lymph nodes via afferent lymphatics
Neutrophil recruitment into the dermis, as in other organs, occurs through postcapillary venules in a process that closely follows the steps of the classical ‘leukocyte adhesion cascade’ [64, 69, 96]. Under inflammatory conditions endothelial cells increase their expression of E- and P-selectins . This allows circulating neutrophils, which express P-selectin glycoprotein ligand-1, to interact with endothelial selectins, and results in neutrophil tethering and rolling along the blood vessel wall (Fig. 9.1b). Neutrophils are then able to interact with chemokines that are presented on the surface of endothelial cells. This triggers conformational changes in integrins, such as LFA-1 and Mac-1 , on neutrophils resulting in high affinity binding of integrins to ICAM1/2 on endothelial cells and, consequently, firm arrest (Fig. 9.1b). Additionally, under some experimental conditions, VLA-4 is also induced on neutrophils and this can interact with VCAM-1 expressed on the endothelium [60]. Thereafter, neutrophils crawl along the luminal surface of the venules in an integrin-dependent manner and search for a preferred point of transmigration. Focal deposits of chemokines along the blood vessel wall are thought to play a role in directed movement, although the precise mechanisms underlying this process are incompletely understood. Firmly adherent neutrophils then traverse the blood vessel wall and enter the dermal interstitial space. This involves negotiating multiple barriers. Initially, the penetration of the endothelial barrier requires neutrophils to either pass through the body of endothelial cells (transcellular pathway), or through the tight junctions between them (paracellular pathway). Upon reaching the subendothelial space, neutrophils migrate along pericyte processes in an integrin-dependent manner before exiting through gaps between pericytes [100]. These gaps become enlarged in response to cytokine stimulation via remodelling of basement membrane proteins [128, 131] and are associated with increased expression of integrin ligands and neutrophil-attracting chemokines on the pericyte [100].
Upon entering the dermis, neutrophils show strong directional migration towards gradients of chemotactic molecules emanating from the site of injury [79, 118]. Capillary and arteriolar pericytes have also been implicated in guiding neutrophils to the site of injury by upregulating ICAM-1 on the pericyte cell surface and releasing macrophage migration-inhibitory factor (MIF) [118]. Depending on the type of noxious insult, a vast array of chemotactic molecules may be present in the interstitium; although activation of endothelial cells and macrophages induces production of various chemokines, a large number of DAMPs and PAMPs emanate from damaged cells or pathogens at the site of injury. It is important to note that recent studies have shown the existence of positive feedback loops which augment the attraction of neutrophils towards the site of injury. For example, neutrophil sensing of nicotinamide dinucleotide (NAD+), which is released from dying cells and acts as a DAMP, has been shown to amplify the accumulation of neutrophils following sterile injury in the dermis [90]. Furthermore, neutrophil-derived leukotriene-B4 (LTB4) acts in a feedforward manner to increase the radius of attraction of the neutrophil population [67, 92]. The multiple chemoattracting cues provided to neutrophils in the interstitial space are integrated, and their migratory behavior is determined by a hierarchical arrangement of signalling patterns [79, 96]. For instance, signalling via formyl peptide receptor-1 supersedes signalling induced via chemokine receptor CXCR2 [79, 96]. Such a hierarchical organisation of signalling patterns assists in the guidance of neutrophils to the precise region of insult.
4 Neutrophil Functions at the Site of Tissue Injury
Neutrophils arriving from circulation need to neutralize invading microorganisms rapidly and clear tissue debris. This is achieved via particle uptake (phagocytosis), release of prestored granule contents, generation of reactive oxygen species (ROS), and the deployment of neutrophil extracellular traps (NETs ; Fig. 9.1c). Circulating neutrophils are transcriptionally inactive but their interaction with pathogens reactivates transcription and triggers the secretion of chemotactic factors that are crucial for recruitment of other immune cells [107]. The amount of chemokines or inflammatory mediators secreted per neutrophil is relatively small compared to other resident immune cells, but their significantly higher numbers at the site of injury compensate for this low expression [88].
4.1 Phagocytosis
During inflammation, neutrophils take up particulates such as microorganisms into membrane-bound vesicles (phagosomes) in a process termed phagocytosis, and particles are then destroyed inside these organelles. The initial process of uptake is strongly enhanced by soluble factors such as antibody or activated complement that mark (‘opsonise’) particulates for phagocytosis. Neutrophils express a range of Fc receptors, in particular FcγIIA and FcγIIIB [42], and aggregation of these receptors by antibodies that are presented in multimeric arrays, such as on microbial cell surfaces, causes neutrophil activation [84, 103]. Additionally, binding of complement-opsonized particles to CR3 (αMβ2-integrin/CD11b/Mac-1) leads to integrin ‘outside-in’ signalling and induction of phagocytosis [72]. Furthermore, phagocytosis can also be initiated nonopsonically through receptors that bind directly to microbial components, the details of which depend on the specific microorganism and the surface products that it expresses. In particular, recognition of microbial carbohydrate structures by neutrophil membrane receptors that contain lectin domains, such as dectin-1, Clec-2, or CR3 can mediate phagocytosis of a range of microorganisms [47, 63, 104]. Once internalised, the antimicrobial arsenal of the neutrophil is focused on the particulates by fusion of the phagosome with neutrophil granules.
4.2 Granule Composition and Release
The direct antimicrobial activity of neutrophils is largely due to the coordinated release of their granule cargo either into the extracellular milieu or the nascent phagosome. In both cases, high concentrations of antimicrobial proteins, as well as reactive oxygen species, are unleashed [2]. The formation of granules and synthesis of their contents occurs in the bone marrow and is tightly regulated during the development and maturation of neutrophils. A list of granules and their antimicrobial contents as well as the mechanism of action are detailed in Table 9.1. The release of these granules is a tightly regulated process that ensures safe delivery of their content only in close vicinity of pathogens, thereby limiting collateral damage to host cells. The mechanism regulating differential mobilization is incompletely understood though the role of intracellular calcium levels has been implicated in the process [110]. Another type of vesicle is secretory vesicles, which are not strictly granules, but are formed by endocytosis of plasma membrane during neutrophil maturation and contain proteins crucial for homing and sensing pathogens [2].
Although neutrophils possess a striking range of granule proteins, their biological activities can be summarised largely based on their mechanism of antimicrobial action [2]. The first group includes cationic proteins that bind directly to bacterial membranes and mediate lysis; these include α-defensins [44], LL37 [125], and bactericidal/permeability-increasing protein (BPI) [68]. In contrast, a number of proteins with enzymatic activity, such as lysozyme [44], neutrophil elastase (NE) [8, 124], and cathepsin G [124] exert antimicrobial activity through enzymatic degradation of microbial products. The products targeted by these enzymes may be essential components of the cell itself (e.g. lysozyme M activity against the Micrococcus luteus peptidoglycan cell wall [45]; neutrophil elastase-mediated degradation of the Escherichia coli outer membrane protein OmpA [8]), or may represent extracellular virulence factors (e.g. neutrophil elastase-mediated degradation of S. flexerni secreted proteins/toxins [132]). Lastly, antimicrobial activity may also be mediated by sequestration of essential nutrients; a key illustration of this is the multiple approaches used by neutrophils to limit iron availability. Lactoferrin, a component of secondary granules, has direct iron-binding activity, which limits free iron available to growing microorganisms [78, 91]. Alternatively, lipocalin (NGAL) is able to bind to a range of bacterial siderophores and inhibits bacterial iron uptake [39]. These multiple proteins act in concert with reactive oxygen species, which are also generated during degranulation, and exert potent microbicidal activity [89].
The second major antimicrobial component of the degranulation response is the production of reactive oxygen species (reviewed in [7, 32, 134]). The assembly of functional NADPH oxidase (phagocytic oxidase/phox) enzyme from individual components is the initiating step in ROS formation. Under resting conditions, Gp91phox and p22phox reside within secondary granules and the other key components of the enzyme, including p47phox, p67phox, and p40phox and the small GTPase rac2, are present within the cytoplasm. Upon neutrophil activation, the cytoplasmic components are translocated to the phagosome that has formed by the fusion of the plasma membrane and granules (or to the plasma membrane), thereby bringing all components of the enzyme together [7, 134]. On becoming functional, NADPH oxidase catalyzes the key initial step in the generation of ROS, which is the conversion of molecular oxygen (O2) to superoxide (2O2 −). Once formed, superoxide rapidly dismutates to hydrogen peroxide (H2O2) and this is acted upon by myeloperoxidase (MPO) , which is also released into the phagosome during degranulation, to form hypochlorous acid (HOCl−). In addition, a range of oxidant species including chloramine, hydroxyl radicals (OH•), singlet oxygen (1O2), and ozone (O3) is formed [127]. Although all of the different ROS produced have varying degrees of antimicrobial activity, hypochlorous acid is particularly potent at causing oxidative damage to most biological molecules. It is generally believed that this powerful oxidation is one of the key mediators of neutrophil antimicrobial activity [32].
4.3 Neutrophil Extracellular Traps (NETs)
Neutrophils can also ‘ensnare’ pathogens by releasing DNA-based molecular traps (referred to as neutrophil extracellular traps or NETs ). These released DNA NETs are associated with histones, myeloperoxidase, neutrophil elastase, lactoferrin, pentraxin-related protein 3 (PTX3), matrix-metalloproteases (MMPs), and other granule proteins all of which can orchestrate antimicrobial functions [15, 16, 137]. DNA NETs can act as molecular scaffolds that entrap pathogens and enhance their interaction with microbicidal proteins. Although neutrophils mostly expel genomic DNA for NET formation [15], mitochondrial DNA can also be used for this process [139]. The process of NET formation is generally considered to be ‘suicidal’ and is also referred to as ‘NETosis ’, similar to other cell death mechanisms such as apoptosis, necrosis, and pyroptosis [16, 137].
The molecular processes required for NET formation are incompletely understood. It has been shown that activation of NADPH oxidase and subsequent generation of ROS is crucial for NET formation [15, 40, 80]. Upon ROS generation, the granule enzymes NE and MPO translocate to the nucleus and degrade histones [93]. Uncondensed nuclear material is then released by perforation of the cell membrane leading to cell lysis [15, 16, 137]. Furthermore, there is increasing evidence for a second type of NETosis that does not require cell membrane perforation and lysis but instead uses vesicular exportation of DNA for release, referred to as ‘vital NETosis ’ [137]. The released ‘anuclear’ neutrophil cytoplast retains pathogen-sensing, chemotactic migration, and phagocytosing capabilities [138]. These differences between ‘suicidal’ and ‘vital’ forms of NETosis could be due to differences in the activating signal or in the ‘maturity’ of neutrophils. Nonetheless, both forms of NETosis are important for mediating neutrophil antibacterial responses [137].
5 Neutrophil Recirculation and Transport of Antigen
Increasing evidence suggests that neutrophils may enter peripheral tissues, including the skin, even under noninflammatory conditions, and that they may traffic into lymphatic vessels to draining lymph nodes both during steady state and inflammation. During their classic lymphatic cannulation studies in sheep, Morris and colleagues observed neutrophils in lymph fluid, in both the lymphatics draining the hind limb [52] and in the efferent lymph of the popliteal lymph node [50]. It was, however, not until the advent of multiphoton microscopy that such ‘scouting’ neutrophils were directly observed within the skin during steady state [90]. Labeled neutrophils intravenously injected into sheep could be recovered from the lymphatics within 3–4 h of intravenous injection, indicating that neutrophil recirculation from the blood into the lymph occurs rapidly, in contrast to the delayed kinetics (6–20 h) of lymphocytes [90]. Although it remains uncertain what proportion of neutrophils recirculates under physiological conditions, a clear consequence of this phenomenon is that, in addition to their role as rapid responders, neutrophils may also serve as a primary sensor population for damage and infection [90].
In addition to this physiological recirculation, numerous studies have reported the trafficking of pathogen-bearing neutrophils from the site of infection to draining lymph nodes, including from the skin to cutaneous lymph nodes [1, 11, 73, 121, 136], and that such migration facilitates antigen delivery for ongoing adaptive immune responses [1, 6, 121]. Only a relatively small proportion of recruited neutrophils appears to traffic into draining lymphatics [121], and it remains unclear what differentiates this population from the majority that remain and die within the infected tissue. Similarly, the cellular and molecular pathways that regulate neutrophil entry into the lymphatics remain largely unknown. Although it has been reported that neutrophil migration to the lymph node is CCR7-dependent [5], the potential role for other chemokine receptors has yet to be investigated. CXCR2 and CXCR4 are of particular interest, given their well-documented role in coordinating neutrophil trafficking from the bone marrow [35, 76]. Moreover, CXCR2 has already been implicated in neutrophil trafficking directly into the lymph node from the blood during inflammation [14], whereas CXCR4 is upregulated on the surface of neutrophils following their extravasation into inflammatory tissue [135].
6 Neutrophil Apoptosis and Resolution of Inflammation
Neutrophils are generally regarded as pro-inflammatory cells but a large body of evidence indicates that they also have a crucial role in resolving inflammation. During the resolution phase, due to their phagocytic activity, neutrophils remove cellular debris from the site of injury, paving the way for tissue repair [64]. MMP-9 released by neutrophils can degrade various intracellular cytoskeletal components released from damaged cells, thereby clearing DAMPs [64], and can also promote vascularisation of tissue by activating vascular endothelial growth factor (VEGF) [9, 25]. Neutrophils also initiate recruitment of monocytes via activating endothelial cells to produce CCL2 or by releasing azurocidin, LL37 and cathepsin G that can activate monocyte formyl peptide receptors (FPRs) (Fig. 9.1d) [114, 116, 117]. These monocytes then phagocytose apoptotic neutrophils during tissue resolution.
Although neutrophils are short-lived cells, factors released at the site of injury can enhance their viability. For example, the presence of LPS or GM-CSF increases the expression of antiapoptotic proteins Mcl-1 and A1, thereby leading to an extended neutrophil lifespan (detailed in [2, 62]). In contrast, the neutralisation of noxious insults and microbes leads to decreased signalling from these pathways and results in increased neutrophil apoptosis. At the initial stages of apoptosis neutrophils secrete ‘find me’ signals to guide monocytes and macrophages to the site of apoptosis and later display ‘eat me’ signals for rapid phagocytic uptake [115]. Phagocytosis of apoptotic neutrophils by tissue-resident macrophages results in decreased production and release of IL-23 leading to decreased IL-17A levels. The reduced IL-17A results in less secretion of G-CSF by bone marrow stromal cells, decreased granulocytopoiesis, and increased retention of mature neutrophils in the bone marrow [119].
Soluble lipid mediators are important both in neutrophil recruitment and in termination of the acute inflammatory response. At early stages of inflammation, prostaglandins (prostaglandin I2) and leukotrienes (LTB4) are produced, resulting in rapid recruitment of neutrophils, but as inflammation progresses, a molecular switch in lipid metabolism results in production of lipoxins, resolvins, and protectins [2, 64, 111]. These mediators assist in monocyte influx and suppress neutrophil recruitment and antimicrobial functions. In addition, lipid mediators result in nonphlogistic uptake of apoptotic cells by monocytes and macrophages resulting in IL-10 and TGF-β production. Together, these processes promote the resolution of inflammation and tissue restitution.
7 Neutrophil Deficiencies
Deficiencies in either the absolute number of circulating neutrophils or their correct functioning typically lead to enhanced susceptibility to infection, particularly within the skin. This is highlighted in acquired neutropenia, such as in patients undergoing chemotherapy, where the severity of neutrophil deficiency is an important determinant of susceptibility to infection with many bacterial and fungal pathogens [10, 74]. A number of molecular deficits that affect neutrophil development or release from the bone marrow can lead to primary neutropenia or functional defects (reviewed in [34]). Such genetic deficiencies include: chronic granulomatous disease (CGD) or MPO deficiency in which ROS generation is affected [54, 127], compromised granule formation/function (e.g. Chédiak–Higashi syndrome) [4], or leukocyte adhesion deficiency syndromes, which result in decreased neutrophil recruitment [51]. Furthermore, defects in cytokine pathways that contribute to the recruitment of neutrophils also manifest as increased susceptibility to cutaneous infection. For example, mutations that lead to either diminished IL-17 production or IL-17 signalling, including in IL17RA, TYK2, STAT3, or STAT1, predispose to chronic mucocutaneous candidiasis [83, 101, 126]. Taken together, the increased susceptibility to infection, particularly to a subset of microorganisms that includes Staphylococcus aureus, Pseudomonas aeruginosa, Candida albicans, and Aspergillus spp, highlights the critical importance of neutrophils in protection from these pathogens.
8 Neutrophilic Dermatoses
Although neutrophils are central to the acute inflammatory response that is initiated following tissue damage or pathogen entry, in some individuals inappropriate recruitment and activation of these cells may occur. This can then contribute to pathology in the absence of an identifiable cause. In some diseases, for example, psoriasis, neutrophils are found in large numbers, but it is unclear whether this represents an epiphenomenon or a true pathogenic event [95, 123]. Alternatively, autoimmune conditions such as lupus erythematosus, cutaneous vasculitides, and neutrophilic dermatoses are prominent examples of conditions where neutrophils appear to contribute to pathology. In vasculitides, the skin can either be the primary organ of involvement, for example, in small vessel leukocytoclastic vasculitis, or part of systemic syndromes, such as in Wegener’s granulomatosis where autoantibodies are directed against neutrophils [19, 59]. These conditions have diverse aetiology and their detailed discussion is beyond the scope of this chapter.
Neutrophilic dermatoses are a set of clinical entities that share the common feature of neutrophil influx within the skin in the absence of infection. Several conditions are recognised, but those most commonly described are Sweet’s syndrome (SS), pyoderma gangrenosum (PG), and subcorneal pustular dermatosis (SCD; reviewed in [28]). In SS and PG, neutrophil influx occurs predominantly within the dermis, although in PG it may extend to subcutaneous tissues. Variable influx of other inflammatory cell populations, including eosinophils, lymphocytes, and giant cells may also occur depending on the subclassification. In contrast, in SCD neutrophils are confined primarily to the subcorneum [27, 28, 105].
The underlying aetiology of neutrophilic dermatoses is unclear; however, there are a number of striking disease correlations. These include, depending on the classification: prior infections, drug treatment, inflammatory bowel disease, autoimmune conditions, and malignancies [27, 28]. A number of lines of evidence implicate Th1 and/or Th17 inflammatory cytokines in mediating disease, at least in SS and PG. Observations supporting Th1 involvement include increased serum IL-2 and IFNγ [46], whereas elevated levels of IL-1, IL-3, IL-6, and IL-8 have also been noted [27, 77]. A clear pro-pathogenic role for TNF, in particular, has been reported, and a number of studies have shown the benefit of anti-TNF therapy [17, 28]. More recently the expression of IL-17 and/or IL-23 in SS and PG has been reported, suggesting potential involvement of this cytokine axis in pathology [38, 49, 77]. Consistent with this, case studies that report the successful use of anti-IL-12/23 therapy for the treatment of PG have begun to emerge [37, 49]. Whether this cytokine–disease link holds true in larger studies remains to be seen.
9 Neutrophils During Cutaneous Infection: S. aureus as a Model Pathogen
The distinct subset of pathogens that cause disease in individuals with neutrophil deficiencies suggests that there are shared protective pathways involved in the responses to these species. In the following section we use the major human pathogen S. aureus as an example to illustrate the role of neutrophils in cutaneous immunity.
Data from clinical, rodent, and in vitro studies show remarkable agreement in support of the essential role of neutrophils in protection from S. aureus. As described in Sect. 9.7 above, a range of defects in either neutrophil number or function is associated with increased susceptibility to cutaneous S. aureus infection. Note that this is mirrored in rodents, as mice deficient in NADPH-oxidase components develop spontaneous cutaneous lesions, or fail to control experimental infection [36, 57, 99]. Moreover, depletion of circulating neutrophils prevents control of cutaneous S. aureus infection [85]. In vitro, phagocytosis of S. aureus by neutrophils is enhanced by serum opsonins [3, 61, 94], and their subsequent killing is mediated by the NADPH-oxidase dependent production of ROS [36, 75]. A number of granule proteins have direct anti-Staphylococcal activity in vitro. These include the α-defensins HNP1-3, as well as LL-37, PG-1, S100A8/9, and lactoferrin [21, 29, 125], whereas others, including azurocidin and lysozyme are ineffective [21]. Lastly, extracellular DNA NETs are released by neutrophils in response to S. aureus. The NETs are capable of trapping bacteria, and the NET-associated histones and granule proteins then mediate killing [15, 40, 98].
Rodent models have been particularly informative in identifying the pathways that lead to neutrophil recruitment. Initial recognition of S. aureus infection by resident immune sentinel cells occurs via TLR2, NOD2, and the NLRP3 inflammasome [55, 82, 112], resulting in the induction of pro-inflammatory cytokines. Dominant amongst the initial cytokines produced are IL-1β [81, 82] and IL-17A/F [23], both of which have been shown to drive neutrophil accumulation and activation in a range of disease models [65, 113]. Both humans [97, 129] and mice [22, 24, 81] with deficits in the TLR/IL-1β signalling pathway control S. aureus infection poorly. The initial source of IL-1β in the skin is unclear: whereas inflammasome-dependent IL-1β release in response to S. aureus in vitro is typically associated with monocyte/macrophages [30, 86, 112], in vivo much of the IL-1β produced appears to be derived from recruited neutrophils, at least during later stages [22]. As with IL-1β, deficiencies in IL-17 signalling (mutations in STAT3 and TYK2) also predispose to S. aureus infection [23, 33, 48, 56, 83, 87]. This IL-17A and IL-17F is produced by skin resident γδ-T cells in an IL-23–dependent manner, and during S. aureus infection γδ-T-cell–deficient mice show decreased neutrophil recruitment [23]. Ultimately, the combination of IL-1β and IL-17A/F stimulates local production of CXCL1 and CXCL2 from resident cells. These chemokines specifically recruit neutrophils to the infection site, and under normal circumstances this results in bacterial clearance [22, 23, 81, 82].
10 Concluding Remarks
The essential role of neutrophils as a key player in the innate immune response to injury/infection within the skin is well recognised, however, it is becoming clear that this is only one aspect in their short life span. Neutrophils under homeostasis perform myriad functions ranging from removing damaged endothelial cells [18] to regulating the stem cell pool in the bone marrow [20]. In particular, the identification of ‘scouting’ neutrophils that are capable of sensing the presence of tissue damage [90], and the finding that they can recirculate [50, 52, 90] and influence the development of immune responses is furthering our understanding of skin immunity. Although these findings suggest that neutrophils may have previously unrecognised functions in the initiation of both innate and adaptive immune responses, the fate of these neutrophils in the lymph nodes is not completely understood. Moreover the presence of these ‘scouting’ neutrophils in uninflamed dermis challenges our current understanding about neutrophil recruitment in the skin. Taken together, the increasingly apparent complexity of neutrophil biology argues that continued investigation into this often underestimated cell is likely to lead to improved strategies for treatment of a broad range of cutaneous diseases.
References
Abadie V, Badell E, Douillard P, Ensergueix D, Leenen PJ, Tanguy M, Fiette L, Saeland S, Gicquel B, Winter N (2005) Neutrophils rapidly migrate via lymphatics after Mycobacterium bovis BCG intradermal vaccination and shuttle live bacilli to the draining lymph nodes. Blood 106(5):1843–1850
Amulic B, Cazalet C, Hayes GL, Metzler KD, Zychlinsky A (2012) Neutrophil function: from mechanisms to disease. Annu Rev Immunol 30:459–489
An J-H, Kurokawa K, Jung D-J, Kim M-J, Kim C-H, Fujimoto Y, Fukase K, Coggeshall KM, Lee BL (2013) Human SAP is a novel peptidoglycan recognition protein that induces complement-independent phagocytosis of Staphylococcus aureus. J Immunol 191(6):3319–3327
Barbosa MD, Nguyen QA, Tchernev VT, Ashley JA, Detter JC, Blaydes SM, Brandt SJ, Chotai D, Hodgman C, Solari RC, Lovett M, Kingsmore SF (1996) Identification of the homologous beige and Chediak-Higashi syndrome genes. Nature 382(6588):262–265
Beauvillain C, Cunin P, Doni A, Scotet M, Jaillon S, Loiry ML, Magistrelli G, Masternak K, Chevailler A, Delneste Y, Jeannin P (2011) CCR7 is involved in the migration of neutrophils to lymph nodes. Blood 117(4):1196–1204
Beauvillain C, Delneste Y, Scotet M, Peres A, Gascan H, Guermonprez P, Barnaba V, Jeannin P (2007) Neutrophils efficiently cross-prime naive T cells in vivo. Blood 110(8):2965–2973
Bedard K, Krause K-H (2007) The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol Rev 87(1):245–313
Belaaouaj A, Kim KS, Shapiro SD (2000) Degradation of outer membrane protein A in Escherichia coli killing by neutrophil elastase. Science 289(5482):1185
Bergers G, Brekken R, McMahon G, Vu TH, Itoh T, Tamaki K, Tanzawa K, Thorpe P, Itohara S, Werb Z, Hanahan D (2000) Matrix metalloproteinase-9 triggers the angiogenic switch during carcinogenesis. Nat Cell Biol 2(10):737–744
Bodey GP, Buckley M, Sathe YS, Freireich EJ (1966) Quantitative relationships between circulating leukocytes and infection in patients with acute leukemia. Ann Intern Med 64(2):328–340
Bonneau M, Epardaud M, Payot F, Niborski V, Thoulouze MI, Bernex F, Charley B, Riffault S, Guilloteau LA, Schwartz-Cornil I (2006) Migratory monocytes and granulocytes are major lymphatic carriers of Salmonella from tissue to draining lymph node. J Leukoc Biol 79(2):268–276
Borregaard N (2010) Neutrophils, from marrow to microbes. Immunity 33(5):657–670
Boxio R, Bossenmeyer-Pourie C, Steinckwich N, Dournon C, Nusse O (2004) Mouse bone marrow contains large numbers of functionally competent neutrophils. J Leukoc Biol 75(4):604–611
Brackett CM, Muhitch JB, Evans SS, Gollnick SO (2013) IL-17 promotes neutrophil entry into tumor-draining lymph nodes following induction of sterile inflammation. J Immunol 8:4348–4357
Brinkmann V, Reichard U, Goosmann C, Fauler B, Uhlemann Y, Weiss DS, Weinrauch Y, Zychlinsky A (2004) Neutrophil extracellular traps kill bacteria. Science 303(5663):1532–1535
Brinkmann V, Zychlinsky A (2007) Beneficial suicide: why neutrophils die to make NETs. Nat Rev Microbiol 5(8):577–582
Brooklyn TN, Dunnill MGS, Shetty A, Bowden JJ, Williams JDL, Griffiths CEM, Forbes A, Greenwood R, Probert CS (2006) Infliximab for the treatment of pyoderma gangrenosum: a randomised, double blind, placebo controlled trial. Gut 55(4):505–509
Carlin LM, Stamatiades EG, Auffray C, Hanna RN, Glover L, Vizcay-Barrena G, Hedrick CC, Cook HT, Diebold S, Geissmann F (2013) Nr4a1-dependent Ly6C(low) monocytes monitor endothelial cells and orchestrate their disposal. Cell 153(2):362–375
Carlson JA (2010) The histological assessment of cutaneous vasculitis. Histopathology 56(1):3–23
Casanova-Acebes M, Pitaval C, Weiss LA, Nombela-Arrieta C, Chevre R, AG N, Kunisaki Y, Zhang D, van Rooijen N, Silberstein LE, Weber C, Nagasawa T, Frenette PS, Castrillo A, Hidalgo A (2013) Rhythmic modulation of the hematopoietic niche through neutrophil clearance. Cell 153(5):1025–1035
Cederlund A, Agerberth B, Bergman P (2010) Specificity in killing pathogens is mediated by distinct repertoires of human neutrophil peptides. J Innate Immun 2(6):508–521
Cho JS, Guo Y, Ramos RI, Hebroni F, Plaisier SB, Xuan C, Granick JL, Matsushima H, Takashima A, Iwakura Y, Cheung AL, Cheng G, Lee DJ, Simon SI, Miller LS (2012) Neutrophil-derived IL-1β is sufficient for abscess formation in immunity against Staphylococcus aureus in mice. PLoS Pathog 8(11):e1003047
Cho JS, Pietras EM, Garcia NC, Ramos RI, Farzam DM, Monroe HR, Magorien JE, Blauvelt A, Kolls JK, Cheung AL, Cheng G, Modlin RL, Miller LS (2010) IL-17 is essential for host defense against cutaneous Staphylococcus aureus infection in mice. J Clin Invest 120(5):1762–1773
Cho JS, Zussman J, Donegan NP, Ramos RI, Garcia NC, Uslan DZ, Iwakura Y, Simon SI, Cheung AL, Modlin RL, Kim J, Miller LS (2011) Noninvasive in vivo imaging to evaluate immune responses and antimicrobial therapy against Staphylococcus aureus and USA300 MRSA skin infections. J Invest Dermatol 131(4):907–915
Christoffersson G, Vagesjo E, Vandooren J, Liden M, Massena S, Reinert RB, Brissova M, Powers AC, Opdenakker G, Phillipson M (2012) VEGF-A recruits a proangiogenic MMP-9-delivering neutrophil subset that induces angiogenesis in transplanted hypoxic tissue. Blood 120(23):4653–4662
Christopher MJ, Link DC (2008) Granulocyte colony-stimulating factor induces osteoblast apoptosis and inhibits osteoblast differentiation. J Bone Miner Res 23(11):1765–1774
Cohen PR (2007) Sweet’s syndrome – a comprehensive review of an acute febrile neutrophilic dermatosis. Orphanet J Rare Dis 2(34):26
Cohen PR (2009) Neutrophilic dermatoses: a review of current treatment options. Am J Clin Dermatol 10(5):301–312
Corbin BD, Seeley EH, Raab A, Feldmann J, Miller MR, Torres VJ, Anderson KL, Dattilo BM, Dunman PM, Gerads R, Caprioli RM, Nacken W, Chazin WJ, Skaar EP (2008) Metal chelation and inhibition of bacterial growth in tissue abscesses. Science 319(5865):962–965
Craven RR, Gao X, Allen IC, Gris D, Bubeck Wardenburg J, McElvania-Tekippe E, Ting JP, Duncan JA (2009) Staphylococcus aureus alpha-hemolysin activates the NLRP3-inflammasome in human and mouse monocytic cells. PLoS One 4(10):0007446
Dancey JT, Deubelbeiss KA, Harker LA, Finch CA (1976) Neutrophil kinetics in man. J Clin Invest 58(3):705–715
Davies MJ, Hawkins CL, Pattison DI, Rees MD (2008) Mammalian heme peroxidases: from molecular mechanisms to health implications. Antioxid Redox Signal 10(7):1199–1234
Donabedian H, Gallin JI (1983) The hyperimmunoglobulin E recurrent-infection (Job’s) syndrome: a review of the NIH experience and the literature. Medicine 62(4):195–208
Donadieu J, Fenneteau O, Beaupain B, Mahlaoui N, Chantelot CB (2011) Congenital neutropenia: diagnosis, molecular bases and patient management. Orphanet J Rare Dis 6:26
Eash KJ, Greenbaum AM, Gopalan PK, Link DC (2010) CXCR2 and CXCR4 antagonistically regulate neutrophil trafficking from murine bone marrow. J Clin Invest 120(7):2423–2431
Ellson CD, Davidson K, Ferguson GJ, O’Connor R, Stephens LR, Hawkins PT (2006) Neutrophils from p40phox-/- mice exhibit severe defects in NADPH oxidase regulation and oxidant-dependent bacterial killing. J Exp Med 203(8):1927–1937
Fahmy M, Ramamoorthy S, Hata T, Sandborn WJ (2012) Ustekinumab for peristomal pyoderma gangrenosum. Am J Gastroenterol 107(5):794–795
Fischer-Stabauer M, Boehner A, Eyerich S, Carbone T, Traidl-Hoffmann C, Schmidt-Weber CB, Cavani A, Ring J, Hein R, Eyerich K (2012) Differential in situ expression of IL-17 in skin diseases. Eur J Dermatol 22(6):781–784
Flo TH, Smith KD, Sato S, Rodriguez DJ, Holmes MA, Strong RK, Akira S, Aderem A (2004) Lipocalin 2 mediates an innate immune response to bacterial infection by sequestrating iron. Nature 432(7019):917–921
Fuchs TA, Abed U, Goosmann C, Hurwitz R, Schulze I, Wahn V, Weinrauch Y, Brinkmann V, Zychlinsky A (2007) Novel cell death program leads to neutrophil extracellular traps. J Cell Biol 176(2):231–241
Furze RC, Rankin SM (2008) The role of the bone marrow in neutrophil clearance under homeostatic conditions in the mouse. FASEB J 22(9):3111–3119
Futosi K, Fodor S, Mocsai A (2013) Neutrophil cell surface receptors and their intracellular signal transduction pathways. Int Immunopharmacol 17(3):638–650
Gallo RL, Hooper LV (2012) Epithelial antimicrobial defence of the skin and intestine. Nat Rev Immunol 12(7):503–516
Ganz T (2003) Defensins: antimicrobial peptides of innate immunity. Nat Rev Immunol 3(9):710–720
Ganz T, Gabayan V, Liao H-I, Liu L, Oren A, Graf T, Cole AM (2003) Increased inflammation in lysozyme M-deficient mice in response to Micrococcus luteus and its peptidoglycan. Blood 101(6):2388–2392
Giasuddin ASM, El-Orfi AHAM, Ziu MM, El-Barnawi NY (1998) Sweet’s syndrome: is the pathogenesis mediated by helper T cell type 1 cytokines? J Am Acad Dermatol 39(6):940–943
Goodridge HS, Reyes CN, Becker CA, Katsumoto TR, Ma J, Wolf AJ, Bose N, Chan ASH, Magee AS, Danielson ME, Weiss A, Vasilakos JP, Underhill DM (2011) Activation of the innate immune receptor Dectin-1 upon formation of a ‘phagocytic synapse’. Nature 472(7344):471–475
Grimbacher B, Holland SM, Gallin JI, Greenberg F, Hill SC, Malech HL, Miller JA, O’Connell AC, Puck JM (1999) Hyper-IgE syndrome with recurrent infections – an autosomal dominant multisystem disorder. N Engl J Med 340(9):692–702
Guenova E, Teske A, Fehrenbacher B, Hoerber S, Adamczyk A, Schaller M, Hoetzenecker W, Biedermann T (2011) Interleukin 23 expression in pyoderma gangrenosum and targeted therapy with ustekinumab. Arch Dermatol 147(10):1203–1205
Hall JG, Morris B (1965) The origin of the cells in the efferent lymph from a single lymph node. J Exp Med 121:901–910
Hanna S, Etzioni A (2012) Leukocyte adhesion deficiencies. Ann N Y Acad Sci 1250:50–55
Heath TJ, Lascelles AK, Morris B (1962) The cells of sheep lymph. J Anat 96:397–408
Heath WR, Carbone FR (2013) The skin-resident and migratory immune system in steady state and memory: innate lymphocytes, dendritic cells and T cells. Nat Immunol 14(10):978–985
Heyworth PG, Cross AR, Curnutte JT (2003) Chronic granulomatous disease. Curr Opin Immunol 15(5):578–584
Hruz P, Zinkernagel AS, Jenikova G, Botwin GJ, Hugot JP, Karin M, Nizet V, Eckmann L (2009) NOD2 contributes to cutaneous defense against Staphylococcus aureus through alpha-toxin-dependent innate immune activation. Proc Natl Acad Sci U S A 106(31):12873–12878
Ishigame H, Kakuta S, Nagai T, Kadoki M, Nambu A, Komiyama Y, Fujikado N, Tanahashi Y, Akitsu A, Kotaki H, Sudo K, Nakae S, Sasakawa C, Iwakura Y (2009) Differential roles of interleukin-17A and -17F in host defense against mucoepithelial bacterial infection and allergic responses. Immunity 30(1):108–119
Jackson SH, Gallin JI, Holland SM (1995) The p47phox mouse knock-out model of chronic granulomatous disease. J Exp Med 182(3):751–758
Jain R, Weninger W (2013) Shedding light on cutaneous innate immune responses: the intravital microscopy approach. Immunol Cell Biol 91(4):263–270
Jennette JC, Falk RJ (1997) Small-vessel vasculitis. N Engl J Med 337(21):1512–1523
Johnston B, Kubes P (1999) The alpha4-integrin: an alternative pathway for neutrophil recruitment? Immunol Today 20(12):545–550
Jung D-J, An J-H, Kurokawa K, Jung Y-C, Kim M-J, Aoyagi Y, Matsushita M, Takahashi S, Lee H-S, Takahashi K, Lee BL (2012) Specific serum Ig recognizing Staphylococcal wall teichoic acid induces complement-mediated opsonophagocytosis against Staphylococcus aureus. J Immunol 189(10):4951–4959
Kennedy AD, DeLeo FR (2009) Neutrophil apoptosis and the resolution of infection. Immunol Res 43(1–3):25–61
Kerrigan AM, Dennehy KM, Mourão-Sá D, Faro-Trindade I, Willment JA, Taylor PR, Eble JA, Brown GD, Reis e Sousa C, Brown GD (2009) CLEC-2 is a phagocytic activation receptor expressed on murine peripheral blood neutrophils. J Immunol 182(7):4150–4157
Kolaczkowska E, Kubes P (2013) Neutrophil recruitment and function in health and inflammation. Nat Rev Immunol 13(3):159–175
Kolls JK, Linden A (2004) Interleukin-17 family members and inflammation. Immunity 21(4):467–476
Kubes P, Hunter J, Granger DN (1992) Ischemia/reperfusion-induced feline intestinal dysfunction: importance of granulocyte recruitment. Gastroenterology 103(3):807–812
Lammermann T, Afonso PV, Angermann BR, Wang JM, Kastenmuller W, Parent CA, Germain RN (2013) Neutrophil swarms require LTB4 and integrins at sites of cell death in vivo. Nature 498(7454):371–375
Levy O, Martin S, Eichenwald E, Ganz T, Valore E, Carroll SF, Lee K, Goldmann D, Thorne GM (1999) Impaired innate immunity in the newborn: newborn neutrophils are deficient in bactericidal/permeability-increasing protein. Pediatrics 104(6):1327–1333
Ley K, Laudanna C, Cybulsky MI, Nourshargh S (2007) Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat Rev Immunol 7(9):678–689
Ley K, Smith E, Stark MA (2006) IL-17A-producing neutrophil-regulatory Tn lymphocytes. Immunol Res 34(3):229–242
Li JL, Ng LG (2012) Peeking into the secret life of neutrophils. Immunol Res 53(1–3):168–181
Lim J, Hotchin NA (2012) Signalling mechanisms of the leukocyte integrin αMβ2: current and future perspectives. Biol Cell 104(11):631–640
Maletto BA, Ropolo AS, Alignani DO, Liscovsky MV, Ranocchia RP, Moron VG, Pistoresi-Palencia MC (2006) Presence of neutrophil-bearing antigen in lymphoid organs of immune mice. Blood 108(9):3094–3102
Malka R, Wolach B, Gavrieli R, Shochat E, Rom-Kedar V (2012) Evidence for bistable bacteria-neutrophil interaction and its clinical implications. J Clin Invest 122(8):3002–3011
Mandell GL, Hook EW (1969) Leukocyte bactericidal activity in chronic granulomatous disease: correlation of bacterial hydrogen peroxide production and susceptibility to intracellular killing. J Bacteriol 100(1):531–532
Martin C, Burdon PC, Bridger G, Gutierrez-Ramos JC, Williams TJ, Rankin SM (2003) Chemokines acting via CXCR2 and CXCR4 control the release of neutrophils from the bone marrow and their return following senescence. Immunity 19(4):583–593
Marzano AV, Cugno M, Trevisan V, Fanoni D, Venegoni L, Berti E, Crosti C (2010) Role of inflammatory cells, cytokines and matrix metalloproteinases in neutrophil-mediated skin diseases. Clin Exp Immunol 162(1):100–107
Masson PL, Heremans JF, Schonne E (1969) Lactoferrin, an iron-binding protein in neutrophilic leukocytes. J Exp Med 130(3):643–658
McDonald B, Pittman K, Menezes GB, Hirota SA, Slaba I, Waterhouse CC, Beck PL, Muruve DA, Kubes P (2010) Intravascular danger signals guide neutrophils to sites of sterile inflammation. Science 330(6002):362–366
Metzler KD, Fuchs TA, Nauseef WM, Reumaux D, Roesler J, Schulze I, Wahn V, Papayannopoulos V, Zychlinsky A (2011) Myeloperoxidase is required for neutrophil extracellular trap formation: implications for innate immunity. Blood 117(3):953–959
Miller LS, O’Connell RM, Gutierrez MA, Pietras EM, Shahangian A, Gross CE, Thirumala A, Cheung AL, Cheng G, Modlin RL (2006) MyD88 mediates neutrophil recruitment initiated by IL-1R but not TLR2 activation in immunity against Staphylococcus aureus. Immunity 24(1):79–91
Miller LS, Pietras EM, Uricchio LH, Hirano K, Rao S, Lin H, O’Connell RM, Iwakura Y, Cheung AL, Cheng G, Modlin RL (2007) Inflammasome-mediated production of IL-1beta is required for neutrophil recruitment against Staphylococcus aureus in vivo. J Immunol 179(10):6933–6942
Minegishi Y (2009) Hyper-IgE syndrome. Curr Opin Immunol 21(5):487–492
Mitchell AJ, Edwards MR, Collins AM (2001) Valency or wahlency: is the epitope diversity of the B-cell response regulated or chemically determined? Immunol Cell Biol 79(5):507–511
Molne L, Verdrengh M, Tarkowski A (2000) Role of neutrophil leukocytes in cutaneous infection caused by Staphylococcus aureus. Infect Immun 68(11):6162–6167
Munoz-Planillo R, Franchi L, Miller LS, Nunez G (2009) A critical role for hemolysins and bacterial lipoproteins in Staphylococcus aureus-induced activation of the Nlrp3 inflammasome. J Immunol 183(6):3942–3948
Myles IA, Fontecilla NM, Valdez PA, Vithayathil PJ, Naik S, Belkaid Y, Ouyang W, Datta SK (2013) Signaling via the IL-20 receptor inhibits cutaneous production of IL-1beta and IL-17A to promote infection with methicillin-resistant Staphylococcus aureus. Nat Immunol 14(8):804–811
Nathan C (2006) Neutrophils and immunity: challenges and opportunities. Nat Rev Immunol 6(3):173–182
Nauseef WM (2007) How human neutrophils kill and degrade microbes: an integrated view. Immunol Rev 219(1):88–102
Ng LG, Qin JS, Roediger B, Wang Y, Jain R, Cavanagh LL, Smith AL, Jones CA, de Veer M, Grimbaldeston MA, Meeusen EN, Weninger W (2011) Visualizing the neutrophil response to sterile tissue injury in mouse dermis reveals a three-phase cascade of events. J Invest Dermatol 131(10):2058–2068
Otto BR, Verweij-van Vught AMJJ, MacLaren DM (1992) Transferrins and heme-compounds as iron sources for pathogenic bacteria. Crit Rev Microbiol 18(3):217–233
Oyoshi MK, He R, Li Y, Mondal S, Yoon J, Afshar R, Chen M, Lee DM, Luo HR, Luster AD, Cho JS, Miller LS, Larson A, Murphy GF, Geha RS (2012) Leukotriene B4-driven neutrophil recruitment to the skin is essential for allergic skin inflammation. Immunity 37(4):747–758
Papayannopoulos V, Metzler KD, Hakkim A, Zychlinsky A (2010) Neutrophil elastase and myeloperoxidase regulate the formation of neutrophil extracellular traps. J Cell Biol 191(3):677–691
Park K-H, Kurokawa K, Zheng L, Jung D-J, Tateishi K, Jin J-O, Ha N-C, Kang HJ, Matsushita M, Kwak J-Y, Takahashi K, Lee BL (2010) Human serum mannose-binding lectin senses wall teichoic acid glycopolymer of Staphylococcus aureus, which is restricted in infancy. J Biol Chem 285(35):27167–27175
Perera GK, Di Meglio P, Nestle FO (2012) Psoriasis. Annu Rev Pathol 7:385–422
Phillipson M, Kubes P (2011) The neutrophil in vascular inflammation. Nat Med 17(11):1381–1390
Picard C, Puel A, Bonnet M, Ku CL, Bustamante J, Yang K, Soudais C, Dupuis S, Feinberg J, Fieschi C, Elbim C, Hitchcock R, Lammas D, Davies G, Al-Ghonaium A, Al-Rayes H, Al-Jumaah S, Al-Hajjar S, Al-Mohsen IZ, Frayha HH, Rucker R, Hawn TR, Aderem A, Tufenkeji H, Haraguchi S, Day NK, Good RA, Gougerot-Pocidalo MA, Ozinsky A, Casanova JL (2003) Pyogenic bacterial infections in humans with IRAK-4 deficiency. Science 299(5615):2076–2079
Pilsczek FH, Salina D, Poon KKH, Fahey C, Yipp BG, Sibley CD, Robbins SM, Green FHY, Surette MG, Sugai M, Bowden MG, Hussain M, Zhang K, Kubes P (2010) A novel mechanism of rapid nuclear neutrophil extracellular trap formation in response to Staphylococcus aureus. J Immunol 185(12):7413–7425
Pollock JD, Williams DA, Gifford MA, Li LL, Du X, Fisherman J, Orkin SH, Doerschuk CM, Dinauer MC (1995) Mouse model of X-linked chronic granulomatous disease, an inherited defect in phagocyte superoxide production. Nat Genet 9(2):202–209
Proebstl D, Voisin M-B, Woodfin A, Whiteford J, D’Acquisto F, Jones GE, Rowe D, Nourshargh S (2012) Pericytes support neutrophil subendothelial cell crawling and breaching of venular walls in vivo. J Exp Med 209(6):1219–1234
Puel A, Cypowyj S, Bustamante J, Wright JF, Liu L, Lim HK, Migaud M, Israel L, Chrabieh M, Audry M, Gumbleton M, Toulon A, Bodemer C, El-Baghdadi J, Whitters M, Paradis T, Brooks J, Collins M, Wolfman NM, Al-Muhsen S, Galicchio M, Abel L, Picard C, Casanova JL (2011) Chronic mucocutaneous candidiasis in humans with inborn errors of interleukin-17 immunity. Science 332(6025):65–68
Rankin SM (2010) The bone marrow: a site of neutrophil clearance. J Leukoc Biol 88(2):241–251
Ravetch JV, Kinet J-P (1991) Fc receptors. Annu Rev Immunol 9(1):457–492
Rubin-Bejerano I, Abeijon C, Magnelli P, Grisafi P, Fink GR (2007) Phagocytosis by human neutrophils is stimulated by a unique fungal cell wall component. Cell Host Microbe 2(1):55–67
Ruocco E, Sangiuliano S, Gravina AG, Miranda A, Nicoletti G (2009) Pyoderma gangrenosum: an updated review. J Eur Acad Dermatol Venereol 23(9):1008–1017
Sadik CD, Kim ND, Luster AD (2011) Neutrophils cascading their way to inflammation. Trends Immunol 32(10):452–460
Scapini P, Lapinet-Vera JA, Gasperini S, Calzetti F, Bazzoni F, Cassatella MA (2000) The neutrophil as a cellular source of chemokines. Immunol Rev 177:195–203
Schwarzenberger P, Huang W, Ye P, Oliver P, Manuel M, Zhang Z, Bagby G, Nelson S, Kolls JK (2000) Requirement of endogenous stem cell factor and granulocyte-colony-stimulating factor for IL-17-mediated granulopoiesis. J Immunol 164(9):4783–4789
Semerad CL, Liu F, Gregory AD, Stumpf K, Link DC (2002) G-CSF is an essential regulator of neutrophil trafficking from the bone marrow to the blood. Immunity 17(4):413–423
Sengelov H, Kjeldsen L, Borregaard N (1993) Control of exocytosis in early neutrophil activation. J Immunol 150(4):1535–1543
Serhan CN (2010) Novel lipid mediators and resolution mechanisms in acute inflammation: to resolve or not? Am J Pathol 177(4):1576–1591
Shimada T, Park BG, Wolf AJ, Brikos C, Goodridge HS, Becker CA, Reyes CN, Miao EA, Aderem A, Götz F, Liu GY, Underhill DM (2010) Staphylococcus aureus evades lysozyme-based peptidoglycan digestion that links phagocytosis, inflammasome activation, and IL-1beta secretion. Cell Host Microbe 7(1):38–49
Sims JE, Smith DE (2010) The IL-1 family: regulators of immunity. Nat Rev Immunol 10(2):89–102
Soehnlein O, Lindbom L (2009) Neutrophil-derived azurocidin alarms the immune system. J Leukoc Biol 85(3):344–351
Soehnlein O, Lindbom L (2010) Phagocyte partnership during the onset and resolution of inflammation. Nat Rev Immunol 10(6):427–439
Soehnlein O, Weber C, Lindbom L (2009) Neutrophil granule proteins tune monocytic cell function. Trends Immunol 30(11):538–546
Soehnlein O, Zernecke A, Eriksson EE, Rothfuchs AG, Pham CT, Herwald H, Bidzhekov K, Rottenberg ME, Weber C, Lindbom L (2008) Neutrophil secretion products pave the way for inflammatory monocytes. Blood 112(4):1461–1471
Stark K, Eckart A, Haidari S, Tirniceriu A, Lorenz M, von Bruhl ML, Gartner F, Khandoga AG, Legate KR, Pless R, Hepper I, Lauber K, Walzog B, Massberg S (2013) Capillary and arteriolar pericytes attract innate leukocytes exiting through venules and ‘instruct’ them with pattern-recognition and motility programs. Nat Immunol 14(1):41–51
Stark MA, Huo Y, Burcin TL, Morris MA, Olson TS, Ley K (2005) Phagocytosis of apoptotic neutrophils regulates granulopoiesis via IL-23 and IL-17. Immunity 22(3):285–294
Sugiyama T, Kohara H, Noda M, Nagasawa T (2006) Maintenance of the hematopoietic stem cell pool by CXCL12-CXCR4 chemokine signaling in bone marrow stromal cell niches. Immunity 25(6):977–988
Sumaria N, Roediger B, Ng LG, Qin J, Pinto R, Cavanagh LL, Shklovskaya E, Fazekas de St Groth B, Triccas JA, Weninger W (2011) Cutaneous immunosurveillance by self-renewing dermal gammadelta T cells. J Exp Med 208(3):505–518
Suratt BT, Petty JM, Young SK, Malcolm KC, Lieber JG, Nick JA, Gonzalo JA, Henson PM, Worthen GS (2004) Role of the CXCR4/SDF-1 chemokine axis in circulating neutrophil homeostasis. Blood 104(2):565–571
Sweeney CM, Tobin AM, Kirby B (2011) Innate immunity in the pathogenesis of psoriasis. Arch Dermatol Res 303(10):691–705
Tkalcevic J, Novelli M, Phylactides M, Iredale JP, Segal AW, Jr R (2000) Impaired immunity and enhanced resistance to endotoxin in the absence of neutrophil elastase and cathepsin G. Immunity 12(2):201–210
Turner J, Cho Y, Dinh N-N, Waring AJ, Lehrer RI (1998) Activities of LL-37, a cathelin-associated antimicrobial peptide of human neutrophils. Antimicrob Agents Chemother 42(9):2206–2214
van de Veerdonk FL, Plantinga TS, Hoischen A, Smeekens SP, Joosten LA, Gilissen C, Arts P, Rosentul DC, Carmichael AJ, Smits-van der Graaf CA, Kullberg BJ, van der Meer JW, Lilic D, Veltman JA, Netea MG (2011) STAT1 mutations in autosomal dominant chronic mucocutaneous candidiasis. N Engl J Med 365(1):54–61
van der Veen BS, de Winther MP, Heeringa P (2009) Myeloperoxidase: molecular mechanisms of action and their relevance to human health and disease. Antioxid Redox Signal 11(11):2899–2937
Voisin MB, Probstl D, Nourshargh S (2010) Venular basement membranes ubiquitously express matrix protein low-expression regions: characterization in multiple tissues and remodeling during inflammation. Am J Pathol 176(1):482–495
von Bernuth H, Picard C, Jin Z, Pankla R, Xiao H, Ku CL, Chrabieh M, Mustapha IB, Ghandil P, Camcioglu Y, Vasconcelos J, Sirvent N, Guedes M, Vitor AB, Herrero-Mata MJ, Arostegui JI, Rodrigo C, Alsina L, Ruiz-Ortiz E, Juan M, Fortuny C, Yague J, Anton J, Pascal M, Chang HH, Janniere L, Rose Y, Garty BZ, Chapel H, Issekutz A, Marodi L, Rodriguez-Gallego C, Banchereau J, Abel L, Li X, Chaussabel D, Puel A, Casanova JL (2008) Pyogenic bacterial infections in humans with MyD88 deficiency. Science 321(5889):691–696
von Vietinghoff S, Ley K (2009) IL-17A controls IL-17F production and maintains blood neutrophil counts in mice. J Immunol 183(2):865–873
Wang S, Voisin MB, Larbi KY, Dangerfield J, Scheiermann C, Tran M, Maxwell PH, Sorokin L, Nourshargh S (2006) Venular basement membranes contain specific matrix protein low expression regions that act as exit points for emigrating neutrophils. J Exp Med 203(6):1519–1532
Weinrauch Y, Drujan D, Shapiro SD, Weiss J, Zychlinsky A (2002) Neutrophil elastase targets virulence factors of enterobacteria. Nature 417(6884):91–94
Wengner AM, Pitchford SC, Furze RC, Rankin SM (2008) The coordinated action of G-CSF and ELR + CXC chemokines in neutrophil mobilization during acute inflammation. Blood 111(1):42–49
Winterbourn CC, Kettle AJ (2013) Redox reactions and microbial killing in the neutrophil phagosome. Antioxid Redox Signal 18(6):642–660
Yamada M, Kubo H, Kobayashi S, Ishizawa K, He M, Suzuki T, Fujino N, Kunishima H, Hatta M, Nishimaki K, Aoyagi T, Tokuda K, Kitagawa M, Yano H, Tamamura H, Fujii N, Kaku M (2011) The increase in surface CXCR4 expression on lung extravascular neutrophils and its effects on neutrophils during endotoxin-induced lung injury. Cell Mol Immunol 8(4):305–314
Yang CW, Strong BS, Miller MJ, Unanue ER (2010) Neutrophils influence the level of antigen presentation during the immune response to protein antigens in adjuvants. J Immunol 185(5):2927–2934
Yipp BG, Kubes P (2013) NETosis: how vital is it? Blood 122(16):2784–2794
Yipp BG, Petri B, Salina D, Jenne CN, Scott BN, Zbytnuik LD, Pittman K, Asaduzzaman M, Wu K, Meijndert HC, Malawista SE, de Boisfleury CA, Zhang K, Conly J, Kubes P (2012) Infection-induced NETosis is a dynamic process involving neutrophil multitasking in vivo. Nat Med 18(9):1386–1393
Yousefi S, Mihalache C, Kozlowski E, Schmid I, Simon HU (2009) Viable neutrophils release mitochondrial DNA to form neutrophil extracellular traps. Cell Death Differ 16(11):1438–1444
Acknowledgments
We thank Drs Maté Biro and Shweta Tikoo for their critical reading and inputs on the paper. This work was supported by grants from the NHMRC and the ARC. WW is a fellow of the Cancer Institute New South Wales.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Japan
About this chapter
Cite this chapter
Jain, R., Mitchell, A.J., Tay, S.S., Roediger, B., Weninger, W. (2016). Neutrophils. In: Kabashima, K. (eds) Immunology of the Skin. Springer, Tokyo. https://doi.org/10.1007/978-4-431-55855-2_9
Download citation
DOI: https://doi.org/10.1007/978-4-431-55855-2_9
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-55853-8
Online ISBN: 978-4-431-55855-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)