Abstract
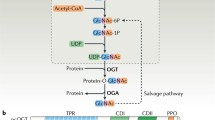
Many nuclear and cytosolic proteins are modified with O-linked β-N-acetylglucosamine (O-GlcNAc) at serine or threonine residues. This O-GlcNAcylation is a dynamic posttranslational modification similar to phosphorylation. In addition, O-GlcNAcylation and phosphorylation have a reciprocal relationship to modulate the function of proteins. There are three types of dynamic interplay between O-GlcNAcylation and phosphorylation. These two modifications occur on same site of the protein or occur on two different sites nearby. Therefore, there is a competitive relationship between O-GlcNAcylation and phosphorylation. The third possibility is these two modifications occur on multiple sites of proteins, and they affect each other or not. In this case, the modification status is changed by cellular signaling. O-GlcNAc modification has vital roles in protein localization, protein degradation, protein stability, transcription, translation, cell cycle regulation, protein-protein interaction, epigenetic regulation, and various diseases including cancer, diabetes, and neurodegenerative diseases. This chapter presents an overview of the function of O-GlcNAc modification in various signaling pathways.
Similar content being viewed by others
References
Dias WB et al (2012) O-GlcNAcylation of kinases. Biochem Biophys Res Commun 422:224–228
Fardini Y et al (2013) O-GlcNAcylation: a new cancer hallmark? Front Endocrinol 4:Acrticle 99, 1–14
Hart GW et al (2011) Cross talk between O-GlcNAcylation and phosphorylation: roles in signaling, transcription, and chronic disease. Annu Rev Biochem 80:825–858
Issad T, Kuo MS (2008) O-GlcNAc modification of transcription factors, glucose sensing and glucotoxicity. Trends Endocrinol Metab 19(10):380–389
Kim EY et al (2012) A role for O-GlcNAcylation in setting circadian clock speed. Genes Dev 26:490–502
Mishra S et al (2011) O-GlcNAc modification: why so intimate associated with phosphorylation? Cell Commun Signal 9:1–4
Ozcan S et al (2010) Modulation of transcription factor function by O-GlcNAc modification. Biochim Biophys Acta 1799:353–364
Ruan HB et al (2013) Cracking the O-GlcNAc code in metabolism. Trends Endocrinol Metab 24(6):301–309
Yang WH et al (2006) Modification of p53 with O-linked N-acetylglucosamine regulates p53 activity and stability. Nat Cell Biol 8(10):1074–1083
Yang X et al (2008) Phosphoinositide signaling links O-GlcNAc transferase to insulin resistance. Nature 451(7181):964–969
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Japan
About this entry
Cite this entry
Park, S., Cho, J.W. (2014). Functional Roles of O-GlcNAc in Cell Signaling. In: Endo, T., Seeberger, P., Hart, G., Wong, CH., Taniguchi, N. (eds) Glycoscience: Biology and Medicine. Springer, Tokyo. https://doi.org/10.1007/978-4-431-54836-2_85-1
Download citation
DOI: https://doi.org/10.1007/978-4-431-54836-2_85-1
Received:
Accepted:
Published:
Publisher Name: Springer, Tokyo
Online ISBN: 978-4-431-54836-2
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences
Publish with us
Chapter history
-
Latest
-GlcNAc in Cell Signaling- Published:
- 16 June 2014
DOI: https://doi.org/10.1007/978-4-431-54836-2_85-2
-
Original
-GlcNAc in Cell Signaling- Published:
- 08 May 2014
DOI: https://doi.org/10.1007/978-4-431-54836-2_85-1