Abstract
Migraine is a common disabling brain disorder whose key manifestations are recurrent attacks of unilateral headache that may be preceded by transient neurological aura symptoms. Missense mutations in CACNA1A, the gene that encodes the pore-forming α1 subunit of human voltage-gated Cav2.1 (P/Q-type) calcium channels, cause a rare form of migraine with aura (familial hemiplegic migraine type 1: FHM1). This chapter, first, briefly summarizes current understanding of the pathophysiological mechanisms that underlie migraine headache, migraine aura, and the onset of a migraine attack. Then, the chapter describes and discusses (i) the functional consequences of FHM1 mutations on the biophysical properties of recombinant human Cav2.1 channels and native Cav2.1 channels in neurons of knockin mouse models carrying the mild R192Q or severe S218L mutations in the orthologous gene, and (ii) the functional consequences of these mutations on neurophysiological processes in the cerebral cortex and trigeminovascular system thought to be involved in the pathophysiology of migraine (including cortical spreading depression, cortical synaptic transmission, and several trigeminal ganglion neuron functions) and the insights into migraine mechanisms obtained from the alterations of these processes in FHM1 knockin mice.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Migraine is an episodic neurological disorder characterized by recurrent attacks of typically throbbing and unilateral, often severe, headache with certain associated features such as nausea, phonophobia, and/or photophobia; in a third of patients the headache is preceded by transient neurological symptoms, that are most frequently visual, but may involve other senses (migraine with aura: MA) (Lipton et al. 2004). Migraine is remarkably common (e.g., it affects 17 % of females and 8 % of males in the European population (Stovner and Hagen 2006), very costly (EUR 1.8 billion per year in Europe) (Olesen et al. 2012), and disabling (one of the 20 most disabling diseases according to the World Health Organization: (Leonardi et al. 2005 ).
Migraine is a complex genetic disorder, with heritability estimates as high as 50 % and a likely polygenic multifactorial inheritance (de Vries et al. 2009). Although recent genome-wide association studies have identified a few risk factors for migraine (Anttila et al. 2010; Chasman et al. 2011; Freilinger et al. 2012), most of our current molecular understanding comes from studies of familial hemiplegic migraine (FHM), a rare monogenic autosomal dominant form of MA (Pietrobon 2007; de Vries et al. 2009; Russell and Ducros 2011).
Three FHM causative genes have been identified, all encoding ion channels or transporters (Ophoff et al. 1996; De Fusco et al. 2003; Dichgans et al. 2005). FHM type 1 (FHM1) is caused by missense mutations in CACNA1A (chromosome 19p13), that encodes the pore-forming α1 subunit of human voltage-gated Cav2.1 (P/Q-type) calcium channels (Ophoff et al. 1996).
The FHM1 mutations produce substitutions of conserved amino acids in important functional regions of the Cav2.1 channel including the pore lining and the voltage sensors (see (Pietrobon 2010; Russell and Ducros 2011; Pietrobon 2013) for recent reviews and references). Figure 1.1 shows the location of FHM1 (and a few sporadic hemiplegic migraine, SHM1) mutations in the secondary structure of the Cav2.1α1 subunit.
Location of FHM1 mutations in the secondary structure of the Cav2.1α1 subunit. FHM1 mutations are indicated with circles; a few SHM1 mutations not found in either controls or parents and affecting conserved amino acids are indicated with rhombs. Reference sequence Genbank Ac. N. X99897. The mutations written in black are those whose effects on the biophysical properties of recombinant Cav2.1 channels have been studied in heterologous expression systems; the mutations underlined are those whose effects have been studied also in transfected neurons from Cav2.1-/- mice expressing human Cav2.1α1 subunits
FHM1 can be considered a model for the common forms of migraine because, apart from the motor weakness or hemiparesis during aura (and the possibly longer aura duration), typical FHM attacks resemble MA attacks (Lipton et al. 2004) and both types of attacks may alternate in patients and co-occur within families (Pietrobon 2007; de Vries et al. 2009; Russell and Ducros 2011). However, several FHM1 families show permanent cerebellar symptoms (such as slowly progressive cerebellar ataxia and/or nystagmus with cerebellar atrophy in some cases), that are usually not observed in common migraines. Pure FHM1 and FHM1 with cerebellar symptoms are usually associated with different mutations. Moreover, in addition to typical attacks, patients of some FHM1 families can have atypical severe attacks with signs of diffuse encephalopathy, impairment of consciousness (coma) or confusion, fever, prolonged hemiplegia lasting several days, and in a few cases seizures (Ducros et al. 2001; Russell and Ducros 2011).
Two different FHM1 knockin mouse models have been generated by introducing the human R192Q and S218L mutations into the orthologous cacna1a Cav2.1 channel gene (van den Maagdenberg et al. 2004, 2010). Whereas mutation R192Q causes in humans pure FHM, mutation S218L causes a particularly dramatic clinical syndrome, that may consists of, in addition to attacks of hemiplegic migraine, slowly progressive cerebellar ataxia and atrophy, epileptic seizures, coma or profound stupor and severe, sometimes fatal, cerebral oedema which can be triggered by only a trivial head trauma (Kors et al. 2001; van den Maagdenberg et al. 2010). Whereas homozygous R192Q and heterozygous S218L knockin mice did not exhibit an overt phenotype, homozygous S218L mice exhibited mild permanent cerebellar ataxia, spontaneous attacks of hemiparesis and/or (sometimes fatal) generalized seizures, and brain edema after only a mild head impact, thus modeling the main features of the severe S218L clinical syndrome (van den Maagdenberg et al. 2004, 2010).
Cav2.1 channels are located in presynaptic terminals and somatodendritic membranes throughout the mammalian brain and spinal cord (Westenbroek et al. 1995), and play a prominent role in initiating action potential (AP)-evoked neurotransmitter release at central nervous system synapses (Pietrobon 2005a). At many central synapses P/Q-, N-, and R-type Ca2+ (Ca) channels cooperate in controlling neurotransmitter release, but P/Q-type channels have a dominant role, partly because of a more efficient coupling to the exocytotic machinery (Mintz et al. 1995; Wu et al. 1999; Li et al. 2007). Moreover, at many central synapses, there is a developmental change in the Ca channel types mediating synaptic transmission, whereby the relative contribution of P/Q-type channels to release increases with postnatal age, until release becomes exclusively dependent on P/Q-type channels (Iwasaki et al. 2000). Among the presynaptic Ca channels, Cav2.1 channels are unique also in their capacity of interacting with and being modulated in a complex manner by a number of Ca-binding proteins ((Catterall and Few 2008) for review and references). As a result, Cav2.1 channels may exhibit both Ca-dependent inactivation and Ca-dependent facilitation. Ca-dependent regulation of presynaptic Cav2.1 channels may play a crucial role in short-term synaptic plasticity during trains of action potentials (Inchauspe et al. 2004; Mochida et al. 2008; Lin et al. 2012). The somatodendritic localization of Cav2.1 channels points to additional postsynaptic roles, e.g., in neural excitability (Pineda et al. 1998; Womack et al. 2004), gene expression (Sutton et al. 1999), and cell survival (Fletcher et al. 2001).
Cav2.1 channels are expressed in all brain regions that have been implicated in the pathogenesis of migraine and/or migraine pain (cf. next Section), including the cerebral cortex (see Sect. 1.5.1), the trigeminal ganglia, and brainstem nuclei involved in the central control of nociception (see Sect. 1.5.2). Their expression is particularly high in the cerebellum, a feature that explains the cerebellar symptoms caused by several FHM1 mutations ((Pietrobon 2010) and Chapter 2 in this book).
2 Migraine Pathophysiology
It is generally recognized that most migraine attacks start in the brain, as suggested by the premonitory symptoms (such as difficulty with speech and reading, increased emotionality, sensory hypersensitivity), that in many patients are highly predictive of the attack although occurring up to 12 h before it, and as suggested also by the nature of some typical migraine triggers such as stress, sleep deprivation, oversleeping, hunger, prolonged sensory stimulation; moreover, psychophysical and neurophysiological studies have provided clear evidence that in the period between attacks migraineurs show hypersensitivity to sensory stimuli and abnormal processing of sensory information (see (Pietrobon and Striessnig 2003; Aurora and Wilkinson 2007; Coppola et al. 2007; Vecchia and Pietrobon 2012; Pietrobon and Moskowitz 2013) for reviews and references).
The neurophysiologial correlate of migraine aura is cortical spreading depression (CSD), a self-sustaining slowly propagating (2–5 mm/min) wave of nearly complete depolarization of a sizable population of brain cells that lasts 1–2 minutes (see (Lauritzen 1994; Pietrobon and Striessnig 2003; Ayata 2009; Charles and Brennan 2009) for reviews and references). CSD can be induced in healthy brain tissue by stimulation of the cerebral cortex with intense depolarizing stimuli. Although the mechanisms of initiation and propagation of CSD remain unclear, the initiation of the positive feedback cycle that ignites CSD and almost zeroes the neuronal membrane potential is thought to depend on the local increase of the extracellular concentration of K+ ions above a critical value and on the activation of a net inward current at the pyramidal cell dendrites (Somjen 2001).
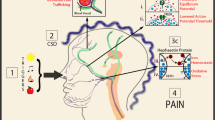
It is generally believed that the headache phase of migraine begins with the activation and sensitization of trigeminal sensory afferents that innervate cranial tissues, in particular the meninges and their large blood vessels; this then leads to sequential activation (and, in most patients, sensitization) of second and third order trigeminovascular neurons (in the trigeminal nucleus caudalis and specific thalamic nuclei, respectively), which in turn activate different areas of the brainstem and forebrain resulting in pain and other symptoms of migraine; the bidirectional signaling between many of these areas contributes to the complexity of the symptoms (see (Olesen et al. 2009; Levy 2010; Akerman et al. 2011; Pietrobon and Moskowitz 2013) for recent reviews and references).
Most dural trigeminovascular afferents can be activated and sensitized by an inflammatory soup and are capsaicin-sensitive, thus exhibiting properties characteristic of nociceptors in other tissues (Strassman et al. 1996; Bove and Moskowitz 1997; Strassman and Levy 2006; Levy et al. 2007; Levy 2010; Vaughn and Gold 2010; Fioretti et al. 2011; Yan et al. 2011). Activation of meningeal nociceptors in vivo leads to release of vasoactive proinflammatory peptides such as CGRP and substance P from their peripheral nerve endings, that produce vasodilation of meningeal blood vessels (mainly due to CGRP), plasma extravasation and local activation of dural mast cells, with ensuing release of cytokines and other inflammatory mediators (neurogenic inflammation) (Waeber and Moskowitz 2005; Levy 2010). Measurements of CGRP levels into the external and internal jugular venous blood have provided evidence that CGRP is released during migraine attacks, and several findings support a pivotal role of CGRP in migraine, including the efficacy of CGRP receptor antagonists in migraine treatment (reviewed in (Recober and Russo 2009; Villalon and Olesen 2009; Ho et al. 2010)). However, the mechanism of actions of CGRP during a migraine attack and the exact sites of action of CGRP receptor antagonists remain unclear and controversial. The localization of CGRP receptors in the trigeminovascular system points to multiple possible mechanisms at both peripheral and central sites (Lennerz et al. 2008).
Although a large body of indirect evidence supports the idea that a sterile meningeal inflammation is a key mechanism that may activate and sensitize perivascular meningeal afferents and lead to migraine pain (reviewed in (Waeber and Moskowitz 2005; Levy 2009, 2010; Vecchia and Pietrobon 2012; Pietrobon and Moskowitz 2013)), the endogenous processes that promote meningeal inflammation and the mechanisms underlying the sustained activation and sensitization of the trigeminovascular system resulting in the typical long-lasting throbbing headache remain unclear.
The mechanisms of the primary brain dysfunction(s) leading to the onset of a migraine attack, to CSD susceptibility and to episodic activation of the trigeminovascular pain pathway remain largely unknown and remain the major open issues in the neurobiology of migraine.
Increasing evidence from animal studies support the idea that CSD can activate trigeminal nociception and thus trigger the headache mechanisms (Vecchia and Pietrobon 2012; Pietrobon and Moskowitz 2013). A direct nociceptive effect of CSD has been demonstrated by the finding that a single CSD can lead to a long-lasting increase in ongoing activity of dural nociceptors and central second order trigeminovascular neurons (Zhang et al. 2010, 2011), and evokes alterations in the meninges and brainstem consistent with the development of pain (Bolay et al. 2002). An interesting recent study provided evidence that CSD leads to opening of neuronal pannexin channels and consequent release of proinflammatory mediators, which initiates a parenchymal inflammatory response leading to sustained release of inflammatory mediators from glia limitans; this CSD-induced inflammatory cascade may provide the sustained stimulus required for sensitization of trigeminal nociceptors and lasting pain (Karatas et al. 2013).
The idea that CSD is noxious and may trigger headache is indirectly supported by the finding that the electrical stimulation threshold for induction of CSD in the rat cortex increases after chronic treatment with five different migraine prophylactic drugs, that are effective in reducing the frequency of migraine attacks (both with and without aura) (Ayata et al. 2006). Moreover, further support to the view of CSD as a key migraine trigger has been provided by the analysis of experimental CSD in FHM knockin mouse models, that revealed a lower electrical stimulation threshold for CSD induction and a higher velocity of CSD propagation in both FHM1 and FHM2 knockin mice (van den Maagdenberg et al. 2004, 2010; Eikermann-Haerter et al. 2009b; Leo et al. 2011) (see Sect. 1.5.1). However, despite the strong support provided by animal studies, the idea that CSD may initiate the headache mechanisms in migraine is not generally accepted, mainly because it seems unable to explain some clinical observations, in particular the lack of a fixed relationship between aura and headache (reviewed in (Pietrobon and Striessnig 2003; Charles 2010; Vecchia and Pietrobon 2012; Pietrobon and Moskowitz 2013)).
The analysis of interictal cortical excitability using psychophysical, electrophysiological, and brain imaging methods has produced contradictory findings and interpretations regarding the mechanisms underlying the abnormal processing of sensory information in migraineurs (see (Pietrobon and Striessnig 2003; Aurora and Wilkinson 2007; Coppola et al. 2007; Vecchia and Pietrobon 2012) for reviews and references). Interestingly, recent transcranial magnetic stimulation studies in MA patients point to deficient regulatory mechanisms of cortical excitability and consequent reduced ability to dynamically maintain the cortical excitatory-inhibitory balance and to prevent excessive increases in cortical excitation, rather than merely hypo- or hyperexcitability, as the mechanisms underlying abnormal sensory processing (Antal et al. 2008; Conte et al. 2010; Siniatchkin et al. 2012). The molecular and cellular mechanisms underlying the abnormal regulation of cortical function and the increased susceptibility to CSD in migraine remain largely unknown. As discussed in Sect. 1.4, the functional analysis of FHM knockin mouse models supports the view of migraine as a disorder of brain excitability characterized by deficient regulation of the cortical excitatory-inhibitory balance, and gives insights into the possible underlying molecular and cellular mechanisms and their relationship to CSD susceptibility.
3 Effect of FHM1 Mutations on the Biophysical Properties of Cav2.1 Channels
The analysis of the P/Q-type calcium current in different neurons (including cortical and TG neurons) of R192Q and S218L FHM1 knockin mice revealed gain-of-function of the Cav2.1 current in a wide range of relatively mild depolarizations, reflecting shifted activation of mutant Cav2.1 channels to more negative voltages (van den Maagdenberg et al. 2004, 2010; Tottene et al. 2009; Inchauspe et al. 2010; Fioretti et al. 2011 Gao et al. 2012). P/Q current densities were similar in knockin and wild-type (WT) neurons at higher voltages (that elicit maximal Cav2.1 channel open probability), indicating similar densities of functional Cav2.1 channels (van den Maagdenberg et al. 2004, 2010; Tottene et al. 2009; Fioretti et al. 2011; Gao et al. 2012). The shift to lower voltages of Cav2.1 channel activation and the gain-of-function of the neuronal Cav2.1 current were about twice as large in homozygous compared to heterozygous knockin mice, revealing an allele-dosage effect consistent with dominance of the mutation in FHM1 patients (van den Maagdenberg et al. 2010). In correlation with the severity of the clinical phenotype, the gain-of-function of the P/Q Ca current at low voltages was larger in cerebellar granule cells of S218L than R192Q knockin mice (van den Maagdenberg et al. 2004, 2010). Two photon microscopy in cerebellar slices of S218L knockin mice revealed increased AP-evoked Ca transients in individual synaptic terminals of cerebellar granule cells, a finding consistent with increased open probability of mutant presynaptic Cav2.1 channels (Adams et al. 2010), that may be due to shifted activation to lower voltages, as shown for the somatic channels (van den Maagdenberg et al. 2010), and/or to basal Ca-dependent facilitation of mutant channels as suggested by the authors (Adams et al. 2010) (see sect. 1.4).
The gain-of-function effect of the FHM1 mutations on native neuronal mouse Cav2.1 channels is in agreement with the increased open probability (mainly due to shifted activation to lower voltages) of recombinant human Cav2.1 channels carrying eight different FHM1 mutations (including R192Q and S218L), that was revealed by single channel recordings (Hans et al. 1999; Tottene et al. 2002; Tottene et al. 2005; Catterall et al. 2008) (our unpublished observations). The larger gain-of-function of the P/Q Ca current in neurons of S218L compared to R192Q knockin mice at low voltages is in agreement with the lower threshold of activation of human S218L Cav2.1 compared to human R192Q Cav2.1 channels (Tottene et al. 2005).
Consistent (and significant for the majority of the 13 FHM1 mutations analyzed so far, shown in black in Fig. 1.1) shifts to lower voltages of activation of recombinant mutant channels were also revealed by measurements of whole-cell current in heterologous expression systems (Kraus et al. 1998, 2000; Hans et al. 1999; Tottene et al. 2002, 2005; Melliti et al. 2003; Mullner et al. 2004; Weiss et al. 2008; Adams et al. 2009; Serra et al. 2009; Garza-Lopez et al. 2012) and transfected neurons (where five FHM1 mutations, underlined in Fig. 1.1, were analyzed) (Tottene et al. 2002, 2005). Moreover, these studies have shown that the FHM1 mutations may also alter the inactivation properties of Cav2.1 channels; the effects were different depending on the mutation and, e.g., resulted in increased, decreased, or unaltered inactivation during train of pulses, depending on the mutation.
For certain mutations, the specific alterations of channel gating, including the magnitude (or even the presence) of the shift to lower voltages of channel activation, depended on the Cav2.1α1 splice variant and the type of coexpressed β subunit (Mullner et al. 2004; Adams et al. 2009). The use of different Cav2.1 splice variants and/or β subunits might explain some discrepancies existing in the literature regarding the functional effects of some FHM1 mutations in transfected cells: e.g., the T666 M mutation was shown to produce a negative shift of Cav2.1 activation in (Kraus et al. 1998; Tottene et al. 2002) in contrast, a lack of effect on activation gating was reported in (Barrett et al. 2005; Tao et al. 2012).
Moreover, the expression of specific Cav2.1 splice variants and/or auxiliary subunits in different neurons might underlie the recent finding of a differential effect of the R192Q mutation on activation gating of native P/Q-type Ca channels in different TG neurons of knockin mice (Fioretti et al. 2011). In small capsaicin-insensitive TG neurons characterized by expression of T-type Ca currents (CI-T neurons) there was a larger P/Q-type Ca current density following mild depolarizations and a larger AP-evoked P/Q calcium current when compared to CI-T neurons from WT mice. In striking contrast, the P/Q-type current density, voltage dependence, and kinetics were not altered by the FHM1 mutation in small capsaicin-sensitive neurons of R192Q knockin mice (Fioretti et al. 2011).
Interestingly, while in cortical pyramidal cells of R192Q KI mice the shift to lower voltages of Cav2.1 channel activation resulted in increased AP-evoked Ca current, a similar shift of mutant R192Q channels at the Calyx of Held synaptic terminals did not alter the AP-evoked Ca current (Inchauspe et al. 2010). The different durations of the AP in pyramidal cells and Calyx (1.8 vs 0.44 ms AP half width) may largely explain the differential effects of the FHM1 mutation on the AP-evoked Ca current; in fact, the AP-evoked Ca current became larger in knockin compared to WT Calyx terminals when the longer duration pyramidal cell AP was used as depolarizing stimulus (Inchauspe et al. 2010).
The findings of (Fioretti et al. 2011) and (Inchauspe et al. 2010) have important general implications for familial migraine mechanisms, in that neuron subtype-specific (and/or subcellular compartment-specific) alterations of Cav2.1 channels and/or AP-evoked Ca influx may help to explain why a mutation in a Ca channel that is widely expressed in the nervous system (Westenbroek et al. 1995) produces the specific neuronal dysfunctions leading to migraine (see Sect. 1.5).
In contrast with the unaltered densities of functional Cav2.1 channels in neurons of FHM1 knockin mice (van den Maagdenberg et al. 2004, 2010; Tottene et al. 2009; Fioretti et al. 2011), overexpression of recombinant mutant Cav2.1 channels in transfected cells leads to alterated (decreased for most mutations) density of functional channels in the membrane, that results in reduced maximal whole-cell P/Q-type Ca current in cells expressing mutant channels (Hans et al. 1999; Tottene et al. 2002; Cao et al. 2004; Barrett et al. 2005; Cao and Tsien 2005; Tottene et al. 2005; Tao et al. 2012), including neurons expressing R192Q and S218L Cav2.1 channels (Tottene et al. 2002; Cao et al. 2004; Cao and Tsien 2005; Tottene et al. 2005) (cf. Fig. 3 in (Pietrobon 2010)).
4 Effect of FHM1 Mutations on Modulation of Cav2.1 Channels
Studies of recombinant human Cav2.1 channels carrying five different FHM1 mutations have shown that the migraine mutations reduce G-protein-mediated inhibitory modulation of Cav2.1 channels (Melliti et al. 2003; Weiss et al. 2008; Serra et al. 2009; Garza-Lopez et al. 2012), an effect that may lead to further gain-of-function of Ca influx through mutant channels during neuromodulation. The only study that investigated voltage-dependent G-protein modulation using physiological-like stimuli revealed a reduced Gβγ-mediated inhibition of the Ca current evoked by AP-like stimuli and a reduced facilitation of the Ca current carried by mutant channels after a train of short depolarizing pulses in cells overexpressing Gβγ subunits; the reduction of G-protein modulation was dependent on the duration of the AP, being larger with APs of longer duration (Serra et al. 2009).
Indirect evidence of reduced G-protein-mediated inhibition of native neuronal Cav2.1 channels in R192Q knockin mice has been recently obtained in a study investigating the effect of the FHM1 mutation on sleep regulation by adenosine receptors in vivo; in fact, the FHM1 mouse models showed reduced responsiveness to either inhibition or enhancement of adenosine receptor activation (Deboer et al. 2013).
Besides G-protein modulation, the FHM1 mutations may also reduce Ca-dependent facilitation (CDF) of Cav2.1 channels. CDF of recombinant Cav2.1 channels was largely reduced by the R192Q mutation and almost eliminated by the S218L mutation (Adams et al. 2010), suggesting that the increase in the open probability of mutant channels (larger for S218L than R192Q: (Tottene et al. 2005)) partially or completely occludes further facilitation. CDF of native Cav2.1 channels in Purkinje cells of S218L knockin mice was also reduced (Adams et al. 2010). In contrast, CDF of native Cav2.1 channels in both Purkinje cells (Adams et al. 2010) and Calyx of Held synaptic terminals (Inchauspe et al. 2010) of R192Q knockin mice was not significantly affected, suggesting that also the effect of FHM1 mutations on CDF likely depends on the specific Cav2.1 variant expressed in a given neuron or neuronal subcompartment.
5 Insights into Migraine Pathophysiology from FHM1 Knockin Mouse Models
5.1 Cortical Synaptic Transmission and Cortical Spreading Depression
In the cerebral cortex, excitatory synaptic transmission at pyramidal cell synapses in different cortical areas depends predominantly on P/Q-type Ca channels (Iwasaki et al. 2000; Koester and Sakmann 2000; Rozov et al. 2001; Ali and Nelson 2006; Zaitsev et al. 2007; Tottene et al. 2009) with a notable exception at synapses between layer five pyramidal cells and burst-firing bipolar interneurons of motor cortex (Ali and Nelson 2006). Activation of the cortex following thalamic stimulation in thalamocortical slices was completely inhibited by blocking P/Q Ca channels (Llinas et al. 2007). The Ca channel pharmacology of cortical inhibitory synapses has been investigated only at fast spiking (FS) interneurons synapses; neurotransmission was found to be exclusively dependent on P/Q-type channels in many cortical areas (Zaitsev et al. 2007; Kruglikov and Rudy 2008; Tottene et al. 2009) (cf also (Sasaki et al. 2006)), but again with the exception of layer five of the motor cortex, where it was exclusively dependent on N-type (Ali and Nelson 2006).
The analysis of cortical excitatory synaptic transmission in neuronal microcultures and in brain slices from R192Q knockin mice revealed enhanced excitatory neurotransmission, due to enhanced action potential evoked Ca influx through mutant presynaptic P/Q Ca channels and enhanced probability of glutamate release at cortical pyramidal cell synapses (Tottene et al. 2009). Short-term synaptic depression during trains of action potentials was also enhanced. Neither amplitude nor frequency of miniature excitatory postsynaptic currents were altered, indicating the absence of homeostatic compensatory mechanisms at excitatory synapses onto pyramidal cells (Tottene et al. 2009). Although indirect, evidence for gain-of-function of excitatory neurotransmission has also been obtained at parallel fibers-Purkinje cell synapses in cerebellar slices of S218L knockin mice (Adams et al. 2010) and at excitatory synapses onto dorsal suprachiasmatic nucleus neurons of R192Q knockin mice (van Oosterhout et al. 2008).
In striking contrast with the enhanced glutamatergic transmission, paired recordings of fast spiking (FS) inhibitory interneurons and layer 2/3 pyramidal cells in acute cortical slices revealed that the inhibitory GABAergic transmission at FS interneuron synapses was not altered in R192Q knockin mice, despite being initiated by P/Q Ca channels (Tottene et al. 2009). The main explanation for the unaltered GABA release at FS interneuron synapses appears to be the specific expression in FS interneurons of a Cav2.1 subtype whose gating properties are little affected by the mutation (Vecchia, Tottene and Pietrobon, unpublished observations).
The investigation of experimental CSD, elicited either by electrical stimulation of the cortex in vivo or high KCl in cortical slices, revealed a lower threshold for CSD induction and an increased velocity of CSD propagation in R192Q and S218L knockin compared to WT mice (van den Maagdenberg et al. 2004, 2010; Tottene et al. 2009). Moreover, a single CSD, elicited by brief epidural application of high KCl, produced more severe and prolonged motor deficits (including hemiplegia) in FHM1 knockin mice, and, in contrast with WT mice, CSD readily propagated into the striatum (Eikermann-Haerter et al. 2009b, 2011). The much higher propensity of CSD to propagate to the striatum in FHM1 mutants compared to WT mice may explain their motor deficits and the hemiplegia typical of FHM1 aura. In agreement with the higher incidence of migraine in females, the velocity of propagation and the frequency of CSDs, elicited by continuous epidural high KCl application, were larger in females than in males of both mutant strains; the sex difference was abrogated by ovariectomy and enhanced by orchiectomy, suggesting that female and male gonadal hormones exert reciprocal effects on CSD susceptibility (Eikermann-Haerter et al. 2009a, b).
The strength of CSD facilitation as well as the severity of the post-CSD neurological motor deficits and the propensity of CSD to propagate into subcortical structures in R192Q and S218L knockin mice were all in good correlation with the strength of the gain-of-function of the Cav2.1 channel and the severity of the clinical phenotype produced by the two FHM1 mutations (van den Maagdenberg et al. 2004, 2010; Tottene et al. 2005; Eikermann-Haerter et al. 2009b, 2011; Kors et al. 2001). Propagation of CSD to the hippocampus and thalamus and repetitive CSD events following a single CSD-inducing stimulus were observed only in S218L mutants (van den Maagdenberg et al. 2010; Eikermann-Haerter et al. 2011). These unique CSD features might account for the severe attacks with seizures, coma, and cerebral edema typical of patients with the S218L mutation. Moreover, the recent finding of increased frequency of periinfarct ischemic depolarizations during acute experimental stroke (and larger critical tissue perfusion level below which infarction ensues) in S218L knockin compared to WT mice (Eikermann-Haerter et al. 2012) suggests that the S218L mutation may lead to increased vulnerability to ischemic stroke as a consequence of increased susceptibility to ischemic depolarizations akin to CSD. The irregular firing patterns and hyperexcitability of Purkinje cells recently uncovered in S218L knockin mice likely contributes to the cerebellar ataxia produced by the S218L mutation in mice and humans (Gao et al. 2012).
Tottene et al. (2009) provided direct evidence that the gain-of-function of glutamate release at synapses onto cortical pyramidal cells may explain the facilitation of experimental CSD in FHM1 knockin mice (Tottene et al. 2009). In fact, the facilitation of CSD in acute cortical slices of R192Q knockin mice was completely eliminated (both CSD threshold and velocity became similar to those in WT slices) when glutamate release at pyramidal cell synapses was brought back to WT values by partially inhibiting P/Q channels (Tottene et al. 2009). The data are consistent with and support a model of CSD initiation in which Cav2.1-dependent release of glutamate from cortical pyramidal cell synapses and activation of NMDA receptors (and possibly postsynaptic Cav2.1 channels) play a key role in the positive feedback cycle that ignites CSD (Pietrobon 2005b; Tottene et al. 2009). This model and in general the specific requirement of Cav2.1 channels in the initiation and propagation of CSD (induced by electrical stimulation or brief pulses of high K+ in healthy tissue) are further supported by the findings that (i) after blockade of either the P/Q-type Ca channels or the NMDA receptors, CSD could not be induced in cortical slices of WT mice even with largely supra threshold depolarizing stimuli; in contrast, blockade of N- or R-type Ca channels had only a small inhibitory effect on CSD threshold and velocity of propagation (Tottene et al. 2011), and (ii) in the spontaneous mouse mutants leaner and tottering, that carry loss-of-function mutations in cacna1a (Pietrobon 2010), the in vivo electrical threshold for CSD initiation was greatly increased and the CSD velocity decreased compared to WT mice (Ayata et al. 2000).
In migraineurs, CSD is not induced by experimental depolarizing stimuli, but arises “spontaneously” in response to specific triggers, that somehow create in the cortex conditions for initiation of the positive feedback cycle that overwhelms the regulatory mechanisms controlling cortical [K+]o and ignites CSD. Insights into how this might occur have been provided by the differential effect of FHM1 mutations on cortical excitatory and inhibitory synaptic transmission (Tottene et al. 2009). This finding suggests that, very likely, the neuronal circuits that dynamically adjust the balance between excitation and inhibition during cortical activity are altered in FHM1 (Tottene et al. 2009). Functional alterations in these circuits are expected to lead to dysfunctional regulation of the cortical excitatory-inhibitory balance and hence to abnormal processing of sensory information (Monier et al. 2003; Shu et al. 2003). It has been hypothesized that this dysregulation may in certain conditions (e.g., in response to migraine triggers such as intense, prolonged sensory stimulation) lead to disruption of the excitatory-inhibitory balance and hyperactivity of cortical circuits, mainly due to excessive recurrent excitation, that may create the conditions for the initiation of “spontaneous” CSDs (e.g., by increasing the extracellular [K+] above a critical value) (Tottene et al. 2009; Vecchia and Pietrobon 2012).
Impairment of the cortical circuits that dynamically adjust the excitatory-inhibitory balance during cortical activity, due to excessive recurrent glutamatergic neurotransmission, might also underlie the abnormal regulation of interictal cortical function in some common migraine subtypes, for which there is indirect evidence consistent with enhanced cortical glutamatergic neurotransmission (Prescot et al. 2009; Siniatchkin et al. 2012) and enhanced cortico–cortical or recurrent excitatory neurotransmission (Siniatchkin et al. 2007; Wilkinson et al. 2008; Conte et al. 2010; Battista et al. 2011).
The gain-of-function effects on cortical pyramidal cell Cav2.1 channels and on cortical glutamatergic synaptic transmission derived from the functional analysis of R192Q and S218L FHM1 knockin mice provide a coherent picture of molecular and cellular mechanisms that may produce increased susceptibility to CSD and thus explain the aura symptoms in FHM1 patients (Fig. 1.2). Most likely, the conclusions are valid for all FHM1 mutations, given that the aura phenotype is common to all FHM1 patients and that eight different FHM1 mutations produce a consistent gain-of-function of single channel Ca influx through recombinant human Cav2.1 channels (see Sect 1.3 ). The opposite conclusions derived from the functional analysis of FHM1 mutations (including the R192Q mutation) in transfected hippocampal neurons overexpressing human Cav2.1α1 subunits (i.e., loss-of-function of Cav2.1 channels with consequent decreased contribution of P/Q Ca channels to glutamatergic synaptic transmission, and unaltered synaptic strength due to compensatory mechanisms (Cao et al. 2004)) provide a picture that seems unable to explain the increased susceptibility to CSD and the aura phenotype of FHM1 patients. Thus, to be able to draw meaningful conclusions regarding the neurophysiological processes involved in the pathogenesis of FHM1 (and probably neuronal channelopathies in general), it seems essential to study these processes in neurons expressing the channels at the native endogenous level.
Functional alterations in the cerebral cortex of a familial hemiplegic migraine type 1 (FHM1) knockin mouse model. Action potential (AP)-evoked glutamate release and excitatory synaptic transmission at pyramidal cell (PC) synapses are increased, due to increased AP-evoked Ca influx through presynaptic Cav2.1 channels consequent to the increased open probability and activation at lower voltages of mutant compared to WT channels. In striking contrast, AP-evoked GABA release and inhibitory synaptic transmission at fast spiking (FS) interneuron synapses are unaltered. The differential effect of the FHM1 mutation on excitatory and inhibitory synaptic transmission likely results in dysfunctional regulation of the cortical excitatory/inhibitory balance. Experimental cortical spreading depression (CSD) is facilitated, as revealed by a decreased threshold for CSD induction, an increased rate of CSD propagation and an increased propensity to propagate into subcortical structures in FHM1 mouse models
5.2 Trigeminal Ganglion Sensory Afferents and Headache Mechanisms
P/Q-type Ca channels account for a large proportion of the Ca current of dissociated trigeminal ganglion (TG) neurons (Borgland et al. 2001; Fioretti et al. 2011; Tao et al. 2012), and are involved in the control of CGRP release from capsaicin-sensitive perivascular terminals of meningeal nociceptors (Hong et al. 1999; Akerman et al. 2003) and of glutamate release from TG neurons in culture (Xiao et al. 2008). P/Q channels are also involved in controlling tonic inhibition of trigeminal nucleus caudalis neurons with input from the dura (Ebersberger et al. 2004), and in descending inhibitory and facilitatory pathways that regulate trigeminal and spinal pain transmission (Knight et al. 2002; Urban et al. 2005). The analysis of the pain responses in Cav2.1−/− null mice revealed a complex role of Cav2.1 channels in pain: a pronociceptive role in inflammatory and neuropathic pain but an antinociceptive role in response to acute non-injurious noxious thermal stimuli (Luvisetto et al. 2006); this complex picture likely reflects the prominent role of P/Q channels in controlling release of both excitatory and inhibitory neurotransmitters and their wide distribution in different regions involved in pain.
As mentioned in Sect. 1.3, the analysis of the P/Q-type Ca current in two defined subpopulations of small (capacitance ≤20 pF) TG neurons from adult R192Q knockin mice showed gain-of-function of the Cav2.1 channel in capsaicin-insensitive TG neurons expressing T-type Ca channels (CI-T neurons), but unaltered Cav2.1 channel gating properties in capsaicin-sensitive TG neurons lacking T-type Ca channels (CS neurons) (Fioretti et al. 2011). WT CI-T and CS neurons were both characterized by APs of long duration with typical shoulder and tonic or slowly adapting firing. While the rheobase (i.e., the minimal current injection that elicits APs) and the frequency of APs were not affected by the FHM1 mutation, the duration of the AP in CI-T neurons of R192Q knockin mice was prolonged, due to a delayed repolarization and more pronounced shoulder that correlated with the larger AP-evoked P/Q-type Ca current measured in these neurons (Fioretti et al. 2011). In contrast, neither the AP shape nor any other excitability property were affected by the FHM1 mutation in small capsaicin-sensitive neurons (in agreement with the unaltered P/Q channel gating and unaltered AP-evoked P/Q current in these TG neurons) (Fioretti et al. 2011).
Measurements of the Ca current in small TG neurons retrogradely labeled from the dura revealed that a major fraction of small dural afferents were CS neurons and none were CI-T neurons (Fioretti et al. 2011). Several lines of evidence indicate that most small capsaicin-sensitive dural afferents are peptidergic neurons expressing CGRP (Jansen et al. 1990; Messlinger et al. 1993; Shimizu et al. 2007). Measurements of CGRP release from dura mater in fluid-filled hemisected skulls revealed that neither basal nor K+ -evoked CGRP release were significantly different in R192Q knockin compared to WT mice (Fioretti et al. 2011); this finding is consistent with and supports lack of effect of the FHM1 mutation on presynaptic P/Q channels at the peripheral terminals of CGRP-expressing (including CS) dural afferents, as demonstrated for P/Q channels at the soma of small CS TG neurons (Fioretti et al. 2011). These data argue against the idea that the facilitation of CGRP-dependent dural vasodilation and CGRP-dependent dural mast cell degranulation contribute to the generation of migraine pain in FHM1 (Vecchia and Pietrobon 2012; Pietrobon and Moskowitz 2013).
Despite the lower percent of CGRP-expressing neurons recently reported in the trigeminal ganglion of R192Q knockin compared to WT mice (Mathew et al. 2011), the FHM1 mutation did increase evoked CGRP release from intact trigeminal ganglia (Fioretti et al. 2011) and both basal and evoked CGRP release from cultured TG neurons (Ceruti et al. 2011) of R192Q knockin mice, suggesting alternative roles. There is in vivo and in vitro evidence for non-synaptic intraganglionic release of CGRP, substance P and ATP from TG neurons cell bodies in response to depolarizing stimuli (Matsuka et al. 2001; Ulrich-Lai et al. 2001). It has been suggested that CGRP-mediated intraganglionic crosstalk between neurons and between neurons and satellite glial cells could promote and maintain a neuron-glia inflammatory cycle that might contribute to peripheral trigeminal sensitization (Villalon and Olesen 2009; Ho et al. 2010). This suggestion is mainly based on the evidence that prolonged application of CGRP to cultures of TG neurons and/or satellite glial cells leads to increased gene expression and/or membrane targeting of specific receptors (e.g., P2X3) in neurons and to increased expression of inflammatory genes and release of inflammatory mediators from satellite glial cells; these inflammatory mediators can sensitize TG neurons and act back on glial cells further activating them ((Vecchia and Pietrobon 2012; Pietrobon and Moskowitz 2013) and references therein). Assuming that similar phenomena occur in the trigeminal ganglion in vivo upon prolonged elevations of CGRP, the enhanced intraganglionic CGRP release measured in FHM1 knockin mice suggests, as a possible working hypothesis, that FHM1 mutations might facilitate peripheral sensitization at the ganglion level. As a matter of fact, there is some evidence suggesting facilitation of CGRP-mediated neuron to glia crosstalk following exposure to proinflammatory stimuli in cultured TG neurons from juvenile R192Q knockin mice (Ceruti et al. 2011). Moreover, a stronger basal activation of macrophages, a larger basal release of TNFα, and an enhanced P2X3 receptor-mediated neuronal current have been recently reported in cultured TG neurons from these mice (Franceschini et al. 2013a; Nair et al. 2010). Also intact trigeminal ganglia of R192Q knockin mice appear enriched in activated macrophages both in the absence and presence of a standard inflammatory stimulus (Franceschini et al. 2013b). On the basis of these findings, it has been suggested that FHM1 mutations might lead to a basal inflammatory milieu within the trigeminal ganglion (Franceschini et al. 2013a) (but of similar levels of inflammatory cytokines in TG extracts from WT and R192Q knockin mice in (Franceschini et al. 2013b)). Interestingly, in both cultured TG neurons and intact ganglia a larger fraction of TG neurons was immunoreactive for active phosphorylated CaMKII in R192Q knockin compared to WT mice. Blockade of P/Q channels in cultured TG neurons eliminated the difference in amount of phosphoprotein between the two genotypes, suggesting facilitation of basal Cav2.1-dependent Ca signaling (Nair et al. 2010).
The functional analysis of TG neurons and dural afferents in R192Q knockin mice supports the conclusions that (i) the FHM1 mutations may lead to gain-of-function of the P/Q Ca current in certain TG neurons without affecting the P/Q current in other TG neuron subtypes, including small capsaicin-sensitive dural afferents; (ii) the FHM1 mutations lead to several gain-of-function effects at the trigeminal ganglion level, but do not alter the functional properties of small capsaicin-sensitive peptidergic dural afferents, including CGRP release from their peripheral terminals at the dura.
Again, as in the case of the cortical neurophysiological processes discussed in Sect. 1.5.1, different conclusions may be drawn from the recent functional analysis of an FHM1 mutation in transfected neonatal TG neurons expressing human mutant Cav2.1α1 subunits (Tao et al. 2012). In these neurons, the P/Q current density was much smaller than in neurons expressing the WT channel, and small CGRP-expressing IB4-negative neurons showed a compensatory increase in low-voltage-activated Ca current, a lower rheobase and higher AP frequency, with unaltered AP duration, compared to neurons expressing the WT channel (Tao et al. 2012). Assuming that this subpopulation of TG neurons innervates the dura, one could predict enhanced CGRP release at the dura as a consequence of the enhanced excitability indirectly produced by the FHM1 mutation in transfected neurons. This prediction contrasts with the unaltered CGRP release measured at the dura of FHM1 knockin mice (Fioretti et al. 2011), further stressing the importance of studying neurophysiological processes potentially involved in the pathogenesis of FHM1 in neurons expressing the Cav2.1 channels at the native endogenous level.
References
Adams PJ, Garcia E, David LS, Mulatz KJ, Spacey SD, Snutch TP (2009) Ca(V)2.1 P/Q-type calcium channel alternative splicing affects the functional impact of familial hemiplegic migraine mutations: implications for calcium channelopathies. Channels (Austin) 3:110–121
Adams PJ, Rungta RL, Garcia E, van den Maagdenberg AM, MacVicar BA, Snutch TP (2010) Contribution of calcium-dependent facilitation to synaptic plasticity revealed by migraine mutations in the P/Q-type calcium channel. Proc Natl Acad Sci U S A 107:18694–18699
Akerman S, Holland PR, Goadsby PJ (2011) Diencephalic and brainstem mechanisms in migraine. Nat Rev Neurosci 12:570–584
Akerman S, Williamson DJ, Goadsby PJ (2003) Voltage-dependent calcium channels are involved in neurogenic dural vasodilatation via a presynaptic transmitter release mechanism. Br J Pharmacol 140:558–566
Ali AB, Nelson C (2006) Distinct Ca2+ channels mediate transmitter release at excitatory synapses displaying different dynamic properties in rat neocortex. Cereb Cortex 16:386–393
Antal A, Lang N, Boros K, Nitsche M, Siebner HR, Paulus W (2008) Homeostatic metaplasticity of the motor cortex is altered during headache-free intervals in migraine with aura. Cereb Cortex 18:2701–2705
Anttila V, Stefansson H, Kallela M, Todt U, Terwindt GM, Calafato MS, Nyholt DR, Dimas AS, Freilinger T, Muller-Myhsok B, Artto V, Inouye M, Alakurtti K, Kaunisto MA, Hamalainen E, de Vries B, Stam AH, Weller CM, Heinze A, Heinze-Kuhn K, Goebel I, Borck G, Gobel H, Steinberg S, Wolf C, Bjornsson A, Gudmundsson G, Kirchmann M, Hauge A, Werge T, Schoenen J, Eriksson JG, Hagen K, Stovner L, Wichmann HE, Meitinger T, Alexander M, Moebus S, Schreiber S, Aulchenko YS, Breteler MM, Uitterlinden AG, Hofman A, van Duijn CM, Tikka-Kleemola P, Vepsalainen S, Lucae S, Tozzi F, Muglia P, Barrett J, Kaprio J, Farkkila M, Peltonen L, Stefansson K, Zwart JA, Ferrari MD, Olesen J, Daly M, Wessman M, van den Maagdenberg AM, Dichgans M, Kubisch C, Dermitzakis ET, Frants RR, Palotie A (2010) Genome-wide association study of migraine implicates a common susceptibility variant on 8q22.1. Nat Genet 42:869–873
Aurora SK, Wilkinson F (2007) The brain is hyperexcitable in migraine. Cephalalgia 27:1442–1453
Ayata C (2009) Spreading depression: from serendipity to targeted therapy in migraine prophylaxis. Cephalalgia 29:1095–1114
Ayata C, Jin H, Kudo C, Dalkara T, Moskowitz MA (2006) Suppression of cortical spreading depression in migraine prophylaxis. Ann Neurol 59:652–661
Ayata C, Shimizu-Sasamata M, Lo EH, Noebels JL, Moskowitz (2000) Impaired neurotransmitter release and elevated threshold for cortical spreading depression in mice with mutations in the a1A subunit of P/Q type calcium channels. Neuroscience 95:639–645
Barrett CF, Cao YQ, Tsien RW (2005) Gating deficiency in a familial hemiplegic migraine type 1 mutant P/Q-type calcium channel. J Biol Chem 280:24064–24071
Battista J, Badcock DR, McKendrick AM (2011) Migraine increases centre-surround suppression for drifting visual stimuli. PLoS ONE 6:e18211
Bolay H, Reuter U, Dunn AK, Huang Z, Boas DA, Moskowitz MA (2002) Intrinsic brain activity triggers trigeminal meningeal afferents in a migraine model. Nat Med 8:136–142
Borgland SL, Connor M, Christie MJ (2001) Nociceptin inhibits calcium channel currents in a subpopulation of small nociceptive trigeminal ganglion neurons in mouse. J Physiol 536:35–47
Bove GM, Moskowitz MA (1997) Primary afferent neurons innervating guinea pig dura. J Neurophysiol 77:299–308
Cao YQ, Piedras-Renteria ES, Smith GB, Chen G, Harata NC, Tsien RW (2004) Presynaptic Ca2+ channels compete for channel type-preferring slots in altered neurotransmission arising from Ca2+ channelopathy. Neuron 43:387–400
Cao YQ, Tsien RW (2005) Effects of familial hemiplegic migraine type 1 mutations on neuronal P/Q-type Ca2+ channel activity and inhibitory synaptic transmission. Proc Natl Acad Sci U S A 102:2590–2595
Catterall WA, Dib-Hajj S, Meisler MH, Pietrobon D (2008) Inherited neuronal ion channelopathies: new windows on complex neurological diseases. J Neurosci 28:11768–11777
Catterall WA, Few AP (2008) Calcium channel regulation and presynaptic plasticity. Neuron 59:882–901
Ceruti S, Villa G, Fumagalli M, Colombo L, Magni G, Zanardelli M, Fabbretti E, Verderio C, van den Maagdenberg AM, Nistri A, Abbracchio MP (2011) Calcitonin gene-related peptide-mediated enhancement of purinergic neuron/glia communication by the algogenic factor bradykinin in mouse trigeminal ganglia from wild-type and R192Q Cav2.1 Knock-in mice: implications for basic mechanisms of migraine pain. J Neurosci 31:3638–3649
Charles A (2010) Does cortical spreading depression initiate a migraine attack? Maybe not. Headache 50:731–733
Charles A, Brennan K (2009) Cortical spreading depression-new insights and persistent questions. Cephalalgia 29:1115–1124
Chasman DI, Schurks M, Anttila V, de Vries B, Schminke U, Launer LJ, Terwindt GM, van den Maagdenberg AM, Fendrich K, Volzke H, Ernst F, Griffiths LR, Buring JE, Kallela M, Freilinger T, Kubisch C, Ridker PM, Palotie A, Ferrari MD, Hoffmann W, Zee RY, Kurth T (2011) Genome-wide association study reveals three susceptibility loci for common migraine in the general population. Nat Genet 43:695–698
Conte A, Barbanti P, Frasca V, Iacovelli E, Gabriele M, Giacomelli E, Aurilia C, Pichiorri F, Gilio F, Inghilleri M (2010) Differences in short-term primary motor cortex synaptic potentiation as assessed by repetitive transcranial magnetic stimulation in migraine patients with and without aura. Pain 148:43–48
Coppola G, Pierelli F, Schoenen J (2007) Is the cerebral cortex hyperexcitable or hyperresponsive in migraine? Cephalalgia 27:1427–1439
De Fusco M, Marconi R, Silvestri L, Atorino L, Rampoldi L, Morgante L, Ballabio A, Aridon P, Casari G (2003) Haploinsufficiency of ATP1A2 encoding the Na+/K+ pump alpha2 subunit associated with familial hemiplegic migraine type 2. Nat Genet 33:192–196
de Vries B, Frants RR, Ferrari MD, van den Maagdenberg AM (2009) Molecular genetics of migraine. Hum Genet 126:115–132
Deboer T, van Diepen HC, Ferrari MD, Van den Maagdenberg AM, Meijer JH (2013) Reduced sleep and low adenosinergic sensitivity in cacna1a R192Q mutant mice. Sleep 36:127–136
Dichgans M, Freilinger T, Eckstein G, Babini E, Lorenz-Depiereux B, Biskup S, Ferrari MD, Herzog J, van den Maagdenberg AM, Pusch M, Strom TM (2005) Mutation in the neuronal voltage-gated sodium channel SCN1A in familial hemiplegic migraine. Lancet 366:371–377
Ducros A, Denier C, Joutel A, Cecillon M, Lescoat C, Vahedi K, Darcel F, Vicaut E, Bousser MG, Tournier-Lasserve E (2001) The clinical spectrum of familial hemiplegic migraine associated with mutations in a neuronal calcium channel. N Engl J Med 345:17–24
Ebersberger A, Portz S, Meissner W, Schaible HG, Richter F (2004) Effects of N-, P/Q- and L-type calcium channel blockers on nociceptive neurones of the trigeminal nucleus with input from the dura. Cephalalgia 24:250–261
Eikermann-Haerter K, Baum MJ, Ferrari MD, van den Maagdenberg AM, Moskowitz MA, Ayata C (2009a) Androgenic suppression of spreading depression in familial hemiplegic migraine type 1 mutant mice. Ann Neurol 66:564–568
Eikermann-Haerter K, Dilekoz E, Kudo C, Savitz SI, Waeber C, Baum MJ, Ferrari MD, van den Maagdenberg AMJM, Moskowitz MA, Ayata C (2009b) Genetic and hormonal factors modulate spreading depression and transient hemiparesis in mouse models of familial hemiplegic migraine type 1. J Clin Investig 119:99–109
Eikermann-Haerter K, Hyun Lee J, Yuzawa I, Liu CH, Zhou Z, Kyoung Shin H, Zheng Y, Qin T, Kurth T, Waeber C, Ferrari MD, van den Maagdenberg AMJM, Moskowitz MA, Ayata C (2012) Migraine mutations increase stroke vulnerability by facilitating ischemic depolarizations/clinical perspective. Circulation 125:335–345
Eikermann-Haerter K, Yuzawa I, Qin T, Wang Y, Baek K, Kim YR, Hoffmann U, Dilekoz E, Waeber C, Ferrari MD, van den Maagdenberg AM, Moskowitz MA, Ayata C (2011) Enhanced subcortical spreading depression in familial hemiplegic migraine type 1 mutant mice. J Neurosci 31:5755–5763
Fioretti B, Catacuzzeno L, Sforna L, Gerke-Duncan MB, van den Maagdenberg AM, Franciolini F, Connor M, Pietrobon D (2011) Trigeminal ganglion neuron subtype-specific alterations of Ca(V)2.1 calcium current and excitability in a Cacna1a mouse model of migraine. J Physiol 589:5879–5895
Fletcher CF, Tottene A, Lennon VA, Wilson SM, Dubel SJ, Paylor R, Hosford DA, Tessarollo L, McEnery MW, Pietrobon D, Copeland NG, Jenkins NA (2001) Dystonia and cerebellar atrophy in Cacna1a null mice lacking P/Q calcium channel activity. Faseb J 15:1288–1290
Franceschini A, Hullugundi SK, van den Maagdenberg AM, Nistri A, Fabbretti E (2013a) Effects of LPS on P2X3 receptors of trigeminal sensory neurons and macrophages from mice expressing the R192Q Cacna1a gene mutation of familial hemiplegic migraine-1. Purinergic Signal 9:7–13
Franceschini A, Vilotti S, Ferrari MD, van den Maagdenberg AM, Nistri A, Fabbretti E (2013b) TNFalpha levels and macrophages expression reflect an inflammatory potential of trigeminal ganglia in a mouse model of familial hemiplegic migraine. PLoS ONE 8:e52394
Freilinger T, Anttila V, de Vries B, Malik R, Kallela M, Terwindt GM, Pozo-Rosich P, Winsvold B, Nyholt DR, van Oosterhout WP, Artto V, Todt U, Hamalainen E, Fernandez-Morales J, Louter MA, Kaunisto MA, Schoenen J, Raitakari O, Lehtimaki T, Vila-Pueyo M, Gobel H, Wichmann E, Sintas C, Uitterlinden AG, Hofman A, Rivadeneira F, Heinze A, Tronvik E, van Duijn CM, Kaprio J, Cormand B, Wessman M, Frants RR, Meitinger T, Muller-Myhsok B, Zwart JA, Farkkila M, Macaya A, Ferrari MD, Kubisch C, Palotie A, Dichgans M, van den Maagdenberg AM (2012) Genome-wide association analysis identifies susceptibility loci for migraine without aura. Nat Genet
Gao Z, Todorov B, Barrett CF, van Dorp S, Ferrari MD, van den Maagdenberg AM, De Zeeuw CI, Hoebeek FE (2012) Cerebellar ataxia by enhanced Ca(V)2.1 currents is alleviated by Ca2+ -dependent K+ -channel activators in Cacna1a(S218L) mutant mice. J Neurosci 32:15533–15546
Garza-Lopez E, Sandoval A, Gonzalez-Ramirez R, Gandini MA, Van den Maagdenberg A, De Waard M, Felix R (2012) Familial hemiplegic migraine type 1 mutations W1684R and V1696I alter G protein-mediated regulation of Ca(V)2.1 voltage-gated calcium channels. Biochim Biophys Acta 1822:1238–1246
Hans M, Luvisetto S, Williams ME, Spagnolo M, Urrutia A, Tottene A, Brust PF, Johnson EC, Harpold MM, Stauderman KA, Pietrobon D (1999) Functional consequences of mutations in the human a1A calcium channel subunit linked to familial hemiplegic migraine. J Neurosci 19:1610–1619
Ho TW, Edvinsson L, Goadsby PJ (2010) CGRP and its receptors provide new insights into migraine pathophysiology. Nat Rev Neurol 6:573–582
Hong KW, Kim CD, Rhim BY, Lee WS (1999) Effect of omega-conotoxin GVIA and omega-agatoxin IVA on the capsaicin-sensitive calcitonin gene-related peptide release and autoregulatory vasodilation in rat pial arteries. J Cereb Blood Flow Metab 19:53–60
Inchauspe CG, Martini FJ, Forsythe ID, Uchitel OD (2004) Functional compensation of P/Q by N-type channels blocks short-term plasticity at the calyx of held presynaptic terminal. J Neurosci 24:10379–10383
Inchauspe CG, Urbano FJ, Di Guilmi MN, Forsythe ID, Ferrari MD, van den Maagdenberg AM, Uchitel OD (2010) Gain of function in FHM-1 Ca(V)2.1 knock-in mice is related to the shape of the action potential. J Neurophysiol 104:291–299
Iwasaki S, Momiyama A, Uchitel OD, Takahashi T (2000) Developmental changes in calcium channel types mediating central synaptic transmission. J Neurosci 20:59–65
Jansen I, Alafaci C, Uddman R, Edvinsson L (1990) Evidence that calcitonin gene-related peptide contributes to the capsaicin-induced relaxation of guinea pig cerebral arteries. Regul Pept 31:167–178
Karatas H, Erdener SE, Gursoy-Ozdemir Y, Lule S, Eren-Kocak E, Sen ZD, Dalkara T (2013) Spreading depression triggers headache by activating neuronal Panx1 channels. Science 339:1092–1095
Knight YE, Bartsch T, Kaube H, Goadsby PJ (2002) P/Q-type calcium-channel blockade in the periaqueductal gray facilitates trigeminal nociception: a functional genetic link for migraine? J Neurosci 22:RC213
Koester HJ, Sakmann B (2000) Calcium dynamics associated with action potentials in single nerve terminals of pyramidal cells in layer 2/3 of the young rat neocortex. J Physiol 529(Pt 3):625–646
Kors EE, Terwindt GM, Vermeulen FL, Fitzsimons RB, Jardine PE, Heywood P, Love S, van den Maagdenberg AM, Haan J, Frants RR, Ferrari MD (2001) Delayed cerebral edema and fatal coma after minor head trauma: role of the CACNA1A calcium channel subunit gene and relationship with familial hemiplegic migraine. Ann Neurol 49:753–760
Kraus RL, Sinnegger M, Koschak A, Glossmann H, Stenirri S, Carrera P, Striessnig J (2000) Three new familial hemiplegic migraine mutants affect P/Q-type Ca2+channel kinetics. J Biol Chem 275:9239–9243
Kraus RL, Sinnegger MJ, Glossmann H, Hering S, Striessnig J (1998) Familial hemiplegic migraine mutations change a1A Ca2+ channel kinetics. J Biol Chem 273:5586–5590
Kruglikov I, Rudy B (2008) Perisomatic GABA release and thalamocortical integration onto neocortical excitatory cells are regulated by neuromodulators. Neuron 58:911–924
Lauritzen M (1994) Pathophysiology of the migraine aura. The spreading depression theory. Brain 117(Pt 1):199–210
Lennerz JK, Ruhle V, Ceppa EP, Neuhuber WL, Bunnett NW, Grady EF, Messlinger K (2008) Calcitonin receptor-like receptor (CLR), receptor activity-modifying protein 1 (RAMP1), and calcitonin gene-related peptide (CGRP) immunoreactivity in the rat trigeminovascular system: differences between peripheral and central CGRP receptor distribution. J Comp Neurol 507:1277–1299
Leo L, Gherardini L, Barone V, De Fusco M, Pietrobon D, Pizzorusso T, Casari G (2011) Increased susceptibility to cortical spreading depression in the mouse model of familial hemiplegic migraine type 2. PLoS Genet 7:e1002129
Leonardi M, Steiner TJ, Scher AT, Lipton RB (2005) The global burden of migraine: measuring disability in headache disorders with WHO’s Classification of Functioning, Disability and Health (ICF). J Headache Pain 6:429–440
Levy D (2009) Migraine pain, meningeal inflammation, and mast cells. Curr Pain Headache Rep 13:237–240
Levy D (2010) Migraine pain and nociceptor activation–where do we stand? Headache 50:909–916
Levy D, Burstein R, Kainz V, Jakubowski M, Strassman AM (2007) Mast cell degranulation activates a pain pathway underlying migraine headache. Pain 130:166–176
Li L, Bischofberger J, Jonas P (2007) Differential gating and recruitment of P/Q-, N-, and R-type Ca2 + channels in hippocampal mossy fiber boutons. J Neurosci 27:13420–13429
Lin KH, Erazo-Fischer E, Taschenberger H (2012) Similar intracellular Ca2 + requirements for inactivation and facilitation of voltage-gated Ca2 + channels in a glutamatergic mammalian nerve terminal. J Neurosci 32:1261–1272
Lipton RB, Bigal ME, Steiner TJ, Silberstein SD, Olesen J (2004) Classification of primary headaches. Neurology 63:427–435
Llinas RR, Choi S, Urbano FJ, Shin HS (2007) Gamma-band deficiency and abnormal thalamocortical activity in P/Q-type channel mutant mice. Proc Natl Acad Sci U S A 104:17819–17824
Luvisetto S, Marinelli S, Panasiti MS, D’Amato FR, Fletcher CF, Pavone F, Pietrobon D (2006) Pain sensitivity in mice lacking the Ca(v)2.1alpha1 subunit of P/Q-type Ca2 + channels. Neuroscience 142:823–832
Mathew R, Andreou AP, Chami L, Bergerot A, van den Maagdenberg AM, Ferrari MD, Goadsby PJ (2011) Immunohistochemical characterization of calcitonin gene-related peptide in the trigeminal system of the familial hemiplegic migraine 1 knock-in mouse. Cephalalgia 31:1368–1380
Matsuka Y, Neubert JK, Maidment NT, Spigelman I (2001) Concurrent release of ATP and substance P within guinea pig trigeminal ganglia invivo. Brain Res 915:248–255
Melliti K, Grabner M, Seabrook GR (2003) The familial hemiplegic migraine mutation R192Q reduces G-protein-mediated inhibition of P/Q-type (Ca(V)2.1) calcium channels expressed in human embryonic kidney cells. J Physiol 546:337–347
Messlinger K, Hanesch U, Baumgartel M, Trost B, Schmidt RF (1993) Innervation of the dura mater encephali of cat and rat: ultrastructure and calcitonin gene-related peptide-like and substance P-like immunoreactivity. Anat Embryol (Berl) 188:219–237
Mintz IM, Sabatini BL, Regehr WG (1995) Calcium control of transmitter release at a cerebellar synapse. Neuron 15:675–688
Mochida S, Few AP, Scheuer T, Catterall WA (2008) Regulation of presynaptic Ca(V)2.1 channels by Ca2 + sensor proteins mediates short-term synaptic plasticity. Neuron 57:210–216
Monier C, Chavane F, Baudot P, Graham LJ, Fregnac Y (2003) Orientation and direction selectivity of synaptic inputs in visual cortical neurons: a diversity of combinations produces spike tuning. Neuron 37:663–680
Mullner C, Broos LA, van den Maagdenberg AM, Striessnig J (2004) Familial Hemiplegic Migraine Type 1 Mutations K1336E, W1684R, and V1696I Alter Cav2.1 Ca2 + Channel Gating: evidence for beta-subunit isoform-specific effects. J Biol Chem 279:51844–51850
Nair A, Simonetti M, Birsa N, Ferrari MD, van den Maagdenberg AM, Giniatullin R, Nistri A, Fabbretti E (2010) Familial hemiplegic migraine Ca(v)2.1 channel mutation R192Q enhances ATP-gated P2X3 receptor activity of mouse sensory ganglion neurons mediating trigeminal pain. Mol Pain 6:48
Olesen J, Burstein R, Ashina M, Tfelt-Hansen P (2009) Origin of pain in migraine: evidence for peripheral sensitisation. Lancet Neurol 8:679–690
Olesen J, Gustavsson A, Svensson M, Wittchen HU, Jonsson B, group Cs, European Brain C (2012) The economic cost of brain disorders in Europe. Eur J Neurol 19:155–162
Ophoff RA, Terwindt GM, Vergouwe MN, van Eijk R, Oefner PJ, Hoffman SMG, Lamerdin JE, Mohrenweiser HW, Bulman DE, Ferrari M, Haan J, Lindhout D, van Hommen G-JB, Hofker MH, Ferrari MD, Frants RR (1996) Familial hemiplegic migraine and episodic ataxia type-2 are caused by mutations in the Ca2+ channel gene CACNL1A4. Cell 87:543–552
Pietrobon D (2005a) Function and dysfunction of synaptic calcium channels: insights from mouse models. Curr Opin Neurobiol 15:257–265
Pietrobon D (2005b) Migraine: new molecular mechanisms. Neuroscientist 11:373–386
Pietrobon D (2007) Familial hemiplegic migraine. Neurotherapeutics 4:274–284
Pietrobon D (2010) Cav2.1 channelopathies. Pflugers Arch 460:375–393
Pietrobon D (2013) Calcium channels and migraine. Biochim Biophys Acta 1828:1655–1665
Pietrobon D, Moskowitz MA (2013) Pathophysiology of migraine. Annu Rev Physiol 75:365–391
Pietrobon D, Striessnig J (2003) Neurobiology of migraine. Nat Rev Neurosci 4:386–398
Pineda JC, Waters RS, Foehring RC (1998) Specificity in the interaction of HVA Ca2 + channel types with Ca2 + -dependent AHPs and firing behavior in neocortical pyramidal neurons. J Neurophysiol 79:2522–2534
Prescot A, Becerra L, Pendse G, Tully S, Jensen E, Hargreaves R, Renshaw P, Burstein R, Borsook D (2009) Excitatory neurotransmitters in brain regions in interictal migraine patients. Mol Pain 5:34
Recober A, Russo AF (2009) Calcitonin gene-related peptide: an update on the biology. Curr Opin Neurol 22:241–246
Rozov A, Burnashev N, Sakmann B, Neher E (2001) Transmitter release modulation by intracellular Ca2 + buffers in facilitating and depressing nerve terminals of pyramidal cells in layer 2/3 of the rat neocortex indicates a target cell-specific difference in presynaptic calcium dynamics. J Physiol 531:807–826
Russell MB, Ducros A (2011) Sporadic and familial hemiplegic migraine: pathophysiological mechanisms, clinical characteristics, diagnosis, and management. Lancet Neurol 10:457–470
Sasaki S, Huda K, Inoue T, Miyata M, Imoto K (2006) Impaired feedforward inhibition of the thalamocortical projection in epileptic Ca2 + channel mutant mice, tottering. J Neurosci 26:3056–3065
Serra SA, Fernandez-Castillo N, Macaya A, Cormand B, Valverde MA, Fernandez–Fernandez JM (2009) The hemiplegic migraine-associated Y1245C mutation in CACNA1A results in a gain of channel function due to its effect on the voltage sensor and G-protein-mediated inhibition. Pflugers Arch 458:489–502
Shimizu T, Toriumi H, Sato H, Shibata M, Nagata E, Gotoh K, Suzuki N (2007) Distribution and origin of TRPV1 receptor-containing nerve fibers in the dura mater of rat. Brain Res 1173:84–91
Shu Y, Hasenstaub A, McCormick DA (2003) Turning on and off recurrent balanced cortical activity. Nature 423:288–293
Siniatchkin M, Kroner-Herwig B, Kocabiyik E, Rothenberger A (2007) Intracortical inhibition and facilitation in migraine—a transcranial magnetic stimulation study. Headache 47:364–370
Siniatchkin M, Sendacki M, Moeller F, Wolff S, Jansen O, Siebner H, Stephani U (2012) Abnormal Changes of Synaptic Excitability in Migraine with Aura. Cereb Cortex 22:2207–2216
Somjen GG (2001) Mechanisms of spreading depression and hypoxic spreading depression-like depolarization. Physiol Rev 81:1065–1096
Stovner LJ, Hagen K (2006) Prevalence, burden, and cost of headache disorders. Curr Opin Neurol 19:281–285
Strassman AM, Levy D (2006) Response properties of dural nociceptors in relation to headache. J Neurophysiol 95:1298–1306
Strassman AM, Raymond SA, Burstein R (1996) Sensitization of meningeal sensory neurons and the origin of headaches. Nature 384:560–564
Sutton KG, McRory JE, Guthrie H, Murphy TH, Snutch TP (1999) P/Q-type calcium channels mediate the activity-dependent feedback of syntaxin-1A. Nature 401:800–804
Tao J, Liu P, Xiao Z, Zhao H, Gerber BR, Cao YQ (2012) Effects of familial hemiplegic migraine type 1 mutation T666 M on voltage-gated calcium channel activities in trigeminal ganglion neurons. J Neurophysiol 107:1666–1680
Tottene A, Conti R, Fabbro A, Vecchia D, Shapovalova M, Santello M, van den Maagdenberg AMJM, Ferrari MD, Pietrobon D (2009) Enhanced excitatory transmission at cortical synapses as the basis for facilitated spreading depression in Ca(v)2.1 knockin migraine mice. Neuron 61:762–773
Tottene A, Fellin T, Pagnutti S, Luvisetto S, Striessnig J, Fletcher C, Pietrobon D (2002) Familial hemiplegic migraine mutations increase Ca(2 +) influx through single human Cav2.1 channels and decrease maximal Cav2.1 current density in neurons. Proc Natl Acad Sci U S A 99:13284–13289
Tottene A, Pivotto F, Fellin T, Cesetti T, van den Maagdenberg AM, Pietrobon D (2005) Specific kinetic alterations of human Cav2.1 calcium channels produced by mutation S218L causing familial hemiplegic migraine and delayed cerebral edema and coma after minor head trauma. J Biol Chem 280:17678–17686
Tottene A, Urbani A, Pietrobon D (2011) Role of different voltage-gated Ca2+ channels in cortical spreading depression: specific requirement of P/Q-type Ca2+ channels. Channels (Austin) 5:110–114
Ulrich-Lai YM, Flores CM, Harding-Rose CA, Goodis HE, Hargreaves KM (2001) Capsaicin-evoked release of immunoreactive calcitonin gene-related peptide from rat trigeminal ganglion: evidence for intraganglionic neurotransmission. Pain 91:219–226
Urban MO, Ren K, Sablad M, Park KT (2005) Medullary N-type and P/Q-type calcium channels contribute to neuropathy-induced allodynia. NeuroReport 16:563–566
van den Maagdenberg AM, Pietrobon D, Pizzorusso T, Kaja S, Broos LA, Cesetti T, van de Ven RC, Tottene A, van der Kaa J, Plomp JJ, Frants RR, Ferrari MD (2004) A Cacna1a knockin migraine mouse model with increased susceptibility to cortical spreading depression. Neuron 41:701–710
van den Maagdenberg AM, Pizzorusso T, Kaja S, Terpolilli N, Shapovalova M, Hoebeek FE, Barrett CF, Gherardini L, van de Ven RC, Todorov B, Broos LA, Tottene A, Gao Z, Fodor M, De Zeeuw CI, Frants RR, Plesnila N, Plomp JJ, Pietrobon D, Ferrari MD (2010) High cortical spreading depression susceptibility and migraine-associated symptoms in Ca(v)2.1 S218L mice. Ann Neurol 67:85–98
van Oosterhout F, Michel S, Deboer T, Houben T, van de Ven RCG, Albus H, Westerhout J, Vansteensel MJ, Ferrari MD, van den Maagdenberg AMJM, Meijer JH (2008) Enhanced circadian phase resetting in R192Q Cav2.1 calcium channel migraine mice. Ann Neurol 64:315–324
Vaughn AH, Gold MS (2010) Ionic mechanisms underlying inflammatory mediator-induced sensitization of dural afferents. J Neurosci 30:7878–7888
Vecchia D, Pietrobon D (2012) Migraine: a disorder of brain excitatory-inhibitory balance? Trends Neurosci 35:507–520
Villalon CM, Olesen J (2009) The role of CGRP in the pathophysiology of migraine and efficacy of CGRP receptor antagonists as acute antimigraine drugs. Pharmacol Ther 124:309–323
Waeber C, Moskowitz MA (2005) Migraine as an inflammatory disorder. Neurology 64:S9–S15
Weiss N, Sandoval A, Felix R, Van den Maagdenberg A, De Waard M (2008) The S218L familial hemiplegic migraine mutation promotes deinhibition of Ca(v)2.1 calcium channels during direct G-protein regulation. Pflugers Arch 457:315–326
Westenbroek RE, Sakurai T, Elliott EM, Hell JW, Starr TVB, Snutch TP, Catterall WA (1995) Immunochemical identification and subcellular distribution of the a1A subunits of brain calcium channels. J Neurosci 15:6403–6418
Wilkinson F, Karanovic O, Wilson HR (2008) Binocular rivalry in migraine. Cephalalgia 28:1327–1338
Womack MD, Chevez C, Khodakhah K (2004) Calcium-activated potassium channels are selectively coupled to P/Q-type calcium channels in cerebellar Purkinje neurons. J Neurosci 24:8818–8822
Wu LG, Westenbroek RE, Borst JGG, Catterall WA, Sakmann B (1999) Calcium channel types with distinct presynaptic localization couple differentially to transmitter release in single calyx-type synapses. J Neurosci 19:726–736
Xiao Y, Richter JA, Hurley JH (2008) Release of glutamate and CGRP from trigeminal ganglion neurons: role of calcium channels and 5-HT1 receptor signaling. Mol Pain 4:12
Yan J, Edelmayer RM, Wei X, De Felice M, Porreca F, Dussor G (2011) Dural afferents express acid-sensing ion channels: a role for decreased meningeal pH in migraine headache. Pain 152:106–113
Zaitsev AV, Povysheva NV, Lewis DA, Krimer LS (2007) P/Q-type, but not N-type, calcium channels mediate GABA release from fast-spiking interneurons to pyramidal cells in rat prefrontal cortex. J Neurophysiol 97:3567–3573
Zhang X, Levy D, Kainz V, Noseda R, Jakubowski M, Burstein R (2011) Activation of central trigeminovascular neurons by cortical spreading depression. Ann Neurol 69:855–865
Zhang X, Levy D, Noseda R, Kainz V, Jakubowski M, Burstein R (2010) Activation of meningeal nociceptors by cortical spreading depression: implications for migraine with aura. J Neurosci 30:8807–8814
Acknowledgements
I acknowledge the support of grants from the University of Padova (Strategic Project: Physiopathology of Signaling in Neuronal Tissue), the Fondazione Cariparo (Excellence Project: Calcium Signaling in Health and Disease) and Telethon-Italy (GGP06234).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Pietrobon, D. (2014). Cav2.1 Channels and Migraine. In: Weiss, N., Koschak, A. (eds) Pathologies of Calcium Channels. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-40282-1_1
Download citation
DOI: https://doi.org/10.1007/978-3-642-40282-1_1
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-40281-4
Online ISBN: 978-3-642-40282-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)